Abstract
Although combination antiretroviral therapy (ART) has reduced mortality and improved lifespan for people living with HIV, it does not provide a cure. Patients must be on ART for the rest of their lives and contend with side effects, unsustainable costs, and the development of drug resistance. A cure for HIV is, therefore, warranted to avoid the limitations of the current therapy and restore full health. However, this cure is difficult to find due to the persistence of latently infected HIV cellular reservoirs during suppressive ART. Approaches to HIV cure being investigated include boosting the host immune system, genetic approaches to disable co-receptors and the viral genome, purging cells harboring latent HIV with latency-reversing latency agents (LRAs) (shock and kill), intensifying ART as a cure, preventing replication of latent proviruses (block and lock) and boosting T cell turnover to reduce HIV-1 reservoirs (rinse and replace). Since most people living with HIV are in Africa, methods being developed for a cure must be amenable to clinical trials and deployment on the continent. This review discusses the current approaches to HIV cure and comments on their appropriateness for Africa.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The HIV pandemic remains one of medicine’s greatest challenges with an estimated 38.0 million people living with HIV (PLWH) of which the vast majority (25.7 million) are in Africa [1]. While antiretroviral therapy (ART) can halt viral replication, reduce mortality, and improve the lifespan of PLWH, this treatment is lifelong, expensive, inaccessible to many, and cannot eradicate the latent virus [2, 3]. It is difficult to find a cure for HIV due to the persistence of latently infected cells that produce the virus following interruption of ART [4, 5], presence of long-lived HIV-infected resting memory CD4 + T cells [6,7,8,9,10], the exhaustion of HIV-specific CD8 + T cells [11, 12] and the difficulty in reaching anatomic sanctuary sites by HIV-specific CD8 + T cells [5, 13]. More so, the current World Health Organization (WHO) eligibility criteria of treating all regardless of CD4 count has increased the funding gap for ART [14]. In addition, with the decline of global funding for HIV [15] and the negative impact of the COVID-19 pandemic on the world economy, sustaining existing HIV/AIDS treatment programs is becoming even more challenging making the quest for a cure more acute. Between 2013 and 2020, despite considerable increase in the number of patients on ART, HIV funding has been generally flat or reduced (Fig. 1).
Three categories of HIV cure approaches have been identified: eradication cure (elimination of all viral reservoirs), functional cure (immune control without reservoir eradication), or a hybrid cure (reservoir reduction with improved immune control) [16]. Approaches to HIV cure under investigation include boosting the host immune system, genetic approaches to disable co-receptors or the viral genome, modification of host cells to resist HIV, and engineered T cells to eliminate HIV-infected cells. Others include therapeutic vaccination, broadly neutralizing antibodies, purging cells harboring latent HIV with latency-reversing agents (LRAs), preventing replication of latent proviruses (block and lock) and boosting T cell change to reduce HIV-1 reservoirs (rinse and replace) [2, 3, 17,18,19,20,21,22,23,24]. As HIV cure research advances, it is important to bear in mind that the vast majority of PLWH who will need these cures live in Africa where resources are limited. Therefore, there is a need to design cure strategies that will be feasible for clinical trial and implementation in Africa. This article reviews ongoing HIV cure approaches, discusses their pros and cons, and suggests how they could be made suitable for Africa.
Progress towards HIV cure
Tremendous successes have been chalked in the fight against HIV since its identification forty years ago [25]. Globally, ART is now available to about 70% of PLWH, thus increasing life span and reducing deaths due to HIV/AIDS [1]. The hope to find a cure for HIV was high after initial reports of both eradication and functional cures. Timothy Brown, the Berlin patient, was the first reported case of eradication cure. He received a bone marrow transplant for acute myeloid leukemia from a donor who was negative for HIV and had a mutation in the CCR5 co-receptor, which is required for HIV entry. He lived free of HIV for close to 10 years [26] and died in 2020 from complications of leukemia. After unsuccessful attempts to replicate the same procedure [27, 28], there was a success in another individual, the London patient, who achieved long-term suppression of HIV-1 [29].
The first reported case of a functional cure was the Mississippi baby who was born to an HIV-positive mother and given a full dose of ART 30 h after birth. Although the family interrupted ART for 18 months, surprisingly HIV remained undetected in the blood [30]. Notwithstanding, there was a rebound of detectable HIV in plasma after 27.6 months [31]. Subsequently, the VISCONTI (Virological and Immunological Studies in Controllers after Treatment Interruption) study in 2013 implied that early initiation of ART enables some patients to maintain a low viral load after ART interruption [32]. Since these initial reports, there has been growing interest in developing curative therapies for HIV.
HIV cure strategies
Stem cell transplants
Ferrebee and Thomas [33] pioneered stem cell therapy which has now become a platform for treating leukemias, lymphomas, and many other malignancies. In people living with HIV, the procedure was first carried out in the 1980s oblivious of the virus since there were no assays to detect HIV [34]. Following this report, several attempts were made to treat HIV using this approach but they were not successful [35,36,37,38]. However, the Berlin patient gave a glimpse of hope that achieving a virus-free state in the absence of ART is possible. Despite that, stem cell transplant for treating HIV is not practical for several reasons. First, stem cell transplant is a high-risk procedure with a high mortality rate and thus only employed in the most desperate situations such as uncontrolled leukemia. The conditioning and bone marrow ablation required for the procedure put the patient at great risk of dying from infections. Second, even in developed countries, the procedure is performed in the most specialized centers where specially trained oncologists, geneticists, hemato-pathologists, and infectious disease specialists are available. These are resources that are mostly not available in Africa. Third, the relative paucity of potential CCR5∆32 donors (less than 1% of Caucasians) make it unattractive as a potential therapy. Thus, while stem cell transplants indicate that HIV is potentially curable, it is not a practical strategy for ending the AIDS pandemic.
Shock and kill
The shock and kill approach uses compounds known as latency-reversing agents (LRAs) to reactivate the latent provirus. The premise is that patients will be given agents that reactivate the latent virus in the resting CD4 + T cells whiles they are on ART (Fig. 2). Since viral replication is usually toxic to CD4 + T cells, it is expected that reactivation will result in the death of these cells, and because patients will be on ART at this stage, any virus produced will not be able to infect bystander cells. In addition, once viral production begins in the resting T cells, they will be recognized by the immune system for clearance. With the clearance of the infected resting T cells, patients could stop taking ART and undergo occasional monitoring. The concept of the shock and kill approach is hinged on reducing the size of the latent reservoir and limiting viral rebound [39]. In theory, any agent that can stimulate resting T cells enough for viral production could be an LRA. Interleukin 2 (IL-2), anti-CD3 antibody, and TNFα were the first LRAs tested in HIV-infected individuals receiving ART [40, 41], however, the results were not promising. The T cell receptor agonist and PMA (phorbol 12-myristate 13-acetate) were used but resulted in global T cell activation. Therefore, an ideal LRA should be able to cause proviral latency reversal without global T cell activation. Latency-reversing agents (LRAs) have been categorized based on their mechanism of action as shown in Table 1 [39]. Of the six categories, the histone deacetylase inhibitors (HDACis) have received much attention with several small early clinical trials completed [39, 42,43,44,45]. Although the shock and kill approach is the most clinically advanced cure strategy, clinical studies have focused mainly on HDACis which are already approved for cancer therapy. In these trials, some adverse effects have been reported [46]. These include but not limited to nausea, vomiting, anorexia [47,48,49,50]; fatigue [51,52,53]; and skin changes [54]. HDACis have been the focus of many researchers and investigated extensively as a potential LRA [55,56,57]. However, several groups have shown that one LRA might not be effective in reducing the size of the reservoir either due to an inadequate reactivation or lack of an effective kill or both [39, 58, 59] and suggested a combination of two or more LRAs to achieve robust viral reactivation and a significant reduction in reservoir size [60,61,62,63,64,65]. Concerning potential combination LRA treatment, one of the most favored is a combination of HDACi and PKC agonists like bryostatin [66, 67]. However, there are fears of severe side effects with bryostatin. Therefore, the idea is to use a lower concentration of bryostatin to synergize with HDACis to achieve potent reactivation while reducing the chances of adverse events. That said, this combination is yet to be tested in clinical trials. Even though, the combination increases the reactivation potential, several obstacles exist in the killing of the reactivated cells. This includes the resistance of the cells to apoptosis, exhaustion of CD8 + cells, and immune escape mutation in chronically infected individuals [68]. Nonetheless, Herzig and his colleagues recently proposed a more effective kill of reactivated cells by utilizing chimeric antigen receptors (CAR) coupled with broadly neutralizing antibodies in an ex vivo study [69].
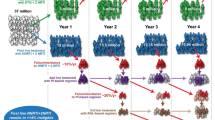
Strategies under development for an HIV cure. A Shock and kill approach using latency-reversing agents (LRAs) to eradicate the latent reservoir. B Gene therapy utilizing CRISPR to target the latent reservoir. C Block and lock approach using latency-inducing agents to induce silencing of the latent reservoir. D Immune-based therapies using therapeutic vaccines, CAR-T cells, and broadly neutralizing antibodies. HDAC Histone deacetylase, HMT histone methyl transferase, PKC protein kinase C, dCA didehydro-cortistatin A, ART antiretroviral therapy
Another scenario where the T cells could be stimulated is the “Rinse and Replace” approach [24]. This is a yet to be tested approach which proposes that under ART, T cell activation could be induced to produce waves of polyclonal T cell differentiation resulting in a situation where latently infected cells are replaced by new uninfected cells. The infected cells are continually “washed out” due to the continuous activation, differentiation, and cell death. This approach is different from the shock and kill approach in that it promotes physiological replacement of frequently activated cells. In addition, only a proportion of latently infected cells are required to express virus that provides adjuvant effects thus helping to induce a potent flux. Furthermore, this functional cure approach aims to combine other cure approaches such as block and lock to prevent viral rebound in the absence of ART. The approach also strongly recommends several rounds of ART and starting ART on time thus raising the concern of negative side effects, adherence, access to ART and cost. Implementing this approach in Africa will be problematic because of the concerns raised on ART. Moreover, clinical trials regarding this approach are yet to commence.
The shock and kill approach could be easily deployed in resource limited settings like Africa, since all that is required is adding another agent to the current ART regimen. That said, an oral regimen will be more preferable to intravenous infusions in an African setting.
The figure was Created with BioRender.com.
Block and lock
The block and lock approach proposes the use of small interfering RNAs (siRNA) [20, 112, 113] or latency-inducing agents [114, 115] to effect transcriptional silencing (block) at the HIV promoter using epigenetic mechanisms to lock the integrated viral genome in a permanent position thereby preventing transcription of new virions even when ART is stopped (Fig. 2). The HIV transactivator of transcription (Tat) protein has been an important target for this approach since it is essential for transcription initiation and elongation. The tat protein which is produced early during the life cycle of HIV promotes the transcription of HIV by binding to the transactivation response element (TAR) and recruiting the positive transcription elongation factor B (P-TEFb) to promote transcriptional elongation [115]. By far the most advanced strategy of the block and lock has been the use of a Tat inhibitor didehydro-cortistatin A (dCA) to enforce HIV latency [21, 115–119]. This compound has shown remarkable success in maintaining viral latency ex vivo in primary T cells from virologically suppressed patients and in vivo in mouse models. In both instances, dCA delayed viral rebound when ART was interrupted [21, 120, 121]. Similar results for dCA were reported in HIV-2 and Simian Immunodeficiency Virus (SIV) [122]. With no obvious adverse effects and the ability to cross the blood brain barrier, dCA may be able to reach the brain and other sanctuary sites of the body where HIV resides [123]. However, induction of resistance to dCA in the laboratory has been reported [124] making it worthwhile to investigate if some strains of HIV may be resistant to the compound. Although there is evidence that dCA induces epigenetic modification of the HIV promoter, the mechanism is not well understood.
Triptolide (TPL), which is predominantly used for the treatment of rheumatoid arthritis, inhibits the function of Tat, thereby promoting latency [125]. Others found that TPL blocks RNAPII and prevents it from initiating transcription [126]. However, the ability of this molecule to interfere with important cellular functions limits its clinical use [118]. Another inhibitor of Tat-mediated viral transcription is Levosimendan, an FDA-approved drug for the treatment of heart-related conditions [127]. The effects of other inhibitors targeting host factors or signaling pathways required for the transcription of viruses, such as P-TEFb, heat shock protein 90 (HSP90), mammalian target of rapamycin (mTOR) complex, facilitates chromatin transactions (FACT), bromodomain-containing protein 4 (BRD4), and xeroderma pigmentosum subtype B (XPB) is reviewed elsewhere [118, 120, 128]. The absence of cellular homologs and the limited off-target effects when Tat-TAR is inhibited makes it an outstanding target for the block and lock approach [119]. The block and lock strategy targets long-term remission of HIV in a more specific manner without global T cell activation, compared to some non-specific shock and kill strategies. Even though the block and lock approach is potentially scalable and could be deployed in Africa if successful, it still awaits human trials to determine feasibility.
Gene therapy to eradicate HIV reservoirs
Innovative ways targeting the genome of HIV in a bid to find a cure for the infection have advanced over the past decades. The idea of gene editing therapy is to alter a selected gene locus to change or interrupt its normal function using engineered nucleases (Fig. 2). This results in deletions or additions at the selected gene target site [22, 129]. Two main repair pathways are involved: (i) non-homologous end-joining (NHEJ) where the break ends are directly ligated without a homologous template and (ii) homology-directed repair pathway (HDR) in which homologous sequences are introduced to guide the repair [130]. The HDR yields limited off-target genome effects [131]. Forms of potential gene editing-based HIV therapies include zinc finger nuclease (ZFN), transcription activator-like nucleases (TALENS), and clustered regularly interspaced short palindromic repeats (CRISPR)-associated protein 9 (CRISPR/Cas9).
The most tried gene therapy approach targeting HIV infection is the ZFN [132]. It is made up of two domains; the FokI endonuclease for cleaving target sequences and the Cys2-His2 zinc-finger proteins (ZFPs) for specific DNA-binding [133, 134]. The initial report utilizing ZFN targeting the C–X–C chemokine receptor type 4 gene (CXCR4) disruption displayed promising results [135, 136]. Given that the mutant CCR5∆32 protein confers resistance to HIV infection and the Berlin patient was cured using a ∆32 stem cell transplant, the CCR5 became an ideal target for HIV gene therapy. Following the initial use of the ZFN approach to disrupt the CCR5 in HIV-infected cells [137, 138], several other works have built upon this strategy [138–141]. More so, there is evidence that ZFN could be used to disrupt the CCR5 in human induced pluripotent stem cells and human embryonic stem cells [142]. Similarly, HIV resistance was achieved when mice were treated with CCR5-disrupted gene hematopoietic stem cells [139]. In a landmark safety study in humans, Tebas et al. used ZFN to edit CCR5 out of CD4 + T cells isolated from HIV patients, and reinfused into the same patients showing that the procedure was safe [143].
In contrast to ZFN, TALENs are engineered proteins that can cleave dsDNA sequences in a single base pair modular fashion [144, 145]. This presupposes that they can bind to wider DNA targets than ZFN. The DNA binding proteins are derived naturally from Xanthomonas, a plant bacterial pathogen [146]. Clinically, TALENS are yet to be applied for the treatment of HIV. However, the success of several experimental studies indicates that it can be scaled up for HIV [147–149]. In comparison to ZFNs, TALENs are cost-effective but difficult to generate, bulkier and the delivery to several cell targets is challenging [150]
The CRISPR/Cas9 uses short-guide RNA (gRNA) to target a specific DNA sequence after cleaving the double-stranded DNA by the Cas9 endonuclease. The double-stranded DNA is then repaired by NHEJ or homologous recombination [151]. The CRISPR/Cas9 approach is being widely investigated as a tool to combat various diseases [152–155]. Ebina et al. first applied this technology to HIV in cell culture to target the integrated provirus [156]. Subsequently, several scientists have explored the potential of the CRISPR/Cas9 technology to target the virus in cell culture systems and mouse models [157–162]. The versatility of the CRISPR/Cas9 technology is such that it can also be used to target co-receptors, restriction factors, and proteins that promote or inhibit HIV latency. It has also been demonstrated that the CCR5 could be silenced in a human embryonic kidney (HEK) 293 T cells transfected with Cas9 and sgRNAs [163]. Subsequently, the piggyBac technology was used to enhance a homozygous ∆32 mutation in induced pluripotent stem cells via the CRISPR/Cas9 system [158]. Wang and his research group also knocked out CCR5 co-receptors using lentivirus vectors to express CCR5-sgRNA and Cas9 [157]. The CXCR4 co-receptor has also been disrupted by the CRISPR/Cas9 technology in CD4 + T cells of humans and rhesus macaque [159]. In addition, this technology has been explored to reactivate HIV. Scientists have used deficient Cas9 (dCas9) coupled with transcription activator domains to trigger the transcription of HIV in latent reservoir cells [164–167].
Since host restriction factors are weakly expressed during HIV infection [168, 169], CRISPR/Cas9 has been utilized in the activation of the expression of these enzymes. For instance, the technology was used in cell culture systems to induce the expression of APOBEC3G (A3G) and APOBFC3B (A3B) to inhibit HIV infection [170]. Disadvantages of the CRISPR/Cas9 technology include potential off-target effects that could induce gene mutations, and the lack of an effective delivery system [164, 166, 169, 171–173]. While adenoviral vectors have been effective in delivering CRISPR/Cas9 [174, 175], lentiviral mode of delivery could increase the risk of off-target effects [157, 176]. Cytotoxicity and immune tolerance are also limitations in the use of the CRISPR-based technology in the fight against HIV [173].
If successful, could gene therapy be widely deployed in Africa? The answer will depend on several key factors. Methods like taking CD4 + T cells from patients, modifying them in the laboratory, and reinfusing them into the same patient (autologous transplant) will be challenging to implement. However, if a simple mode of delivery is found for CRISPR/Cas9 for instance, such a method could be deployed widely if the cost is reasonable. The critical issue for gene therapy however is whether patients doing well on ART will accept gene modification with unknown risks for mutations, malignancies, and other potentially serious adverse effects.
Intensification of antiretroviral therapy as a cure
The serendipitous report of the HIV-positive Mississippi baby who was put on ART 30 h after birth and was able to maintain viral suppression off treatment for 27.6 months raised the hopes that early ART could lead to a possible functional cure, especially if started early after the initial infection [30, 31]. A similar report was also observed in a French girl who has gone into remission for over 12 years after starting and interrupting ART at 3 months and 6.5 years old respectively [177]. Recently, a report from South Africa indicates that a child who started treatment at 2 months and discontinued it after 10 months has remained in remission for 9.5 years [178]. Using an analytic treatment interruption (ATI) method, remission was also observed in some children in South Africa who started ART as early as 14 days after birth [181]. Furthermore, in adults, it has been shown that initiating ART earlier results in HIV remission and smaller reservoir size [32, 179–184].
Bearing in mind that the norm is viral rebound within weeks of interrupting ART in the majority of HIV-infected persons, these findings highlight the potential benefits of early ART. Nonetheless, very early ART on its own is not likely to achieve sustained virologic remission in the majority of HIV-infected persons. A combination of early ART with other curative strategies such as broadly neutralizing antibodies, and therapeutic vaccines is likely to be the ultimate approach. Initiating early ART should be based on improving clinical outcomes rather than achieving remission [57, 185].
The main problem with this approach is that most patients in Africa and elsewhere live with HIV for years before they recognize they have the infection, by which time the reservoir is well established and ART alone will not result in long-term remission. Moreso, access to ART and funding still remains a challenge and thus its sustainability on the African continent is questionable.
Immune-based interventions
HIV causes severe damage to the immune system of the host [186, 187], destroys CD4 T cells, and evades immune responses [188]. The aim here is to compensate for the loss of CD4 T cells [189], augment the anti-viral effects of CD8 + T cells (CTLs), and enhance neutralizing antibody-mediated killing of infected cells [190–192]. In this section, therapeutic vaccines, broadly neutralizing antibodies (bNab), and chimeric antigen receptors will be highlighted (Fig. 2). Therapeutic vaccines aim to increase the magnitude and function of anti-HIV immunity by facilitating long-term viral control without ART [193]. They are administered after a disease or infection has already occurred using the patient’s immune system to fight the infection [192]. In this case, they would produce HIV-specific immune responses to better control the virus when ART is interrupted [194] by eliciting anti-viral CD8 T cells (CTLs), and neutralizing antibodies [190–192, 195]. Therapeutic vaccines may also produce polyfunctional T cells which will release multiple cytokines and perform effector functions [196, 197].
Therapeutic vaccines are used to augment CTLs to increase their cytotoxicity capacity [198, 199]. CTLs are a major component of the host response to HIV [200–203] and are usually exhausted due to their persistent exposure to HIV which impairs their killing ability [204, 205]. Even during HIV control using ART, there is reduced virus-specific CD8 T cell responses [206, 207], implying that to control HIV, T cells need to be augmented. So far, therapeutic vaccine trials have failed to achieve functional HIV cure and sustained viral control after ART was stopped [208–211]
Research has also shown that broadly neutralizing antibodies (bNAbs) can control HIV replication [212–217]. All bnAbs target the HIV-1 Envelope (Env) glycoprotein 120 (gp120) and gp41 [218]. The first-generation bNabs (b12, 447-52D, 2G12, 17b, 2F5, 4E10 and Z13) [219–221] generated little clinical effect on HIV [222]. Next, antibodies targeting the CD4 binding site (VRC01, 3BNC117, VRC01-LS, and VRC07-523LS), the glycan-rich V3 loop (10–1074 and PGT121), the V2-glycan site (PGDM1400, CAP256-VRC26.25) and MPER epitope (10E8) were identified [218, 223, 224]. All these bNAbs have shown different levels of protection against Simian Immunodeficiency Virus (SHIV) [225–228]. Research has shown that combining two or more bNAbs is effective in enhancing a broad viral coverage [229–233]. bnAbs against HIV have shown significant promise for their potential use in the control of HIV, [218, 223], however, one setback is to identify combinations of bnAbs that will increase the breadth to cover circulating variants [218, 234]. In terms of HIV cure or remission, bNAbs could be given to patients who are virologically suppressed on ART to keep the virus undetected after withdrawal of ART. Issues of broad coverage against HIV variants and how long these antibodies last in vivo will be critical for success. They could also be deployed as part of a therapeutic vaccine strategy as discussed above.
Chimeric antigen receptors (CAR), following their breakthrough in the treatment of cancers [235–237], are currently being employed to enhance recognition and the killing of cells infected with HIV [69, 238]. They are produced by first removing patient T cells and inserting into them a CAR against a specific antigen [239] and then reinfusing the CAR-T cells into the patient [240], thus giving T cells a new ability to target a specific protein. This technology has seen the production of four generations so far [241–245]. The CAR T cell therapy can be used in conjunction with LRA to enhance the killing of reactivated cells, thus reducing the size of the reservoir [69].
Even though CAR-T cell therapy has proven to be beneficial, it faces some challenges. The cost of treating a single patient as well as the technical expertise in designing the therapy limits its use not only in Africa but even in the developed world [246].
Another important limitation is the lack of universality since they need to be produced from autologous T cells. More so, the long process in the design and treatment of patients makes it difficult to implement in developing countries [247]
These limitations notwithstanding, researchers are working to produce universal CAR T cells that could be taken off the shelf and given to any patient. It is expected that over time, the technology will mature, become easier to manage and less expensive.
Which HIV cure strategies are appropriate for Africa?
It is worth mentioning that, none of the HIV cure strategies discussed here has been tried on the African soil. Only a few targeting therapeutic vaccines and HIV treatment have been tried in African as shown in Table 2.
As cure strategies are developed, several factors inherent to the strategies themselves will determine how appropriate they are for Africa. These factors include the type of cure offered (functional versus complete), ease of deployment, perceived risks, and cost.
First, the type of cure offered, whether complete or functional could be important in determining patient acceptance. Ideally, a complete cure for HIV is desirable as it gives peace of mind to patients that the virus is gone. However, some form of functional cure is likely to be more feasible in the near future. How will PLWH in Africa perceive sustained remission or functional cure? Such a cure will mean patients need to have periodic follow-up. Moreover, will PLWH in Africa accept that they are ‘cured’ but there is still virus in the body as approaches like block and lock are likely to provide? Will they see sustained remission as a major improvement over daily ART, and or will anxieties about potential viral rebounds keep patients away from such remedies? These are important questions that need to be answered now before these remedies become realities. Knowing patient and caregiver perspectives on these issues could inform the design of clinical trials for remission strategies and ultimately the kind of cures made available to patients. Second, a cure that is easy to deploy is more likely to gain ground in Africa. Gene therapy, especially the type that requires pheresis of white cells from patients, modification and reintroduction will be expensive and logistically difficult to implement in Africa. When it comes to gene therapy, efforts should concentrate on developing delivery vehicles for methods like CRISPR/Cas9 for easy administration. Methods like shock and kill and block and lock or even therapeutic vaccines that require adding another agent to ART will be easier to deploy. Even for these, the route of administration will be important. Treatments that are given orally or simple intramuscular injections will be easier than methods that require admission and intravenous infusions at a medical center. Therefore, all these factors should be taken into consideration as scientists develop these cure strategies. Third, the perceived risks and benefits of the cure method offered in Africa will be critical in determining uptake or even participation in clinical trials. How will methods such as CRISPR/Cas9-mediated gene therapy that modify part of the patient’s genome be viewed in Africa? With the current COVID-19 pandemic, there is a lot of misinformation and resistance to mRNA vaccines in Africa even though they do not modify the genome. This will call for close collaboration and education for both providers and patients about trial methods prior to their availability in clinical trials. Such education and interactions may even help modify some of the methods that are eventually brought to patients. Finally, the cost of the intervention will also be crucial in ensuring wide deployment in Africa. In Africa, HIV treatment is heavily dependent on donor support with the constant threat that this support could be cut off. The ideal HIV cure for Africa should therefore be affordable, easy to administer, have short treatment time and as be low risk as possible. These factors should be at the forefront as scientists develop HIV cures. African scientists should be involved in the development and clinical trials of these strategies to engender confidence among patients. Of all the cure strategies discussed, the shock and kill block and lock, therapeutic vaccines, and perhaps CRISPR/Cas9-based treatment with easy delivery systems will be most appropriate for the African continent.
Data availability statement
Not applicable.
References
UNAIDS/WHO estimates (2020) Fact sheet - Latest global and regional statistics on the status of the AIDS epidemic. Accessed: Dec 10, 2021. https://www.unaids.org/en/resources/fact-sheet
Ananworanich J (2015) What will it take to cure HIV? Topics Antiviral Med 23(2):80–84
Passaes CP, Sáez-Cirión A (2014) HIV cure research: advances and prospects. Virology 454–455(1):340–352. https://doi.org/10.1016/j.virol.2014.02.021 (Academic Press Inc.)
Banga R et al (2016) PD-1+ and follicular helper T cells are responsible for persistent HIV-1 transcription in treated aviremic individuals. Nat Med 22(7):754–761. https://doi.org/10.1038/nm.4113
Fukazawa Y et al (2015) B cell follicle sanctuary permits persistent productive simian immunodeficiency virus infection in elite controllers. Nat Med 21(2):132–139. https://doi.org/10.1038/nm.3781
Chun TW et al (1997) Presence of an inducible HIV-1 latent reservoir during highly active antiretroviral therapy. Proc Natl Acad Sci USA 94(24):13193–13197. https://doi.org/10.1073/pnas.94.24.13193
Finzi D et al (1997) Identification of a reservoir for HIV-1 in patients on highly active antiretroviral therapy. Science (1979) 278(5341):1295–1300. https://doi.org/10.1126/science.278.5341.1295
Jd S et al (2003) Long-term follow-up studies confirm the stability of the latent reservoir for HIV-1 in resting CD4+ T cells. Nat Med. https://doi.org/10.1038/NM880
Chomont N et al (2009) HIV reservoir size and persistence are driven by T cell survival and homeostatic proliferation. Nat Med 15(8):893–900. https://doi.org/10.1038/nm.1972
Buzon MJ et al (2014) HIV-1 persistence in CD4+ T cells with stem cell-like properties. Nat Med 20(2):139–142. https://doi.org/10.1038/nm.3445
Petrovas C et al (2006) PD-1 is a regulator of virus-specific CD8+ T cell survival in HIV infection. J Exp Med 203(10):2281–2292. https://doi.org/10.1084/jem.20061496
Trautmann L et al (2006) Upregulation of PD-1 expression on HIV-specific CD8+ T cells leads to reversible immune dysfunction. Nat Med 12(10):1198–1202. https://doi.org/10.1038/nm1482
Connick E et al (2007) CTL Fail to Accumulate at Sites of HIV-1 Replication in Lymphoid Tissue. J Immunol 178(11):6975–6983. https://doi.org/10.4049/jimmunol.178.11.6975
Dutta A, Barker C, Kallarakal A (2015) The HIV treatment gap: estimates of the financial resources needed versus available for scale-up of antiretroviral therapy in 97 countries from 2015 to 2020. PLoS Med 12(11):e1001907. https://doi.org/10.1371/JOURNAL.PMED.1001907
Joint United Nations Programme on HIV/AIDS, “Joint United Nations Programme on HIV/AIDS. The Gap Report,” Geneva, 2014. Accessed: May 23, 2022. https://www.unaids.org/en/resources/documents/2014/20140716_UNAIDS_gap_report
Cillo AR, Mellors JW (2016) Which therapeutic strategy will achieve a cure for HIV-1? Curr Opin Virol 18:14–19. https://doi.org/10.1016/j.coviro.2016.02.001
Battistini A, Sgarbanti M (2014) HIV-1 latency: an update of molecular mechanisms and therapeutic strategies. Viruses 6(4):1715–1758. https://doi.org/10.3390/v6041715 (MDPI AG)
Choudhary SK, Margolis DM (2011) Curing HIV: pharmacologic approaches to target HIV-1 latency. Annu Rev Pharmacol Toxicol 51:397–418. https://doi.org/10.1146/annurev-pharmtox-010510-100237
Kwarteng A, Ahuno ST, Kwakye-Nuako G (2017) The therapeutic landscape of HIV-1 via genome editing. AIDS Res Ther 14(1):32. https://doi.org/10.1186/s12981-017-0157-8
Méndez C, Ledger S, Petoumenos K, Ahlenstiel C, Kelleher AD (2018) RNA-induced epigenetic silencing inhibits HIV-1 reactivation from latency. Retrovirology 15(1):1–18. https://doi.org/10.1186/s12977-018-0451-0
Mousseau G, Kessing CF, Fromentin R, Trautmann L, Chomont N, Valente ST (2015) The tat inhibitor didehydro-cortistatin a prevents HIV-1 reactivation from latency. MBio 6(4):1–14. https://doi.org/10.1128/mBio.00465-15
Ognenovska K et al (2019) Mechanisms for controlling HIV-1 infection: a gene therapy approach. In Vivo Ex Vivo Gene Ther Inherited Non-Inherited Disord. https://doi.org/10.5772/intechopen.79669
Kyei GB et al (2018) Splicing factor 3B subunit 1 interacts with HIV tat and plays a role in viral transcription and reactivation from latency. MBio 9(6):1–14. https://doi.org/10.1128/mBio.01423-18
Grossman Z et al (2020) ‘Rinse and replace’: boosting T cell turnover to reduce HIV-1 reservoirs. Trends Immunol 41(6):466–480. https://doi.org/10.1016/J.IT.2020.04.003
Barré-Sinoussi JC, Chermann F, Rey F, Nugeyre MT, Chamaret S, Gruest J, Montagnier L (1983) Isolation of a T-lymphotropic retrovirus from a patient at risk for acquired immune deficiency syndrome (AIDS). Science (1979) 220(4599):868–871
Hütter G et al (2009) Long-term control of HIV by CCR5 Delta32/ Delta32 stem-cell transplantation
Abbate A, Gold KJ, Goldman JWEBD, Kathryn L, Moseley MPH (2014) More on shift of HIV tropism in stem-cell transplantation with CCR5 Delta32/Delta32 mutation. N Engl J Med 371(25):2437–2438. https://doi.org/10.1056/NEJMc1412279
Kordelas L, Verheyen J, Esser S (2014) Shift of HIV tropism in stem-cell transplantation with CCR5 Delta32 mutation. N Engl J Med 371(9):880–882. https://doi.org/10.1056/NEJMc1405805
Gupta RK et al (2020) Evidence for HIV-1 cure after CCR5Δ32/Δ32 allogeneic haemopoietic stem-cell transplantation 30 months post analytical treatment interruption: a case report. The Lancet HIV 7(5):e340–e347. https://doi.org/10.1016/S2352-3018(20)30069-2
Persaud D et al (2013) Absence of detectable HIV-1 viremia after treatment cessation in an infant. N Engl J Med 369(19):1828–1835. https://doi.org/10.1056/nejmoa1302976
Luzuriaga K et al (2015) Viremic relapse after HIV-1 remission in a perinatally infected child. N Engl J Med 372(8):786–788. https://doi.org/10.1056/nejmc1413931
Sáez-Cirión A et al (2013) Post-treatment HIV-1 controllers with a long-term virological remission after the interruption of early initiated antiretroviral therapy ANRS VISCONTI study. PLoS Pathog. https://doi.org/10.1371/journal.ppat.1003211
Ferrebee JW, Thomas ED (1960) Transplantation of marrow in man. Arch Intern Med 106(4):523–531. https://doi.org/10.1001/archinte.1960.03820040061007
Hassett J, Zaroulis C, Greenberg M, Fiegal F (1983) Bone-marrow transplantation in AIDS. N Engl J Med 309(11):665–665. https://doi.org/10.1056/NEJM198309153091114
Verdonck LF, De Gast GC, Lange JMA, Schuurman HJ, Dekker AW, Bast BJEG (1988) Syngeneic leukocytes together with suramin failed to improve immunodeficiency in a case of transfusion-associated AIDS after syngeneic bone marrow transplantation. Blood 71(3):666–671. https://doi.org/10.1182/blood.v71.3.666.666
Furlini G, Re MC, Bandini G, Albertazzi L, La Placa M (1988) Antibody response to human immunodeficiency virus after infected bone marrow transplant. Eur J Clin Microbiol Infect Dis 7(5):664–666. https://doi.org/10.1007/BF01964248
Lane HC et al (1984) Partial immune reconstitution in a patient with the acquired immunodeficiency syndrome. N Engl J Med 311(17):1099–1103. https://doi.org/10.1056/nejm198410253111706
Vilmer E et al (1987) Clinical and immunological restoration in patients with aids after marrow transplantation, using lymphocyte transfusions from the marrow donor. Transplantation 44(1):25–29. https://doi.org/10.1097/00007890-198707000-00007
Abner E, Jordan A (2019) HIV ‘shock and kill’ therapy: in need of revision. Antiviral Res 166(November 2018):19–34. https://doi.org/10.1016/j.antiviral.2019.03.008
Chun TW et al (1999) Effect of interleukin-2 on the pool of latently infected, resting CD4+ T cells in HIV-1-infected patients receiving highly active anti-retroviral therapy. Nat Med 5(6):651–655. https://doi.org/10.1038/9498
Prins JM et al (1999) Immune-activation with anti-CD3 and recombinant human IL-2 in HIV-1-infected patients on potent antiretroviral therapy. AIDS 13(17):2405–2410. https://doi.org/10.1097/00002030-199912030-00012
Archin NM et al (2012) Administration of vorinostat disrupts HIV-1 latency in patients on antiretroviral therapy. Nature 487(7408):482–485. https://doi.org/10.1038/nature11286
Archin NM, Margolis DM (2014) Emerging strategies to deplete the HIV reservoir. Curr Opin Infect Dis. https://doi.org/10.1097/QCO.0000000000000026
Aid M et al (2018) Follicular CD4 T helper cells as a major HIV reservoir compartment: a molecular perspective. Front Immunol. https://doi.org/10.3389/fimmu.2018.00895
Delagrèverie HM, Delaugerre C, Lewin SR, Deeks SG, Li JZ (2016) Ongoing clinical trials of human immunodeficiency virus latency-reversing and immunomodulatory agents. Open Forum Infect Dis. https://doi.org/10.1093/ofid/ofw189 (Oxford University Press)
Subramanian S, Bates SE, Wright JJ, Espinoza-Delgado I, Piekarz RL (2010) Clinical toxicities of histone deacetylase inhibitors. Pharmaceuticals 2010 3:2751–2767. https://doi.org/10.3390/PH3092751
Patnaik A et al (2002) A phase I study of pivaloyloxymethyl butyrate, a prodrug of the differentiating agent butyric acid, in patients with advanced solid malignancies. Clin Cancer Res 8(7):2142–2148
Camacho LH, Olson J, Tong WP, Young CW, Spriggs DR, Malkin MG (2007) Phase I dose escalation clinical trial of phenylbutyrate sodium administered twice daily to patients with advanced solid tumors. Invest New Drugs 25(2):131–138. https://doi.org/10.1007/S10637-006-9017-4
Atmaca A et al (2007) Valproic acid (VPA) in patients with refractory advanced cancer: a dose escalating phase I clinical trial. Br J Cancer 97(2):177–182. https://doi.org/10.1038/SJ.BJC.6603851
Gimsing P et al (2008) A phase I clinical trial of the histone deacetylase inhibitor belinostat in patients with advanced hematological neoplasia. Eur J Haematol 81(3):170–176. https://doi.org/10.1111/j.1600-0609.2008.01102.x
Kummar S et al (2007) Phase I trial of MS-275, a histone deacetylase inhibitor, administered weekly in refractory solid tumors and lymphoid malignancies. Clin Cancer Res 13(18 Pt 1):5411–5417. https://doi.org/10.1158/1078-0432.CCR-07-0791
Olsen EA et al (2007) Phase IIb multicenter trial of vorinostat in patients with persistent, progressive, or treatment refractory cutaneous T-cell lymphoma. J Clin Oncol 25(21):3109–3115. https://doi.org/10.1200/JCO.2006.10.2434
Crump M et al (2008) Phase II trial of oral vorinostat (suberoylanilide hydroxamic acid) in relapsed diffuse large-B-cell lymphoma. Ann Oncol 19(5):964–969. https://doi.org/10.1093/ANNONC/MDN031
Galanis E et al (2009) Phase II trial of vorinostat in recurrent glioblastoma multiforme: a north central cancer treatment group study. J Clin Oncol 27(12):2052–2058. https://doi.org/10.1200/JCO.2008.19.0694
Lehrman G et al (2005) Depletion of latent HIV-1 infection in vivo: a proof-of-concept study. Lancet 366(9485):549–555. https://doi.org/10.1016/S0140-6736(05)67098-5
Margolis DM (2011) Histone deacetylase inhibitors and HIV latency. Curr Opin HIV AIDS 6(1):25–29. https://doi.org/10.1097/COH.0b013e328341242d
Chun TW, Moir S, Fauci AS (2015) HIV reservoirs as obstacles and opportunities for an HIV cure. Nat Immunol 16(6):584–589. https://doi.org/10.1038/ni.3152
Chen HC, Martinez JP, Zorita E, Meyerhans A, Filion GJ (2017) Position effects influence HIV latency reversal. Nat Struct Mol Biol 24(1):47–54. https://doi.org/10.1038/NSMB.3328
Battivelli E et al (2018) Distinct chromatin functional states correlate with HIV latency reactivation in infected primary CD4+ T cells. Elife. https://doi.org/10.7554/ELIFE.34655
Bouchat S et al (2012) Histone methyltransferase inhibitors induce HIV-1 recovery in resting CD4(+) T cells from HIV-1-infected HAART-treated patients. AIDS 26(12):1473–1482. https://doi.org/10.1097/QAD.0B013E32835535F5
Reuse S et al (2009) Synergistic activation of HIV-1 expression by deacetylase inhibitors and prostratin: implications for treatment of latent infection. PLoS ONE. https://doi.org/10.1371/JOURNAL.PONE.0006093
Tripathy MK, McManamy MEM, Burch BD, Archin NM, Margolis DM (2015) H3K27 demethylation at the proviral promoter sensitizes latent HIV to the effects of vorinostat in ex vivo cultures of resting CD4+ T cells. J Virol 89(16):8392–8405. https://doi.org/10.1128/JVI.00572-15
Abdel-Mohsen M et al (2016) Human Galectin-9 Is a Potent Mediator of HIV Transcription and Reactivation. PLoS Pathog. https://doi.org/10.1371/JOURNAL.PPAT.1005677
Rochat MA, Schlaepfer E, Speck RF (2017) Promising role of toll-like receptor 8 agonist in concert with prostratin for activation of silent HIV. J Virol. https://doi.org/10.1128/JVI.02084-16
Das B et al (2018) Estrogen receptor-1 is a key regulator of HIV-1 latency that imparts gender-specific restrictions on the latent reservoir. Proc Natl Acad Sci USA 115(33):E7795–E7804. https://doi.org/10.1073/PNAS.1803468115
Albert BJ et al (2017) Combinations of isoform-targeted histone deacetylase inhibitors and bryostatin analogues display remarkable potency to activate latent HIV without global T-cell activation. Sci Rep. https://doi.org/10.1038/S41598-017-07814-4
Perez M et al (2010) Bryostatin-1 synergizes with histone deacetylase inhibitors to reactivate HIV-1 from latency. Curr HIV Res 8(6):418–429. https://doi.org/10.2174/157016210793499312
Kim Y, Anderson JL, Lewin SR (2018) Getting the ‘Kill’ into ‘Shock and Kill’: strategies to eliminate latent HIV. Cell Host Microbe 23(1):14–26. https://doi.org/10.1016/j.chom.2017.12.004
Herzig E et al (2019) Attacking latent HIV with convertibleCAR-T cells, a highly adaptable killing platform. Cell 179(4):880-894.e10. https://doi.org/10.1016/j.cell.2019.10.002
Wei D et al (2014) Histone deacetylase inhibitor romidepsin induces HIV expression in CD4 T cells from patients on suppressive antiretroviral therapy at concentrations achieved by clinical dosing. PLoS Pathog. https://doi.org/10.1371/JOURNAL.PPAT.1004071
Albert BJ et al (2017) Combinations of isoform-targeted histone deacetylase inhibitors and bryostatin analogues display remarkable potency to activate latent HIV without global T-cell activation. Sci Rep 7(1):1–12. https://doi.org/10.1038/s41598-017-07814-4
Wightman F et al (2013) Entinostat is a histone deacetylase inhibitor selective for class 1 histone deacetylases and activates HIV production from latently infected primary T cells. AIDS 27(18):2853–2862. https://doi.org/10.1097/QAD.0000000000000067
Contreras X et al (2009) Suberoylanilide hydroxamic acid reactivates HIV from latently infected cells. J Biol Chem 284(11):6782. https://doi.org/10.1074/JBC.M807898200
Matalon S, Rasmussen TA, Dinarello CA (2011) Histone deacetylase inhibitors for purging HIV-1 from the latent reservoir. Mol Med 17(5–6):466–472. https://doi.org/10.2119/molmed.2011.00076
Bullen C, Laird G, Durand C, Siliciano J, Siliciano R (2014) New ex vivo approaches distinguish effective and ineffective single agents for reversing HIV-1 latency in vivo. Nat Med 20(4):425–429. https://doi.org/10.1038/NM.3489
Stoszko M et al (2015) Small Molecule Inhibitors of BAF; a promising family of compounds in HIV-1 latency reversal. EBioMedicine 3:108–121. https://doi.org/10.1016/J.EBIOM.2015.11.047
Gallastegui E et al (2012) Combination of biological screening in a cellular model of viral latency and virtual screening identifies novel compounds that reactivate HIV-1. J Virol 86(7):3795–3808. https://doi.org/10.1128/JVI.05972-11
Abner E et al (2018) A new quinoline BRD4 inhibitor targets a distinct latent HIV-1 reservoir for reactivation from other ‘Shock’ drugs. J Virol. https://doi.org/10.1128/JVI.02056-17
Banerjee C et al (2012) BET bromodomain inhibition as a novel strategy for reactivation of HIV-1. J Leukoc Biol 92(6):1147–1154. https://doi.org/10.1189/JLB.0312165
Zhu J et al (2012) Reactivation of latent HIV-1 by inhibition of BRD4. Cell Rep 2(4):807–816. https://doi.org/10.1016/J.CELREP.2012.09.008
Kauder SE, Bosque A, Lindqvist A, Planelles V, Verdin E (2009) Epigenetic regulation of HIV-1 latency by cytosine methylation. PLoS Pathog. https://doi.org/10.1371/journal.ppat.1000495
Blazkova J et al (2009) CpG methylation controls reactivation of HIV from latency. PLoS Pathog. https://doi.org/10.1371/journal.ppat.1000554
Micheva-Viteva S et al (2011) High-throughput screening uncovers a compound that activates latent HIV-1 and acts cooperatively with a histone deacetylase (HDAC) inhibitor. J Biol Chem 286(24):21083–21091. https://doi.org/10.1074/JBC.M110.195537
Bosque A et al (2017) Benzotriazoles reactivate latent HIV-1 through inactivation of STAT5 SUMOylation. Cell Rep 18(5):1324–1334. https://doi.org/10.1016/J.CELREP.2017.01.022
Kobayashi Y, Gélinas C, Dougherty J (2017) Histone deacetylase inhibitors containing a benzamide functional group and a pyridyl cap are preferentially effective human immunodeficiency virus-1 latency-reversing agents in primary resting CD4+ T cells. J Gen Virol 98(4):799–809. https://doi.org/10.1099/JGV.0.000716
Marsden MD et al (2018) Characterization of designed, synthetically accessible bryostatin analog HIV latency reversing agents. Virology 520:83–93. https://doi.org/10.1016/j.virol.2018.05.006
Vlach J, Pitha P (1992) Activation of human immunodeficiency virus type 1 provirus in T-cells and macrophages is associated with induction of inducer-specific NF-κB binding proteins. Virology 187(1):63–72. https://doi.org/10.1016/0042-6822(92)90295-Z
Folks TM et al (1988) Characterization of a promonocyte clone chronically infected with HIV and inducible by 13-phorbol-12-myristate acetate. J Immunol 140(4):1117–1122
Kulkosky J et al (2001) Prostratin: activation of latent HIV-1 expression suggests a potential inductive adjuvant therapy for HAART. Blood 98(10):3006–3015. https://doi.org/10.1182/BLOOD.V98.10.3006
Jiang G et al (2014) Reactivation of HIV latency by a newly modified Ingenol derivative via protein kinase Cδ-NF-κB signaling. AIDS 28(11):1555–1566. https://doi.org/10.1097/QAD.0000000000000289
Pandeló José D et al (2014) Reactivation of latent HIV-1 by new semi-synthetic ingenol esters. Virology 462–463(1):328–339. https://doi.org/10.1016/J.VIROL.2014.05.033
Alvarez-Carbonell D et al (2017) Toll-like receptor 3 activation selectively reverses HIV latency in microglial cells. Retrovirology. https://doi.org/10.1186/S12977-017-0335-8
Macedo A et al (2018) Dual TLR2 and TLR7 agonists as HIV latency-reversing agents. JCI Insight. https://doi.org/10.1172/JCI.INSIGHT.122673
Thibault S, Imbeault M, Tardif MR, Tremblay MJ (2009) TLR5 stimulation is sufficient to trigger reactivation of latent HIV-1 provirus in T lymphoid cells and activate virus gene expression in central memory CD4+ T cells. Virology 389(1–2):20–25. https://doi.org/10.1016/J.VIROL.2009.04.019
Schlaepfer E, Audigé A, Joller H, Speck RF (2006) TLR7/8 triggering exerts opposing effects in acute versus latent HIV infection. J Immunol 176(5):2888–2895. https://doi.org/10.4049/JIMMUNOL.176.5.2888
Winckelmann AA et al (2013) Administration of a Toll-like receptor 9 agonist decreases the proviral reservoir in virologically suppressed HIV-infected patients. PLoS ONE. https://doi.org/10.1371/JOURNAL.PONE.0062074
López-Huertas M et al (2017) The CCR5-antagonist Maraviroc reverses HIV-1 latency in vitro alone or in combination with the PKC-agonist Bryostatin-1. Sci Rep. https://doi.org/10.1038/S41598-017-02634-Y
Madrid-Elena N et al (2018) Maraviroc is associated with latent HIV-1 reactivation through NF-κB activation in resting CD4 + T cells from HIV-infected individuals on suppressive antiretroviral therapy. J Virol. https://doi.org/10.1128/JVI.01931-17
Tong-Starksen SE, Luciw PA, Peterlin BM (1989) Signaling through T lymphocyte surface proteins, TCR/CD3 and CD28, activates the HIV-1 long terminal repeat. J Immunol 142(2):702–707
Spina CA et al (2013) An in-depth comparison of latent HIV-1 reactivation in multiple cell model systems and resting CD4+ T cells from aviremic patients. PLoS Pathog 9(12):1–15. https://doi.org/10.1371/JOURNAL.PPAT.1003834
Calvanese V, Chavez L, Laurent T, Ding S, Verdin E (2013) Dual-color HIV reporters trace a population of latently infected cells and enable their purification. Virology 446(1–2):283–292. https://doi.org/10.1016/J.VIROL.2013.07.037
Siekevitz M, Josephs SF, Dukovich M, Peffer N, Wong-Staal F, Greene WC (1987) Activation of the HIV-1 LTR by T cell mitogens and the trans-activator protein of HTLV-I. Science 238(4833):1575–1578. https://doi.org/10.1126/SCIENCE.2825351
Doyon G, Sobolewski MD, Huber K, McMahon D, Mellors JW, Sluis-Cremer N (2014) Discovery of a small molecule agonist of phosphatidylinositol 3-kinase p110α that reactivates latent HIV-1. PLoS ONE. https://doi.org/10.1371/JOURNAL.PONE.0084964
Lim H et al (2017) Synergistic reactivation of latent HIV-1 provirus by PKA activator dibutyryl-cAMP in combination with an HDAC inhibitor. Virus Res 227:1–5. https://doi.org/10.1016/J.VIRUSRES.2016.09.015
Xing S et al (2012) Novel structurally related compounds reactivate latent HIV-1 in a bcl-2-transduced primary CD4+ T cell model without inducing global T cell activation. J Antimicrob Chemother 67(2):398–403. https://doi.org/10.1093/JAC/DKR496
Kapewangolo P, Omolo JJ, Fonteh P, Kandawa-Schulz M, Meyer D (2017) Triterpenoids from Ocimum labiatum activates latent HIV-1 expression in vitro: potential for use in adjuvant therapy. Molecules. https://doi.org/10.3390/MOLECULES22101703
Research Toward a Cure Trials – Treatment Action Group. https://www.treatmentactiongroup.org/cure/trials/ (accessed May 23, 2022)
Two-year virologic outcomes of very early art for infants in the impact P1115 STUDY. https://www.croiconference.org/abstract/two-year-virologic-outcomes-of-very-early-art-for-infants-in-the-impaact-p1115-study/ (accessed May 23, 2022).
Treatment with broadly neutralizing antibodies in children with HIV in Botswana. https://www.croiconference.org/abstract/treatment-with-broadly-neutralizing-antibodies-in-children-with-hiv-in-botswana/ (accessed May 23, 2022).
Safety and pharmacokinetics of vrc01ls and 10–1074 among children in Botswana. https://www.croiconference.org/abstract/safety-and-pharmacokinetics-of-vrc01ls-and-10-1074-among-children-in-botswana/ (accessed May 23, 2022).
Román VRG et al (2013) Therapeutic vaccination using cationic liposome-adjuvanted HIV type 1 peptides representing HLA-supertype-restricted subdominant T cell epitopes: safety, immunogenicity, and feasibility in guinea-bissau. AIDS Res Hum Retroviruses 29(11):1504–1512. https://home.liebertpub.com/aid, https://doi.org/10.1089/AID.2013.0076
Suzuki K et al (2008) Closed chromatin architecture is induced by an RNA duplex targeting the HIV-1 promoter region. J Biol Chem 283(34):23353–23363. https://doi.org/10.1074/jbc.M709651200
Ahlenstiel C et al (2015) Novel RNA duplex locks HIV-1 in a latent state via chromatin-mediated transcriptional silencing. Mol Ther- Nucleic Acids 4(10):e261. https://doi.org/10.1038/mtna.2015.31
Gallo RC (2016) Shock and kill with caution. Science (1979) 354(6309):177–178. https://doi.org/10.1126/science.aaf8094
Mousseau G, Valente ST (2016) Didehydro-Cortistatin A: a new player in HIV-therapy? Expert Rev Anti Infect Ther 14(2):145–148. https://doi.org/10.1586/14787210.2016.1122525
Mousseau G et al (2012) An analog of the natural steroidal alkaloid cortistatin A potently suppresses tat-dependent HIV transcription. Cell Host Microbe 12(1):97–108. https://doi.org/10.1016/j.chom.2012.05.016
Mediouni S et al (2019) Didehydro-cortistatin a inhibits HIV-1 by specifically binding to the unstructured basic region of tat. MBio 10(1):1–19. https://doi.org/10.1128/MBIO.02662-18
Moranguinho I, Valente ST (2020) Block-and-lock: new horizons for a cure for HIV-1. Viruses. https://doi.org/10.3390/v12121443 (NLM (Medline))
Li C, Mori L, Valente ST (2021) The block-and-lock strategy for human immunodeficiency virus cure: lessons learned from didehydro-cortistatin A. J Infect Dis 223(1):46–53. https://doi.org/10.1093/infdis/jiaa681
Mori L, Valente ST (2020) Key players in HIV-1 transcriptional regulation: targets for a functional cure. Viruses. https://doi.org/10.3390/v12050529 (MDPI AG)
Kessing CF et al (2017) In vivo suppression of HIV rebound by didehydro-cortistatin A, a ‘block-and-lock’ strategy for HIV-1 treatment. Cell Rep 21(3):600–611. https://doi.org/10.1016/J.CELREP.2017.09.080
Mediouni S et al (2019) The Tat inhibitor didehydro-cortistatin A suppresses SIV replication and reactivation. FASEB J 33(7):8280–8293. https://doi.org/10.1096/fj.201801165R
Mediouni S et al (2015) Didehydro-cortistatin A inhibits HIV-1 tat mediated neuroinflammation and prevents potentiation of cocaine reward in tat transgenic mice. Curr HIV Res 13(1):64–79. https://doi.org/10.2174/1570162x13666150121111548
Mousseau G et al (2019) Resistance to the tat inhibitor didehydro-cortistatin A is mediated by heightened basal HIV-1 transcription. MBio. https://doi.org/10.1128/MBIO.01750-18
Wan Z, Chen X (2014) Triptolide inhibits human immunodeficiency virus type 1 replication by promoting proteasomal degradation of Tat protein. Retrovirology. https://doi.org/10.1186/s12977-014-0088-6
Titov DV et al (2011) XPB, a subunit of TFIIH, is a target of the natural product triptolide. Nat Chem Biol 7(3):182–188. https://doi.org/10.1038/nchembio.522
Hayashi T, Jean M, Huang H, Simpson S, Santoso NG, Zhu J (2017) Screening of an FDA-approved compound library identifies levosimendan as a novel anti-HIV-1 agent that inhibits viral transcription. Antiviral Res 146:76–85. https://doi.org/10.1016/j.antiviral.2017.08.013
Vansant G, Bruggemans A, Janssens J, Debyser Z (2020) Block-and-lock strategies to cure HIV infection. Viruses. https://doi.org/10.3390/v12010084 (MDPI AG)
Stone D, Kiem HP, Jerome KR (2013) Targeted gene disruption to cure HIV. Curr Opin HIV AIDS 8(3):217–223. https://doi.org/10.1097/COH.0b013e32835f736c (NIH Public Access)
Zaboikin M, Zaboikina T, Freter C, Srinivasakumar N (2017) Non-homologous end joining and homology directed DNA repair frequency of double-stranded breaks introduced by genome editing reagents. PLoS ONE. https://doi.org/10.1371/journal.pone.0169931
Wang CX, Cannon PM (2016) The clinical applications of genome editing in HIV. Blood 127(21):2546–2552. https://doi.org/10.1182/blood-2016-01-678144 (American Society of Hematology)
Owens B (2014) Zinc-finger nucleases make the cut in HIV. Nat Rev Drug Discovery 13(5):321–322. https://doi.org/10.1038/nrd4316
Carroll D (2011) Genome engineering with zinc-finger nucleases. Genetics 188(4):773–782. https://doi.org/10.1534/genetics.111.131433
Kwarteng A, Ahuno ST, Kwakye-Nuako G (2017) The therapeutic landscape of HIV-1 via genome editing. AIDS Res Ther. https://doi.org/10.1186/s12981-017-0157-8 (BioMed Central Ltd.)
Wilen CB et al (2011) Engineering HIV-resistant human CD4+ T cells with CXCR4-specific zinc-finger nucleases. PLoS Pathog 7(4):e1002020. https://doi.org/10.1371/journal.ppat.1002020
Yuan J et al (2012) Zinc-finger nuclease editing of human cxcr4 promotes HIV-1 CD4 T cell resistance and enrichment. Mol Ther 20(4):849–859. https://doi.org/10.1038/mt.2011.310
Perez EE et al (2008) Establishment of HIV-1 resistance in CD4+ T cells by genome editing using zinc-finger nucleases. Nat Biotechnol 26(7):808–816. https://doi.org/10.1038/nbt1410
Holt N et al (2010) Human hematopoietic stem/progenitor cells modified by zinc-finger nucleases targeted to CCR5 control HIV-1 in vivo. Nat Biotechnol 28(8):839–847. https://doi.org/10.1038/nbt.1663
Li L et al (2013) Genomic editing of the HIV-1 coreceptor CCR5 in adult hematopoietic stem and progenitor cells using zinc finger nucleases. Mol Ther 21(6):1259–1269. https://doi.org/10.1038/mt.2013.65
Wang J et al (2012) Targeted gene addition to a predetermined site in the human genome using a ZFN-based nicking enzyme. Genome Res 22(7):1316–1326. https://doi.org/10.1101/gr.122879.111
Badia R, Riveira-Muñoz E, Clotet B, Esté JA, Ballana E (2014) Gene editing using a zinc-finger nuclease mimicking the CCR5Δ32 mutation induces resistance to CCR5-using HIV-1. J Antimicrob Chemother 69(7):1755–1759. https://doi.org/10.1093/jac/dku072
Yao Y et al (2012) Generation of CD34+ cells from CCR5-disrupted human embryonic and induced pluripotent stem cells. Hum Gene Ther 23(2):238–242. https://doi.org/10.1089/hum.2011.126
Tebas P et al (2014) Gene editing of CCR5 in autologous CD4 T cells of persons infected with HIV. N Engl J Med 370(10):901–910. https://doi.org/10.1056/nejmoa1300662
Moscou MJ, Bogdanove AJ (2009) A simple cipher governs DNA recognition by TAL effectors. Science (1979) 326(5959):1501. https://doi.org/10.1126/science.1178817
Boch J et al (2009) Breaking the code of DNA binding specificity of TAL-type III effectors. Science (1979) 326(5959):1509–1512. https://doi.org/10.1126/science.1178811
Bogdanove AJ, Schornack S, Lahaye T (2010) TAL effectors: finding plant genes for disease and defense. Curr Opin Plant Biol 13(4):394–401. https://doi.org/10.1016/j.pbi.2010.04.010
Shi B et al (2017) TALEN-mediated knockout of CCR5 confers protection against infection of human immunodeficiency virus. J Acquir Immune Defic Syndr 74(2):229–241. https://doi.org/10.1097/QAI.0000000000001190
Strong CL, Guerra HP, Mathew KR, Roy N, Simpson LR, Schiller MR (2015) Damaging the integrated HIV proviral DNA with TALENs. PLoS ONE 10(5):e0125652. https://doi.org/10.1371/journal.pone.0125652
Kishida T, Ejima A, Mazda O (2016) Specific destruction of HIV proviral p17 gene in T lymphoid cells achieved by the genome editing technology. Front Microbiol. https://doi.org/10.3389/fmicb.2016.01001
Mock U et al (2014) Novel lentiviral vectors with mutated reverse transcriptase for mRNA delivery of TALE nucleases. Sci Rep 4(1):1–8. https://doi.org/10.1038/srep06409
Sternberg SH, Doudna JA (2015) Expanding the biologist’s toolkit with CRISPR-Cas9. Mol Cell 58(4):568–574. https://doi.org/10.1016/j.molcel.2015.02.032 (Cell Press)
Stadtmauer EA et al (2020) CRISPR-engineered T cells in patients with refractory cancer. Science (1979). https://doi.org/10.1126/science.aba7365
Frangoul H (2020) Safety and efficacy of CTX001 in patients with transfusion-dependent β-thalassemia and sickle cell disease: early results from the climb THAL-111 and climb SCD-121 studies of autologous CRISPR-CAS9–modified CD34+ hematopoietic stem and progenitor cells. ASH
Geurts MH et al (2020) CRISPR-based adenine editors correct nonsense mutations in a cystic fibrosis organoid biobank. Cell Stem Cell 26(4):503–510. https://doi.org/10.1016/j.stem.2020.01.019 (e7)
Long C et al (2018) Correction of diverse muscular dystrophy mutations in human engineered heart muscle by single-site genome editing. Sci Adv 4(1):eaap9004. https://doi.org/10.1126/sciadv.aap9004
Ebina H, Misawa N, Kanemura Y, Koyanagi Y (2013) Harnessing the CRISPR/Cas9 system to disrupt latent HIV-1 provirus. Sci Rep 3(1):1–7. https://doi.org/10.1038/srep02510
Wang W, Ye C, Liu J, Zhang D, Kimata JT, Zhou P (2014) CCR5 gene disruption via lentiviral vectors expressing Cas9 and single guided RNA renders cells resistant to HIV-1 infection. PLoS ONE. https://doi.org/10.1371/journal.pone.0115987
Ye L et al (2014) Seamless modification of wild-type induced pluripotent stem cells to the natural CCR5Δ32 mutation confers resistance to HIV infection. Proc Natl Acad Sci USA 111(26):9591–9596. https://doi.org/10.1073/pnas.1407473111
Hou P et al (2015) Genome editing of CXCR4 by CRISPR/cas9 confers cells resistant to HIV-1 infection. Sci Rep. https://doi.org/10.1038/srep15577
Kang HJ, Minder P, Park MA, Mesquitta WT, Torbett BE, Slukvin II (2015) CCR5 disruption in induced pluripotent stem cells using CRISPR/Cas9 provides selective resistance of immune cells to CCR5-tropic HIV-1 virus. Mol Ther-Nucleic Acids 4(12):e268. https://doi.org/10.1038/mtna.2015.42
Chen S, Yu X, Guo D (2018) CRISPR-Cas targeting of host genes as an antiviral strategy. Viruses. https://doi.org/10.3390/v10010040
Qi C et al (2018) Inducing CCR5Δ32/Δ32 homozygotes in the human jurkat CD4+ cell line and primary CD4+ cells by CRISPR-Cas9 genome-editing technology. Mol Ther- Nucleic Acids 12:267–274. https://doi.org/10.1016/j.omtn.2018.05.012
Cho SW, Kim S, Kim JM, Kim JS (2013) Targeted genome engineering in human cells with the Cas9 RNA-guided endonuclease. Nat Biotechnol 31(3):230–232. https://doi.org/10.1038/nbt.2507
Zhang Y et al (2015) CRISPR/gRNA-directed synergistic activation mediator (SAM) induces specific, persistent and robust reactivation of the HIV-1 latent reservoirs. Sci Rep. https://doi.org/10.1038/srep16277
Bialek JK et al (2016) Targeted HIV-1 latency reversal using CRISPR/Cas9-derived transcriptional activator systems. PLoS ONE. https://doi.org/10.1371/journal.pone.0158294
Kim V, Mears B, Powell B, Witwer K (2017) Mutant Cas9-transcriptional activator activates HIV-1 in U1 cells in the presence and absence of LTR-specific guide RNAs. Matters (Zur). https://doi.org/10.19185/matters.201611000027
Limsirichai P, Gaj T, Schaffer DV (2016) CRISPR-mediated activation of latent HIV-1 expression. Mol Ther 24(3):499–507. https://doi.org/10.1038/mt.2015.213
Chemudupati M et al (2019) From APOBEC to ZAP: diverse mechanisms used by cellular restriction factors to inhibit virus infections. Biochem Biophys Acta 1866(3):382–394. https://doi.org/10.1016/j.bbamcr.2018.09.012 (Elsevier B.V.)
Xiao Q, Guo D, Chen S (2019) Application of CRISPR/Cas9-based gene editing in HIV-1/AIDS therapy. Front Cell Infect Microbiol 9(MAR):1–15. https://doi.org/10.3389/fcimb.2019.00069
Bogerd HP, Kornepati AVR, Marshall JB, Kennedy EM, Cullen BR (2015) Specific induction of endogenous viral restriction factors using CRISPR/Cas-derived transcriptional activators. Proc Natl Acad Sci USA 112(52):E7249–E7256. https://doi.org/10.1073/pnas.1516305112
Han Y, Li Q (2016) Application progress of CRISPR/Cas9 genome editing technology in the treatment of HIV-1 infection. Yi chuan = Hereditas / Zhongguo yi chuan xue hui bian ji 38(1):9–16. https://doi.org/10.16288/j.yczz.15-284 (Yi Chuan)
Kimberland ML et al (2018) Strategies for controlling CRISPR/Cas9 off-target effects and biological variations in mammalian genome editing experiments. J Biotechnol 284:91–101. https://doi.org/10.1016/j.jbiotec.2018.08.007 (Elsevier B.V.)
Herrera-Carrillo E, Gao Z, Berkhout B (2019) CRISPR therapy towards an HIV cure. Brief Funct Genomics 00(00):1–8. https://doi.org/10.1093/bfgp/elz021
Wold W, Toth K (2014) Adenovirus vectors for gene therapy, vaccination and cancer gene therapy. Curr Gene Ther 13(6):421–433. https://doi.org/10.2174/1566523213666131125095046
Afkhami S, Yao Y, Xing Z (2016) Methods and clinical development of adenovirus-vectored vaccines against mucosal pathogens. Mol Ther Methods Clin Dev 3:16030. https://doi.org/10.1038/mtm.2016.30 (Elsevier Inc)
Khalili K, White MK, Jacobson JM (2017) Novel AIDS therapies based on gene editing. Cell Mol Life Sci 74(13):2439–2450. https://doi.org/10.1007/s00018-017-2479-z (Birkhauser Verlag AG)
Frange P et al (2016) HIV-1 virological remission lasting more than 12 years after interruption of early antiretroviral therapy in a perinatally infected teenager enrolled in the French ANRS EPF-CO10 paediatric cohort: a case report. The Lancet HIV 3(1):e49–e54. https://doi.org/10.1016/S2352-3018(15)00232-5
Violari A et al (2019) A child with perinatal HIV infection and long-term sustained virological control following antiretroviral treatment cessation. Nat Commun. https://doi.org/10.1038/s41467-019-08311-0
Stöhr W et al (2013) Duration of HIV-1 viral suppression on cessation of antiretroviral therapy in primary infection correlates with time on therapy. PLoS ONE. https://doi.org/10.1371/journal.pone.0078287
Williams JP et al (2014) HIV-1 DNA predicts disease progression and post-treatment virological control. Elife 3:e03821. https://doi.org/10.7554/eLife.03821
Namazi G et al (2018) The control of HIV after antiretroviral medication pause (CHAMP) study: posttreatment controllers identified from 14 clinical studies. J Infect Dis 218(12):1954–1963. https://doi.org/10.1093/infdis/jiy479
Luzuriaga K et al (2014) HIV Type 1 (HIV-1) Proviral reservoirs decay continuously under sustained Virologic control in HIV-1-infected children who received early treatment. J Infect Dis 210(10):1529–1538. https://doi.org/10.1093/infdis/jiu297
Van Zyl GU, Bedison MA, Van Rensburg AJ, Laughton B, Cotton MF, Mellors JW (2015) Early antiretroviral therapy in South African children reduces HIV-1-infected cells and cell-associated HIV-1 RNA in blood mononuclear cells. J Infect Dis 212(1):39–43. https://doi.org/10.1093/infdis/jiu827
Kuhn L et al (2018) Age at antiretroviral therapy initiation and cell-associated HIV-1 DNA levels in HIV-1infected children. PLoS ONE. https://doi.org/10.1371/journal.pone.0195514
Kuhn L et al (2020) Early antiretroviral treatment of infants to attain HIV remission. EClin Med. https://doi.org/10.1016/j.eclinm.2019.100241
Graziosi C, Soudeyns H, Rizzardi GP, Bart PA, Chapuis A and Pantaleo G (1998) Immunopathogenesis of HIV infection. AIDS Res Hum Retroviruses 14(Suppl 2)
Pantaleo G, Fauci AS (1996) Immunopathogenesis of HIV infection. Ann Rev Microbiol 50:825–854. https://doi.org/10.1146/annurev.micro.50.1.825
Letvin NL, Walker BD (2003) Immunopathogenesis and immunotherapy in AIDS virus infections. Nat Med 9(7):861–866
Arduino RC et al (2004) CD4 cell response to 3 doses of subcutaneous interleukin 2: Meta-analysis of 3 vanguard studies. Clin Infect Dis 39(1):115–122. https://doi.org/10.1086/421775
Lévy Y et al (2006) Sustained control of viremia following therapeutic immunization in chronically HIV-1-infected individuals. AIDS 20(3):405–413. https://doi.org/10.1097/01.aids.0000206504.09159.d3
Bart PA, Pantaleo G (2006) Immune-based interventions in HIV infection: doing the right studies, getting the right answers. AIDS 20(4):617–618. https://doi.org/10.1097/01.aids.0000199827.79983.66
Autran B, Carcelain G, Combadiere B, Debre P (2004) Therapeutic vaccines for chronic infections. Science 305(5681):205–208. https://doi.org/10.1126/science.1100600
Avettand-Fenoel V et al (2008) HIV-DNA in rectal cells is well correlated with HIV-DNA in blood in different groups of patients, including long-term non-progressors. AIDS 22(14):1880–1882. https://doi.org/10.1097/QAD.0b013e32830fbdbc
Barouch DH, Deeks SG (2014) Immunologic strategies for HIV-1 remission and eradication. Science 345(6193):169–174. https://doi.org/10.1126/science.1255512 (American Association for the Advancement of Science)
Deng K et al (2015) Broad CTL response is required to clear latent HIV-1 due to dominance of escape mutations. Nature 517(7534):381–385. https://doi.org/10.1038/nature14053
Betts MR et al (2006) HIV nonprogressors preferentially maintain highly functional HIV-specific CD8+ T cells. Blood 107(12):4781–4789. https://doi.org/10.1182/blood-2005-12-4818
Kannanganat S, Ibegbu C, Chennareddi L, Robinson HL, Amara RR (2007) Multiple-cytokine-producing antiviral CD4 T cells are functionally superior to single-cytokine-producing cells. J Virol 81(16):8468–8476. https://doi.org/10.1128/jvi.00228-07
Shan L et al (2012) Stimulation of HIV-1-specific cytolytic T lymphocytes facilitates elimination of latent viral reservoir after virus reactivation. Immunity 36(3):491–501. https://doi.org/10.1016/j.immuni.2012.01.014
Migueles SA et al (2008) Lytic granule loading of CD8+ T cells is required for HIV-infected cell elimination associated with immune control. Immunity 29(6):1009. https://doi.org/10.1016/J.IMMUNI.2008.10.010
Walker BD et al (1988) HIV-specific cytotoxic T lymphocytes in seropositive individuals. Nature 328(6128):345–348. https://doi.org/10.1038/328345a0
Koup RA et al (1994) Temporal association of cellular immune responses with the initial control of viremia in primary human immunodeficiency virus type 1 syndrome. J Virol 68(7):4650–4655. https://doi.org/10.1128/jvi.68.7.4650-4655.1994
Gandhi RT, Walker BD (2002) Immunologic control of HIV-1. Ann Rev Med 53:149–172. https://doi.org/10.1146/annurev.med.53.082901.104011
Hersperger AR, Migueles SA, Betts MR, Connors M (2011) Qualitative features of the HIV-specific CD8+ T-cell response associated with immunologic control. Curr Opin HIV AIDS 6(3):169–173. https://doi.org/10.1097/COH.0b013e3283454c39
Fenwick C et al (2019) T-cell exhaustion in HIV infection. Immunol Rev 292(1):149–163. https://doi.org/10.1111/imr.12823 (Blackwell Publishing Ltd)
Mylvaganam G, Yanez AG, Maus M, Walker BD (2019) Toward T cell-mediated control or elimination of HIV reservoirs: lessons from cancer immunology. Front Immunol 10:2109. https://doi.org/10.3389/fimmu.2019.02109
Dalod M et al (1999) Broad, intense anti-Human Immunodeficiency Virus (HIV) ex vivo CD8+ responses in HIV Type 1-infected patients: comparison with anti-Epstein-Barr virus responses and changes during antiretroviral therapy. J Virol 73(9):7108–7116. https://doi.org/10.1128/jvi.73.9.7108-7116.1999
Addo MM et al (2003) Comprehensive Epitope Analysis of Human Immunodeficiency Virus Type 1 (HIV-1)-specific T-cell responses directed against the entire expressed HIV-1 genome demonstrate broadly directed responses, but no correlation to viral load. J Virol 77(3):2081–2092. https://doi.org/10.1128/jvi.77.3.2081-2092.2003
Kinloch-De Loes S et al (2005) Impact of therapeutic immunization on HIV-1 viremia after discontinuation of antiretroviral therapy initiated during acute infection. J Infect Dis 192(4):607–617. https://doi.org/10.1086/432002
Autran B et al (2008) Greater viral rebound and reduced time to resume antiretroviral therapy after therapeutic immunization with the ALVAC-HIV vaccine (vCP1452). AIDS 22(11):1313–1322. https://doi.org/10.1097/QAD.0b013e3282fdce94
García F et al (2013) A dendritic cell-based vaccine elicits T cell responses associated with control of HIV-1 replication. Sci Transl Med. https://doi.org/10.1126/scitranslmed.3004682
Pollard RB et al (2014) Safety and efficacy of the peptide-based therapeutic vaccine for HIV-1, Vacc-4x: a phase 2 randomised, double-blind, placebo-controlled trial. Lancet Infect Dis 14(4):291–300. https://doi.org/10.1016/S1473-3099(13)70343-8
Burton DR, Poignard P, Stanfield RL, Wilson IA (2012) Broadly neutralizing antibodies present new prospects to counter highly antigenically diverse viruses. Science 337(6091):183–186. https://doi.org/10.1126/science.1225416 (American Association for the Advancement of Science)
Barouch DH et al (2013) Therapeutic efficacy of potent neutralizing HIV-1-specific monoclonal antibodies in SHIV-infected rhesus monkeys. Nature 503(7475):224–228. https://doi.org/10.1038/nature12744
Halper-Stromberg A et al (2014) Broadly neutralizing antibodies and viral inducers decrease rebound from HIV-1 latent reservoirs in humanized mice. Cell 158(5):989–999. https://doi.org/10.1016/j.cell.2014.07.043
Klein F et al (2012) HIV therapy by a combination of broadly neutralizing antibodies in humanized mice. Nature 492(7427):118–122. https://doi.org/10.1038/nature11604
Shingai M et al (2013) Antibody-mediated immunotherapy of macaques chronically infected with SHIV suppresses viraemia. Nature 503(7475):277–280. https://doi.org/10.1038/nature12746
Rasmussen TA et al (2014) Panobinostat, a histone deacetylase inhibitor, for latent virus reactivation in HIV-infected patients on suppressive antiretroviral therapy: a phase 1/2, single group, clinical trial. The Lancet HIV 1(1):e13–e21. https://doi.org/10.1016/S2352-3018(14)70014-1
Walsh SR, Seaman MS (2021) Broadly Neutralizing Antibodies for HIV-1 Prevention. Front Immunol. https://doi.org/10.3389/FIMMU.2021.712122
Gama L, Koup RA (2018) New-generation high-potency and designer antibodies: role in HIV-1 treatment. Ann Rev Med 69:409–419. https://doi.org/10.1146/annurev-med-061016-041032 (Annual Reviews Inc.)
Thali M et al (1993) Characterization of conserved human immunodeficiency virus type 1 gp120 neutralization epitopes exposed upon gp120-CD4 binding. J Virol 67(7):3978–3988. https://doi.org/10.1128/jvi.67.7.3978-3988.1993
Zwick MB et al (2001) Broadly neutralizing antibodies targeted to the membrane-proximal external region of human immunodeficiency virus type 1 glycoprotein gp41. J Virol 75(22):10892–10905. https://doi.org/10.1128/jvi.75.22.10892-10905.2001
West AP, Scharf L, Horwitz J, Klein F, Nussenzweig MC, Bjorkman PJ (2013) Computational analysis of anti-HIV-1 antibody neutralization panel data to identify potential functional epitope residues. Proc Natl Acad Sci USA 110(26):10598–10603. https://doi.org/10.1073/pnas.1309215110
Liu Y, Cao W, Sun M, Li T (2020) Broadly neutralizing antibodies for HIV-1: efficacies, challenges and opportunities. Emerg Microb Infect 9(1):194–206. https://doi.org/10.1080/22221751.2020.1713707 (Taylor and Francis Ltd)
Wang Q, Zhang L (2020) Broadly neutralizing antibodies and vaccine design against HIV-1 infection. Front Med 14(1):30–42. https://doi.org/10.1007/s11684-019-0721-9 (Higher Education Press)
Gautam R et al (2016) A single injection of anti-HIV-1 antibodies protects against repeated SHIV challenges. Nature 533(7601):105–109. https://doi.org/10.1038/NATURE17677
Pegu A et al (2014) Neutralizing antibodies to HIV-1 envelope protect more effectively in vivo than those to the CD4 receptor. Sci Transl Med. https://doi.org/10.1126/SCITRANSLMED.3008992
Julg B et al (2017) Protective efficacy of broadly neutralizing antibodies with incomplete neutralization activity against simian-human immunodeficiency virus in rhesus monkeys. J Virol. https://doi.org/10.1128/JVI.01187-17
Julg B et al (2017) Broadly neutralizing antibodies targeting the HIV-1 envelope V2 apex confer protection against a clade C SHIV challenge. Sci Transl Med. https://doi.org/10.1126/SCITRANSLMED.AAL1321
Caskey M, Klein F, Nussenzweig MC (2019) Broadly neutralizing anti-HIV-1 monoclonal antibodies in the clinic. Nat Med 25(4):547–553. https://doi.org/10.1038/s41591-019-0412-8
Caskey M et al (2015) Viraemia suppressed in HIV-1-infected humans by broadly neutralizing antibody 3BNC117. Nature 522(7557):487–491. https://doi.org/10.1038/nature14411
Lynch RM et al (2015) Virologic effects of broadly neutralizing antibody VRC01 administration during chronic HIV-1 infection. Sci Transl Med. https://doi.org/10.1126/scitranslmed.aad5752
Scheid JF et al (2016) HIV-1 antibody 3BNC117 suppresses viral rebound in humans during treatment interruption. Nature 535(7613):556–560. https://doi.org/10.1038/nature18929
Mendoza P et al (2018) Combination therapy with anti-HIV-1 antibodies maintains viral suppression. Nature 561(7724):479–484. https://doi.org/10.1038/s41586-018-0531-2
Hsu DC, Mellors JW, Vasan S (2021) Can broadly neutralizing HIV-1 antibodies help achieve an ART-free remission? Front Immunol. https://doi.org/10.3389/FIMMU.2021.710044/FULL
Dana H et al (2021) CAR-T cells: early successes in blood cancer and challenges in solid tumors. Acta Pharmaceutica Sinica B 11(5):1129–1147. https://doi.org/10.1016/J.APSB.2020.10.020
Almåsbak H, Aarvak T, Vemuri MC (2016) CAR T cell therapy: a game changer in cancer treatment. J Immunol Res. https://doi.org/10.1155/2016/5474602
Miliotou A, Papadopoulou L (2018) CAR T-cell therapy: a new era in cancer immunotherapy. Curr Pharm Biotechnol 19(1):5–18. https://doi.org/10.2174/1389201019666180418095526
Mu W, Carrillo MA, Kitchen SG (2020) Engineering CAR T cells to target the HIV reservoir. Front Cell Infect Microbiol. https://doi.org/10.3389/FCIMB.2020.00410
Guedan S, Calderon H, Posey A, Maus M (2018) Engineering and design of chimeric antigen receptors. Mol Ther Methods Clin Dev 12:145–156. https://doi.org/10.1016/J.OMTM.2018.12.009
Pule M, Finney H, Lawson A (2003) Artificial T-cell receptors. Cytotherapy 5(3):211–226. https://doi.org/10.1080/14653240310001488
Chmielewski M, Abken H (2015) TRUCKs: the fourth generation of CARs. Expert Opin Biol Ther 15(8):1145–1154. https://doi.org/10.1517/14712598.2015.1046430
Chmielewski M, Hombach AA, Abken H (2014) Of CARs and TRUCKs: chimeric antigen receptor (CAR) T cells engineered with an inducible cytokine to modulate the tumor stroma. Immunol Rev 257(1):83–90. https://doi.org/10.1111/IMR.12125
Hurton LV et al (2016) Tethered IL-15 augments antitumor activity and promotes a stem-cell memory subset in tumor-specific T cells. Proc Natl Acad Sci 113(48):E7788–E7797. https://doi.org/10.1073/PNAS.1610544113
Maher J, Brentjens R, Gunset G, Rivière I, Sadelain M (2002) Human T-lymphocyte cytotoxicity and proliferation directed by a single chimeric TCRzeta /CD28 receptor. Nat Biotechnol 20(1):70–75. https://doi.org/10.1038/NBT0102-70
Savoldo B et al (2011) CD28 costimulation improves expansion and persistence of chimeric antigen receptor–modified T cells in lymphoma patients. J Clin Investig 121(5):1822–1826. https://doi.org/10.1172/JCI46110
Sarkar RR, Gloude NJ, Schiff D, Murphy JD (2019) Cost-effectiveness of chimeric antigen receptor T-cell therapy in pediatric relapsed/refractory B-cell acute lymphoblastic leukemia. J Natl Cancer Inst 111(7):719–726. https://doi.org/10.1093/JNCI/DJY193
Kharsany ABM, Karim QA (2016) HIV infection and AIDS in sub-Saharan Africa: current status, challenges and opportunities. Open AIDS J 10(1):34–48. https://doi.org/10.2174/1874613601610010034
Acknowledgements
The authors acknowledge the support of the Massive Action Team of the HIV Cure Research Infrastructure Study (H-CRIS).
Funding
This paper is part of the EDCTP2 program supported by the European Union (Grant Number TMA2017SF-1955, H-CRIS) to GBK. The funder had no role in this publication.
Author information
Authors and Affiliations
Contributions
Writing—original draft preparation, CZ-YA; writing—review and editing, CZ-YA, GBK, EYB, and HL; supervision, GBK; funding acquisition, GBK. All the authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest. The funders had no role in the writing of the manuscript.
Institutional review board statement
Not applicable.
Informed consent
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Abana, C.ZY., Lamptey, H., Bonney, E.Y. et al. HIV cure strategies: which ones are appropriate for Africa?. Cell. Mol. Life Sci. 79, 400 (2022). https://doi.org/10.1007/s00018-022-04421-z
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00018-022-04421-z