Abstract
Pulmonary neuroendocrine (NE) cells represent a small population in the airway epithelium, but despite this, hyperplasia of NE cells is associated with several lung diseases, such as congenital diaphragmatic hernia and bronchopulmonary dysplasia. The molecular mechanisms causing the development of NE cell hyperplasia remains poorly understood. Previously, we showed that the SOX21 modulates the SOX2-initiated differentiation of epithelial cells in the airways. Here, we show that precursor NE cells start to develop in the SOX2 + SOX21 + airway region and that SOX21 suppresses the differentiation of airway progenitors to precursor NE cells. During development, clusters of NE cells start to form and NE cells mature by expressing neuropeptide proteins, such as CGRP. Deficiency in SOX2 resulted in decreased clustering, while deficiency in SOX21 increased both the numbers of NE ASCL1 + precursor cells early in development, and the number of mature cell clusters at E18.5. In addition, at the end of gestation (E18.5), a number of NE cells in Sox2 heterozygous mice, did not yet express CGRP suggesting a delay in maturation. In conclusion, SOX2 and SOX21 function in the initiation, migration and maturation of NE cells.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The airway epithelium consists of different cell types, such as basal, secretory and ciliated cells. A rare (< 1%) subpopulation of airway epithelial cells, the pulmonary neuroendocrine (NE) cell, is associated with a variety of lung diseases [1]. These lung diseases may have a complex origin, showing an inflammatory phenotype like bronchopulmonary dysplasia (BPD), asthma and chronic obstructive pulmonary disease (COPD) [2,3,4] or may have a congenital origin like congenital diaphragmatic hernia (CDH) [5, 6]. In addition, NE cells have proliferative potential and are the cell of origin driving development of human small cell lung cancer [7]. The various diseases with a disturbed NE cell phenotype suggests that involvement of NE cells is of a complex nature.
Pulmonary NE cells are chemo-sensory cells, which monitor different aspects of lung physiology, such as changes in oxygen, other chemicals and changes in mechanical forces [8]. Changes registered by the NE cells are relayed to the brain by sensory neurons [9]. In order to exert biological responses to the detected changes, NE cells contain dense core vesicles containing neuropeptides, such as Calcitonin gene-related peptide (CGRP) [8]. In the mouse lung, solitary NE cells are scattered throughout the airway epithelium, whereas clusters of NE cells, called neuroendocrine bodies (NEBs), are often found at airway bifurcations [10]. In mouse models it was shown that upon allergen exposure, NE cells are activated, and initiate goblet cell hyperplasia and infiltration of immune cells into the lung [3]. The increased mucus production due to goblet cell hyperplasia is initiated by the production and secretion of GABA by NE cells, which is completely abrogated upon loss of neuronal innervation [11, 12]. Besides the importance of NE cell innervation, a proper clustering of NE cells was shown to be essential in restricting immune infiltration in the neonatal lung [13], showing a clear difference in functionality between solitary NE cells and NEBs.
NE cells are endoderm-derived, and together with basal cells are the earliest cells originating from the airway progenitors. Notch signaling plays an active role in distinguishing between a NE and non-NE cell fate [14,15,16]. In non-NE cells, active Notch signaling represses the transcription of Achaete-scute homolog 1 (Ascl1), which is a master regulator in the formation of NE cells [17]. Previously, we showed that overexpression of the transcription factor sex determining region Y-box 2 (SOX2) in the developing lung epithelium resulted in increased expression of Ascl1 and an increased number of NE cells [18]. Moreover, we showed that early in development the expression levels of both SOX2 and SOX21 determine the fate of airway progenitor to basal cells [19]. In neural and olfactory precursor cells, ASCL1 has shown to be a transcriptional target of SOX2 [20, 21]. However, if and how SOX2 and SOX21 play a role in the initial differentiation and distribution of NE cells during lung development is not yet determined.
Here, we show that SOX2 and SOX21 have distinct roles in the development and maturation of NE cells. We show that reduced levels of SOX21, but not SOX2, affect the initiation of the formation of ASCL1 + NE precursor cells. Both, SOX2 and SOX21 deficiency resulted in an aberrant clustering of NE cells compared to wild-type conditions. In addition, deficiency of SOX2 levels delayed CGRP + NE cell maturation, while absence of SOX21 promoted NE cell maturation.
Results
SOX21 inhibits SOX2 progenitor differentiation to precursor NE cells
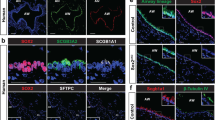
In previous studies we suggested a relation between SOX2 and NE cell development [18]. A potential interaction between SOX2 and SOX21 within the development of NE cells was determined by evaluating whether NE precursors, marked by ASCL1 expression, appear in the SOX2 + SOX21 + proximal airway region [19]. At early stages of development (E13.5 and E14.5), solitary and clustered (~ 10–30 cells) NE precursor cells (ASCL1 +) emerge in the proximal airways, consistent with previous published data [10, 22] (Fig. 1A). We found that precursor NE cells were observed in the region positive for SOX21, at both E13.5 and E14.5 (Fig. 1B). We assessed SOX2 and SOX21 expression levels in ASCL1 + NE and ASCL- non-NE cells and calculated the mean fluorescence intensity (MFI) of SOX2 and SOX21 in ASCL + and ASCL1- nuclei (Fig. 1C). This showed no correlation between ASLC1 + precursor NE cells and MFI of SOX2 or SOX21 (Fig. 1C). At E14.5, SOX2 + airway progenitor cells also differentiate to TRP63 + basal cells (Transformation Related Protein 63 positive cells). Similar to the precursor NE cells, we did not observe a correlation, between TRP63 + and TRP- nuclei and MFI of SOX2 or SOX21 at this stage of development (Fig. 1D). Thus, similar to basal cells, precursor NE cells start appearing in the airway regions positive for SOX2 and SOX21, but SOX21 or SOX2 abundancy does not correlate with either basal or NE cells early in lung development.
Differentiation to NE cells of SOX2 + airway progenitor cells occurs in SOX21 + airway region. A Immunofluorescence staining of ASCL1 (red) and DAPI (blue) at gestational ages E13.5 and E14.5. C = cluster, S = solitary. Scale bar = 50 µm. B Immunofluorescence staining of ASCL1 (red), SOX21 (green) and SOX2 (blue) at gestational age E13.5 and E14.5. Cells positive for ASCL1 are encircled. Scale bar = 25 µm. C Immunofluorescence staining of ASCL1 (red), SOX21 (green) and SOX2 (blue) at gestational age E14.5. In the encircled cells the MFI of ASCL1, SOX21 and SOX2 is shown at that specific region. Scale bar = 25 µm. The graph shows the MFI of ASCL1, SOX21 and SOX2 in ASCL- and ASCL + airway epithelium. Data are represented as mean ± SD. Two-way ANOVA (n = 3, *** p < 0.001). D Immunofluorescence staining of TRP63 (violet), ASCL1 (red), SOX21 (green) and DAPI (blue) at gestational age E14.5. Cells positive for TRP63 are encircled. Scale bar = 25 µm. The graph shows the MFI of TRP63, SOX21 and SOX2 in TRP63- and TRP63 + airway epithelium. Data are represented as mean ± SD. Two-way ANOVA (n = 3, *** p < 0.001)
We next determined whether levels of SOX2 or SOX21 play a role in the differentiation of SOX2 + airway progenitors to NE cells. The number of precursor NE cells were quantified at E14.5 in lungs of SOX21 heterozygous (Sox21+/−), knock-out (Sox21−/−) and SOX2 heterozygous (Sox2+/−) mice. Comparable numbers of precursor NE cells were observed in lungs of wild type (WT), Sox21+/− and Sox2+/−. Although the numbers of NE cells were not significantly different in lungs of Sox21+/− mice, complete ablation of SOX21 resulted in increased numbers of precursor NE cells (Fig. 2). So, absence of SOX21 increased the differentiation of SOX2 + airway progenitors to NE cells, indicating that SOX21 may control the emergence of NE cells. In addition, we observed that in both the Sox21+/− and Sox21−/−, NE cells were more solitary distributed throughout the airway epithelium (Fig. 2).
Loss of SOX21 increases differentiation of SOX2 + airway progenitor cell to NE precursor cells. A Immunofluorescence staining of ASCL1 (red) on the first bifurcation of the main bronchus at gestational age E14.5 of wild-type (WT), Sox2+/−, Sox21+/− and Sox21−/− mice. B Quantification of the percentage of NE precursor cells in a box of 400 µm2 around the first bifurcation of the main bronchus. Data are represented as mean ± SEM. One-way ANOVA (n WT = 5, n Sox2+/− = 4, Sox21+/− = 3, Sox21−/− = 3, **p < 0.01, ***p < 0.001, ****p < 0.0001)
Expression levels of SOX21 decrease during the maturation of NE cells
To gain more insight in the role for SOX2 and SOX21 in the formation of NE cells during lung development, we looked at the development of NE cells at later gestational ages. Functional NE cells are innervated by sensory neurons and express the neuropeptide protein, CGRP. Innervation of NE cells was determined by the presence of synaptic vesicle protein 2 (SV2), a general marker of synaptic vesicles in neuronal cells [23, 24]. SV2 and CGRP were detected weakly at E16.5 and more abundantly expressed at E18.5 (Fig. 3A). From E15.5 onward, NE cells started to appear in the distal airways, probably due to migration of the NE cells in a proximal to distal fashion [22]. Distal airway epithelial cells express SOX2, but not SOX21, especially later in lung development (> E15.5) [19]. At E15.5, SOX21 was observed both in NEBs in the main bronchi and in the distal airways (Fig. 3A–C). Resembling E14.5, in the main bronchi of E15.5, the MFI of SOX2 and SOX21 was similar in NE cells compared to the adjacent non-NE airway epithelium (Fig. 3B, C). At E16.5, when the earliest expression of CGRP could be detected (Fig. 3A), NE cells in the main bronchi showed a lower MFI of SOX21 and a slightly lower MFI of SOX2 compared to adjacent non-NE airway epithelium (Fig. 3B, C). Distal airway epithelium lacks SOX21, and we did not observe expression of SOX21 in NE cells at E16.5 (Fig. 3E) [19]. At E18.5, NEBs were both SOX2 and SOX21 positive in proximal and distal airway epithelium (Fig. 3B–D). However, the MFI of SOX21 was slightly lower in NE cells compared to adjacent non-NE airway epithelium (Fig. 3B, C). In conclusion, the levels of SOX2 and SOX21 are dynamically expressed during formation and differentiation of the NE cells. NE cells start to express CGRP around E16.5 and this is accompanied by a decrease in SOX21 levels. We also observed a small, but non-significant decrease in SOX2 levels at this developmental stage. At E18.5, SOX21 is observed again in NEBs (Fig. 3E).
Different abundancy of SOX2 and SOX21 during the maturation of NEBs. A Immunofluorescence staining of SV2 (violet), ASCL1 (red), CGRP (green) and DAPI (blue) at gestational ages E15.5, E16.5 and E18.5. Scale bar = 50 µm. B Immunofluorescence staining of ASCL1 (red), SOX21 (green) and SOX2 (blue) at gestational age E15.5, E16.5 and E18.5. The proximal airway epithelium is characterized by abundant expression of SOX21. Cells positive for ASCL1 are encircled. Scale bar = 25 µm. C The graph shows the MFI of ASCL1, SOX21 and SOX2 in ASCL- and ASCL + airway epithelium in proximal airway epithelium at E15.5, E16.5 and E18.5. Data are represented as mean ± SD. Two-way ANOVA (n = 3, *p < 0.05, **p < 0.01, ***p < 0.001). D Immunofluorescence staining of ASCL1 (red), SOX21 (green) and SOX2 (blue) at gestational age E15.5, E16.5 and E18.5 in the distal SOX2 + SOX21- airway epithelium. Cells positive for ASCL1 are encircled. Scale bar = 25 µm. E Schematic representation of SOX2 and SOX21 protein levels in neuroendocrine bodies (NEBs) at different gestational ages. SOX21 shows a dip and SOX2 a slight decrease in expression at E16.5, when CGRP expression starts and NEBs become innervated (SV2)
SOX2 and SOX21 are important in the maturation and clustering of NE cells
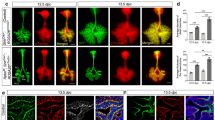
Next, the roles of SOX2 and SOX21 in the emergence and differentiation of NE cells later in lung development at E18.5 were investigated by analyzing the total number of ASCL1 + lineage committed NE cells and CGRP + differentiated NE cells. Contrary to E14.5, Sox2+/− lungs at E18.5 showed a decreased number of ASCL1 + NE cells compared to the wild type (Fig. 4A, B). However, no difference was observed in the number of ASCL1 + NE cells in lungs of hetero- and homozygous Sox21 mice. Quantification of the number of CGRP + NE cells revealed a reduced number of mature NE cells in Sox2+/− lungs, corresponding to the reduced number of ASCL1 + NE cells. Remarkably, an increase in the number of mature CGRP + NE cells was found in the airways of both Sox21+/− and Sox21−/− (Fig. 4B). In addition, nearly all ASCL1 + cells in the Sox21−/− airways also expressed high levels of CGRP, whereas the WT, and Sox2+/− airways showed mainly CGRP expression at the basal side of the NE cells and a substantial number of ASCL1 + NE cells that did not express CGRP (Fig. 4C). Generally, all CGRP + NE cells were ASCL1 + , and no single CGRP + cells were found. In conclusion, SOX2 is important in the lineage specification or commitment towards the NE lineage, since Sox2+/− mice showed a reduced number of ASCL1 positive NE cells. SOX21 suppresses the initiation of NE cells early in lung development, but later in development it modulates the terminal differentiation of committed NE cells.
Loss of SOX2 decreases and of SOX21 increases the number NE cells at E18.5. A Immunofluorescence staining of CGRP (green), SOX2 (RED) and DAPI (blue) on lung sections at gestational age E18.5 of wild-type (WT), Sox2+/−, Sox21+/− and Sox21−/− mice. Scale bar = 200 µm. B Quantification of the number of NE cells on E18.5 lung sections, normalized to the area of SOX2 + area measured. Data are represented as mean ± SEM. One-way ANOVA on ASCL1 + cells (left) (n WT = 4, n Sox2+/− = 4, Sox21+/− = 4, Sox21−/− = 3), One-way ANOVA on CGRP + cells (right) (n WT = 7, n Sox2+/− = 7, Sox21+/− = 7, Sox21−/− = 6) (*p < 0.05 **p < 0.01, ***p < 0.001, ****p < 0.0001). C Immunofluorescence staining of ASCL1 (red), CGRP (green) and SOX2 (blue) on lung sections at gestational age E18.5 of wild-type (WT), Sox2+/−, Sox21+/− and Sox21−/− mice. Scale bar = 50 µm
In the mouse lung, NE cells are found solitary, in small clusters (2–5 PNECs) or big clusters/NEBs (> 5 NE cells). The distribution and innervation of NE cells has been shown to be important for their function [11, 13]. We observed, that both clusters and single CGRP + NE cells show neuronal innervation (SV2 +) in WT, Sox2+/−, Sox21+/− and Sox21−/− lungs (Fig. 5A). Next, we determined the distribution of mature NE cells upon reduced levels of Sox2 and Sox21 expression (Fig. 5A). In WT lungs, NE cells were mostly present in small and big clusters, while in Sox2+/− lungs, NE cells were slightly more present as solitary cells although most of the NE cells were present in small clusters (Fig. 5B). While reduced levels of SOX21 did not show any differences compared to the wild type, complete absence of SOX21 resulted in a high percentage of NE cells in NEBs (Fig. 5B). All genotypes contained a similar number of mature cells in NEBs (Fig. 5C). Taken together, the data show that SOX21 levels play a role in initiation of NE cell differentiation and maturation, and the distribution of cells in NEBs, while SOX2 levels are involved in the maturation of NE cells and the clustering of NE cells in NEBs.
Loss of SOX2 decreases and of SOX21 increases the number large NE clusters. A Immunofluorescence staining of CGRP (green) and SV2 (RED) on lung sections at gestational age E18.5 of wild-type (WT), Sox2+/−, Sox21+/− and Sox21−/− mice. Scale bar = 15 µm. B The percentage of CGRP + NE cells that are present as single NE cells, small clusters (2–5 cells) or large clusters (> 5). Data are represented as mean ± SEM. Two-way ANOVA (n WT = 7, n Sox2+/− = 7, Sox21+/− = 7, Sox21−/− = 6, *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001). C Representation of the average number of CGRP + NE cells in clusters > 5. Data are represented as mean ± SEM. One- way ANOVA (n WT = 7, n Sox2+/− = 3 (4 did not show any clusters > 5), Sox21+/− = 7, Sox21−/− = 6)
Discussion
SOX2 and SOX21 were known to be co-expressed in developing proximal airway epithelium [19], but their individual functions in the development of NE cells were not known. In this study, we investigated their potential involvement in the initiation, maturation and distribution of NE cells in the mouse lung. Deficiency in either SOX2 or SOX21 expression, shows that SOX21 suppresses the initial differentiation to NE cells early in development while SOX2 levels are mainly important in the maintenance and maturation of NE cells later during lung development.
During airway development, airway progenitor cells differentiate to basal, secretory, ciliated and NE cells. Complete deficiency of the Notch signaling pathway, through deletion of Rbpjk (Notch transcriptional effector), Pofut1 (essential for Notch-ligand binding) [25] or deletion of all Notch receptors [15], resulted in the expansion of NE cells. The initial stages of airway progenitor differentiation to NE cells, takes place in the SOX2 + SOX21 + intrapulmonary airways. Here, we show that SOX21 is important in suppressing NE cell differentiation, which is dependent on the level of expression as demonstrated by SOX21 hetero- and homozygous knockout mice.
Based on our data we hypothesize that a competition between SOX2 and SOX21 exists and that subtle changes in the balance between these factors determines cell fate decisions. This is supported by the physically interaction between SOX2 and SOX21 [26, 27], by the transcriptional activation of SOX21 by SOX2 [18, 26], and by the facts that SOX2 and SOX21 bind the same genomic locations, and SOX21 can compete SOX2 binding to suppress gene activation [19]. Together, these data support the hypothesis that SOX2 and SOX21 compete for DNA binding sites, and thus changes in the balance between SOX2 or SOX21 directly influence the binding of these factors to DNA. As a result, targets genes are either activated, or not, because SOX2 has a C-terminal transactivation domain, whereas SOX21 harbors a transcriptional repressor motif in the C-terminal part of the protein [28].
In addition, Sox2+/− mice did not show an increase or decrease in the number of NE precursor cells, showing that deficiency in SOX2 or SOX21 does not affect the initial differentiation to NE cells. In neuronal stem cell differentiation, SOX21 deletion also increased differentiation to ASCL1 + precursor cells through suppression of Notch target HES5 [29]. NE cell differentiation in the developing lung is repressed by HES1 in non-NE airway epithelial cells [14, 16].
We previously showed that the balance between SOX2 and SOX21 is important for specification of several epithelial cells [19]. Moreover, SOX2 and SOX21 are expressed in NE and non-NE cells, as we show in Fig. 1C. Hence, the question arises whether SOX2 and SOX21 work autonomously in NE cell fate specification, or whether SOX2 and SOX21 act in non-NE cells to affect NE cell differentiation and maturation non-cell autonomously. As described before, NE cell fate is determined by lateral inhibition of NOTCH-HES1 signalling [14, 15, 22, 25]. Ascl1 + NE cells express the Notch ligand Dll, whereas the neighbouring non-NE cells express Notch, and one of the downstream targets Hes1. Interference with Notch signalling by inhibition leads to more NEs, or more NEs after ectopic activation of Notch signalling [30]. Although non-NE cells do not contribute to the formation of NEBs, it could be possible that these cells influence the maturation of NEs and NEBs [22].
Although we did not observe a difference in the number of precursor NE cells upon reduced levels of SOX2, we did show that SOX2 plays an important role in the further expansion of NE cells and subsequent maturation of NE cells at the end of lung development. These results indicate that high levels of SOX2 are not necessary for the induction of Ascl1 but are necessary for the maintenance and maturation of NE cells. Furthermore, reduced levels of SOX2 showed a higher frequency of single scattered NE cells in the airway epithelium. In contrast, reduced levels of SOX21resulted in an increase in mature CGRP + NE cells and in NE cluster formation, indicated by the presence of more NEBs. Since there is no increase in the total number of ASCL1 + NE cells in the SOX21 deprived cells, we suggest that SOX21 is mainly involved in repressing maturation rather than maintenance of NE cells in later stages of development. In addition, a difference in abundancy of SOX2 and SOX21 was observed within NEBs at different stages of NE development. This, all together suggests that SOX2 and SOX21 might function differently during different stages of NE development; initiation, migration and maturation.
In addition to the function of the NE cell as a chemo-sensory cell, a subset of NE cells was shown to proliferate and repair surrounding airway epithelium upon response to injury [31, 32]. In several mouse models regulation of maintenance and proliferation of NE cells is important, because NE cells were found to be the origin for small cell lung cancer (SCLC) [33, 34]. It has long been speculated that SCLC arise from NE cells due to similarities in morphology and expression of NE markers in the tumor [7]. It was recently established that specific NE stem cells are the source of tumor formation within NEBs [32]. Multiple studies have shown an increase in Sox2 expression in SCLC, and a few also indicated elevated levels of Sox21 [35,36,37,38].
In conclusion, this study shows distinct involvement of SOX2 and SOX21 in the development and maturation of pulmonary NE cells. SOX21 suppresses initiation of NE cell differentiation early in development, preventing NE cell hyperplasia within the lung. In contrast, high SOX2 levels are mostly involved in the maintenance, maturation, and clustering of NE cells. Given that hyperplasia of NE cells and defective clustering are associated with several lung diseases and NE function respectively, investigating the molecular mechanisms downstream of SOX2 and SOX21 may provide new insight in NE function and development of new therapeutic approaches.
Materials and methods
Mice
All animal experimental protocols were approved by the animal welfare committee of the veterinary authorities of the Erasmus Medical Center (SP2100184). Mice were kept under standard conditions. Mouse strains bioSOX2flox [39], SOX2-cre-ERT [40] and SOX21-KO (gift of Stavros Malas) were used. Wild-type animals were C57BL/6.
Immunofluorescence
Mouse embryonic lungs were fixed overnight in 4% w/v PFA at 4 ºC. Post-fixation, samples were washed with PBS, de-hydrated to 100% ethanol, transferred to xylene and processed to paraffin wax for embedding. Paraffin blocks were sectioned at 5 µm and dried overnight at 37 ºC.
Sections were deparaffinized by 3 times 2 min xylene washes, followed by rehydration in distilled water. Antigen retrieval was performed by boiling the slides in Tris–EDTA (10 mM Tris, 1 mM EDTA) buffer pH = 9.0 for 15 min at 600W. Slides were cooled down for 30 min and transferred to PBS. For SOX21 and ASCL1 staining, the Tyramide Signal Amplification (TSA) kit was used (Invitrogen, B40922, according to manufacturer’s protocol). When using the TSA kit, a hydrogen peroxide (1.5% in PBS) blocking step was performed after boiling. For co-staining of SOX21 and ASCL1, first ASCL1 was stained (first and secondary antibodies plus TSA reaction), sections were boiled again in Tris–EDTA (10 mM Tris, 1 mM EDTA) buffer pH = 9.0 for 15 min at 300W and subsequently SOX21 was stained. Sections were blocked for 1 h at room temperature (RT) in 5% Elk (Campina) for ASCL1 staining or 3% BSA, 0.05% Tween in PBS. Primary antibodies (Table 1) were diluted in blocking buffer and incubated with the sections overnight at 4 ºC. The next day, sections were washed 3 times for 5 min at room temperature (RT) in PBS. Secondary antibodies (Table 1) were added in blocking buffer and incubated for 1 h at RT. DAPI (4′,6-Diamidino-2-Phenylindole) solution (BD Pharmingen, 564907, 1:2000) was added to the secondary antibodies for nuclear staining. After incubation, 3 times 5 min washes in PBS, sections were mounted using Mowiol reagent (For 100 mL: 2,4% w/v Mowiol, 4,75% w/v glycerol, 12% v/v Tris 0.2 M pH = 8.5 in dH2O till 100 mL). All sections were imaged on a Leica SP5 confocal microscope.
Image analysis
Neuroendocrine precursor cells (ASCL1 +) were counted on E14.5, 5 µm thick sections, in a square of 400 µm2 around the first branch of the main bronchi. Of each genotype and each n, 3 sections were counted and the percentage of NE cells were calculated based on the total number of DAPI + nuclei in the airway.
Neuroendocrine cells (ASCL1 + or ASCL1 + /CGRP +) were counted on E18.5, 5 µm thick sections. Of each genotype and each n, 1 or 2 sections of the left lobe were counted and the number of NE cells were normalized to airway (SOX2 +) area present in the section.
Fluorescent intensity of SOX2 and SOX21 was measured in three different location which contained ASCL1 + NEBs, in proximal SOX2 + SOX21 + airway epithelium. The mean fluorescence intensity (MFI) was calculated by dividing the fluorescence intensity of ASCL1 + or ASLC1- region compared to the fluorescence intensity of the total airway epithelium at that specific location.
Fluorescent intensity of TRP63, SOX2 and SOX21 was measured in a 150 µm2 box in the main bronchi, in 3 sections of 3 different mouse lungs. The MFI was calculated by dividing the fluorescence intensity of TRP63 + or TRP63- region compared to the fluorescence intensity of the total airway epithelium at that specific location.
Image J was used to analyze the pictures.
Data availability
No large datasets nave been generated during the current study.
References
Garg A, Sui P, Verheyden JM, Young LR, Sun X (2019) Consider the lung as a sensory organ: a tip from pulmonary neuroendocrine cells. Curr Top Dev Biol 132:67–89. https://doi.org/10.1016/bs.ctdb.2018.12.002. (Epub 2019/02/25. PubMed PMID: 30797518)
Gu X, Karp PH, Brody SL, Pierce RA, Welsh MJ, Holtzman MJ et al (2014) Chemosensory functions for pulmonary neuroendocrine cells. Am J Respir Cell Mol Biol 50(3):637–646. https://doi.org/10.1165/rcmb.2013-0199OC. (Epub 2013/10/19. PubMed PMID: 24134460; PubMed Central PMCID: PMC4068934)
Sui P, Wiesner DL, Xu J, Zhang Y, Lee J, Van Dyken S et al (2018) Pulmonary neuroendocrine cells amplify allergic asthma responses. Science. https://doi.org/10.1126/science.aan8546. (Epub 2018/03/31. PubMed PMID: 29599193; PubMed Central PMCID: PMC6387886)
Cutz E, Yeger H, Pan J (2007) Pulmonary neuroendocrine cell system in pediatric lung disease-recent advances. Pediatr Dev Pathol 10(6):419–435. https://doi.org/10.2350/07-04-0267.1. (Epub 2007/11/16. PubMed PMID: 18001162)
Ijsselstijn H, Gaillard JL, de Jongste JC, Tibboel D, Cutz E (1997) Abnormal expression of pulmonary bombesin-like peptide immunostaining cells in infants with congenital diaphragmatic hernia. Pediatr Res 42(5):715–720. https://doi.org/10.1203/00006450-199711000-00026. (Epub 1997/11/14. PubMed PMID: 9357948)
IJsselstijn H, Hung N, de Jongste JC, Tibboel D, Cutz E (1998) Calcitonin gene-related peptide expression is altered in pulmonary neuroendocrine cells in developing lungs of rats with congenital diaphragmatic hernia. Am J Respir Cell Mol Biol 19(2):278–285. https://doi.org/10.1165/ajrcmb.19.2.2853. (Epub 1998/08/12. PubMed PMID: 9698600)
van Meerbeeck JP, Fennell DA, De Ruysscher DK (2011) Small-cell lung cancer. Lancet 378(9804):1741–1755. https://doi.org/10.1016/S0140-6736(11)60165-7. (Epub 2011/05/14. PubMed PMID: 21565397)
Cutz E, Pan J, Yeger H, Domnik NJ, Fisher JT (2013) Recent advances and contraversies on the role of pulmonary neuroepithelial bodies as airway sensors. Semin Cell Dev Biol 24(1):40–50. https://doi.org/10.1016/j.semcdb.2012.09.003. (Epub 2012/10/02. PubMed PMID: 23022441)
Brouns I, Oztay F, Pintelon I, De Proost I, Lembrechts R, Timmermans JP et al (2009) Neurochemical pattern of the complex innervation of neuroepithelial bodies in mouse lungs. Histochem Cell Biol 131(1):55–74. https://doi.org/10.1007/s00418-008-0495-7. (Epub 2008/09/03. PubMed PMID: 18762965)
Kuo CS, Krasnow MA (2015) Formation of a neurosensory organ by epithelial cell slithering. Cell 163(2):394–405. https://doi.org/10.1016/j.cell.2015.09.021. (Epub 2015/10/06. PubMed PMID: 26435104; PubMed Central PMCID: PMC4597318)
Barrios J, Patel KR, Aven L, Achey R, Minns MS, Lee Y et al (2017) Early life allergen-induced mucus overproduction requires augmented neural stimulation of pulmonary neuroendocrine cell secretion. FASEB J 31(9):4117–4128. https://doi.org/10.1096/fj.201700115R. (Epub 2017/06/02. PubMed PMID: 28566470; PubMed Central PMCID: PMC5572694)
Barrios J, Kho AT, Aven L, Mitchel JA, Park JA, Randell SH et al (2019) Pulmonary neuroendocrine cells secrete gamma-aminobutyric acid to induce goblet cell hyperplasia in primate models. Am J Respir Cell Mol Biol 60(6):687–694. https://doi.org/10.1165/rcmb.2018-0179OC. (Epub 2018/12/21. PubMed PMID: 30571139; PubMed Central PMCID: PMCPMC6543741)
Branchfield K, Nantie L, Verheyden JM, Sui P, Wienhold MD, Sun X (2016) Pulmonary neuroendocrine cells function as airway sensors to control lung immune response. Science 351(6274):707–710. https://doi.org/10.1126/science.aad7969. (Epub 2016/01/09. PubMed PMID: 26743624)
Ito T, Udaka N, Yazawa T, Okudela K, Hayashi H, Sudo T et al (2000) Basic helix-loop-helix transcription factors regulate the neuroendocrine differentiation of fetal mouse pulmonary epithelium. Development 127(18):3913–3921 (PubMed PMID: 10952889)
Morimoto M, Nishinakamura R, Saga Y, Kopan R (2012) Different assemblies of Notch receptors coordinate the distribution of the major bronchial Clara, ciliated and neuroendocrine cells. Development 139(23):4365–4373. https://doi.org/10.1242/dev.083840. (Epub 2012/11/08. PubMed PMID: 23132245; PubMed Central PMCID: PMC3509731)
Jia S, Wildner H, Birchmeier C (2015) Insm1 controls the differentiation of pulmonary neuroendocrine cells by repressing Hes1. Dev Biol 408(1):90–98. https://doi.org/10.1016/j.ydbio.2015.10.009. (Epub 2015/10/11. PubMed PMID: 26453796)
Borges M, Linnoila RI, van de Velde HJ, Chen H, Nelkin BD, Mabry M et al (1997) An achaete-scute homologue essential for neuroendocrine differentiation in the lung. Nature 386(6627):852–855 (PubMed PMID: 9126746)
Gontan C, de Munck A, Vermeij M, Grosveld F, Tibboel D, Rottier R (2008) Sox2 is important for two crucial processes in lung development: branching morphogenesis and epithelial cell differentiation. Dev Biol 317(1):296–309. https://doi.org/10.1016/j.ydbio.2008.02.035. (Epub 2008/04/01. PubMed PMID: 18374910)
Eenjes E, Buscop-van Kempen M, Boerema-de Munck A, Edel GG, Benthem F, de Kreij-de BL et al (2021) SOX21 modulates SOX2-initiated differentiation of epithelial cells in the extrapulmonary airways. Elife. https://doi.org/10.7554/eLife.57325. (Epub 2021/07/22. PubMed PMID: 34286693; PubMed Central PMCID: PMC8331192)
Tucker ES, Lehtinen MK, Maynard T, Zirlinger M, Dulac C, Rawson N et al (2010) Proliferative and transcriptional identity of distinct classes of neural precursors in the mammalian olfactory epithelium. Development 137(15):2471–2481. https://doi.org/10.1242/dev.049718. (Epub 2010/06/25. PubMed PMID: 20573694; PubMed Central PMCID: PMCPMC2927697)
Niu W, Zang T, Smith DK, Vue TY, Zou Y, Bachoo R et al (2015) SOX2 reprograms resident astrocytes into neural progenitors in the adult brain. Stem Cell Reports 4(5):780–794. https://doi.org/10.1016/j.stemcr.2015.03.006. (Epub 2015/04/30. PubMed PMID: 25921813; PubMed Central PMCID: PMCPMC4437485)
Noguchi M, Sumiyama K, Morimoto M (2015) Directed migration of pulmonary neuroendocrine cells toward airway branches organizes the stereotypic location of neuroepithelial bodies. Cell Rep 13(12):2679–2686. https://doi.org/10.1016/j.celrep.2015.11.058. (Epub 2015/12/30. PubMed PMID: 26711336)
Weichselbaum M, Everett AW, Sparrow MP (1996) Mapping the innervation of the bronchial tree in fetal and postnatal pig lung using antibodies to PGP 9.5 and SV2. Am J Respir Cell Mol Biol 15(6):703–710. https://doi.org/10.1165/ajrcmb.15.6.8969263. (Epub 1996/12/01. PubMed PMID: 8969263)
Pan J, Yeger H, Cutz E (2004) Innervation of pulmonary neuroendocrine cells and neuroepithelial bodies in developing rabbit lung. J Histochem Cytochem 52(3):379–389. https://doi.org/10.1177/002215540405200309. (Epub 2004/02/18. PubMed PMID: 14966205)
Tsao PN, Vasconcelos M, Izvolsky KI, Qian J, Lu J, Cardoso WV (2009) Notch signaling controls the balance of ciliated and secretory cell fates in developing airways. Development 136(13):2297–2307 (PubMed PMID: 19502490)
Kuzmichev AN, Kim SK, D’Alessio AC, Chenoweth JG, Wittko IM, Campanati L et al (2012) Sox2 acts through Sox21 to regulate transcription in pluripotent and differentiated cells. Curr Biol 22(18):1705–1710. https://doi.org/10.1016/j.cub.2012.07.013. (Epub 2012/08/21. PubMed PMID: 22902753)
Mallanna SK, Ormsbee BD, Iacovino M, Gilmore JM, Cox JL, Kyba M et al (2010) Proteomic analysis of Sox2-associated proteins during early stages of mouse embryonic stem cell differentiation identifies Sox21 as a novel regulator of stem cell fate. Stem Cells 28(10):1715–1727. https://doi.org/10.1002/stem.494. (Epub 2010/08/06. PubMed PMID: 20687156; PubMed Central PMCID: PMC3260005)
Kamachi Y, Kondoh H (2013) Sox proteins: regulators of cell fate specification and differentiation. Development 140(20):4129–4144. https://doi.org/10.1242/dev.091793. (Epub 2013/10/03. PubMed PMID: 24086078)
Matsuda S, Kuwako K, Okano HJ, Tsutsumi S, Aburatani H, Saga Y et al (2012) Sox21 promotes hippocampal adult neurogenesis via the transcriptional repression of the Hes5 gene. J Neurosci 32(36):12543–12557. https://doi.org/10.1523/JNEUROSCI.5803-11.2012. (Epub 2012/09/08. PubMed PMID: 22956844; PubMed Central PMCID: PMCPMC6621257)
Stupnikov MR, Yang Y, Mori M, Lu J, Cardoso WV (2019) Jagged and Delta-like ligands control distinct events during airway progenitor cell differentiation. Elife. https://doi.org/10.7554/eLife.50487. (Epub 2019/10/22. PubMed PMID: 31631837; PubMed Central PMCID: PMC6887486)
Reynolds SD, Hong KU, Giangreco A, Mango GW, Guron C, Morimoto Y et al (2000) Conditional clara cell ablation reveals a self-renewing progenitor function of pulmonary neuroendocrine cells. Am J Physiol Lung Cell Mol Physiol 278(6):L1256–L1263. https://doi.org/10.1152/ajplung.2000.278.6.L1256. (Epub 2000/06/03. PubMed PMID: 10835332)
Ouadah Y, Rojas ER, Riordan DP, Capostagno S, Kuo CS, Krasnow MA (2019) Rare pulmonary neuroendocrine cells are stem cells regulated by Rb, p53, and notch. Cell 179(2):403–16 e23. https://doi.org/10.1016/j.cell.2019.09.010. (Epub 2019/10/05. PubMed PMID: 31585080)
Song H, Yao E, Lin C, Gacayan R, Chen MH, Chuang PT (2012) Functional characterization of pulmonary neuroendocrine cells in lung development, injury, and tumorigenesis. Proc Natl Acad Sci USA 109(43):17531–17536. https://doi.org/10.1073/pnas.1207238109. (Epub 2012/10/11. PubMed PMID: 23047698; PubMed Central PMCID: PMC3491514)
Sutherland KD, Proost N, Brouns I, Adriaensen D, Song JY, Berns A (2011) Cell of origin of small cell lung cancer: inactivation of Trp53 and Rb1 in distinct cell types of adult mouse lung. Cancer Cell 19(6):754–764. https://doi.org/10.1016/j.ccr.2011.04.019. (Epub 2011/06/15. PubMed PMID: 21665149)
Rudin CM, Durinck S, Stawiski EW, Poirier JT, Modrusan Z, Shames DS et al (2012) Comprehensive genomic analysis identifies SOX2 as a frequently amplified gene in small-cell lung cancer. Nat Genet 44(10):1111–1116. https://doi.org/10.1038/ng.2405. (Epub 2012/09/04. PubMed PMID: 22941189; PubMed Central PMCID: PMCPMC3557461)
Zhu Y, Li Y, Jun Wei JW, Liu X (2012) The role of Sox genes in lung morphogenesis and cancer. Int J Mol Sci 13(12):15767–15783. https://doi.org/10.3390/ijms131215767. (Epub 2013/02/28. PubMed PMID: 23443092; PubMed Central PMCID: PMCPMC3546660)
Gure AO, Stockert E, Scanlan MJ, Keresztes RS, Jager D, Altorki NK et al (2000) Serological identification of embryonic neural proteins as highly immunogenic tumor antigens in small cell lung cancer. Proc Natl Acad Sci USA 97(8):4198–4203 (PubMed PMID: 10760287)
Titulaer MJ, Klooster R, Potman M, Sabater L, Graus F, Hegeman IM et al (2009) SOX antibodies in small-cell lung cancer and Lambert-Eaton myasthenic syndrome: frequency and relation with survival. J Clin Oncol 27(26):4260–4267. https://doi.org/10.1200/JCO.2008.20.6169. (Epub 2009/08/12. PubMed PMID: 19667272)
Schilders K, Eenjes E, Edel G, de Munck AB, van Kempen MB, Demmers J et al (2018) Generation of a biotinylatable Sox2 mouse model to identify Sox2 complexes in vivo. Transgenic Res 27(1):75–85. https://doi.org/10.1007/s11248-018-0058-1. (Epub 2018/02/01. PubMed PMID: 29383478)
Arnold K, Sarkar A, Yram MA, Polo JM, Bronson R, Sengupta S et al (2011) Sox2(+) adult stem and progenitor cells are important for tissue regeneration and survival of mice. Cell Stem Cell 9(4):317–329. https://doi.org/10.1016/j.stem.2011.09.001. (Epub 2011/10/11. PubMed PMID: 21982232; PubMed Central PMCID: PMC3538360)
Acknowledgements
We like to thank Frank Grosveld and Niels Galjart for critically reading the manuscript (Department of Cell Biology, Erasmus MC).
Funding
This work was supported by grants from the Sophia Foundation for Medical Research S14-12 (EE) and S22-38 (FB).
Author information
Authors and Affiliations
Contributions
EE and FB performed the experiments, wrote the main manuscript and prepared the figures; ABM and MBK provided technical support, preformed cell culture experiments. DT and RJR contributed to the experimental set-up, data interpretation and wrote the main manuscript. RJR acquired funding support. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare to have no competing financial and/or non-financial interests in relation to the work described.
Ethics approval
All animal experimental protocols were approved by the animal welfare committee of the veterinary authorities of the Erasmus Medical Center (SP2100184).
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Eenjes, E., Benthem, F., Boerema-de Munck, A. et al. Distinct roles for SOX2 and SOX21 in differentiation, distribution and maturation of pulmonary neuroendocrine cells. Cell. Mol. Life Sci. 80, 79 (2023). https://doi.org/10.1007/s00018-023-04731-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00018-023-04731-w