Abstract
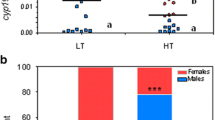
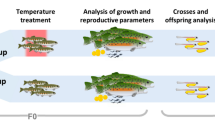
Environmental changes alter the sex fate in about 15% of vertebrate orders, mainly in ectotherms such as fish and reptiles. However, the effects of temperature changes on the endocrine and molecular processes controlling gonadal sex determination are not fully understood. Here, we provide evidence that thyroid hormones (THs) act as co-players in heat-induced masculinization through interactions with the stress axis to promote testicular development. We first demonstrated that the thyroid axis (through thyroid-related genes and T3 levels) is highly active in males during the gonadal development in medaka (Oryzias latipes). Similarly, T3 treatments promoted female-to-male sex reversal in XX embryos. Subsequently, embryonic exposure to temperature-induced stress up-regulated the genes related to the thyroid and stress axes with a final increase in T3 levels. In this context, we show that blocking the stress axis response by the loss of function of the corticotropin-releasing hormone receptors suppresses thyroid-stimulating hormone expression, therefore, heat-induced activation of the thyroid axis. Thus, our data showed that early activation of the stress axis and, in consequence, the TH axis, too, leaves us with that both being important endocrine players in inducing female-to-male reversal, which can help predict possible upcoming physiological impacts of global warming on fish populations.





Similar content being viewed by others
Data availability
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
Abbreviations
- amh:
-
Anti-müllerian hormone
- amhrII:
-
Anti-müllerian hormone receptor type II
- ar:
-
Androgen receptor
- BPI:
-
Brain-pituitary-interrenal
- BPT:
-
Brain-pituitary-thyroid
- CRH:
-
Corticotropin-releasing hormone
- crhb:
-
Corticotropin-releasing hormone b
- Crhr2:
-
Corticotropin-releasing hormone receptor 2
- cyp19a1a:
-
Cytochrome P450, family 19, subfamily A, polypeptide 1a; ovary marker
- dio1:
-
Deiodinase type 1
- dio2:
-
Deiodinase type 2
- dio3:
-
Deiodinase type 3
- foxl2:
-
Forkhead box protein L2
- gsdf:
-
Gonadal soma derived factor; testicular marker
- MM:
-
Methimazole
- R:
-
RU486; mifepristone
- T:
-
Testosterone
- T2:
-
Diiodothyronine; 3,5- diiodotyrosine
- T3:
-
Triiodothyronine; 3, 5, 3´-L-triiodothyronine
- T4:
-
Thyroxine; 3, 5, 3´, 5´-L-tetra-iodothyronine
- THs:
-
Thyroid hormones
- TRH:
-
Thyrotropin-releasing hormone
- TSH:
-
Thyroid-stimulating hormone
- thra:
-
Thyroid hormone receptors a
- thrb:
-
Thyroid hormone receptors b
- TRs:
-
Thyroid receptors
- tshba:
-
Thyroid-stimulating hormone subunit beta a
- tshr:
-
Thyroid-stimulating hormone receptors
References
Capel B (2017) Vertebrate sex determination: evolutionary plasticity of a fundamental switch. Nat Rev Genet 18:675–689
Ashman TL, Bachtrog D, Blackmon H, Goldberg EE, Hahn MW, Kirkpatrick M, Kitano J, Mank JE, Mayrose I, Ming R, Otto SP, Peichel CL, Pennell MW, Perrin N, Ross L, Valenzuela N, Vamosi JC (2014) Tree of sex: a database of sexual systems. Sci Data 1:140015. https://doi.org/10.1038/sdata.2014.15
Hattori RS, Castañeda-Cortés DC, Arias Padilla LF, Strobl-Mazzulla PH, Fernandino JI (2020) Activation of stress response axis as a key process in environment-induced sex plasticity in fish. Cell Mol Life Sci 77:4223–4236. https://doi.org/10.1007/s00018-020-03532-9
Dahlke FT, Wohlrab S, Butzin M, Pörtner H-O (2020) Thermal bottlenecks in the life cycle define climate vulnerability of fish. Science 369:65–70. https://doi.org/10.1126/science.aaz3658
Castañeda Cortés DC, Arias Padilla LF, Langlois VS, Somoza GM, Fernandino JI (2019) The central nervous system acts as a transducer of stress-induced masculinization through corticotropin-releasing hormone B. Dev 146:1–10. https://doi.org/10.1242/dev.172866
Hattori RS, Fernandino JI, Kishil A, Kimura H, Kinno T, Oura M, Somoza GM, Yokota M, Strüssmann CA, Watanabe S (2009) Cortisol-induced masculinization: Does thermal stress affect gonadal fate in pejerrey, a teleost fish with temperature-dependent sex determination? PLoS ONE. https://doi.org/10.1371/journal.pone.0006548
Hayashi Y, Kobira H, Yamaguchi T, Shiraishi E, Yazawa T, Hirai T, Kamei Y, Kitano T (2010) High temperature causes masculinization of genetically female medaka by elevation of cortisol. Mol Reprod Dev 77:679–686. https://doi.org/10.1002/mrd.21203
Yamaguchi T, Yoshinaga N, Yazawa T, Gen K, Kitano T (2010) Cortisol is involved in temperature-dependent sex determination in the Japanese flounder. Endocrinology 151:3900–3908. https://doi.org/10.1210/en.2010-0228
Goikoetxea A, Todd EV, Gemmell NJ (2017) Stress and sex: does cortisol mediate sex change in fish? Reproduction 154:R149–R160. https://doi.org/10.1530/REP-17-0408
Nozu R, Nakamura M (2015) Cortisol Administration induces sex change from ovary to testis in the protogynous wrasse, Halichoeres trimaculatus. Sex Dev 9:118–124. https://doi.org/10.1159/000373902
Chen J, Peng C, Yu Z, Xiao L, Yu Q, Li S, Zhang H, Lin H, Zhang Y (2020) The administration of cortisol induces female-to-male sex change in the protogynous orange-spotted grouper Epinephelus coioides. Front Endocrinol (Lausanne) 11:12. https://doi.org/10.3389/fendo.2020.00012
Fernandino JI, Hattori RS, Kishii A, Strüssmann CA, Somoza GM (2012) The cortisol and androgen pathways cross talk in high temperature-induced masculinization: the 11β-hydroxysteroid dehydrogenase as a key enzyme. Endocrinology 153:6003–6011. https://doi.org/10.1210/en.2012-1517
Castañeda-Cortés DC, Zhang J, Boan AF, Langlois VS, Fernandino JI (2020) High temperature stress response is not sexually dimorphic at the whole-body level and is dependent on androgens to induce sex reversal. Gen Comp Endocrinol 299:113605. https://doi.org/10.1016/j.ygcen.2020.113605
Fernandino JI, Hattori RS, Moreno Acosta OD, Strüssmann CA, Somoza GM (2013) Environmental stress-induced testis differentiation: androgen as a by-product of cortisol inactivation. Gen Comp Endocrinol 192:36–44. https://doi.org/10.1016/j.ygcen.2013.05.024
Simi S, Peter VS, Peter MCS (2017) Zymosan-induced immune challenge modifies the stress response of hypoxic air-breathing fish (Anabas testudineus Bloch): Evidence for reversed patterns of cortisol and thyroid hormone interaction, differential ion transporter functions and non-specific imm. Gen Comp Endocrinol 251:94–108. https://doi.org/10.1016/j.ygcen.2016.11.009
Peter MCS (2011) The role of thyroid hormones in stress response of fish. Gen Comp Endocrinol 172:198–210. https://doi.org/10.1016/j.ygcen.2011.02.023
Little AG, Seebacher F (2014) The evolution of endothermy is explained by thyroid hormone-mediated responses to cold in early vertebrates. J Exp Biol 217:1642–1648. https://doi.org/10.1242/jeb.088880
Mukhi S, Torres L, Patiño R (2007) Effects of larval-juvenile treatment with perchlorate and co-treatment with thyroxine on zebrafish sex ratios. Gen Comp Endocrinol 150:486–494. https://doi.org/10.1016/j.ygcen.2006.11.013
Sharma P, Tang S, Mayer GD, Patiño R (2016) Effects of thyroid endocrine manipulation on sex-related gene expression and population sex ratios in zebrafish. Gen Comp Endocrinol 235:38–47. https://doi.org/10.1016/j.ygcen.2016.05.028
Helmreich DL, Tylee D (2011) Thyroid hormone regulation by stress and behavioral differences in adult male rats. Horm Behav 60:284–291. https://doi.org/10.1016/j.yhbeh.2011.06.003
Westphal NJ, Evans RT, Seasholtz AF (2009) Novel expression of type 1 corticotropin-releasing hormone receptor in multiple endocrine cell types in the murine anterior pituitary. Endocrinology 150:260–267. https://doi.org/10.1210/en.2008-0630
De Groef B, Goris N, Arckens L, Kühn ER, Darras VM (2003) Corticotropin-releasing hormone (CRH)-induced thyrotropin release is directly mediated through CRH receptor type 2 on thyrotropes. Endocrinology 144:5537–5544. https://doi.org/10.1210/en.2003-0526
Denver RJ (1993) Acceleration of anuran amphibian metamorphosis by corticotropin-releasing hormone-like peptides. Gen Comp Endocrinol 91:38–51. https://doi.org/10.1006/gcen.1993.1102
Denver RJ (2021) Stress hormones mediate developmental plasticity in vertebrates with complex life cycles. Neurobiol Stress 14:100301. https://doi.org/10.1016/j.ynstr.2021.100301
Miranda LA, Affanni JM, Paz DA (2000) Corticotropin-releasing factor accelerates metamorphosis in Bufo arenarum: effect on pituitary ACTH and TSH cells. J Exp Zool 286:473–480. https://doi.org/10.1002/(SICI)1097-010X(20000401)286:5%3c473::AID-JEZ4%3e3.0.CO;2-H
Larsen DA, Swanson P, Dickey JT, Rivier J, Dickhoff WW (1998) In vitro thyrotropin-releasing activity of corticotropin-releasing hormone-family peptides in coho salmon, Oncorhynchus kisutch. Gen Comp Endocrinol 109:276–285. https://doi.org/10.1006/gcen.1997.7031
Murata K, Kinoshita M, Naruse K, Tanaka M, Kamei Y (2019) Medaka: biology, management, and experimental protocols. Wiley
Iwamatsu T (2004) Stages of normal development in the medaka Oryzias latipes. Mech Dev 121:605–618. https://doi.org/10.1016/j.mod.2004.03.012
Saito D, Morinaga C, Aoki Y, Nakamura S, Mitani H, Furutani-Seiki M, Kondoh H, Tanaka M (2007) Proliferation of germ cells during gonadal sex differentiation in medaka: Insights from germ cell-depleted mutant zenzai. Dev Biol 310:280–290. https://doi.org/10.1016/j.ydbio.2007.07.039
Arias Padilla LF, Castañeda-Cortés DC, Rosa IF, Moreno Acosta OD, Hattori RS, Nóbrega RH, Fernandino JI (2021) Cystic proliferation of germline stem cells is necessary to reproductive success and normal mating behavior in medaka. Elife 10:e62757. https://doi.org/10.7554/eLife.62757
Nanda I, Kondo M, Hornung U, Asakawa S, Winkler C, Shimizu A, Shan Z, Haaf T, Shimizu N, Shima A, Schmid M, Schartl M (2002) A duplicated copy of DMRT1 in the sex-determining region of the Y chromosome of the medaka, Oryzias latipes. Proc Natl Acad Sci U S A 99:11778–11783. https://doi.org/10.1073/pnas.182314699
Pfaffl MW, Horgan GW, Dempfle L (2002) Relative expression software tool (REST ©) for group-wise comparison and statistical analysis of relative expression results in real-time PCR. Nucleic Acids Res 30:36e. https://doi.org/10.1093/nar/30.9.e36
Sato T, Endo T, Yamahira K, Hamaguchi S, Sakaizumi M (2005) Induction of female-to-male sex reversal by high temperature treatment in medaka, Oryzias latipes. Zool Sci 22:985–988. https://doi.org/10.2108/zsj.22.985
Hattori RS, Gould RJ, Fujioka T, Saito T, Kurita J, Strüssmann CA, Yokota M, Watanabe S (2007) Temperature-dependent sex determination in Hd-rR Medaka Oryzias latipes: gender sensitivity, thermal threshold, critical period, and DMRT1 expression profile. Sex Dev 1:138–146. https://doi.org/10.1159/000100035
Ansai S, Kinoshita M (2014) Targeted mutagenesis using CRISPR/Cas system in medaka. Biol Open 3:362–371. https://doi.org/10.1242/bio.20148177
Vijayan MM, Reddy PK, Leatherland JF, Moon TW (1994) The effects of cortisol on hepatocyte metabolism in rainbow trout: a study using the steroid analogue RU486. Gen Comp Endocrinol 96:75–84
Bandyopadhyay U, Biswas K, Banerjee RK (2002) Extrathyroidal actions of antithyroid thionamides. Toxicol Lett 128:117–127
Sharma P, Patiño R (2013) Regulation of gonadal sex ratios and pubertal development by the thyroid endocrine system in zebrafish (Danio rerio). Gen Comp Endocrinol 184:111–119. https://doi.org/10.1016/j.ygcen.2012.12.018
Kumai Y, Nesan D, Vijayan M, Perry S (2012) Cortisol regulates Na+ uptake in zebrafish, Danio rerio, larvae via the glucocorticoid receptor. Mol Cell Endocrinol 364:113–125. https://doi.org/10.1016/j.mce.2012.08.017
Chen SX, Bogerd J, García-López Á, de Jonge H, de Waal PP, Hong WS, Schulz RW (2010) Molecular cloning and functional characterization of a zebrafish nuclear progesterone receptor1. Biol Reprod 82:171–181. https://doi.org/10.1095/biolreprod.109.077644
Castañeda Cortés DC, Langlois VS, Fernandino JI (2014) Crossover of the hypothalamic pituitary-adrenal/interrenal, -thyroid, and -gonadal axes in testicular development. Front Endocrinol (Lausanne) 5:139. https://doi.org/10.3389/fendo.2014.00139
Chu Y-D, Yeh C-T (2020) The molecular function and clinical role of thyroid stimulating hormone receptor in cancer cells. Cells 9:1730. https://doi.org/10.3390/cells9071730
Szkudlinski MW, Fremont V, Ronin C, Weintraub BD (2002) Thyroid-stimulating hormone and thyroid-stimulating hormone receptor structure-function relationships. Physiol Rev 82:473–502. https://doi.org/10.1152/physrev.00031.2001
Flood DEK, Fernandino JI, Langlois VS (2013) Thyroid hormones in male reproductive development: evidence for direct crosstalk between the androgen and thyroid hormone axes. Gen Comp Endocrinol 192:2–14
Tovo-Neto A, da Silva RM, Habibi HR, Nóbrega RH (2018) Thyroid hormone actions on male reproductive system of teleost fish. Gen Comp Endocrinol 265:230–236. https://doi.org/10.1016/j.ygcen.2018.04.023
Rodrigues MS, Tovo-Neto A, Rosa IF, Doretto LB, Fallah HP, Habibi HR, Nóbrega RH (2022) Thyroid hormones deficiency impairs male germ cell development: a cross talk between hypothalamic-Pituitary-thyroid, and—gonadal axes in Zebrafish. Front Cell Dev Biol 10:865948. https://doi.org/10.3389/fcell.2022.865948
Poppe K, Velkeniers B, Glinoer D (2008) The role of thyroid autoimmunity in fertility and pregnancy. Nat Clin Pract Endocrinol Metab 4:394–405. https://doi.org/10.1038/ncpendmet0846
Wagner MS, Wajner SM, Maia AL (2008) The role of thyroid hormone in testicular development and function. J Endocrinol 199:351–365. https://doi.org/10.1677/JOE-08-0218
Krassas GE, Poppe K, Glinoer D (2010) Thyroid function and human reproductive health. Endocr Rev 31:702–755
Rodrigues MS, Fallah HP, Zanardini M, Malafaia G, Habibi HR, Nóbrega RH (2021) Interaction between thyroid hormones and gonadotropin inhibitory hormone in ex vivo culture of zebrafish testis: An approach to study multifactorial control of spermatogenesis. Mol Cell Endocrinol. https://doi.org/10.1016/j.mce.2021.111331
Swapna I, Rajasekhar M, Supriya A, Raghuveer K, Sreenivasulu G, Rasheeda MK, Majumdar KC, Kagawa H, Tanaka H, Dutta-Gupta A, Senthilkumaran B (2006) Thiourea-induced thyroid hormone depletion impairs testicular recrudescence in the air-breathing catfish, Clarias gariepinus. Comp Biochem Physiol Part A Mol Integr Physiol 144:1–10. https://doi.org/10.1016/j.cbpa.2006.01.017
Morais RDVS, Nóbrega RH, Gómez-González NE, Schmidt R, Bogerd J, França LR, Schulz RW (2013) Thyroid hormone stimulates the proliferation of sertoli cells and single type A spermatogonia in adult zebrafish (Danio rerio) testis. Endocrinology 154:4365–4376. https://doi.org/10.1210/en.2013-1308
Akhlaghi A, Zamiri MJ (2007) Effect of transient prepubertal hypothyroidism on serum testosterone level and seminal characteristics of chickens. Iran J Vet Res 8:23–31. https://doi.org/10.22099/ijvr.2007.2704
Weng Q, Saita E, Watanabe G, Takahashi S, Sedqyar M, Suzuki AK, Taneda S, Taya K (2007) Effect of methimazole-induced hypothyroidism on adrenal and gonadal functions in male Japanese quail (Coturnix coturnix japonica). J Reprod Dev. https://doi.org/10.1262/jrd.19081
Chiao Y-C, Cho W-L, Wang PS (2002) Inhibition of testosterone production by propylthiouracil in rat leydig cells. Biol Reprod 67:416–422. https://doi.org/10.1095/biolreprod67.2.416
Maran RRM (2003) Thyroid hormones: their role in testicular steroidogenesis. Arch Androl 49:375–388. https://doi.org/10.1080/01485010390204968
Buglo E, Sarmiento E, Martuscelli NB, Sant DW, Danzi MC, Abrams AJ, Dallman JE, Züchner S (2020) Genetic compensation in a stable slc25a46 mutant zebrafish: a case for using F0 CRISPR mutagenesis to study phenotypes caused by inherited disease. PLoS ONE 15:e0230566
Galas L, Raoult E, Tonon M-C, Okada R, Jenks BG, Castaño JP, Kikuyama S, Malagon M, Roubos EW, Vaudry H (2009) TRH acts as a multifunctional hypophysiotropic factor in vertebrates. Gen Comp Endocrinol 164:40–50. https://doi.org/10.1016/j.ygcen.2009.05.003
Tagawa M, Hirano T (1991) Effects of thyroid hormone deficiency in eggs on early development of the medaka, Oryzias latipes. J Exp Zool 257:360–366. https://doi.org/10.1002/jez.1402570309
Arjona FJ, Vargas-Chacoff L, Martín Del Río MP, Flik G, Mancera JM, Klaren PHM (2011) Effects of cortisol and thyroid hormone on peripheral outer ring deiodination and osmoregulatory parameters in the Senegalese sole (Solea senegalensis). J Endocrinol 208:323–330. https://doi.org/10.1530/JOE-10-0416
Vijayan MM, Flett PA, Leatherland JF (1988) Effect of cortisol on the in vitro hepatic conversion of thyroxine to triiodothyronine in brook charr (Salvelinus fontinalis Mitchill). Gen Comp Endocrinol 70:312–318. https://doi.org/10.1016/0016-6480(88)90151-7
Szisch V, Papandroulakis N, Fanouraki E, Pavlidis M (2005) Ontogeny of the thyroid hormones and cortisol in the gilthead sea bream, Sparus aurata. Gen Comp Endocrinol 142:186–192. https://doi.org/10.1016/j.ygcen.2004.12.013
Galton VA (1990) Mechanisms underlying the acceleration of thyroid hormone-induced tadpole metamorphosis by corticosterone*. Endocrinology 127:2997–3002. https://doi.org/10.1210/endo-127-6-2997
Politis SN, Servili A, Mazurais D, Zambonino-Infante J-L, Miest JJ, Tomkiewicz J, Butts IAE (2018) Temperature induced variation in gene expression of thyroid hormone receptors and deiodinases of European eel (Anguilla anguilla) larvae. Gen Comp Endocrinol 259:54–65. https://doi.org/10.1016/j.ygcen.2017.11.003
Zak MA, Manzon RG (2019) Expression and activity of lipid and oxidative metabolism enzymes following elevated temperature exposure and thyroid hormone manipulation in juvenile lake whitefish (Coregonus clupeaformis). Gen Comp Endocrinol 275:51–64. https://doi.org/10.1016/j.ygcen.2019.02.001
Acknowledgements
We thank Technician Gabriela C. López and Dr. Leandro A. Miranda (INTECH) for helping with histological preparations. We also thank Dr. Tania Rodriguez for technical support and Technician Javier Herdman (INTECH) for fish handling. We thank NBRP Medaka (https://shigen.nig.ac.jp/medaka/) for providing HNI (Strain ID: MT835).
Funding
This work was supported by the Agencia Nacional de Promoción Científica y Tecnológica Grants 2501/15, 1875/18 and 3231/20 (to J.I.F.). VSL holds a Canada Research Chair (950-232235). RHN and IFR were supported by São Paulo Research Foundation (FAPESP), Brazil (grant numbers 14/07620-7; 18/10265-5; 20/15237-0; 21/06742-5). DCCC, AFB and NP were supported by a PhD scholarship from the National Research Council (CONICET). JIF and PHSM are members of the Research Scientist Career at the CONICET.
Author information
Authors and Affiliations
Contributions
DCCC: conceptualization, methodology, visualization and editing; IFR: conceptualization, methodology and editing; AFB: methodology; DM: methodology; NP: methodology; MAO: methodology MSR: methodology; LB: methodology; CS: methodology; JTJ: methodology, DFC: methodology, SD: methodology; PHS: editing; VSL: conceptualization, supervision; writing-reviewing and editing; RHN: conceptualization, investigation, supervision, writing-reviewing and editing, JIF: writing- original draft preparation, funding acquisition, conceptualization, investigation, supervision, writing-reviewing and editing.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
Fish were handled under the ethical regulations of the institutional committee for the Brazilian legislation regulated by the Ethical Principles in Animal Research (Protocol n. 1868-CEUA) and the Institutional Committee for the Care and Use of Experimental Animals from Universidad de San Martín, Argentina (CICUAE-UNSAM 010/2021).
Impact statement
Crosstalk Between the Thyroid and Stress Axes in Thermal-Induced Masculinization in Fish.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Castañeda-Cortés, D.C., Rosa, I.F., Boan, A.F. et al. Thyroid axis participates in high-temperature-induced male sex reversal through its activation by the stress response. Cell. Mol. Life Sci. 80, 253 (2023). https://doi.org/10.1007/s00018-023-04913-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00018-023-04913-6