Abstract
De novo generation of T cells depends on continual colonization of the thymus by bone marrow-derived progenitors. Thymus seeding progenitors (TSPs) constitute a heterogeneous population comprising multipotent and lineage-restricted cell types. Entry into the thymic microenvironment is tightly controlled and recent quantitative studies have revealed that the adult murine thymus only contains approximately 160 niches to accommodate TSPs. Of these niches only about 6% are open for seeding on average at steady-state. Here, I review the state of understanding of colonization of the adult murine thymus with a particular focus on past and current controversies in the field. Improving thymus colonization and/or maintaining intact TSP niches during the course of pre-conditioning regimens are likely to be critical for efficient T-cell regeneration after hematopoietic stem cell transplantation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
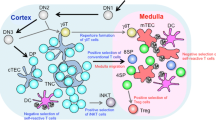
T cells are generated from bone marrow (BM)-derived progenitor cells. Whereas most hematopoietic cells complete development in bone marrow, T-cell progenitors undergo lineage commitment, selection as well as maturation in the thymus. These processes occur at phenotypically defined developmental stages. The most immature population of thymocytes are termed early T-lineage progenitors (ETPs) (Allman et al. 2003). They are negative for CD8, express low levels of CD4, and are otherwise characterized as CD117hiCD44hiCD25−. ETPs give rise to double-negative (negative for the co-receptors CD4 and CD8) two (DN2; CD117hiCD44hiCD25+) cells, at which stage T-lineage commitment is completed and somatic recombination of T-cell receptor (TCR) genes begins. The DN3 stage (CD44lo/−CD25+) is characterized by the pre-TCR checkpoint, followed by generation of double-positive (DP) cells, which are positive for CD4 and CD8 and comprise the largest population of thymocytes. At the DP stage, TCRα genes are rearranged, commitment to the CD4 and CD8 T lineages occurs combined with positive selection and the beginning of negative selection. The latter is completed in single-positive cells, which mature and ultimately egress from the thymus to contribute to the peripheral T-cell pool.
Under physiologic conditions, intrathymic T-cell development depends on continual colonization of the thymus by BM-derived progenitors (Donskoy and Goldschneider 1992). In hematopoietic stem cell transplantation (HSCT), entry of donor-derived progenitors may constitute a major limitation for de novo generation of naive T cells (Chaudhry et al. 2016; Zlotoff et al. 2011). Thus, conditioning therapies should be designed to limit injury to the recipient’s thymus. In addition, strategies to improve T-lineage regeneration through co-transplantation of in vitro-differentiated progenitors must take into account the thymus homing potential of such donor cells.
Here, I review the current understanding of physiologic colonization of the adult mouse thymus with a particular focus on the nature of thymus seeding progenitors (TSPs), their mode of thymus entry and, finally, quantitative aspects of thymus seeding.
Who?
The type of BM-derived progenitor cells colonizing the thymus has been a matter of intense debate. Some of the fundamental arguments of this debate can be recapitulated in two seminal review articles (Kawamoto and Katsura 2009; Schlenner and Rodewald 2010). With the characterization of a common lymphoid progenitor (CLP) population almost exclusively carrying lymphoid lineage potential in vivo, it appeared obvious that these progenitors should constitute the physiologic T lineage progenitor in the adult (Kondo et al. 1997). Such a scenario would best fit to a model of progressive lineage restriction throughout the hematopoietic developmental tree. However, certain in vitro differentiation systems supported a different path of lineage commitment with the presence of bipotent myeloid/B lineage and myeloid/T lineage progenitors but the distinct lack of bipotent B/T lineage progenitors (Lu et al. 2002). Additional questions concerning the role of CLPs constituting the physiologic T-cell progenitors were raised in a paper showing that Ikaros-deficient mice were able to generate T cells despite a complete absence of CLPs (Allman et al. 2003). Phenotypic similarities between BM-derived multipotent progenitors (MPPs) and intrathymic ETPs, most notably the absence of surface expression of interleukin (IL)-7R on both populations further supported a model suggesting that MPPs rather than CLPs were the physiologic TSPs. Thus, phenotypic similarity does not constitute a reliable criterion to establish pre- and intrathymic precursor–progeny relationships.
Physiologic T-lineage progenitors must fulfill three criteria that can be experimentally addressed: (1) they must possess T-lineage developmental potential; (2) they must be able to enter the thymus; and (3) they must have the capacity to leave BM and, therefore, be found in circulation.
T-lineage potential has been identified for a number of progenitor populations, including hematopoietic stem cells (HSCs), MPPs and their various subpopulations as well as more restricted progenitors, such as CLPs or even T–lineage-restricted progenitors.
A variety of in vitro assays can be employed to test T-lineage potential, such as fetal thymic organ cultures or co-cultures with BM stromal cells expressing delta-like Notch ligands (Schmitt and Zuniga-Pflucker 2002). In vivo, T-lineage potential can be assessed through direct intrathymic injection of candidate progenitors (Goldschneider et al. 1986). Whereas it is comparatively easy to determine T-lineage potential as such, it remains difficult to quantitatively assess T-lineage potential compared to the potential of a progenitor to enter alternative fates. This is mostly due to the fact that true clonal assays for multilineage potential comprising the T lineage remain a challenge due to the strong inductive character of Notch signals required to promote T-lineage fate. Conversely, it has been shown that even CLPs, which are strictly lymphoid-restricted in vivo, can give rise to myeloid cells in vitro under appropriate conditions (Łyszkiewicz et al. 2015; Serwold et al. 2009). In the thymus, only ablation of Notch signals allows for adoption of alternative lineage fates, such as intrathymic B cell and dendritic cell (DC) development (Feyerabend et al. 2009). In turn, in a somewhat artificial experimental approach it could be demonstrated that even committed DC progenitors could be, albeit very inefficiently, reprogrammed into the T lineage upon direct intrathymic transfer (Łyszkiewicz et al. 2015). Thus, the concept of T-lineage potential alone is not sufficient for progenitor cells to qualify as T-cell precursors.
Migration to the thymus is generally tested upon administration of BM-derived candidate progenitors into the bloodstream. However, the outcome here also depends to a large degree on experimental conditions. Transfer into non-manipulated mice constitutes the gold-standard, but such recipients are largely refractory to thymus seeding and, therefore, preclude easy quantitative analysis. However, pre-conditioning or use of mutant mice can significantly alter the result of such experiments. For instance, HSCs can directly colonize the thymus of irradiated animals, but not that of non-irradiated mice (Schwarz et al. 2007). IL-7R-deficient, P-selectin glycoprotein (PSGL)-1-deficient, and CCR7/CCR9 double-deficient mice have proven to be particularly receptive for BM-derived progenitors (Prockop and Petrie 2004; Rossi et al. 2005; Zietara et al. 2015). Notably, though, use of these mice as recipients has generated conflicting results with regard to cellular feedback mechanisms governing thymus colonization. The second factor to be taken account when analyzing thymus colonization is the time point of analysis. Short-term assays prevent potential misinterpretation of results obtained by progenitor cells recolonizing BM and undergoing differentiation there prior to entering thymus. However, such assays frequently detect cell numbers much larger than the number of cells actually capable of seeding the thymus at any given time (Mori et al. 2001; Scimone et al. 2006). In contrast, analysis after prolonged periods of time only detects true T-lineage progenitors but cannot ultimately exclude a detour via BM. Recently, it has been attempted to circumvent this conundrum by combining short-term transfers with prolonged differentiation in vitro, to exclusively capture progenitors that have both the capacity of thymus colonization and T-lineage potential (Saran et al. 2010; Zhang et al. 2014).
Presence in peripheral blood was first described for MPPs of the lineage negative, Sca-1+, c-kit+ (LSK) phenotype, which could be successfully isolated from peripheral blood in low numbers (Schwarz and Bhandoola 2004). Circulating LSK cells phenotypically and functionally resembled their BM counterparts. They displayed phenotypic heterogeneity in terms of Flt3 expression, suggesting that blood LSK cells comprised HSCs and MPPs, including lymphoid-primed LMPPs. In addition, they displayed T-lineage potential in vivo and colonized the thymus of irradiated hosts. Later, multiple groups reported the presence of CLPs in peripheral blood as well (Saran et al. 2010; Schwarz et al. 2007; Serwold et al. 2009; Umland et al. 2007). A third T-lineage progenitor population termed circulating T-lineage committed progenitor was isolated from peripheral blood at numbers similar to LSK cells (Krueger and von Boehmer 2007). Unlike other adult progenitors but similar to fetal T-lineage progenitors, these cells were largely restricted to the T lineage prior to thymus colonization.
Conflicting data on the nature of TSPs due to variations in assay conditions prompted a number of groups to devise more rigorous experiments to define true TSPs. Thus, to avoid confounding effects of infusing excessive numbers of progenitors, selective depletion of candidate progenitors rather than enrichment was employed to generate donor cell samples (Saran et al. 2010; Serwold et al. 2009). These assays revealed minimal surface expression characteristics for progenitors, as complete loss of thymus colonization of non-irradiated hosts was observed when donor populations were depleted of cells expressing CD27 and/or CD135 (Flt3). Furthermore, such experiments showed that TSPs constitute a heterogeneous population comprising at least both (L)MPPs as well as CLPs (Fig. 1a). Genetic fate mapping traces the developmental history of a cell without the need of transfer experiments. As ETPs lack surface expression of IL-7R (CD127) and also express very low amounts of Il7r mRNA, IL-7R-Cre-mediated fate mapping was chosen to test whether ETPs were derived from IL-7R expressing committed lymphoid cells or directly derived from IL-7R-negative multipotent cells (Schlenner et al. 2010). Indeed, the majority of ETPs (75–80%) in such mice displayed a history of IL-7R expression indicating that mostly lymphoid-committed progenitors rather than multipotent cells constituted physiologic TSPs. Interestingly, in neonatal mice a small subset of LMPPs was described expressing high levels of Flt3, CCR9 and a Rag1-driven GFP reporter gene, which also expressed considerable levels of IL-7R (Luc et al. 2012). These IL-7R+ LMPPs had transcriptional profiles very similar to CLPs as well as to ETPs. Furthermore, the presence of these cells is consistent with data from IL-7R-Cre fate mapping experiments showing a higher frequency of fate-map positive cells among LMPPs when compared to HSCs or myeloid committed cells (Schlenner et al. 2010). However, this model is characterized by a clear dichotomy of peripheral fate-map positive cells exclusively belonging to the lymphoid lineage and fate-map negative cells virtually exclusively belonging to the myeloid lineage. Thus, IL-7R-Cre fate mapping clearly separates lymphoid cells from other lineages and indicates that in vivo IL-7R+ LMPPs are bona fide lymphoid progenitors.
Phenotype and homing of TSPs. a The heterogeneous population of TSPs is characterized by the shared markers CD135 (Flt3) and CD27. Subsets of TSPs comprise multipotent progenitors (MPPs) expressing high levels of CD117 (c-kit), common lymphoid progenitors (CLPs) expressing low levels of CD117 and CD127 (IL-7Rα) and most likely other cell types as well. b Surface receptors on TSPs involved in thymus seeding
The biological significance of the heterogeneity of TSPs remains elusive. However, it has been demonstrated that MPPs and CLPs generate T-lineage progeny with different kinetics and it has been suggested that colonization of the adult thymus occurs in waves (Foss et al. 2001; Saran et al. 2010). Thus, heterogeneity of TSPs might account for continuous de novo production of T cells despite discontinuous thymus colonization.
How?
To colonize the thymus, TSPs must leave the bloodstream through the vascular endothelium. The port of entry for TSPs is most likely located at the cortico-medullary junction (Lind et al. 2001). The basic mechanism of progenitor transit through the endothelium is very similar to that of other immune cells leaving the circulation into peripheral tissues or lymph nodes and involves the adhesion molecule CD44, selectins, integrins, and chemokine receptors (Fig. 1b).
CD44 has been implicated in thymus colonization already shortly after its identification as a marker of early thymocytes (Graham et al. 2007; Wu et al. 1993). In short-term homing assays, a multistep adhesion cascade for thymus colonization was characterized. P-selectin—PSGL-1 interactions form the first step of adhesion of blood borne progenitors to the thymic vascular endothelium (Rossi et al. 2005; Scimone et al. 2006). Requirement of this interaction for efficient thymus seeding was shown in long-term and short-term assays as well as in parabiotic mice. Of note, loss of this interaction did not fully abrogate thymus colonization.
As a second step of progenitor homing to the thymus at steady state, integrins are activated through chemokine receptor signals and the requirement for such interactions was demonstrated by inhibition of chemokine receptor signal-transducing Gαi proteins by pertussis toxin (Scimone et al. 2006). Blocking of CCL25 or loss of CCR9 resulted in a partial block of thymus homing and reduced numbers of ETPs suggesting that the CCL25/CCR9 axis plays a critical role in thymus colonization (Scimone et al. 2006; Svensson et al. 2008; Uehara et al. 2002). In contrast, blocking of CXCL12/CXCR4 had no such effect (Scimone et al. 2006).
At present, it remains unclear whether direct migration along chemokine gradients plays a dominant role in chemokine receptor function during thymus colonization. It has been reported that pre-treatment of progenitor cells with CCL25 was able to promote recruitment into the irradiated thymus, suggesting that chemokine-directed migration is not the sole function of chemokine receptor signaling during thymus colonization (Zhang et al. 2014).
Finally, blocking of α4β1/VCAM-1 and αLβ2/ICAM-1 interactions resulted in a partial loss of homing capacity to the thymus (Scimone et al. 2006).
Interfering with individual selectin, chemokine receptor, or integrin interactions was characterized by a partial loss of homing capacity at best, suggesting that most, if not all, steps of thymus colonization display molecular redundancy. Such redundancy has been best characterized for chemokine receptors involved in thymus seeding. Despite the fact that CCR7-deficient mice do not display a deficiency in numbers of ETPs, competitive transfers showed that BM-derived progenitors from these mice also had a partial defect in thymus colonization (Krueger et al. 2010; Zlotoff et al. 2010). These findings prompted several laboratories to test whether CCR7 and CCR9 together might control thymus colonization completely. Indeed, compound loss of CCR7 and CCR9 resulted in almost complete absence of ETPs at steady state as well as a complete block in thymus colonization in a competitive setting (Krueger et al. 2010; Zlotoff et al. 2010). Surprisingly, total thymus cellularity was virtually normal in CCR7/CCR9 double-deficient mice, showing an almost exclusive loss of ETPs and DN2 cells with only a moderate reduction of DN3 cell numbers. The reason for recovery of thymus size in these mice has not been fully explained, as some investigators but not others provided evidence for compensatory proliferation at the level of DN3 thymocytes.
TSPs as well as ETPs, which constitute the earliest phenotypically discernable intrathymic population, express the cytokine receptors Flt3 (CD135) and c-kit (CD117). Prethymic signals through Flt3 are required for expression of CCR9 on TSPs (Schwarz et al. 2007). Consistently, Flt3-deficient progenitors have a competitive disadvantage in thymus colonization. Once TSPs have entered the thymic microenvironment, a number of factors contribute to their maintenance in what can be tentatively described as TSP niche. Recently, it has been shown that vascular endothelial cells that depend on cross-talk with thymocytes via lymphotoxin β-receptor signaling are critical for thymus colonization, possibly by providing a suitable niche for TSPs (Lucas et al. 2016; Shi et al. 2016). Consistently, these cells express membrane-bound stem cell factor, the ligand for c-kit, thus providing signals to ETPs and it is likely that TSPs as their immediate precursors are receiving such signals as well (Buono et al. 2016).
How Many?
Quantification of the number of TSPs colonizing the adult thymus has been hampered by a number of factors. First, the most immature intrathymic progenitor population, the ETPs, share phenotypic similarity only with some, but not other BM-derived progenitors. Thus, upon entry into the thymic microenvironment, TSPs undergo rapid phenotypic and presumably also functional changes, which are most likely to be induced through strong Notch signals (Krueger et al. 2006; Schwarz et al. 2007). These changes preclude direct observation of TSPs without manipulation. Second, failure to directly detect BM-derived progenitors after thymus colonization indicated that very few progenitors are able to colonize the thymus at any given time. This idea was supported by transfer experiments followed by microscopic analysis, which failed to detect more than few individual cells in microscopic sections (Lind et al. 2001). Further evidence for low numbers of TSPs was obtained already multiple decades ago. Estimates on the amount of thymus colonization were obtained by titration of donor BM into irradiated hosts and from mixed BM transplantation experiments (Ezine et al. 1984; Kadish and Basch 1976; Wallis et al. 1975). The latter experiments showed that both thymic lobes were colonized by unequal mixtures of cells and could be mathematically analyzed using binomial equations. The numbers obtained for colonization of the irradiated thymus were in the range 10–300 progenitors (for review see Scollay et al. 1986). Major limitations of these early studies were the use of irradiated recipients and an extended phase of experimental inaccessibility, because the only marker for donor cells was the Thy1 antigen (CD90), which is only expressed at beginning of the DN2 stage, i.e. 10–12 days after colonization. This black box period precluded faithful separation of true colonization events with intrathymic proliferation or cell death. To prevent such a scenario, short-term assays have been developed, looking at thymus colonization within days or even hours after transfer (Martin et al. 2003; Mori et al. 2001; Scimone et al. 2006). However, there is evidence that in these assays considerable cell loss occurs resulting in over-estimation of thymus seeding.
Recently, a novel approach termed multicongenic fate mapping was employed to re-assess thymus colonization (Zietara et al. 2015). This approach was based on transfer of defined mixtures of congenically and transgenically marked donor cells followed by flow cytometric analysis once donor-derived cells had reached the most numerous DP stage of development. Late analysis thus guaranteed restriction to progenitors entering the T lineage and excluding those that were incapable of seeding TSP niches. Mathematical analysis was then based on counting the number of congenic subsets missing at the time point of analysis, thus converting cell numbers into simple present/absent calls and excluding intrathymic proliferation as confounding factor. These experiments revealed that on average ten TSP niches were available for colonization at steady-state in wild-type mice (Fig. 2). To assess the number of TSP niches in a virtually empty thymus, CCR7/CCR9 double-deficient mice were used as recipients (Zietara et al. 2015). Due to a near complete failure of endogenous progenitors to colonize the thymus, these mice almost completely lack ETPs and most likely TSPs as well (Krueger et al. 2010; Zlotoff et al. 2010). Despite these defects, thymus cellularity is essentially normal as is the surface area of the cortico-medullary junction and the levels of expression of key chemokines and cytokines required for homing. Together, these factors qualify CCR7/CCR9 double-deficient mice as a suitable model to quantitate TSP niches. Multicongenic fate mapping revealed the presence of 160 TSP niches in these mice and a comparable number of open niches in irradiated recipients as well (Fig. 2). Together, these experiments indicated that the adult murine thymus contains approximately 160 progenitor niches, 6% of which can be refilled on average at steady-state at any given time (Zietara et al. 2015). Interestingly, these findings were in extremely good accordance with estimates made 3–4 decades earlier.
Low numbers of TSP niches suggested that thymus colonization is a tightly regulated process. However, the limiting factors in this process remain largely elusive. It has been proposed that thymus colonization occurs in a discontinuous periodic manner. Transfers of progenitors into non-irradiated recipients of defined age combined with studies of timed separation of parabiotic mice revealed a 4-week periodicity with 1 week of thymic receptivity followed by three refractory weeks (Foss et al. 2001). A more detailed study, albeit in irradiated recipients, concluded that thymus colonization occurred within 4 and 24 h after transfer followed by progressive and ultimately complete refractoriness. TSP niches are vacated again after 7–14 days (Foss et al. 2002).
To uncover potential molecular mechanisms of control of thymus colonization, mutant mouse strains were tested for thymus receptivity. Comparison of Rag-deficient mice, which are as refractory as wild-type mice, with Il7r-deficient mice, which are receptive for TSPs, led to the conclusion that DN3 thymocytes provided negative feedback to the TSP niche (Prockop and Petrie 2004). In contrast, comparison of CCR7-deficient (refractory), CCR9-deficient (mildly susceptible) and CCR7/CCR9 double-deficient recipients showed that receptivity correlated best with low numbers of ETPs and DN2, but not DN3, thymocytes (Zietara et al. 2015). This study also revealed a periodicity of 9–12 days in thymus colonization of non-manipulated wild-type as well as CCR7/CCR9-deficient mice, consistent with the estimated life-time of ETPs (Krueger et al. 2017; Porritt et al. 2003). Of note, this study failed to confirm age-dependent 4-week periodicity of thymus colonization for as yet unknown reasons. Similarly, another study reported a high degree of thymus receptivity in PSGL-1-deficient mice correlating with reduced numbers of ETPs but not DN3 thymocytes (Rossi et al. 2005). Furthermore, multicongenic fate mapping had shown that non-manipulated Il7r-deficient mice had only slightly elevated numbers of free TSP niches compared to wild-type mice, suggesting that thymic receptivity in these mice is not exclusively due to availability of TSP niches. Together these findings suggest that ETPs provide cellular feedback to thymus colonization, presumably by competing for TSP niches.
The molecular mechanisms governing thymic receptivity remain largely elusive. It has been reported that some mutant mouse strains with elevated thymus receptivity, including Il7r-deficient and PSGL-1-deficient mice, expressed higher levels of CCL25 and P-selectin, and that P-selectin and CCL25 levels were subject to periodic alterations (Gossens et al. 2009). Thus, this study suggested that CCL25 and P-selectin might control gated thymus colonization. However, elevated levels of CCL25 were not detectable in the thymus of receptive CCR7/CCR9 double-deficient mice (Zietara et al. 2015). Interestingly, modulation of P-selectin levels might constitute a means of communication between thymus and peripheral T-cell numbers via an S1P-dependent feedback mechanism (Gossens et al. 2009). Thymus receptivity in PSGL-1-deficient mice coincided with a defect in thymic egress and concomitant peripheral lymphopenia. Rescue of the lymphopenic state was sufficient to dampen thymus homing in these mice. Conversely, peripheral T-cell depletion in wild-type mice promoted thymus receptivity. Of note, there is no overt paucity in peripheral T cells in CCR7/CCR9 double-deficient mice, although alterations in T-cell subsets remain to be characterized. Taken together, these studies suggest that distinct steps of thymus colonization, such as crossing the endothelial barrier and occupation of TSP niches, might be governed by independent feedback mechanisms with distinct functions.
Conclusion
Thymus colonization has been extensively characterized over the past four decades. Thus, a picture is emerging suggesting that thymus is colonized by a heterogeneous mixture of TSPs comprising both (L)MPPs and CLPs and possibly other progenitors as well. However, it remains an open question to what extent each individual sub-population eventually contributes to T-cell output. We have hypothesized that progenitor heterogeneity ensures continuous output despite discontinuous thymus colonization (Saran et al. 2010). Furthermore, it is by now well-established that colonization is a rare event with the adult murine thymus providing only approximately 160 niches for TSPs, of which the majority is occupied at any given time point. TSP niche occupation is a periodic event, although controversies exist with regard to the time-span of each wave (9–12 days or 4 weeks or both). Whereas the short-term periodicity is likely to be directly controlled through competition with ETPs, no plausible explanation exists yet for gated import at 4-week intervals.
Thymus colonization is critical not only for developing strategies to improve T-lineage regeneration after HSCT. Continuous replacement of TSPs is also critical to prevent malignant transformation of thymocytes. It has been shown that upon inhibition of thymus colonization for extended periods of time, e.g. via transplantation of wild-type thymus into progenitor-deficient hosts, intrathymic progenitors adopt stem-cell-like characteristics and begin supporting T-cell development for extended periods of time (Martins et al. 2012; Peaudecerf et al. 2012). Ultimately, removal of competition for intrathymic progenitor niches results in formation of leukemia (Martins et al. 2014).
Despite its relevance for T-cell regeneration after HSCT, thymus colonization in humans remains poorly understood. A number of candidate human TSP populations as well as intrathymic ETPs have been characterized. These comprise CD34hiCD45RAhiCD7+ multipotent cells of fetal origin as well as postnatal CD34+CD10+CD7− lymphoid-restricted cells (Haddad et al. 2006; Six et al. 2007). Transcriptional programs of lineage commitment of such progenitors have been increasingly well characterized (Casero et al. 2015; Laurenti et al. 2013). However, it remains a central problem in the study of human thymus colonization that migration cannot be studied directly in vivo. Humanized mouse models represent excellent tools to study human immune system development in vivo (Shultz et al. 2012). However, thymus colonization in these mice is complicated by mutations in the Rag and cytokine receptor γ chain genes required to induce murine immunodeficiency. These mutations result in development of a very small, structurally abnormal thymus, the size of which does not fully recover during human progenitor cell reconstitution. Nevertheless, development of improved mouse strains for humanization, for instance by introducing transgenes for human cytokines and chemokines to limit requirements of cross-reactivity between human and mouse receptor-ligand pairs, remains a promising approach to improve our understanding of human thymus colonization.
References
Allman DM, Sambandam A, Kim S et al (2003) Thymopoiesis independent of common lymphoid progenitors. Nat Immunol 4:168–174
Buono M, Facchini R, Matsuoka S et al (2016) A dynamic niche provides Kit ligand in a stage-specific manner to the earliest thymocyte progenitors. Nat Cell Biol 18:157–167
Casero D, Sandoval S, Seet CS et al (2015) Long non-coding RNA profiling of human lymphoid progenitor cells reveals transcriptional divergence of B cell and T cell lineages. Nat Immunol 16:1282–1291
Chaudhry MS, Velardi E, Dudakov JA et al (2016) Thymus: the next (re)generation. Immunol Rev 271:56–71
Donskoy E, Goldschneider I (1992) Thymocytopoiesis is maintained by blood-borne precursors throughout postnatal life. A study in parabiotic mice. J Immunol 148:1604–1612
Ezine S, Weissman IL, Rouse RV (1984) Bone marrow cells give rise to distinct cell clones within the thymus. Nature 309:629–631
Feyerabend TB, Terszowski G, Tietz A et al (2009) Deletion of Notch1 converts pro-T cells to dendritic cells and promotes thymic B cells by cell-extrinsic and cell-intrinsic mechanisms. Immunity 30:67–79
Foss D, Donskoy E, Goldschneider I (2001) The importation of hematogenous precursors by the thymus is a gated phenomenon in normal adult mice. J Exp Med 193:365–374
Foss D, Donskoy E, Goldschneider I (2002) Functional demonstration of intrathymic binding sites and microvascular gates for prothymocytes in irradiated mice. Int Immunol 14:331–338
Goldschneider I, Komschlies KL, Greiner DL (1986) Studies of thymocytopoiesis in rats and mice. I. Kinetics of appearance of thymocytes using a direct intrathymic adoptive transfer assay for thymocyte precursors. J Exp Med 163:1–17
Gossens K, Naus S, Corbel SY et al (2009) Thymic progenitor homing and lymphocyte homeostasis are linked via S1P-controlled expression of thymic P-selectin/CCL25. J Exp Med 206:761–778
Graham VA, Marzo AL, Tough DF (2007) A role for CD44 in T cell development and function during direct competition between CD44+ and CD44− cells. Eur J Immunol 37:925–934
Haddad R, Guimiot F, Six EM et al (2006) Dynamics of thymus-colonizing cells during human development. Immunity 24:217–230
Kadish JL, Basch RS (1976) Hematopoietic thymocyte precursors. I. Assay and kinetics of the appearance of progeny. J Exp Med 143:1082–1099
Kawamoto H, Katsura Y (2009) A new paradigm for hematopoietic cell lineages: revision of the classical concept of the myeloid-lymphoid dichotomy. Trends Immunol 30:193–200
Kondo M, Weissman IL, Akashi K (1997) Identification of clonogenic common lymphoid progenitors in mouse bone marrow. Cell 91:661–672
Krueger A, von Boehmer H (2007) Identification of a T lineage-committed progenitor in adult blood. Immunity 26:105–116
Krueger A, Garbe AI, von Boehmer H (2006) Phenotypic plasticity of T cell progenitors upon exposure to Notch ligands. J Exp Med 203:1977–1984
Krueger A, Willenzon S, Lyszkiewicz M et al (2010) CC chemokine receptor 7 and 9 double-deficient hematopoietic progenitors are severely impaired in seeding the adult thymus. Blood 115:1906–1912
Krueger A, Zietara N, Łyszkiewicz M (2017) T cell development by the numbers. Trends Immunol 38:128–139
Laurenti E, Doulatov S, Zandi S et al (2013) The transcriptional architecture of early human hematopoiesis identifies multilevel control of lymphoid commitment. Nat Immunol 14:756–763
Lind EF, Prockop SE, Porritt HE et al (2001) Mapping precursor movement through the postnatal thymus reveals specific microenvironments supporting defined stages of early lymphoid development. J Exp Med 194:127–134
Lu M, Kawamoto H, Katsube Y et al (2002) The common myelolymphoid progenitor: a key intermediate stage in hemopoiesis generating T and B cells. J Immunol 169:3519–3525
Luc S, Luis TC, Boukarabila H et al (2012) The earliest thymic T cell progenitors sustain B cell and myeloid lineage potential. Nat Immunol 13:412–419
Lucas B, James KD, Cosway EJ et al (2016) Lymphotoxin β receptor controls T cell progenitor entry to the thymus. J Immunol 197:2665–2672
Łyszkiewicz M, Zietara N, Föhse L et al (2015) Limited niche availability suppresses murine intrathymic dendritic-cell development from noncommitted progenitors. Blood 125:457–464
Martin C, Aifantis I, Scimone ML et al (2003) Efficient thymic immigration of B220 + lymphoid-restricted bone marrow cells with T precursor potential. Nat Immunol 4:866–873
Martins VC, Ruggiero E, Schlenner SM et al (2012) Thymus-autonomous T cell development in the absence of progenitor import. J Exp Med 209:1409–1417
Martins VC, Busch K, Juraeva D et al (2014) Cell competition is a tumour suppressor mechanism in the thymus. Nature 509:465–470
Mori S, Shortman K, Wu L (2001) Characterization of thymus-seeding precursor cells from mouse bone marrow. Blood 98:696–704
Peaudecerf L, Lemos S, Galgano A et al (2012) Thymocytes may persist and differentiate without any input from bone marrow progenitors. J Exp Med 209:1401–1408
Porritt HE, Gordon KM, Petrie HT (2003) Kinetics of steady-state differentiation and mapping of intrathymic-signaling environments by stem cell transplantation in nonirradiated mice. J Exp Med 198:957–962
Prockop SE, Petrie HT (2004) Regulation of thymus size by competition for stromal niches among early T cell progenitors. J Immunol 173:1604–1611
Rossi FMV, Corbel SY, Merzaban JS et al (2005) Recruitment of adult thymic progenitors is regulated by P-selectin and its ligand PSGL-1. Nat Immunol 6:626–634
Saran N, Łyszkiewicz M, Pommerencke J et al (2010) Multiple extrathymic precursors contribute to T-cell development with different kinetics. Blood 115:1137–1144
Schlenner SM, Rodewald HR (2010) Early T cell development and the pitfalls of potential. Trends Immunol 31:303–310
Schlenner SM, Madan V, Busch K et al (2010) Fate mapping reveals separate origins of T cells and myeloid lineages in the thymus. Immunity 32:426–436
Schmitt TM, Zuniga-Pflucker JC (2002) Induction of T cell development from hematopoietic progenitor cells by delta-like-1 in vitro. Immunity 17:749–756
Schwarz B, Bhandoola A (2004) Circulating hematopoietic progenitors with T lineage potential. Nat Immunol 5:953–960
Schwarz BA, Sambandam A, Maillard I et al (2007) Selective thymus settling regulated by cytokine and chemokine receptors. J Immunol 178:2008–2017
Scimone ML, Aifantis I, Apostolou I et al (2006) A multistep adhesion cascade for lymphoid progenitor cell homing to the thymus. Proc Natl Acad Sci USA 103:7006–7011
Scollay R, Smith J, Stauffer V (1986) Dynamics of early T cells: prothymocyte migration and proliferation in the adult mouse thymus. Immunol Rev 91:129–157
Serwold T, Ehrlich LIR, Weissman IL (2009) Reductive isolation from bone marrow and blood implicates common lymphoid progenitors as the major source of thymopoiesis. Blood 113:807–815
Shi Y, Wu W, Chai Q et al (2016) LTβR controls thymic portal endothelial cells for haematopoietic progenitor cell homing and T-cell regeneration. Nat Commun 7:12369
Shultz LD, Brehm MA, Garcia-Martinez JV et al (2012) Humanized mice for immune system investigation: progress, promise and challenges. Nat Rev Immunol 12:786–798
Six EM, Bonhomme D, Monteiro M et al (2007) A human postnatal lymphoid progenitor capable of circulating and seeding the thymus. J Exp Med 204:3085–3093
Svensson M, Marsal J, Uronen-Hansson H et al (2008) Involvement of CCR9 at multiple stages of adult T lymphopoiesis. J Leukoc Biol 83:156–164
Uehara S, Grinberg A, Farber JM et al (2002) A role for CCR9 in T lymphocyte development and migration. J Immunol 168:2811–2819
Umland O, Mwangi WN, Anderson BM et al (2007) The blood contains multiple distinct progenitor populations with clonogenic B and T lineage potential. J Immunol 178:4147–4152
Wallis VJ, Leuchars E, Chwalinski S, Davies AJ (1975) On the sparse seeding of bone marrow and thymus in radiation chimaeras. Transplantation 19:2–11
Wu L, Kincade PW, Shortman K (1993) The CD44 expressed on the earliest intrathymic precursor population functions as a thymus homing molecule but does not bind to hyaluronate. Immunol Lett 38:69–75
Zhang SL, Wang X, Manna S et al (2014) Chemokine treatment rescues profound T-lineage progenitor homing defect after bone marrow transplant conditioning in mice. Blood 124:296–304
Zietara N, Łyszkiewicz M, Puchałka J et al (2015) Multicongenic fate mapping quantification of dynamics of thymus colonization. J Exp Med 212:1589–1601
Zlotoff DA, Sambandam A, Logan TD et al (2010) CCR7 and CCR9 together recruit hematopoietic progenitors to the adult thymus. Blood 115:1897–1905
Zlotoff DA, Zhang SL, De Obaldia ME et al (2011) Delivery of progenitors to the thymus limits T-lineage reconstitution after bone marrow transplantation. Blood 118:1962–1970
Acknowledgements
This work was supported by grants from the German Research Foundation (Deutsche Forschungsgemeinschaft) (KR2320/3-1, KR2320/5-1, SFB902-B15, and EXC62 “REBIRTH”).
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Krueger, A. Thymus Colonization: Who, How, How Many?. Arch. Immunol. Ther. Exp. 66, 81–88 (2018). https://doi.org/10.1007/s00005-017-0503-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00005-017-0503-5