Abstract
In this study, we assessed six Oklahoma streams for Enterococcus sediment and water concentrations along with water quality, sediment, hydrologic and geographical factors. We also conducted a microcosm experiment from two stream sediments to evaluate Enterococcus survivability under stable laboratory conditions. Stream sites exhibited common relationships between Enterococcus and other environmental factors, including significant correlations to antecedent dry period, Escherichia coli, impervious area, dissolved oxygen, and turbidity. These correlations were found for Enterococcus in both water and sediment. Specifically for Enterococcus in sediment, concentrations were also significantly correlated to turbidity and sediment percent organic matter, but not to hydrological conditions. Conversely, concentrations of Enterococcus in water exhibited significant moderate correlations to precipitation, antecedent dry period, drainage area, impervious area, and discharge, as well as streambed particle size. High variability between geographical attributes and stream conditions increased uncertainties and relationships between Enterococcus concentrations in the stream among most factors. However, when grouping sites by similar watershed and sediment characteristics, strong significant relationships for water-quality parameters and Enterococcus concentrations in water and sediment were observed. The microcosm study indicated that sediment Enterococcus concentrations for two streams with contrasting sediment properties were stable, except for a considerable increase between day 0 and day 1, with no decay shown for a 31 day period. Collectively, our field and laboratory results revealed that Enterococcus can survive for extended periods under both dynamic and stable sediment and water conditions, and that environmental factors can be used to characterize freshwater streams and rivers for Enterococcus concentrations in freshwater streams and rivers.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Pathogens from environmental and anthropogenic sources have the potential to degrade water quality below that required for beneficial use of streams and rivers (Holcomb and Stewart 2020). Fecal indicator bacteria (FIB)—Escherichia coli and Enterococcus—are commonly used as a measure to determine potential fecal contamination in freshwater streams and rivers for the United States Environmental Protection Agency (USEPA) 303(d) impairment determination and beneficial uses (OWRB 2017; USEPA 1986; USEPA 2012). FIB concentrations used to determine human-health risk, PBCR, were established by the USEPA through a series of studies of marine and freshwater beaches in the 1980’s (USEPA 2012). Water thresholds for impairment are related to number of gastrointestinal illnesses versus FIB concentrations (UESPA 1986, 2012). From these previous studies, Enterococcus has been assumed and established as a quality indicator of human-health risk for all recreational waters, including streams and rivers. E. coli is well documented in literature as a quality indicator bacterium in freshwater for predicting human-health hazards and fecal contamination in both lentic and lotic freshwater bodies (Odonkor and Ampofi 2013). Conversely, limited information is available to understand the dynamics of Enterococcus populations in freshwater lotic waterbodies.
There is evidence to suggest from related research, including recent USEPA work, that Enterococcus in freshwater bodies from environmental and animal sources may not be the best indicator for human-health risk (Cloutier and McLellan 2017; USEPA 2010). Previous studies have deducted that due to the number of non-human Enterococcus sources in the environment such as animal feces, soils, plants, and decaying matter that Enterococcus can replicate and survive outside of enteric environments (Boehm and Sassoubre 2014; Byappanahalli et al. 2012; Devane et al. 2020). Enterococcus is often used as a primary stable indicator in brackish or saline waters, whereas E. coli is often considered a more sensitive indicator in freshwater environments for fecal contamination (Jin et al. 2004). Recent research indicates that temporal and spatial factors due to climate change, seasonality and environmental conditions may impact how we currently assess waterbodies for fecal contamination, specifically Enterococcus as an indicator in freshwater (Petersen and Hubbart 2020; Teixeira et al. 2020). Furthermore, these factors may have an impact on FIB concentrations in streams and rivers as bacteria colonies have been shown as dynamic and in constant flux between the sediment and stream column (Litton et al. 2010; Stocker et al. 2016).
Bed sediments in streams are known to be stable reservoirs for the persistence and proliferation of Enterococcus in the environment that have the potential to be reintroduced into the water column through flow changes and bed disturbances within the stream (Bradshaw et al. 2016; Brinkmeyer et al. 2015; Haller et al. 2009). A study by Stocker et al. (2019) indicated that FIB can display persistence in periphyton and can contribute as a source of Enterococcus in sediment and the water column. Sediment and submerged aquatic vegetation may provide a reservoir of Enterococcus populations that do not correspond with external contaminant sources (Badgley et al. 2010). Therefore, additional research is needed to quantify environmental factors that may play roles in Enterococcus survivability.
Our research team is not currently aware of similar research that has been completed in for Enterococcus in freshwater streams in Oklahoma or elsewhere. With more than 260,000 km of rivers and streams and 88,000 km of shoreline, Oklahoma is known for its water recreation and tourism opportunities (OWRB 2020). However, Oklahoma currently has over 12,000 km of streams that are listed on the 2020 303(d) list for both E. coli and Enterococcus, which are used for primary body contact recreation indicators (PBCR) as defined in Chapter 45 of Oklahoma Water Quality Standards (ODEQ 2020; OWRB 2020). The results of this study are intended provide insight on how to approach fecal indicator bacteria analyses for beneficial use criteria and identify any factors that can be used to predict bacteria loads when developing monitoring strategies in freshwater streams and rivers. In this paper, we describe results from a field and microcosm study in six Oklahoma streams to evaluate Enterococcus survivability in streams and potential environmental factors that have influence on their persistence in the environment.
Methods
Field study
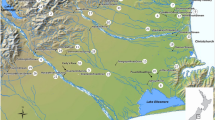
Six perennial Oklahoma streams representing variable sediment types, flow conditions and ecoregions were monitored for FIB and water quality weekly for 10 weeks from July–September 2021 in the stream water column and benthic substrate. Water quality parameters, sediment and water samples, and hydrologic measurements were collected to evaluate and compare the stream reaches. Stream sampling points were selected to represent varied site conditions (e.g., urban, rural, ecoregion) and associated stream reaches were listed as impaired for both Enterococcus and E. coli in the most recent 2020 USEPA 303(d) list (ODEQ 2020). The sites monitored were located in the Upper Neosho-Grand (n = 5) and Upper Canadian basins (n = 1) to provide a geographic contrast to evaluate between varied stream types in Oklahoma (Fig. 1). Stream sediments were characterized during site reconnaissance and ranged from silty sand to medium gravel. A particle size distribution using method ASTM 6913 was performed in the laboratory to confirm the median particle size (D50) for each stream. Drainage area for each site was calculated from the pour point at the site location and delineated using U.S. Geological Survey (USGS) Stream Stats (Smith and Esralew 2010). Percent impervious area was calculated using the 2019 National Land Cover Dataset Imperviousness class from the USGS and using the Extract by Mask tool in ArcGIS 10.8 to clip the raster layer to the watershed area as delineated in USGS Stream Stats. Percent imperviousness for the purposes of this study was determined where percent imperviousness of the layer was greater than or equal to 10% of the raster grids, which is a typically used cutoff for when rivers and streams begin to erode, and sediment has the potential to be transported in the stream (Chithra et al. 2015). Sediment samples were sent to an external laboratory to determine percent organic matter for each sediment using Loss on Ignition (LOI) methods (Ball et al. 1964). Sampling visits were conducted during expected baseflow conditions to reduce potential variability of external influences (i.e., runoff), based on historical precipitation and stream flow for the sampling period of July through September in Oklahoma. However, samples were collected weekly, regardless of precipitation or change in streamflow, which allowed for analysis of variable hydrologic conditions.
Site information collected on ten weekly occasions included hydrologic conditions, water samples, sediment samples, water-quality parameters and other relevant watershed and stream information. Specifically, water-quality parameters (pH, dissolved oxygen [DO], water temperature [T], specific conductance [SC], turbidity, total suspended solids [TSS]), and hydrologic parameters (stream discharge, and precipitation [nearest Oklahoma Mesonet station 24 h precipitation]) were collected along with bacteria water and sediment samples. Bacteria samples were collected based on USGS methods for collecting water samples (USGS 2014). A representative water sample (well-mixed, with adequate flow) was collected at the thalweg of the stream. Samples were stored on ice during field transport and lab storage and were processed within a 24 h hold time. Additional stream water was collected in 1-l polypropylene bottles and cooled to < 6 °C for total suspended solids and turbidity analysis. Antecedent dry period days were calculated from daily Oklahoma Mesonet rainfall (Brock et al. 1995; McPherson et al. 2007). Water quality parameters (pH, DO, SC, T) were measured in-situ using an Orion Star™ A325 portable multimeter (Thermo Fisher Scientific, Waltham, MA). Turbidity was measured with a Hach® 2100Q (Hach Company, Loveland, CO) portable turbidity meter. TSS was calculated by following ASTM 2540D. Stream discharge was measured using a SonTek FlowTracker2® Handheld-ADV® (SonTek YSI, San Diego, CA).
Microcosm study
A microcosm study was conducted from two streams, Cat and Walnut, to replicate stream conditions in a controlled environment to understand survivability of bacteria cells. Sample water and sediment from the upper 5 cm of the benthic stream substrate in the thalweg were collected in 290 ml sterile polystyrene bottles during April 2021. Stream temperatures in the water column were 17 °C and 16 °C, respectively, for Cat and Walnut during time of sediment collection. A total of 22 bottles for each site were collected with approximately 150 ml of stream water and 100 ml of sediment by volume. The microcosm trial was set up in an analogous manner as those routinely performed using soils (Schmidt and Scow 1997). The microcosm container tops (polystyrene collection bottles) were loosely covered with aluminum foil and the sides and bottoms were wrapped tight with aluminum foil to simulate a controlled, dark environment. The containers were held at a constant room temperature of 22 °C in a laboratory and placed on a horizontal orbital shaker at 100 revolutions per minute to encourage aerobic mixing as found in natural streambed conditions. For each day of analysis, two bottles from each site were removed randomly from the storage location on days 0, 1, 2, 5, 10, 17, 24 and 31, and preserved and processed using methods described below in Sect. “Microbiological analysis”. Day 0, when the samples were collected, were processed upon arrival to the lab within 24 h to determine initial concentrations of FIB from stream conditions. A decay rate was calculated for a 31 day period for each microcosm based on methods from Anderson et al. (2005) and Badgley et al. (2010) to understand and relate decay rates to similar studies using the following equation:
where r = decay rate, Nt = Enterococcus Most Probable Number (MPN) 100 ml−1 at time t, N0 = Enterococcus MPN 100 ml−1 at time zero, and t = time (31 days). The magnitude of the r value is relative where a positive value indicates positive growth, and a negative value indicates decay.
Microbiological analysis
Bacteria water samples were processed using the IDEXX most probable number (MPN) methods as defined under SM9223B and SM9230D for E. coli and Enterococcus concentrations, respectively (Baird et al. 2012). Sediment samples were stored in the dark at < 6 °C and processed within 24 h for Enterococcus using sodium pyrophosphate microbial detachment and soil dispersion methods modified from Ogram et al. (2007). A 2% sodium pyrophosphate solution was developed by mixing the tetrasodium pyrophosphate with sterile, reverse osmosis (RO) water, and then adjusting the pH to 7.0. Sediment samples were processed by carefully decanting the water from the top of the bottle using a sterile serological pipetter. Next, 200 ml of pyrophosphate solution was added to the bottle containing saturated sediment and the samples were dispersed by capping the bottles and manually shaking them vigorously for two minutes before they were placed on a horizontal orbital shaker at 200 revolutions per minute for 15 min. Serial dilutions were made (1:100, 1:500, 1:1000) from the sediment and pyrophosphate samples with buffered (7.0 pH) sterile RO water. Diluted samples were processed as water samples using standard methods (SM9230D) as previously described in this section.
Quality control
Quality control samples, which included duplicates, field blanks and lab blanks, for field FIB enumeration were conducted at a rate of 5% for all water and sediment samples collected. Average duplicate results were ± 22% (SD = 15%) for all samples and no positive counts resulted from field or laboratory blanks. All field and laboratory water-quality meters were calibrated per specification standards once per week. Turbidity readings were taken five times for each sample and the median value was reported. TSS sample blanks and duplicates were performed at a rate of 5% of samples. TSS sample duplicates were within ± 5% for each sample and lab blanks reported a < 0.01% change in filter mass.
Data analysis
Data were analyzed using Microsoft Excel® software and R Studio statistical software to evaluate statistical inferences between bacteria, water quality and hydrologic metrics. Correlations between water quality, sediment, hydrology, and bacteria parameters were analyzed using the ‘stats’ package within R using the correlation and Pearson functions. A Pearson correlation matrix was used to evaluate trends, specifically for parameters related to Enterococcus sediment and water concentrations, and a Welch’s t test was used to determine significance. Prior to the correlation analysis, data were evaluated for skewness and log-transformations were performed (Helsel et al. 2020). A Kruskal–Wallis test in R was performed to evaluate differences among means of watershed characteristics from Table 1 (n = 6) (R Core Team 2013). For bacteria concentrations, a geometric mean was used to normalize right skewed data and is often used for regulatory limit reports for primary body contact recreation for Enterococcus and E. coli (OWRB 2017). A log-linear regression (α = 0.05) was used to evaluate the field sampling time-series Enterococcus sediment concentration data (R Core Team 2013). An antecedent dry period was calculated using a custom script in R by calculating consecutive run length of days less than 2.54 mm precipitation for the sampling days and site locations. The closest Oklahoma Mesonet stations were spatially matched to the water-quality sampling locations in ArcGIS.
Results and discussion
Site information collected for watershed and soil characteristics are presented in Table 1. Drainage area (DA), impervious drainage percentage (IA), particle size (D50) and percentage organic matter (OM) were analyzed by ranking parameters. A Kruskal–Wallis test was performed, and a significant difference (p < 0.001) was shown between all sites for all parameters. From these results, two groups were identified for further exploration of factors based on significant differences (p < 0.001) in DA, D50 and particle texture class from a Kruskal–Wallis test between the groups. No significant difference (p > 0.05) between the groups for all sites was identified for impervious drainage and percent organic matter. However, two of the smaller sites (Location [ID] 2 and 5) were subdrainages of the larger drainages of ID 2 and 3, respectively (Table 1). Hogshooter and Walnut were hydrologically disconnected from the other watersheds. The two groups identified for further analysis based on similar stream characteristics were Group 1: site ID 1 (Bird), 5 (Hominy) and 6 (Walnut), and Group 2: site ID 2 (Cat), 3 (Dog) and 4 (Hogshooter).
Field study
Summary statistics from sediment and water samples from six creeks were monitored for ten weekly sampling events for FIB and water-quality parameters and results are presented in Table 2.
Enterococcus in sediment
Samples were collected over 10 consecutive weeks to identify trends between weeks and understand background levels of Enterococcus in streambed sediments during a primary body contact recreation sampling period. Overall, sediment Enterococcus concentrations were variable between sampling weeks for all locations. Only one significant relationship was determined from a log-linear regression for concentration versus time at Hominy (p < 0.001), where a decreasing trend was shown over the sampling period. No other significant relationships (p > 0.05) were determined for the other locations. The maximum recorded concentration for all sites was Bird on July 26 at 3951 MPN/g wet sediment, and the minimum concentration for all sites was 1.3 MPN/g wet sediment at Walnut on August 31. The geometric mean for all sites was 122 MPN/g wet sediment and geometric means ranged from 18 MPN/g wet sediment at Walnut to 472 MPN/g wet sediment at Bird (Fig. 2).
Notably, sediment samples from the week of July 27 and August 17 showed a mean increase of 79% (SD ± 16%) in Enterococcus sediment concentrations. Average ADP at the nearest Oklahoma Mesonet station for those days at all sampling locations was 1.6 days for precipitation less than 2.5 mm and an average precipitation of 3.6 mm whereas on all the other sampling dates the ADP was 3.3 days and an average precipitation of 1.3 mm in the previous 24 h period. The relationship between ADP and FIB concentrations is evaluated further in Sect. “Environmental factor correlation”. The time-series results indicate that Enterococcus concentrations in sediment, regardless of location or time, are consistent with previous studies showing viable streambed populations that have the potential to interact with the water column (Brinkmeyer et al. 2015).
Environmental factor correlation
Summary statistics (mean, minimum, maximum and standard deviation) were calculated for the 12 stream metrics collected over a period of 10 sampling visits (Table 2). Log transformations were performed for discharge, turbidity, total suspended solids, E. coli water, Enterococcus water, and Enterococcus sediment based on methods from Helsel et al. (2020) to reduce skewness. Results from the Pearson correlation matrix values ranged from a strong linear positive correlation of 1 to a strong linear negative correlation of − 1 and significance was determined as p < 0.05. Values between ± 0.3 and 0.7 are moderately correlated, values less than ± 0.3 are weakly correlated and values greater than ± 0.7 are strongly correlated, in respect to positive or negative values. Factors were explored by each stream for all factors and by the two groups previously identified by site characteristics. A correlation matrix with significant parameters is displayed in Fig. 3.
Pearson correlation matrix of all parameters measured from field water samples and associated soil, geographical and hydrologic data. The correlation matrix values range from a strong linear positive correlation of 1 indicating a very strong positive correlation to a negative correlation of − 1 indicating a very strong negative linear relationship. Values and blocks shown are those that showed a significant (p < 0.05) relationship from a t test. Acronym explanation: T water temperature, SC specific conductance, DO dissolved oxygen, ADP antecedent dry period, OM organic matter, D50 median particle size, DA drainage area, IA impervious area. Parameters that were log-transformed are indicated by “log” before the description
Significant parameters (p < 0.05) related to Enterococcus water concentrations included moderate positive correlations to E. coli water concentrations (0.5), median particle size (D50) (0.47), percent impervious area (IA) (0.42), and 24 h precipitation (0.41). Weak negative correlations were observed for Enterococcus water concentrations to percent drainage area (DA) (− 0.5), dissolved oxygen (DO) (− 0.41), discharge (− 0.4) and antecedent dry period (ADP) (− 0.38). Enterococcus sediment concentrations displayed moderate positive correlations to E. coli (0.45), Enterococcus water (0.4), and weak positive correlations to IA (0.38), percent organic matter (OM%) (0.38), and turbidity (0.36). Negative weak correlations were shown for Enterococcus sediment to conductivity (− 0.38) and dissolved oxygen (DO) (− 0.38). Based on correlation comparisons, Enterococcus in the sediment does not appear to be significantly influenced by hydrology but does appear to correlate to sediment differences such as OM% and turbidity. However, Enterococcus water concentrations and precipitation and discharge were moderately correlated, which corresponds to previous studies showing precipitation influence on FIB water concentrations in freshwater streams from external influences (Ibekwe et al. 2011). ADP displayed a moderate negative correlation for both water and sediment Enterococcus concentrations, indicating that potential dry periods allow for concentrations to decrease in the water column due to reduced fluctuation in hydrology that could disrupt sediment from increased runoff. Previous research similarly found that ADP has the potential to influence FIB by creating a flushing effect in the stream sediments and can potentially be used as a predicting factor for bacteria concentrations (Christian et al. 2020; Phillips et al. 2011).
Relationships between E. coli and Enterococcus in both the sediment and water are key considerations for evaluating fecal indicators as E. coli is used as a primary indicator of recreational criteria and is often related to Enterococcus concentrations (Stocker et al. 2019). We found that E. coli was not significantly related to sediment (TSS, OM, D50) or other water-quality parameters except for turbidity (0.48). However, E. coli was moderately correlated to sediment and water Enterococcus concentrations, which corresponds to results in previous studies of freshwater streams where sediment was found to be a significant contributor to E. coli and Enterococcus in the water column (Brinkmeyer et al. 2015). Similarly, others have indicated relationships between watershed characteristics (i.e., percent imperviousness and watershed area) and E. coli. that corresponds with our correlation results for both fecal indicator bacteria (Chen and Chang 2014). Correlation of these two fecal indicators is important because there is evidence that suggests water-quality monitoring for human health can be impacted by naturalized bacteria that are potential reservoirs and sources of contamination in freshwaters (Devane et al. 2020).
Hydrologic characteristics within watersheds and stream reaches are often used to evaluate water-quality trends for abiotic and biotic factors (Bojarczuk et al. 2018; Economy et al. 2019). Discharge showed a significant moderate negative correlation (− 0.4) for Enterococcus water concentrations and no correlation with Enterococcus concentrations, potentially indicating that small fluctuations in flow are not as representative for evaluating Enterococcus concentrations without additional water-quality parameters measured such as turbidity and TSS. Discharges for this study were magnitudes smaller than what would typically occur during the spring or fall precipitation events and discharge could play a more important role for correlating FIB concentrations during high flow conditions (Garbrecht et al. 2004). Additionally, smaller order streams and mixed land use, as most of the streams in this study represent, potentially have higher hydrologic variability and influence from precipitation events than higher order streams on FIB concentrations (Dila et al. 2018; Zhang et al. 2020). Furthermore, drainage area showed a moderate negative correlation to Enterococcus water correlations, which could be due to potential dilution from precipitation and other watershed inputs (i.e., mixed land use) that could influence Enterococcus concentrations in the stream column (Islam et al. 2017). No significant relationship for E. coli, Enterococcus sediment or water and TSS was determined for this study, which may be indicative of the distribution of streambed particle sizes and external influences of suspended particles. Other studies have shown that turbidity is often a stronger predictor of fecal indicators, and that the particle-bound Enterococcus relationship is not well-understood (Suter et al. 2011).
Based on Group 1 and Group 2 identified earlier in Sect. “Field study”, factors between each group were explored to determine if watershed and water-quality characteristics potentially influenced Enterococcus concentrations in streams. A log–log regression was performed to evaluate prediction between Enterococcus sediment and water. Results showed that Group 1 had an R2 of 0.003 and Group 2 had an R2 of 0.51 and a significant difference (p < 0.05) was shown between groups for Enterococcus water and sediment from a Welch’s t test (Fig. 4).
Scatter plot of log Enterococcus water versus log Enterococcus sediment concentrations from water and sediment samples collected during the field study over a period of 10 sampling events for Group 1: site ID 1 (Bird), 5 (Hominy), and 6 (Walnut), and Group 2: site ID 2 (Cat), 3 (Dog), and 4 (Hogshooter). Linear trend lines are displayed and next to each line are associated equations and R2 values. MPN most probable number
Stream characteristics between Group 1 and Group 2 were evaluated to determine if relationships existed between Enterococcus water and sediment concentrations. Significant differences between means (p < 0.05) resulted for DO, pH, discharge, Enterococcus water, OM, TSS, D50, DA, IA, turbidity, and E. coli water. Conversely, no statistical differences (p > 0.05) were found for Enterococcus sediment, temperature, conductivity, precipitation, and ADP.
Relationships within each group were then compared to determine where relationships exist, if any, and if any parameters correlated to Enterococcus sediment and water concentrations. Results indicate that within Group 1, discharge (0.56), turbidity (0.56), OM (0.54), DA (0.53), and D50 (0.45) showed significant moderate correlations within Enterococcus sediment. Escherichia coli (0.43) and turbidity (0.4) resulted in a moderate positive correlation with Enterococcus water concentration, and conductivity (SC) (− 0.52) and ADP (− 0.54) were moderately negative correlated with Enterococcus sediment. Within Group 2, however, Enterococcus sediment indicated significant positive moderate correlations for OM (0.54), E. coli (0.48), IA (0.46), water temperature (0.46), and a negative moderate correlation for D50 (− 0.5) and ADP (− 0.48), while Enterococcus water concentrations showed positive moderate correlations between precipitation (0.55), E. coli water (0.48), turbidity, (0.5), TSS (0.48)), water temperature (0.41) and a negative moderate correlation for ADP (− 0.52). The relationship within Group 2 for Enterococcus water and Enterococcus sediment had a significant strong positive correlation (0.71), whereas no significant relationship was found in Group 1. The similarities between groups for both Enterococcus sediment and water concentrations were turbidity and E. coli, and ADP and OM for Enterococcus sediment.
Previous research has indicated that sediment and particles are related to persistence of Enterococcus in the water column, and our results similarly demonstrate that smaller drainages may be easier to predict the concentrations of Enterococcus from hydrological (precipitation and discharge) and sediment characteristics (turbidity, TSS, D50) (Myers and Juhl 2020). From the sediment characteristics, TSS, turbidity, OM and D50 were significantly different between each group. Brinkmeyer et al. (2015) found that most Enterococcus in the water column were correlated with suspended sediment from silt to fine sand grains, and Haller et al. (2009) showed that smaller particles have the potential to resuspend FIB and have higher interaction with the water column. Between the two groups, Group 2 had a larger mean size substrate (gravel size particles), lower OM, lower discharge, and lower mean turbidity and TSS, which resulted in reduced variability between Enterococcus concentrations in the sediment and water. In larger streams and river drainages, where suspended sediment and higher OM from higher turbulent discharge is possible, such as found in Group 2 in this study, Enterococcus concentrations in sediment and water may be highly variable due to the continuous interaction between the streambed and water column (Grant et al. 2011).
Differences between sites were highly variable for all site characteristics, hydrology, and water-quality parameters versus Enterococcus concentrations in sediment and water. Conversely, similarities existed between groupings of sites (i.e., drainage area) and could provide insight when selecting sites for monitoring and evaluation of water quality and impairment for Enterococcus. Similar research has revealed that grouping watershed and stream characteristics can be important when developing spatial and temporal monitoring studies (Piorkowski et al. 2014; Stocker et al. 2016). We found that when evaluating streams for Enterococcus concentrations, hydrologic and geologic factors such as discharge, sediment (OM, turbidity, TSS, D50), antecedent dry period, and drainage and impervious area may be influential on where to monitor streams and expected relationships of environmental factors. Furthermore, water-quality parameters (TDO and pH) were shown in this study to be significantly different depending on the watershed and could be important considerations when evaluating Enterococcus levels within in stream. The variability of these characteristics was shown to increase uncertainty of predictors for determining Enterococcus concentrations, regardless of stream conditions (i.e., water-quality parameters).
Microcosm study
A microcosm study for two streams, Cat, and Walnut, was performed for a period of 31 day to investigate the sediment Enterococcus concentrations. The geometric means for Enterococcus concentrations were 729 MPN/g and 7 MPN/g wet sediment (n = 8), for Cat and Walnut, respectively. Maximum and minimum values were 2055 MPN/g and 439 MPN/g wet sediment for Cat, and 17 MPN/g and 4 MPN/g wet sediment for Walnut. A time-series plot for both microcosms is presented in Fig. 5. A Pearson correlation with a paired t test was performed to determine correlation between Cat and Walnut Enterococcus sediment concentrations and results indicated a significant (p < 0.01) strong positive correlation (0.76). For both microcosms, the Enterococcus concentrations increased between Day 0 and Day 1 before exponentially declining to stabilization around Day 10–17. The decay rates for Cat and Walnut were calculated for Day 0 and Day 31 from Eq. 1 and resulted in r = − 0.032 and r = 0.001, respectively. From these values, no discernable difference in concentrations was shown for the study period with a slight decay in Cat and neutral growth for Walnut.
Throughout the 31 day study period, both microcosms with contrasting substrate types, organic matter and Enterococcus concentrations showed persistence in Enterococcus viability under stable conditions. A comparable study to our results by Kim and Wuertz (2015) indicated a rapid tenfold increase in Enterococcus counts for the initial 2–3 days followed by a gradual decay and stabilization in numbers over a 40 day period. Similarly, decay rates and survival of Enterococcus in a microcosm study by Haller et al. (2009) were shown for a period of 50 day, whereas E. coli and total coliforms appeared to decrease to non-detectable concentrations. Other related research conducted in mesocosm studies have shown that Enterococcus and FIB decay was significantly reduced in sediment and organic matter (Tiwari 2019). Furthermore, aquatic vegetation such as periphyton has been shown to play a key role in Enterococcus survivability and growth (Stocker et al. 2019). Sediment for our study was collected from the upper benthic substrate and had the potential for inclusion of periphyton and biofilm that accumulated from the natural stream conditions.
Since our microcosm experiments were under no-light conditions, the Enterococcus colonies may have experienced the rapid growth shown early in the time period due to abundance of organic and plant matter before metabolizing the available nutrients resulting in a decay and stabilization in concentrations and is analogous to conclusions from studies by Kim and Wuertz (2015) and Zimmer-Faust et al. (2017). Enterococcus has been found to persist in many different environments (e.g., soil and plant matter) regardless of external inputs such as fecal contamination (Staley et al. 2014). Additionally, organic carbon and nutrient inputs have been found to stimulate growth of FIB in stream sediments and enhance population stability (Korajkic et al. 2019). Many of the streams in the study region have dense canopy cover, high potential for nutrient and organic carbon inputs and mobile substrates, which could enhance Enterococcus survivability.
Our microcosm experiment along with previous experiments provide convincing evidence that benthic streambed sediments under stable conditions have the potential to be reservoirs and sources of Enterococcus. Given the dynamic nature of mobile streambeds, resuspension of these sediment-laden Enterococcus could increase the potential for these fecal indicators to persist within the stream water column for extended periods without external inputs (e.g., stormwater runoff) as evident in studies relating E. coli and streambed sediments (Garzio-Hadzick et al. 2010; Stephenson et al. 1982). Therefore, sediment sources could create interferences with accurately assessing human-health risk and stream impairment criteria. More research is needed to understand the in-situ relationship of streambed sediment influences on Enterococcus concentrations in freshwater streams.
Conclusions
Enterococcus is often used to determine recreational water-quality for the purposes of limiting or preventing potential gastrointestinal illness. However, questions remain on the validity of using Enterococcus to make regulatory decisions given the potential for persistence in the environment without external inputs of fecal sources. Additionally, limited information exists on the relationships between water quality, geography, stream substrate properties, and hydrologic conditions that have the potential to influence Enterococcus concentrations in the stream water column. Our study aimed to understand stream dynamics in the field and laboratory to assess potential persistence in the environment and relate stream factors to Enterococcus concentrations. Results indicate, in general, that hydrologic conditions, watershed area, sediment properties and multiple water-quality parameters are correlated to Enterococcus concentrations in the water column and sediments. Furthermore, relationships between sediment and water Enterococcus sediment existed when grouping sites by geographical and sediment characteristics. The microcosm Enterococcus sediment study corresponded with the field sampling study in that concentrations remained stable throughout the study period except for during the first day after the start of the trial. Conditions as in the first days of the microcosm study could also occur in the streambed where variations in sediment Enterococcus concentrations may exist due to external inputs (e.g., rainfall runoff) and mobile beds. Implications from this work emphasize that more research is needed to evaluate Enterococcus as a regulatory indicator, given counts have the potential to remain viable in recreational freshwater streams and are often ubiquitous in concentrations above the regulatory thresholds for a majority of the recreational season. This study indicates that monitoring plans should consider environmental factors as influencers on Enterococcus concentrations within freshwater streams.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Anderson KL, Whitlock JE, Harwood VJ (2005) Persistence and differential survival of fecal indicator bacteria in subtropical waters and sediments. Appl Environ Microbiol 71(6):3041–3048. https://doi.org/10.1128/AEM.71.6.3041-3048.2005
Badgley BD, Nayak BS, Harwood VJ (2010) The importance of sediment and submerged aquatic vegetation as potential habitats for persistent strains of enterococci in a subtropical watershed. Water Res 44(20):5857–5866. https://doi.org/10.1016/j.watres.2010.07.005
Baird R et al (2012) Standard methods for the analysis of water and wastewater (22nd edn.) Section 9223B and 2540D: Washington, D.C. American Public Health Association, American Public Health Association, American Water Works Association, and Water Pollution Control Federation
Ball DF (1964) Loss-on-ignition as an estimate of organic matter and organic carbon in non-calcareous soils. J Soil Sci 15:84–92
Boehm AB, Sassoubre LM (2014) Enterococci as indicators of environmental fecal contamination. In: Enterococci: from commensals to leading causes of drug resistant infection [Internet]. Massachusetts eye and ear infirmary. https://www.ncbi.nlm.nih.gov/sites/books/NBK190421/. Accessed 12 Aug 2022
Bojarczuk A, Jelonkiewicz Ł, Lenart-Boroń A (2018) The effect of anthropogenic and natural factors on the prevalence of physicochemical parameters of water and bacterial water quality indicators along the river Białka, southern Poland. Environ Sci Pollut Res 25(10):10102–10114. https://doi.org/10.1007/s11356-018-1212-2
Bradshaw JK, Snyder BJ, Oladeinde A, Spidle D, Berrang ME, Meinersmann RJ, Oakley B, Sidle RC, Sullivan K, Molina M (2016) Characterizing relationships among fecal indicator bacteria, microbial source tracking markers, and associated waterborne pathogen occurrence in stream water and sediments in a mixed land use watershed. Water Res 101:498–509. https://doi.org/10.1016/j.watres.2016.05.014
Brinkmeyer R, Amon RMW, Schwarz JR, Saxton T, Roberts D, Harrison S, Ellis N, Fox J, DiGuardi K, Hochman M, Duan S, Stein R, Elliott C (2015) Distribution and persistence of Escherichia coli and Enterococci in stream bed and bank sediments from two urban streams in Houston, TX. Sci Total Environ 502:650–658. https://doi.org/10.1016/j.scitotenv.2014.09.071
Brock FV, Crawford KC, Elliott RL, Cuperus GW, Stadler SJ, Johnson HL, Eilts MD (1995) The Oklahoma Mesonet: a technical overview. J Atmos Oceanic Technol 12:5–19
Byappanahalli MN, Nevers MB, Korajkic A, Staley ZR, Harwood VJ (2012) Enterococci in the environment. Microbiol Mol Biol Rev 76(4):685–706. https://doi.org/10.1128/MMBR.00023-12
Chen HJ, Chang H (2014) Response of discharge, TSS, and E. coli to rainfall events in urban, suburban, and rural watersheds. Environ Sci 16(10):2313–2324. https://doi.org/10.1039/C4EM00327F
Cloutier DD, McLellan SL (2017) Distribution and differential survival of traditional and alternative indicators of fecal pollution at freshwater beaches. Appl Environ Microbiol. https://doi.org/10.1128/AEM.02881-16
Chithra SV, Nair MH, Amarnath A, Anjana NS (2015) Impacts of impervious surfaces on the environment. Int J Eng Sci Invention 4(5):27–31
Christian L, Epps T, Diab G, Hathaway J (2020) Pollutant concentration patterns of in-stream urban stormwater runoff. Water 12(9):2534. https://doi.org/10.3390/w12092534
Devane ML, Moriarty E, Weaver L, Cookson A, Gilpin B (2020) Fecal indicator bacteria from environmental sources; strategies for identification to improve water quality monitoring. Water Res 185:116204. https://doi.org/10.1016/j.watres.2020.116204
Dila DK, Corsi SR, Lenaker PL, Baldwin AK, Bootsma MJ, McLellan SL (2018) Patterns of host-associated fecal indicators driven by hydrology, precipitation, and land use attributes in great lakes watersheds. Environ Sci Technol 52(20):11500–11509. https://doi.org/10.1021/acs.est.8b01945
Economy LM, Wiegner TN, Strauch AM, Awaya JD, Gerken T (2019) Rainfall and streamflow effects on estuarine Staphylococcus aureus and fecal indicator bacteria concentrations. J Environ Qual 48(6):1711–1721. https://doi.org/10.2134/jeq2019.05.0196
Garbrecht J, Van Liew M et al (2004) Trends in precipitation, streamflow, and evapotranspiration in the Great Plains of the United States. J Hydrol Eng 9(5):360–367
Garzio-Hadzick A, Shelton DR, Hill RL, Pachepsky YA, Guber AK, Rowland R (2010) Survival of manure-borne E. coli in streambed sediment: effects of temperature and sediment properties. Water Res 44(9):2753–2762. https://doi.org/10.1016/j.watres.2010.02.011
Grant SB, Litton-Mueller RM, Ahn JH (2011) Measuring and modeling the flux of fecal bacteria across the sediment-water interface in a turbulent stream. Water Resour Res. https://doi.org/10.1029/2010WR009460
Haller L, Amedegnato E, Poté J, Wildi W (2009) Influence of freshwater sediment characteristics on persistence of fecal indicator bacteria. Water Air Soil Pollut 203(1):217–227. https://doi.org/10.1007/s11270-009-0005-0
Helsel DR, Hirsch RM, Ryberg KR, Archfield SA, Gilroy EJ (2020) Statistical methods in water resources: U.S. Geological Survey Techniques and Methods, book 4, chap. A3, 458 p., https://doi.org/10.3133/tm4a3
Holcomb DA, Stewart JR (2020) Microbial indicators of fecal pollution: recent progress and challenges in assessing water quality. Curr Environ Health Rep 7(3):311–324. https://doi.org/10.1007/s40572-020-00278-1
Ibekwe AM, Lesch SM, Bold RM, Leddy MB, Graves AK (2011) Variations of indicator bacteria in a large urban watershed. Trans ASABE 54:10
Islam MMM, Hofstra N, Islam MdA (2017) The Impact of environmental variables on Faecal indicator bacteria in the Betna River Basin. Bangladesh Environ Processes 4(2):319–332. https://doi.org/10.1007/s40710-017-0239-6
Jin G, Englande AJ, Bradford H, Jeng H-W (2004) Comparison of E. coli, Enterococci, and Fecal Coliform as indicators for Brackish water quality assessment. Water Environ Res 76(3):245–255. https://doi.org/10.2175/106143004X141807
Kim M, Wuertz S (2015) Survival and persistence of host-associated Bacteroidales cells and DNA in comparison with Escherichia coli and Enterococcus in freshwater sediments as quantified by PMA-qPCR and qPCR. Water Res 87:182–192. https://doi.org/10.1016/j.watres.2015.09.014
Korajkic A, Wanjugi P, Brooks L, Cao Y, Harwood VJ (2019) Persistence and decay of fecal microbiota in aquatic habitats. Microbiol Mol Biol Rev 83(4):e00005-19. https://doi.org/10.1128/MMBR.00005-19
Litton RM, Ahn JH, Sercu B, Holden PA, Sedlak DL, Grant SB (2010) Evaluation of chemical, molecular, and traditional markers of Fecal contamination in an effluent dominated urban stream (world). American Chemical Society, ACS Publications. https://doi.org/10.1021/es101092g
McPherson RA, Fiebrich C, Crawford KC, Elliott RL, Kilby JR, Grimsley DL, Martinez JE, Basara JB, Illston BG, Morris DA, Kloesel KA, Stadler SJ, Melvin AD, Sutherland AJ, Shrivastava H (2007) Statewide monitoring of the mesoscale environment: a technical update on the Oklahoma Mesonet. J Atmos Oceanic Technol 24:301–321
Myers EM, Juhl AR (2020) Particle association of Enterococcus sp. increases growth rates and simulated persistence in water columns of varying light attenuation and turbulent diffusivity. Water Res 186:116140. https://doi.org/10.1016/j.watres.2020.116140
ODEQ (Oklahoma Department of Environmental Quality). (2020) Water Quality in Oklahoma 2020 Integrated Report. https://www.deq.ok.gov/water-quality-division/watershed-planning/integrated-report/. Accessed 25 June 2022
Odonkor ST, Ampofi JK (2013) Escherichia coli as an indicator of bacteriological quality of water: an overview. Microbiol Res 4:1–11
Ogram A, Castro H, Chauhan A (2007) Methods of soil microbial community analysis, p 652–662. In: Hurst CJ, Crawford RL, Garland JL, Lipson DA, Mills AL, Stetzenbach LD (eds) Manual of environmental microbiology, 3rd edn. https://doi.org/10.1128/9781555815882.ch53
OWRB (Oklahoma Water Resources Board) (2017) Oklahoma water quality standards. Oklahoma administrative Code 785:45. oar.state.ok.us. Accessed 25 June 2022
OWRB (Oklahoma Water Resources Board). (2020) Status of Water Quality Monitoring in Oklahoma 2019–2020. https://www.owrb.ok.gov/quality/monitoring/StatusReport.pdf. Accessed 25 June 2022
Petersen F, Hubbart JA (2020) Advancing understanding of land use and physicochemical impacts on fecal contamination in mixed-land-use watersheds. Water 12(4):1094. https://doi.org/10.3390/w12041094
Piorkowski GS, Jamieson RC, Hansen LT, Bezanson GS, Yost CK (2014) Characterizing spatial structure of sediment E. coli populations to inform sampling design. Environ Monitor Assess 186(1):277–291. https://doi.org/10.1007/s10661-013-3373-2
Phillips MC, Solo-Gabriele HM, Reniers AJHM, Wang JD, Kiger RT, Abdel-Mottaleb N (2011) Pore water transport of Enterococci out of beach sediments. Mar Pollut Bull 62(11):2293–2298. https://doi.org/10.1016/j.marpolbul.2011.08.049
R Core Team (2013) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. ISBN 3-900051-07-0, URL: http://www.R-project.org/. Accessed 12 Aug 2022
Schmidt SK, Scow KM (1997) Use of soil bioreactors and microcosms in bioremediation research. In: Hurst CJ, Knudsen GR, McInerney MJ, Stetzenbach LD, Walter MV (eds) Manual of environmental microbiology. Published by ASM Press, Washington, D.C, pp 822–829
Smith SJ, Esralew RA (2010) StreamStats in Oklahoma—drainage-basin characteristics and peak-flow frequency statistics for ungaged streams (Report No. 2009–5255; Scientific Investigations Report). USGS Publications Warehouse. https://doi.org/10.3133/sir20095255
Staley C, Dunny GM, Sadowsky MJ (2014) Chapter four—environmental and animal-associated Enterococci. In: Sariaslani S, Gadd GM (Eds) Advances in Applied Microbiology (Vol. 87, pp. 147–186). Academic Press. https://doi.org/10.1016/B978-0-12-800261-2.00004-9
Stephenson GR, Rychert RC (1982) Bottom sediment: a reservoir of Escherichia coli in rangeland streams. Rangeland Ecol Manag/j Range Manag Archiv 35(1):119–123
Stocker MD, Rodriguez-Valentín JG, Pachepsky YA, Shelton DR (2016) Spatial and temporal variation of fecal indicator organisms in two creeks in Beltsville. Maryland Water Quality Res J 51(2):167–179. https://doi.org/10.2166/wqrjc.2016.044
Stocker MD, Smith JE, Hernandez C, Macarisin D, Pachepsky Y (2019) Seasonality of E. coli and Enterococci concentrations in creek water, sediment, and periphyton. Water Air Soil Poll 230(9):223. https://doi.org/10.1007/s11270-019-4263-1
Suter E, Juhl A, O’Mullan G (2011) Particle association of Enterococcus and total bacteria in the lower hudson River Estuary, USA. J Water Resour Proect. https://doi.org/10.4236/jwarp.2011.310082
Teixeira P, Salvador D, Brandão J, Ahmed W, Sadowsky MJ, Valério E (2020) Environmental and adaptive changes necessitate a paradigm shift for indicators of fecal contamination. Microbiol Spectr. https://doi.org/10.1128/microbiolspec.ERV-0001-2019
Tiwari A, Kauppinen A, Pitkänen T (2019) Decay of Enterococcus faecalis, Vibrio cholerae and MS2 coliphage in a laboratory mesocosm under brackish beach conditions. Front Public Health 7:269
USEPA (United States Environmental Protection Agency). (1986) Bacteriological Water Quality Criteria for Marine and Fresh Recreational Waters. EPA- 440/5-84-002. US Environmental Protection Agency, Office of Water Regulations and Standards, Cincinnati, OH, USA
USEPA (United States Environmental Protection Agency). (2010) Sampling and Consideration of Variability (Temporal and Spatial) for Monitoring of Recreational Waters. EPA-823-R-10-005 US Environmental Protection Agency, Washington, DC, USA
USEPA (United States Environmental Protection Agency). (2012) Recreational Water Quality Criteria. Office of Water 820-F-12-058. https://www.epa.gov/sites/production/files/2015-10/documents/rwqc2012.pdf. Accessed 12 Aug 2022
USGS (U.S. Geological Survey) (2014) National field manual for the collection of water-quality data: U.S. Geological Survey Techniques of Water-Resources Investigations, book 9, chaps. A1-A10. http://pubs.water.usgs.gov/twri9A
Zhang X, Zhi X, Chen L, Shen Z (2020) Spatiotemporal variability and key influencing factors of river fecal coliform within a typical complex watershed. Water Res 178:115835. https://doi.org/10.1016/j.watres.2020.115835
Zimmer-Faust AG, Thulsiraj V, Marambio-Jones C, Cao Y, Griffith JF, Holden PA, Jay JA (2017) Effect of freshwater sediment characteristics on the persistence of fecal indicator bacteria and genetic markers within a Southern California watershed. Water Res 119:1–11. https://doi.org/10.1016/j.watres.2017.04.028
Acknowledgements
The authors would like to acknowledge contributions from research assistants at the Oklahoma Water Survey: Julie Bahr, Paul Baumann, Hannah Curtis, Alejandra Hernandez-Santana, Derek Kendzor, Kassidy Long, Jacob McDoulett, and Yvette Wiley.
Funding
This research was partially funded by a USGS 104(b) project—2020 OK198B and through research funding provided by the Office of the Vice President for Research at the University of Oklahoma to the Oklahoma Water Survey at the University of Oklahoma.
Author information
Authors and Affiliations
Contributions
All authors contributed to study conception and design; GMG and RST: collected and analyzed data; JRV: provided project oversight; GMG wrote the manuscript; all authors edited and reviewed manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Graves, G.M., Vogel, J.R. & Tanner, R.S. Investigation of environmental factors on Enterococcus survival in Oklahoma streams. Aquat Sci 85, 34 (2023). https://doi.org/10.1007/s00027-023-00933-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00027-023-00933-3