Abstract
Background
The role of inflammation in coronary artery disease (CAD) pathogenesis is well recognized. Moreover, smoking inhalation increases the activity of inflammatory mediators through an increase in leukotriene synthesis essential in atherosclerosis pathogenesis.
Aim
The aim of this study is to investigate the effect of “selected” genetic variants within the leukotriene (LT) pathway and other variants on the development of CAD.
Methods
CAD was detected by cardiac catheterization. Logistic regression was performed to investigate the association of smoking and selected susceptibility variants in the LT pathway including ALOX5AP, LTA4H, LTC4S, PON1, and LTA as well as CYP1A1 on CAD risk while controlling for age, gender, BMI, family history, diabetes, hyperlipidemia, and hypertension.
Results
rs4769874 (ALOX5AP), rs854560 (PON1), and rs4646903 (CYP1A1 MspI polymorphism) are significantly associated with an increased risk of CAD with respective odds ratios of 1.53703, 1.67710, and 1.35520; the genetic variant rs9579646 (ALOX5AP) is significantly associated with a decreased risk of CAD (OR 0.76163). Moreover, a significant smoking-gene interaction is determined with CYP1A1 MspI polymorphism rs4646903 and is associated with a decreased risk of CAD in current smokers (OR 0.52137).
Conclusion
This study provides further evidence that genetic variation of the LT pathway, PON1, and CYP1A1 can modulate the atherogenic processes and eventually increase the risk of CAD in our study population. Moreover, it also shows the effect of smoking-gene interaction on CAD risk, where the CYP1A1 MspI polymorphism revealed a decreased risk in current smokers.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The role of inflammation in coronary artery disease (CAD) pathogenesis is well recognized, and investigating genes involved in inflammatory processes provide further insight into the pathogenic pathways leading to CAD [1]. Oxidative modification of low-density lipoprotein-C (LDL-C), leukotriene-induced vasoconstriction, and platelet aggregation, as well as LDL accumulation in the arterial wall, are three of the main processes that lead to CAD [2]. Furthermore, it has been proposed that cigarette smoking causes lipid peroxidation which elevates atherosclerosis risk [3]. Cigarette smoke contains reactive peroxy radicals [4] and induces the polymorphonuclear leukocytes to produce oxygen-free radicals [5], further increasing oxidative damage, and atheromatous aggregation. These oxygen-free radicals oxidize LDL molecules [6] and form oxidized LDLs (oxLDLs) which are believed to be important initiators of atherosclerosis (Fig. 1) [2].
The relation between arachidonic acid (AA) metabolism and PON1 gene activation. A defective PON1 gene variant may lead to disrupted activity of the paraoxonase enzyme that normally inhibits the oxidation of low-density lipoproteins (LDL) into oxidized LDL (oxLDL), which is at the basis of atherogenesis by formation of foamy macrophages. Moreover, the AA metabolism will shift from the cycloxygenase (COX) pathway and favor the activation of the 5-LO pathway enzymes, particularly FLAP. This will lead to decreased activation of PPARγ by COX products, particularly prostaglandins (PGs), and thus, the activation of PON1 gene and production of PON1 by the liver will likely decrease. The activation of 5-lipoxygenase (5-LO) pathway of AA metabolism by the enzymes 5-lipoxygenase activating protein (FLAP) and 5-lipoxygenase (5-LO) will catalyze the conversion of AA into the chemokine, leukotriene A4 (LTA4), which will be converted into other leukotrienes by the enzymes LTA4 hydrolase (LTA4H) and LTC4 synthase (LTC4S). These leukotrienes act on leukotriene receptors in lymphocytes, endothelial, and smooth muscle cells, which further enhance inflammatory reactions and subsequently, atherogenesis. SNPs with (+) confer higher risk of CAD, SNPs with (−) are cardio-protective, and SNPs with dashed box indicate no significant association. Asterisk (*) indicates significant association and dashed lines indicate blocked pathway
Several enzymatic pathways play a significant role in the pathogenesis of CAD through their direct interaction with cigarette smoke metabolism. One of the major studied enzymes is CYP1A1, a member of cytochrome P-450 enzymes responsible for detoxification of polycyclic aromatic hydrocarbons (PAHs) released from the cigarette smoke in addition to neutralization of peroxy radicals as byproducts of metabolism [7, 8]. Variability in individual susceptibility to cigarette smoking-induced atherogenesis could be contributed to CYP1A1 variants. For example, CYP1A1 MspI variant (rs4646903—rare CC genotype) was shown to aggravate smoking risk with an increased triple vessel disease risk (OR 3.44; P = 0.0046) in light smokers (<20 pack-year) among a Caucasian population in Australia [9].
The leukotriene (LT) pathway is one of the most important inflammatory pathways widely attributed to atherosclerosis. Class four leukotrienes are potent pro-inflammatory mediators synthesized from arachidonic acid, an omega-6 polyunsaturated fatty acid (PUFA) [10]. ALOX5AP, LTA4H, and LTC4S are the major products of this pathway leading to vasoconstriction and leukocyte attraction and activation (Fig. 1) [11]. Nonetheless, there has been conflicting evidence in the literature regarding the association between ALOX5AP gene variants and cardiovascular disease, suggesting that this variability may be due to genetic differences that exit between populations with different ancestries [12]. In Central European patients with stroke, a significant association with several ALOX5AP SNPs was found [13]. Studies in North Americans, however, failed to show a significant association of the same SNPs with either stroke or MI [14, 15].
Another major contributing inflammatory mechanism involved in CAD progression is LDL accumulation in the arterial wall. This process activates several immune cells and pro-inflammatory cytokines, creating a local inflammatory state [16]. The HDL-associated paraoxonase 1 (PON1) decreases oxidation of LDL [29] and therefore inhibits the progression of atherosclerosis, which in turn reduces monocyte activation and prevents their adhesion to endothelial surface [17]. Several studies showed a significant association between Q192R and L55M variants of PON1 gene and risk to CAD [18, 19]. Moreover, the interaction between PON1 activity and cigarette smoking is well documented on the biological and genetic levels. Robertson et al. [20] showed a higher smoking associated risk in men carrying the PON1 55 M allele, and hence the LL genotype conferred a degree of protection from smoking-induced CAD risks.
The aim of our study is to investigate both the effect of genetic variants in the leukotriene pathway, PON1 and CYP1A1 on the risk of CAD, and the interaction between smoking and these genetic variants on the occurrence of CAD in our study population.
Methods
Study subjects
Based on complete data availability (Table 1), 1259 subjects were selected from a total of 7000 Lebanese patients undergoing cardiac catheterization and enrolled as part of a multi center cross-sectional study after they signed a consent form that was explained to them [21]. Samples with any of the following information missing were excluded from the study: age, gender, BMI, diagnosed diabetes, diagnosed hypertension, diagnosed hyperlipidemia, family history of CAD, and smoking status (non-smoker, ex-smoker, and current-smoker). Whole genome data were obtained for the selected subjects. The medical charts of all patients were reviewed for demographics, medical history, surgical and pharmacological interventions, and laboratory test results. The Institutional Review Board at the Lebanese American University approved the study protocol.
Variants selection
After reviewing the literature for CAD candidate genes, six genetic variants reported to influence to pathogenesis of CAD were selected for the purpose of this study. Given the significant role of inflammation in the pathogenesis of CAD, four variants (ALOX5AP: rs4769874 and rs9579646; LTA4H: rs6538697; LTC4S: rs730012) related to genes encoding crucial enzymes of a major inflammatory pathway, the leukotriene pathway, were chosen. Increased activity of ALOX5AP enzyme leads to accumulation of leukotrienes LTA4 and LTC4 that are important inflammatory mediators that lead to enhanced atherosclerosis (Fig. 1). Given the direct link of oxidized LDL accumulation and metabolism with atherosclerotic plaque formation, PON1 gene variant (rs854560) encoding for the protective enzyme paraoxonase enzyme was studied as well. In addition, CYP1A1 MspI (rs4646903) was selected for the extensive evidence on its interaction with smoking and CAD risk. Since these gene variants were proven to have population-specific association with CAD risk, they were hence investigated in the Lebanese population (Table 1).
Statistical analysis
The genotyping results of ALOX5AP, PON1, CYP1A1, LTC4S, and LTA4H variants were used from the GWAS data generated previously in our laboratory [21–24]. Actual genotype date were obtained for two of the variants (LTA4H: rs6538697 and LTC4S: rs730012), while the remaining ones were imputed using Impute2 [25] from the 1000 genomes reference panel. The imputed regions consisted of an area of 2.75 Mbps overlapping each of the five genes. Only the imputed variants with an imputation information score of >0.9 (PON1: rs854572, CYP1A1: rs4646903, ALOX5AP: rs9579646 and rs4769874) were included in the subsequent analysis. Odds ratios (ORs) with their 95 % confidence interval and p values were calculated for the susceptible variants in order to test their association with CAD using the logistic regression model while controlling for the covariates: gender, BMI, age, family history of CAD, hypertension, hyperlipidemia, diabetes, ex-smoker, and current smoker. Interactions among the 6 SNPs and smoking status were also tested in the regression model. Stepwise regression was then performed and the best-reduced model was obtained using the Akaike Information Criterion (AIC) in order to keep the most significant variants and covariates in the mode. The R statistical package version 3.0.3 [26] was used for all data analysis.
Results
Patients’ clinical and demographic characteristics are listed in Table 2. The total number of samples was reduced to 1959 after removing all those with missing data. The average age was found to be significantly different between the control group (60.9 ± 11.07, n = 589) and the CAD group (63.9 ± 10.31, n = 1370) (p value = 1.31 × 10−8). No significant difference was found for the BMI levels between control group (27.7 ± 5.1) and the CAD group (27.4 ± 3.9) (p value >0.05).
The model presented in Table 3 is the one obtained after fitting a stepwise regression model. Current-smoker and ex-smoker showed positive association with CAD and had the highest odds ratios (OR) of 8.415 with a p value of 6.39 × 10−4 and 4.751 with a p value of 1.81 × 10−2, respectively. All known risk factors were also found to be significantly associated with the occurrence of CAD with the exception of BMI (OR 0.98, p value >0.05). This is mainly due to the fact that there is no significant difference in BMI levels between the control and CAD groups. The female gender was found to be significantly negatively associated with CAD occurrence (OR 0.215, p value = 7.11 × 10−36), while age, family history of CAD, hypertension, diabetes, and hyperlipidemia were found to be significantly positively associated with the occurrence of CAD.
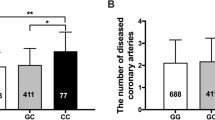
The variants rs6538697 and rs730012 in the genes LTA4H and LTC4S were not found to be significantly associated with CAD (p value >0.05). The variants rs4769874 and rs9579646 were found to be significantly associated with CAD (p values <0.05). In addition, variants rs4646903 and rs854560 in the genes CYP1Aa MspI and PON1 were both significantly positively associated with CAD with Odds Ratios of 1.677 (p value = 1.94 × 10−2) and 1.355 (p value = 1.69 × 10−2), respectively. We found that there is significant interaction between the “current-smoker” status and the variant rs4646903 of the gene CYP1A1 with an OR of 0.521 (p value = 3.66 × 10−2). Although the variant rs4646903 and the status “current-smoker” are both significantly associated with CAD with OR of 1.367 and 8.45, respectively, a reduced CAD risk is observed in current smokers carrying the rs rs4646903 variant.
Discussion
The pathogenic role of the inflammation leukotriene pathway in CAD was investigated. There have been conflicting results in the literature about the association between ALOX5AP gene variants and CAD suggesting some variability among populations with different ancestries [12, 13, 27, 28]. Our study demonstrated that the variant rs4769874 significantly increased the risk of CAD, while the variant rs9579646 was associated with decreased CAD risk. The rs4769874 variant is most likely linked to the enhanced expression of ALOX5AP and its gene product FLAP, hence amplifying the leukotriene pathway inflammatory reactions and atherogenic effects (Fig. 1). In contrast the rs9579646 variant revealed a significantly lower CAD risk in our study population suggesting that this variant may lead to a decreased expression of the ALOX5AP gene and its gene product FLAP, diminishing the drastic effects of the leukotriene pathway.
The role of paraoxonase (PON1) pathway in atherogenesis is shown in Fig. 1. Interactions between lipoproteins and immunity are crucial in atherosclerosis [17]. One of the major pathogenic mechanisms initiating and accelerating the process of atherosclerosis is the oxidative modification of native LDL-C in the arterial wall [29]. This eventually leads to the accumulation of foamy macrophages as these oxidized LDL molecules are taken up by the native macrophages. PON1 enzyme (paraoxonase) can prevent this pathological mechanism by interfering at two levels: inhibiting the oxidation of LDL into oxLDL and hydrolyzing oxLDL (Fig. 1) [30–32]. The role of PON1 in inflammation and in modulating the adverse oxidizing effect of smoking was investigated. PON1 L55M polymorphism rs854560 was significantly associated with an increased risk of CAD among patients in our study population. This risk may be due to a decreased catalytic activity in the PON1 L55M variant, impairing its ability to reduce the damage instigated by the oxidized LDL molecules in the arterial wall resulting in higher accumulation of oxidized LDL, enhancing atherogenesis and eventually CAD risk [33].
Of the major detoxifying enzymes in the body, the CYP enzymes are capable of both qualitatively and quantitatively altering the metabolism of exogenous toxins that are generated during cigarette smoking including PAHs [8]. Specifically, individual sensitivity to cigarette smoking is modulated by the CYP1A1 gene a phase I cytochrome P450 enzyme [9, 34–39]. The role of CYP1A1 variants has been investigated yielding conflicting results as to its impact on CAD [40]. In our study, CYP1A1 Msp I polymorphism rs4646903 was significantly associated with an increased risk of CAD, suggesting that the highly inducible form of CYP1A1 as previously reported aggravates atherosclerosis and eventually elevates the risk of CAD [9].
When the smoking-gene interaction was investigated in our study, CYP1A1 MspI polymorphism surprisingly presented an association with a reduced risk of CAD after correcting for all other factors. This effect was prominent especially with current smokers since they significantly showed nearly 50 % lower CAD risk. This association was also observed in ex-smokers but it was not significant. Our CYP1A1 and PON1 results are not aligned with much of the literature concerning the smoking-gene interaction since most of the studies found either no association or increased risk of CAD among smokers. These results warrant additional thorough investigation, of smoking-gene interaction in CAD in several populations before drawing solid conclusion on the effect of such interactions on CAD. Our results can also be explained by the possibility that this interaction is population-specific and the observed associations are due to other non-investigated genetic markers that are unique to this population.
Smoking-gene interaction model
Hazardous smoking effects form oxidized LDL, which will aggravate the inflammation by forming a progressive atherosclerotic plaque, eventually cause atherosclerosis and CAD [4]. Cigarette smoke contains reactive peroxy radicals [4] that result in the oxidation of LDL [6], leading to initiation of atherosclerosis [2]. An augmented expression of 5-lipoxygenase (5-LO) and downstream enzymes LTA4H and LTC4S was demonstrated in the macrophages from atherosclerotic lesions [41]. This evidence provides the link between smoking and the formation of ox-LDL and the ultimate formation of lipid-laden (foamy) macrophages with increased leukotriene synthesis leading to the atherosclerotic lesion formation. As shown in Fig. 1, LTs promote leukocyte chemotaxis, which will exacerbate cardiovascular inflammation by recruiting leukocytes into the arterial wall and atherosclerotic lesions.
In order to illustrate the overall interaction between the LT-pathway and PON1 activation, a common pathway linking the two inflammatory processes is proposed in Fig. 1. The PON1 gene is activated by peroxisome proliferator-activated receptors γ (PPAR-γ), leading to the synthesis and release of PON 1 enzyme from the liver [42]. Recently, inhibition of 5-LO by a 5-LO inhibitor has been shown to increase COX-2 expression and PGE2 production, which stimulates PPAR-γ expression [43]. This is further proven in a recent study showing that ALOX5 knock-out mice exhibits increased expression of COX-2 and PPAR-γ [44]. Overall, when the leukotriene is deactivated by the absence of 5-LO, the arachidonic acid metabolism will shift into the cyclooxygenase pathway and toward PG production, thus toward enhanced PPAR-γ function and PON1 production [45].
We deduce from our results that the inflammatory effect of the leukotriene pathway is intensified by the suggested enhanced expression of ALOX5AP rs4769874 variant and the decreased paraoxonase activity of the PON1 rs854560 variant. In addition, the reduced activity of CYP1A1 rs4646903 variant in detoxifying cigarette toxins will all contribute to a greater risk of CAD in subjects carrying these polymorphisms.
Limitations
Imputation accuracy might be a limitation in this study as imputed values are predicted values and only a proxy of the true values and the actual allele association in our model, which should be accounted for when estimating the variance and the odds ratios. Besides, the difference of our results compared to other population studies may be due to improper selection and categorization and selection of the cases and/or controls. Furthermore, it has been suggested that obesity is a significant contributor to LT pathway activation [46, 47], which might be a confounding factor in this study population. Finally, this remains a genetically focused study without functional phenotypic data.
Conclusion
This study provides further evidence that genetic variants of the LT pathway, PON1, and CYP1A1 can enhance the atherogenic processes and eventually increase the risk of CAD. Moreover, smoking inhalation increases the activity of inflammatory mediators by inducing the formation of oxidized LDL-driven foam cells. These foam cells have high leukotriene synthesizing capacity, which will augment the inflammatory mediators production in atherosclerosis. As shown in Fig. 1, many of basic researches leading to the lipoxygenase hypothesis point toward an involvement in early events of atheroma development, through LTB4-mediated migration and activation of monocyte/macrophages, as well as lipoxygenase-mediated LDL oxidation. An important finding in this study was the lower risk in carriers of both PON1 and CYP1A1 investigated variants among ex-smokers and current smokers with respect to the smoking-gene interaction among the Lebanese cohort, as opposed to studies demonstrating the absence or increased risk among other populations. Hence, population-specific studies on these genetic variants are essential to understand such disparities in results.
References
Cipollone F, Mezzetti A, Fazia ML, Cuccurullo C, Iezzi A, Ucchino S, et al. Association between 5-lipoxygenase expression and plaque instability in humans. Arterioscler Thromb Vasc Biol. 2005;25:1665–70.
Steinberg D. The LDL modification hypothesis of atherogenesis: an update. J Lipid Res. 2009;50(Suppl):S376–81.
Steinberg FM, Chait A. Antioxidant vitamin supplementation and lipid peroxidation in smokers. Am J Clin Nutr. 1998;68:319–27.
Church DF, Pryor WA. Free-radical chemistry of cigarette smoke and its toxicological implications. Environ Health Perspect. 1985;64:111–26.
Webster RO, Hong SR, Johnston RB Jr, Henson PM. Biologial effects of the human complement fragments C5a and C5ades Arg on neutrophil function. Immunopharmacology. 1980;2:201–19.
Niki E. Do free radicals play causal role in atherosclerosis? Low density lipoprotein oxidation and vitamin E revisited. J Clin Biochem Nutr. 2011;48:3–7.
Guengerich FP. Cytochromes P-450. Comp Biochem Physiol, C: Comp Pharmacol Toxicol. 1988;89:1–4.
Guengerich FP. Roles of cytochrome P-450 enzymes in chemical carcinogenesis and cancer chemotherapy. Cancer Res. 1988;48:2946–54.
Wang XL, Greco M, Sim AS, Duarte N, Wang J, Wilcken DE. Effect of CYP1A1 MspI polymorphism on cigarette smoking related coronary artery disease and diabetes. Atherosclerosis. 2002;162:391–7.
Peters-Golden M, Henderson WR Jr. Leukotrienes. N Engl J Med. 2007;357:1841–54.
Lotzer K, Funk CD, Habenicht AJ. The 5-lipoxygenase pathway in arterial wall biology and atherosclerosis. Biochim Biophys Acta. 2005;1736:30–7.
Zhang SY, Xu ML, Zhang CE, Qu ZY, Zhang BB, Zheng ZY, et al. Association of ALOX5AP gene single nucleotide polymorphisms and cerebral infarction in the Han population of northern China. BMC Med Genet. 2012;13:61.
Lohmussaar E, Gschwendtner A, Mueller JC, Org T, Wichmann E, Hamann G, et al. ALOX5AP gene and the PDE4D gene in a central European population of stroke patients. Stroke. 2005;36:731–6.
Zee RY, Cheng S, Hegener HH, Erlich HA, Ridker PM. Genetic variants of arachidonate 5-lipoxygenase-activating protein, and risk of incident myocardial infarction and ischemic stroke: a nested case-control approach. Stroke. 2006;37:2007–11.
Meschia JF, Brott TG, Brown RD Jr, Crook R, Worrall BB, Kissela B, et al. Phosphodiesterase 4D and 5-lipoxygenase activating protein in ischemic stroke. Ann Neurol. 2005;58:351–61.
Hansson GK, Libby P. The immune response in atherosclerosis: a double-edged sword. Nat Rev Immunol. 2006;6:508–19.
Aharoni S, Aviram M, Fuhrman B. Paraoxonase 1 (PON1) reduces macrophage inflammatory responses. Atherosclerosis. 2013;228:353–61.
Ruiz J, Blanche H, James RW, Garin MC, Vaisse C, Charpentier G, et al. Gln-Arg192 polymorphism of paraoxonase and coronary heart disease in type 2 diabetes. Lancet. 1995;346:869–72.
Schmidt H, Schmidt R, Niederkorn K, Gradert A, Schumacher M, Watzinger N, et al. Paraoxonase PON1 polymorphism leu-Met54 is associated with carotid atherosclerosis: results of the Austrian Stroke Prevention Study. Stroke. 1998;29:2043–8.
Robertson KS, Hawe E, Miller GJ, Talmud PJ, Humphries SE, Northwick Park Heart Study II. Human paraoxonase gene cluster polymorphisms as predictors of coronary heart disease risk in the prospective Northwick Park Heart Study II. Biochim Biophys Acta. 2003;1639:203–12.
Youhanna S, Platt DE, Rebeiz A, Lauridsen M, Deeb ME, Nasrallah A, et al. Parental consanguinity and family history of coronary artery disease strongly predict early stenosis. Atherosclerosis. 2010;212:559–63.
Ghassibe-Sabbagh M, Platt DE, Youhanna S, Abchee AB, Stewart K, Badro DA, et al. Genetic and environmental influences on total plasma homocysteine and its role in coronary artery disease risk. Atherosclerosis. 2012;222:180–6.
Alwan A, Youhanna SC, Platt DE, El-Sibai M, Yerezian JS, Deeb ME, et al. ALOX5AP gene variants show differential association with coronary artery disease in different populations. J Community Genet. 2010;1:107–15.
Saade S, Cazier JB, Ghassibe-Sabbagh M, Youhanna S, Badro DA, Kamatani Y, et al. Large scale association analysis identifies three susceptibility loci for coronary artery disease. PLoS One. 2011;6:e29427.
Howie BN, Donnelly P, Marchini J. A flexible and accurate genotype imputation method for the next generation of genome-wide association studies. PLoS Genet. 2009;5:e1000529.
Team RC. R: A language and environment for statistical computing. Vienna: Foundation for Statistical Computing; 2014.
Kaushal R, Pal P, Alwell K, Haverbusch M, Flaherty M, Moomaw C, et al. Association of ALOX5AP with ischemic stroke: a population-based case-control study. Hum Genet. 2007;121:601–7.
Domingues-Montanari S, Fernandez-Cadenas I, del Rio-Espinola A, Corbeto N, Krug T, Manso H, et al. Association of a genetic variant in the ALOX5AP with higher risk of ischemic stroke: a case-control, meta-analysis and functional study. Cerebrovasc Dis (Basel, Switzerland). 2010;29:528–37.
Navab M, Ananthramaiah GM, Reddy ST, Van Lenten BJ, Ansell BJ, Fonarow GC, et al. The oxidation hypothesis of atherogenesis: the role of oxidized phospholipids and HDL. J Lipid Res. 2004;45:993–1007.
Mackness B, Durrington PN, Mackness MI. The paraoxonase gene family and coronary heart disease. Curr Opin Lipidol. 2002;13:357–62.
Cao H, Girard-Globa A, Berthezene F, Moulin P. Paraoxonase protection of LDL against peroxidation is independent of its esterase activity towards paraoxon and is unaffected by the Q–> R genetic polymorphism. J Lipid Res. 1999;40:133–9.
Shih DM, Xia YR, Wang XP, Miller E, Castellani LW, Subbanagounder G, et al. Combined serum paraoxonase knockout/apolipoprotein E knockout mice exhibit increased lipoprotein oxidation and atherosclerosis. J Biol Chem. 2000;275:17527–35.
Gupta N, Singh S, Maturu VN, Sharma YP, Gill KD. Paraoxonase 1 (PON1) polymorphisms, haplotypes and activity in predicting cad risk in North-West Indian Punjabis. PLoS One. 2011;6:e17805.
Iwanari M, Nakajima M, Kizu R, Hayakawa K, Yokoi T. Induction of CYP1A1, CYP1A2, and CYP1B1 mRNAs by nitropolycyclic aromatic hydrocarbons in various human tissue-derived cells: chemical-, cytochrome P450 isoform-, and cell-specific differences. Arch Toxicol. 2002;76:287–98.
Kerzee JK, Ramos KS. Activation of c-Ha-ras by benzo(a)pyrene in vascular smooth muscle cells involves redox stress and aryl hydrocarbon receptor. Mol Pharmacol. 2000;58:152–8.
Kerzee JK, Ramos KS. Constitutive and inducible expression of Cyp1a1 and Cyp1b1 in vascular smooth muscle cells: role of the Ahr bHLH/PAS transcription factor. Circ Res. 2001;89:573–82.
McManus ME, Burgess WM, Veronese ME, Huggett A, Quattrochi LC, Tukey RH. Metabolism of 2-acetylaminofluorene and benzo(a)pyrene and activation of food-derived heterocyclic amine mutagens by human cytochromes P-450. Cancer Res. 1990;50:3367–76.
McNamara P, FitzGerald GA. Smoking-induced vascular disease: a new twist on an old theme. Circ Res. 2001;89:563–5.
Roberts-Thomson SJ, McManus ME, Tukey RH, Gonzalez FF, Holder GM. The catalytic activity of four expressed human cytochrome P450s towards benzo[a]pyrene and the isomers of its proximate carcinogen. Biochem Biophys Res Commun. 1993;192:1373–9.
Ingelman-Sundberg M. Genetic susceptibility to adverse effects of drugs and environmental toxicants. The role of the CYP family of enzymes. Mutat Res. 2001;482:11–9.
Silva AR, Pacheco P, Vieira-de-Abreu A, Maya-Monteiro CM, D’Alegria B, Magalhaes KG, et al. Lipid bodies in oxidized LDL-induced foam cells are leukotriene-synthesizing organelles: a MCP-1/CCL2 regulated phenomenon. Biochim Biophys Acta. 2009;1791:1066–75.
Khateeb J, Gantman A, Kreitenberg AJ, Aviram M, Fuhrman B. Paraoxonase 1 (PON1) expression in hepatocytes is upregulated by pomegranate polyphenols: a role for PPAR-gamma pathway. Atherosclerosis. 2010;208:119–25.
Avis I, Hong SH, Martinez A, Moody T, Choi YH, Trepel J, et al. Five-lipoxygenase inhibitors can mediate apoptosis in human breast cancer cell lines through complex eicosanoid interactions. FASEB J. 2001;15:2007–9.
Le P, Kawai M, Bornstein S, DeMambro VE, Horowitz MC, Rosen CJ. A high-fat diet induces bone loss in mice lacking the Alox5 gene. Endocrinology. 2012;153:6–16.
Kwak HJ, Park KM, Choi HE, Lim HJ, Park JH, Park HY. The cardioprotective effects of zileuton, a 5-lipoxygenase inhibitor, are mediated by COX-2 via activation of PKC delta. Cell Signal. 2010;22:80–7.
Back M, Avignon A, Stanke-Labesque F, Boegner C, Attalin V, Leprieur E, et al. Leukotriene production is increased in abdominal obesity. PLoS One. 2014;9:e104593.
Stanke-Labesque F, Pepin JL, Gautier-Veyret E, Levy P, Back M. Leukotrienes as a molecular link between obstructive sleep apnoea and atherosclerosis. Cardiovasc Res. 2014;101:187–93.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standards
This study has been approved by the Lebanese American University Institutional Review Board and has been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. We would like to state that all the study participants gave their informed and written consent prior to their inclusion in the study.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Responsible Editor: John Di Battista.
M. Merhi, S. Demirdjian, E. Hariri and N. Sabbah contributed equaly.
Rights and permissions
About this article
Cite this article
Merhi, M., Demirdjian, S., Hariri, E. et al. Impact of inflammation, gene variants, and cigarette smoking on coronary artery disease risk. Inflamm. Res. 64, 415–422 (2015). https://doi.org/10.1007/s00011-015-0821-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-015-0821-1