Abstract
Phosphorus (P) is a vital nutrient for algal growth. Aside from soluble reactive P (SRP), organic P (OP) is used by algae via alkaline phosphatase (AP) hydrolysis, which can play an important role in supplying P. Enzymatically-hydrolysable OP (EHP) can potentially be used as an indicator of bioavailability of P other than SRP in natural waters. We investigated the ecological significance of alkaline phosphatase activity (APA), EHP concentration and P turnover time in the inflowing rivers of Lake Taihu (Taihu) during three hydrologic periods. Results indicated high SRP concentration and low SRP demand by algal suppressed APA in the inflowing rivers, the highest proportion of OP mineralization rate (v) to the maximum reaction velocity of AP (Vmax) is only 14.7%. P turnover time of the inflowing rivers was generally from 3 to 7 days and in exceptional cases, it could exceed 10 days. The high EHP reserve and the sufficient AP for OP mineralization render the rivers a significant source of utilizable OP, further exacerbating eutrophication of Taihu.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Eutrophication is one of the most challenging environmental problems in nutrient-enriched waters (Qin 2009; Smith and Schindler 2009; Wang and Wang 2009; Zhu et al. 2015; Schindler et al. 2016). It is generally accepted that key nutrient inputs include nitrogen (N) and phosphorus (P) (Hecky and Kilham 1988; Lowery 1998; Gurkan et al. 2006; Bhagowati and Ahamad 2018). Phosphorus is an essential element for plant growth and its input has long been recognized as necessary to maintain primary production. P is also a vital nutrient stimulating problematic harmful cyanobacterial blooms and so its control is of prime importance in mitigating the global proliferation of such blooms in freshwater ecosystems (Daniel et al. 1998; Schindler and Vallentyne 2008).
P in water can be separated into numerous forms, such as total P (TP), total dissolved P (TDP), organic phosphorus (OP), molybdate reactive P or soluble reactive P (SRP), molybdate unreactive P (Xie et al. 2011). Among the various forms of P, SRP is the most biologically available and readily usable for phytoplankton and bacteria (Cembella et al. 1982; Kwon et al. 2011). However, OP often represents the largest phosphorus pool in water and can greatly exceed SRP concentrations (Shun et al. 1994; Oh et al. 2005; Lin et al. 2018). OP may also be an important P storage for phytoplankton when depletion of SRP occurs in the surface water (Oh et al. 2002; Kwon et al. 2011; Lin et al. 2018).
Microbial utilization of OP through enzymatic hydrolysis is one of the most important pathways in the P cycle (Cembella et al. 1982; Suzumura et al. 1998). Alkaline phosphatase (AP) hydrolyzes OP to SRP under SRP depleted conditions (Kuenzler and Perras 1965; Jin et al. 2006; Xie et al. 2011; Li et al. 2015; Lin et al. 2018). Enzyme assays with soluble substrate showed that extracellular AP is important in providing P for phytoplankton growth, particularly during blooms induced by nutrient over-enrichment (Harke et al. 2012; Okubo et al. 2014; Ivančić et al. 2016). Alkaline phosphatase-hydrolyzable OP or enzymatically-hydrolysable OP (EHP) have potential use as indicators of bioavailability of P other than SRP in natural waters (Shun et al. 1994), and the enzyme plays an important role in P supply (Zhang et al. 2007). The most common practice to evaluate OP utilization under P limited conditions is to determine alkaline phosphatase activity (APA) (Hoppe 2003; Ryzhakov and Stepanova 2016).
Lake Taihu (Taihu) is the third largest freshwater lake in China, located in the Changjiang (Yangtze) Delta. It has a surface area of about 2338 km2 and mean depth of 1.9 m (Qin et al. 2007). Taihu serves as a drinking water source for approximately 20 million local inhabitants and plays an important role in the Jiangsu Province economy. However, in recent decades, cyanobacterial blooms occur regularly from late spring through fall throughout the lake (Chen et al. 2003; Xu et al. 2015). P is the primary limiting nutrient in winter and spring in Taihu, while in summer and autumn N and P co-limit algal growth (Xu et al. 2010). Controlling P supply is thus a critical step in reversing eutrophication and the restoration of the lake (Zhu et al. 2013).
Inflowing rivers are a key source of nutrient pollution in Taihu (Zhao et al. 2011). Previous studies have shown that about 80% of the nutrients discharged to Taihu came from these rivers and surface runoff (Xie et al. 2007; Wang et al. 2011). Therefore, monitoring and controlling nutrient discharge from inflowing rivers is a necessary component of effective lake management (Zhao et al. 2011; Wang and Bi 2016).
Past research and development of water quality standards have focused on TP, TDP and SRP. However, few studies have investigated the contributions of EHP and APA to P cycling in rivers discharging to Taihu. In view of their potential ecological significance, we measured EHP, APA, OP mineralization rate (v) and P turnover time (t) in inflowing rivers of Taihu during various hydrologic periods. The objective of the study was to evaluate the rate of P turnover, P bioavailability and mobility. These results could help show the impact of inflowing rivers on Taihu’s algal bloom potential.
Materials and methods
Sampling times and sites
The rainy season in the Taihu Basin begins in April, with water levels usually reaching their peak in late July, while the lowest water levels occur between February and March every year, according to long-term hydrology data from the Lake Taihu Basin Authority of Ministry of Water Resources (http://www.tba.gov.cn/channels/43.html). Therefore, we chose February, May and August 2009 to represent low, moderate and high flow periods. Samples were collected from 21 to 25 February, from 24 to 26 May and from 26 to 28 August of 2009. Nine rivers discharging to Taihu (30°55′40″–31°32′58″N; 119°52′32″–120°36′10″E) were selected for this study. In1 (Zhihu port), In2 (Hengtang river) and In3 (Wangyu river) are polluted by industry and they are located on the north side of Taihu. Se4 (Xujiang river), Se5 (Zhi port) and Se6 (Cao port) are polluted by domestic sewage and they are located on the east side of Taihu. Ag7 (Zhongtang river), Ag8 (Changxing port) and Ag9 (Jiapu port) are heavily influenced by agricultural pollution and they are located on the southwest side of Taihu (Fig. 1). Three sampling sites were set up along the upper, middle and lower reaches of each river, amounting to a total of 27 sample sites examined in our study. The downstream sampling sites were at the intersection of each river and Taihu.
Sample collection and laboratory analysis
Water samples were collected at 0.5 m below the surface. The transparency of water was measured by Secchi depth (SD). Water temperature, pH, dissolved oxygen (DO), turbidity and chlorophyll-a (Chl-a) were measured in situ using a Yellow Springs Instruments (YSI) 6600 V2 multi-sensor sonde. TP, TDP, SRP, EHP and APA were measured in the laboratory. TP and TDP were determined by persulfate digestion and then determined by spectrophotometry at 700 nm after reaction with molybdate, ascorbic acid, and trivalent antimony (Jin and Tu 1990). A flow-injection system (Skalar Co., http://www.skalar.com) was used to determine SRP concentrations in Whatman GF/F membrane-filtered water samples.
EHP was measured as follows: 1 mL 1.0 mol/L Tris buffer (pH 8.2) and 5 mL pure chloroform was added to 100 mL of initial water sample, with subsequent incubation at 30 °C for 5 days (Peters 1981; Chrost et al. 1986). Subsamples were filtered on Whatman GF/F filters, followed by SRP concentration measurements using a flow-injection system (Skalar Co., http://www.skalar.com). EHP concentration was calculated as the difference between the SRP concentration in the incubated subsample and the initial water sample. All samples were run in triplicate.
APA was determined by analyzing the production of p-nitrophenol (pNP) from model substrate p-nitrophenyl phosphate (pNPP) hydrolyzed by AP in the initial water samples (Berman 1970; Sayler et al. 1979; Joner et al. 2000). In the assay, 1 mL 0.5 mol/L Tris butter (pH 8.4) and 2 mL millimolar pNPP were added to 2 mL of sample, followed by incubation at 30 °C for 6 h in the dark (Gao et al. 2006). The absorbance at 410 nm was measured and enzymatic activity was calculated from absorbance using a standard curve based on pNP. Eight gradient millimolar pNPP concentrations ranging from 0.02 to 2.0 mmol/L were chosen to determine the following kinetic constants and APA was determined at the final substrate concentration of 1.0 mmol/L. All samples were run in triplicate.
It was found that the reaction rate was determined by enzyme concentration and substrate concentration if other conditions remained constant. If enzyme concentration remains unchanged, the reaction rate presents a complex process of change when substrate concentration is changed. The Michaelis–Menten equation is a velocity equation of the relationship between the initial enzymatic reaction rate and substrate concentration of the enzymatic reaction. It includes the enzymatic reaction rate and other factors, such as inhibitors. The affinity between enzyme and substrate as well as potential reaction rate of extracellular AP hydrolysis can be inferred by using the Michaelis–Menten equation (Tabatabai and Bremner 1971; Manzoni and Porporato 2009; Allison et al. 2010; Wang and Post 2013; Hui et al. 2013):
where v (nmol/L/min) is OP mineralization rate; Vmax (nmol/L/min) is the maximum reaction velocity of AP; Km (μmol/L) is the Michaelis constant; [S] (μmol/L) is substrate concentration. We used Lineweaver–Burk transformation (\(\frac{1}{v} = \frac{{K_{m} }}{{V_{\hbox{max} } }} \cdot \frac{1}{\left[ S \right]} + \frac{1}{{V_{\hbox{max} } }}\)) of the Michaelis–Menten equation to calculate Vmax and Km (Zhou et al. 2004). Km and Vmax were computed by linear regression analysis on the values obtained in the assay (Han and Srinivasan 1969; Zhou et al. 2004; Xu et al. 2018). v was calculated by using Km, Vmax combined with the EHP as substrate concentration. P turnover time t (min) was calculated with EHP and v (Larionova et al. 2007; Tischer et al. 2015).
Mean value and standard deviation of the variation of the upper, middle and lower reaches of each river were all calculated using Microsoft Excel 2007 software. Pearson correlation coefficients were determined with a Statistical Program for Social Sciences (SPSS) 16.0 software. Significance levels were reported as significant (0.01 ≤ p < 0.05) or highly significant (p < 0.01).
Results
Dynamic parameters (K m, V max) of AP in the inflowing rivers of Taihu
Average Km value of AP in the nine inflowing rivers ranged from 14 to 61 µmol/L during low flow period, from 19 to 52 µmol/L during moderate flow period and from 10 to 34 µmol/L during high flow period. The highest Km values of AP appeared in rivers polluted by domestic sewage (Se4, Se5, Se6) and the lowest Km values of AP appeared in rivers polluted by agriculture (Ag7, Ag8, Ag9) during the various hydrologic periods. A comparison of average SRP concentrations of the three types of rivers (Gao et al. 2016) shows that SRP concentrations in the rivers polluted by domestic sewage (Se4, Se5, Se6) contained the highest SRP and rivers polluted by agriculture (Ag7, Ag8, Ag9) were the lowest.
Km values of AP showed no significant difference (p = 0.691) between the low flow period and the moderate flow period. Km values of AP in the high flow period showed a significant difference with the low flow period (p = 0.021) and with the moderate flow period (p = 0.024). The linearly dependent coefficients between Km and SRP are 0.65, 0.60 and 0.54 during low, moderate and high flow periods. Statistical results showed there were no significant differences in pH among the low, moderate and high flow periods (p ≥ 0.068). Km values of AP were negatively dependent on temperature (linearly dependent coefficient r = − 0.87).
Average Vmax of AP in the inflowing rivers ranged from 2.7 to 10.6 nmol/L/min during the low flow period, from 1.6 to 10.9 nmol/L/min during the moderate flow period and from 1.5 to 13.6 nmol/L/min during the high flow period. There was no significant difference in the Vmax of AP among the low, moderate and high flow periods (p ≥ 0.644).
Organic P mineralization rate (v) in the inflowing rivers of Taihu
OP mineralization rate v in the inflowing rivers increased gradually from the low flow period to the high flow period. There was no significant difference between the low and moderate flow periods (p = 0.289) as well as between the moderate and high flow periods (p = 0.098), but there was a significant difference between the low and high flow periods (p = 0.032). There was no significant difference among the three types of rivers (p > 0.9). It shows that OP mineralization rate v was much lower than Vmax of AP in the inflowing rivers by comparing Vmax of AP with v in the nine sampled rivers of Taihu. The highest proportion of v to Vmax (v/Vmax) is only 14.7%. The average v/Vmax for rivers polluted by industry ranging from 2.6 to 12.5%, for rivers polluted by agriculture ranging from 0 to 11.3% and for rivers polluted by domestic sewage ranging from 1.3 to 14.7%. v/Vmax increased gradually from the low flow period to the high flow period, which means that the AP enzymatic hydrolysis reaction in the inflowing rivers increased from the low flow period to the high flow period.
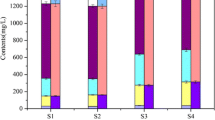
v and Vmax/Km had similar variation trend during the low, moderate and high flow periods (Fig. 3). The variation trends of EHP concentration and OP mineralization rate v in the inflowing rivers were also similar (Fig. 4), and OP mineralization rate increased with the increase of EHP concentration. The proportion of EHP to TP (EHP/TP) was nearly equal to the proportion of SRP to TP (SRP/TP) during the moderate flow period and EHP/TP was higher than SRP/TP during the high flow period in the three types of inflowing rivers (Fig. 5).
P turnover time (t) in the inflowing rivers of Taihu
We measured P turnover time in the inflowing rivers of Taihu and there were no significant differences among low, moderate and high flow periods (p ≥ 0.167), nor were there significant differences among the three types of rivers (p ≥ 0.08). EHP concentrations in the inflowing rivers ranged from 0.011 to 0.113 mg/L and it generally needed half a week to a week (range 3–7 days) to be mineralized (Fig. 6).
Discussion
Characteristics of AP in the inflowing rivers of Taihu
Michaelis constant Km and Vmax of AP are responsible for the major part of the phosphatase activity (Bogé et al. 2014). Km is used to measure the affinity between enzyme and substrate (Tischer et al. 2015). A smaller Km value represents a stronger affinity (Wang et al. 2006; Xiao et al. 2018). The temporal patterns showed that Affinity between AP and substrate in the inflowing rivers during high flow period was higher than that during low and moderate flow periods. Vmax is used to characterize the intrinsic catalytic rate of the enzyme. When the substrate is combined completely with the enzyme, the maximum reaction velocity can be obtained.
In Fig. 2a–c it appears that Km values of AP are also high when SRP concentrations are high. The variation characteristic of SRP concentrations and Km values in the nine inflowing rivers during low, moderate and high flow periods (Fig. 2a–c) indicates that affinity between AP and substrate is inversely related to SRP concentrations in the inflowing rivers. Natural environmental conditions such as pH, substrate concentration and temperature could markedly affect the APA (Hui et al. 2013). However, there were no significant differences in pH among the low, moderate and high flow periods and Km values of AP were negatively dependent on temperature indicated that the high water temperature (average 31 °C) during high flow period increased the affinity between AP and substrate in the inflowing rivers. These results were similar to Gershenson et al. (2009) and Tan et al. (2018).
Based on different habitats of Taihu, Lu et al. (2009) reported the Km values of AP in the grass type zone, Gonghu Bay zone, Zhushan Bay zone, Meiliang Bay zone, the central zone and estuary zone of the western bank were 20.50, 20.11, 15.70, 13.34, 11.75 and 9.17 µmol/L respectively. Km values of AP in Taihu were lower than those in the inflowing rivers and it indicated that affinity between AP and substrate in Taihu was higher than that in the inflowing rivers. Vmax of AP in Taihu in 2008 ranged from 7.70 to 26.9 nmol/L/min and the average Vmax was 13.70 nmol/L/min (Lu et al. 2009). The average Vmax of AP in Taihu in 1999 was 191 nmol/L/min (Gao et al. 2004). The average Vmax of AP in Taihu in 2011 was 16.0 nmol/L/min under no turbulent condition (Ding et al. 2016). The Vmax of AP in Taihu in 2014 increased from 39 nmol/L/min under calm condition to 458 nmol/L/min under long-term moderate wind condition, averaging 184 nmol/L/min (Chao et al. 2017). Vmax of AP in Taihu was far higher than that in the inflowing rivers. Taihu is a typical shallow hyper-eutrophic lake, with an average depth of 1.9 m and it is disturbed by wind-induced wave most of time (Qin et al. 2007). Wind-induced wave disturbances can cause rapid sediment resuspension (Tammeorg et al. 2013). APA and TP in sediment were resuspended in the water column with the sediment resuspension, adding P cycling rate and bio-available P (Chao et al. 2017). As a result, algae largely grew and reproduced, accompanying high Vmax of AP.
AP is often repressed at high SRP concentrations and reactive at low SRP concentrations (Jansson et al. 1988). APA has therefore been used as a SRP deficiency indicator in algae and in natural plankton populations. Previous studies indicate that phytoplankton and bacterioplankton began to synthesize AP to hydrolyze phosphate esters when SRP was depleted (Berman 1970; Chrost et al. 1986). Previous work carried out on Taihu showed that APA was significantly increased when SRP concentrations were less than 0.020 mg/L and 0.020 mg/L is the SRP threshold to activate APA of phytoplankton (Gao et al. 2000). SRP concentrations were far higher than 0.020 mg/L in most of the inflowing rivers (Fig. 2a–c) and APA was repressed in the rivers. However, SRP was only about 8% of TP and the average SRP concentration was 0.0137 mg/L in Taihu (Gao et al. 2004). As a result, Vmax of AP in Taihu was far higher than that in the inflowing rivers.
Phytoplankton are influenced by a wide range of factors including water temperature, water column stability, flushing rate, irradiance, and available nutrients (Havens et al. 2019; Islam et al. 2013). Many types of algae flourish in lakes with poor water flow and a flush of water suppress blooms (Islam et al. 2013; Havens et al. 2019). It has been documented that, on a global scale, cyanobacteria are favored by warmer water temperatures (Kosten et al. 2012). However, water temperature of the nine inflowing rivers had no significant difference with Taihu during low (p = 0.274), moderate (p = 0.830) and high flow periods (p = 0.425). Water transparency of the nine inflowing rivers had no significant difference with Taihu during low (p = 0.812), moderate (p = 0.074) and high flow periods (p = 0.826). So, water temperature and irradiance were not the main causes for slow algal growth in the inflowing rivers. Taihu is relative stable because of the water control gates (Wu and Kong 2009) and the water retention time of the lake is approximately 180 days (Xu et al. 2015).The water retention time of the inflowing rivers were short because of their high flow velocity. Though the algal cells are in a thick (low Reynolds number) environment and cannot sense the movement of water, algae cannot make full use of SRP in the inflowing rivers because of the short water retention time. Therefore, high flow velocity of the inflowing rivers indirectly affected the growth of algae. High flow velocity of rivers is not suitable for algae growth compared with the lake, so the demand for SRP by algae in the inflowing rivers is lower than that in Taihu. This helps explain why Vmax of AP in Taihu was far higher than that in the inflowing rivers. High SRP concentration and low demand for SRP by algae lead to Vmax of AP in the inflowing rivers being lower than that in Taihu.
Impact of OP mineralization rate in the inflowing rivers of Taihu
Km and Vmax represent the characteristics of AP itself, and OP mineralization rate v is the actual reaction rate between AP and substrate EHP. v is affected by the catalytic efficiency of AP, substrate concentration and SRP concentration according to the reversible enzyme-driven reaction principle. However, the two catalytic property parameters Km and Vmax have different characteristics, the decrease of Km and the increase of Vmax means enhanced OP mineralization by simultaneously increasing the reaction velocity v and improving the affinity for the substrate. In order to analyze the relationship between OP mineralization rate v and catalytic efficiency of AP in the inflowing rivers, Vmax/Km is used here as a proxy for the catalytic efficiency of AP (Wang et al. 2018). Figure 3 indicates that OP mineralization rate increases with the increase of AP catalytic efficiency. v accounts for only a small proportion of Vmax and v can potentially increase. The results of Fig. 3 indicate that AP in the inflowing rivers is sufficient for OP mineralization and AP is not the reason for the low OP mineralization rate.
Another possible controlling factor of v is substrate concentration. The substrate refers to phosphatase-hydrolyzable OP in our study, namely EHP. OP mineralization rate increased with the increase of EHP concentration (Fig. 4) indicated that substrate concentration was enough for the enzyme-driven reaction and it was not the reason for the low OP mineralization rates.
The reason for the low OP mineralization rate v in the inflowing rivers was not due to the catalytic efficiency of AP or the substrate (EHP) concentration according to the above analysis, but rather the inhibition by the higher SRP concentration. Compared with Taihu, algae cannot make full use of SRP in the inflowing rivers because of the short water retention time. As a result, SRP in rivers was utilized slowly by algae and the existent higher SRP concentration inhibited the reversible enzyme-driven reaction. Thus, OP mineralization rate v in the inflowing rivers was very low. The high EHP reserve (Fig. 5) and the sufficient AP for OP mineralization point to the inflowing rivers as a potential threat to water quality in Taihu.
The difference of P turnover time (t) between Taihu and its inflowing rivers
Halemejko and Chrost (1984)’s study on Lake Glebokie in Poland found that phosphatases participated in the mineralization of approximately 70% OP in the spring seston after a 60 day decomposition period. During the summer, seston decomposition was accompanied by 90% mineralization of P after 30 days (Halemejko and Chrost 1984). During autumn, after 60 days, 96% of the initial content of OP in the algae was mineralized (Halemejko and Chrost 1984). Hudson et al. (1999) measured phosphorus regeneration rates from 20 temperate freshwater lakes from three major physiographical regions of North America and found regeneration could release an amount of P equal to particulate P pool every 5 days (range 3.5–7 days). P turnover time was from 275.9 to 20.3 min in different ecotype sites of Taihu and the most rapid P turnover time was on the order of several minutes (Gao et al. 2006). P turnover time in the inflowing rivers of Taihu was far longer than the P turnover time of Taihu (Fig. 6). One reason was that Taihu had longer hydraulic residence times, more algal biomass and higher demand for SRP by algae than inflowing rivers. The difference in structure and form of OP compounds might be another reason for the difference in P turnover time. A variety of soluble organic P compounds is present in the upper layer of water (Halemejko and Chrost 1984). These compounds include phospholipids, phosphomononucleotides and their derivatives (Kleerekoper 1953; Lean and Nalewajko 1976). Sugar phosphates and aminophosphoric acids contain very stable C–P bonds (Demain et al. 1965). Phosphoric monoester or diesters contain easily hydrolysable P–O–C bonds (Halemejko and Chrost 1984). Further studies will focus on the relationship between the specific molecular structure of EHP and P turnover time in different types of inflowing rivers.
Conclusions
The affinity between AP and substrate is inversely related to SRP concentration in the inflowing rivers and the affinity in Taihu was higher than that in the inflowing rivers. High SRP concentration and low SRP demand by algae suppressed APA in the inflowing rivers. As a result, the highest proportion of v to Vmax is only 14.7%. The EHP concentrations in the inflowing rivers ranged from 0.011 to 0.113 mg/L and half a week to a week (range 3–7 days) was needed to mineralize it. In exceptional cases, the P turnover time could exceed 10 days. The high EHP reserve and the sufficient AP for OP mineralization render the inflowing rivers a potential threat to water quality and cyanobacterial bloom formation in Taihu, especially during warm, maximum irradiance summer periods when maximum bloom potential exists.
References
Allison SD, Wallenstein MD, Bradford MA (2010) Soil-carbon response to warming dependent on microbial physiology. Nat Geosci 3:336–340
Berman T (1970) Alkaline phosphatases and phosphorus availability in Lake Kinneret. Limnol Oceanogr 15:663–674
Bhagowati B, Ahamad KU (2018) A review on lake eutrophication dynamics and recent developments in lake modeling. Ecohydrol Hydrobiol. https://doi.org/10.1016/j.ecohyd.2018.03.002
Bogé G, Lespilette M, Jamet D, Jamet JL (2014) Analysis of the role of DOP on the particulate phosphatase activity in Toulon Bay (N.W. Mediterranean Sea, France). Mar Pollut Bull 86:342–348
Cembella AD, Antia NJ, Harrison PJ (1982) The utilization of inorganic and organic phosphorus compounds as nutrients by eukaryotic microalgae: a multidisciplinary perspective: part I. CRC Crit Rev Microbiol 10:317–391
Chao JY, Zhang YM, Kong M, Zhuang W, Wang LM, Shao KQ, Gao G (2017) Long-term moderate wind induced sediment resuspension meeting phosphorus demand of phytoplankton in the large shallow eutrophic Lake Taihu. PLoS One 12:e0173477
Chen YW, Qin BQ, Teubner K, Dokulil MT (2003) Long-term dynamics of phytoplankton assemblages: Microcystis-domination in Lake Taihu, a large shallow lake in China. J Plankton Res 25:445–453
Chrost RJ, Siuda W, Albrecht D, Overbeck J (1986) A method for determining enzymatically hydrolyzable phosphate (EHP) in natural waters. Limnol Oceanogr 31:662–667
Daniel TC, Sharpley AN, Lemunyon JL (1998) Agricultural phosphorus and eutrophication: a symposium overview. J Environ Qual 27:251–257
Demain AL, Burg RW, Hendlin D (1965) Excretion and degradation of ribonucleic acid by Bacillus subtilis. J Bacteriol 89:640–646
Ding YQ, Qin BQ, Xu H, Wang XD (2016) Effects of sediment and turbulence on alkaline phosphatase activity and photosynthetic activity of phytoplankton in the shallow hyper-eutropic Lake Taihu, China. Environ Sci Pollut Res 23:16183–16193
Gao G, Gao XY, Qin BQ (2000) Experimental study on the PO43–P threshold of the alkaline phosphatase activity in Taihu Lake. J Lake Sci 12:353–358 (in Chinese)
Gao G, Qin BQ, Zhu GW, Fan CX, Ji J (2004) Seasonal variation of alkaline phosphatase activity in Meiliang Bay, Lake Taihu. J Lake Sci 16:245–251 (in Chinese)
Gao G, Zhu GW, Qin BQ, Chen J, Wang K (2006) Alkaline phosphatase activity and the phosphorus mineralization rate of Lake Taihu. Sci China Ser D 49:176–185
Gao YX, Song YZ, Yu JH, Zhu GW (2016) Spatial and temporal distribution characteristics of different forms of phosphorus in three sorts of rivers around Lake Taihu. Environ Sci 37:1404–1412 (in Chinese)
Gershenson A, Bader NE, Cheng WX (2009) Effects of substrate availability on the temperature sensitivity of soil organic matter decomposition. Glob Change Biol 15:176–183
Gurkan Z, Zhang JJ, Jørgensen SE (2006) Development of a structurally dynamic model for forecasting the effects of restoration of Lake Fure, Denmark. Ecol Model 197:89–102
Halemejko GZ, Chrost RJ (1984) The role of phosphatases in phosphorus mineralization during decomposition of lake phytoplankton blooms. Arch Hydrobiol 101:489–502
Han YW, Srinivasan VR (1969) Purification and characterization of β-glucosidase of alcaligenes faecalis. J Bacteriol 100:1355–1363
Harke MJ, Berry DL, Ammerman JW, Gobler CJ (2012) Molecular response of the bloom-forming cyanobacterium, Microcystis aeruginosa, to phosphorus limitation. Microb Ecol 63:188–198
Havens KE, Ji GH, Beaver JR, Fulton IIIRS, Teacher CE (2019) Dynamics of cyanobacteria blooms are linked to the hydrology of shallow Florida lakes and provide insight into possible impacts of climate change. Hydrobiologia 829:43–59
Hecky RE, Kilham P (1988) Nutrient limitation of phytoplankton in freshwater and marine environments: a review of recent evidence on the effects of enrichment. Limnol Oceanogr 33:796–822
Hoppe HG (2003) Phosphatase activity in the sea. Hydrobiologia 493:187–200
Hudson JJ, Taylor WD, Schindler DW (1999) Planktonic nutrient regeneration and cycling efficiency in temperate lakes. Nature 400:659–661
Hui DF, Mayes MA, Wang GS (2013) Kinetic parameters of phosphatase: a quantitative synthesis. Soil Biol Biochem 65:105–113
Islam MN, Kitazawa D, Hamill T, Park HD (2013) Modeling mitigation strategies for toxic cyanobacteria blooms in shallow and eutrophic Lake Kasumigaura, Japan. Mitig Adapt Strat Glob Change 18:449–470
Ivančić I, Pfannkuchen M, Godrijan J, Djakovac T, Pfannkuchen DM, Korlević M, Gašparović B, Najdek M (2016) Alkaline phosphatase activity related to phosphorus stress of microphytoplankton in different trophic conditions. Prog Oceanogr 146:175–186
Jansson M, Olsson H, Pettersson K (1988) Phosphatases; origin, characteristics and function in lakes. Hydrobiologia 170:157–175
Jin XC, Tu QY (1990) Standard for lake eutrophication investigation. Chinese Environmental Science Press, Beijing (in Chinese)
Jin XC, Wang SR, Zhao HC, Bu QY, Chu JZ, Cui Z, Zhou XN, Wu FC (2006) Effect of lake sediments of different trophic states on alkaline phosphatase activity. Lakes Reserv 11:169–176
Joner EJ, Van Aarle IM, Vosatka M (2000) Phosphatase activity of extra-radical arbuscular mycorrhizal hyphae: a review. Plant Soil 226:199–210
Kleerekoper H (1953) The mineralization of plankton. J Fish Res Board Can 10:283–291
Kosten S, Huszar VLM, Bécares E, Costa LS, Donk EV, Hansson LA, Jeppesen E, Kruk C, Lacerot G, Mazzeo N, Meester LD, Moss B, Lürling M, Nõges T, Romo S, Scheffer M (2012) Warmer climates boost cyanobacterial dominance in shallow lakes. Glob Change Biol 18:118–126
Kuenzler EJ, Perras JP (1965) Phosphatases of marine algae. Biol Bull 128:271–284
Kwon HK, Oh SJ, Yang HS (2011) Ecological significance of alkaline phosphatase activity and phosphatase-hydrolyzed phosphorus in the northern part of Gamak Bay, Korea. Mar Pollut Bull 62:2476–2482
Larionova AA, Yevdokimov IV, Bykhovets SS (2007) Temperature response of soil respiration is dependent on concentration of readily decomposable C. Biogeosciences 4:1073–1081
Lean DRS, Nalewajko C (1976) Phosphate exchange and organic phosphorus excretion by freshwater algae. J Fish Res Board Can 33:1312–1323
Li JH, Wang ZW, Cao X, Wang ZF, Zheng Z (2015) Effect of orthophosphate and bioavailability of dissolved organic phosphorus compounds to typically harmful cyanobacterium Microcystis aeruginosa. Mar Pollut Bull 92:52–58
Lin WT, Zhao DD, Luo JF (2018) Distribution of alkaline phosphatase genes in cyanobacteria and the role of alkaline phosphatase on the acquisition of phosphorus from dissolved organic phosphorus for cyanobacterial growth. J Appl Phycol 30:839–850
Lowery TA (1998) Modelling estuarine eutrophication in the context of hypoxia, nitrogen loadings, stratification and nutrient ratios. J Environ Manag 52:289–305
Lu N, Hu WP, Deng JC, Zhai SH, Chen XM, Zhou XP (2009) Spatial distribution characteristics and ecological significance of alkaline phosphatase in water column of Taihu Lake. Environ Sci 30:2898–2903 (in Chinese)
Manzoni S, Porporato A (2009) Soil carbon and nitrogen mineralization: theory and models across scales. Soil Biol Biochem 41:1355–1379
Oh SJ, Yamamoto T, Kataoka Y, Matsuda O, Matsuyama Y, Kotani Y (2002) Utilization of dissolved organic phosphorus by the two toxic dinoflagellates, Alexandrium tamarense and Gymnodinium catenatum (Dinophyceae). Fish Sci 68:416–424
Oh SJ, Yoon YH, Yamamoto T, Matsuyama Y (2005) Alkaline phosphatase activity and phosphatase hydrolyzable phosphorus for phytoplankton in hiroshima bay, Japan. Ocean Sci J 40:183–190
Okubo Y, Inoue T, Yokota K, Ngoc NM (2014) Availability of different phosphorus forms in agricultural soil to Microcystis aeruginosa. Water Sci Technol 69:1205–1211
Peters RH (1981) Phosphorus availability in Lake Memphremagog and its tributaries. Limnol Oceanogr 26:1150–1161
Qin BQ (2009) Lake eutrophication: control countermeasures and recycling exploitation. Ecol Eng 35:1569–1573
Qin BQ, Xu PZ, Wu QL, Luo LC, Zhang YL (2007) Environmental issues of Lake Taihu, China. Hydrobiologia 581:3–14
Ryzhakov AV, Stepanova IA (2016) Determination of rate of inorganic phosphorus consumption in natural waters using alkaline phosphatase inhibitors. Russ J Gen Chem 86:3015–3017
Sayler GS, Puziss M, Silver M (1979) Alkaline phosphatase assay for freshwater sediments: application to perturbed sediment systems. Appl Environ Microbiol 38:922–927
Schindler DW, Vallentyne JR (2008) The algal bowl: overfertilization of the world’s freshwaters and estuaries. University of Alberta Press, Edmonton
Schindler DW, Carpenter SR, Chapra SC, Hecky RE, Orihel DM (2016) Reducing phosphorus to curb lake eutrophication is a success. Environ Sci Technol 50:8923–8929
Shun Y, Mckelvie ID, Hart BT (1994) Determination of alkaline phosphatase—hydrolyzable phosphorus in natural water systems by enzymatic flow injection. Limnol Oceanogr 39:1993–2000
Smith VH, Schindler DW (2009) Eutrophication science: where do we go from here? Trends Ecol Evol 24:201–207
Suzumura M, Ishikawa K, Ogawa H (1998) Characterization of dissolved organic phosphorus in coastal seawater using ultrafiltration and phosphohydrolytic enzymes. Limnol Oceanogr 43:1553–1564
Tabatabai MA, Bremner JM (1971) Michaelis constants of soil enzymes. Soil Biol Biochem 3:317–323
Tammeorg O, Niemistö J, Möls T, Laugaste R, Panksep K, Kangur K (2013) Wind-induced sediment resuspension as a potential factor sustaining eutrophication in large and shallow Lake Peipsi. Aquat Sci 75:559–570
Tan XP, Machmuller MB, Wang ZQ, Li XD, He WX, Cotrufo MF, Shen WJ (2018) Temperature enhances the affinity of soil alkaline phosphatase to Cd. Chemosphere 196:214–222
Tischer A, Blagodatskaya E, Hamer U (2015) Microbial community structure and resource availability drive the catalytic efficiency of soil enzymes under land-use change conditions. Soil Biol Biochem 89:226–237
Wang C, Bi J (2016) TMDL development for the Taihu Lake’s influent rivers, China using variable daily load expressions. Stoch Env Res Risk Assess 30:911–921
Wang G, Post WM (2013) A note on the reverse Michaelis–Menten kinetics. Soil Biol Biochem 57:946–949
Wang HJ, Wang HZ (2009) Mitigation of lake eutrophication: loosen nitrogen control and focus on phosphorus abatement. Prog Nat Sci 19:1445–1451
Wang J, Liu B, Guo N, Xie P (2006) Alkaline phosphatase activity in four Microcystis aeruginosa species and their responses to nonylphenol stress. Bull Environ Contam Toxicol 76:999–1006
Wang FE, Tian P, Yu J, Lao GM, Shi TC (2011) Variations in pollutant fluxes of rivers surrounding Taihu Lake in Zhejiang Province in 2008. Phys Chem Earth 36:366–371
Wang ZQ, Tian HX, Lu GN, Zhao YM, Yang R, Megharaj M, He WX (2018) Catalytic efficiency is a better predictor of arsenic toxicity to soil alkaline phosphatase. Ecotoxicol Environ Saf 148:721–728
Wu XD, Kong FX (2009) Effects of light and wind speed on the vertical distribution of Microcystis aeruginosa colonies of different sizes during a summer bloom. Int Rev Hydrobiol 94:258–266
Xiao J, Wang SY, Zhou ZJ, Zhang Y, Song CL, Zhou YY, Cao XY (2018) An enzymatic mechanism for balancing the stoichiometry of nitrogen and phosphorus in a shallow Chinese eutrophic lake. Sci Total Environ 630:1071–1077
Xie YX, Xiong ZQ, Xing GX, Sun GQ, Zhu ZL (2007) Assessment of nitrogen pollutant sources in surface waters of Taihu Lake region. Pedosphere 17:200–208
Xie CS, Zhao J, Tang J, Xu J, Lin XY, Xu XH (2011) The phosphorus fractions and alkaline phosphatase activities in sludge. Bioresour Technol 102:2455–2461
Xu H, Paerl HW, Qin BQ, Zhu GW, Gao G (2010) Nitrogen and phosphorus inputs control phytoplankton growth in eutrophic Lake Taihu, China. Limnol Oceanogr 55:420–432
Xu H, Paerl HW, Qin BQ, Zhu GW, Hall NS, Wu Y (2015) Determining critical nutrient thresholds needed to control harmful cyanobacterial blooms in eutrophic Lake Taihu, China. Environ Sci Technol 49:1051–1059
Xu ZR, Wang SB, Wang YA, Zhang J (2018) Growth, extracellular alkaline phosphatase activity, and kinetic characteristic responses of the bloom-forming toxic cyanobacterium, Microcystis aeruginosa, to atmospheric particulate matter (PM2.5, PM2.5-10, and PM>10). Environ Sci Pollut Res 25:7358–7368
Zhang TX, Wang XR, Jin XC (2007) Variations of alkaline phosphatase activity and P fractions in sediments of a shallow Chinese eutrophic lake (Lake Taihu). Environ Pollut 150:288–294
Zhao DH, Cai Y, Jiang H, Xu DL, Zhang WG, An SQ (2011) Estimation of water clarity in Taihu Lake and surrounding rivers using Landsat imagery. Adv Water Resour 34:165–173
Zhou YY, Li JQ, Song CL, Cao XY (2004) Variations and possible source of potentially available phosphorus in a Chinese shallow eutrophic lake. J Freshw Ecol 19:87–96
Zhu MY, Zhu GW, Li W, Zhang YL, Zhao LL, Gu Z (2013) Estimation of the algal-available phosphorus pool in sediments of a large, shallow eutrophic lake (Taihu, China) using profiled SMT fractional analysis. Environ Pollut 173:216–223
Zhu GW, Cui Y, Han XX, Li HY, Zhu MY, Deng JM, Li HP, Chen WM (2015) Response of phytoplankton to nutrient reduction in Shahe Reservoir, Taihu catchment, China. J Freshw Ecol 30:41–58
Funding
This study was funded by the National Natural Science Foundation of China (Grant numbers 41671494, 41501532, 41471446), the Key Research Program of Frontier Sciences, CAS (Grant number QYZDJ-SSW-DQC008), the “One-Three-Five” Strategic Planning of Nanjing Institute of Geography and Limnology, Chinese Academy of Sciences (Grant number NIGLAS2017GH04), Jiangsu Overseas Research & Training Program for University Prominent Young & Middle-aged Teachers and Presidents. H. Paerl was supported by the US National Science Foundation Dimensions in Biodiversity Program (Project 1831096) and the International Science & Technology Cooperation Program of China (Grant number 2015DFG91980).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gao, Y., Zhu, G., Paerl, H.W. et al. A study of bioavailable phosphorus in the inflowing rivers of Lake Taihu, China. Aquat Sci 82, 1 (2020). https://doi.org/10.1007/s00027-019-0673-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00027-019-0673-9