Abstract
Background
The imbalance between osteogenic and adipogenic differentiation of bone marrow mesenchymal stem cells (BMSCs) is not only the primary pathological feature but also a major contributor to the pathogenesis of steroid-induced osteonecrosis of the femoral head (SONFH). Cellular senescence is one of the main causes of imbalanced BMSCs differentiation. The purpose of this study was to reveal whether cellular senescence could participate in the progression of SONFH and the related mechanisms.
Methods
The rat SONFH model was constructed, and rat BMSCs were extracted. Aging-related indicators were detected by SA-β-Gal staining, qRT-PCR and Western Blot experiments. Using H2O2 to construct a senescent cell model, and overexpressing and knocking down miR-601 and SIRT1 in hBMSCs, the effect on BMSCs differentiation was explored by qRT-PCR, Western Blot experiment, oil red O staining (ORO), alizarin red staining (ARS), and luciferase reporter gene experiment. A rat SONFH model was established to test the effects of miR-601 and metformin in vivo.
Results
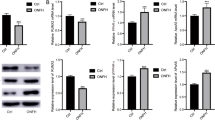
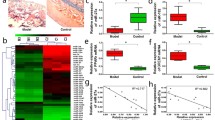
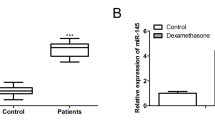
The current study showed that glucocorticoids (GCs)-induced BMSCs senescence, which caused imbalanced osteogenesis and adipogenesis of BMSCs, was responsible for the SONFH progression. Further, elevated miR-601 caused by GCs was demonstrated to contribute to BMSCs senescence through targeting SIRT1. In addition, the anti-aging drug metformin was shown to be able to alleviate GCs-induced BMSCs senescence and SONFH progression.
Conclusions
Considering the role of BMSCs aging in the progression of SONFH, this provides a new idea for the prevention and treatment of SONFH.







Similar content being viewed by others
Availability of data and materials
The data that support the findings of this study are available from the Corresponding authors upon reasonable request.
Abbreviations
- ALP:
-
Alkaline phosphatase
- ARS:
-
Alizarin red staining
- BMSCs:
-
Bone marrow mesenchymal stem cells
- BV/TV:
-
Bone volume per tissue volume
- C/EBPɑ:
-
CCAAT-enhancer-binding proteins ɑ
- FABP4:
-
Fatty acid-binding protein 4
- FNF:
-
Femoral neck fracture
- GCs:
-
Glucocorticoids
- H&E:
-
Hematoxylin and eosin staining
- IHC:
-
Immunohistochemistry
- LPS:
-
Lipopolysaccharide
- MPS:
-
Methylprednisolone
- OPN:
-
Osteopontin
- ORO:
-
Oil red O staining
- PPARγ:
-
Peroxisome proliferators-activated receptors γ
- RUNX2:
-
Runt-related transcription factor 2
- SIRT1:
-
Sirtuin1
- SONFH:
-
Steroid-induced osteonecrosis of the femoral head
- Tb.N:
-
Trabecular number
- Tb.Sp:
-
Trabecular separation
- Tb.Th:
-
Trabecular thickness
References
Zaidi M, Sun L, Robinson LJ, Tourkova IL, Liu L, Wang Y et al (2010) ACTH protects against glucocorticoid-induced osteonecrosis of bone. Proc Natl Acad Sci USA 107(19):8782–8787
Zhao D, Xie H, Xu Y, Wang Y, Yu A, Liu Y et al (2017) Management of osteonecrosis of the femoral head with pedicled iliac bone flap transfer: a multicenter study of 2190 patients. Microsurgery 37(8):896–901
Zhao D, Zhang F, Wang B, Liu B, Li L, Kim SY et al (2020) Guidelines for clinical diagnosis and treatment of osteonecrosis of the femoral head in adults (2019 version). J Orthop Transl 21:100–110
Han L, Wang B, Wang R, Gong S, Chen G, Xu W (2019) The shift in the balance between osteoblastogenesis and adipogenesis of mesenchymal stem cells mediated by glucocorticoid receptor. Stem Cell Res Ther 10(1):377
Zhu W, Guo M, Yang W, Tang M, Chen T, Gan D et al (2020) CD41-deficient exosomes from non-traumatic femoral head necrosis tissues impair osteogenic differentiation and migration of mesenchymal stem cells. Cell Death Dis 11(4):293
Tan G, Kang PD, Pei FX (2012) Glucocorticoids affect the metabolism of bone marrow stromal cells and lead to osteonecrosis of the femoral head: a review. Chin Med J (Engl) 125(1):134–139
Muñoz-Espín D, Serrano M (2014) Cellular senescence: from physiology to pathology. Nat Rev Mol Cell Biol 15(7):482–496
Chen Q, Shou P, Zheng C, Jiang M, Cao G, Yang Q et al (2016) Fate decision of mesenchymal stem cells: adipocytes or osteoblasts? Cell Death Differ 23(7):1128–1139
Hernandez-Segura A, Nehme J, Demaria M (2018) Hallmarks of cellular senescence. Trends Cell Biol 28(6):436–453
Moerman EJ, Teng K, Lipschitz DA, Lecka-Czernik B (2004) Aging activates adipogenic and suppresses osteogenic programs in mesenchymal marrow stroma/stem cells: the role of PPAR-gamma2 transcription factor and TGF-beta/BMP signaling pathways. Aging Cell 3(6):379–389
Chen H, Liu X, Chen H, Cao J, Zhang L, Hu X et al (2014) Role of SIRT1 and AMPK in mesenchymal stem cells differentiation. Ageing Res Rev 13:55–64
Chen C, Zhou M, Ge Y, Wang X (2020) SIRT1 and aging related signaling pathways. Mech Ageing Dev 187:111215
Sasaki T, Maier B, Bartke A, Scrable H (2006) Progressive loss of SIRT1 with cell cycle withdrawal. Aging Cell 5(5):413–422
Xu C, Wang L, Fozouni P, Evjen G, Chandra V, Jiang J et al (2020) SIRT1 is downregulated by autophagy in senescence and ageing. Nat Cell Biol 22(10):1170–1179
McBurney MW, Yang X, Jardine K, Hixon M, Boekelheide K, Webb JR et al (2003) The mammalian SIR2alpha protein has a role in embryogenesis and gametogenesis. Mol Cell Biol 23(1):38–54
Kulkarni AS, Gubbi S, Barzilai N (2020) Benefits of metformin in attenuating the hallmarks of aging. Cell Metab 32(1):15–30
Ramis MR, Esteban S, Miralles A, Tan DX, Reiter RJ (2015) Caloric restriction, resveratrol and melatonin: Role of SIRT1 and implications for aging and related-diseases. Mech Ageing Dev 146–148:28–41
Finkel T, Deng CX, Mostoslavsky R (2009) Recent progress in the biology and physiology of sirtuins. Nature 460(7255):587–591
Barnes PJ, Baker J, Donnelly LE (2019) Cellular senescence as a mechanism and target in chronic lung diseases. Am J Respir Crit Care Med 200(5):556–564
Ong ALC, Ramasamy TS (2018) Role of Sirtuin1-p53 regulatory axis in aging, cancer and cellular reprogramming. Ageing Res Rev 43:64–80
Duan JL, Ruan B, Song P, Fang ZQ, Yue ZS, Liu JJ et al (2022) Shear stress-induced cellular senescence blunts liver regeneration through Notch-sirtuin 1–P21/P16 axis. Hepatology 75(3):584–599
Song W, Wenhui Z, Ruiqiang Y, Hu X, Shi T, Wang M et al (2021) Long noncoding RNA PP7080 promotes hepatocellular carcinoma development by sponging mir-601 and targeting SIRT1. Bioengineered 12(1):1599–1610
Wei W, Ji S (2018) Cellular senescence: molecular mechanisms and pathogenicity. J Cell Physiol 233(12):9121–9135
Chen K, Liu Y, He J, Pavlos N, Wang C, Kenny J et al (2020) Steroid-induced osteonecrosis of the femoral head reveals enhanced reactive oxygen species and hyperactive osteoclasts. Int J Biol Sci 16(11):1888–1900
McGarry T, Biniecka M, Veale DJ, Fearon U (2018) Hypoxia, oxidative stress and inflammation. Free Radic Biol Med 125:15–24
Sung YJ, Kao TY, Kuo CL, Fan CC, Cheng AN, Fang WC et al (2018) Mitochondrial Lon sequesters and stabilizes p53 in the matrix to restrain apoptosis under oxidative stress via its chaperone activity. Cell Death Dis 9(6):697
Hwang JW, Yao H, Caito S, Sundar IK, Rahman I (2013) Redox regulation of SIRT1 in inflammation and cellular senescence. Free Radic Biol Med 61:95–110
Campisi J, d’Adda-di-Fagagna F (2007) Cellular senescence: when bad things happen to good cells. Nat Rev Mol Cell Biol 8(9):729–740
Pignolo RJ, Law SF, Chandra A (2021) Bone aging, cellular senescence, and osteoporosis. JBMR Plus 5(4):e10488
Du J, Xu M, Kong F, Zhu P, Mao Y, Liu Y et al (2022) CB2R attenuates intervertebral disc degeneration by delaying nucleus pulposus cell senescence through AMPK/GSK3β pathway. Aging Dis 13(2):552–567
Hsu YK, Chen HY, Wu CC, Huang YC, Hsieh CP, Su PF et al (2021) Butein induces cellular senescence through reactive oxygen species-mediated p53 activation in osteosarcoma U-2 OS cells. Environ Toxicol 36(5):773–781
González-Osuna L, Sierra-Cristancho A, Rojas C, Cafferata EA, Melgar-Rodríguez S, Cárdenas AM et al (2021) Premature senescence of T-cells favors bone loss during osteolytic diseases. A new concern in the osteoimmunology arena. Aging Dis 12(5):1150–1161
Sui B, Hu C, Liao L, Chen Y, Zhang X, Fu X et al (2016) Mesenchymal progenitors in osteopenias of diverse pathologies: differential characteristics in the common shift from osteoblastogenesis to adipogenesis. Sci Rep 6:30186
Pryor R, Cabreiro F (2015) Repurposing metformin: an old drug with new tricks in its binding pockets. Biochem J 471(3):307–322
Ou Z, Kong X, Sun X, He X, Zhang L, Gong Z et al (2018) Metformin treatment prevents amyloid plaque deposition and memory impairment in APP/PS1 mice. Brain Behav Immun 69:351–363
Langan RC, Goodbred AJ (2017) Vitamin B12 deficiency: recognition and management. Am Fam Physician 96(6):384–389
McCreight LJ, Stage TB, Connelly P, Lonergan M, Nielsen F, Prehn C et al (2018) Pharmacokinetics of metformin in patients with gastrointestinal intolerance. Diabetes Obes Metab 20(7):1593–1601
Kular J, Tickner J, Chim SM, Xu J (2012) An overview of the regulation of bone remodelling at the cellular level. Clin Biochem 45(12):863–873
Ngo D, Beaulieu E, Gu R, Leaney A, Santos L, Fan H et al (2013) Divergent effects of endogenous and exogenous glucocorticoid-induced leucine zipper in animal models of inflammation and arthritis. Arthritis Rheum 65(5):1203–1212
Funding
The study was supported by the Natural Science Foundation of China (No. 32271179, 82102610); Chongqing Technology Innovation and Application Development Project (CQYC202010); the Natural Science Foundation of Chongqing Science and Technology Commission (No. cstc2020jcyj-msxmX0179); the Science Foundation for Top-notch Young Scholars of the First Affiliated Hospital of Chongqing Medical University (No. BJRC2021-02); the Science and Technology Research Project of Chongqing Sports Bureau (No. A202206, A202013, C202124); and the Science and Technology Research Project of Chongqing Education Commission (No. KJQN202200404). All the funding body played no role in the design of the study and collection, analysis, and interpretation of data and in writing the manuscript.
Author information
Authors and Affiliations
Contributions
J.Z, C.L. and B.T. participated in the design of the study and carried out experiments. B.T., Y.C., M.S., H.Z., and D.X. conceived the experiments. B.T, P.Z, W.Y, X.H. and T.C. analyzed the data. B.T., C.L. and J.Z. wrote the manuscript. All authors were involved in writing the paper and had final approval of the submitted and published versions.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval and consent to participate
(1) Title of the approved project: The role and molecular mechanism of miRNA-601 in steroid-induced necrosis of the femoral head; (2) Name of the institutional approval committee or unit: The First Affiliated Hospital of Chongqing Medical University and the Ethical Committee of Chongqing Medical University; (3) Approval number: 2022–005; (4) Date of approval: March 23, 2022.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tang, B., Chen, Y., Zhao, P. et al. MiR-601-induced BMSCs senescence accelerates steroid-induced osteonecrosis of the femoral head progression by targeting SIRT1. Cell. Mol. Life Sci. 80, 261 (2023). https://doi.org/10.1007/s00018-023-04903-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00018-023-04903-8