Abstract
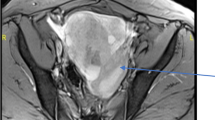
Struma ovarii (SO), a rare tumor containing at least 50% of thyroid tissue, represents approximately 5% of all ovarian teratomas; its malignant transformation rate is reported to occur in up to 10% of cases and metastases occur in about 5–6% of them. We describe a 36-year old woman who underwent laparoscopic left annessectomy two years earlier because of an ovarian cyst. Follow-up imaging revealed a right adnexal mass, ascitis and peritoneal nodes that were diagnosed as comprising a malignant SO with peritoneal secondary localizations at histopathology performed after intervention. Restaging with 18F-FDG-PET/CT scan, abdominal CT and ultrasonography showed abnormalities in the perihepatic region and presacral space and left hypochondrium localizations. The patient underwent thyroidectomy, hepatic nodulectomy and cytoreductive peritonectomy: histopathological examination did not show any malignant disease in the thyroid and confirmed the presence of peritoneal localizations due to malignant SO; molecular analysis detected NRAS Q61K mutation in exon 3, whereas no mutations were identified on the BRAF gene. The patient underwent radioiodine treatment: serum Tg was decreased at first follow-up after three months of 131I-therapy. We believe that our case raises some interesting considerations. First, pathologists should be aware of this entity and should check for the presence of point mutations suggesting an aggressive disease behavior, which could be beneficial for an optimal therapeutic approach. Second, although most of the knowledge in this field comes from case reports, efforts should be made to standardize the management of patients affected by malignant SO, including use of practice guidelines.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Roth LM, Talerman A, 2007 The enigma of struma ovarii. Pathology 39: 139–146.
Pick L, 1903 Beitrag zur Lehre von den Greschwülsten über struma thyroidaea ovarii aberrata. Verhandlungen der Berliner Medizinischen Geselschaft 33: 139–146.
Rosenblum NG, LiVolsi VA, Edmonds PR, Mikuta JJ, 1989 Malignant struma ovarii. Gynecol Oncol 32: 224–227.
Volpi E, Ferrero A, Nasi PG, Sismondi P, 2003 Malignant struma ovarii: a case report of laparoscopic management. Gynecol Oncol 90: 191–194.
Dardik RB, Dardik M, Westra W, Montz FJ, 1999 Malignant struma ovarii: two case reports and a review of the literature. Gynecol Oncol 73: 447–451.
Yassa L, Sadow P, Marqusee E, 2008 Malignant struma ovarii. Nat Clin Pract Endocrinol Metab 4: 469–472.
Goffredo P, Sawka AM, Pura J, Adam MA, Roman SA, Sosa JA, 2015 Malignant struma ovarii: a population-level analysis of a large series of 68 patients. Thyroid 25: 211–215.
Yoo SC, Chang KH, Lyu MO, Chang SJ, Ryu HS, Kim HS, 2008 Clinical characteristics of struma ovarii. J Gynecol Oncol 19: 135–138.
Matsuda K, Maehama T, Kanazawa K, 2001 Malignant struma ovarii with thyrotoxicosis. Gynecol Oncol 82: 575–577.
Ciccarelli A, Valdes-Socin H, Parma J, et al, 2004 Thyrotoxic adenoma followed by atypical hyperthyroidism due to struma ovarii: clinical and genetic studies. Eur J Endocrinol 150: 431–437.
De Simone CP, Lele SM, Modesitt SC, 2003 Malignant struma ovarii: a case report and analysis of cases reported in the literature with focus on survival and I-131 therapy. Gynecol Oncol 89: 543–548.
Dane C, Ekmez M, Karaca A, Ak A, Dane B, 2012 Follicular variant of papillary thyroid carcinoma arising from a dermoid cyst: a rare malignancy in young women and review of the literature. Taiwan J Obstet Gynecol 51: 421–425.
Uzum AK, Iyibozkurt C, Canbaz B, et al, 2013 Management and follow-up results of an incidental thyroid carcinoma in a young woman with ovarian teratoma. Gynecol Endocrinol 29: 724–726.
Wolff EF, Hughes M, Merino MJ, et al, 2010 Expression of benign and malignant thyroid tissue in ovarian teratomas and the importance of multimodal management as illustrated by a BRAF-positive follicular variant of papillary thyroid cancer. Thyroid 20: 981–987.
Zhu Y, Wang C, Zhang G-N, et al, 2016 Papillary thyroid cancer located in malignant struma ovarii with omentum metastasis: a case report and review of the literature. World J Surg Oncol 14: 17.
Makani S, Kim W, Gaba AR, 2004 Struma Ovarii with a focus of papillary thyroid cancer: a case report and review of the literature. Gynecol Oncol 94: 835–839.
Robboy S J, Shaco-Levy R, Peng RY, et al, 2009 Malignant struma ovarii: an analysis of 88 cases, including 27 with extraovarian spread. Int J Gynecol Pathol 28: 405–422.
Marti JL, Clark VE, Harper H, Chhieng DC, Sosa JA, Roman SA, 2012 Optimal surgical management of well-differentiated thyroid cancer arising in struma ovarii: a series of 4 patients and a review of 53 reported cases. Thyroid 22: 400–406.
Roth LM, Miller AW 3rd, Talerman A, 2008 Typical thyroid-type carcinoma arising in struma ovarii: a report of 4 cases and review of the literature. Int J Gynecol Pathol 27: 496–506.
Elisei R, Romei C, Castagna MG, et al, 2005 RET/PTC3 rearrangement and thyroid differentiation gene analysis in a struma ovarii fortuitously revealed by elevated serum thyroglobulin concentration. Thyroid 15: 1355–1361.
Flavin R, Smyth P, Crotty P, et al, 2007 BRAF T1799A mutation occurring in a case of malignant struma ovarii. Int J Surg Pathol 15: 116–120.
Schmidt J, Derr V, Heinrich MC, et al, 2007 BRAF in papillary thyroid carcinoma of ovary (struma ovarii). Am J Surg Pathol 31: 1337–1343.
Coyne C, Nikiforov YE, 2010 RAS mutation-positive follicular variant of papillary thyroid carcinoma arising in a struma ovarii. Endocr Pathol 21: 144–147.
Celestino R, Magalhães J, Castro P, et al, 2009 A follicular variant of papillary thyroid carcinoma in struma ovarii. Case report with unique molecular alterations. Histopathology 55: 482–487.
Boutross-Tadross O, Saleh R, Asa SL, 2007 Follicular variant papillary thyroid carcinoma arising in struma ovarii. Endocr Pathol 18: 182–186.
Tate G, Tajiri T, Suzuki T, Mitsuya T, 2009 Mutations of the KIT gene and loss of heterozygosity of the PTEN region in a primary malignant melanoma arising from a mature cystic teratoma of the ovary. Cancer Genet Cytogenet 190: 15–20.
Melo M, Gaspar da Rocha A, Batista R, et al, 2017 TERT, BRAF, and NRAS in Primary Thyroid Cancer and Metastatic Disease. J Clin Endocrinol Metab 102: 1898–1907.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gobitti, C., Sindoni, A., Bampo, C. et al. Malignant struma ovarii harboring a unique NRAS mutation: case report and review of the literature. Hormones 16, 322–327 (2017). https://doi.org/10.1007/BF03401528
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03401528