Abstract
The world’s population has been increasing at a tremendous rate causing an increase in the demand for food. Seafood is a major source utilized by almost 3 billion people worldwide. With this massive consumption of seafood, waste generation is an increasing risk to the environment; both capture fisheries and aquacultures have been generating increasing amounts of waste over the past few years. The current objective of most countries is to enforce strict regulations to monitor the amount of waste generated and pursuit of appropriate storage, utilization, and disposal techniques. Traditional disposal techniques like landfills and ocean dumping are an easy form of discarding waste, but they have been hugely criticized by environmentalists, researchers, and governments because of leaching and an increase in biochemical oxygen demand and chemical oxygen demand in the water. This has led to the development of new techniques for waste utilization and improvements to the older disposal techniques. Previously, fish remnants were utilized to generate products such as fishmeal, fish oils, and isinglass while lately researchers have been attempting to isolate various bioactive compounds. These have found exponential use in the pharmaceutical and nutraceutical industries due to their high potential as natural medicines.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
11.1 Introduction
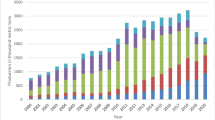
Fish is consumed as a good source of protein and other vital nutrients by millions of people around the globe. Consumption of fish over the years has increased dramatically such that more than 3.2 billion people relied on fish for over 20% of their protein requirement during the year 2016 (Zhou 2017). According to past 20 years data, the maximum production of fisheries is found from coastal regions like China, Indonesia, and India (as mentioned in Fig. 11.1). India has seen a massive jump in the amount of seafood being consumed especially between the years 1990 and 2010. To cope with the increasing demand for fish and other seafood, the farming of marine organisms has boomed over the past few decades (Rao 2010). The increase in the consumption of seafood has resulted in the generation of high amounts of waste; more than 27% of the captured fish are either thrown away or rot before they can be sold (Ryder 2018; Kruijssen et al. 2020). Whole fish and shellfish are rarely consumed; most people only consume the flesh (40%), while the discard bones, shells is roughly 60% of total weight (Agustin et al. 2021). India being one of the largest fish-producing nations, generates 2 metric million tonnes of fish waste annually according to a survey conducted. The data on fish waste generation in the major coastal cities of India showed that Mumbai (22.60%) is the largest contributor followed by Chennai (15.10%) and Goa (12.50%) (Ahmad and Bhuimbar 2019). The improper discard of these wastes causes pollution to the surrounding environment and disrupts the coastal ecosystem. The waste management process can be best optimized by proper disposal or finding a specific utility for it. For finding an economically valuable purpose of the byproducts, it is very important to know the source of fish waste production (Boziaris 2014; Munari et al. 2016).
11.2 Sources of Fish Waste Production
11.2.1 Aquaculture and Fisheries
The fisheries industry is dependent on two major forms of fishing: (i) capture fisheries and (ii) aquaculture. Capture fisheries consist of the traditional form of catching fish in the wild while aquaculture is an advanced technique which involves building enclosures for the fish, crustaceans, mollusks, and aqueous plants in the water bodies. The fish in these enclosures are given high protein and other nutrient feeds which enhances their growth rate (Ryder 2018). However, this comes with a consequence of impact on its surrounding environment, when an attempt to produce a higher number of fish per unit area is made. The attempt for increasing profits must be made in accordance with the amount of waste that can be managed to make the process sustainable. Thus, the fish must be fed according to its size, digestibility, excretion, and other health and physiological factors to ensure an optimal growth process (Bowyer et al. 2012).
The fish which are unable to survive the culture process accumulate as solid waste or suspended solid waste. This accumulation could alter the physiochemical balance of the aquaculture including biological oxygen demand (BOD) and chemical oxygen demand, which would further degrade the habitability of the aquaculture. Due to the high BOD of the fish waste, it is imperative to perform timely rectification to avoid health hazards in the aquacultures and oceans (Dauda et al. 2019). The environment protection authority (EPA) in Victoria, Seychelles has tested fish wastes and has declared them safe for biological decomposition under controlled conditions. They can be used for land applications after being subjected to appropriate treatments, irrespective of their shape and size as they fall under prescribed industrial waste. Thus, these waste materials can be processed in a designated composting facility after acquiring EPA Works Approval and License. The composting facility is built in proximity to the aquaculture if it has optimal leachate, odor, moisture, and surface run-off conditions (Venugopal 2021).
11.2.2 Seafood Industry
The seafood industry usually discards inedible components like skin, fins, bones, and scales as waste. These components however can be used for the production of value-added products. Organs like liver, gills, and kidney are highly susceptible to contamination by toxins like tetrodotoxin (TTX) and conotoxin, a powerful neurotoxin, which makes them unsuitable for consumption and are thus usually removed from seafood to avoid seafood poisoning (Saravanan et al. 2009; Lago et al. 2015; Schneider et al. 2018). Consumption of crustaceans such as crab, shrimp, and lobster gives rise to an enormous amount of shell waste (Joshi et al. 2020). It has been estimated that annually about 6–8 million tons of shell waste are being produced in the world out of which, Southeast Asia itself accounts for about 1.5 million tonnes, which is an alarming amount of waste (Yan and Chen 2015). Despite having high amounts of calcium, carbohydrates, and proteins, they are not considered to be of much value and are sold. Currently, these wastes are being utilized for the production of microparticles used for various applications including anti-inflammatory drugs, chitin production, and auxiliary fertilizer or as a component of animal feed. A large amount of research is being carried out to value chitosan as a natural biopolymer and its potential to be part of high-quality bioplastic (Hamed et al. 2016).
11.2.3 Fishing Harbors
Fishing harbors are one of the highest accumulators of fish waste for two main reasons: (i) during transportation of fish from harbor to market, fish and other marine organisms are accidentally dropped, resulting in random displacement of fish and fish remnants all around the harbor; (ii) the damaged fish are thrown either back to the ocean or in the harbor shore (Balde et al. 2020). The vendors are also known to sell only the edible part of the fish to the customers, and throw the inedible parts in their vicinity which can lead to the accumulation of fish offal and blood water waste (Fig. 11.2). This can produce an unpleasant sight and odor, degrading the esthetic value of the place. Mostly, the damaged fish are collected for animal feed. But, due to high yield per day, fish waste continues to be dumped in the harbor area, though disposal of that high level of waste is a difficult task (Read and Fernandes 2003).
11.2.4 Industrial Fish-Based Processes
Many industries using fish and other marine organisms generate huge amounts of semi-processed marine waste. This includes fish and shrimp processing units subjected to chemical treatments like calcium hypochlorite, or cleaning using soaps and detergents (Mathew et al. 2021). Each part is processed separately according to its specific needs for the products they are capable of synthesizing. The carcass of the fish is rich in meat, processing of which is first started by the removal of swim bladder, which has no significant contribution to human diet. Other parts like skin, bones, etc., are separated from the edible parts and are processed separately for production of leather and other valuable products. As a result, a huge amount of waste is generated when only the essential parts of the fish and other marine organisms are used (Sasidharan and Mathew 2011; Willis et al. 2018). The fish and shrimp processing units generate wastes which are released in nearby water bodies creating an imbalance in various parameters like BOD, suspended solids, etc., and thus polluting it and making it unfit for use (Dauda et al. 2019).
11.3 Classification of Fish Waste
Marine wastes can be categorized as solid and liquid waste; both solid and liquid wastes have their respective problems and disposal issues. The following flowchart (Fig. 11.3) explains the classification of marine waste along with their method of treatment/disposal. Aquaculture waste is categorized into three categories 1, 2, and 3 ranging from the highest to lowest risk, respectively. Waste disposal is carried out under strict regulations to prevent any health hazard. Category 3 consists of the fish that are caught for the sole purpose of being converted to fish meal or other by-products (Sharp and Mariojouls 2012). These are of least harm to the environment, but still require licensed facilities containing the proper apparatus for disposal. Mortalities that are not caused by intentional slaughter for human consumption are classified as Category 2, these should be disposed of through legal disposal routes. Fish and other marine organisms affected by communicable diseases belong to Category 1, posing the highest risk and require appropriate disposal methods (Dauda et al. 2019; Miller and Semmens 2002). Table 11.1 describes all the categories of aquaculture/fishing waste:
11.4 Waste Treatment
11.4.1 Primary Waste Treatment
The main objective of primary treatment is to remove the large sized solid particles that either float on the surface or settle at the bottom. It involves treatment of the wastewater using three basic physical processes. (i) Screening: Screening involves passing the liquid through a sieve-like structure, through which solid particles are trapped in the sieve, while the liquid flows out. The size of the sieve can be changed depending on the size of particles present in the liquid. (ii) Sedimentation: Sedimentation is the tendency of particles to settle in a solution. Particles settle due to the difference between their density and that of the solution. Higher density particles settle while lower density ones remain afloat. This concept is very useful in the removal of solid particles present in a liquid solution. (iii) Flotation: As opposed to sedimentation, during flotation particles float over the liquid surface (Fig. 11.4). These suspended or lower density particles can be collected and removed (Cristóvão et al. 2014).
11.4.2 Secondary Waste Treatment
It involves the use of microbes for the degradation of wastewater. This is done through the usage of aerobic as well as anaerobic microbes for efficient treatment of the waste. Aerobic microbes digest the organic matter in the presence of oxygen to release carbon dioxide along with biomass, while anaerobic microbes produce methane, carbon dioxide, and water. Following this, the physicochemical process (Coagulation-flocculation) utilizes substances known as flocculants/coagulants to cause aggregation of the particulate matter to form “flocs” which then settle down (Mack et al. 2004).
11.5 Fish Waste Disposal
The importance of organized fish waste management is increasing due to concern over health and sanitation issues. Fish waste which is disposed and circulated improperly can lower the oxygen levels in water bodies and as water decomposes it leads to foul odor. Small-scale fish industries produce around 20–80% of fish waste (Islam et al. 2004; Arvanitoyannis and Tserkezou 2014).
11.5.1 Fish Disposal Treatment Methods
They are classified mainly into two different types: traditional fish waste disposal and anaerobic digestion as illustrated in Table 11.2. Traditional methods are the conventional methods which are used to dispose of fish waste (Arvanitoyannis and Kassaveti 2008; Green and Mattick 1977). These methods have major drawbacks and are not commonly used.
11.5.2 Anaerobic Treatment
Fish canning industries produce a large amount of anaerobically digested solid waste. Most of this solid waste can be used to produce methane in reasonable amounts. Research has shown that anaerobic digestion and co-digestion of fish waste has significant potential for bio-methane production (van Rijn et al. 1995; Nges et al. 2012). Fish waste has seen very limited use as substrate in anaerobic digestion due to two major issues: (i) variable composition (up to 60% protein, 20% fat, calcium and hydroxyapatite from bones and scale, palmitic acid, mono-saturated acids); (ii) release of high quantities of ammonia as a result of fish waste digestion inhibits digestion of the substrate and can also result in the accumulation of volatile fatty acids. In contrast, co-digestion has been found to be a suitable option to overcome these issues by increasing the organic content of the substrate through the addition of another substrate, such as cow manure, water hyacinth, and sisal pulp (Boziaris 2014; Mshandete et al. 2004).
11.6 Utilization of Marine Waste
As previously stated, the incorrect disposal of marine waste leads to many disagreeable circumstances. Thus, there is a need for novel techniques for the utilization of waste to produce useful products (Fig. 11.5). Utilization of marine waste can be of two broad categories: (i) for the production of nutritional supplements or consumable products, and (ii) non-nutritional uses (Plazzotta and Manzocco 2019; Radziemska et al. 2019).
11.6.1 Nutritional Supplements or Consumable Products
11.6.1.1 Fishmeal and Fish Oils
Fishmeal is commercially used nutritive feed for aquaculture and farm animals. It is a powdered product produced from fish waste or whole fish of any species through drying and grinding which are generally not used for human consumption. When combined with fish oil, it is a rich source of protein, vitamins, and essential oils (Han 2015). The production of fishmeal is initiated by pressing cooked fish waste or fish, yielding a cake and the removed liquid. This cake along with any remaining solids, which can be separated from the removed liquid, is dried to produce the fishmeal. This liquid is then subjected to centrifugation to separate the oil from water (Fig. 11.6). The oil thus obtained can be further processed and sold commercially as fish oil (Das et al. 2011; Mo et al. 2018).
Fish oils are of two types: Oil present in the muscle of the fish and the oil present in the visceral mass of the fish including the liver. It is also possible to extract fish oils from the heads, fins, and scales in fish waste (Aidos et al. 2001). During fishmeal production, the crude oil obtained consists of both muscle and visceral oil components which are then purified and used in aquacultures or for bulk production of products such as margarine, ink, and rubber. They have also been used in the treatment of hide in leather production. Products such as cod liver or shark liver oils are most commonly used due to their high content of omega-3 fatty acids, Vitamins D and A (Bockisch 2015). Crude oil obtained during fishmeal production is washed and centrifuged for usage in animal feed followed by purification if it is intended for human consumption. The process begins with alkaline refining to prevent the rancidity of the oil during storage, followed by the removal of the clear layer formed upon settling of the mixture. Following this, bleaching of the oil is carried out to remove undesirable fatty acids and pigments. Hydrogenation is then carried out using hydrogen in the presence of a catalyst (Fig. 11.7). In succession, vacuum steam distillation is carried out to remove more volatile compounds and stabilize the flavor of the oil (Archer 2001).
11.6.1.2 Silage
Fish silage is a liquid product used in animal feed similar to fishmeal due to its high nutritious value. It can be produced from any type of fish or from fish waste through low value, relatively inexpensive fish that are generally used for the production of silage. Silage can be produced through the enzymatic digestion of fish waste or through the addition of acid for degradation (Ke et al. 2017). Production of fish silage by acid digestion consists of four main stages. The first stage involves the rupture of cells to release intracellular enzymes through mincing of the fish. This is followed by acidification of the sample using 3.5% Formic acid, though mineral acids can be used as cheaper alternatives. This addition of acid initiates the production of the silage which is enhanced by intermittent mixing (Fig. 11.8). The last two stages of the process are removal of oil from the silage through heating followed by centrifugation (Arruda et al. 2007).
Silage production through enzymatic digestion involves the addition of an inexpensive carbohydrate source and lactobacilli to the fish waste. The bacteria cause acidification of the system by converting the sugars to lactic acid while simultaneously producing additional products associated with the deterrence of spoilage. This mixture is then incubated at 25–30 °C and the oil is removed through the same processes as silage produced by acid digestion. The major drawback of silage is the inconvenience faced during its transport (Ghaly et al. 2013; Olsen and Toppe 2017).
11.6.1.3 Fish Protein Hydrolysate (FPH)
FPH is a powder produced by proteolytic digestion of fish to yield amino acids of numerous lengths as mentioned in Fig. 11.9. It is used in animal feed or as an emulsifying agent due to its high gelling and whipping properties (Kristinsson and Rasco 2000). The production of FPH is either through an autolytic process or through the addition of external proteolytic enzymes, i.e., accelerated hydrolysis (Balde et al. 2021). While the autolytic process depends on the enzymes of the fish itself, accelerated hydrolysis is generally preferred as there is no interfering enzyme activity. The autolytic process is time-consuming and involves the breakdown of peptides over a period of time ranging between a few days to a few months. Preservative agents such as organic acids or salts are added to prevent spoilage (Rustad and Hayes 2012; Yoshida et al. 1999, 2003). FPH production using external enzymes involves the mincing of the fish waste followed by agitation with the addition of a commercial protease (Rustad et al. 2011). Optimum conditions for the protease activity are then maintained through the addition of an acid or alkali and maintenance of temperature. The resultant broth is then filtered and spray-dried for commercialization (Anal et al. 2013). FPH may also be utilized further for the production of bioactive peptides and proteins showing various biological activities (Narayanasamy et al. 2020).
11.6.1.4 Fish Protein Concentrate (FPC)
FPC is a powder similar to FPH which is intended for human consumption. It is often used to increase the protein content of food or even as a food product in itself (Ahmad et al. 2019). The FPC is of three grades: Type A, B, and C. Type A FPC is used in a variety of edible products such as biscuits, bread, and soups while Type B is used as a nutritional supplement in some Asian countries. Both are manufactured through a multistage solvent extraction method followed by the removal of fats and odor components. Type C FPC is produced through a more hygienic fishmeal process (Archer 2001).
11.6.2 Non-Nutritional Uses
11.6.2.1 Collagen
Collagen is the major structural protein in the extracellular matrix found abundantly in skin, bones, and fins of fish. It is used by the food, cosmetics, biomedical, and pharmaceutical industries as well as being considered for a novel wound dressing material in the form of films (Govindharaj et al. 2019). Isolation of collagen is in three general stages: Sample preparation, Extraction, and Recovery. The fish waste is prepared by cleaning and pretreatment using an alkali-like sodium hydroxide as described in Fig. 11.10. Extraction is achieved by agitation with an acid to give acid-soluble collagen (ASC). Pepsin is also used to improve the yield of collagen; this is pepsin soluble collagen (PSC) (Sampath Kumar and Nazeer 2013). Recovery of the extracted collagen is through salt precipitation or salting out of the protein (Kumar et al. 2012; Kim and Mendis 2006).
11.6.2.2 Gelatin
Gelatin is a voraciously used substance in the food industry for the formation of gels, for the improvement of elasticity, and stability of food. Fish gelatin has found application in food, formation of coatings, and photograph processing (Kouhdasht et al. 2018). It is produced by hydrolysis of collagen. Fish skin is soaked in an alkali solution to remove pigments and undesirable proteins after which it is washed and soaked. The process is accompanied by continuous gentle stirring in acetic acid followed by distilled water upon swelling of the skins. The product is filtered and the filtrate is lyophilized to give gelatin powder (Nazeer and Deepthi 2013; Nazeer and Suganya 2014).
11.6.2.3 Chitin and Chitosan
Chitin is a biopolymer composed of repeating N-acetylgluoseamine units that are primarily manufactured from shellfish waste. It is converted to the more stable chitosan through deacetylation by alkali treatment which is used in effluent treatment, paper, food, and cosmetic industries (Morin-Crini et al. 2019). Extraction of chitin is initiated by crushing the waste shells followed by the extraction of pigments from the shell waste using a suitable solvent to produce chitin (Fig. 11.11). Chitosan is produced by deacetylation of chitin using a strong alkali (Sudhakar et al. 2020).
11.6.2.4 Leather, Carotenoid Pigments, and Isinglass
Leather is made from fish skin similar to animal hide to make clothing, bags, shoes, etc. The skins of larger fish such as cod, shark, and salmon are generally preferred for the leather-making process. Fish leather is often lighter, strong, and attractively patterned and is thus becoming more prevalent in the fashion industry through prominent designer clothing brands (Duraisamy et al. 2016).
Carotenoids are pigments which contribute to the characteristic pink-red color of fish and shellfish. They are extracted from shellfish waste through solvent extraction during the extraction of chitin and are essential to aquacultures as they cannot be synthesized by the fish and thus must be taken up through food. Astaxanthin is a commercially marketed carotenoid used in aquacultures and as a nutraceutical product for the treatment of a variety of conditions including Alzheimer’s disease, Parkinson’s, and sunburns (Stepnowski et al. 2004).
Isinglass is a tough leather-like product sold commercially as a powder or paste. It is produced from the swim bladder of fish and is used as a common additive to beverages such as beer and in the food industry as a replacement for gelatin. Production of isinglass begins with the cleaning of the swim bladders of the fish followed by salting for 1–3 months (Asty et al. 2018). After slating, the swim bladders are washed thoroughly and the fat-rich tissue is removed. They are then stretched out and dried in sunlight to remove all water. The innermost collagenous layer is separated from the other two layers and sundried. The product at this stage is called fish maw. This maw is moisturized in water and flattened by pressing between iron rollers to form thin sheets that is the final isinglass (Shahidi et al. 2019).
11.6.2.5 Biodiesel and Fertilizer Production
Fish and other marine organisms are usually rich in fats and oil. This is the primary requirement for the synthesis of biodiesel and is a suitable alternative fuel for the current conventional sources, which is currently being rapidly depleted. Fish oil can be converted to biodiesel by suitable preliminary treatments (Ahmad and Bhuimbar 2019). Fish oils are less dense than commercially used vegetable oils, which lessens the carbon deposition and viscosity, making it a better source for biodiesel production. Ozone pretreatment is one of the most essential steps in biodiesel production. It aids in structural modifications of various types of biomasses, which would result in release of carbohydrates and other substrates for hydrolytic processes, resulting in production of biodiesel. The filtration processes are carried out in high-grade kaolin filters. The biodiesel produced is tested for various parameters like flash point, density, percentage of sulfur, etc. (Travaini et al. 2016).
The harmful side effects of synthetic fertilizers has encouraged the development of new innovative organic fertilizers. A mixture of agricultural waste and fish waste has proven to have very good nutritional properties and rapid degradability, which makes it a good alternative component of fertilizers (Radziemska et al. 2019). Though this application is only recently put into research spotlight, it has been traditionally used as fertilizers by Native Americans, using Herring fish. This technique can be proven to be very useful, especially in islands and remote coastal areas, where access to commercial fertilizers is bleak. Any fertilizer must essentially have NPK elements, i.e., nitrogen, phosphorus, and potassium, respectively. Apart from this, they should enable the soil to regenerate other minerals in the required proper ratio. Amalgamation of fish waste and seaweed was proven to be a good fertilizer after being tried in North-west Spain coast plantations (López-Mosquera et al. 2011). This way, we can get rid of seaweed and also improve crop yield. The fertilizer was synthesized by composting for months together, to derive the required nutrient ratio, enzyme degradability, moisture, and various other parameters. Fish waste has also proven to improve the C to N ratio of soil. As the proteins in the fish remains start degrading on burial, the essential nutrients, nitrogen and carbon get released into the soil. It is important that before being brought to use, it must be subjected to phytotoxicity tests (like root growth inhibition) and derive clearance. However, it must be noted that fish fertilizers are notorious for attracting animals like bear and raccoons. Thus, it should be buried deeper in the soil, or completely avoided if there is any risk of these animals (López-Mosquera et al. 2011).
11.7 Conclusion
Fish consumption has rapidly increased over the years, which has led to a complementary increase in fish waste accumulation as a large percentage of fish is inedible. Various sources of fish wastes have been identified including poorly maintained aquaculture, fisheries, and inedible fish remnants from seafood industries. Harbors are also a site of fish waste accumulation, degrading their esthetic value. Industries release untreated or partially treated fish wastes in water bodies leading to a decline in its quality index. Waste segregation practices are gradually being adapted based on the treatment and disposal methods used. The most commonly practiced disposal techniques include acid hydrolysis and anaerobic digestion. Various traditional methods of waste disposal have been put into practice including landfills, ocean disposal, incineration, direct feeding, and its usage in silage and mince-based products. Currently, this waste is used for the production of commercially valuable products. Mostly, nutritional values are tried to be exploited by making fishmeal for aquaculture and farm animals, extract vitamins and omega-rich oil, preparing silage, fish protein hydrolysates for commercial purposes, and fish protein concentrates which are used in edible products. Non-nutritional products like collagen, gelatin, chitin and chitosan, leather, fertilizer, biodiesel, isinglass, and carotenoids are also extracted from fish waste and can solve various environmental issues.
Abbreviations
- ASC:
-
Acid soluble collagen
- BOD:
-
Biological oxygen demand
- EPA:
-
Environment protection authority
- FPC:
-
Fish protein concentrate
- FPH:
-
Fish protein hydrolysate
- PSC:
-
Pepsin soluble collagen
- TSE:
-
Transmissible spongiform encephalopathy
- TTX:
-
Tetrodotoxin
References
Agustin, V., Husni, A., & Putra, M. M. P. (2021). Antioxidant activity of protein hydrolysate from snakehead fish (Channa striata) viscera obtained by enzymatic process. In IOP conference series: earth and environmental science, vol 919, no 1. IOP Publishing, p 012046
Ahmad W, Bhuimbar S (2019) Current status of fish waste management in Karwar city. Int J Res Appl Sci Eng Technol 7(4):3563–3568. https://doi.org/10.22214/ijraset.2019.4597
Ahmad R, Oterhals Å, Xue Y, Skodvin T, Samuelsen TA (2019) Impact of fish protein concentrate on apparent viscosity and physical properties of soy protein concentrate subjected to thermomechanical treatment. J Food Eng 259:34–43. https://doi.org/10.1016/j.jfoodeng.2019.04.014
Aidos I, van der Padt A, Boom RM, Luten JB (2001) Upgrading of maatjes herring byproducts: production of crude fish oil. J Agric Food Chem 49(8):3697–3704. https://doi.org/10.1021/jf001513s
Anal AK, Noomhorm A, Vongsawasdi P (2013) Protein hydrolysates and bioactive peptides from seafood and crustacean waste: their extraction, bioactive properties and industrial perspectives. In: Marine proteins and peptides: biological activities and applications, pp 709–735. https://doi.org/10.1002/9781118375082.ch36
Archer M (2001) Fish waste production in the United Kingdom: the quantities produced and opportunities for better utilisation. Sea Fish Industry Authority
Arruda LFD, Borghesi R, Oetterer M (2007) Use of fish waste as silage: a review. Braz Arch Biol Technol 50:879–886. https://doi.org/10.1590/S1516-89132007000500016
Arvanitoyannis IS, Kassaveti A (2008) Fish industry waste: treatments, environmental impacts, current and potential uses. Int J Food Sci Technol 43(4):726–745. https://doi.org/10.1111/j.1365-2621.2006.01513.x
Arvanitoyannis IS, Tserkezou P (2014) Fish waste management. In: Seafood processing–technology, quality and safety, pp 263–309
Asty WN, Yasman Y, Wardhana W (2018) Characteristic of semisolid isinglass from catfish (Pangasius sp.) swimbladder based on different concentration in acetic and citric acid solution. In AIP conference proceedings (Vol. 2023, 1, p. 020131). AIP publishing LLC. https://doi.org/10.1063/1.5064128
Balde A, Hasan A, Joshi I, Nazeer RA (2020) Preparation and optimization of chitosan nanoparticles from discarded squilla (Carinosquilla multicarinata) shells for the delivery of anti-inflammatory drug: diclofenac. J Air Waste Manage Assoc 70(12):1227–1235. https://doi.org/10.1080/10962247.2020.1727588
Balde A, Raghavender P, Dasireddy S, Abraham J, Prasad S, Joshi I, Abdul NR (2021) Crab Pentapeptide and its anti-inflammatory activity on macrophage cells. Int J Pept Res Ther 27:2595–2605. https://doi.org/10.1007/s10989-021-10276-y
Bockisch M (ed) (2015) Fats and oils handbook (Nahrungsfette und Öle). Elsevier
Bowyer JN, Qin JG, Smullen RP, Stone DAJ (2012) Replacement of fish oil by poultry oil and canola oil in yellowtail kingfish (Seriola lalandi) at optimal and suboptimal temperatures. Aquaculture 356:211–222. https://doi.org/10.1016/j.aquaculture.2012.05.014
Boziaris IS (2014) Introduction to seafood processing-assuring quality and safety of seafood. In: Seafood Processing—Technology, Quality, and Safety, pp 1–18
Cristóvão RO, Botelho CM, Martins RJ, Loureiro JM, Boaventura RA (2014) Primary treatment optimization of a fish canning wastewater from a Portuguese plant. Water Resour Ind 6:51–63. https://doi.org/10.1016/j.wri.2014.07.002
Das M, Ghosh S, Maheswarudu G (2011) Tuna fish waste as an aquafeed substitute at Visakhapatnam. Mar Fish Infor Serv 209:3–3. http://eprints.cmfri.org.in/id/eprint/9120
Dauda AB, Ajadi A, Tola-Fabunmi AS, Akinwole AO (2019) Waste production in aquaculture: sources, components and managements in different culture systems. Aquac Fish J 4(3):81–88. https://doi.org/10.1016/j.aaf.2018.10.002
Duraisamy R, Shamena S, Berekete AK (2016) A review of bio-tanning materials for processing of fish skin into leather. Int J Eng Trends Technol 39(1):10–20
Ghaly AE, Ramakrishnan VV, Brooks MS, Budge SM, Dave D (2013) Fish processing wastes as a potential source of proteins. Amino acids and oils: a critical review. J Microb Biochem Technol 5(4):107–129. https://doi.org/10.4172/1948-5948.1000110
Govindharaj M, Roopavath UK, Rath SN (2019) Valorization of discarded marine eel fish skin for collagen extraction as a 3D printable blue biomaterial for tissue engineering. J Clean Prod 230:412–419. https://doi.org/10.1016/j.jclepro.2019.05.082
Green JH, Mattick JF (1977) Possible methods for the utilization or disposal of fishery solid wastes 1. J Food Qual 1(3):229–251. https://doi.org/10.1111/j.1745-4557.1977.tb00943.x
Hamed I, Özogul F, Regenstein JM (2016) Industrial applications of crustacean by-products (chitin, chitosan, and chitooligosaccharides): a review. Trends Food Sci Technol 48:40–50. https://doi.org/10.1016/j.tifs.2015.11.007
Han DH (2015) A recycling method of rotten fish wastes. Indian J Sci Technol 8(12):134–141. https://doi.org/10.17485/ijst/2015/v8i33/85283
Islam MS, Khan S, Tanaka M (2004) Waste loading in shrimp and fish processing effluents: potential source of hazards to the coastal and nearshore environments. Mar Pollut Bull 49(1–2):103–110. https://doi.org/10.1016/j.marpolbul.2004.01.018
Joshi I, Janagaraj K, Nazeer RA (2020) Isolation and characterization of angiotensin I-converting enzyme (ACE-I) inhibition and antioxidant peptide from by-catch shrimp (Oratosquilla woodmasoni) waste. Biocatal Agric Biotechnol 29:101770. https://doi.org/10.1016/j.bcab.2020.101770
Ke WC, Ding WR, Xu DM, Ding LM, Zhang P, Li FD, Guo XS (2017) Effects of addition of malic or citric acids on fermentation quality and chemical characteristics of alfalfa silage. J Dairy Sci 100(11):8958–8966. https://doi.org/10.3168/jds.2017-12875
Kim SK, Mendis E (2006) Bioactive compounds from marine processing byproducts–a review. Food Res Int 39(4):383–393. https://doi.org/10.1016/j.foodres.2005.10.010
Kouhdasht AM, Moosavi-Nasab M, Aminlari M (2018) Gelatin production using fish wastes by extracted alkaline protease from bacillus licheniformis. J Food Sci Technol 55(12):5175–5180. https://doi.org/10.1007/s13197-018-3449-7
Kristinsson HG, Rasco BA (2000) Fish protein hydrolysates: production, biochemical, and functional properties. Crit Rev Food Sci Nutr 40(1):43–81. https://doi.org/10.1080/10408690091189266
Kruijssen F, Tedesco I, Ward A, Pincus L, Love D, Thorne-Lyman AL (2020) Loss and waste in fish value chains: a review of the evidence from low and middle-income countries. Glob Food Sec 26:100434. https://doi.org/10.1016/j.gfs.2020.100434
Kumar NS, Nazeer RA, Jaiganesh R (2012) Wound healing properties of collagen from the bone of two marine fishes. Int J Pept Res Ther 18(3):185–192. https://doi.org/10.1007/s10989-012-9291-2
Lago J, Rodríguez LP, Blanco L, Vieites JM, Cabado AG (2015) Tetrodotoxin, an extremely potent marine neurotoxin: distribution, toxicity, origin and therapeutical uses. Mar Drugs 13(10):6384–6406. https://doi.org/10.3390/md13106384
López-Mosquera ME, Fernández-Lema E, Villares R, Corral R, Alonso B, Blanco C (2011) Composting fish waste and seaweed to produce a fertilizer for use in organic agriculture. Procedia Environ Sci 9:113–117. https://doi.org/10.1016/j.proenv.2011.11.018
Mack D, Huntington T, Curr C, Joensen J (2004) Evaluation of fish waste management techniques. Report to the Scottish Environment Protection Agency. Poseidon Aquatic Resource Management Ltd, Lymington, Hampshire
Mathew GM, Sukumaran RK, Sindhu R, Binod P, Pandey A (2021) Green remediation of the potential hazardous shellfish wastes generated from the processing industries and their bioprospecting. Environ Technol Innov 24:101979. https://doi.org/10.1016/j.eti.2021.101979
Miller D, Semmens K (2002) Waste management in aquaculture. West Virginia University Extension Service Publication No. AQ02-1. USA 8
Mo WY, Man YB, Wong MH (2018) Use of food waste, fish waste and food processing waste for China's aquaculture industry: needs and challenge. Sci Total Environ 613:635–643. https://doi.org/10.1016/j.scitotenv.2017.08.321
Morin-Crini N, Lichtfouse E, Torri G, Crini G (2019) Applications of chitosan in food, pharmaceuticals, medicine, cosmetics, agriculture, textiles, pulp and paper, biotechnology, and environmental chemistry. Environ Chem Lett 17(4):1667–1692. https://doi.org/10.1007/s10311-019-00904-x
Mshandete A, Kivaisi A, Rubindamayugi M, Mattiasson BO (2004) Anaerobic batch co-digestion of sisal pulp and fish wastes. Bioresour Technol 95(1):19–24. https://doi.org/10.1016/j.biortech.2004.01.011
Munari C, Corbau C, Simeoni U, Mistri M (2016) Marine litter on Mediterranean shores: analysis of composition, spatial distribution and sources in north-western Adriatic beaches. Waste Manag 49:483–490. https://doi.org/10.1016/j.wasman.2015.12.010
Narayanasamy A, Balde A, Raghavender P, Shashanth D, Abraham J, Joshi I, Nazeer RA (2020) Isolation of marine crab (Charybdis natator) leg muscle peptide and its anti-inflammatory effects on macrophage cells. Biocatal Agric Biotechnol 25:101577. https://doi.org/10.1016/j.bcab.2020.101577
Nazeer RA, Deepthi MK (2013) Physicochemical and nanostructural properties of gelatin from uneconomical marine cornet fish (Fistularia petimba). Food Sci Biotechnol 22(1):9–14. https://doi.org/10.1007/s10068-013-0002-3
Nazeer RA, Suganya US (2014) Porous scaffolds of gelatin from the marine gastropod Ficus variegata with commercial cross linkers for biomedical applications. Food Sci Biotechnol 23(2):327–335. https://doi.org/10.1007/s10068-014-0046-z
Nges IA, Mbatia B, Björnsson L (2012) Improved utilization of fish waste by anaerobic digestion following omega-3 fatty acids extraction. J Environ Manag 110:159–165. https://doi.org/10.1016/j.jenvman.2012.06.011
Olsen RL, Toppe J (2017) Fish silage hydrolysates: not only a feed nutrient, but also a useful feed additive. Trends Food Sci Technol 66:93–97. https://doi.org/10.1016/j.tifs.2017.06.003
Plazzotta S, Manzocco L (2019) Food waste valorization. In: Saving food. Academic Press, pp 279–313. https://doi.org/10.1016/B978-0-12-815357-4.00010-9
Radziemska M, Vaverková MD, Adamcová D, Brtnický M, Mazur Z (2019) Valorization of fish waste compost as a fertilizer for agricultural use. Waste and Biomass Valorization 10(9):2537–2545. https://doi.org/10.1007/s12649-018-0288-8
Rao GS (2010) Current status and prospects of fishery resources of the Indian continental shelf. Society of Fisheries Technologists, pp 1–13. http://eprints.cmfri.org.in/id/eprint/8748
Read P, Fernandes T (2003) Management of environmental impacts of marine aquaculture in Europe. Aquaculture 226(1–4):139–163. https://doi.org/10.1016/S0044-8486(03)00474-5
Rustad T, Hayes M (2012) Marine bioactive peptides and protein hydrolysates: generation, isolation procedures, and biological and chemical characterizations. In: Marine bioactive compounds. Springer, Boston, MA. https://doi.org/10.1007/978-1-4614-1247-2_3
Rustad T, Storrø I, Slizyte R (2011) Possibilities for the utilisation of marine by-products. Int J Food Sci Technol 46(10):2001–2014. https://doi.org/10.1111/j.1365-2621.2011.02736.x
Ryder J (2018) Aquaculture and trade. FAO Aquaculture Newslett 58:II–III
Sampath Kumar NS, Nazeer RA (2013) Characterization of acid and pepsin soluble collagen from the skin of horse mackerels (Magalaspis cordyla) and croaker (Otolithes ruber). Int J Food Prop 16(3):613–621. https://doi.org/10.1080/10942912.2011.557796
Saravanan R, Sambasivam S, Shanmugam A, Kumar DS, Vanan T, Nazeer RA (2009) Isolation, purification and biochemical characterization of conotoxin from Conus figulinus Linnaeus (1758). http://nopr.niscair.res.in/handle/123456789/4738
Sasidharan A, Mathew S (2011) Current status of fish waste management in Kerala, India. Waste Manag 31(8):1898–1900
Schneider F, Parsons S, Clift S, Stolte A, McManus MC (2018) Collected marine litter—a growing waste challenge. Mar Pollut Bull 128:162–174. https://doi.org/10.1016/j.marpolbul.2018.01.011
Shahidi F, Varatharajan V, Peng H, Senadheera R (2019) Utilization of marine by-products for the recovery of value-added products. J Food Bioact 6. https://doi.org/10.31665/JFB.2019.6184
Sharp M, Mariojouls C (2012) Waste not, want not: better utilisation of fish waste in the Pacific. SPC Fish Newslett 138:44–48
Stepnowski P, Olafsson G, Helgason H, Jastorff B (2004) Recovery of astaxanthin from seafood wastewater utilizing fish scales waste. Chemosphere 54(3):413–417. https://doi.org/10.1016/S0045-6535(03)00718-5
Sudhakar S, Chandran SV, Selvamurugan N, Nazeer RA (2020) Biodistribution and pharmacokinetics of thiolated chitosan nanoparticles for oral delivery of insulin in vivo. Int J Biol Macromol 150:281–288. https://doi.org/10.1016/j.ijbiomac.2020.02.079
Travaini R, Martín-Juárez J, Lorenzo-Hernando A, Bolado-Rodríguez S (2016) Ozonolysis: an advantageous pretreatment for lignocellulosic biomass revisited. Bioresour Technol 199:2–12. https://doi.org/10.1016/j.biortech.2015.08.143
van Rijn J, Fonarev N, Berkowitz B (1995) Anaerobic treatment of intensive fish culture effluents: digestion of fish feed and release of volatile fatty acids. Aquaculture 133(1):9–20. https://doi.org/10.1016/0044-8486(94)00386-3
Venugopal V (2021) Valorization of seafood processing discards: bioconversion and bio-refinery approaches. Front Sustain Food Syst 5:132. https://doi.org/10.3389/fsufs.2021.611835
Willis K, Maureaud C, Wilcox C, Hardesty BD (2018) How successful are waste abatement campaigns and government policies at reducing plastic waste into the marine environment? Mar Policy 96:243–249. https://doi.org/10.1016/j.marpol.2017.11.037
Yan N, Chen X (2015) Sustainability: don't waste seafood waste. Nature News 524(7564):155. https://doi.org/10.1038/524155a
Yoshida H, Terashima M, Takahashi Y (1999) Production of organic acids and amino acids from fish meat by sub-critical water hydrolysis. Biotechnol Prog 15(6):1090–1094. https://doi.org/10.1021/bp9900920
Yoshida H, Takahashi Y, Terashima M (2003) A simplified reaction model for production of oil, amino acids, and organic acids from fish meat by hydrolysis under sub-critical and supercritical conditions. J Chem Eng Japan 36(4):441–448. https://doi.org/10.1252/jcej.36.441
Zhou X (2017) Considerations from the aquaculture statistician. FAO Aquaculture Newslett 57:7–8
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Nazeer, R.A., Joshi, I., Mahankali, S., Mazumdar, A., Sridharan, B., Sankar, S.J. (2023). Is Marine Waste a Boon or Bane? An Insight on Its Source, Production, Disposal Consequences, and Utilization. In: Samuel Jacob, B., Ramani, K., Vinoth Kumar, V. (eds) Applied Biotechnology for Emerging Pollutants Remediation and Energy Conversion. Springer, Singapore. https://doi.org/10.1007/978-981-99-1179-0_11
Download citation
DOI: https://doi.org/10.1007/978-981-99-1179-0_11
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-99-1178-3
Online ISBN: 978-981-99-1179-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)