Abstract
Background
Anti-inflammatory n-3 fatty acids (FA) like docosahexaenoic acid (DHA) opposed to the pro-inflammatory n-6 FA arachidonic acid (AA) might modulate lipid rafts within the cell membrane by differential incorporation. In inflammation, monocyte adhesion to endothelial cells is a crucial step mediated by intracellular calcium changes. We investigated whether lipid rafts mediate FA-induced modulation of adhesion and intracellular calcium.
Methods
In isolated human monocytes and monocytic U937 cells we measured adhesion to human umbilical vein endothelial cells (HUVEC) using a parallel flow chamber and a static assay, adhesion molecules by FACScan, and intracellular calcium by fluorescence. Monocyte lipid rafts were isolated by ultracentrifugation and submitted to gas chromatography for FA analysis.
Results
Pre-incubation with AA or DHA resulted in a predominant incorporation of the respective FA into raft compared to non-raft fraction. DHA as compared to AA significantly reduced monocyte adhesion and calcium release after stimulation with TNF-α while expression of adhesion molecules remained unchanged. Pre-treatment with a calcium chelator abolished the effect of FA on calcium and adhesion. Disruption of lipid rafts prevented FA-induced modulations.
Conclusion
Incorporation of FA into lipid rafts seem to be crucial for modulation of adhesion under inflammatory conditions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Enhanced transmigration of circulating blood monocytes across the vascular endothelium is considered to be an important contributor to the pathogenesis of chronic and acute inflammatory diseases. Here, atherosclerosis [1] as well as sepsis and multi-organ failure [2] are the most prominent examples [3]. This process of vascular inflammation requires adhesion of leukocytes to and migration through endothelium. Interaction between several adhesion molecules were shown to be involved in the transendothelial migration of monocytes, as well as enhanced calcium signalling, and other pro-inflammatory mediators like tumor-necrosis factor (TNF)-α [4–6]. It has also been demonstrated that TNF-α mediated nuclear factor-κB activation is dependent on lipid rafts [7].
Lipid rafts are sphingolipid- and cholesterol-rich signalling platforms within the plasma membrane. These membrane microdomains are characterized by being detergent resistant and liquid ordered [8]. They can organize domains on the inner leaflet, coupling events between the outer leaflet to others in the inner leaflet and the cell cytoplasm. Membrane microdomains influence membrane functions by concentrating signalling molecules in these particular regions of the surface for facilitated interaction. Otherwise, by excluding molecules from the rafts their interaction and subsequent signalling may be prevented [9, 10]. It has also been demonstrated in U937-monocytes [11] and in human monocytes [12] that rafts are important for inflammatory signal transduction [13, 14].
In the context of lipids and inflammation, eicosanoids represent a focus of interest due to their strong pro-inflammatory and anti-inflammatory properties [15, 16]. Different groups reported a major influence of nutrition including n-3 fatty acids (FA) on morbidity and mortality of intensive care patients as well as patients suffering from coronary artery disease [17, 18]. The family of n-6 FA, including arachidonic acid (AA), are the predominant polyunsaturated fatty acids (PUFA) in common western diet. The n-3 FA, including docosahexaenoic acid (DHA), are the predominant fat in cold-water fish and seal meat. N-3 vis-à-vis n-6 FA incorporation into membrane (phospho)-lipid pools was suggested to influence lipid-related intracellular signaling events [19].
It has been demonstrated that FA are capable of modulating lipid raft composition and raft related signalling [20]. Due to the ability of PUFA to inhibit palmitoylation, n-3 vs. n-6 FA might possess different abilities to target signalling proteins into rafts [21, 22]. Modulation of membrane composition due to incorporation of different FA represents a means by which PUFA could differentially influence rafts and subsequently raft-dependent signal transduction. Reports demonstrate that in particular anti-inflammatory capacity of n-3 FA might be in part due to raft modulation, namely due to their potential to alter both the composition of signalling molecules and the lipid composition within the rafts [22, 23].
The aim of this study was firstly, to investigate whether n-3 vs. n-6 FA differentially influence lipid raft fatty acid composition in U937-monocytes as well as in freshly isolated human blood monocytes. Secondly, we investigated whether a differential fatty acid composition of lipid rafts influenced intracellular calcium and monocyte adhesion to human umbilical venous endothelial cells (HUVEC) after TNF-α stimulation.
Methods
Materials
Chemicals of highest purity were obtained from Merck (Darmstadt, Gernany). AA and DHA were obtained from Sigma Chemical (Deisenhofen, Germany). Tissue culture plastic supplies were purchased from Becton–Dickinson (Heidelberg, Germany). Cell culture reagents, buffer, and media were from PAN (Aidenbach, Germany). Lyn and Fyn antibody for Western Blots were obtained from BD Biosciences (Pharmingen, Germany). Secondary antibodies were purchased from Santa Cruz (Heidelberg, Germany). Blot analysis was performed with BioDoc II Biometra (Göttingen, Germany) with Scan Pack 3.0 software.
Experimental protocol
U937-monocytes were cultured, human blood monocytes were isolated and HUVEC were grown to confluence. Then, the culture medium was exchanged, and free fatty acids (AA and DHA) dissolved in ethanol (final volume ≤1 %, vol/vol), or vehicle were admixed to the culture medium at a final concentration of 10 µmol/l in all experiments with U937 and fresh monocytes. The concentration was derived from our previous experiments and due to the fact that this concentration of free AA and DHA is similar to that detected in human plasma [24–26]. Controls were sham incubated with solvent only.
TNF-α was added after FA pre-incubation to U937 and to fresh monocytes in all experiments at a concentration of 1 ng/ml. In static adhesion experiments with U937, additionally a second concentration of 10 ng/ml was applied. In dynamic flow adhesion investigations using freshly isolated monocytes, HUVEC were incubated with 10 ng/ml TNF-α for 20 h prior to the beginning of the experiments.
FA pre-incubation times were 24 h in all experiments with U937, and 2 h in all experiments with freshly isolated monocytes. Due to the nature of the tumor cell line, pre-incubation and stimulation time as well as the kind of adhesion assay varied between U937 and freshly isolated human blood monocytes.
After FA pre-incubation, TNF-α stimulation followed for 4 h in experiments with U937 and for 2 h in experiments with freshly isolated monocytes. Only in experiments for calcium measurements in U937, TNF-α stimulation time was 1 h.
In experiments with calcium-chelator [10 µmol/l 1,2-bis(o-aminophenoxy)ethane-N,N,N′,N′-tetraacetic acid tetra acetoxymethyl-ester(BAPTA); (Calbiochem; Bad Soden, Germany)] a co-incubation with BAPTA and FA was performed. In experiments with disruption of lipid rafts, methyl-ß-cyclodextrin (MCD, Sigma, Dreisenhofen, Germany) was used at a concentration of 10 mmol/l for 10 min at 37 °C after FA incubation and prior to TNF-α stimulation.
Cell culture
Endothelial cells were obtained from human umbilical veins according to the method described by Jaffe et al. [27]. Cells of the passages 1–4 were used in all experiments.
The monocyte tumor cell line U937 was cultured in RPMI culture medium with supplementation of 5 % fetal calf serum (FCS), and 1 % penicillin/streptomycin (both PAA, Linz, Austria). Cells were splitted 1–4 every 48 h.
Fluorescence imaging: calcium measurement using a microplate reader
Intracellular Ca2+ concentrations ([Ca2+]i) were determined using the fluorescent Ca2+ indicator Fluo-3-AM (Molecular Probes, Leiden, Netherlands). Monocytes were labelled with 5 µmol/l Fluo-3-AM. After an incubation period of 90 min, extracellular Fluo-3-AM was removed and the medium was exchanged using PBS buffer. Changes of [Ca2+]i in monocytes were analysed by a “GENios Plus multi-detection microplate reader with enhanced fluorescence” (Tecan Inc., Research Triangle Park, NC) after 1 h stimulation with 1 ng/ml TNF-α. The excitation wavelength was set at 485 nm and emitted light was detected at 535 nm. Fluorescence background determination and calibration were performed according to methods described by Fowler [28] and Baskin [29].
Static adhesion assay
The static interaction assay between monocytes and endothelial cells has been described previously [30]. Briefly, after FA pre-incubation, U937-monocytes were labelled with 3 μg/ml 2,-7-bis2carboxyethyl5and6carbox fluorescein, acetoxymethylester (BCECF-AM) (Molecular Probes, Leiden, Netherlands) at 37 °C for 30 min, washed, and resuspended in medium. 250 000 BCECF-AM-loaded U937-monocytes per well were co-incubated with confluent HUVEC for 4 h in a 24-well plate after stimulation with 1 or 10 ng/ml TNF-α. After aspiration of the medium, three washing steps with PBS followed to remove non-adherent cells. Adherent cells were lysed using 1 mol/l NaOH for 45 min. Fluorescence was measured by multi-detection microplate reader including fluorescence background determination and calibration as detailed above. All measurements were corrected by subtraction of the fluorescence of non-treated cells as background.
Isolation of human blood monocytes
Human monocytes were isolated from platelet pheresis residues by Ficoll-Hypaque density gradient centrifugation, followed by counterflow centrifugation elutriation using a Beckman JE-5.0 rotor as described [31]. Monocyte purity (88–98 %) was confirmed by light scatter [fluorescence-activated cell sorter (FACS) scan; Becton–Dickinson]. Cell viability was above 96 % throughout the study measured by trypan blue test.
Flow-chamber adhesion assay
Adhesion of freshly isolated human blood monocytes to HUVEC was determined as described previously [26] using a parallel plate flow chamber according to Lawrence and Springer [32]. Confluent endothelial monolayers were pre-incubated with fatty acids and TNF-α according to the experimental protocol. HUVEC were pre-incubated with 10 ng/ml TNF-α for 20 h [26]. A suspension of 2 × 106 monocytes was perfused through the chamber at a constant wall shear stress of 1.0 dyn/cm2 (syringe pump sp100i, WPI; Sarasota, FL). Interactions were visualized using a phase contrast video microscope (IMT-2, Olympus Optical, Hamburg, Germany, with a KP-C551 CCD camera, Hitachi, Rodgau, Germany) and videotaped (JVC HR-S7000, JVC; Friedberg, Germany) over the entire time period of monocyte perfusion. Rolling in the parallel plate flow chamber was measured in one high-power field for each experiment. “Rolling” was expressed as the number of rolling cells per high power field (20× objective) during a 10-min observation period. Monocytes were considered to be adherent after 30 s of stable contact with the monolayer. Adhesion was determined after 10 min of perfusion by analysis of five random high magnification fields (20×) from videotape. Results are expressed as adherent cells per high magnification field.
FACScan immunofluorescence analysis of adhesion molecules
U937-monocytes and freshly isolated human monocytes were subjected to immunofluorescence staining by incubation of samples with appropriately diluted PE-labeled anti-CD11b (Mac-2)mAb or anti-CD18 mAb or anti-CD49d mAb (very late antigen (VLA)-4) mAb (R&D Systems, Wiesbaden, Germany) for 30 min at ambient temperature followed by flow cytometric analysis using a FACScan flow cytometer (BD Biosciences, San Jose, CA, USA). Data analysis was performed using the CellQuest software package (BD Biosciences).
Detergent-resistant membrane (lipid raft) partitioning
Lipid raft isolation was performed by a modified method according to Waheed and Jones [33]. Briefly, cell pellets from U937-monocytes and from freshly isolated human monocytes were resuspended and incubated for 20 min at 4 °C in ice cold buffer containing 0.5 % Triton X-100 to give a detergent to protein ratio of 5:1. The samples were adjusted to 35 % (v/v) OptiPrep (Sigma; Dreisenhofen, Germany) and the lipid raft fraction separated by centrifugation on a 5/30/35 % OptiPrep sucrose-gradient. After ultra-centrifugation, nine fractions were collected. Determination of two raft-marker (Scr-family protein tyrosine kinases Lyn and Fyn) by Western Blot was performed demonstrating detection of Lyn and Fyn within fraction one and two. Thereby we defined fractions one and two as raft fractions and fractions three to nine as non-raft fractions.
Membrane fatty acid analysis
Membrane fatty acid analysis of U937-monocytes and freshly isolated human monocytes of pooled raft fractions one and two and the pooled non-raft fractions three to nine was done by gas chromatography as described [34] after lipid extraction according to Bligh and Dyer [35].
Statistics
Data are given as mean ± SEM. One-way analysis of variance and Student–Newman–Keuls post hoc test was performed to test for differences between different experimental groups in experiments with U937-monocytes. Since freshly isolated human monocytes also underwent experiments where fatty acids were examined without TNF-α, two-way analysis of variance and Student–Newman–Keuls post hoc test was performed to test for differences between different experimental groups. Data generated from rolling experiments after treatment with MCD were not distributed normally. Therefore, log-transformation was applied. Probability (p) values <0.05 were considered to indicate statistical significance. Analysis was carried out using SigmaStat® version 3.5.
Results
Fatty acid composition of raft and non-raft membranes of U937-monocytes
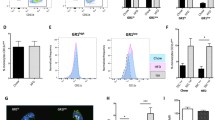
U937-monocytes were pre-incubated with FA and stimulated with 1 ng/ml TNF-α followed by separation of raft and non-raft membrane fractions as described in the method section. A western blot of the monocyte raft marker Lyn and Fyn is depicted. Lyn and Fyn are situated in the first and second lane (raft fraction). Both proteins cannot be detected in the non-raft fractions and but in the “pellet” fractions to demonstrate a proper separation of raft and non-raft parts of the membrane (Fig. 1a, b). Subsequently, raft and non-raft fractions were submitted to FA analysis as described.
Impact of AA and DHA-incubation on U937-monocyte and freshly isolated human blood monocyte membrane FA composition: analysis of raft and non-raft fraction. Membrane fatty acid analysis of U937- and freshly isolated human blood monocytes raft fraction and non-raft fraction was performed after lipid raft separation. A western blot depicting monocyte raft marker Lyn (a) and Fyn (b) in the first two of nine fractions is shown. Fractions one and two were analysed as raft fraction; fractions three to nine as non-raft fraction. In U937-monocytes, arachidonic acid (AA) pre-incubation and TNF-α stimulation resulted in a non-significant rise of AA in non-raft fraction (c) whereas a highly significant rise was noted in raft fraction (d, p < 0.001). In U937-monocytes, docosahexaenoic acid (DHA) pre-incubation and TNF-α stimulation induced a highly significant rise in both non-raft (e) and raft (f) fraction (p < 0.001) though with a marked enhanced incorporation into raft fraction. In freshly isolated human blood monocytes, AA pre-incubation and TNF-α stimulation resulted in a significant rise of AA in non-raft fraction (g) though in raft fraction a significant rise of AA compared to TNF-α and compared to DHA was detected which was more pronounced (h, p < 0.05). In freshly isolated human blood monocytes, DHA pre-incubation and TNF-α stimulation induced a highly significant rise in both non-raft (i) and raft (j) fraction (p < 0.001) though with a marked enhanced incorporation into raft fraction. Two-way analysis of variance revealed no significant impact of TNF-α on the incorporation of AA into raft or non-raft fractions as compared to control conditions. While DHA incorporation into the non-raft fraction was not influenced by TNF-α, DHA was significantly increased under inflammatory conditions in the raft fraction (j; §, p < 0.05). Data are given as mean ± SEM
Incubation with AA and TNF-α resulted in a 1.4-fold but not significant increase of AA in total fatty acids (TFA) of the non-raft membrane fraction to 13.11 ± 2.78 % compared to sole TNF-α stimulation with 9.12 ± 2.74 % (n = 3–5; Fig. 1c). In contrast, incubation with AA and TNF-α induced a highly significant 2.1-fold rise in AA in raft TFA to 7.15 ± 0.64 % compared to stimulation with TNF-α (3.39 ± 0.33 %, p < 0.001; n = 4–7; Fig. 1d). In line with these findings, stimulation with DHA and TNF-α lead to a 3.0-fold rise in DHA to 8.34 ± 0.41 % compared to TNF-α stimulated cells with 2.75 ± 0.04 % of TFA in the non-raft membrane fraction (p < 0.001; n = 3–7; Fig. 1e). Though in raft membrane fraction, incubation with DHA and TNF-α enhanced DHA of TFA even stronger (6.8-fold) to 7.14 ± 0.72 % compared to TNF-α with 1.05 ± 0.18 % (p < 0.001; n = 3–7; Fig. 1f).
Fatty acid composition of raft and non-raft membranes of freshly isolated human blood monocytes
Freshly isolated human blood monocytes were pre-incubated with FA and stimulated with 1 ng/ml TNF-α followed by separation of rafts and non-raft membrane and subsequently submitted to FA analysis as described.
Incubation of AA without TNF-α lead to a significant rise of AA in total fatty acids (TFA) to 15.51 ± 1.023 % compared to control and DHA in the non-raft fraction (p < 0.05, Fig. 1g; n = 3–7). Incubation with AA and TNF-α resulted also in a 1.4-fold increase of AA in TFA of the non-raft membrane fraction to 15.16 ± 1.03 % compared to sole TNF-α stimulation with 10.78 ± 1.03 % (p < 0.05; Fig. 1g; n = 3–7).
In raft-fraction, incubation of AA without TNF-α lead to a not significant rise of AA in TFA to 4.77 ± 0.75 % compared to control with 2.72 ± 0.58 %. Notably, incubation with AA and TNF-α induced a significant 1.9-fold rise in AA in raft TFA to 5.66 ± 0.75 % compared to control cells stimulated with TNF-α (3.00 ± 0.58 %, p < 0.05; Fig. 1h; n = 3–6).
In line with these findings, in non-raft fraction, incubation of DHA without TNF-α lead to a highly significant rise of DHA in TFA to 5.82 ± 0.40 % compared to control with 1.66 ± 0.40 % (p < 0.001).
Pre-incubation with DHA and subsequent stimulation by TNF-α lead to a 4.5-fold rise in DHA to 6.44 ± 0.40 % compared to TNF-α stimulated cells with 1.42 ± 0.40 % of TFA in the non-raft membrane fraction (p < 0.001; Fig. 1i; n = 3).
Though in raft membrane fraction, incubation of DHA without TNF-α lead to a significant rise of DHA in TFA to 2.30 ± 0.34 % compared to control with 0.51 ± 0.29 % (p < 0.05).
Incubation with DHA + TNF-α enhanced DHA of TFA 5.7-fold to 5.64 ± 0.34 % compared to TNF-α with 0.99 ± 0.29 % (p < 0.001; Fig. 1j; n = 3–4).
Two-way analysis of variance revealed no significant impact of TNF-α on the incorporation of AA into raft or non-raft fractions as compared to control conditions. While DHA incorporation into the non-raft fraction was not influenced by TNF-α, DHA was significantly increased under inflammatory conditions in the raft fraction (p < 0.05; Fig. 1j).
Impact of fatty acids on static adhesion of U937-monocytes after TNF-α stimulation
Adhesion of unstimulated U937-monocytes to HUVEC was determined as 492.73 ± 40.46 fluorescence units (FU) which was highly significantly different from all TNF-α treated groups (p < 0.001, Fig. 2a). Stimulation of U937-monocytes with 10 ng/ml TNF-α for 4 h increased adhesion to HUVEC (3335.77 ± 111.84 FU). Pre-incubation with AA + TNF-α resulted in a comparable adhesion (3212.02 ± 288.73 FU). However, DHA + TNF-α pre-treatment induced a significant reduction of fluorescence (2900.48 ± 142.65 FU) compared to AA + TNF-α and compared to TNF-α (p < 0.05, n = 7–11; Fig. 2a).
Effect of FA pre-incubation on U937- and on human monocytes adhesion and rolling. U937-monocytes were pre-incubated with arachidonic acid (AA), docosahexaenoic acid (DHA), or vehicle followed by stimulation with TNF-α. Adhesion of U937 to HUVEC was measured using a fluorescence-based static assay. After stimulation with 10 ng/ml TNF-α adhesion was significantly different between the FA groups (a, p < 0.05). After stimulation with 1 ng/ml TNF-α FA induced a highly significant modulation of adhesion (b, p < 0.001). Freshly isolated human monocytes were pre-incubated with arachidonic acid (AA), docosahexaenoic acid (DHA), or vehicle followed by stimulation with 1 ng/ml TNF-α. Rolling and adhesion to HUVEC under laminar flow conditions was measured using a parallel plate flow chamber. Pre-incubation with DHA induced a highly significant reduction compared to TNF-α and a significant down-regulation compared to AA-pre-incubation and TNF-α stimulation (c, p < 0.001 and p < 0.05, respectively). DHA induced significantly less rolling after stimulation with 1 ng/ml TNF-α than AA and highly significantly less rolling than sole TNF-α stimulation (d, p < 0.05 and p < 0.001, respectively). Data are given as mean ± SEM
Stimulation of U937-cells with 1 ng/ml TNF-α for 4 h increased subsequent adhesion to endothelial cells. Fluorescence increased from 168.30 ± 4.94 FU (control) to 183.564 ± 2.14 FU. Pre-incubation of monocytes with DHA + TNF-α resulted in a slightly reduced adhesion (181.24 ± 3.00 FU) compared to TNF-α. In contrast, incubation of U937-monocytes with AA + TNF-α induced a highly significant difference in adhesion compared to DHA and compared to TNF-α [(274.16 ± 6.61 FU); (p < 0.001, n = 4–5; Fig. 2b)]. Since we aimed at investigating the FA-induced modulation, we continued our experiments using the lower TNF-α dose (1 ng/ml) that allowed a highly significant difference in adhesion between FA groups and did not provoke a ceiling effect as observed with 10 ng/ml TNF-α.
Impact of fatty acids on dynamic flow-chamber adhesion of freshly isolated human monocytes after TNF-α stimulation
Adhesion of unstimulated freshly isolated human monocytes under flow conditions to HUVEC was determined as 22 ± 3 adherent cells per high magnification field, and increased upon pre-incubation of freshly isolated human monocytes with 10 µM of AA to 40 ± 3 adherent cells and with DHA to 37 ± 3 adherent cells per high magnification field. AA and DHA differed highly significantly from unstimulated control. Stimulation of freshly isolated human monocytes with TNF-α increased adhesion to HUVEC highly significantly to 41 ± 3 adherent cells per high magnification field compared to unstimulated control. Pre-incubation with AA + TNF-α augmented adhesion to 43 ± 3 adherent cells per high magnification field. DHA + TNF-α pre-treatment induced a highly significant reduction to 27 ± 3 adherent cells per high magnification field compared to TNF-α and a significant reduction compared to TNF-α + AA as well as to DHA without TNF-α (p < 0.001 and p < 0.05; n = 6–10; Fig. 2c).
Impact of fatty acids on dynamic flow-chamber rolling of freshly isolated human monocytes after TNF-α stimulation
Rolling of unstimulated freshly isolated human monocytes under flow conditions to HUVEC was determined as 10 ± 1 rolling cells in 10 min, and increased upon pre-incubation of freshly isolated human monocytes with 10 µM of AA to 12 ± 2 rolling cells per high magnification field and with DHA to 11 ± 2 rolling cells per high magnification field. Stimulation of freshly isolated human monocytes with TNF-α increased rolling to HUVEC highly significantly to 18 ± 2 rolling cells per high magnification field compared to unstimulated control. Pre-incubation with AA + TNF-α resulted in rolling of 16 ± 2 cells per high magnification field. DHA + TNF-α pre-treatment induced a highly significant reduction to 9 ± 2 rolling cells per high magnification field compared to TNF-α and a significant reduction to TNF-α + AA (p < 0.001 and p < 0.05, n = 5–9; Fig. 2d).
Analysis of surface expression of adhesion molecules on U937-monocytes
Immunofluorescence analysis of CD11b (Mac-2), CD18, or CD49d (very late antigen (VLA)-4) on U937-monocytes by FACScan under the above mentioned conditions did not show a significant modulation between any groups (data not shown).
Analysis of surface expression of adhesion molecules on freshly isolated human monocytes
Immunofluorescence analysis of CD11b (Mac-2), CD18, or CD49d (very late antigen (VLA)-4) on freshly isolated human monocytes by FACScan under the above mentioned conditions did not show a significant modulation between any groups (data not shown).
Effect of the calcium-chelator BAPTA on FA-modulation of U937- and freshly isolated human monocyte adhesion to HUVEC after TNF-α exposure
Co-incubation of U937- and human monocytes with 10 µmol/l BAPTA and AA or DHA subsequent stimulation with 1 ng/ml TNF-α induced a similar increase in adhesion as TNF-α when compared to unstimulated cells (negative control) and were no longer able to differentially modulate adhesion. AA or DHA incubation of HUVEC without TNF-α-stimulation did not differ from negative control (data not shown).
Differential impact of FA on intracellular calcium in U937-monocytes after TNF-α stimulation
Stimulation of U937-monocytes with 1 ng/ml TNF-α induced an increase to 191.80 ± 8.25 % of intracellular calcium measured after 1 h with Fluo-3-AM-fluorescence with unstimulated U937-cells (negative control) set as 100 %. Pre-incubation of U937 with 10 µM DHA for 24 h and subsequent stimulation with TNF-α induced a significant reduction in calcium release to 153.97 ± 10.16 % compared to TNF-α and TNF-α + AA with 191.80 ± 8.25 % and 190.33 ± 7.34 %, respectively (p < 0.05; n = 7–8; Fig. 3a). Co-stimulation with FA and calcium-chelator BAPTA did not result in any significant modulation of intracellular calcium after TNF-α stimulation (data not shown).
Intracellular calcium is modulated by U937- and freshly isolated human monocyte pre-incubation with FA. U937-monocytes were pre-incubated arachidonic acid (AA), docosahexaenoic acid (DHA), or vehicle followed by stimulation with 1 ng/ml TNF-α. Intracellular calcium was measured using a fluorescence-based assay. DHA induced a significant reduction of intracellular calcium compared to AA and compared to TNF-α alone (a, p < 0.05). Using the same assay to measure intracellular calcium but examining freshly isolated human monocytes, pre-incubation with DHA and stimulation with 1 ng/ml TNF-α resulted in a highly significant reduced value of intracellular calcium compared to AA-pre-incubation with TNF-α stimulation and sole TNF-α stimulation (b, p < 0.001). Data are given as mean ± SEM
Differential impact of FA on intracellular calcium in freshly isolated human monocytes after TNF-α stimulation
Stimulation of freshly isolated human monocytes with 1 ng TNF-α induced a highly significant increase to 4270 ± 297 Fluorescence Units(FU) of intracellular calcium measured with Fluo-3-AM-fluorescence compared to unstimulated human monocytes with 2643 ± 364 FU. FA incubation without TNF-α resulted in equal levels of intracellular calcium as negative control with AA inducing 2966 ± 269 FU and DHA inducing 2777 ± 269 FU. Pre-incubation of human monocytes with 10 µM DHA and subsequent stimulation with TNF-α induced a highly significant reduction in calcium release to 2349 ± 269 FU compared to TNF-α and TNF-α + AA with 4270 ± 297 FU and 3690 ± 269 FU, respectively (p < 0.001; n = 6–11; Fig. 3b). Co-stimulation with FA and calcium-chelator BAPTA did not result in any significant modulation of intracellular calcium after TNF-α stimulation (data not shown).
Effect of raft disruption by MCD on FA-induced modulation of U937-monocyte adhesion and intracellular calcium after TNF-α stimulation
Adding 10 mmol/l MCD for 30 min prior to stimulation with 1 ng/ml TNF-α of U937-monocytes for cholesterol depletion and thus lipid raft disruption resulted in a reduced adhesion of U937-cells to HUVEC. With adhesion of MCD-treated but unstimulated monocytes set as 100 %, TNF-α increased adhesion to 151.51 ± 1.57 %. Under these conditions, AA (154.41 ± 1.16 %) and DHA (156.43 ± 1.31 %) failed to modulate adhesion (n = 5–7; Fig. 4a).
Disruption of U937- and freshly isolated human monocyte lipid rafts by MCD abolishes FA-induced modulation of adhesion, intracellular calcium and human monocyte rolling. Treatment of U937-monocytes with MCD prior to TNF-α stimulation abolished FA-induced effect on adhesion (a) and intracellular calcium (b). Treatment of freshly isolated human monocytes with MCD prior to TNF-α stimulation prevented a significant modulation of adhesion under flow conditions (c), intracellular calcium (d) and of rolling under flow conditions (e) by fatty acids. Data are given as mean ± SEM
After MCD-treatment, addition of TNF-α induced an increase to 116.27 ± 3.33 % of intracellular calcium measured with Fluo-3-AM-fluorescence with unstimulated monocytes set as 100 %. Under conditions of raft-disruption, pre-incubation with DHA (113.87 ± 1.00 %) or AA (112.21 ± 1.44 %) did not modulate TNF-α-induced calcium release (n = 4–7; Fig. 4b).
Effect of raft disruption by MCD on FA-induced modulation of freshly isolated human monocyte adhesion under flow conditions and intracellular calcium after TNF-α stimulation
Adding MCD prior to stimulation with TNF-α of freshly isolated human monocytes for cholesterol depletion and thus lipid raft disruption resulted in a reduced adhesion under flow conditions of human monocytes to HUVEC with 14.25 ± 1.66 cells compared to 22.90 ± 1.39 cells without MCD. Applying MCD, control without TNF-α treatment was at 10.50 ± 1.66 adherent cells with MCD. After MCD-treatment without TNF-α, AA and DHA induced 8.57 ± 1.26 cells and 8.2 ± 1.49 cells to adhere, respectively. After MCD treatment, TNF-α stimulation induced a significant difference to control (p < 0.05, n = 4–7; Fig. 4c).
After MCD-treatment, intracellular calcium control was elevated to 1595 ± 136 FU(fluorescence units). MCD-treatment increased TNF-α stimulated calcium to 1797 ± 122 FU. Without addition of TNF-α, MCD induced an increase of intracellular calcium measured with Fluo-3-AM-fluorescence after AA- and DHA-treatment to 1899 ± 157 FU and to 1876 ± 157 FU, respectively. Notably, MCD-treatment plus TNF-α stimulation resulted in no significant differences in calcium between AA pretreated cells with 2034 ± 122 FU, DHA pretreated cells with 1588 ± 136 FU and TNF-α with 1797 ± 122 FU. Under conditions of raft-disruption, pre-incubation with DHA or AA did not modulate TNF-α-induced calcium release (n = 3–5; Fig. 4d).
Effect of raft disruption by MCD on fatty acids induced modulation of dynamic flow-chamber rolling of freshly isolated human monocytes after TNF-α stimulation
Rolling of unstimulated freshly isolated human monocytes after MCD-treatment under flow conditions to HUVEC was determined as 1.33 ± 0.62 rolling cells in 10 min, and increased upon pre-incubation of freshly isolated human monocytes with 10 µM of AA to 1.67 ± 0.62 rolling cells per high magnification field and with DHA to 1.40 ± 0.59 rolling cells per high magnification field. Control cells without MCD-treatment showed a rolling of 2.86 ± 0.71 cells which increased after TNF-α-treatment to 8.78 ± 0.62 rolling cells. Stimulation of freshly isolated MCD-treated human monocytes with TNF-α increased rolling to HUVEC to 3.88 ± 0.66 rolling cells per high magnification field compared to unstimulated control. Pre-incubation with MCD + DHA + TNF-α pre-treatment induced 2.43 ± 0.71 rolling cells per high magnification field. MCD + AA + TNF-α resulted in a rolling of 3.83 ± 0.54 cells per high magnification field. Within the MCD-treated cells, rolling did not differ significantly (n = 7–11; Fig. 4e).
Discussion
The present study elucidates pathways influenced by n-3 (DHA) and n-6 (AA) FA in vascular inflammation. We could demonstrate that AA and DHA effectively modulate U937- and human monocyte lipid raft composition. As judged by FA analysis of raft and non-raft membrane fraction incorporation of DHA and AA was more pronounced in lipid rafts than in non-raft membrane fraction. Adhesion of U937- and freshly isolated human monocytes to HUVEC as well as human monocyte rolling was modulated differentially by n-3 and n-6 FA: AA pre-incubation of U937-monocytes induced a highly significant difference in adhesion compared to DHA after stimulation with 1 ng/ml TNF-α. Using 10 ng/ml TNF-α, AA did not further increase adhesion and DHA lead only to a small reduction. To prevent this “ceiling”-effect and since we aimed at investigating FA-induced modulation, we continued our experiments using 1 ng/ml TNF-α where a greater difference between FA could be observed. Analysis of adhesion molecules CD11b, CD18, and CD49d of U937- and human monocytes did not reveal any modulation in quantitative expression thus ruling out an impact of AA or DHA on expression of adhesion molecules under our conditions.
Next, our study proved calcium to play a role in FA-induced change in adhesion since differential modulation was abolished by pre-treatment with the calcium-chelator BAPTA. In addition, we found that disruption of U937- and freshly isolated human monocyte-lipid rafts by MCD also abolished the modulation of intracellular calcium and monocyte-endothelial adhesion as well as differential influence on human monocyte rolling by FA. We conclude that modulation of monocyte-endothelial adhesion and intracellular calcium by n-3 and n-6 FA is at least in part dependent on lipid rafts and their modulation of FA composition.
We could show that the approved method of lipid raft isolation using Triton X-100 and a sucrose-gradient was efficient using U937- and human monocytes [33]. After ultra-centrifugation, nine fractions were collected and raft-marker detection (Lyn and Fyn) by Western Blot was performed demonstrating fraction one and two as raft-fraction and fraction three to nine as non-raft fraction.
Polyunsaturated FA as AA and DHA are readily incorporated into lipid rafts, a feature not described for monocytes up to now. Incorporation of AA and DHA in U937- and human monocytes was higher in raft fraction than in non-raft fraction compared to TNF-α control with DHA being even more readily incorporated. This contrasts current ideas, because rafts are characterised by containing mostly saturated FA [8]. An explanation might be that T-cells lipid rafts are different in their ability to incorporate polyunsaturated FA as compared to monocyte lipid rafts. Another explanation would be that increased availability of free FA as used in our study—contrasting the esterified FA used in most other studies—targets polyunsaturated FA preferentially to naturally PUFA-deprived lipid rafts. Particular attention should be paid to the inflammatory setting in which incorporation of FA into membranes is different than under resting conditions [36]. The fact that TNF-α significantly increases the content of DHA in the raft fractions of the membrane is an interesting finding. One might speculate that this preferred allocation into the signalling platform might be a part of the altered inflammatory behaviour of monocytes after exposure to n-3 fatty acids. Furthermore, studying the effect of n-3 FA on lipid rafts in a biophysical model might also lead to different results than studying living cells. Though, reports also demonstrate a modulation of lipid rafts in different cells by n-3 FA some of them owing attention to the fact that n-3 FA incorporation could inhibit lipid raft signalling by altering its size or by simply disturbing its order [37–41].
We found that there is a differential influence of FA on adhesion and rolling with DHA exerting a reduction also compared to TNF-α except for the static U937 adhesion assay which might be due to the lack of flow conditions and the nature of the tumor cell line. The finding that monocyte adhesion and rolling to endothelial cells is modulated by n-3 and n-6 FA is well in line with previous reports from our group and others [25, 42, 43]. However, in this study we did not pre-incubate endothelial cells [25] but monocytes with FA. The finding that AA and DHA had no impact on expression of adhesion molecules in U937- and human monocytes contrasts the finding of other investigators in isolated monocytes [44] and endothelial cells [42]. However, different experimental conditions may fully compensate for the failure as we incubated monocytes in vitro (contrasting ingestion of fish oil by volunteers), and used low physiologic concentrations of free fatty acids as opposed to e.g., supraphysiologic concentrations of triglycerides. However, the finding is in line with previous reports of our group and others that did not show an impact of fatty acids on expression of adhesion molecules in endothelial cells or isolated monocytes derived after infusion of fish oil in volunteers [25, 31, 45]. Despite failure to detect an overall change in adhesion molecules recruitment of integrins into lipid rafts may be differentially modulated by fatty acids due to their effect on lipid composition of rafts. FA might alter adhesion via lipid rafts by exclusion or inclusion of signalling proteins into lipid rafts or changes of avidity or affinity of receptors which we did not investigate [46–48]. In inflammatory activated cells, also the high turnover of the membranes might play a significant role in modulation of signal transduction by FA [49]. There is evidence that the PAF–PAF-receptor system as well as the PI3-kinase system is affected by fatty acids in the process of adhesion [25, 43]. These receptor systems might also be affected in their affinity to lipid rafts by FA. Monocytes were already shown to be influenced by FA as judged by presentation of antigens in addition to adhesion in vitro as well as in vivo [31, 50]. Modulation of the inflammatory activated cell correlates with the situation of the intensive care patient suffering from an acute inflammatory disease requiring parenteral nutrition. Since parenteral nutrition consists of lipid emulsions based on triglycerides, fatty acids need to be liberated from triglycerides in order to become active. In septic conditions with catecholamines and heparin in the bloodstream, free fatty acids are augmented by order of magnitude compared to non-inflammatory conditions [31, 51]. It has been demonstrated that FA differently affect inflammatory activated cells and modulate various lipid-dependent signalling pathways [15, 19].
Control of adhesion by calcium signalling is generally recognized [52] and the fact that FA or their metabolites might have an impact on cellular calcium signalling has been investigated [53]. Several arachidonic acid-derived derivatives were identified that trigger Ca2+ entry into B cells, including the 5-lipoxygenase derived 5-hydroperoxyeicosatetranenoic acid and the cytochrome P450 hydroxylase product 20-hydroxyeicosatetraenoic. Influence of FA on U937-monocyte calcium has been observed [54] However, investigations demonstrating a calcium dependency of FA modulation of monocyte-endothelial adhesion like in our setting were missing until now. Our experiments demonstrated that DHA induces a decline of intracellular calcium in U937- and human monocytes in an inflammatory setting. This effect was more prominent using human monocytes. Here, we focus on modulation of adhesion by FA and were able to show a calcium-dependency of diverging effects of AA and DHA on adhesion since it was abolished after pre-treatment of monocytes with calcium-chelator BAPTA. Co-incubation of FA and BAPTA lead to a diminished calcium peak and abolished differences in intracellular calcium content between FA. We conclude that FA-influence on intracellular calcium contributes to modulation of monocyte-endothelial adhesion seen with FA. In U937-monocytes, we observed an enhanced intracellular calcium after TNF-α stimulation but in the static adhesion assay did not translate into an enhanced adhesion of U937-monocytes. This may be due to the non-physiological situation of the tumor cell line U937 combined with the static adhesion assay. Using freshly isolated human monocytes and a more physiological adhesion assay under flow conditions we could demonstrate a concordant significant difference between control and TNF-α stimulation in adhesion, rolling and intracellular calcium.
Reports demonstrating calcium dependency of lipid raft signalling have been published [55]. It could be depicted in endothelial cells that DHA reduces calcium in a raft-dependent manner [56]. In this study, we could demonstrate that disrupting monocyte lipid rafts with MCD abolished FA modulation of calcium signalling and FA influence on monocyte-endothelial adhesion and rolling. The significant difference between control and TNF-α stimulated monocytes seen in adhesion, rolling and calcium experiments could not always be detected after MCD treatment. An explanation would be that by destroying lipid rafts with MCD part of the raft-dependent-inflammatory signalling is lost. A toxic effect of MCD is to be neglected since dose and duration of MCD treatment was used as published previously and monocytes were tested for viability [57]. This would also be reasonable with respect to the decreased TNF-α-effect. Here, a hint is given at the recognized role of lipid rafts for inflammation in general [58]. But a more detailed investigation would be beyond the scope of this manuscript. Interesting work revealed an impact of FA on lipid raft dependent signal transduction. Though, most studies were performed in T-cells [20–23]. Modification of proteins by acylation as N-myristoylation and palmitoylation serves to target proteins into lipid rafts [59]. A possible mechanism of interference by FA with lipid raft signalling—besides altering raft FA composition—is the interaction with acylation of signalling or target proteins [21]. Signal transduction components like PAF(platelet-activated-factor)-PAF-receptor pathway or lipoprotein lipase A2, interacting with FA could be dependent on lipid rafts and be less influence able by FA when lipid rafts are destroyed. This may also explain why calcium modulation by FA is lost after lipid raft disruption [60].
To our knowledge, this study is the first report demonstrating in U937- and human monocytes that modulation of intracellular calcium and adhesion by FA is dependent on lipid rafts. It remains to be clarified how monocyte lipid rafts influence calcium signalling and via which route exactly intracellular calcium affects monocyte-endothelial adhesion.
Conclusions
We could demonstrate that n-6 and n-3 FA modulate FA content of monocyte lipid rafts to a greater extent than non-raft membrane fraction. Modulation of TNF-α-induced adhesion, rolling and intracellular calcium signalling by FA is abolished by disruption of lipid rafts. Therefore, lipid rafts seem to be crucial for inflammatory modulation by n-6 and n-3 FA in monocytes. This could help elucidating the role of FA in inflammatory diseases.
References
Mattila KJ, Valtonen VV, Nieminen MS, Asikainen S. Role of infection as a risk factor for atherosclerosis, myocardial infarction, and stroke. Clin Infect Dis Off Publ Infect Dis Soc Am. 1998;26(3):719–34.
Semeraro N, Ammollo CT, Semeraro F, Colucci M. Sepsis, thrombosis and organ dysfunction. Thromb Res. 2012;129(3):290–5.
Aird WC. Spatial and temporal dynamics of the endothelium. J Thromb Haemost. 2005;3(7):1392–406.
Schildberger A, Rossmanith E, Weber V, Falkenhagen D. Monitoring of endothelial cell activation in experimental sepsis with a two-step cell culture model. Innate Immun. 2010;16(5):278–87.
Burgazli KM, Venker CJ, Mericliler M, Atmaca N, Parahuleva M, Erdogan A. Importance of large conductance calcium-activated potassium channels (BKCa) in interleukin-1b-induced adhesion of monocytes to endothelial cells. Eur Rev Med Pharmacol Sci. 2014;18(5):646–56.
Sprague AH, Khalil RA. Inflammatory cytokines in vascular dysfunction and vascular disease. Biochem Pharmacol. 2009;78(6):539–52.
Legler DF, Micheau O, Doucey MA, Tschopp J, Bron C. Recruitment of TNF receptor 1 to lipid rafts is essential for TNFalpha-mediated NF-kappaB activation. Immunity. 2003;18(5):655–64.
Simons K, Vaz WL. Model systems, lipid rafts, and cell membranes. Annu Rev Biophys Biomol Struct. 2004;33:269–95.
Laude AJ, Prior IA. Plasma membrane microdomains: organization, function and trafficking. Mol Membr Biol. 2004;21(3):193–205.
Schmitz G, Orso E. CD14 signalling in lipid rafts: new ligands and co-receptors. Curr Opin Lipidol. 2002;13(5):513–21.
Charruyer A, Grazide S, Bezombes C, Muller S, Laurent G, Jaffrezou JP. UV-C light induces raft-associated acid sphingomyelinase and JNK activation and translocation independently on a nuclear signal. J Biol Chem. 2005;280(19):19196–204.
Hillyard DZ, Jardine AG, McDonald KJ, Cameron AJ. Fluvastatin inhibits raft dependent Fcgamma receptor signalling in human monocytes. Atherosclerosis. 2004;172(2):219–28.
Bouillon M, El Fakhry Y, Girouard J, Khalil H, Thibodeau J, Mourad W. Lipid raft-dependent and -independent signaling through HLA-DR molecules. J Biol Chem. 2003;278(9):7099–107.
NavarreteSantos A, Roentsch J, Danielsen EM, Langner J, Riemann D. Aminopeptidase N/CD13 is associated with raft membrane microdomains in monocytes. Biochem Biophys Res Commun. 2000;269(1):143–8.
Heller A, Koch T, Schmeck J, van Ackern K. Lipid mediators in inflammatory disorders. Drugs. 1998;55(4):487–96.
Mayer K, Seeger W, Grimminger F. Clinical use of lipids to control inflammatory disease. Curr Opin Clin Nutr Metab Care. 1998;1(2):179–84.
Gadek JE, DeMichele SJ, Karlstad MD, Pacht ER, Donahoe M, Albertson TE, et al. Effect of enteral feeding with eicosapentaenoic acid, gamma-linolenic acid, and antioxidants in patients with acute respiratory distress syndrome. Enteral Nutrition in ARDS Study Group. Crit Care Med. 1999;27(8):1409–20.
Marchioli R, Barzi F, Bomba E, Chieffo C, Di Gregorio D, Di Mascio R, et al. Early protection against sudden death by n-3 polyunsaturated fatty acids after myocardial infarction: time-course analysis of the results of the Gruppo Italiano per lo Studio della Sopravvivenza nell’Infarto Miocardico (GISSI)-Prevenzione. Circulation. 2002;105(16):1897–903.
Calder PC. n-3 polyunsaturated fatty acids, inflammation, and inflammatory diseases. Am J Clin Nutr. 2006;83(6 Suppl):1505S–19S.
Fan YY, McMurray DN, Ly LH, Chapkin RS. Dietary (n-3) polyunsaturated fatty acids remodel mouse T-cell lipid rafts. J Nutr. 2003;133(6):1913–20.
Stulnig TM, Berger M, Sigmund T, Raederstorff D, Stockinger H, Waldhausl W. Polyunsaturated fatty acids inhibit T cell signal transduction by modification of detergent-insoluble membrane domains. J Cell Biol. 1998;143(3):637–44.
Stulnig TM, Huber J, Leitinger N, Imre EM, Angelisova P, Nowotny P, et al. Polyunsaturated eicosapentaenoic acid displaces proteins from membrane rafts by altering raft lipid composition. J Biol Chem. 2001;276(40):37335–40.
Zeyda M, Staffler G, Horejsi V, Waldhausl W, Stulnig TM. LAT displacement from lipid rafts as a molecular mechanism for the inhibition of T cell signaling by polyunsaturated fatty acids. J Biol Chem. 2002;277(32):28418–23.
Grimminger F, Mayer K, Kramer HJ, Stevens J, Walmrath D, Seeger W. Differential vasoconstrictor potencies of free fatty acids in the lung vasculature: 2-versus 3-series prostanoid generation. J Pharmacol Exp Ther. 1993;267(1):259–65.
Mayer K, Merfels M, Muhly-Reinholz M, Gokorsch S, Rosseau S, Lohmeyer J, et al. Omega-3 fatty acids suppress monocyte adhesion to human endothelial cells: role of endothelial PAF generation. Am J Physiol Heart Circ Physiol. 2002;283(2):H811–8.
Schaefer MB, Wenzel A, Mohr A, Bi MH, Zaanen I, Braun-Dullaeus R, et al. Free fatty acids (FA) differentially influence PI3-kinase signalling in HUVEC. Faseb J. 2005;19(4):A170.
Jaffe EA, Nachman RL, Becker CG, Minick CR. Culture of human endothelial cells derived from umbilical veins. Identification by morphologic and immunologic criteria. J Clin Invest. 1973;52(11):2745–56.
Fowler MR, Naz JR, Graham MD, Bru-Mercier G, Harrison SM, Orchard CH. Decreased Ca2+ extrusion via Na+/Ca2+ exchange in epicardial left ventricular myocytes during compensated hypertrophy. Am J Physiol Heart Circ Physiol. 2005;288(5):H2431–8.
Baskin DS, Ngo H, Didenko VV. Thimerosal induces DNA breaks, caspase-3 activation, membrane damage, and cell death in cultured human neurons and fibroblasts. Toxicol Sci. 2003;74(2):361–8.
Fischer D, Bhattacharya R, Osburg B, Bickel U. Inhibition of monocyte adhesion on brain-derived endothelial cells by NF-kappaB decoy/polyethylenimine complexes. J Gene Med. 2005;7(8):1063–76.
Mayer K, Meyer S, Reinholz-Muhly M, Maus U, Merfels M, Lohmeyer J, et al. Short-time infusion of fish oil-based lipid emulsions, approved for parenteral nutrition, reduces monocyte proinflammatory cytokine generation and adhesive interaction with endothelium in humans. J Immunol. 2003;171(9):4837–43.
Lawrence MB, Springer TA. Leukocytes roll on a selectin at physiologic flow rates: distinction from and prerequisite for adhesion through integrins. Cell. 1991;65(5):859–73.
Waheed AA, Jones TL. Hsp90 interactions and acylation target the G protein Galpha 12 but not Galpha 13 to lipid rafts. J Biol Chem. 2002;277(36):32409–12.
Mayer K, Gokorsch S, Fegbeutel C, Hattar K, Rosseau S, Walmrath D, et al. Parenteral nutrition with fish oil modulates cytokine response in patients with sepsis. Am J Respir Crit Care Med. 2003;167(10):1321–8.
Bligh EG, Dyer WJ. A rapid method of total lipid extraction and purification. Can J Biochem Physiol. 1959;37(8):911–7.
Mayer K, Fegbeutel C, Hattar K, Sibelius U, Kramer HJ, Heuer KU, et al. Omega-3 vs. omega-6 lipid emulsions exert differential influence on neutrophils in septic shock patients: impact on plasma fatty acids and lipid mediator generation. Intensive Care Med. 2003;29(9):1472–81.
Duraisamy Y, Lambert D, O’Neill CA, Padfield PJ. Differential incorporation of docosahexaenoic acid into distinct cholesterol-rich membrane raft domains. Biochem Biophys Res Commun. 2007;360(4):885–90.
Hou TY, Monk JM, Fan YY, Barhoumi R, Chen YQ, Rivera GM, et al. n-3 polyunsaturated fatty acids suppress phosphatidylinositol 4,5-bisphosphate-dependent actin remodelling during CD4+ T-cell activation. Biochem J. 2012;443(1):27–37.
Langelier B, Linard A, Bordat C, Lavialle M, Heberden C. Long chain-polyunsaturated fatty acids modulate membrane phospholipid composition and protein localization in lipid rafts of neural stem cell cultures. J Cell Biochem. 2010;110(6):1356–64.
Park J, Yamaura T, Kawamoto J, Kurihara T, Sato SB. Reciprocal modulation of surface expression of annexin A2 in a human umbilical vein endothelial cell-derived cell line by eicosapentaenoic acid and docosahexaenoic acid. PLoS One. 2014;9(1):e85045.
Rockett BD, Teague H, Harris M, Melton M, Williams J, Wassall SR, et al. Fish oil increases raft size and membrane order of B cells accompanied by differential effects on function. J Lipid Res. 2012;53(4):674–85.
Weber C, Erl W, Pietsch A, Danesch U, Weber PC. Docosahexaenoic acid selectively attenuates induction of vascular cell adhesion molecule-1 and subsequent monocytic cell adhesion to human endothelial cells stimulated by tumor necrosis factor-alpha. Arterioscler Thromb Vasc Biol. 1995;15(5):622–8.
Schaefer MB, Wenzel A, Fischer T, Braun-Dullaeus RC, Renner F, Dietrich H, et al. Fatty acids differentially influence phosphatidylinositol 3-kinase signal transduction in endothelial cells: impact on adhesion and apoptosis. Atherosclerosis. 2008;197(2):630–7.
Hughes DA, Pinder AC, Piper Z, Johnson IT, Lund EK. Fish oil supplementation inhibits the expression of major histocompatibility complex class II molecules and adhesion molecules on human monocytes. Am J Clin Nutr. 1996;63(2):267–72.
Issekutz AC, Chuluyan HE, Lopes N. CD11/CD18-independent transendothelial migration of human polymorphonuclear leukocytes and monocytes: involvement of distinct and unique mechanisms. J Leukoc Biol. 1995;57(4):553–61.
Aziz M, Ishihara S, Ansary MU, Sonoyama H, Tada Y, Oka A, et al. Crosstalk between TLR5 and Notch1 signaling in epithelial cells during intestinal inflammation. Int J Mol Med. 2013;32(5):1051–62.
Lee EJ, Yun UJ, Koo KH, Sung JY, Shim J, Ye SK, et al. Down-regulation of lipid raft-associated onco-proteins via cholesterol-dependent lipid raft internalization in docosahexaenoic acid-induced apoptosis. Biochim Biophys Acta. 2014;1841(1):190–203.
Carman CV, Springer TA. Integrin avidity regulation: are changes in affinity and conformation underemphasized? Curr Opin Cell Biol. 2003;15(5):547–56.
Calder PC. Long-chain fatty acids and inflammation. Proc Nutr Soc. 2012;71(2):284–9.
Hughes DA, Pinder AC. N-3 polyunsaturated fatty acids modulate the expression of functionally associated molecules on human monocytes and inhibit antigen-presentation in vitro. Clin Exp Immunol. 1997;110(3):516–23.
Suchner U, Katz DP, Furst P, Beck K, Felbinger TW, Thiel M, et al. Impact of sepsis, lung injury, and the role of lipid infusion on circulating prostacyclin and thromboxane A(2). Intensive Care Med. 2002;28(2):122–9.
van der Voort van Zyp J, Conway WC, Thamilselvan V, Polin L, Basson MD. Divalent cations influence colon cancer cell adhesion in a murine transplantable tumor model. Am J Surg. 2005;190(5):701-7.
Zhu P, Liu X, Labelle EF, Freedman BD. Mechanisms of hypotonicity-induced calcium signaling and integrin activation by arachidonic acid-derived inflammatory mediators in B cells. J Immunol. 2005;175(8):4981–9.
Weber C, Aepfelbacher M, Lux I, Zimmer B, Weber PC. Docosahexaenoic acid inhibits PAF and LTD4 stimulated [Ca2+]i-increase in differentiated monocytic U937 cells. Biochim Biophys Acta. 1991;1133(1):38–45.
Cuschieri J, Bulger E, Garcia I, Maier RV. Oxidative-induced calcium mobilization is dependent on annexin VI release from lipid rafts. Surgery. 2005;138(2):158–64.
Ye S, Tan L, Ma J, Shi Q, Li J. Polyunsaturated docosahexaenoic acid suppresses oxidative stress induced endothelial cell calcium influx by altering lipid composition in membrane caveolar rafts. Prostaglandins Leukot Essent Fatty Acids. 2010;83(1):37–43.
Triantafilou M, Miyake K, Golenbock DT, Triantafilou K. Mediators of innate immune recognition of bacteria concentrate in lipid rafts and facilitate lipopolysaccharide-induced cell activation. J Cell Sci. 2002;115(Pt 12):2603–11.
Laurenzana A, Fibbi G, Chilla A, Margheri G, Del Rosso T, Rovida E, et al. Lipid rafts: integrated platforms for vascular organization offering therapeutic opportunities. Cell Mol Life Sci CMLS. 2015;72(8):1537–57.
Pike LJ. Lipid rafts: heterogeneity on the high seas. Biochem J. 2004;378(Pt 2):281–92.
Poisson C, Rollin S, Veronneau S, Bousquet SM, Larrivee JF, Le Gouill C, et al. Caveolae facilitate but are not essential for platelet-activating factor-mediated calcium mobilization and extracellular signal-regulated kinase activation. J Immunol. 2009;183(4):2747–57.
Acknowledgments
Dr. Martina B. Schaefer and Dr. Christian A. Schaefer were holders of post-doctoral grants from the Medical Faculty of the Justus-Liebig University, Giessen. This manuscript includes portions of the doctoral thesis of Stefanie Schifferings. Supported by: Deutsche Forschungsgemeinschaft, Transregio-SFB 84 innate immunity of the lung, Projekt C3 to KM and by a grant of the Novartis Foundation to KM.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible Editor: Mauro Teixeira.
Rights and permissions
About this article
Cite this article
Schaefer, M.B., Schaefer, C.A., Schifferings, S. et al. N-3 vs. n-6 fatty acids differentially influence calcium signalling and adhesion of inflammatory activated monocytes: impact of lipid rafts. Inflamm. Res. 65, 881–894 (2016). https://doi.org/10.1007/s00011-016-0971-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-016-0971-9