Abstract
The clinical application of stem cells continues to fascinate the scientific and clinical communities. Despite the controversies surrounding this field, it is clear that stem cells have revolutionized regenerative medicine. Cell therapy is a progressively growing field that is moving fast from preclinical model development to clinical application. In this regard, outcomes obtained from clinical trials reveal the therapeutic potential of stem cell-based therapy that deals with unmet medical treatment for several disorders with no therapeutic alternatives. The application of stem cells in regenerative medicine is addressing a wide range of clinical conditions using various types of stem cells. Mesenchymal stromal cells (MSCs) have been established as promising candidate sources of universal donor cells for cell therapy due to their contributions to tissue and organ homeostasis, repair, and support by self-renewal and multi-differentiation, as well as by their anti-inflammatory, anti-proliferative, immunomodulatory, trophic, and pro-angiogenic properties. Various diseases have been successfully treated by MSCs in animal models. Additionally, hundreds of clinical trials related to the potential benefits of MSCs are in progress or have concluded satisfactorily. However, although all MSCs are considered suitable to exert these functions, dissimilarities have been found among MSCs derived from different tissues. The same levels of efficacy and desired outcomes have not always been achieved in the diverse studies that have been performed thus far. Therefore, collecting information regarding the characteristics of MSCs obtained from different sources and the influence of other medical and physiological conditions on MSCs is important for assuring the feasibility, safety, and efficacy of cell-based therapies. This chapter will update and discuss the state of the art in MSCs’ cell-based therapies and provide relevant information regarding factors to consider for the clinical application of MSCs.
Access provided by Autonomous University of Puebla. Download reference work entry PDF
Similar content being viewed by others
Keywords
- Advanced therapy
- Cell therapy
- Clinical trial
- Good manufacturing practice
- Immunomodulation
- Inflammation
- Medicinal products
- Mesenchymal stromal cells
- Trophic factors
Introduction
Regenerative medicine is a novel emerging medical approach that drives the current understanding of biological and medical processes and suggests new treatments. As defined by the European Medicines Agency (EMA) and the US Food and Drug Administration (FDA), advanced therapies include cell and gene therapy and tissue engineering (Iglesias-López et al. 2019). Advanced therapies open up a broad set of translational fields and targets in areas of unmet medical need. In this regard, cell-based therapy through the application of cells, either alone or engineered, as a pharmacologically active substance seeks to restore the functioning of damaged tissues or organs through the protection of cellular integrity, the replacement of damaged cells, and the promotion of trophic, anti-inflammatory, and immunomodulatory effects among others. However, while the progression of cell-based therapy in early-phase clinical trials with patients has progressed promisingly, the translation from laboratory to bedside to late-phase clinical trials has not been as rapid as expected. It is necessary to consider that these new therapeutic alternatives also involve unknown side effects that must be detected and characterized in-depth to improve and ensure safety, feasibility, and efficacy of cell application (García-Bernal et al. 2021; Hmadcha et al. 2020; Soria-Juan et al. 2019; Escacena et al. 2015; Gálvez et al. 2013).
In this regard, mesenchymal stromal cells (MSCs) are the most common cell type used in cell-based therapy due to their unique biological properties, including easy expansion and culture. The MSC-Committee of the International Society for Cellular Therapy (ISCT-MSC) first proposed that plastic-adherent cells of the bone marrow (BM) generally described as “mesenchymal stem cells” should be defined as “multipotent mesenchymal stromal cells.” In contrast, the term “mesenchymal stem cells” should be reticent for a subset of these cells that show stem cell activity by clearly stated criteria. As the acronym MSCs may be used to define both cell populations, the combined definition “mesenchymal stem/stromal cells” is probably more appropriate, especially when the “stemness” of the whole MSC population is not demonstrated (Horwitz et al. 2005). Recently, this committee offers a position statement to clarify the nomenclature of “mesenchymal stem/stromal cells.” The ISCT-MSC committee continues to support the use of the acronym “mesenchymal stromal cells” but recommends that this should be complemented by the tissue origin from which the cells were derived, which would highlight tissue-specific properties: that they be referred to as “stromal” unless there are rigorous in vitro and in vivo evidence of their stemness supplemented by a robust matrix of functional assays to demonstrate the “mesenchymal stromal cells” properties. Thus, they should not be defined generically, but based on the intended therapeutic mode of action (Viswanathan et al. 2019).
The MSCs are now considered as “cellular medicament” but are widely accepted to represent a heterogeneous population of multipotent non-hematopoietic progenitor cells with varying degrees of stemness, which mean that they have self-renewal and multi-differentiation abilities, the capability to differentiate into multiple cell types, including adipocytes, chondrocytes, and osteoblasts, depending on in vitro culture conditions (Soria-Juan et al. 2019).
The MSCs reside in almost all tissues, are found in virtually all post-natal organs and tissues, and are derived from the mesodermal germ layer. Furthermore, MSCs can be obtained from easily accessible sources by minimally invasive methods (e.g., peripheral blood, adipose tissue) and can be rapidly expanded in large scale for clinical use (Escacena et al. 2015). This allows producing a patient-specific cellular medicament (e.g., autologous medicinal product) within a therapeutic time window. In addition, the possibility of obtaining MSCs from adult tissue circumvents the ethical issues associated with the use of embryonic source (Lo and Parham 2009; Ramos-Zúñiga et al. 2012). MSCs are inexpensively isolated and are easily expanded in vitro due to their fibroblastic characteristic and high adherence to plastic. MSCs are characterized by a specific pattern of membrane markers, consisting of the expression of CD73, D90, and CD105 and the absence of expression of CD14, CD34, CD45, and human leukocyte antigen-DR (HLA-DR), making them promising candidate sources of donor cells for use in cell-based therapy and transplantation (Horwitz et al. 2005).
MSCs function in tissue repair and support, contributing to tissue homeostasis. Even though the exact origin of MSCs remains elusive, there is strong evidence that MSC progenitors are found in the perivascular zone (Escacena et al. 2015) in an environment that promotes a quiescent state, ensuring the maintenance of homeostasis. Upon tissue damage, MSCs enter the bloodstream and are attracted to pro-inflammatory cytokines in the areas of injury. Therefore, MSCs have been termed “guardians of inflammation” (Prockop and Oh 2012). The cytoskeleton, extracellular matrix molecules, cell-cell contacts, adhesion ligands, and receptors are involved in the repair process. While the exact mechanisms related to MSCs’ migration to specific sites and through the endothelial cell layer are still unknown, chemokines and their receptors may play a role in this process (Hmadcha et al. 2020; Petrie et al. 2009).
Furthermore, MSCs’ survival, permanent engraftment, and differentiation into resident cells were thought, initially, to be necessary to obtain the beneficial effects of these cells, and clinical experience and several experiments have shown that one of the primary functions of MSCs, most likely their critical function, is to secrete several bioactive molecules related to the microenvironment “niche” in which these cells are located. Consequently, the secretome reproduces most of the effects of MSCs transiently; in this sense, MSCs secrete a wide variety of pro-inflammatory and anti-inflammatory cytokines, chemokines, growth factors, and prostaglandins under resting and inflammatory conditions (Hmadcha et al. 2009).
These molecules are associated with immunomodulation (indoleamine-2,3-dioxygenase (IDO), prostaglandin-E2 (PGE-2), transforming growth factor beta (TGF-β), human leukocyte antigen-G5 (HLA-G5), and hepatocyte growth factor (HGF)), anti-apoptosis (vascular endothelial growth factor (VEGF), granulocyte-macrophage colony-stimulating factor (GM-CSF), TGF-β, stanniocalcin-1 (STC1), and insulin-like growth factor 1 (IGF-1)), angiogenesis (VEGF, monocyte chemoattractant protein 1 (MCP-1), and IGF-1), local stem and progenitor cell growth and differentiation support (CSF complex, angiopoietin-1, and stromal cell-derived factor 1 (SDF-1)), anti-fibrosis (HGF and basic fibroblast growth factor (bFGF)), and chemoattraction (chemokine (C-C motif) ligands 2 and 4 (CCL2, CCL4) and C-X-C motif chemokine 12 (CXCL12 also called SDF1)) (Meirelles Lda et al. 2009).
MSCs display a low expression of major histocompatibility complex class I human leukocyte antigen (MHC-HLA class I), while they are constitutively negative for HLA-class II; likewise, they do not express costimulatory molecules such as CD80, CD86, CD40, and CD40L. However, MSCs share the expression of surface markers, such as vascular cell adhesion protein 1 (VCAM-1), intercellular adhesion molecule 2 (ICAM-2), and lymphocyte function-associated antigen 3 (LFA-3 or CD58) with the thymic epithelium, which is crucial for the interaction with T cells (Hmadcha et al. 2009; Le Blanc 2003). Whereas MSCs remain in a quiescent state showing anti-apoptotic properties and contributing to homeostasis, in an inflammatory environment (presence of IFN-γ, TNF-α, IL-1α, and IL-1β), they begin to exercise their immunomodulation abilities, inhibiting the proliferation of effector cells and their cytokine production. In the same way, MSCs can block various immune cell functions (Hmadcha et al. 2009; Cagliani et al. 2017).
There is a complex “cross-talk” interaction between MSCs and endothelial cells (ECs). MSCs increase the proliferation and migration of the ECs, promoting early events of angiogenesis and decreasing the permeability of the monolayer of the ECs. In direct co-cultures of MSCs and ECs, MSCs increased the persistence of pre-existing blood vessels in a dose-dependent manner (Duffy et al. 2009). Moreover, beneficial therapeutic effects of the use of conditioned media of MSCs have been reported; even it is therapeutically better than the cells themselves (Burlacu et al. 2013; Shrestha et al. 2013) and to stimulate the proliferation of local ECs (Potapova et al. 2007). Likewise, in addition to direct “cell-cell” contact, speculation has been made with a possible transfer of mitochondria or vesicular components (secretome) that contain mRNA, microRNA, and proteins (Tan et al. 2021). Not only have this, the exosomes, secretory extracellular vesicles (EVs) from MSCs, also been identified to produce the same immunomodulatory activity as MSCs (Haider and Aramini 2020). Targeting the MSCs’ secretome as an acellular therapeutic agent could provide several advantages over the use of cell-based therapies for various diseases paving the way for cell-free therapy (Haider and Aslam 2018; Bari et al. 2019).
Altogether, these features constitute an area of research in expansion in the last decade and make MSCs an eligible therapeutic candidate to be evaluated within clinical trials for a plethora of diseases such as diabetes and diabetes complication and cardiovascular and neurological diseases; in immune-mediated disorders, such as graft-versus-host diseases (GVHD), multiple sclerosis (MS), Crohn’s disease (CD), and osteoarthritis (OA); and even in immune-dysregulating infectious diseases such as the novel coronavirus disease 2019 (COVID-19) (see Chap. 6, “Mesenchymal Stromal Cells for COVID-19 Critical Care Patients,” of this book for review on COVID-19).
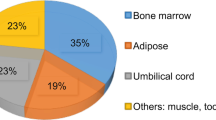
When writing this chapter (May 2021), 1.276 × 103 publicly and privately funded clinical studies worldwide in which MSCs have been used have been reported and registered in the US National Library of Medicine database (NIH-ClinicalTrials.gov). Although the therapeutic efficacy of MSCs has been demonstrated in different disease animal models and numerous human phase 1/2 clinical trials and generally communicated, only very few (84 studies) phase 3/4 clinical trials using MSCs are registered (Table 1) and have demonstrated the expected potential therapeutic benefit. Almost all registered clinical trials are early phase 1/2 with safety as the primary objective. For efficacy and effectiveness issues, other advanced phases are mandatory. In all cases, one cannot consider these issues (efficacy nor effectiveness) unless phase 3 clinical trials are developed (García-Bernal et al. 2021) (Fig. 1).
The MSCs have multiple potential advantages for their clinical application. Among other advantages, MSCs can be isolated from various sources, are produced on a large scale, differentiate into various cell types, and have pleiotropic effects. Such advantages make MSCs suitable for clinical application in different disease conditions, such as neurological damage, liver disorders, cardiac ischemia, diabetes, or skin problems. (Re-published from Hmadcha 2020, article published under CC-BY terms). Abbreviations: bFGF basic fibroblast growth factor, CXCL12 C-X-C motif chemokine 12, EPO erythropoietin, GM-CSF granulocyte-macrophage colony-stimulating factor, HGF hepatocyte growth factor, HLA-DR major histocompatibility complex class II DR, iDC immature dendritic cell, IDO indoleamine-2,3-dioxygenase, IGF1 insulin-like growth factor 1, IL-10 interleukin-10, IL-12 interleukin-12, IL-4 interleukin-4, IL-6 interleukin-6, INF-γ interferon-γ, iNOS inducible nitric oxide synthase, KGF keratinocyte growth factor, MCP1 monocyte chemoattractant protein 1, MIP macrophage inflammatory protein, MMP matrix metalloproteinases, MN monocyte, NK natural killer cell, SFRP2 secreted frizzled-related protein 2, STC1 stanniocalcin 1, TF tissue factor, TGF-β transforming growth factor beta, TIMP tissue inhibitor of metalloproteinases, TNF-α tumor necrosis factor α, TRAIL TNF-related apoptosis-inducing ligand, Treg regulatory T cell, VEGF vascular endothelial growth factor
Even though MSCs and their EVs have been shown to have high potential benefits in regenerative medicine and cell-free-based therapy, their clinical application remains controversial; thus, considerations and determination of possible side effects need to be addressed to optimize the clinical application of this double-edged sword cellular medicament. This chapter updates and discusses the state of the art in MSCs’ cell-based therapies and provides relevant information regarding factors to consider for the clinical application of MSCs.
Biological Characteristics
Phenotypic Profile
Since Friedenstein and colleagues first isolated a colony-forming unit fibroblast (CFU-F) from BM, BM has been widely used as a source of MSCs for many investigations and clinical trials. In addition to BM (BM-derived MSCs), MSCs have been isolated from different tissues, such as adipose tissue (Ad-MSCs), umbilical cord (UCB-MSCs), dental pulp, synovial liquid, and amniotic fluid. All these tissues vary in their cellular components, signals, and factors secreted, resulting in different immediate microenvironment conditions, thus developing several physiological niches (Hmadcha et al. 2009; Escacena et al. 2015). Although isolated and long-term cultured MSCs of most tissues show similar immunophenotypic characteristics, some differences have been found among MSCs of different tissue origins according to data obtained by in vitro experiments. In 2006, the International Society for Cellular Therapy (ISCT) published the minimal criteria to define MSCs by nomenclature and by biological characteristics to allow studies from different groups to be compared and contrasted. These criteria include the co-expression of markers such as CD73, CD90, and CD105, a lack of expression of hematopoietic markers (CD45, CD34, and CD14) and HLA-DR, multipotent differentiation potential, and adherence to plastic (Horwitz et al. 2005). However, several researchers have noted that Ad-MSCs express CD34 and CD54 in early passages and have lower expression of CD106 and that umbilical cord blood-derived MSCs (UCB-MSCs) express CD90 and CD105. Other markers have been used in different studies, and other differences have emerged, such as VEGFR-2 (Flk-1) expression, which was significantly higher in periosteum-derived cells than in adipose tissue- and muscle-derived cells, or the rate of NGFR positivity, which was much higher in muscle-derived cells than in other mesenchymal tissue-derived cells (Escacena et al. 2015).
Although some immunophenotypic differences have been documented, many researchers consider that these differences could be due to distinct extraction methods and different culture methodologies, resulting in variations of MSC surface markers. Therefore, this chapter aimed to investigate markers and characteristics that are more specific to select better sources of MSCs for clinical applications. Likewise, expanding the cells in vitro is necessary to obtain the desired numbers for therapeutic approaches. Changes in the proteomic phenotype of MSCs have been observed during high passages, although no proper approaches to examine the state of cells continuously during long-term in vitro culture have been established (Capra et al. 2012). Some researchers ascribe these variations to the adaptation of cells to the environment; thus, determining the biomolecular markers that are involved in these variations is essential for obtaining a better phenotypic characterization of these cells and thus for achieving more effective cell therapy in the future (Escacena et al. 2015).
MSCs’ Proliferation
The proliferative activity of MSCs is another feature that may be affected by the different origins of MSCs. The rate and persistence of MSC proliferation appear to vary between source tissues. MSCs are considered adult stem cells, and unlike embryonic stem cells (ESCs), these cells have a limited proliferative capacity. Physiological niches maintain adult stem cells in an undifferentiated state; however, when MSCs are cultured in vitro, they age, which affects their therapeutic properties, such as alterations in phenotype, differentiation potential, global gene expression patterns, miRNA profiles, and even chromosomal abnormalities, particularly after long-term culture or when cells of multiple doublings are used (Escacena et al. 2015). Large numbers of MSCs are needed for therapeutic applications, and in vitro expansion is required to produce the desired MSC numbers. In vivo, MSCs represent 0.0001% of nucleated BM cells, and their number decreases with the donor’s age. The quantity of MSCs (CFU-Fs) among nucleated BM cells decreases with age from one MSC in 104 BM cells in newborn to one MSC in 105 cells in teenagers and one MSC in 106 cells in older individuals (Caplan 2009).
Furthermore, MSCs from older human donors differ significantly from younger donors in morphology, replicative lifespan, doubling time, healing capacity, and differentiation potential. Sufficient evidence has indicated that MSCs from older donors have limited therapeutic efficacy. Some studies have suggested that the difference between preclinical and clinical findings is due to the donor age (Stenderup et al. 2003; Escacena et al. 2015). Therefore, considering that several age-related diseases exist and that elderly patients are potential users of cell therapy, understanding the molecular and biological effects of aging on MSCs is essential for developing safe and effective MSC-based autologous cell therapy. Meanwhile, the use of allogeneic MSCs may be a treatment option for these specific patients. As commented below, MSCs elude allogeneic rejection, and their infusion is feasible and well-tolerated, with no adverse effects (McAuley et al. 2014; Liang et al. 2010).
Differentiation Capacity
MSCs can differentiate in vitro into several mesenchymal lineages, including adipose tissue, bone, cartilage, and muscle (Pittenger et al. 1999; Prockop 1997; Bruder et al. 1997). Furthermore, MSCs can differentiate into ECs, neurons, and glial cells because MSCs express genes related to specific lineages rather than those of the mesenchymal lineage (Woodbury et al. 2002). Although multilineage differentiation is another minimal criterion advised by the ISCT and undoubtedly represents a fundamental property of MSCs, this ability depends primarily on the source tissue from which these cells are derived. As such, Sakaguchi and colleagues (Sakaguchi et al. 2005) compared human MSCs isolated from BM, synovium, periosteum, skeletal muscle, and adipose tissue. The cells were expanded by similar processes; synovium-derived cells had the most remarkable ability for chondrogenesis; adipose- and synovium-derived cells, for adipogenesis; and BM-, synovium-, and periosteum-derived cells, for osteogenesis. In another comparative analysis, UCB-MSCs showed no adipogenic differentiation capacity compared to BM- and Ad-MSCs (Kern et al. 2006).
As discussed by Horwitz (Horwitz et al. 2002), who used differentiated MSCs in a study to test the regeneration of damaged tissues, BM-derived MSCs can engraft after transplantation, differentiate to functional osteoblasts, and contribute to the formation of new dense bone in children with osteogenesis imperfecta. Most likely, the microenvironment in which MSCs are transplanted directly influences in their distinct differentiation pathways. New insights into the biological characteristics of MSCs are needed to achieve future therapies.
Cellular Transformation
In general, successive passages or long-term cultures induce genetic instability and cell transformation. Several authors have described that MSCs cultivated in vitro can be expanded multiple times without an apparent loss of differentiation potential or chromosomal alterations and even that long-term MSC cultures can develop chromosomal abnormalities but without an obvious potential for transformation (Koç et al. 2000; Le Blanc et al. 2004; Ringdén et al. 2006; Fang et al. 2006; Ning et al. 2008). Although no tumor formation in humans has been reported after the administration of MSCs, several factors must be considered that can contribute significantly to the induction of cytogenetic abnormalities, such as aspects related to the manufacturing process of the cellular medicine (e.g., culture conditions and duration of cell expansion) and heterogeneity of the MSC population (e.g., cells in different stages of duplication). The tumorigenic potential of a cell therapy medicament may depend on intrinsic and extrinsic factors, such as the administration site in the patient (due to the receptor’s microenvironment) and/or the manipulation of the culture ex vivo.
Mechanism of Action
Cell Migration Toward Damaged Tissues
The success of an advanced therapy medicinal product initially depends on its ability to reach target tissues. MSCs possess inherent tropism toward damaged sites controlled by many factors and mechanisms, including chemoattractant signals. For instance, the C-X-C motif chemokine ligand 12 (CXCL12) is a frequent triggering factor at the injury site. It has been demonstrated that a subpopulation of MSCs expresses the C-X-C chemokine receptor type 4 (CXCR4) that binds to its ligand, the CXCL12, to mediate cell migration (Wynn et al. 2004; Ma et al. 2015). Aside from CXCR4, MSCs express other chemokine receptors, such as CCR1, CCR2, CCR4, CCR7, CCR8, CCR9, CCR10, CXCR1, CXCR2, CXCR3, CXCR4, CXCR5, CXCR6, and CX3CR1 (Sordi et al. 2005; Von Lüttichau et al. 2005; Honczarenko et al. 2006; Ringe et al. 2007). These receptors are essential to respond to triggering factors at the site of injury. In addition, MSCs also express cell adhesion molecules, including CD49d, CD44, CD54, CD102, and CD106 (De Ugarte et al. 2003). These chemokines and cell adhesion molecules orchestrate the mobilization of MSCs’ injury sites in a similar manner to white blood cells (Kolaczkowska and Kubes 2013). MSC mobilization is a multistep process that encompasses the attachment of free circulating MSCs in the bloodstream to transmigrate between ECs with the ultimate goal of migrating and engrafting to the target tissue.
Tissue Repair
Once recruited in the injured site, MSCs contribute to tissue repair and regeneration by activating several mechanisms. A growing body of research has demonstrated that MSCs display pleiotropic effects, which give them enormous therapeutic potential. MSCs secrete various mediators of tissue repair in response to injury signals, including anti-apoptotic, anti-inflammatory, immunomodulatory, anti-fibrotic, and angiogenic agents (Caplan and Dennis 2006; Meirelles Lda et al. 2009; Maltman et al. 2011; Escacena et al. 2015). Among pleiotropic effects, anti-inflammatory and immunomodulatory properties are mainly responsible for the therapeutic benefits of MSCs. As sensors of inflammation, MSCs release soluble factors, such as TGF-β, IDO, TNF-α, IL-10, and INF-γ, which interfere with the immune system and modify the inflammatory landscape (Prockop and Oh 2012). Pivotal studies showed that MSCs inhibit the proliferation of T and B cells (Di Nicola et al. 2002; Corcione et al. 2006; Song et al. 2019), suppress the activation of natural killer cells (Sotiropoulou et al. 2006), and prevent the generation and maturation of monocyte-derived dendritic cells (English et al. 2008; Spaggiari et al. 2009). Furthermore, MSCs can promote the generation of regulatory T cells (Maccario et al. 2005), which exert immunosuppressive effects. Although soluble factors play a key role in the immunosuppressive activity of MSCs, cell-to-cell contact also influences immune responses (Ren et al. 2010; Li et al. 2019). For instance, direct contact between MSCs and pro-inflammatory macrophages has been shown to induce immune tolerance by inducing tumor necrosis factor-stimulated gene-6 (TSG-6) production (Li et al. 2019). MSC mediated modulations of the immune response set in motion essential inflammatory processes that significantly promote tissue repair and regeneration by driving healing, scarring, and fibrosis (Julier et al. 2017).
Immunomodulatory Potential
The immunomodulatory properties of MSCs and their immune-privileged condition make these cells good candidates for use in several clinical trials related to chronic, inflammatory, and autoimmune diseases. MSCs interact with cells of the innate or adaptive immune system (T cells, B cells, NK cells, monocyte-derived dendritic cells, and neutrophils) (Di Nicola et al. 2002; Raffaghello et al. 2008). For a cell to be recognized by the immune system, the expression of major histocompatibility complex (MHC) and co-stimulatory molecules is necessary. MHC class I and class II human leukocyte antigens (HLAs) are master triggers of robust immunological rejection of grafts because they present antigens to cytolytic T lymphocytes (CTL). Human mesenchymal stem cells (hMSCs) are characterized by low expression of MHC class I HLAs but are constitutively negative for class II HLCs; these cells do not express co-stimulatory molecules such as B7-1, B7-2, CD80, CD86, CD40, and CD40L (Hmadcha et al. 2009; Le Blanc 2003). However, similar to the thymic epithelium, MSCs express the surface markers VCAM-1, ICAM-2, and LFA-3 (Le Blanc 2003; Conget and Minguell 1999), which are crucial for T-cell interactions.
Although a T-cell response should be expected, hMSCs can modulate the activation and proliferation of both CD4+ and CD8+ cells in vitro by arresting T cells in G0/G1 phase (Glennie et al. 2005; Benvenuto et al. 2007). Different studies have suggested that cell-cell interactions and certain soluble factors are the mechanisms used by MSCs to mediate the immune response. Factors, such as IDO, TGF-β1, IFN-γ, IL-1β, TNF-α, IL-6, IL-10, PGE-2, HGF, HLA-G5, and others, are secreted by MSCs or released after interactions with target cells. As mentioned above, MSCs remain in a resting state, display anti-apoptotic properties, and maintain different cells such as hematopoietic stem cells (HSCs), thus contributing to tissue homeostasis. However, in an inflammatory environment such as that created by cytokines such as IFN-γ, TNF-α, IL-1α, and IL-1β, MSCs begin to exert their immunosuppressive effects and polarize, inhibiting the proliferation of effector cells and their production of cytokines. In this regard, IFN-γ is postulated as a “licensing” agent for MSC anti-proliferative action. MSCs may also acquire behavior as antigen-presenting cells (APCs) under specific concentrations of IFN-γ (Stagg et al. 2006; Uccelli et al. 2008). However, no consensus regarding what concentration of IFN-γ is more necessary for MSCs to show inhibitory or APC functions exists.
Likewise, TNF-α is another pro-inflammatory cytokine involved in the MSC immune response, and TNF-α enhances the effect of IFN-γ. IFN-γ, with or without the help of TNF-α, stimulates the production of IDO by MSCs, inhibiting the proliferation of activated T or NK cells and thus enhancing the homing potential and reparative properties of these cells; however, some potential risks are associated with the role of IFN-γ (Krampera et al. 2006; Sivanathan et al. 2014). Some authors have maintained that the immunomodulatory properties of MSCs are comparable, while others have argued that MSCs of different tissue origins or species cannot have equivalent and comparable immunomodulatory properties (Najar et al. 2010; Yoo et al. 2009; Ricciardi et al. 2012; Krampera 2011). For example, MSCs from perinatal sources (umbilical cord and amniotic membrane) show a higher immunomodulatory capacity, differential gene expression profiles, and paracrine factor secretion compared to BM-MSCs (Wegmeyer et al. 2013). Lee and colleagues found that HLA-G, a specific MHC-I antigen that is critical for maintaining the immune-tolerant state of pregnancy and that is a contributing factor to the induction of more substantial immunosuppression, is strongly positive only in placenta-derived MSCs (PD-MSCs) (Lee et al. 2012a). This is in contrast to BM-derived MSCs and Ad-MSCs and suggests that the immunophenotype of PD-MSCs may be superior to other MSCs in terms of their immunosuppressive function (Hunt et al. 2005). Nonetheless, some authors claimed that BM-derived MSCs were more immunomodulatory than PD-MSCs (Fazekasova et al. 2011). And others concluded that the immunomodulatory capacities of BM-derived MSCs and Ad-MSCs are similar but that differences in cytokine secretion cause Ad-MSCs to have more potent immunomodulatory effects than BM-derived MSCs (Melief et al. 2013).
Bartholomew and colleagues (Bartholomew et al. 2002) showed that allogeneic MSCs prolonged skin graft survival in baboons. Mouse MSCs have been used in related experiments; these cells use inducible nitric oxide synthase (iNOS) for immunosuppression instead of IDO. These findings indicate that MSCs differ between species (Ren et al. 2009). Since then, several preclinical models have been used to analyze the biological effects of MSCs and their ability to modulate immune responses, considering that not all animal models mimic human diseases. Once more, these differences could be due to isolation procedures, to culture methodology, or, more likely, to differences in the microenvironments where cells reside. These and other findings lead us to conclude that determining whether these differences may be relevant for clinical applications and whether MSCs of a particular tissue type are more appropriate for specific therapies or diseases.
Preclinical Applications
Preclinical models are essential for clinicians, researchers, and both national and international regulatory agencies to demonstrate the safety and efficacy of MSC-based therapies (Krampera et al. 2013). Because MSCs can exert immunomodulatory properties and act on different immune cells in vitro and in vivo, these cells have begun to be used against autoimmune diseases based on multiple autoimmune experimental models. Pioneer studies in experimental autoimmune encephalomyelitis (EAE), a model for multiple sclerosis, reported that MSCs derived from numerous tissue origins show efficacy against neurodegenerative disorders (Zappia et al. 2005; Rafei et al. 2009; Constantin et al. 2009; Bai et al. 2009; Zhang et al. 2005). BM-MSC and UCB-MSC treatments have improved clinical and laboratory parameters in systemic lupus erythematosus (SLE) (Sun et al. 2010). Furthermore, ameliorating effects have been observed in experimental mouse models of rheumatoid arthritis (RA) (González et al. 2009). Diabetes is another autoimmune disorder in which MSCs have been employed (Jurewicz et al. 2010; Lee et al. 2006). Although promising results and progress have been observed in this field, the interspecies differences, and contradictory experimental outcomes, and the inability to recreate the complete pathophysiology of some diseases make it necessary to search for new animal models for comparable results.
MSC-Based Therapy for Autoimmune Diseases
The MSCs are being used to facilitate the engraftment of transplanted HSCs and treat graft-versus-host disease (GvHD) after allogeneic hematopoietic stem cell transplantation (HSCT) based on their immunomodulatory properties and provide appropriate conditions. However, preclinical and clinical experiments with MSCs do not always show similar results for the prevention and treatment of GvHD. In a study using a mouse model of GvHD (Sudres et al. 2006), MSCs suppressed alloantigen-induced T-cell proliferation in vitro in a dose-dependent manner but yielded no clinical benefit regarding the incidence or severity of GvHD. Instead, when UCB-MSCs were administered in weekly doses in a xenogenic model of GvHD, a marked decrease in human T-cell proliferation was observed, and none of the mice developed GvHD. No therapeutic effect was obtained when UCB-MSCs were administered at the onset of GvHD (Tisato et al. 2007). In the same line of research, serial infusions of mouse AD-MSCs could efficiently control the lethal GvHD that occurred in recipients transplanted with haploidentical hematopoietic grafts (Yañez et al. 2006). Mixed results have also been achieved in human patients. One study found that the co-transplantation of culture-expanded MSCs and HSCs from HLA-identical sibling donors after myeloablative therapy accelerated hematopoietic engraftment (Lazarus et al. 2005); however, a significant reduction of GvHD symptoms was not shown, although the incidence or severity of GvHD did not increase. Koç and colleagues (Koç et al. 2000) reported a positive impact of MSCs on hematopoiesis; rapid hematopoietic recovery was observed in a clinical study with breast cancer patients who received autologous HSCT together with autologous MSCs.
Therapeutic effects have also been reported at the onset of GvHD, such as the case of a 9-year-old boy with severe treatment-resistant GvHD after allogeneic HSCT for acute lymphocytic leukemia who received haploidentical MSCs derived from his mother. He showed improvement after two administrations of MSCs (Le Blanc et al. 2004). Similar results have been obtained in steroid-refractory GvHD pilot studies with BM-MSCs and AD-MSCs (Ringdén et al. 2006; Fang et al. 2006). Several infusions appear to be required to maintain the level of active immunomodulation by MSCs. Similarly, the expression of pro-inflammatory cytokines such as IFN-γ in the environment at the time of MSC administration is required by these cells to exert their immunosuppressive effect. A lack of MSC “licensing” can result in the absence of the desired therapeutic effect.
While evidence that MSCs are effective in combination or after HSCT in specific hematological and non-hematological diseases has been shown, adverse reactions and risk factors intrinsic to this practice have been reported. In a pilot study, HLA-identical sibling-matched HSCs were transplanted with or without MSCs in hematological malignancy patients. Although MSCs were well-tolerated and this treatment effectively prevented GVHD, six patients (60%) in the MSC group and three (20%) in the non-MSC group had 3-year disease-free survival rates of 30 and 66.7%, respectively. The relapse rate in the experimental group was higher than that in the control group, suggesting that MSCs may impair the therapeutic graft-versus-leukemia (GVL) effect (Ning et al. 2008). In vitro and in vivo studies regarding the relationship between the immunosuppressive properties of MSCs and the stimulation of cancer growth have been performed. Mouse MSCs from the BM, spleen, and thymus injected together with a genetically modified tumor cell vaccine could equally prevent the onset of an anti-tumor memory immune response, thus leading to tumor growth in normally resistant mice (Krampera et al. 2007). In another in vivo experiment with a murine melanoma tumor model, the authors observed that the subcutaneous injection of B16 melanoma cells led to tumor growth in allogeneic recipients only when MSCs were co-injected (Djouad et al. 2003). The functions of MSCs can be influenced by the existing microenvironment, making them acquire supportive properties toward cancer cells and decrease immune reactions (Galiè et al. 2008). Therefore, potential risks related to the growth support and enhancement of undetected or “resident” cancer exist, and the administration of MSCs in these patients must be thoroughly evaluated.
MSCs for Cancer Treatment
The therapeutic benefits of MSCs have prompt their use in cell-based strategies to treat different diseases, including cancer (Hmadcha et al. 2020). Similar to damaged tissues, tumors exert chemoattractant effects on MSCs that influence their recruitment to tumor sites. The CXCL12/CXCR4 axis is one of the most frequently studied signaling pathways in mobilizing MSCs to the tumor microenvironment (Gao et al. 2009; Xu et al. 2009; Lourenco et al. 2015; Wobus et al. 2015; Kalimuthu et al. 2017). However, the ability of MSCs to migrate toward cancerous tissue is also controlled by other agents, including diffusible cytokines, such as IL-8, growth factors such as TGF-β1 or platelet-derived growth factor (PDGF), and extracellular matrix molecules, such as matrix metalloproteinase 2 (MMP-2) (Nakamizo et al. 2005; Birnbaum et al. 2007; Bhoopathi et al. 2011). Once the tumor niche is reached, MSCs interact with cancer cells via direct and indirect mechanisms that affect tumor development. The paracrine action of MSCs is one of the main mechanisms involved in cancer regulation and is mediated by multiple factors, including growth factors and cytokines. These paracrine factors affect cellular processes involving the tumor cell cycle (e.g., cell proliferation), cell survival, angiogenesis, and immunosuppression/immunomodulation, allowing MSCs to regulate cancer.
The paracrine agents can be directly secreted into the extracellular space or packaged into EVs for spreading in the tumor milieus (Rani et al. 2015). The interaction of MSCs with the tumor cell cycle is the most commonly accepted process by which MSCs exert their therapeutic effects (Fathi et al. 2019). By inhibiting proliferation-related signaling pathways, such as the phosphatidylinositol 3-kinase/protein kinase B (PI3K/AKT), MSCs can induce cell cycle arrest and reduce cancer growth (Lu et al. 2019). In addition, MSCs can undergo differentiation into other cell types, such as cancer-associated fibroblasts (CAFs), to directly contribute to cancer progression (Jotzu et al. 2011; Barcellos-de-Souza et al. 2016; Aoto et al. 2018).
Accumulating evidence indicates that the cross-talk between MSCs and tumor cells results in both pro-tumor and anti-tumor effects, raising safety concerns for clinical application in oncology (Barkholt et al. 2013). The discrepancies in the ability of MSCs to promote or suppress tumor development may be attributable to differences in experimental tumor models, MSC tissue source, dose or timing of the MSC treatment, cell delivery method, control group chosen, and other experimental conditions (Bortolotti et al. 2015; Bajetto et al. 2017). In this regard, a study demonstrated that direct (cell-to-cell contact) or indirect (released soluble factors) interaction between umbilical cord MSCs and glioblastoma stem cells produces divergent effects on cell growth, invasion, and migration (Bajetto et al. 2017). Moreover, the application of MSCs for cancer patients is a more complex situation in which other factors have to be taken into consideration. For instance, the pathological conditions of each patient may induce cellular and molecular changes in MSCs that interfere with their therapeutic effects (Capilla-González et al. 2018; Pérez et al. 2018; Rivera et al. 2019). Therefore, it is important to be cautious while drawing conclusions from a single study regarding the therapeutic effects of MSCs in cancer.
The Anti-tumor Activity of MSCs
Although compelling evidence shows a pro-tumorigenic role of MSCs, these cells also have potent tumor-suppressive effects that have been exploited as cancer therapeutics. Previous studies have demonstrated that MSCs release cytotoxic agents, such as TNF-related apoptosis-inducing ligand (TRAIL) that selectively induces apoptosis in different types of cancer (Wiley et al. 1995; Hao et al. 2001; Takeda et al. 2001; Akimoto et al. 2013). Recently, a report indicated that BM-derived MSCs promote apoptosis and suppress the growth of glioma U251 cells through downregulation of the PI3K/AKT signaling pathway (Lu et al. 2019). Likewise, intravenously transplanted MSCs were found to suppress tumor growth by blocking AKT activation in a Kaposi sarcoma mouse model (Khakoo et al. 2006). In mammary carcinomas, umbilical cord MSCs attenuated cell growth and triggered apoptosis through inhibiting ERK1/2 and AKT activation (Ganta et al. 2009). The Wnt signaling pathway has also been involved in the ability of MSCs to inhibit tumor cell proliferation (Qiao et al. 2008a, b). A mechanistic study of the inhibitory effect of MSCs on breast cancer cells demonstrated that the protein dickkopf-1 (Dkk-1) released from MSCs blocks tumor growth via depression of Wnt signaling (Qiao et al. 2008a). In contrast to investigations describing the pro-angiogenic effect of MSCs (Zhang et al. 2013; Li et al. 2016), the anti-tumor activity of MSCs via the inhibition of tumor angiogenesis has also been documented. A study reported that BM-derived MSCs restrict vascular growth in 1Gli36 glioma xenograft through the downregulation of the PDGF/PDGFR axis (Ho et al. 2013). In particular, the expression of PDGF-BB protein was significantly reduced in tumor lysates when treated with MSCs, which correlated with reduced levels of activated PDGFR-b and the active isoform of its downstream target AKT (Ho et al. 2013).
In a melanoma mouse model, transplanted MSCs inhibited angiogenesis in a concentration-dependent manner, leading to reduced tumor growth (Otsu et al. 2009). Confirmatory in vitro studies suggested that the anti-angiogenic effect was due to MSC-induced capillary degeneration (Otsu et al. 2009). Furthermore, MSCs have elicited anti-tumor immune responses through released inflammatory mediators, such as the multifunctional cytokine TGF-β. Like several signaling molecules, TGF-β plays a dual role in cancer development (Bierie and Moses 2006). Besides the aforementioned pro-tumor functions, TGF-β signaling exhibits suppressive effects in cancer (Dong et al. 2007; Guasch et al. 2007). While the expression of type III TGF-β receptor (TbRIII) decreases during breast cancer progression, restoring TbRIII expression suppresses tumorigenicity (Dong et al. 2007).
The Pro-tumor Activity of MSCs
The pleiotropic effects of MSCs that promote tissue repair and regeneration may also confer pro-tumor functions to these cells. For instance, metastatic human breast carcinoma cells were found to induce the secretion of the chemokine (C-C motif) ligand 5 (CCL5) from MSCs, which enhanced tumor invasion (Karnoub et al. 2007). Seminal reports demonstrated that MSCs could also inhibit apoptosis in tumor cells by secreting pro-survival factors such as VEGF and bFGF (König et al. 1997; Dias et al. 2002). Numerous studies converged on the finding that MSCs contribute to cancer pathogenesis by releasing inflammatory factors that promote immunosuppressive effects. For example, an in vitro study showed that MSCs isolated from gastric tumors mediate cancer progression through the secretion of IL-8 (Li et al. 2015). This pro-inflammatory chemokine favors the recruitment of leukocytes. It is known that recruited leukocytes, such as macrophages and neutrophils, facilitate cancer initiation and progression (Guo et al. 2017; Powell et al. 2018). Similarly, MSCs are able to secrete TGF-β that promotes macrophage infiltration at the tumor site and facilitates tumor escape from immune surveillance (Kim et al. 2006; Byrne et al. 2008). Compelling evidence indicates that MSCs can also support tumor angiogenesis, an essential process in cancer progression that supplies tumors with oxygen and nutrients. For instance, MSCs recruited in breast and prostate tumors were found to increase the expression of angiogenic factors, including TGF-β, VEGF, and IL-6, which contribute to tumor growth and vascularization (Zhang et al. 2013).
Similarly, a correlation between increased expression of TGF-β and higher microvessel density was observed in hepatocellular carcinomas of mice receiving intravenous injections of human MSCs (Li et al. 2016), which further supports that MSCs may enhance tumor angiogenesis via TGF-β. Furthermore, MSCs can also respond to soluble factors secreted from cancer cells and differentiate into CAFs, a cell type within the tumor microenvironment capable of promoting tumorigenesis (Mishra et al. 2008). In particular, TGF-β secreted from cancer cells plays a critical role in the differentiation of MSCs into CAFs (Jotzu et al. 2011; Barcellos-de-Souza et al. 2016; Aoto et al. 2018).
It is known that the transition of MSCs into CAFs contributes to tumor progression in part by their active secretome. Profiling of the secretome shows that it is rich in many bioactive molecules, including immune-modulating agents (CXCL12, granulocyte-macrophage colony-stimulating factor), pro-angiogenic factors (VEGF, TGF-β, PDGF), pro-survival factors (hepatocyte growth factor, insulin-like growth factor 1, interleukin 6), and extracellular matrix modulators (MMP, tissue inhibitor of metalloproteinases) among others (Kalluri 2016). Cell engulfment has also been identified as an interacting process between MSCs and cancer cells that enhances tumor aggressiveness. A recent report demonstrated that breast cancer cell engulfment of MSCs leads to changes in the transcriptome profile of tumor cells. These changes are mainly associated with oncogenic pathways. This MSC engulfment enhances epithelial-to-mesenchymal transition, stemness, invasion, and metastasis of breast cancer (Chen et al. 2019).
The Imprint of Disease on MSCs
One of the strategies to obtain MSCs for therapeutic purposes is an autologous approach. These cells are collected from patients by more or less invasive methods, isolated, seeded in culture under good manufacturing practice (GMP) quality standards, and re-injected into the patient. Nevertheless, when body’s repair mechanisms are insufficient or ineffective, this treatment results in a homeostatic imbalance in the organism, producing degradation and disease and compromising the pool of endogenous cells, thus resulting in low efficacy. Some conditions/diseases provoke changes in the BM microenvironment, which is one of the primary sources of MSCs, thus producing changes in the endogenous pool of MSCs and altering their biological features (Mazzanti et al. 2008). MSCs from patients with acute myeloid leukemia showed abnormal biological properties, including morphological heterogeneity, limited proliferation capacity, and impaired differentiation and hematopoiesis supportability (Zhao et al. 2007).
MSCs derived from patients with multiple myeloma showed impaired immune-inhibitory effects on T cells, decreasing their osteogenic potential (Li et al. 2010). Poor proliferation, differentiation potential, and cytokine release defect were found in BM-derived MSCs derived from patients with aplastic anemia, another hematopoietic disorder (Chao et al. 2010; Bacigalupo et al. 2005). Although the mechanisms remain unknown, MSCs appear to be involved in autoimmune pathologies. For instance, MSCs derived from patients with autoimmune diseases display the following altered functions; MSCs from rheumatoid arthritis (RA) patients have an impaired ability to support hematopoiesis and lower proliferative and clonogenic potentials (Papadaki et al. 2002; Kastrinaki et al. 2008). MSCs from immune thrombocytopenic purpura (ITP) patients have a reduced proliferative capacity and a lower inhibitory effect on T-cell proliferation than MSCs from healthy donors (Pérez-Simón et al. 2009). MSCs from systemic lupus erythematous (SLE) patients display deficient growth, abnormal morphology, and upregulated telomerase activity (Nie et al. 2010; Sun et al. 2007). MSCs from systemic sclerosis (SSc) patients display early senescence (Cipriani et al. 2007). In metabolic diseases such as diabetes, alterations in autologous MSCs have also been documented.
A study using MSCs from type 2 diabetic mice showed that the number of these cells was diminished and their proliferation and survival abilities were impaired in vitro. Moreover, diabetic MSC engraftment produced only limited improvement in the diabetic subjects and could not produce the same therapeutic outcomes as in their nondiabetic counterparts in vivo (Shin and Peterson 2012). Advanced glycation end products (AGEs) accumulate in the tissues of aged people, and these products are involved in diabetes and musculoskeletal diseases. In 2005, Kume and colleagues (Kume et al. 2005) investigated the effect of AGEs on MSCs. They showed that AGEs inhibited MSC proliferation, induced MSC apoptosis, and interfered with MSCs’ differentiation into adipose tissue, cartilage, and bone. Type 2 diabetes-derived Ad-MSCs have been found to have functional impairments in their multilineage potential and proliferative capacity because of prolonged exposure to high glucose concentrations (Cramer et al. 2010).
Diabetic-derived Ad-MSCs have an altered phenotype related to plasminogen activator inhibitor-1 (PAI-1) expression levels and display reduced fibrinolytic activity (Acosta et al. 2013), which suggests that the immunogenicity of MSCs could have associated effects on the coagulation system (Wang et al. 2012; Moll et al. 2012). Thus, MSC-based therapy could lead to thrombotic events in particular recipients. Although the possibility of healing with autologous cells is desirable, little is known regarding the influence of different disease states and concomitant medications on MSCs (Benvenuti et al. 2007; Lee et al. 2009). Thus, although the use of autologous MSCs for cell therapy is widespread, their use in humans must be handled with extreme caution. Researching and analyzing both the risks and benefits of this therapy in individual patients and for each disease are necessary.
Considerations for Clinical Applications
Several clinical trials are in progress to ensure the safety and efficacy of MSCs used as medicaments. For cell-based products, it must be considered that cells are living products and that their interactions with body fluids remain unclear (Acosta et al. 2013; Moll et al. 2014). Phase 1 clinical trials are the first step in investigating a new drug. They include pharmacokinetic and pharmacodynamic studies in which the patient’s safety plays an essential role in the development of medicaments. The primary goal of phase 2 clinical trials is to provide preliminary information regarding the drug efficacy and safety supplement data obtained in phase 1 trials. For efficacy and effectiveness issues, other advanced phases are mandatory. In all cases, one cannot consider these issues (efficacy nor effectiveness) unless phase 3 clinical trials are developed (García-Bernal et al. 2021; Hmadcha et al. 2020; Escacena et al. 2015). Usually, safety evaluations are based on possible complications derived from the procedure in a time-dependent manner after administering the cells. Efficacy parameters focus on the improvement of clinical effects at a given time. MSC-based cell therapy is a relatively new therapeutic option for certain diseases, and data regarding the long-term monitoring of patients remain lacking.
Nevertheless, the administration of MSCs is considered a feasible and safe procedure with no adverse events reported. However, the risks associated with stem cell therapy (Herberts et al. 2011) must be considered because these risks increase the probability of an adverse event. The cell source, donor origin, product manufacturing, and recipient disease status are important factors related to the safety and efficacy of MSCs. In this regard, the use of bovine proteins in the medium used to culture these cells (Horwitz et al. 2002) and the observed formation of ectopic tissue in animal models (Breitbach et al. 2007; Kunter et al. 2007), as well as malignant transformation (Wang et al. 2005; Røsland et al. 2009) and immune responses, must be evaluated before wider clinical applications and registration are accepted.
Safety Concerns
Cell therapy is incredibly complex due to the nature of the product. The mode of action is not always clear, and the potency tests are imprecise, by which it might not be possible to predict the risks thoroughly. When considering the use of expanded MSCs ex vivo for clinical applications, it is necessary to consider a series of potential risks that could affect the cellular product.
The administration of stem cells could affect the host’s immune system. These cells could directly influence the immune system (e.g., pro-inflammatory environment) or have an immunomodulatory effect. Although MSCs have been considered immune-privileged in this regard, long-term exposure to the culture medium can make them more immunogenic by positively regulating the normal set of histocompatibility molecules (Moll et al. 2011, 2014). On the one hand, the allogeneic use of the cells entails a greater risk of rejection by the immune system. This rejection could lead to a loss of the function of the administered cells, and consequently, their therapeutic activity could be compromised. The use of immunosuppressants could limit these risks, but, in turn, could cause adverse reactions due to immunosuppressive medication.
On the other hand, MSCs isolated from healthy donors have shown uniform and consistent properties, while patients with some degenerative and inflammatory disease differ in their biological and functional characteristics (Capilla-González et al. 2018; Rennert et al. 2014). In this regard, studies with MSCs from diabetic patients suggest that the hyperglycemic environment and other metabolic disorders associated with diabetes affect the endogenous cellular reserve and their proliferation, differentiation, and angiogenic capacity, among other cellular characteristics (Minteer et al. 2015; Rennert et al. 2014; George et al. 2018; Moll et al. 2019). Once infused in the recipient, the cells come into direct contact with the tissues, bloodstream, and other host cells; the cell-recipient interaction process still needs a thorough investigation and characterization.
Physiologically, MSCs reside in the perivascular compartment of almost every tissue (Bianco et al. 2008; Crisan et al. 2008); however, one of the hurdles to the sustained success of their therapeutic effect is early cell loss. This is primarily due to the incompatibility responses after systemic infusion of cells, a reaction termed as instant blood-mediated inflammatory reaction (IBMIR) suggesting that the immune and inflammatory system reacts to cells that generally are not in contact with the blood circulation (Gupta et al. 2014; Moll et al. 2011, 2014, 2019, 2020; Bianco et al. 2008; Crisan et al. 2008; Nilsson et al. 2014). Even more, it has been further shown that different MSC products display varying levels of highly pro-coagulant tissue factor, a decrease in tissue plasminogen activator (tPA), or an increase in PAI-1 and may adversely trigger the IBMIR or microthrombosis in the target tissue (Acosta et al. 2013; Moll et al. 2019). Although MSCs are considered to be safe, they can promote fibrinolysis (Hashi et al. 2007; Neuss et al. 2010; Moll et al. 2020).
Safety and efficacy are the basic pillars that support the viability of clinical application to treat any disease. Except for hematopoietic stem cell transplants, stem cell therapies used to treat any disease are considered medicinal products; therefore, their development, approval, and use must be per the specific standards established nationally and internationally for such medicines. Thus, regulatory authorities guarantee the safety of the studies (Fig. 2).
The MSCs possess a wide range of paracrine effects, including anti-inflammatory, immunomodulatory, trophic, anti-apoptotic, and anti-fibrotic properties. These are mostly mediated by molecules released by MSCs, but also by direct cell-cell contacts. For cell-based therapy, the paracrine properties of MSCs have beneficial effects for the patient. However, the interaction between MSCs and the host can lead to adverse side effects. (Re-published from Soria-Juan 2019, article published under CC-BY terms). Abbreviations: B-cell B lymphocyte, CXCL C-X-C motif chemokine ligand, DC dendritic cell, G-CSF granulocyte colony-stimulating factor, HGF hepatocyte growth factor, IL interleukin, INF-γ interferon-γ, MSC mesenchymal stem cells, NK natural killer cells, T-cell T lymphocyte, TGF-α transforming growth factor α, Treg regulatory T cell
Cell Manufacturing for Clinical Use
Except for hematopoietic stem cell transplants, stem cell therapies used to treat any disease are considered drugs; therefore, their development, approval, and use must be per the specific standards established for such medicines nationally and internationally. In this context, MSCs are now considered as “cellular medicament” and are called advanced therapy medicinal products (ATMPs) and are under regulation No. 1394/2007 (Escacena et al. 2015; Gálvez et al. 2013). Relating production processes and development staff, clinicians and researchers must achieve GMP procedures under European regulations (Sensebé et al. 2013; Gálvez et al. 2014). Currently, no standardized manufacturing platform exists, although most facilities employ standard release criteria to measure sterility, viability, and chromosomal stability to meet European or FDA regulations (Phinney 2012; Iglesias-López et al. 2019).
Although regulation establishes common parameters to follow, different protocols are used to isolate these cells, and the processes, plating densities, and reagents used cause the results to differ from each other. Donor selection in terms of age and disease status is another variable to consider due to known MSC donor-to-donor heterogeneity (Phinney et al. 1999). The cell source is another important factor related to the efficacy of the product. As reported previously, MSCs derived from different tissues do not consistently achieve the same level of efficacy. Additionally, culture media used for the production of MSCs could affect the basic characteristics of cells; thus, designing a fully defined medium free of animal and human origins is crucial.
Thus far, no MSC-based medicine product has marketing authorization in the European Union, although four gene and cell-based products have a valid marketing authorization awarded by the EMA. However, since 2011, three MSC products have received marketing approval in other regions (Ancans 2012). The MSCs’ field continues its upward progression, with a growing number of established companies established and ongoing clinical trials, but remaining challenges must be overcome. Bottlenecks exist regarding donor selection, cell sources, isolation protocols, culture media used, open-culture systems, bioreactors, and recipient disease status. Establishing a standardized and comparable process is also crucial to ensure biological and functional equivalence between product lots.
Considerations for Cellular Medicament
General Considerations
The cell expansion and culture protocol are not standardized, although the regulatory agencies (e.g., EMA, FDA) recommend a set of standards to be followed to produce cellular drugs. Currently, there is no protocol or universal definition for stem cell culture and expansion. The different sources of origin, and the different methodologies for obtaining tissue cells, make it very difficult to compare research groups in search of the fastest, most effective, economical, high-yielding, efficient, and clinical-grade quality method. Cell viability after the infusion is poor; in this regard, it is known that very few cells survive after infusion. Although the in vivo follow-up is ethically and technically complicated, it is necessary to continue investigating this line to understand the intrinsic mechanisms of integrating the infused cells in the concrete microenvironment.
The cellular dose to obtain the desired effects is also unknown. Investigations with HSCs have revealed that the administration of sufficient cells promotes faster cell recovery and reduces hospitalizations (Mohty et al. 2011). Preclinical studies using murine animal models have established a minimum dose of 1 × 106 cells/kg of weight, a quantity necessary to obtain quantifiable but weak benefits (Shabbir et al. 2009; Mastri et al. 2012). The dose for cellular treatment is probably influenced by the patient’s body weight and the biodistribution of paracrine factors secreted by MSCs in the human body; however, most clinical trials use a similar cell dose (Tan et al. 2012; Jiang et al. 2011). The doses used have been insufficient in most cases to show clear therapeutic benefits. This fact leads us to design future trials to test different cell doses. Likewise, the frequency of administration is currently unknown. The effectiveness of cell therapy is probably related to the number of others applications (Cobellis et al. 2008; Teraa et al. 2015; Molavi et al. 2016), similar to that established with conventionally used medications. The timing and the ideal number of cellular applications are still unknown. Since conventional medicines are depending on the dose, cellular therapy may need to be adjusted accordingly. The most suitable cell type remains a challenge for regenerative medicine. Knowing which cell type is most appropriate for each particular pathology or if a combination of these would be more recommended is another big issue in cell therapy.
The method for the cellular administration continues without giving conclusive results because cell viability must be preserved as much as possible, and compromised tissue is often associated with ischemia, fibrosis and inflammation, which could impair cell survival, therapeutic delivery of stem cells in the distal areas to the damaged tissue appear to offer some advantage. There are no conclusive findings of a more significant benefit within the existing modes of administration, so this is another variable to have into account for future clinical trials. The desired therapeutic effect depends on many factors since mechanism of action of stem cells in tissue regeneration is likely to be multifaceted. Cellular competition can be dictated by the ability of injected cells to migrate, survive, integrate, differentiate, and produce functional paracrine mediators (“cell-cell interactions”). It is known that many diseases (e.g., diabetes, cancer, etc.) affect the phenotypic and therapeutic properties of stem cells. Finally, for the therapy to be effective, the recipient tissue must respond favorably to the injected cells, which would result in the activation of endogenous regeneration mechanisms (Lee 2010). Understanding integration of the exogenous mechanisms (injected cells) with the endogenous (host) will play a decisive role in the future clinical use of adult stem cells (Acosta et al. 2013; Moll et al. 2019).
Attempts to Improve the Therapeutic Outcomes of Cellular Medicament
Advances in the production compliance under good manufacturing practices (GMP) standards of more sophisticated cellular products are now opening up the way for the second generation of cell therapy clinical trials. One of the reasons why unmodified MSCs have not shown the therapeutic efficacy expected in human clinical trials is that, after their systemic infusion (intravenous), these cells become trapped in the vascular filters (fundamentally the liver and lung) and only a small percentage reach the target tissues. Therefore, strategies must be designed that favor migration, nesting, and localization in the inflammatory and/or infectious focus to increase their effectiveness. Biodistribution and long-term follow-up of these cells in animal models show that only a few cells remain after long periods. This will support the idea that most of the effects of MSCs are based on a “hit and run effect.”
To increase the concentration of ATMPs in the injured tissue, the CD44 antigen on MSCs’ cell membrane by enzymatic fucosylation has been converted into hematopoietic cell E-selectin/L-selectin ligand (HCELL) glycoform (Dimitroff et al. 2001; Pachón-Peña et al. 2017). This molecular change favored the migration of the MSCs to the inflamed tissues (Sackstein et al. 2008; García-Bernal et al. 2020). This method, called glycosyltransferase-programmed stereo substitution (GPS) of cell surface glycans, has been optimized for its clinical application so that the reagents used (glycosyltransferases and buffers) have been specifically formulated to preserve cell viability and phenotype (García-Bernal et al. 2021). Moreover, this modification not only increases the adhesion of the MSCs to the endothelium, but it also enhances their transmigration through it by activating the alfa4/beta1 integrin in the absence of chemokine stimulation (López-Lucas et al. 2018). Therefore, this modification by fucosylation could improve the efficacy of the treatment with MSCs by increasing the migratory capacity of the cells to the inflamed tissues after being administered systemically (García-Bernal et al. 2020). Other strategies may include expressing CXCR4. These strategies will help to engineer new generation of MSCs for use when both increased migration and targeting and an increased power are required. Expression of the CXCR4 receptor will increase the migration of the MSCs toward the inflammatory focus (Zhu et al. 2021). On the other hand, the co-expression of the anti-inflammatory cytokine IL-10 and/or the anti-infectious cytokine IL-7 will increase the anti-inflammatory effect (IL-10) and even the anti-infective effect (IL-7) (Mao et al. 2017).
Furthermore, the extensive use of fetal bovine serum (FBS) in the MSC-expansion media represents an explicit limitation for the introduction of ATMP at the clinical level. Currently, cell expansion is carried out in culture media supplemented with FBS (Gottipamula et al. 2013). The SFB used must be a clinical-grade (free of animal pathogens). Associated with the growing demand for MSCs, this has led to a series of technical and ethical conditions of production (using a high number of bovine fetuses) and geographic (zones free of prion diseases), which have had an impact on their price (Kinzebach and Bieback 2013; Wessman and Levings 1999). The substitution of FBS by human serum and platelet lysate also represents technical limitations mainly related to the supply of human material and the absence of uniformity of the lots. All these considerations force the development of robust processes of production of MSC in chemically defined culture media free of animal and human components. These media are supplemented with recombinant proteins (albumin, insulin, TGF-β, and bFGF), iron, selenium, and an antioxidant system (2-mercaptoethanol) (Badenes et al. 2016; Jayme and Smith 2000). Although several serum-free media are found in the literature and market (Chase et al. 2010; Ishikawa et al. 2009), there is still no effective means of functioning.
The therapeutic efficacy of MSCs has been further optimized by genetically modifying MSCs to produce trophic cytokines or other beneficial gene products in numerous preclinical models by transfecting MSCs with viral or non-viral vectors (Jiang et al. 2006; Haider et al. 2008; Kim et al. 2012a, b). These MSCs have been successfully modified to express therapeutic peptides and proteins to express therapeutic peptides and proteins in animal models (Zhou et al. 2021). For example, MSCs expressing thioredoxin-1 (Trx1, a potent antioxidant, transcription factor, and growth factor regulator) improved cardiac function in post-myocardial infarction rat models (Suresh et al. 2015). Simultaneous overexpression of Akt and Ang-1 in BM-derived MSC not only enhanced their reparability of the infarcted myocardium with sustained beneficial effect (Jiang et al. 2006, 2008), but it also led to non-hypoxic stabilization of HIF-1 to enhance their endothelial commitment (Lai et al. 2012a) and increased their proliferation potential via the involvement of miR-143 (Lai et al. 2012b). The MSCs expressing IL-12 showed potent anticancer activity against melanoma, breast cancer, and hepatoma (Gao et al. 2010; Han et al. 2014). In addition, interferon-γ-expressing MSCs inhibited tumor growth in mouse models of neuroblastoma and lung carcinoma (Relation et al. 2018; Seo et al. 2011). Similar to these advances achieved in animal models, several MSC-based therapies are under clinical development.
Both viral and non-viral vectors, however, have some limitations (Kim and Haider 2001). Non-viral vectors exhibit transient gene expression and low transfection efficiency. In contrast, viral transduction is associated with an increased risk of chromosomal instability, insertional mutagenesis, and proto-oncogene activation, despite the inherent high transfection efficiency (Cheng et al. 2019). It has been reported that adverse immune reactions induced by viral transduction impair transgene stability (Mingozzi and High 2013; Wang et al. 2018). Thus, limitations and adverse responses must be assessed when modifying MSCs by transfection. Several studies have sought to use MSCs derived from induced pluripotent stem cells (iPSCs) to obtain better expansion capacity. In fact, therapeutic transgenes could be inserted into iPSC-derived MSCs before MSC derivation. Such a strategy could eliminate insertional mutations and ensure stable expression of transgenes during a prolonged expansion (Zhao et al. 2015). Therefore, MSCs derived from iPSCs may be a renewable source of MSCs for theranostic applications. It is pertinent to mention that BM-derived MSCs have also been successfully reprogrammed to pluripotent status and used for the efficient repair of infarcted myocardium in an experimental animal model (Buccini et al. 2012).
Interestedly, CRISPR-Cas9 technology was used to obtain highly homogeneous MSCs. Genetic modifications of MSCs can be performed with greater efficiency and specificity using CRISPR/Cas9 technology (Gerace et al. 2017). This is faster, cost-efficient, and easier to use compared to alternatives such as transcription activator nucleases (TALENs) and zinc finger nucleases (ZFNs) (Faulkner et al. 2020). CRISPR/Cas9 has been widely employed in the stem cell field, particularly in MSC research, including knock-in, knock-out, gene activation, or gene silencing. In this regard, the application of CRISPR/Cas9 in MSCs has demonstrated its efficacy in treating diseases, such as myocardial infarction (Golchin et al. 2020). Targeting gene knock-in further promoted the differentiation capacity of MSCs and, in turn, improved the insufficiency of functional cells at local sites (Miwa and Era 2018). Genetically modified MSCs have been evaluated in clinical trials, such as the “TREAT-ME-1” clinical trial, an open-label, multicenter, first-in-human phase 1/2 trial, which aimed to evaluate the safety, tolerability, and efficacy of the application of genetically modified autologous MSCs-apceth-101 in patients with advanced gastrointestinal adenocarcinoma (von Einem et al. 2019). Despite promising advances in this field, further research is still needed to obtain solid evidence on the differentiation and regenerative potentials of MSCs in vivo. Undoubtedly, the next-generation sequencing and genotyping techniques could serve as valuable tools to improve the efficacy of targeting specific cell types for personalized medicine.
Besides, priming MSCs with exogenous small molecules has been found to boost their therapeutic function. Since current MSC manufacturing cannot meet the requirements of clinical trials in terms of production scale, the alternative is to enhance the function of limited cells by priming MSCs. Cell priming, or cell preconditioning, is a commonly used concept in the field of immunology and has been adapted to the stem cell arena (Lu et al. 2010; Haider and Ashraf 2012; Carvalho et al. 2013; Noronha et al. 2019) by ex vivo addition to MSCs of pro-inflammatory cytokines, such as IFN-γ, TNF-α, IL-1α, and IL-1β. More priming approaches are currently being proposed and optimized to improve MSC function, proliferation, survival, and therapeutic efficacy (Afzal et al. 2010; Kim et al. 2012a, b; Lu et al. 2012; Kim et al. 2018; Mead et al. 2020). In this regard and as mentioned before, other approaches are focused on enhancing the therapeutic effects of cell therapy products regulating their biological characteristics (Mangi et al. 2003; Mei et al. 2007; Lee et al. 2012b; Liao et al. 2017).
The beneficial effects of PDGF-BB to restore the defective phenotype of therapeutic MSCs derived from type 2 diabetic patients have been demonstrated. The pretreatment with PDGF-BB potentiates proliferation, migration, and homing of defective MSCs and recovers their impaired fibrinolytic ability. Furthermore, PDGF-BB has been found to exert its beneficial effects through the ERK-SMAD pathway. Therefore, the pretreatment with PDGF-BB represents a suitable strategy to produce more effective MSCs for autologous therapies (Capilla-González et al. 2018).
Concluding Remarks and Future Perspective
Treatments based on the use of human stem cells are novel and promising therapeutic alternatives for some diseases. Currently, the use of living cells as a medicinal product is becoming realistic. Cell therapy should be safe, pure, stable, and efficient. Cell-based products are more complex and depend on the physiological and genetic heterogeneity of the patient. Obtaining as much information as possible with the appropriate and available technology at our disposal is essential for ensuring the safety, reliability, quality, and effectiveness of the manufactured product. MSCs are leading the way into a new era of regenerative medicine, and their multifaceted features make them powerful candidates to become tools to treat several diseases. However, their indiscriminate use has resulted in mixed outcomes in preclinical and clinical studies. While MSCs derived from diverse tissues share some common properties, they markedly differ in terms of their differentiation abilities, growth rates, healing capacity, and gene expression profile.
Similarly, the disease status of donors and recipients is a critical factor to consider when using MSCs as therapeutic agents because factors such as the MSC behavior with body fluids and specific disease environments remain unclear. Available data suggest that some tissue-specific MSCs are more appropriate than others according to particular pathologies. Although no severe adverse effects related to the application and testing of MSCs in humans have been reported to date, some evidence has indicated that specific patient profiles are not suitable to be treated with these therapies. Thus, multiple bottlenecks for the standardization of therapeutic protocols exist. Future well-designed clinical trials, advanced-phase clinical trials (phase 3/4), and long-term monitoring of patients are crucial for obtaining additional information regarding the therapeutic use of MSCs.
Cross-References
Abbreviations
- Ad-MSCs:
-
Adipose tissue-derived MSCs
- AGEs:
-
Advanced glycation end products
- ATMPs:
-
Advanced therapy medicinal products
- bFGF:
-
Basic fibroblast growth factor
- BM-MSCs:
-
Bone marrow-derived MSCs
- CAFs:
-
Cancer-associated fibroblasts
- Cas9:
-
CRISPR-associated protein 9
- CCL:
-
Chemokine (C-C motif) ligand
- CFU-F:
-
Colony-forming unit fibroblast
- CRISPR:
-
Clustered regularly interspaced short palindromic repeats
- CXCL12:
-
C-X-C motif chemokine 12 (or SDF1)
- Dkk-1:
-
Dickkopf-1
- ECs:
-
Endothelial cells
- EMA:
-
European Medicines Agency
- ESCs:
-
Embryonic stem cells
- EVs:
-
Extracellular vesicles
- FBS:
-
Fetal bovine serum
- FDA:
-
Food and Drug Administration
- GM-CSF:
-
Granulocyte-macrophage colony-stimulating factor
- GMP:
-
Good manufacturing practice
- GPS:
-
Glycotransferase-programmed stereo substitution
- GVHD:
-
Graft-versus-host diseases
- GVL:
-
Graft-versus-leukemia
- HCELL:
-
Hematopoietic cell E-selectin/L-selectin ligand
- HGF:
-
Hepatocyte growth factor
- HLA-DR:
-
Human leukocyte antigen-DR isotype
- HLA-G5:
-
Human leukocyte antigen-G5
- HSCT:
-
Hematopoietic stem cell transplantation
- IBMIR:
-
Instant blood-mediated inflammatory reaction
- ICAM-2:
-
Intercellular adhesion molecule 2
- IDO:
-
Indoleamine-2,3-dioxygenase
- IFN-γ:
-
Interferon-gamma
- IGF-1:
-
Insulin-like growth factor 1
- IL:
-
Interleukin
- IL-1α:
-
Interleukin-1 alpha
- IL-1β:
-
Interleukin-1 beta
- iPSCs:
-
Induced pluripotent stem cells
- ISCT:
-
International Society for Cellular Therapy
- ITP:
-
Immune thrombocytopenic purpura
- LFA-3:
-
Lymphocyte function-associated antigen 3 (or CD58)
- MCP-1:
-
Monocyte chemoattractant protein 1
- MHC-HLA:
-
Major histocompatibility complex-human leukocyte antigen
- MMP-2:
-
Matrix metalloproteinase 2
- MSCs:
-
Mesenchymal stromal cells
- PAI-1:
-
Plasminogen activator inhibitor-1
- PDGF:
-
Platelet-derived growth factor
- PD-MSCs:
-
Placenta-derived MSCs
- PGE-2:
-
Prostaglandin-E2
- RA:
-
Rheumatoid arthritis
- SDF-1:
-
Stromal cell-derived factor 1
- SLE:
-
Systemic lupus erythematosus
- SSc:
-
Systemic sclerosis
- STC1:
-
Stanniocalcin-1
- TALENs:
-
Transcription activator nucleases
- TbRIII:
-
Type III TGF-β receptor
- TGF-β:
-
Transforming growth factor beta
- TNF-α:
-
Tumor necrosis factor alpha
- tPA:
-
Tissue plasminogen activator
- TRAIL:
-
TNF-related apoptosis-inducing ligand
- Trx1:
-
Thioredoxin-1
- TSG-6:
-
Tumor necrosis factor-stimulated gene-6
- UCB-MSCs:
-
Umbilical cord-derived MSCs
- VCAM-1:
-
Vascular cell adhesion protein 1
- VEGF:
-
Vascular endothelial growth factor
- ZFNs:
-
Zinc finger nucleases
References
Acosta L, Hmadcha A, Escacena N, Pérez-Camacho I, de la Cuesta A, Ruiz-Salmeron R, Gauthier BR et al (2013) Adipose mesenchymal stromal cells isolated from type 2 diabetic patients display reduced fibrinolytic activity. Diabetes 62(12):4266–4269. https://doi.org/10.2337/db13-0896
Afzal MR, Haider KH, Idris NM, Jiang S, Ahmed RPH, Ashraf M (2010) Preconditioning promotes survival and angiomyogenic potential of mesenchymal stem cells in infarcted heart via NF-кB signaling. Antioxid Redox Signal 12(6):693–702. https://doi.org/10.1089/ars.2009.2755
Akimoto K, Kimura K, Nagano M, Takano S, To'a Salazar G, Yamashita T, Ohneda O (2013) Umbilical cord blood-derived mesenchymal stem cells inhibit, but adipose tissue-derived mesenchymal stem cells promote, glioblastoma multiforme proliferation. Stem Cells Dev 22(9):1370–1386. https://doi.org/10.1089/scd.2012.0486
Ancans J (2012) Cell therapy medicinal product regulatory framework in Europe and its application for MSC-based therapy development. Front Immunol 3:253. https://doi.org/10.3389/fimmu.2012.00253
Aoto K, Ito K, Aoki S (2018) Complex formation between platelet-derived growth factor receptor β and transforming growth factor β receptor regulates the differentiation of mesenchymal stem cells into cancer-associated fibroblasts. Oncotarget 9(75):34090–34102. https://doi.org/10.18632/oncotarget.26124
Bacigalupo A, Valle M, Podestà M, Pitto A, Zocchi E, De Flora A, Pozzi S et al (2005) T-cell suppression mediated by mesenchymal stem cells is deficient in patients with severe aplastic anemia. Exp Hematol 33(7):819–827. https://doi.org/10.1016/j.exphem.2005.05.006
Badenes SM, Fernandes TG, Cordeiro CS, Boucher S, Kuninger D, Vemuri MC, Diogo MM et al (2016) Defined essential 8™ medium and vitronectin efficiently support scalable xeno-free expansion of human induced pluripotent stem cells in stirred microcarrier culture systems. PLoS One 11(3):e0151264. https://doi.org/10.1371/journal.pone.0151264
Bai L, Lennon DP, Eaton V, Maier K, Caplan AI, Miller SD, Miller RH (2009) Human BM-derived MSCs induce Th2-polarized immune response and promote endogenous repair in animal models of multiple sclerosis. Glia 57(11):1192–1203. https://doi.org/10.1002/glia.20841
Bajetto A, Pattarozzi A, Corsaro A, Barbieri F, Daga A, Bosio A, Gatti M et al (2017) Different effects of human umbilical cord mesenchymal stem cells on glioblastoma stem cells by direct cell interaction or via released soluble factors. Front Cell Neurosci 11:312. https://doi.org/10.3389/fncel.2017.00312
Barcellos-de-Souza P, Comito G, Pons-Segura C, Taddei ML, Gori V, Becherucci V, Bambi F et al (2016) Mesenchymal stem cells are recruited and activated into carcinoma-associated fibroblasts by prostate cancer microenvironment-derived TGF-β1. Stem Cells 34(10):2536–2547. https://doi.org/10.1002/stem.2412
Bari E, Perteghella S, Catenacci L, Sorlini M, Croce S, Mantelli M, Avanzini MA et al (2019) Freeze-dried and GMP-compliant pharmaceuticals containing exosomes for acellular mesenchymal stromal cell immunomodulant therapy. Nanomedicine (Lond) 14(6):753–765. https://doi.org/10.2217/nnm-2018-0240
Barkholt L, Flory E, Jekerle V, Lucas-Samuel S, Ahnert P, Bisset L, Büscher D et al (2013) Risk of tumorigenicity in mesenchymal stromal cell-based therapies--bridging scientific observations and regulatory viewpoints. Cytotherapy 15(7):753–759. https://doi.org/10.1016/j.jcyt.2013.03.005
Bartholomew A, Sturgeon C, Siatskas M, Ferrer K, McIntosh K, Patil S, Hardy W et al (2002) Mesenchymal stem cells suppress lymphocyte proliferation in vitro and prolong skin graft survival in vivo. Exp Hematol 30(1):42–48. https://doi.org/10.1016/s0301-472x(01)00769-x
Benvenuti S, Cellai I, Luciani P, Deledda C, Baglioni S, Giuliani C, Saccardi R et al (2007) Rosiglitazone stimulates adipogenesis and decreases osteoblastogenesis in human mesenchymal stem cells. J Endocrinol Investig 30(9):RC26–RC30. https://doi.org/10.1007/BF03350807
Benvenuto F, Ferrari S, Gerdoni E, Gualandi F, Frassoni F, Pistoia V, Mancardi G et al (2007) Human mesenchymal stem cells promote survival of T cells in a quiescent state. Stem Cells 25(7):1753–1760. https://doi.org/10.1634/stemcells.2007-0068
Bhoopathi P, Chetty C, Gogineni VR, Gujrati M, Dinh DH, Rao JS, Lakka SS (2011) MMP-2 mediates mesenchymal stem cell tropism towards medulloblastoma tumors. Gene Ther 18(7):692–701. https://doi.org/10.1038/gt.2011.14
Bianco P, Robey PG, Simmons PJ (2008) Mesenchymal stem cells: revisiting history, concepts, and assays. Cell Stem Cell 2(4):313–319. https://doi.org/10.1016/j.stem.2008.03.002
Bierie B, Moses HL (2006) Tumour microenvironment: TGFbeta: the molecular Jekyll and Hyde of cancer. Nat Rev Cancer 6(7):506–520. https://doi.org/10.1038/nrc1926
Birnbaum T, Roider J, Schankin CJ, Padovan CS, Schichor C, Goldbrunner R, Straube A (2007) Malignant gliomas actively recruit bone marrow stromal cells by secreting angiogenic cytokines. J Neuro-Oncol 83(3):241–247. https://doi.org/10.1007/s11060-007-9332-4
Bortolotti F, Ukovich L, Razban V, Martinelli V, Ruozi G, Pelos B, Dore F et al (2015) In vivo therapeutic potential of mesenchymal stromal cells depends on the source and the isolation procedure. Stem Cell Rep 4(3):332–339. https://doi.org/10.1016/j.stemcr.2015.01.001
Breitbach M, Bostani T, Roell W, Xia Y, Dewald O, Nygren JM, Fries JW et al (2007) Potential risks of bone marrow cell transplantation into infarcted hearts. Blood 110(4):1362–1369. https://doi.org/10.1182/blood-2006-12-063412
Bruder SP, Jaiswal N, Haynesworth SE (1997) Growth kinetics, self-renewal, and the osteogenic potential of purified human mesenchymal stem cells during extensive subcultivation and following cryopreservation. J Cell Biochem 64(2):278–294. https://doi.org/10.1002/(sici)1097-4644(199702)64:2<278::aid-jcb11>3.0.co;2-f
Buccini S, Haider KH, Ahmed RPH, Jiang S, Ashraf M (2012) Cardiac progenitors derived from reprogrammed mesenchymal stem cells contribute to angiomyogenic repair of the infarcted heart. Basic Res Cardiol 107(6):301. https://doi.org/10.1007/s00395-012-0301-5
Burlacu A, Grigorescu G, Rosca AM, Preda MB, Simionescu M (2013) Factors secreted by mesenchymal stem cells and endothelial progenitor cells have complementary effects on angiogenesis in vitro. Stem Cells Dev 22(4):643–653. https://doi.org/10.1089/scd.2012.0273
Byrne SN, Knox MC, Halliday GM (2008) TGFbeta is responsible for skin tumour infiltration by macrophages enabling the tumours to escape immune destruction. Immunol Cell Biol 86(1):92–97. https://doi.org/10.1038/sj.icb.7100116
Cagliani J, Grande D, Molmenti EP, Miller EJ, Rilo HLR (2017) Immunomodulation by mesenchymal stromal cells and their clinical applications. J Stem Cell Regen Biol 3(2). https://doi.org/10.15436/2471-0598.17.022
Capilla-González V, López-Beas J, Escacena N, Aguilera Y, de la Cuesta A, Ruiz-Salmerón R, Martín F et al (2018) PDGF restores the defective phenotype of adipose-derived mesenchymal stromal cells from diabetic patients. Mol Ther 26(11):2696–2709. https://doi.org/10.1016/j.ymthe.2018.08.011
Caplan AI (2009) Why are MSCs therapeutic? New data: new insight. J Pathol 217(2):318–324. https://doi.org/10.1002/path.2469
Caplan AI, Dennis JE (2006) Mesenchymal stem cells as trophic mediators. J Cell Biochem 98(5):1076–1084. https://doi.org/10.1002/jcb.20886
Capra E, Beretta R, Parazzi V, Viganò M, Lazzari L, Baldi A, Giordano R (2012) Changes in the proteomic profile of adipose tissue-derived mesenchymal stem cells during passages. Proteome Sci 10(1):46. https://doi.org/10.1186/1477-5956-10-46
Carvalho JL, Braga VB, Melo MB, Campos AC, Oliveira MS, Gomes DA, Ferreira AJ et al (2013) Priming mesenchymal stem cells boosts stem cell therapy to treat myocardial infarction. J Cell Mol Med 17(5):617–625. https://doi.org/10.1111/jcmm.12036
Chao YH, Peng CT, Harn HJ, Chan CK, Wu KH (2010) Poor potential of proliferation and differentiation in bone marrow mesenchymal stem cells derived from children with severe aplastic anemia. Ann Hematol 89(7):715–723. https://doi.org/10.1007/s00277-009-0892-6
Chase LG, Lakshmipathy U, Solchaga LA, Rao MS, Vemuri MC (2010) A novel serum-free medium for the expansion of human mesenchymal stem cells. Stem Cell Res Ther 1(1):8. https://doi.org/10.1186/scrt8
Chen YC, Gonzalez ME, Burman B, Zhao X, Anwar T, Tran M, Medhora N et al (2019) Mesenchymal stem/stromal cell engulfment reveals metastatic advantage in breast cancer. Cell Rep 27(13):3916–3926.e5. https://doi.org/10.1016/j.celrep.2019.05.084
Cheng S, Nethi SK, Rathi S, Layek B, Prabha S (2019) Engineered mesenchymal stem cells for targeting solid tumors: therapeutic potential beyond regenerative therapy. J Pharmacol Exp Ther 370(2):231–241. https://doi.org/10.1124/jpet.119.259796
Cipriani P, Guiducci S, Miniati I, Cinelli M, Urbani S, Marrelli A, Dolo V et al (2007) Impairment of endothelial cell differentiation from bone marrow-derived mesenchymal stem cells: new insight into the pathogenesis of systemic sclerosis. Arthritis Rheum 56(6):1994–2004. https://doi.org/10.1002/art.22698
Cobellis G, Silvestroni A, Lillo S, Sica G, Botti C, Maione C, Schiavone V et al (2008) Long-term effects of repeated autologous transplantation of bone marrow cells in patients affected by peripheral arterial disease. Bone Marrow Transplant 42(10):667–672. https://doi.org/10.1038/bmt.2008.228
Conget PA, Minguell JJ (1999) Phenotypical and functional properties of human bone marrow mesenchymal progenitor cells. J Cell Physiol 181(1):67–73. https://doi.org/10.1002/(SICI)1097-4652(199910)181:1<67::AID-JCP7>3.0.CO;2-C
Constantin G, Marconi S, Rossi B, Angiari S, Calderan L, Anghileri E, Gini B et al (2009) Adipose-derived mesenchymal stem cells ameliorate chronic experimental autoimmune encephalomyelitis. Stem Cells 27(10):2624–2635. https://doi.org/10.1002/stem.194
Corcione A, Benvenuto F, Ferretti E, Giunti D, Cappiello V, Cazzanti F, Risso M et al (2006) Human mesenchymal stem cells modulate B-cell functions. Blood 107(1):367–372. https://doi.org/10.1182/blood-2005-07-2657
Cramer C, Freisinger E, Jones RK, Slakey DP, Dupin CL, Newsome ER, Alt EU et al (2010) Persistent high glucose concentrations alter the regenerative potential of mesenchymal stem cells. Stem Cells Dev 19(12):1875–1884. https://doi.org/10.1089/scd.2010.0009
Crisan M, Yap S, Casteilla L, Chen CW, Corselli M, Park TS, Andriolo G et al (2008) A perivascular origin for mesenchymal stem cells in multiple human organs. Cell Stem Cell 3(3):301–313. https://doi.org/10.1016/j.stem.2008.07.003
De Ugarte DA, Alfonso Z, Zuk PA, Elbarbary A, Zhu M, Ashjian P, Benhaim P et al (2003) Differential expression of stem cell mobilization-associated molecules on multi-lineage cells from adipose tissue and bone marrow. Immunol Lett 89(2–3):267–270. https://doi.org/10.1016/s0165-2478(03)00108-1
Di Nicola M, Carlo-Stella C, Magni M, Milanesi M, Longoni PD, Matteucci P, Grisanti S (2002) Human bone marrow stromal cells suppress T-lymphocyte proliferation induced by cellular or nonspecific mitogenic stimuli. Blood 99(10):3838–3843. https://doi.org/10.1182/blood.v99.10.3838
Dias S, Shmelkov SV, Lam G, Rafii S (2002) VEGF(165) promotes survival of leukemic cells by Hsp90-mediated induction of Bcl-2 expression and apoptosis inhibition. Blood 99(7):2532–2540. https://doi.org/10.1182/blood.v99.7.2532
Dimitroff CJ, Lee JY, Rafii S, Fuhlbrigge RC, Sackstein R (2001) CD44 is a major E-selectin ligand on human hematopoietic progenitor cells. J Cell Biol 153(6):1277–1286. https://doi.org/10.1083/jcb.153.6.1277
Djouad F, Plence P, Bony C, Tropel P, Apparailly F, Sany J, Noël D et al (2003) Immunosuppressive effect of mesenchymal stem cells favors tumor growth in allogeneic animals. Blood 102(10):3837–3844. https://doi.org/10.1182/blood-2003-04-1193
Dong M, How T, Kirkbride KC, Gordon KJ, Lee JD, Hempel N, Kelly P et al (2007) The type III TGF-beta receptor suppresses breast cancer progression. J Clin Invest 117(1):206–217. https://doi.org/10.1172/JCI29293
Duffy GP, Ahsan T, O’Brien T, Barry F, Nerem RM (2009) Bone marrow-derived mesenchymal stem cells promote angiogenic processes in a time- and dose-dependent manner in vitro. Tissue Eng Part A 15(9):2459–2470. https://doi.org/10.1089/ten.TEA.2008.0341
English K, Barry FP, Mahon BP (2008) Murine mesenchymal stem cells suppress dendritic cell migration, maturation and antigen presentation. Immunol Lett 115(1):50–58. https://doi.org/10.1016/j.imlet.2007.10.002
Escacena N, Quesada-Hernández E, Capilla-González V, Soria B, Hmadcha A (2015) Bottlenecks in the efficient use of advanced therapy medicinal products based on mesenchymal stromal cells. Stem Cells Int 2015:895714. https://doi.org/10.1155/2015/895714
Fang B, Song YP, Liao LM, Han Q, Zhao RC (2006) Treatment of severe therapy-resistant acute graft-versus-host disease with human adipose tissue-derived mesenchymal stem cells. Bone Marrow Transplant 38(5):389–390. https://doi.org/10.1038/sj.bmt.1705457
Fathi E, Sanaat Z, Farahzadi R (2019) Mesenchymal stem cells in acute myeloid leukemia: a focus on mechanisms involved and therapeutic concepts. Blood Res 54(3):165–174. https://doi.org/10.5045/br.2019.54.3.165
Faulkner J, Jiang P, Farris D, Walker R, Dai Z (2020) CRISPR/CAS9-mediated knockout of Abi1 inhibits p185Bcr-Abl-induced leukemogenesis and signal transduction to ERK and PI3K/Akt pathways. J Hematol Oncol 13(1):34. https://doi.org/10.1186/s13045-020-00867-5
Fazekasova H, Lechler R, Langford K, Lombardi G (2011) Placenta-derived MSCs are partially immunogenic and less immunomodulatory than bone marrow-derived MSCs. J Tissue Eng Regen Med 5(9):684–694. https://doi.org/10.1002/term.362
Galiè M, Konstantinidou G, Peroni D, Scambi I, Marchini C, Lisi V, Krampera M et al (2008) Mesenchymal stem cells share molecular signature with mesenchymal tumor cells and favor early tumor growth in syngeneic mice. Oncogene 27(18):2542–2551. https://doi.org/10.1038/sj.onc.1210920
Gálvez P, Clares B, Bermejo M, Hmadcha A, Soria B (2014) Standard requirement of a microbiological quality control program for the manufacture of human mesenchymal stem cells for clinical use. Stem Cells Dev 23(10):1074–1083. https://doi.org/10.1089/scd.2013.0625
Gálvez P, Clares B, Hmadcha A, Ruiz A, Soria B (2013) Development of a cell-based medicinal product: regulatory structures in the European Union. Br Med Bull 105:85–105. https://doi.org/10.1093/bmb/lds036
Ganta C, Chiyo D, Ayuzawa R, Rachakatla R, Pyle M, Andrews G, Weiss M, Tamura M, Troyer D et al (2009) Rat umbilical cord stem cells completely abolish rat mammary carcinomas with no evidence of metastasis or recurrence 100 days post-tumor cell inoculation. Cancer Res 69(5):1815–1820. https://doi.org/10.1158/0008-5472.CAN-08-2750
Gao P, Ding Q, Wu Z, Jiang H, Fang Z (2010) Therapeutic potential of human mesenchymal stem cells producing IL-12 in a mouse xenograft model of renal cell carcinoma. Cancer Lett 290(2):157–166. https://doi.org/10.1016/j.canlet.2009.08.031
Gao H, Priebe W, Glod J, Banerjee D (2009) Activation of signal transducers and activators of transcription 3 and focal adhesion kinase by stromal cell-derived factor 1 is required for migration of human mesenchymal stem cells in response to tumor cell-conditioned medium. Stem Cells 27(4):857–865. https://doi.org/10.1002/stem.23
García-Bernal D, García-Arranz M, Yáñez RM, Hervás-Salcedo R, Cortés A, Fernández-García M, Hernando-Rodríguez M et al (2021) The current status of mesenchymal stromal cells: controversies, unresolved issues and some promising solutions to improve their therapeutic efficacy. Front Cell Dev Biol 9:650664. https://doi.org/10.3389/fcell.2021.650664
García-Bernal D, García-Arranz M, García-Guillén AI, García-Hernández AM, Blanquer M, García-Olmo D, Sackstein R et al (2020) Exofucosylation of adipose mesenchymal stromal cells alters their secretome profile. Front Cell Dev Biol 8:584074. https://doi.org/10.3389/fcell.2020.584074
George MJ, Prabhakara K, Toledano-Furman NE, Wang YW, Gill BS, Wade CE, Olson SD et al (2018) Clinical cellular therapeutics accelerate clot formation. Stem Cells Transl Med 7(10):731–739. https://doi.org/10.1002/sctm.18-0015
Gerace D, Martiniello-Wilks R, Nassif NT, Lal S, Steptoe R, Simpson AM (2017) CRISPR-targeted genome editing of mesenchymal stem cell-derived therapies for type 1 diabetes: a path to clinical success? Stem Cell Res Ther 8(1):62. https://doi.org/10.1186/s13287-017-0511-8
Glennie S, Soeiro I, Dyson PJ, Lam EW, Dazzi F (2005) Bone marrow mesenchymal stem cells induce division arrest anergy of activated T cells. Blood 105(7):2821–2827. https://doi.org/10.1182/blood-2004-09-3696
Golchin A, Shams F, Karami F (2020) Advancing mesenchymal stem cell therapy with CRISPR/Cas9 for clinical trial studies. Adv Exp Med Biol 1247:89–100. https://doi.org/10.1007/5584_2019_459
González MA, Gonzalez-Rey E, Rico L, Büscher D, Delgado M (2009) Treatment of experimental arthritis by inducing immune tolerance with human adipose-derived mesenchymal stem cells. Arthritis Rheum 60(4):1006–1019. https://doi.org/10.1002/art.24405
Gottipamula S, Muttigi MS, Kolkundkar U, Seetharam RN (2013) Serum-free media for the production of human mesenchymal stromal cells: a review. Cell Prolif 46(6):608–627. https://doi.org/10.1111/cpr.12063
Guasch G, Schober M, Pasolli HA, Conn EB, Polak L, Fuchs E (2007) Loss of TGFbeta signaling destabilizes homeostasis and promotes squamous cell carcinomas in stratified epithelia. Cancer Cell 12(4):313–327. https://doi.org/10.1016/j.ccr.2007.08.020
Guo X, Zhao Y, Yan H, Yang Y, Shen S, Dai X, Ji X et al (2017) Single tumor-initiating cells evade immune clearance by recruiting type II macrophages. Genes Dev 31(3):247–259. https://doi.org/10.1101/gad.294348.116
Gupta NK, Armstrong EJ, Parikh SA (2014) The current state of stem cell therapy for peripheral artery disease. Curr Cardiol Rep 16(2):447. https://doi.org/10.1007/s11886-013-0447-2
Haider KH, Ashraf M (2012) Preconditioning approach in stem cell therapy for the treatment of infarcted heart. Prog Mol Biol Transl Sci 111:323–356. https://doi.org/10.1016/B978-0-12-398459-3.00015-0
Haider KH, Aramini B (2020) Mircrining the injured heart with stem cell-derived exosomes: an emerging strategy of cell-free therapy. Stem Cell Res Ther 11(1):23. https://doi.org/10.1186/s13287-019-1548-7
Haider KH, Aslam M (2018) Cell-free therapy with stem cell secretions: protection, repair and regeneration of the injured myocardium. In: Haider KH, Aziz S (eds) Stem cells: from hype to real hope. Medicine & Life Sciences, DE GRUYTER, Berlin. (Published, 2018). https://doi.org/10.1515/9783110642438
Haider KH, Jiang S, Niagara MI, Ashraf M (2008) IGF-I over expressing mesenchymal stem cells accelerate bone marrow stem cell mobilization via paracrine activation of SDF-1α/CXCR4 signaling to promote myocardial repair. Circulation Res 103:1300–1308. https://doi.org/10.1161/CIRCRESAHA.108.186742
Han J, Zhao J, Xu J, Wen Y (2014) Mesenchymal stem cells genetically modified by lentivirus-mediated interleukin-12 inhibit malignant ascites in mice. Exp Ther Med 8(4):1330–1334. https://doi.org/10.3892/etm.2014.1918
Hao C, Beguinot F, Condorelli G, Trencia A, Van Meir EG, Yong VW, Parney IF et al (2001) Induction and intracellular regulation of tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) mediated apoptosis in human malignant glioma cells. Cancer Res 61(3):1162–1170
Hashi CK, Zhu Y, Yang GY, Young WL, Hsiao BS, Wang K, Chu B et al (2007) Antithrombogenic property of bone marrow mesenchymal stem cells in nanofibrous vascular grafts. Proc Natl Acad Sci U S A 104(29):11915–11920. https://doi.org/10.1073/pnas.0704581104
Herberts CA, Kwa MS, Hermsen HP (2011) Risk factors in the development of stem cell therapy. J Transl Med 9:29. https://doi.org/10.1186/1479-5876-9-29
Hmadcha A, Dominguez-Bendala J, Wakeman J, Arredouani M, Soria B (2009) The immune boundaries for stem cell based therapies: problems and prospective solutions. J Cell Mol Med 13(8A):1464–1475. https://doi.org/10.1111/j.1582-4934.2009.00837.x
Hmadcha A, Martin-Montalvo A, Gauthier BR, Soria B, Capilla-González V (2020) Therapeutic potential of mesenchymal stem cells for cancer therapy. Front Bioeng Biotechnol 8:43. https://doi.org/10.3389/fbioe.2020.00043
Ho IA, Toh HC, Ng WH, Teo YL, Guo CM, Hui KM, Lam PY (2013) Human bone marrow-derived mesenchymal stem cells suppress human glioma growth through inhibition of angiogenesis. Stem Cells 31(1):146–155. https://doi.org/10.1002/stem.1247
Honczarenko M, Le Y, Swierkowski M, Ghiran I, Glodek AM, Silberstein LE (2006) Human bone marrow stromal cells express a distinct set of biologically functional chemokine receptors. Stem Cells 24(4):1030–1041. https://doi.org/10.1634/stemcells.2005-0319
Horwitz EM, Gordon PL, Koo WK, Marx JC, Neel MD, McNall RY, Muul L et al (2002) Isolated allogeneic bone marrow-derived mesenchymal cells engraft and stimulate growth in children with osteogenesis imperfecta: implications for cell therapy of bone. Proc Natl Acad Sci U S A 99(13):8932–8937. https://doi.org/10.1073/pnas.132252399
Horwitz EM, Le Blanc K, Dominici M, Mueller I, Slaper-Cortenbach I, Marini FC, Deans RJ et al (2005) Clarification of the nomenclature for MSC: the International Society for Cellular Therapy position statement. Cytotherapy 7(5):393–395. https://doi.org/10.1080/14653240500319234
Hunt JS, Petroff MG, McIntire RH, Ober C (2005) HLA-G and immune tolerance in pregnancy. FASEB J 19(7):681–693. https://doi.org/10.1096/fj.04-2078rev
Iglesias-López C, Agustí A, Obach M, Vallano A (2019) Regulatory framework for advanced therapy medicinal products in Europe and United States. Front Pharmacol 10:921. https://doi.org/10.3389/fphar.2019.00921
Ishikawa I, Sawada R, Kato Y, Tsuji K, Shao J, Yamada T, Kato R et al (2009) Effectivity of the novel serum-free medium STK2 for proliferating human mesenchymal stem cells. Yakugaku Zasshi 129(3):381–384. Japanese. https://doi.org/10.1248/yakushi.129.381
Jayme DW, Smith SR (2000) Media formulation options and manufacturing process controls to safeguard against introduction of animal origin contaminants in animal cell culture. Cytotechnology 33(1–3):27–36. https://doi.org/10.1023/A:1008133717035
Jiang R, Han Z, Zhuo G, Qu X, Li X, Wang X, Shao Y et al (2011) Transplantation of placenta-derived mesenchymal stem cells in type 2 diabetes: a pilot study. Front Med 5(1):94–100. https://doi.org/10.1007/s11684-011-0116-z
Jiang S, Haider KH, Niagara MI, Salim A, Ashraf M (2006) Supportive interaction between cell survival signaling and angio-competent factors enhances donor cell survival and promotes angiomyogenesis for cardiac repair. Circulation Res 99:776–784. https://doi.org/10.1161/01.RES.0000244687.97719.4f
Jiang S, Haider KH, Niagara MI, Lu G, Ashraf M (2008) Stable therapeutic effects of mesenchymal stem cell based multiple gene delivery for cardiac repair. Cardiovasc Res 77(3):525–533. https://doi.org/10.1093/cvr/cvm077
Jotzu C, Alt E, Welte G, Li J, Hennessy BT, Devarajan E, Krishnappa S et al (2011) Adipose tissue derived stem cells differentiate into carcinoma-associated fibroblast-like cells under the influence of tumor derived factors. Cell Oncol (Dordr) 34(1):55–67. https://doi.org/10.1007/s13402-011-0012-1
Julier Z, Park AJ, Briquez PS, Martino MM (2017) Promoting tissue regeneration by modulating the immune system. Acta Biomater 53:13–28. https://doi.org/10.1016/j.actbio.2017.01.056
Jurewicz M, Yang S, Augello A, Godwin JG, Moore RF, Azzi J, Fiorina P et al (2010) Congenic mesenchymal stem cell therapy reverses hyperglycemia in experimental type 1 diabetes. Diabetes 59(12):3139–3147. https://doi.org/10.2337/db10-0542
Kalimuthu S, Oh JM, Gangadaran P, Zhu L, Lee HW, Rajendran RL, Baek SH et al (2017) In vivo tracking of chemokine receptor CXCR4-engineered mesenchymal stem cell migration by optical molecular imaging. Stem Cells Int 2017:8085637. https://doi.org/10.1155/2017/8085637
Kalluri R (2016) The biology and function of fibroblasts in cancer. Nat Rev Cancer 16(9):582–598. https://doi.org/10.1038/nrc.2016.73
Karnoub AE, Dash AB, Vo AP, Sullivan A, Brooks MW, Bell GW, Richardson AL et al (2007) Mesenchymal stem cells within tumour stroma promote breast cancer metastasis. Nature 449(7162):557–563. https://doi.org/10.1038/nature06188
Kastrinaki MC, Sidiropoulos P, Roche S, Ringe J, Lehmann S, Kritikos H, Vlahava VM et al (2008) Functional, molecular and proteomic characterisation of bone marrow mesenchymal stem cells in rheumatoid arthritis. Ann Rheum Dis 67(6):741–749. https://doi.org/10.1136/ard.2007.076174
Kern S, Eichler H, Stoeve J, Klüter H, Bieback K (2006) Comparative analysis of mesenchymal stem cells from bone marrow, umbilical cord blood, or adipose tissue. Stem Cells 24(5):1294–1301. https://doi.org/10.1634/stemcells.2005-0342
Khakoo AY, Pati S, Anderson SA, Reid W, Elshal MF, Rovira II, Nguyen AT et al (2006) Human mesenchymal stem cells exert potent antitumorigenic effects in a model of Kaposi’s sarcoma. J Exp Med 203(5):1235–1247. https://doi.org/10.1084/jem.20051921
Kim CK, Haider KH, Lim SJ (2001) Gene medicine: a new field of molecular medicine. Arch Pharmacol Res 24(1):1–15. https://doi.org/10.1007/BF02976486
Kim DS, Jang IK, Lee MW, Ko YJ, Lee DH, Lee JW, Sung KW et al (2018) Enhanced immunosuppressive properties of human mesenchymal stem cells primed by interferon-γ. EBioMedicine 28:261–273. https://doi.org/10.1016/j.ebiom.2018.01.002
Kim HW, Malik F, Durrani S, Ashraf M, Jiang S, Haider KH (2012a) Concomitant Activation of miR-107/PDCD10 and Hypoxamir-210/Casp8ap2 and Their Role in Cytoprotection During Ischemic Preconditioning of Stem Cells. Antioxidant Redox Signal 17(8):1053–1065. https://doi.org/10.1089/ars.2012.4518
Kim HW, Ashraf M, Jiang S, Haider KH (2012b) Stem cell based delivery of Hypoxamir-210 to the infarcted heart: implications on stem cell survival and preservation of the infarcted heart function. J Mol Med 90(9):997–1010. https://doi.org/10.1007/s00109-012-0920-1
Kim JS, Kim JG, Moon MY, Jeon CY, Won HY, Kim HJ, Jeon YJ et al (2006) Transforming growth factor-beta1 regulates macrophage migration via RhoA. Blood 108(6):1821–1829. https://doi.org/10.1182/blood-2005-10-009191
Kinzebach S, Bieback K (2013) Expansion of mesenchymal stem/stromal cells under xenogenic-free culture conditions. Adv Biochem Eng Biotechnol 129:33–57. https://doi.org/10.1007/10_2012_134
Koç ON, Gerson SL, Cooper BW, Dyhouse SM, Haynesworth SE, Caplan AI, Lazarus HM (2000) Rapid hematopoietic recovery after coinfusion of autologous-blood stem cells and culture-expanded marrow mesenchymal stem cells in advanced breast cancer patients receiving high-dose chemotherapy. J Clin Oncol 18(2):307–316. https://doi.org/10.1200/JCO.2000.18.2.307
Kolaczkowska E, Kubes P (2013) Neutrophil recruitment and function in health and inflammation. Nat Rev Immunol 13(3):159–175. https://doi.org/10.1038/nri3399
König A, Menzel T, Lynen S, Wrazel L, Rosén A, Al-Katib A, Raveche E et al (1997) Basic fibroblast growth factor (bFGF) upregulates the expression of bcl-2 in B cell chronic lymphocytic leukemia cell lines resulting in delaying apoptosis. Leukemia 11(2):258–265. https://doi.org/10.1038/sj.leu.2400556
Krampera M, Cosmi L, Angeli R, Pasini A, Liotta F, Andreini A, Santarlasci V et al (2006) Role for interferon-gamma in the immunomodulatory activity of human bone marrow mesenchymal stem cells. Stem Cells 24(2):386–398. https://doi.org/10.1634/stemcells.2005-0008
Krampera M, Galipeau J, Shi Y, Tarte K, Sensebe L, MSC Committee of the International Society for Cellular Therapy (ISCT) (2013) Immunological characterization of multipotent mesenchymal stromal cells--The International Society for Cellular Therapy (ISCT) working proposal. Cytotherapy 15(9):1054–1061. https://doi.org/10.1016/j.jcyt.2013.02.010
Krampera M (2011) Mesenchymal stromal cell ‘licensing’: a multistep process. Leukemia 25(9):1408–1414. https://doi.org/10.1038/leu.2011.108
Krampera M, Sartoris S, Liotta F, Pasini A, Angeli R, Cosmi L, Andreini A et al (2007) Immune regulation by mesenchymal stem cells derived from adult spleen and thymus. Stem Cells Dev 16(5):797–810. https://doi.org/10.1089/scd.2007.0024
Kume S, Kato S, Yamagishi S, Inagaki Y, Ueda S, Arima N, Okawa T et al (2005) Advanced glycation end-products attenuate human mesenchymal stem cells and prevent cognate differentiation into adipose tissue, cartilage, and bone. J Bone Miner Res 20(9):1647–1658. https://doi.org/10.1359/JBMR.050514
Kunter U, Rong S, Boor P, Eitner F, Müller-Newen G, Djuric Z, van Roeyen CR et al (2007) Mesenchymal stem cells prevent progressive experimental renal failure but maldifferentiate into glomerular adipocytes. J Am Soc Nephrol 18(6):1754–1764. https://doi.org/10.1681/ASN.2007010044
Lai VK, Afzal MR, Ashraf M, Jiang S, Haider KH (2012a) Non-hypoxic stabilization of Hif-1α during coordinated interaction between Akt and angiopoietin-1 enhances endothelial commitment of bone marrow stem cells. J Mol Med 90(6):719–730. https://doi.org/10.1007/s00109-011-0852-1
Lai VK, Ashraf M, Jiang S, Haider KH (2012b) MicroRNA-143 is critical regulator of cell cycle activity in stem cells with co-overexpression of Akt and angiopoietin-1 via transcriptional regulation of Erk5/Cyclin D1 signaling. Cell Cycle 11(4):767–677. https://doi.org/10.4161/cc.11.4.19211
Lazarus HM, Koc ON, Devine SM, Curtin P, Maziarz RT, Holland HK, Shpall EJ et al (2005) Cotransplantation of HLA-identical sibling culture-expanded mesenchymal stem cells and hematopoietic stem cells in hematologic malignancy patients. Biol Blood Marrow Transplant 11(5):389–398. https://doi.org/10.1016/j.bbmt.2005.02.001
Le Blanc K (2003) Immunomodulatory effects of fetal and adult mesenchymal stem cells. Cytotherapy 5(6):485–489. https://doi.org/10.1080/14653240310003611
Le Blanc K, Rasmusson I, Sundberg B, Götherström C, Hassan M, Uzunel M, Ringdén O (2004) Treatment of severe acute graft-versus-host disease with third party haploidentical mesenchymal stem cells. Lancet 363(9419):1439–1441. https://doi.org/10.1016/S0140-6736(04)16104-7
Lee JM, Jung J, Lee HJ, Jeong SJ, Cho KJ, Hwang SG, Kim GJ (2012a) Comparison of immunomodulatory effects of placenta mesenchymal stem cells with bone marrow and adipose mesenchymal stem cells. Int Immunopharmacol 13(2):219–224. https://doi.org/10.1016/j.intimp.2012.03.024
Lee RH, Seo MJ, Reger RL, Spees JL, Pulin AA, Olson SD, Prockop DJ (2006) Multipotent stromal cells from human marrow home to and promote repair of pancreatic islets and renal glomeruli in diabetic NOD/scid mice. Proc Natl Acad Sci U S A 103(46):17438–17443. https://doi.org/10.1073/pnas.0608249103
Lee T (2010) Host tissue response in stem cell therapy. World J Stem Cells 2(4):61–66. https://doi.org/10.4252/wjsc.v2.i4.61
Lee S, Park JR, Seo MS, Roh KH, Park SB, Hwang JW, Sun B et al (2009) Histone deacetylase inhibitors decrease proliferation potential and multilineage differentiation capability of human mesenchymal stem cells. Cell Prolif 42(6):711–720. https://doi.org/10.1111/j.1365-2184.2009.00633.x
Lee RH, Yoon N, Reneau JC, Prockop DJ (2012b) Preactivation of human MSCs with TNF-α enhances tumor-suppressive activity. Cell Stem Cell 11(6):825–835. https://doi.org/10.1016/j.stem.2012.10.001
Li GC, Zhang HW, Zhao QC, Sun LI, Yang JJ, Hong L, Feng F et al (2016) Mesenchymal stem cells promote tumor angiogenesis via the action of transforming growth factor β1. Oncol Lett 11(2):1089–1094. https://doi.org/10.3892/ol.2015.3997
Li B, Fu J, Chen P, Zhuang W (2010) Impairment in immunomodulatory function of mesenchymal stem cells from multiple myeloma patients. Arch Med Res 41(8):623–633. https://doi.org/10.1016/j.arcmed.2010.11.008
Li W, Zhou Y, Yang J, Zhang X, Zhang H, Zhang T, Zhao S et al (2015) Gastric cancer-derived mesenchymal stem cells prompt gastric cancer progression through secretion of interleukin-8. J Exp Clin Cancer Res 34(1):52. https://doi.org/10.1186/s13046-015-0172-3
Li Y, Zhang D, Xu L, Dong L, Zheng J, Lin Y, Huang J et al (2019) Cell-cell contact with proinflammatory macrophages enhances the immunotherapeutic effect of mesenchymal stem cells in two abortion models. Cell Mol Immunol 16(12):908–920. https://doi.org/10.1038/s41423-019-0204-6
Liang J, Zhang H, Hua B, Wang H, Lu L, Shi S, Hou Y et al (2010) Allogenic mesenchymal stem cells transplantation in refractory systemic lupus erythematosus: a pilot clinical study. Ann Rheum Dis 69(8):1423–1429. https://doi.org/10.1136/ard.2009.123463
Liao L, Shi B, Chang H, Su X, Zhang L, Bi C, Shuai Y et al (2017) Heparin improves BMSC cell therapy: anticoagulant treatment by heparin improves the safety and therapeutic effect of bone marrow-derived mesenchymal stem cell cytotherapy. Theranostics 7(1):106–116. https://doi.org/10.7150/thno.16911
Lo B, Parham L (2009) Ethical issues in stem cell research. Endocr Rev 30(3):204–213. https://doi.org/10.1210/er.2008-0031
López-Lucas MD, Pachón-Peña G, García-Hernández AM, Parrado A, Sánchez-Salinas D, García-Bernal D, Algueró MDC et al (2018) Production via good manufacturing practice of exofucosylated human mesenchymal stromal cells for clinical applications. Cytotherapy 20(9):1110–1123. https://doi.org/10.1016/j.jcyt.2018.07.001
Lourenco S, Teixeira VH, Kalber T, Jose RJ, Floto RA, Janes SM (2015) Macrophage migration inhibitory factor-CXCR4 is the dominant chemotactic axis in human mesenchymal stem cell recruitment to tumors. J Immunol 194(7):3463–3474. https://doi.org/10.4049/jimmunol.1402097
Lu G, Ashraf M, Haider KH (2012) Insulin-Like Growth Factor-1 Preconditioning Accentuates Intrinsic Survival Mechanism in Stem Cells to Resist Ischemic Injury by Orchestrating Protein Kinase Cα–Erk1/2 Activation. Antioxidant Red Signaling 16(3):217–227. https://doi.org/10.1089/ars.2011.4112
Lu G, Haider KH, Porollo A, Ashraf M (2010) Mitochondria specific transgenic overexpression of connexin-43 simulates preconditioning induced cytoprotection of stem cells. Cardiovasc Res 88:277–286. https://doi.org/10.1093/cvr/cvq293
Lu L, Chen G, Yang J, Ma Z, Yang Y, Hu Y, Lu Y et al (2019) Bone marrow mesenchymal stem cells suppress growth and promote the apoptosis of glioma U251 cells through downregulation of the PI3K/AKT signaling pathway. Biomed Pharmacother 112:108625. https://doi.org/10.1016/j.biopha.2019.108625
Ma J, Liu N, Yi B, Zhang X, Gao BB, Zhang Y, Xu R et al (2015) Transplanted hUCB-MSCs migrated to the damaged area by SDF-1/CXCR4 signaling to promote functional recovery after traumatic brain injury in rats. Neurol Res 37(1):50–56. https://doi.org/10.1179/1743132814Y.0000000399
Maccario R, Podestà M, Moretta A, Cometa A, Comoli P, Montagna D, Daudt L et al (2005) Interaction of human mesenchymal stem cells with cells involved in alloantigen-specific immune response favors the differentiation of CD4+ T-cell subsets expressing a regulatory/suppressive phenotype. Haematologica 90(4):516–525
Maltman DJ, Hardy SA, Przyborski SA (2011) Role of mesenchymal stem cells in neurogenesis and nervous system repair. Neurochem Int 59(3):347–356. https://doi.org/10.1016/j.neuint.2011.06.008
Mangi AA, Noiseux N, Kong D, He H, Rezvani M, Ingwall JS, Dzau VJ (2003) Mesenchymal stem cells modified with Akt prevent remodeling and restore performance of infarcted hearts. Nat Med 9(9):1195–1201. https://doi.org/10.1038/nm912
Mao F, Wu Y, Tang X, Kang J, Zhang B, Yan Y, Qian H et al (2017) Exosomes derived from human umbilical cord mesenchymal stem cells relieve inflammatory bowel disease in mice. Biomed Res Int 2017:5356760. https://doi.org/10.1155/2017/5356760
Mastri M, Shah Z, McLaughlin T, Greene CJ, Baum L, Suzuki G, Lee T (2012) Activation of toll-like receptor 3 amplifies mesenchymal stem cell trophic factors and enhances therapeutic potency. Am J Physiol Cell Physiol 303(10):C1021–C1033. https://doi.org/10.1152/ajpcell.00191.2012
Mazzanti B, Aldinucci A, Biagioli T, Barilaro A, Urbani S, Dal Pozzo S, Amato MP et al (2008) Differences in mesenchymal stem cell cytokine profiles between MS patients and healthy donors: implication for assessment of disease activity and treatment. J Neuroimmunol 199(1–2):142–150. https://doi.org/10.1016/j.jneuroim.2008.05.006
McAuley DF, Curley GF, Hamid UI, Laffey JG, Abbott J, McKenna DH, Fang X et al (2014) Clinical grade allogeneic human mesenchymal stem cells restore alveolar fluid clearance in human lungs rejected for transplantation. Am J Physiol Lung Cell Mol Physiol 306(9):L809–L815. https://doi.org/10.1152/ajplung.00358.2013
Mead B, Chamling X, Zack DJ, Ahmed Z, Tomarev S (2020) TNFα-mediated priming of mesenchymal stem cells enhances their neuroprotective effect on retinal ganglion cells. Invest Ophthalmol Vis Sci 61(2):6. https://doi.org/10.1167/iovs.61.2.6
Mei SH, McCarter SD, Deng Y, Parker CH, Liles WC, Stewart DJ (2007) Prevention of LPS-induced acute lung injury in mice by mesenchymal stem cells overexpressing angiopoietin 1. PLoS Med 4(9):e269. https://doi.org/10.1371/journal.pmed.0040269
Meirelles Lda S, Fontes AM, Covas DT, Caplan AI (2009) Mechanisms involved in the therapeutic properties of mesenchymal stem cells. Cytokine Growth Factor Rev 20(5–6):419–427. https://doi.org/10.1016/j.cytogfr.2009.10.002
Melief SM, Zwaginga JJ, Fibbe WE, Roelofs H (2013) Adipose tissue-derived multipotent stromal cells have a higher immunomodulatory capacity than their bone marrow-derived counterparts. Stem Cells Transl Med 2(6):455–463. https://doi.org/10.5966/sctm.2012-0184
Mingozzi F, High KA (2013) Immune responses to AAV vectors: overcoming barriers to successful gene therapy. Blood 122(1):23–36. https://doi.org/10.1182/blood-2013-01-306647
Minteer DM, Young MT, Lin YC, Over PJ, Rubin JP, Gerlach JC, Marra KG (2015) Analysis of type II diabetes mellitus adipose-derived stem cells for tissue engineering applications. J Tissue Eng 6:2041731415579215. https://doi.org/10.1177/2041731415579215
Mishra PJ, Mishra PJ, Humeniuk R, Medina DJ, Alexe G, Mesirov JP, Ganesan S et al (2008) Carcinoma-associated fibroblast-like differentiation of human mesenchymal stem cells. Cancer Res 68(11):4331–4339. https://doi.org/10.1158/0008-5472.CAN-08-0943
Miwa H, Era T (2018) Tracing the destiny of mesenchymal stem cells from embryo to adult bone marrow and white adipose tissue via Pdgfrα expression. Development 145(2):dev155879. https://doi.org/10.1242/dev.155879
Mohty M, Duarte RF, Croockewit S, Hübel K, Kvalheim G, Russell N (2011) The role of plerixafor in optimizing peripheral blood stem cell mobilization for autologous stem cell transplantation. Leukemia 25(1):1–6. https://doi.org/10.1038/leu.2010.224
Molavi B, Zafarghandi MR, Aminizadeh E, Hosseini SE, Mirzayi H, Arab L, Baharvand H et al (2016) Safety and efficacy of repeated bone marrow mononuclear cell therapy in patients with critical limb ischemia in a pilot randomized controlled trial. Arch Iran Med 19(6):388–396
Moll G, Ankrum JA, Kamhieh-Milz J, Bieback K, Ringdén O, Volk HD, Geissler S et al (2019) Intravascular mesenchymal stromal/stem cell therapy product diversification: time for new clinical guidelines. Trends Mol Med 25(2):149–163. https://doi.org/10.1016/j.molmed.2018.12.006
Moll G, Hult A, von Bahr L, Alm JJ, Heldring N, Hamad OA, Stenbeck-Funke L et al (2014) Do ABO blood group antigens hamper the therapeutic efficacy of mesenchymal stromal cells? PLoS One 9(1):e85040. https://doi.org/10.1371/journal.pone.0085040
Moll G, Jitschin R, von Bahr L, Rasmusson-Duprez I, Sundberg B, Lönnies L, Elgue G et al (2011) Mesenchymal stromal cells engage complement and complement receptor bearing innate effector cells to modulate immune responses. PLoS One 6(7):e21703. https://doi.org/10.1371/journal.pone.0021703
Moll G, Drzeniek N, Kamhieh-Milz J, Geissler S, Volk HD, Reinke P (2020) MSC therapies for COVID-19: importance of patient coagulopathy, thromboprophylaxis, cell product quality and mode of delivery for treatment safety and efficacy. Front Immunol 11:1091. https://doi.org/10.3389/fimmu.2020.01091
Moll G, Rasmusson-Duprez I, von Bahr L, Connolly-Andersen AM, Elgue G, Funke L, Hamad OA et al (2012) Are therapeutic human mesenchymal stromal cells compatible with human blood? Stem Cells 30(7):1565–1574. https://doi.org/10.1002/stem.1111
Najar M, Raicevic G, Boufker HI, Fayyad Kazan H, De Bruyn C, Meuleman N, Bron D et al (2010) Mesenchymal stromal cells use PGE2 to modulate activation and proliferation of lymphocyte subsets: combined comparison of adipose tissue, Wharton’s Jelly and bone marrow sources. Cell Immunol 264(2):171–179. https://doi.org/10.1016/j.cellimm.2010.06.006
Nakamizo A, Marini F, Amano T, Khan A, Studeny M, Gumin J, Chen J et al (2005) Human bone marrow-derived mesenchymal stem cells in the treatment of gliomas. Cancer Res 65(8):3307–3318. https://doi.org/10.1158/0008-5472.CAN-04-1874
Neuss S, Schneider RK, Tietze L, Knüchel R, Jahnen-Dechent W (2010) Secretion of fibrinolytic enzymes facilitates human mesenchymal stem cell invasion into fibrin clots. Cells Tissues Organs 191(1):36–46. https://doi.org/10.1159/000215579
Nie Y, Lau C, Lie A, Chan G, Mok M (2010) Defective phenotype of mesenchymal stem cells in patients with systemic lupus erythematosus. Lupus 19(7):850–859. https://doi.org/10.1177/0961203309361482
Nilsson B, Teramura Y, Ekdahl KN (2014) The role and regulation of complement activation as part of the thromboinflammation elicited in cell therapies. Mol Immunol 61(2):185–190. https://doi.org/10.1016/j.molimm.2014.06.009
Ning H, Yang F, Jiang M, Hu L, Feng K, Zhang J, Yu Z et al (2008) The correlation between cotransplantation of mesenchymal stem cells and higher recurrence rate in hematologic malignancy patients: outcome of a pilot clinical study. Leukemia 22(3):593–599. https://doi.org/10.1038/sj.leu.2405090
Noronha NC, Mizukami A, Caliári-Oliveira C, Cominal JG, Rocha JLM, Covas DT, Swiech K et al (2019) Priming approaches to improve the efficacy of mesenchymal stromal cell-based therapies. Stem Cell Res Ther 10(1):131. https://doi.org/10.1186/s13287-019-1224-y
Otsu K, Das S, Houser SD, Quadri SK, Bhattacharya S, Bhattacharya J (2009) Concentration-dependent inhibition of angiogenesis by mesenchymal stem cells. Blood 113(18):4197–4205. https://doi.org/10.1182/blood-2008-09-176198
Pachón-Peña G, Donnelly C, Ruiz-Cañada C, Katz A, Fernández-Veledo S, Vendrell J, Sackstein R (2017) A glycovariant of human CD44 is characteristically expressed on human mesenchymal stem cells. Stem Cells 35(4):1080–1092. https://doi.org/10.1002/stem.2549
Papadaki HA, Kritikos HD, Gemetzi C, Koutala H, Marsh JC, Boumpas DT, Eliopoulos GD (2002) Bone marrow progenitor cell reserve and function and stromal cell function are defective in rheumatoid arthritis: evidence for a tumor necrosis factor alpha-mediated effect. Blood 99(5):1610–1619. https://doi.org/10.1182/blood.v99.5.1610
Pérez-Simón JA, Tabera S, Sarasquete ME, Díez-Campelo M, Canchado J, Sánchez-Abarca LI, Blanco B et al (2009) Mesenchymal stem cells are functionally abnormal in patients with immune thrombocytopenic purpura. Cytotherapy 11(6):698–705. https://doi.org/10.3109/14653240903051558
Pérez LM, de Lucas B, Gálvez BG (2018) Unhealthy stem cells: when health conditions upset stem cell properties. Cell Physiol Biochem 46(5):1999–2016. https://doi.org/10.1159/000489440
Petrie RJ, Doyle AD, Yamada KM (2009) Random versus directionally persistent cell migration. Nat Rev Mol Cell Biol 10(8):538–549. https://doi.org/10.1038/nrm2729
Phinney DG (2012) Functional heterogeneity of mesenchymal stem cells: implications for cell therapy. J Cell Biochem 113(9):2806–2812. https://doi.org/10.1002/jcb.24166
Phinney DG, Kopen G, Righter W, Webster S, Tremain N, Prockop DJ (1999) Donor variation in the growth properties and osteogenic potential of human marrow stromal cells. J Cell Biochem 75(3):424–436
Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, Moorman MA et al (1999) Multilineage potential of adult human mesenchymal stem cells. Science 284(5411):143–147. https://doi.org/10.1126/science.284.5411.143
Potapova IA, Gaudette GR, Brink PR, Robinson RB, Rosen MR, Cohen IS, Doronin SV (2007) Mesenchymal stem cells support migration, extracellular matrix invasion, proliferation, and survival of endothelial cells in vitro. Stem Cells 25(7):1761–1768. https://doi.org/10.1634/stemcells.2007-0022
Powell D, Lou M, Barros Becker F, Huttenlocher A (2018) Cxcr1 mediates recruitment of neutrophils and supports proliferation of tumor-initiating astrocytes in vivo. Sci Rep 8(1):13285. https://doi.org/10.1038/s41598-018-31675-0
Prockop DJ (1997) Marrow stromal cells as stem cells for nonhematopoietic tissues. Science 276(5309):71–74. https://doi.org/10.1126/science.276.5309.71
Prockop DJ, Oh JY (2012) Mesenchymal stem/stromal cells (MSCs): role as guardians of inflammation. Mol Ther 20(1):14–20. https://doi.org/10.1038/mt.2011.211
Qiao L, Xu ZL, Zhao TJ, Ye LH, Zhang XD (2008a) Dkk-1 secreted by mesenchymal stem cells inhibits growth of breast cancer cells via depression of Wnt signalling. Cancer Lett 269(1):67–77. https://doi.org/10.1016/j.canlet.2008.04.032
Qiao L, Xu Z, Zhao T, Zhao Z, Shi M, Zhao RC, Ye L et al (2008b) Suppression of tumorigenesis by human mesenchymal stem cells in a hepatoma model. Cell Res 18(4):500–507. https://doi.org/10.1038/cr.2008.40
Rafei M, Birman E, Forner K, Galipeau J (2009) Allogeneic mesenchymal stem cells for treatment of experimental autoimmune encephalomyelitis. Mol Ther 17(10):1799–1803. https://doi.org/10.1038/mt.2009.157
Raffaghello L, Bianchi G, Bertolotto M, Montecucco F, Busca A, Dallegri F, Ottonello L et al (2008) Human mesenchymal stem cells inhibit neutrophil apoptosis: a model for neutrophil preservation in the bone marrow niche. Stem Cells 26(1):151–162. https://doi.org/10.1634/stemcells.2007-0416
Ramos-Zúñiga R, González-Pérez O, Macías-Ornelas A, Capilla-González V, Quiñones-Hinojosa A (2012) Ethical implications in the use of embryonic and adult neural stem cells. Stem Cells Int 2012:470949. https://doi.org/10.1155/2012/470949
Rani S, Ryan AE, Griffin MD, Ritter T (2015) Mesenchymal stem cell-derived extracellular vesicles: toward cell-free therapeutic applications. Mol Ther 23(5):812–823. https://doi.org/10.1038/mt.2015.44
Relation T, Yi T, Guess AJ, La Perle K, Otsuru S, Hasgur S, Dominici M et al (2018) Intratumoral delivery of interferonγ-secreting mesenchymal stromal cells repolarizes tumor-associated macrophages and suppresses neuroblastoma proliferation in vivo. Stem Cells 36(6):915–924. https://doi.org/10.1002/stem.2801
Ren G, Su J, Zhang L, Zhao X, Ling W, L’huillie A, Zhang J et al (2009) Species variation in the mechanisms of mesenchymal stem cell-mediated immunosuppression. Stem Cells 27(8):1954–1962. https://doi.org/10.1002/stem.118
Ren G, Zhao X, Zhang L, Zhang J, L’Huillier A, Ling W, Roberts AI et al (2010) Inflammatory cytokine-induced intercellular adhesion molecule-1 and vascular cell adhesion molecule-1 in mesenchymal stem cells are critical for immunosuppression. J Immunol 184(5):2321–2328. https://doi.org/10.4049/jimmunol.0902023
Rennert RC, Sorkin M, Januszyk M, Duscher D, Kosaraju R, Chung MT, Lennon J et al (2014) Diabetes impairs the angiogenic potential of adipose-derived stem cells by selectively depleting cellular subpopulations. Stem Cell Res Ther 5(3):79. https://doi.org/10.1186/scrt468
Ricciardi M, Malpeli G, Bifari F, Bassi G, Pacelli L, Nwabo Kamdje AH, Chilosi M et al (2012) Comparison of epithelial differentiation and immune regulatory properties of mesenchymal stromal cells derived from human lung and bone marrow. PLoS One 7(5):e35639. https://doi.org/10.1371/journal.pone.0035639
Ringdén O, Uzunel M, Rasmusson I, Remberger M, Sundberg B, Lönnies H, Marschall HU et al (2006) Mesenchymal stem cells for treatment of therapy-resistant graft-versus-host disease. Transplantation 81(10):1390–1397. https://doi.org/10.1097/01.tp.0000214462.63943.14
Ringe J, Strassburg S, Neumann K, Endres M, Notter M, Burmester GR, Kaps C et al (2007) Towards in situ tissue repair: human mesenchymal stem cells express chemokine receptors CXCR1, CXCR2 and CCR2, and migrate upon stimulation with CXCL8 but not CCL2. J Cell Biochem 101(1):135–146. https://doi.org/10.1002/jcb.21172
Rivera FJ, de la Fuente AG, Zhao C, Silva ME, Gonzalez GA, Wodnar R, Feichtner M et al (2019) Aging restricts the ability of mesenchymal stem cells to promote the generation of oligodendrocytes during remyelination. Glia 67(8):1510–1525. https://doi.org/10.1002/glia.23624
Røsland GV, Svendsen A, Torsvik A, Sobala E, McCormack E, Immervoll H, Mysliwietz J et al (2009) Long-term cultures of bone marrow-derived human mesenchymal stem cells frequently undergo spontaneous malignant transformation. Cancer Res 69(13):5331–5339. https://doi.org/10.1158/0008-5472.CAN-08-4630
Sackstein R, Merzaban JS, Cain DW, Dagia NM, Spencer JA, Lin CP, Wohlgemuth R (2008) Ex vivo glycan engineering of CD44 programs human multipotent mesenchymal stromal cell trafficking to bone. Nat Med 14(2):181–187. https://doi.org/10.1038/nm1703
Sakaguchi Y, Sekiya I, Yagishita K, Muneta T (2005) Comparison of human stem cells derived from various mesenchymal tissues: superiority of synovium as a cell source. Arthritis Rheum 52(8):2521–2529. https://doi.org/10.1002/art.21212
Sensebé L, Gadelorge M, Fleury-Cappellesso S (2013) Production of mesenchymal stromal/stem cells according to good manufacturing practices: a review. Stem Cell Res Ther 4(3):66. https://doi.org/10.1186/scrt217
Seo SH, Kim KS, Park SH, Suh YS, Kim SJ, Jeun SS, Sung YC (2011) The effects of mesenchymal stem cells injected via different routes on modified IL-12-mediated antitumor activity. Gene Ther 18(5):488–495. https://doi.org/10.1038/gt.2010.170
Shabbir A, Zisa D, Suzuki G, Lee T (2009) Heart failure therapy mediated by the trophic activities of bone marrow mesenchymal stem cells: a noninvasive therapeutic regimen. Am J Physiol Heart Circ Physiol 296(6):H1888–H1897. https://doi.org/10.1152/ajpheart.00186.2009
Shin L, Peterson DA (2012) Impaired therapeutic capacity of autologous stem cells in a model of type 2 diabetes. Stem Cells Transl Med 1(2):125–135. https://doi.org/10.5966/sctm.2012-0031
Shrestha C, Zhao L, Chen K, He H, Mo Z (2013) Enhanced healing of diabetic wounds by subcutaneous administration of human umbilical cord derived stem cells and their conditioned media. Int J Endocrinol 2013:592454. https://doi.org/10.1155/2013/592454
Sivanathan KN, Gronthos S, Rojas-Canales D, Thierry B, Coates PT (2014) Interferon-gamma modification of mesenchymal stem cells: implications of autologous and allogeneic mesenchymal stem cell therapy in allotransplantation. Stem Cell Rev Rep 10(3):351–375. https://doi.org/10.1007/s12015-014-9495-2
Song JY, Kang HJ, Ju HM, Park A, Park H, Hong JS, Kim CJ et al (2019) Umbilical cord-derived mesenchymal stem cell extracts ameliorate atopic dermatitis in mice by reducing the T cell responses. Sci Rep 9(1):6623. https://doi.org/10.1038/s41598-019-42964-7
Sordi V, Malosio ML, Marchesi F, Mercalli A, Melzi R, Giordano T, Belmonte N et al (2005) Bone marrow mesenchymal stem cells express a restricted set of functionally active chemokine receptors capable of promoting migration to pancreatic islets. Blood 106(2):419–427. https://doi.org/10.1182/blood-2004-09-3507
Soria-Juan B, Escacena N, Capilla-González V, Aguilera Y, Llanos L, Tejedo JR, Bedoya FJ et al (2019) Cost-effective, safe, and personalized cell therapy for critical limb ischemia in type 2 diabetes mellitus. Front Immunol 10:1151. https://doi.org/10.3389/fimmu.2019.01151
Sotiropoulou PA, Perez SA, Gritzapis AD, Baxevanis CN, Papamichail M (2006) Interactions between human mesenchymal stem cells and natural killer cells. Stem Cells 24(1):74–85. https://doi.org/10.1634/stemcells.2004-0359
Spaggiari GM, Abdelrazik H, Becchetti F, Moretta L (2009) MSCs inhibit monocyte-derived DC maturation and function by selectively interfering with the generation of immature DCs: central role of MSC-derived prostaglandin E2. Blood 113(26):6576–6583. https://doi.org/10.1182/blood-2009-02-203943
Stagg J, Pommey S, Eliopoulos N, Galipeau J (2006) Interferon-gamma-stimulated marrow stromal cells: a new type of nonhematopoietic antigen-presenting cell. Blood 107(6):2570–2577. https://doi.org/10.1182/blood-2005-07-2793
Stenderup K, Justesen J, Clausen C, Kassem M (2003) Aging is associated with decreased maximal life span and accelerated senescence of bone marrow stromal cells. Bone 33(6):919–926. https://doi.org/10.1016/j.bone.2003.07.005
Sudres M, Norol F, Trenado A, Grégoire S, Charlotte F, Levacher B, Lataillade JJ et al (2006) Bone marrow mesenchymal stem cells suppress lymphocyte proliferation in vitro but fail to prevent graft-versus-host disease in mice. J Immunol 176(12):7761–7767. https://doi.org/10.4049/jimmunol.176.12.7761
Sun LY, Zhang HY, Feng XB, Hou YY, Lu LW, Fan LM (2007) Abnormality of bone marrow-derived mesenchymal stem cells in patients with systemic lupus erythematosus. Lupus 16(2):121–128. https://doi.org/10.1177/0961203306075793
Sun L, Wang D, Liang J, Zhang H, Feng X, Wang H, Hua B et al (2010) Umbilical cord mesenchymal stem cell transplantation in severe and refractory systemic lupus erythematosus. Arthritis Rheum 62(8):2467–24675. https://doi.org/10.1002/art.27548
Suresh SC, Selvaraju V, Thirunavukkarasu M, Goldman JW, Husain A, Alexander Palesty J, Sanchez JA et al (2015) Thioredoxin-1 (Trx1) engineered mesenchymal stem cell therapy increased pro-angiogenic factors, reduced fibrosis and improved heart function in the infarcted rat myocardium. Int J Cardiol 201:517–528. https://doi.org/10.1016/j.ijcard.2015.08.117
Takeda K, Hayakawa Y, Smyth MJ, Kayagaki N, Yamaguchi N, Kakuta S, Iwakura Y et al (2001) Involvement of tumor necrosis factor-related apoptosis-inducing ligand in surveillance of tumor metastasis by liver natural killer cells. Nat Med 7(1):94–100. https://doi.org/10.1038/83416
Tan J, Wu W, Xu X, Liao L, Zheng F, Messinger S, Sun X et al (2012) Induction therapy with autologous mesenchymal stem cells in living-related kidney transplants: a randomized controlled trial. JAMA 307(11):1169–1177. https://doi.org/10.1001/jama.2012.316
Tan KL, Chia WC, How CW, Tor YS, Show PL, Looi QHD, Foo JB (2021) Benchtop isolation and characterisation of small extracellular vesicles from human mesenchymal stem cells. Mol Biotechnol. https://doi.org/10.1007/s12033-021-00339-2
Teraa M, Sprengers RW, Schutgens RE, Slaper-Cortenbach IC, van der Graaf Y, Algra A, van der Tweel I et al (2015) Effect of repetitive intra-arterial infusion of bone marrow mononuclear cells in patients with no-option limb ischemia: the randomized, double-blind, placebo-controlled Rejuvenating Endothelial Progenitor Cells via Transcutaneous Intra-arterial Supplementation (JUVENTAS) trial. Circulation 131(10):851–860. https://doi.org/10.1161/CIRCULATIONAHA.114.012913
Tisato V, Naresh K, Girdlestone J, Navarrete C, Dazzi F (2007) Mesenchymal stem cells of cord blood origin are effective at preventing but not treating graft-versus-host disease. Leukemia 21(9):1992–1999. https://doi.org/10.1038/sj.leu.2404847
Uccelli A, Moretta L, Pistoia V (2008) Mesenchymal stem cells in health and disease. Nat Rev Immunol 8(9):726–736. https://doi.org/10.1038/nri2395
Viswanathan S, Shi Y, Galipeau J, Krampera M, Leblanc K, Martin I, Nolta J et al (2019) Mesenchymal stem versus stromal cells: International Society for Cell & Gene Therapy (ISCT®) Mesenchymal Stromal Cell committee position statement on nomenclature. Cytotherapy 21(10):1019–1024. https://doi.org/10.1016/j.jcyt.2019.08.002
von Einem JC, Guenther C, Volk HD, Grütz G, Hirsch D, Salat C, Stoetzer O et al (2019) Treatment of advanced gastrointestinal cancer with genetically modified autologous mesenchymal stem cells: results from the phase 1/2 TREAT-ME-1 trial. Int J Cancer 145(6):1538–1546. https://doi.org/10.1002/ijc.32230
Von Lüttichau I, Notohamiprodjo M, Wechselberger A, Peters C, Henger A, Seliger C, Djafarzadeh R et al (2005) Human adult CD34- progenitor cells functionally express the chemokine receptors CCR1, CCR4, CCR7, CXCR5, and CCR10 but not CXCR4. Stem Cells Dev 14(3):329–336. https://doi.org/10.1089/scd.2005.14.329
Wang J, Liu X, Qiu Y, Shi Y, Cai J, Wang B, Wei X et al (2018) Cell adhesion-mediated mitochondria transfer contributes to mesenchymal stem cell-induced chemoresistance on T cell acute lymphoblastic leukemia cells. J Hematol Oncol 11(1):11. https://doi.org/10.1186/s13045-018-0554-z
Wang Y, Huso DL, Harrington J, Kellner J, Jeong DK, Turney J, McNiece IK (2005) Outgrowth of a transformed cell population derived from normal human BM mesenchymal stem cell culture. Cytotherapy 7(6):509–519. https://doi.org/10.1080/14653240500363216
Wang B, Wu SM, Wang T, Liu K, Zhang G, Zhang XQ, Yu JH et al (2012) Pre-treatment with bone marrow-derived mesenchymal stem cells inhibits systemic intravascular coagulation and attenuates organ dysfunction in lipopolysaccharide-induced disseminated intravascular coagulation rat model. Chin Med J 125(10):1753–1759
Wegmeyer H, Bröske AM, Leddin M, Kuentzer K, Nisslbeck AK, Hupfeld J, Wiechmann K et al (2013) Mesenchymal stromal cell characteristics vary depending on their origin. Stem Cells Dev 22(19):2606–2618. https://doi.org/10.1089/scd.2013.0016
Wessman SJ, Levings RL (1999) Benefits and risks due to animal serum used in cell culture production. Dev Biol Stand 99:3–8
Wiley SR, Schooley K, Smolak PJ, Din WS, Huang CP, Nicholl JK, Sutherland GR et al (1995) Identification and characterization of a new member of the TNF family that induces apoptosis. Immunity 3(6):673–682. https://doi.org/10.1016/1074-7613(95)90057-8
Wobus M, List C, Dittrich T, Dhawan A, Duryagina R, Arabanian LS, Kast K et al (2015) Breast carcinoma cells modulate the chemoattractive activity of human bone marrow-derived mesenchymal stromal cells by interfering with CXCL12. Int J Cancer 136(1):44–54. https://doi.org/10.1002/ijc.28960
Woodbury D, Reynolds K, Black IB (2002) Adult bone marrow stromal stem cells express germline, ectodermal, endodermal, and mesodermal genes prior to neurogenesis. J Neurosci Res 69(6):908–917. https://doi.org/10.1002/jnr.10365
Wynn RF, Hart CA, Corradi-Perini C, O’Neill L, Evans CA, Wraith JE, Fairbairn LJ et al (2004) A small proportion of mesenchymal stem cells strongly expresses functionally active CXCR4 receptor capable of promoting migration to bone marrow. Blood 104(9):2643–2645. https://doi.org/10.1182/blood-2004-02-0526
Xu WT, Bian ZY, Fan QM, Li G, Tang TT (2009) Human mesenchymal stem cells (hMSCs) target osteosarcoma and promote its growth and pulmonary metastasis. Cancer Lett 281(1):32–41. https://doi.org/10.1016/j.canlet.2009.02.022
Yañez R, Lamana ML, García-Castro J, Colmenero I, Ramírez M, Bueren JA (2006) Adipose tissue-derived mesenchymal stem cells have in vivo immunosuppressive properties applicable for the control of the graft-versus-host disease. Stem Cells 24(11):2582–2591. https://doi.org/10.1634/stemcells.2006-0228
Yoo KH, Jang IK, Lee MW, Kim HE, Yang MS, Eom Y, Lee JE et al (2009) Comparison of immunomodulatory properties of mesenchymal stem cells derived from adult human tissues. Cell Immunol 259(2):150–156. https://doi.org/10.1016/j.cellimm.2009.06.010
Zappia E, Casazza S, Pedemonte E, Benvenuto F, Bonanni I, Gerdoni E, Giunti D et al (2005) Mesenchymal stem cells ameliorate experimental autoimmune encephalomyelitis inducing T-cell anergy. Blood 106(5):1755–1761. https://doi.org/10.1182/blood-2005-04-1496
Zhang J, Li Y, Chen J, Cui Y, Lu M, Elias SB, Mitchell JB et al (2005) Human bone marrow stromal cell treatment improves neurological functional recovery in EAE mice. Exp Neurol 195(1):16–26. https://doi.org/10.1016/j.expneurol.2005.03.018
Zhang T, Lee YW, Rui YF, Cheng TY, Jiang XH, Li G (2013) Bone marrow-derived mesenchymal stem cells promote growth and angiogenesis of breast and prostate tumors. Stem Cell Res Ther 4(3):70. https://doi.org/10.1186/scrt221
Zhao Q, Gregory CA, Lee RH, Reger RL, Qin L, Hai B, Park MS et al (2015) MSCs derived from iPSCs with a modified protocol are tumor-tropic but have much less potential to promote tumors than bone marrow MSCs. Proc Natl Acad Sci U S A 112(2):530–535. https://doi.org/10.1073/pnas.1423008112
Zhao ZG, Liang Y, Li K, Li WM, Li QB, Chen ZC, Zou P (2007) Phenotypic and functional comparison of mesenchymal stem cells derived from the bone marrow of normal adults and patients with hematologic malignant diseases. Stem Cells Dev 16(4):637–648. https://doi.org/10.1089/scd.2007.0008
Zhou T, Yuan Z, Weng J, Pei D, Du X, He C, Lai P (2021) Challenges and advances in clinical applications of mesenchymal stromal cells. J Hematol Oncol 14(1):24. https://doi.org/10.1186/s13045-021-01037-x
Zhu J, Liu Y, Chen C, Chen H, Huang J, Luo Y, Zhao K et al (2021) Cyasterone accelerates fracture healing by promoting MSCs migration and osteogenesis. J Orthop Translat 28:28–38. https://doi.org/10.1016/j.jot.2020.11.004
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Section Editor information
Rights and permissions
Copyright information
© 2022 Springer Nature Singapore Pte Ltd.
About this entry
Cite this entry
Hmadcha, A., Soria, B., Tejedo, J.R., Bedoya, F.J., Sempere-Ortells, J.M., Smani, T. (2022). Considerations for Clinical Use of Mesenchymal Stromal Cells. In: Haider, K.H. (eds) Handbook of Stem Cell Therapy. Springer, Singapore. https://doi.org/10.1007/978-981-19-2655-6_3
Download citation
DOI: https://doi.org/10.1007/978-981-19-2655-6_3
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-19-2654-9
Online ISBN: 978-981-19-2655-6
eBook Packages: Biomedical and Life SciencesReference Module Biomedical and Life Sciences