Abstract
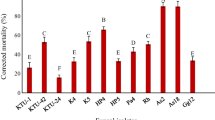
The application of entomopathogenic fungi against weevils of the genus Otiorhynchus spp. might represent an environmentally friendly management option for biological control of these insects, which have recently increased in their importance as pests in a variety of horticultural plants. As only little information is available on the susceptibility of different weevils of this genus towards various fungal species, laboratory experiments were conducted to evaluate the control potential of three different commercially available entomopathogenic fungi (Beauveria bassiana, Isaria fumosorosea, Metarhizium anisopliae) against adults of O. sulcatus, O. raucus, O. dieckmanni, O. rugosostriatus and O. crataegi. Mortality rates were dependent on fungal and weevil species, respectively. Application of Naturalis® containing B. bassiana as the active ingredient caused a significantly higher mortality rate than evident in control groups in four of the five Otiorhynchus species assessed. Adults of O. rugosostriatus were apparently not susceptible to any of the fungi tested. Efficacy of a single application of Naturalis® against adult O. sulcatus was also assessed in three independent semi-field experiments. Weevils got infected during the first 14 days after application, but mortality rates did not substantially increase until 2 months after application. Even 6 months after application, mortality of weevils was caused by the respective applied B. bassiana strain, which was confirmed via an amplification of isolate-specific microsatellite markers. Thus, a single application of a B. bassiana-based product in the field could have long-term effects on population densities of respective weevils.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Abbott WS, 1925. A method of computing the effectiveness of an insecticide. J Econ Entomol 18, 265–267.
Ansari MA & Butt TM, 2013. Influence of the application methods and doses on the susceptibility of black vine weevil larvae Otiorhynchus sulcatus to Metarhizium anisopliae in field-grown strawberries. BioControl 58, 257–267.
Ansari MA, Shah FA & Butt TM, 2008. Combined use of entomopathogenic nematodes and Metarhizium anisopliae as a new approach for black vine weevil, Otiorhynchus sulcatus, control. Entomol Exp Appl 129, 340–347.
Bouchard P, Lesage L, Goulet H, Bostanian NJ, Vincent C, Zmudzinska A & Lasnier J, 2005. Weevil (Coleoptera: Curculionoidea) diversity and abundance in two Quebec vineyards. Ann Entomol Soc Am 98, 565–574.
Bruck DJ, 2007. Efficacy of Metarhizium anisopliae as a curative application for black vine weevil, (Otiorhynchus sulcatus) infesting container-grown nursery crops. J Environ Hort 25, 15–156.
Bruck DJ & Donahue KM, 2007. Persistence of Metarhizium anisopliae incorporated into soilless potting media for control of the black vine weevil, Otiorhynchus sulcatus in container-grown ornamentals. J Invertebr Pathol 95, 146–150.
Cowles RS, 1995. Black Vine Weevil biology and management. J Am Rhod Soc 49, 83–97.
Cowles RS, 2001. Protecting container-grown crops from black vine weevil larvae with bifenthrin. J Environ Hort 19, 184–189.
Dubovskiy IM, Whitten MMA, Yaroslavtseva ON, Greig C, Kryukov VY, Grizanova EV, Mukherjee K, Vilcinskas A, Glupov VV & Butt TM, 2013. Can insects develop resistance to insect pathogenic fungi? PLoS One 8.
Enkerli J, Widmer F & Keller S, 2004. Long-term field persistence of Beauveria brongniartii strains applied as biocontrol agents against European cockchafer larvae in Switzerland. Biol Control 29, 115–123.
Enriquez-Vara JN, Cordoba-Aguilar A, Guzman-Franco AW, Alatorre-Rosas R & Contreras-Garduno J, 2012. Is survival after pathogen exposure explained by host’s immune strength? A test with two species of White Grubs (Coleoptera: Scarabaeidae) exposed to fungal infection. Environ Entomol 41, 959–965.
Hajek A & Delalibera I, 2010. Fungal pathogens as classical biological control agents against arthropods. BioControl 55, 147–158.
Haukeland S & Lola-Luz T, 2010. Efficacy of the entomopathogenic nematodes Steinernema kraussei and Heterorhabditis megidis against the black vine weevil Otiorhynchus sulcatus in open field-grown strawberry plants. Agric Forest Entomol 12, 363–369.
Hirsch J, Sprick P & Reineke A, 2010. Molecular identification of larval stages of Otiorhynchus (Coleoptera: Curculionidae) species based on PCR-RFLP analysis. J Econ Entomol 103, 898–907.
Hirsch J, Strohmeier S, Pfannkuchen M & Reineke A, 2012. Assessment of bacterial endosymbiont diversity in Otio rhynchus spp. (Coleoptera: Curculionidae) larvae using a multitag 454 pyrosequencing approach. BMC Microbiol 12, S6.
Jackson M, Dunlap C & Jaronski S, 2010. Ecological considerations in producing and formulating fungal entomopathogens for use in insect biocontrol. BioControl 55, 129–145.
Kakouli-Duarte T, Labuschagne L & Hague NGM, 1997. Biological control of the black vine weevil, Otiorhynchus sulcatus (Coleoptera: Curculionidae) with entomopathogenic nematodes (Nematoda: Rhabditida). Ann Appl Biol 131, 11–27.
Kepler RM & Bruck DJ, 2006. Examination of the interaction between the black vine weevil (Coleoptera: Curculionidae) and an entomopathogenic fungus reveals a new tritrophic interaction. Environ Entomol 35, 1021–1029.
Kraaijeveld AR & Godfray HCJ, 2008. Selection for resistance to a fungal pathogen in Drosophila melanogaster. Heredity 100, 400–406.
Łukasik P, van Asch M, Guo H, Ferrari J & Godfray HCJ, 2013. Unrelated facultative endosymbionts protect aphids against a fungal pathogen. Ecol Lett 16, 214–218.
Lundmark M, 2010. Otiorhynchus sulcatus, an autopolyploid general-purpose genotype species? Hereditas 147, 278–282.
Lykouressis DP, Perdikis DC & Drilias AG, 2004. Temporal dynamics of Otiorhynchus schlaeflini (Coleoptera: Curculionidae) adults and damage assessment on two wine grape cultivars. J Econ Entomol 97, 59–66.
Majka CG & MacIvor JS, 2009. Otiorhynchus porcatus (Coleoptera: Curculionidae): a European root weevil newly discovered in the Canadian Maritime Provinces. J Acad Entomol Soc 5, 27–31.
Moorhouse ER, Charnley AK & Gillespie AT, 1992. A review of the biology and control of the vine weevil, Otiorhynchus sulcatus (Coleoptera: Curculionidae). Ann Appl Biol 121, 431–454.
Nankinga CM & Moore D, 2000. Reduction of banana weevil populations using different formulations of the entomopathogenic fungus Beauveria bassiana. Biocontrol Sci Technol 10, 645–657.
Parker BJ, Spragg CJ, Altincicek B & Gerardo NM, 2013. Symbiont-mediated protection against fungal pathogens in pea aphids: a role for pathogen specificity? Appl Environ Microbiol 79, 2455–2458.
Reding ME & Persad AB, 2009. Systemic insecticides for control of black vine weevil (Coleoptera: Curculionidae) in container- and field-grown nursery crops. J Econ Entomol 102, 927–933.
Reding ME & Ranger CM, 2011. Systemic insecticides reduce feeding, survival, and fecundity of adult Black Vine Weevils (Coleoptera: Curculionidae) on a variety of ornamental nursery crops. J Econ Entomol 104, 405–413.
Rehner SA & Buckley EP, 2003. Isolation and characterization of microsatellite loci from the entomopathogenic fungus Beauveria bassiana (Ascomycota: Hypocreales). Mol Ecol Notes 3, 409–411.
Reineke A, Bischoff-Schaefer M, Rondot Y, Galidevara S, Hirsch J & Uma Devi K, 2014. Microsatellite markers to monitor a commercialized isolate of the entomopatho-genic fungus Beauveria bassiana in different environments: Technical validation and first applications. Biol Control 70, 1–8.
Reineke A, Hirsch J & Kubach G, 2011. Aggregation, abundance and dispersal capabilities of Otiorhynchus rugosostriatus Goeze and Otiorhynchus raucus Fabricius (Coleoptera: Curculionidae) in plantations of ornamental plants. J Pest Sci 84, 297–302.
Sabbahi R, Merzouki A & Guertin C, 2008. Efficacy of Beauveria bassiana against the strawberry pests, Lygus lineolaris, Anthonomus signatus and Otiorhynchus ovatus. J Appl Entomol 132, 151–160.
Scheepmaker JWA & Butt TM, 2010. Natural and released inoculum levels of entomopathogenic fungal biocontrol agents in soil in relation to risk assessment and in accordance with EU regulations. Biocontrol Sci Technol 20, 503–552.
Shah FA, Ansari MA, Prasad M & Butt TM, 2007. Evaluation of black vine weevil (Otiorhynchus sulcatus) control strategies using Metarhizium anisopliae with sublethal doses of insecticides in disparate horticultural growing media. Biol Control 40, 246–252.
Sprick P & Stüben PE, 2012. Rüsselkäfer in anthropogenen Lebensräumen — Studies on taxonomy, biology and ecology of Curculionidae. Snudebillerextra. Curculio-Institute, Mönchengladbach, Germany.
Tinsley MC, Blanford S & Jiggins FM, 2006. Genetic variation in Drosophila melanogaster pathogen susceptibility. Parasitology 132, 767–773.
Vainio A & Hokkanen HMT, 1993. The potential of entomopathogenic fungi and nematodes against Otiorhynchus ovatus L. and O. dubius Ström (Col., Curculionidae) in the field. J Appl Entomol 115, 379–387.
Vanninen I, Tyni-Juslin J & Hokkanen H, 2000. Persistence of augmented Metarhizium anisopliae and Beauveria bassiana in Finnish agricultural soils. BioControl 45, 201–222.
Wang YD, Yang PC, Cui F & Kang L, 2013. Altered immunity in crowded locust reduced fungal (Metarhizium anisopliae) pathogenesis. PLoS Pathog 9.
Wheeler AG, 1999. Otiorhynchus ovatus, O. rugosostriatus, and O. sulcatus (Coleoptera: Curculionidae): Exotic weevils in natural communities, mainly mid-Appalachian shale barrens and outcrops. Proc Entomol Soc Wash 101, 689–692.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hirsch, J., Reineke, A. Efficiency of commercial entomopathogenic fungal species against different members of the genus Otiorhynchus (Coleoptera: Curculionidae) under laboratory and semi-field conditions. J Plant Dis Prot 121, 211–218 (2014). https://doi.org/10.1007/BF03356513
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03356513