Abstract
Diabetes mellitus, a chronic metabolic dysfunction found in people of different age groups worldwide, is now seriously threatening mankind’s health. Despite the application of insulin and other synthetic oral anti-diabetic drugs, there is a great need for the discovery and development of novel anti-diabetic drugs of plant origin as the synthetic drugs have more side effects in long-term use. Therefore, researchers engaged in discovering novel bioactive compounds from plants bearing anti-diabetic potential also have fewer unwanted side effects than conventional drugs. In this chapter, an attempt has been made to discuss the prospective medicinal plants comprising either plant extracts or isolated bioactive phyto-constituents bearing anti-diabetic potential, which has been reported in several in vitro, in vivo, or clinical studies. Because of this, the mechanism of action and the management of diabetes will be valuable to scientists, chemists, and pharmaceutical corporations for the discovery of novel anti-diabetic drugs in the future.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Plant-human interaction has long been a part of the history of civilization (Mukherjee et al., 2007). About 60% of medicine is naturally derived, i.e. directly plant or plant-based secondary metabolites. The recent trend shows more experimental work based on plants, around triple the amount from the past. Diabetes is a steadily rising trend globally, with around 422 million adults inflicted in 2014 compared to 108 million adults in 1980, which from 4.7% doubled to 8.5%. Early life was lost in around 43% of all deaths due to high blood glucose (World Health Organization (WHO) Global report on diabetes, 2020). In 2021, the International Diabetes Federation (IDF) Atlas 10 edition highlighted 537 million adults with diabetes between 20 and 79 years, and this number of cases will rise to around 643 million and 783 million by 2030 and 2045 years, respectively, and more will be affected in low- and middle-income countries, with loss of life accounting for 6.7 million.
It has traditionally been the most difficult process to find drugs from natural plants through product identification and proper application of that to a “lead drug,” but recent multidisciplinary research approaches have placed a high priority on developing natural drugs for a variety of molecular targets and the treatment of various illnesses and disease conditions. In ancient times, people used rawer extraction, a purification technique, for plant drug discovery. At that time, an empirical technique was preferred over a rational method for discovering new medicines. Evidence from recorded history suggests that the Mesopotamian culture, which dates back to 2600 BC, utilized medicine derived from 1000 different plant species. Egyptian medicine dates back to 2900 BC, and some medical documents called the ‘Ebers Papyrus’, which were discovered around 1550 BC, describe 700 medications derived from plants. Ayurveda from India dates back to the first millennium BC, whereas the Chinese medical system is over a thousand years old. Greek and Roman cultures of medicine contributed significantly to the western world’s extensive knowledge of herbal medicine. These cultures contribute their self-knowledge to the Arabian medicinal system, part of which comes from India’s and China’s old medical practices (Atanas et al., 2015). Though individuals still use them as unrecognized drug states, ethnographic data on a plant with medical benefits is a big interest to new research perspectives.
Ayurveda is a notable example of this source and is best suited for anti-diabetic drugs—metformin and more biguanide drug were made by copying like galegine from Galega officinalis L. plant. Plant phytochemical for drug testing is related to traditional in vitro and in vivo approaches. This pharmacological method is divided into forwarding pharmacological and reverse pharmacological approaches. The primary step in identifying and evaluating a pre-drug plant is to acquire animal data based on signs and symptoms or in vitro experiments. If the available biomolecules are active and interact with the target protein in vivo, or if searching phytochemical data for specific functional molecules and their application in animals or other species, then this is an example of a ‘reverse approach’ to the process of discovering new drugs from plants. The preliminary step in screening plant bioactives is the extraction of either aqueous or organic solvents. After that, fractionalization for the isolation of interactive compounds is performed using methods such as column chromatography, which is a traditional method. However, the most sensitive characterization is performed using nuclear magnetic resonance (NMR), gas chromatography-mass spectroscopy (GCMS), or high-performance liquid chromatography (HPLC), which can distinguish secondary plant metabolites more accurately (Atanas et al., 2015).
2 History of Indian Medicinal Plants for Diabetes Control
Traditional herbal medicine has existed worldwide since the prehistoric era. It was recorded in Ayurveda, Chinese, Greek, and Egyptian for various therapeutic uses; meanwhile, African and American native people use herbs in their cultural rituals. Ayurveda has become one of the ancient alternative Indian medicine systems in the period between 4000 BC and 1500 BC, where the history of medical knowledge about plants or herbal therapy was well recognized in two early textbooks, i.e. Charaka Samhita and Sushruta Samhita. Around 1000 BC, the Indian medical practice of Ayurveda referred to diabetes mellitus (DM) as madhumeha, which is a type of Prameha. At that time diabetes was a mythological view mentioned eating Havisha, a special type of food i.e., offered during the Yagna occasion by Daksha Prajapati. In the Vedic period, around 600 BC, this illness was referred to as ‘Asrava’, and its detailed description was recorded in Charaka Samhita, Sushruta Samhita, and Vagbhatta (Singh, 2011). Since then, indigenous herbal medicines as raw forms have been strong reflections. For example, Gymnema sylvestre (Asclepiadaceae) is native to the tropical forest of South West India. This plant leaf extract was used as anti-diabetic medicine in the contemporary period of Sushruta (600 BC) (Laha & Paul, 2019). But in the early nineteenth century, scientific advancement with time beings industrialists, scientists, and researchers focused on synthetic pharmaceuticals, which show the declinature of herbal value. However, recent initiatives are being utilized to revive and widely promote traditional plant medicine in the mainstream medical system while considering quality and safety issues (Parasuraman et al., 2014). A WHO-estimated report says that about 80% of the world population from third-world countries trust traditional plant medicine for health care (Mathew & Babu, 2011; Amalraj & Gopi, 2016). About 60% of rural people in India use herbal medicine (Amalraj & Gopi, 2016). The Indian subcontinent is a rich diversity of 45,000 plant species, and 15,000 medicinal flora have been recognized. Knowingly, out of the medicinal plants, 7000–7500 species have been used by community people to cure diseases. Ayurveda recorded about 700 types of medicinal plants used in the ancient era (Parasuraman et al., 2014). Importantly, literature data shows more than 400 hypoglycaemic plants have been discovered, but still surprising to discover new diabetic plants serve as an alternative in the most attractive field. Information from an ethnobotanical survey suggests that 800 plants may pose potent anti-diabetic properties. Out of these, three have been evaluated as beneficial for treating type-2 diabetes mellitus—Trigonella foenum-graecum, Pterocarpus marsupium, and Momordica charantia (Patel et al., 2012; Ponnusamy et al., 2011). Because even the most advanced allopathic system cannot entirely cure diabetes and oral hypoglycaemic medicines have caused a number of unpleasant side effects, the entire globe is concentrating its efforts now on herbal treatments for the disease (Srivastava et al., 2012). That is why in recent years, considerable importance has been given to plant herbal medicine for diabetes management. Herbal preparation makes more polyherbal formulations due to extra therapeutic benefits than a single herbal formulation. The combination of herbs improves the pharmacological activity of polyherbal formulation (Parasuraman et al., 2014). Examples include the Indian company Diabet (Herbal Galenicals), which manufactures anti-diabetic herbal medicines using a variety of plant extracts, including Strychnos potatorum, Tamarindus indica, Tribulus terrestris, Curcuma longa, Coscinium fenestratum, and Phyllanthus reticulates, among others (Umamaheswari et al., 2010). Other anti-diabetic treatments include Diabrid, Diakyur, Dihar, Dianex, Diashis, Diasulin, and Diasol, all of which have been shown to have effects that are compatible with those of regular allopathic drugs (Srivastava et al., 2012).
3 Pharmacological Management of Diabetes Mellitus
According to the World Health Organization (WHO), diabetes mellitus is a chronic and metabolic disorder characterized by a rise in blood glucose (or blood sugar) level, which leads over time to serious damage to the eyes, blood vessels, kidneys, heart, nerves, etc. This disease is divided into type-1 diabetes and type-2 diabetes. In patients with type-1 diabetes, once known as insulin-dependent diabetes or juvenile diabetes chronic conditions, the body does not produce enough (or any) insulin; that is why these patients are required to inject insulin directly. Type-2 diabetes is the most common type of diabetes, which is usually found in adults. Diabetes develops when the human body either develops insulin resistance or fails to produce sufficient amounts of insulin. After a while, diabetes mellitus is accompanied by specific vascular and neuropathic complications (Ngugi et al., 2012). The frequency of type-2 diabetes in the population has dramatically risen in the past three decades in countries of all income levels. It is critical to the survival of people with diabetes, although they access affordable treatment, including insulin. There is a global consensus that the rate of increase in diabetes and obesity should be slowed down by the year 2025 (WHO, 2022a, 2022b). Thus, nowadays, there is a great need to manage diabetes to avoid or reduce chronic complications arising from it and to prevent acute complications like hyper- and hypoglycemia, blindness, limb amputation, and cardiovascular diseases (CVDs).
4 Pharmacological Management
Over 61 million people in India have diabetes, so it can be said that India is the ‘capital of diabetes’. It is challenging to treat diabetes and manage all the complications simultaneously because of inadequate health care and a lack of sufficient facilities (Jacob & Narendhirakannan, 2019). In the past, medical practitioners used insulin and sulfonylureas to treat patients with diabetes mellitus type 2. But nowadays, several other medications are available on the market to treat type-2 diabetes. These are acarbose, migital, repaglinide, metformin hydrochloride, troglitazone, and rosiglitazone. In the near future, hopefully several other agents will be available to treat patients with diabetes. To control diabetic patients’ blood sugar levels without requiring insulin injections, physicians will need to use these agents. Here are some common uses of these drugs.
4.1 Insulin Therapy
Insulin is a peptide hormone secreted by beta cells located in the islets of the pancreas. Insulin is released into the bloodstream as a consequence of the ingestion of food particles. This step assists in the movement of glucose from the food particles we have consumed into cells so that it can be used as a source of energy.
4.2 For Patients with Type-1 Diabetes
Due to the autoimmune reaction of the body of patients with type-1 diabetes mellitus, insulin-producing cells have been destroyed, so the body produces little or no insulin. In this scenario, it is necessary to replace the insulin in the body on a daily basis by injecting it.
4.3 For Patients with Type-2 Diabetes
In the case of type-2 diabetes, the body produces insulin but does not work correctly. In order to maintain stable blood sugar levels and postpone the requirement for tablets and insulin, lifestyle changes are necessary. When the patient requires insulin, it is important to understand that this is just the condition’s natural progression (Diabetes Australia, 2022).
4.4 Sulfonylureas
Patients who still have some function in their pancreatic beta cells may experience a reduction in their blood glucose level as a result of the ability of sulfonylureas to stimulate increased insulin secretion from their pancreatic beta cells. The stimulation of pancreatic beta cells by sulfonylureas can result in an increase in the amount of insulin that is secreted. In the earlier stages of type-2 diabetes, sulfonylureas are a more effective medication for patients who have an increased pancreatic beta-cell function (Costello & Shivkumar, 2021).
4.5 Metformin Hydrochloride
Metformin is prescribed to patients who have type-2 diabetes. Metformin is an anti-diabetic medication that is available in both immediate-release and extended-release forms. Additionally, it is frequently combined with other anti-diabetic medications in a variety of combination products. The treatment and prevention of polycystic ovary syndrome (PCOS), the management of antipsychotic-induced weight gain, gestational diabetes, the prevention of type-2 diabetes, etc. are some of the off-label indications that have been approved for the use of this medication. Metformin is currently the only anti-diabetic medication that is recommended for pre-diabetic patients by the American Diabetes Association. Anti-aging effects, cancer prevention, and neuroprotection are just some of the possible applications for this compound.
4.6 Acarbose
Acarbose is a medication prescribed to patients with type-2 diabetes. This medication should be taken orally before each meal to reduce the amount of sugar that remains in the blood. Diabetes mellitus can lead to many complications over time, but taking this medication can help prevent those complications.
5 Potential Plant Used for Diabetes Control
Diabetes is a chronic lifestyle disease that is not completely curable but controllable and manifests macronutrients–carbohydrate, protein, and fat metabolic impairments. It will become the world’s most-wanted killer disease in the coming 25 years. Literature source describes over 400 medicinal plant species that have hypoglycaemic effects. Plants are diverse sources of important bioactive components; many of these are potentially used in diabetes management (Malviya et al., 2010). The following figure describes the potential plants with their family bearing anti-diabetic activity (Fig. 1).
6 Medicinal Plants and Their Active Parts in Diabetes Mellitus Management
Due to the presence of therapeutically important active photo components, medicinal plants have a significant potential to treat a wide range of diseases and conditions. In recent years, various developing and underdeveloped countries have relied on medicinal plants to cure several diseases since herbal medicines are accessible, cost-effective, and without any or fewer side effects. Diabetes mellitus is a serious metabolic disorder in human beings. There are several medicines available on the market to alleviate the disease. However, these drugs are more expensive and are associated with several complications to the human body. So contemporary researchers have investigated various medicinal plants with anti-diabetic activities. Here, we will discuss various active medicinal plant parts used to treat diabetes mellitus (Tables 1, 2 and 3).
7 Phyto-Chemistry of Medicinal Plants
Plants used for medicinal purposes are an abundant source of various vital bioactive photo components as well as bio-nutrients. These bioactive phytochemicals are generally called active principles or plant secondary metabolites. Plant secondary metabolites generally group according to their biosynthetic pathways and can be grouped into three large groups: alkaloids, phenolic compounds, and terpenes (Francesca et al., 2019).
7.1 Alkaloids
The word ‘alkaloid’ comes from the Arabic word ‘al-qali’, which refers to the plant from which soda was first isolated. The term ‘alkaloid’ has been in use since the nineteenth century (Kaur & Arora, 2015). The term ‘alkaloid’ was first used in 1819 by the German chemist Carl F. W. Meissner to refer to a substance derived from plants that possessed alkaline characteristics. According to, alkaloids are base-type compounds that contain an N-atom at any position in the molecule and do not include N in an amide or peptide bond. Furthermore, alkaloids are distinguished from amines and peptides by the absence of an amide bond. According to Gutiérrez et al.’s research from 2020, alkaloids are abundant in bacteria, fungi, plants, and animals, and they are formed when acids combine with alkaloids to make salts. One of the defining characteristics of alkaloids is that they do not contain nitrogen in the amide or peptide bonds that they form. Since ancient times, purgatives, anti-tussives, and sedatives that are derived from plants have been used as part of folklore medicine to treat a wide variety of diseases and ailments. Alkaloids have played a significant role in the treatment of these conditions. The low MW (molecular weight) structures that makeup alkaloids account for approximately 20% of the secondary metabolites that are plant-derived. It is estimated that approximately 12,000 different alkaloids have been isolated to this point from various plant families across the kingdom of plants (Kaur & Arora, 2015). Alkaloids are typically produced by plants in order to facilitate their existence in the ecosystem, the formation of seeds, and their escape from a variety of predators. Alkaloids are the most abundant and diverse group of secondary metabolites. They also have anti-diabetic activity because they inhibit enzymes such as beta-amylase, beta-glucosidase, dipeptidyl peptidase-IV, aldose reductase, and protein tyrosine phosphatase-1B, among others.
The anti-hyperglycaemic activity of the roots of Aerva lanata Linn. (Amaranthaceae) was investigated in an experiment conducted by Agrawal et al. (2013). The results of the experiment showed that the plant exhibits this kind of activity due to the presence of alkaloids known as canthin-6-one derivatives. Sangeetha, Priya, and Vasanthi conducted an investigation in vitro for the purpose of determining the anti-hyperglycaemic potential of the stem of Tinospora cordifolia in 2013. Because it contains palmatine and alkaloid, the stem of Tinospora cordifolia has been shown to have anti-hyperglycaemic properties when it has been extracted with petroleum ether.
The anti-diabetic potential of alkaloids such as vindoline, vindolidine, vindolicine, and vindolinine was demonstrated by Tiong et al. (2013) when they were extracted from the leaves of Catharanthus roseus. In another experiment, discovered that the seed of Brassica oleracea var. capitata contains 2,3-Dicyano-5,6-diphenyl pyrazine, which is an alkaloid that has anti-diabetic activities.
7.2 Phenolic Compounds
Phenolic compounds, phenolics, and polyphenolics are all umbrella terms that refer to the same thing: chemical compounds that have at least one aromatic ring and one or more hydroxyl substituents. Phenolic compounds, phenolics, and polyphenolics are all examples of these types of compounds. This encompasses functional derivatives like esters, methyl ethers, glycosides, and other compounds that are very similar to these. Over 8000 distinct types of phenolic compounds can be found in the plant kingdom (Bhuyan & Basu, 2017; Ho, 1992; Cartea et al., 2011). Whereas the term ‘plant phenolics’ refers to the natural secondary metabolites that arise through biogenesis from either the shikimic acid pathway or the phenylpropanoid pathway, which directly provides phenylpropanoids, or the ‘polyketide’ acetate/malonate pathway, which also accomplishes a very wide range of physiological roles in medicinal plants, the phenylpropanoids that are directly provided by the phenylpropa (Quideau et al., 2011; Harborne, 1989).
Ever since the course of evolutionary consequences of various plant lineages, higher plants have synthesized several thousand known phenolic compounds. This has allowed plants to tolerate adverse environmental challenges over the course of evolutionary time. When plants are subjected to a variety of environmental stresses, such as pathogen infections, herbivores, low temperatures, high light, and nutrient deficiency, the phenolic compounds found in the plants play an important role in the defence mechanisms that the plants employ. This can increase the number of free radicals and other oxidative species produced by the plants (Lattanzio, 2013).
7.3 Terpenes
Terpenes are the most numerous and diverse group of naturally occurring chemical compounds that can be found almost exclusively in plants. They are also known as terpenoids and isoprenoids. Terpenes, such as sterols and squalenes, have also been discovered in animal tissues. The majority of terpenes have structures that are multicyclic and contain oxygen-containing functional groups. Terpenoids account for approximately 60% of all known natural products that play a role in the secondary metabolism of plants. Despite the fact that the nomenclature of this compound is sometimes used interchangeably with ‘terpenes’, the compounds in question are hydrocarbons, whereas terpenoids have additional functional groups, most of which contain the element oxygen. Plants have a wide variety of pigments, aromas, and flavours, all of which are caused by the presence of terpenes (Cox-Georgian et al., 2019). The presence of terpenoids in plants is one factor that contributes to their aromatic qualities, and as a result, they play an important role in traditional herbal medicine. In addition, terpenes serve a number of other important functions in plants, including signalling, thermoprotection, flavouring, pigmentation, and solvent production. Terpenes also have a number of applications in the medical field (Cox-Georgian et al., 2019) (Table 4).
8 Mechanism of Action in Diabetes Management
-
1.
Role in the Normalization of Insulin Production: The pharmacokinetic activity of Indian traditional plants, especially insulin production from pancreatic beta cells or similar insulin-related functions, is sourced from various literature surveys (Patel et al., 2012) (Table 5).
-
2.
Role in Insulin Resistance and Associated Metabolic Disorders: Noninsulin-dependent diabetes mellitus (NIDDM) or insulin resistance can lead to carbohydrate, protein, and fat metabolic derangement or syndrome, which is at the forefront of today’s research (Wilcox, 2005). Therapeutic drugs alone challenge to manage this syndrome. There has been a long history of traditional plants used in diabetic medicine, evident in Ayurveda, Unani, and Siddha medical systems (Ozturk, 2018). However, the mechanism of action in various parts of the medicinal plant will interest to know the importance of these plants, which mentioning bellows.
8.1 Terminalia arjuna (Roxb.)
The common name of Terminalia arjuna (Roxb.) is arjuna. It is used traditionally in various medical applications, such as for anti-microbial purposes, human immunodeficiency virus (HIV) treatment, bone fracture treatment, hypolipidaemic drugs, anti-inflammatory, cardio protection, antioxidant, and diabetes treatment. This plant is 60–80 ft in height and is distributed in India, Myanmar, Sri Lanka, and Mauritius. Tannins, polyphenols, flavonoids, saponins, triterpenoids, sterols, and mineral constituents are just some phytochemicals found in arjuna in high concentrations. The active parts of this plant are stem bark and root bark (Amalraj & Gopi, 2016). In normal conditions, insulin decreases carbohydrate catabolism and promotes an anabolic pathway. Arjuna bark extract has similar functions in the condition of insulin limitation. These are enhanced hexokinase, glucokinase, and phosphofructokinase enzyme activity. Hexokinase presents all cells and involves the phosphorylation of glucose to glucose 6-phosphates. On the other hand, it lowers the activity of gluconeogenic enzymes, viz. glucose-6-phosphatase and fructose-1, 6-diphosphatase, in kidney and liver cells. This may indicate insulin secretion by secretagogue in bark extract; otherwise, enzyme controlling function cannot proceed (Ragavan & Krishnakumari, 2006).
The medicinal use of arjuna has evidence of documentation from Charaka to his decedent Chakradatta, Bhavamishra, and present-day Ayurveda practitioners. Medication with T. arjuna preparation may be effective in CVD. It has been suggested that the soluble fibre, sitostanol content, and flavonoid antioxidant found in this plant are responsible for its hypocholesterolemic effects. It reduces platelet aggregation by inhibiting ca2+ release and CD62P gene expression in platelets (Maulik & Talwar, 2012). Applying bark extract on streptozotocin (STZ) induced diabetic rats in 8 weeks positively impacted left ventricular pressure, reducing inflammatory cytokines, oxidative stress, blood lipid profile, and myocardial injuries. In conclusion, it reduces diabetic-associated cardiopathy, vascular thrombosis, and inflammation (Amalraj & Gopi, 2016).
8.2 Aloe vera
Bioactive AIII and prototinosaponin AIII compounds increase glucose utilization through liver gluconeogenesis (Bnouham et al., 2006).
8.3 Allium cepa
Allium cepa is commonly known as onion, and it benefits patients with type-2 diabetes mellitus. Insulin insensitivity is a consequence of type-2 diabetes mellitus and may also cause hypoglycemia as a result of the exhaustion of pancreatic beta cells. During the last 10 years, research focuses on the pathogenic role of type-2 DM and insulin resistance due to lipid toxicity and low-grade inflammation. Because a higher number of free fatty acids may directly activate macrophage cells to release pro-inflammatory cytokines like TNF-α, IL-1β and IL6. Excessive stimulation of inflammatory cytokines leads to direct insulin resistance in muscle cells’ insulin receptors. In addition, an increase in free fatty acid may deposit in the liver cell, causing fatty liver, free radicals induce oxidative stress, and activation of stress induce signalling pathway making storage glycogen breakdown and hyper-glycemia. Onion peel extract may ameliorate hyperglycaemia and insulin resistance by increasing glucose uptake in the peripheral tissue by stimulating GLUT-4 and INSR gene expression in muscle tissues. Onion peel extract is highly evidenced in quercetin in the dry outer part, which increases insulin sensitization capabilities as well as lowered plasma free fatty, suppression of inflammatory cytokines and oxidative injury of liver cells (Jung et al., 2011; Noor et al., 2013; Ozougwu, 2011).
8.4 Aegle marmelos
Aegle marmelos, whose common name is bael, is an Indian traditional medicinal plant. Its leaf and fruit have been used for treating diabetes since ancient times. In recent times, advanced research established that the ingestion of bael fruit extracts heals the beta cells of the pancreatic gland and causes insulin sensitivity, gives antioxidant effects and lowers the risk of diabetic-associated hyperlipidemia. In type-2 DM, the insulin resistance triggers are higher free fatty acid (as a source of fatty meal diet), inflammatory cytokines like IL-6, TNF-β, oxidative stress induce beta-cell destruction, down-regulation insulin receptors, etc. For compensatory mechanisms, insulin over-secretion and reduced insulin extraction by the liver lead to hyperinsulinemia, which indicates beta-cell abnormality and hyperglycemia. Recent research suggests that Aegle marmelos fruit extracts increase insulin sensitivity through the up-regulation of the peroxisome proliferator-activated receptor-γ expression (PPARγ) and phosphoinositide 3-kinase (PI3 kinase) and increase the tyrosine phosphorylation of insulin receptors. As a result, the amount of glucose taken up by muscle, liver, and peripheral tissues increases. Dyslipidemia is a complication of insulin resistance and is evidence of increased free fatty acid flow, total cholesterol, total triglyceride, LDL-c, and lower HDL-c. The risk factor analysis suggests a high-fat diet and insulin resistance. Because insulin resistance increases 3-hydroxy-3-methylglutaryl (HMG), coenzyme A (CoA) re-educates enzyme synthesis and inhibits lipoprotein lipase (LPL) enzyme activity so that a higher amount of free fatty acid mobilized from peripheral tissue deport. Normally, insulin promotes lipogenesis and inhibits lipolysis. So the possible mechanisms of Aegle marmelos fruit extracts are for increasing liver triglyceride (TG) synthesis, activating LPL enzymes for the clearance of TG and free fatty acids from the blood vessels to the peripheral organs and tissues, and decreasing dietary cholesterol absorption and synthesis. These are the anti-dyslipidemia effects of bael fruit extracts. The antioxidant enzyme activity of superoxide dismutase (SOD) is also increased by bael fruit extracts, which helps prevent lipid peroxidation by interfering with the formation of superoxides and hydroperoxides. And finally, bael fruit extracts protect pancreas integrity and prevent beta-cell destruction due to their antioxidant nature (Sharma et al., 2011; Sabu & Kuttan, 2004).
8.5 Azadirachta indica (Neem)
Insulin resistance is synonymous with metabolic syndrome and includes the collective effects of obesity, insulin resistance, hypertension, high blood cholesterol, triglyceride, etc. Neem in parts of leaf, bark, stem, and flower, are very effective in high blood pressure, insulin resistance, antioxidants, dyslipidemia, etc. For blood pressure lowering, several possible mechanisms demonstrate that:
-
1.
Leave extract of neem up to regulate gene expression of the extracellular signal-regulated kinase (ERK 1 and 2) in the smooth endothelial muscle of blood vessels and blocking of ca2+ channel for smooth muscle contraction (Shah et al., 2014; Omóbòwálé et al., 2018),
-
2.
Gene expression of nuclear factor erythroid 2–related factor 2 (Nrf2) as a transcription factor for synthesis of anti-oxidant enzymes that prevent high blood pressure (Howden, 2013).
-
3.
Elevation of nitric oxide (vasodilator) (Omóbòwálé et al., 2019).
The fact of neem leaf extract in lipid-lowering describes that prevent free radical induce (ROS) oxidation of cholesterol, TG, fatty acid, and phospholipid otherwise oxidation leads to raise bad cholesterol level and reduce HDL-c. This decreases LDL-c, TG, and phospholipids and increases high-density lipoprotein (HDL) levels (Zuraini et al., 2006; Peer et al., 2008). The stem bark and root extract of neem applied for the control of obesity inhibit pancreatic lipase and α-glycosidase, which are responsible for weight gain (Mukherjee & Sengupta, 2013). Neem powder is used as a dietary supplement in the treatment of type-2 diabetes mellitus. It better controls blood glucose and HbA1c in patients ages 18–70 years. This leaf extract reduces glucose production during starch digestion by inactivating lingual α-amylase and intestinal glycosidase and induces the glycolysis pathway by activating hexokinase (Tadera et al., 2006). This is related to controlling high blood glucose during insulin resistance. Neem is used to prepare poly-herbal formulas (PHFs) and treat DM. This extract also up-regulates the GLUT-4 gene in muscle cells, increasing glucose uptake from blood vessels and maintaining blood glucose homeostasis (Yarmohammadi et al., 2021).
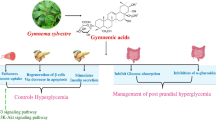
8.6 Gymnema sylvestre
The leaf of Gymnema sylvestre is an Indian traditional medicinal plant mentioned in the Sushruta (600 BC) and is now also recommended by many Ayurveda practitioners. At that time, this plant belonged to the ‘sala saradi’ group and was preferred for type-2 DM treatment with dietary restriction. This plant is also known as a sugar destroyer. The anti-diabetic role of the leaf extract is active compound gymnemic acid which has anti-hyperinsulinemia, insulin secretion, regeneration or repair of pancreatic β cells, and liver and kidney cell protection. The suggested mechanisms are as follows:
-
1.
Managing high blood sugar during hyperinsulinemia through liver glycogen synthesis by activating the glycogen synthetase enzyme and inhibiting glucose 6-phosphatase, which converts glucose 6-phosphate to glucose; it decreases the activity of other insulin-independent enzymes, viz. glycogen phosphorylase, gluconeogenic enzymes, fructose 1,6- diphosphatase, and sorbitol dehydrogenase, by which glucose release in to blood vessels is lower (Baskaran et al., 1990; Khan et al., 2019).
-
2.
Reducing glycosylated haemoglobin (HbA1c).
-
3.
Preventing glucose absorption by the small intestine, thus lowering fasting sugar in the blood; in lowering glucose absorption, the gymnemic acid matches structurally with a glucose molecule, which is why it blocks glucose-absorbing receptors in the small intestine (Khan et al., 2019).
-
4.
Reducing high lipid profile, such as LDL-c, VLDL-c, TG, and HDL-c, and the secondary development of atherosclerosis risks; this is due to the presence of acidic polyphenolic substances such as flavonoids, saponins, tannins, etc. (Khan et al., 2019).
8.7 Momordica charantia
Momordica charantia is commonly known as bitter gourd, karela, etc. It plays an important role in anti-hyperglycaemic and hypoglycaemic conditions and diabetic complications. It has bioactive compounds in fruits derived P insulin, charantin, and seed-derived vicine. Charantin is a mixture of two steroidal sitosteryl glucoside and stigmasteryl glucoside which have significant hypoglycaemic effects. The oral dose of 50 mg/kg charantin has 42% lower blood glucose. Bitter gourd fruits extract vital anti-diabetic functions in a number of ways:
-
1.
It reduces Na+- and K+-dependent glucose absorption in the intestine.
-
2.
Karela juice has clinical insulin-mimetic functions, such as enhancing glucose uptake in muscle cells and in the liver by increasing glycogenesis.
-
3.
Karela seed protein extracts stimulate lipogenesis or inhibit lipolysis, neoglucogenesis, etc.
-
4.
Hepatic and red blood cell glucose-6-phosphate dehydrogenase (G6PD-6-PDH) activity utilized excess glucose in hexose mono phosphate shunt pathway (HMP) that is the role of hypoglycaemic. It inhibits the activities of liver fructose 1, 6-diphosphatase and glucose-6-phosphatase that prevent free glucose release into blood stream i.e., anti-hypoglycaemic role.
-
5.
Insulin sensitization through AMP-activated protein kinase phosphorylation in the target receptors may affect karela juice consumption. (Joseph & Jini, 2013; Raman & Lau, 1996).
8.8 Syzygium cumini
Both the seeds and bark of the Jamun stem show promise as an anti-diabetic medicine in humans. The various parts (leaves, bark, fruits, and seeds) of Syzygium cumini are traditionally used for diabetes treatment. It may reduce blood glucose by inhibiting α-glycosidase for di- and oligosaccharide digestion and inhibitory effects on pancreatic amylase. The activities of plant seeds control blood sugar through anti-hyperglycaemic and hypoglycaemic, where increased glycogen synthesis in liver and muscle cells and inactivation of hepatic gluconeogenesis (glucose-6-phosphatase) is called anti-hyperglycaemic and increased activity of key glycolytic enzymes (hexokinase) called hypoglycemia (Zulcafli et al., 2020). Mycaminose component shows the anti-hyperglycaemic effect of seeds. Oral administration of seed powder continues for 3 months, effectively managing various diabetic symptoms such as polyurea, polyphagia, weakness and weight loss. This seed extract increases cellular glucose uptake by up-regulating gene expression of GLUT4 and protecting β-cell dysfunction. The pathogenesis of β-cell dysfunction is due to insulin resistance inducing inflammatory cytokines such TNF-α, IL-6, etc. Apart from glucose homeostasis, this seed extract possibly reduces dyslipidemia through the gene expression of peroxisome proliferator-activated receptor alpha (PPARα) and peroxisome proliferator-activated receptor gamma (PPAR γ) activities in the liver. That will affect insulin sensitization through increased mobilization of free fatty acid and storage. Endogenous cholesterol synthesis is inhibited by inactive HMG CoA reductase and decreased intestinal fat absorption by the function of seed extract (Baliga et al., 2011; Grover et al., 2002).
8.9 Ficus benghalensis
During diabetes, insulin resistance causes changes in the metabolic enzymes found in the liver. These changes can include glycogen synthase, glucokinase, lactate dehydrogenase (LDH), succinate dehydrogenase, and malate dehydrogenase. That leads to impaired glycogenesis, tricarboxylic acid (TCA) cycle, etc., which may be alleviated by bark extract. This suggests that gene expression of enzyme protein in response to the released insulin and utilizes blood glucose (Gayathri & Kannabiran, 2008). Because of its amylase inhibitory action during the digestion of starch, the pelargonidin derivative glycoside that was isolated from the bark extract has the potential to lower fasting blood glucose levels. Leucocyanidin derivative, which was isolated from the bark of F. benghalensis, has hypoglycaemic properties with a dose of 100 mg/kg in normal, which is why it is better to manage diabetes as combination therapy with insulin, and even lower the requirement of insulin therapy (Grover et al., 2002).
8.10 Trigonella foenum-graecum
Fenugreek seeds are similarly medicinal plants good for diabetic management and associated chronic illnesses. With regard to blood lipid control, methi seed extracts could reduce LDLL-c, VLDL-c, and TG and increase HDL-c. This is possible due to the crude fibres and saponin in methi seeds as these components may increase faecal bile and cholesterol excretion (Geberemeskel et al., 2019). Fenugreek seeds manage blood glucose and lipid profile in several ways:
-
1.
They increase glucose uptake through the gene expression of the GLUT-2 transporter.
-
2.
They enhance the messenger RNA (mRNA) transcription of CCAAT/enhancer-binding protein (C/EBPδ) and peroxisome proliferator-activated receptor-γ (PPAR-γ) protein. These protein receptors are located in the liver, adipocyte, intestines, lungs, kidneys, and myeloid cells. Due to the activation of C/EBP and PPAR-γ family receptors in adipose tissue and liver, increased lipogenesis and excess FFA, TG and cholesterol will mobilize.
-
3.
They activate C/EBPα-mediated glycogen synthetase enzymes, which promote glycogenesis (Ramji & Foka, 2002; Baset et al., 2020).
-
4.
Galactomannan, a carbohydrate-containing bioactive substances (45–60%), reduces postprandial blood glucose by blocking α-amylase and pancreatic lipase.
-
5.
An oral dose of fenugreek seeds may reduce renal complications by protecting the glomerular basement membrane and lowering the creatinine level. It also reduces TG and increases HDL-c levels. Fenugreek seeds have an antioxidant potential; the legumes raise the level of glutathione peroxidase and catalase, preventing lipid peroxidation (Baset et al., 2020).
8.11 Coccinia indica
It has been used in Indian traditional medicine from Ayurveda and the Unani systems. Beta-sitosterol, an active compound in leaves and pectins in fruits, has hypoglycaemic effects. Pectins hypoglycaemic mechanism in normal conditions is activated through glycogen synthetase and inactivation of glycogen phosphorylase. Leaf-containing beta-sitosterol deactivates liver fructose 1, 6 bisphosphatase, glucose 6-phosphatase, and LDH and increases lipoprotein lipase activity which is anti-hyperglycemic anti-cholesterolemic effect. Oral administration of leaf extract may reduce fasting sugar due to halting starch-breaking enzymes, insulin secretion, etc. (Grover et al., 2002; Balaraman et al., 2010) (Fig. 2).
9 Future Prospects of Indian Medicinal Plants in Diabetes
It is estimated that approximately 33 million adults have been suffering from diabetes mellitus in recent years in India, and it is likely to increase by 57.2 million by 2025 (Manukumar et al., 2016). Plant-derived active ingredients are applied to patients, along with some medications, to treat diabetes mellitus. But these active principles are practically extractable with different solvent phases, so it is essential to isolate individual phyto-components to confirm their activity on diabetes mellitus. Therefore, more research is needed. Several medicinal plants play an important role in treating diabetes mellitus and its associated complications, but herbal formulations’ major drawback is that these active ingredients are not well defined. Their molecular interactions and mode of action should be more carefully investigated for future novel drug discoveries. Recently, more initiatives have been undertaken to fight diabetes mellitus. Abdulazeez, in the year 2013, remarked that the cells that produce insulin might be derived from stem cells, and he is confident that shortly scientists may come to light with solutions for the use of stem cells as therapeutic agents to cure diabetes (Abdulazeez, 2013). Pancreas and cell of islet transplantation is currently an outstanding achievement at the University of Illinois Chicago (UIC).
10 Conclusion
In this chapter, we discussed the Indian traditional medicinal plants for the treatment of diabetes. There are so many Indian traditional plants with anti-diabetic roles that have been reported. The effect is attributed to the plant phytochemicals, single compounds as well as extracts. The main plant phytochemicals responsible for anti-diabetic activity are alkaloids, phenolic acids, flavonoids, glycosides, etc. Identification of anti-diabetic properties on Indian traditional ethnographic plants is remarkable progress for human welfare and evidence-based short-term and long-term implication has also been integrated in past, present and future also. To see the observable effects, multiple in vitro and in vivo experiments have been conducted. Various mechanisms are described for the anti-diabetes role of medicinal plant parts, explaining the beneficial effects of phytochemicals, such as their role in the normalization of Insulin production, Role in insulin resistance and associated metabolic disorders, regulation of glucose and lipid metabolism, stimulating β cells and ROS protective action. However, still many aspects such as a mark popularization over pharmaceutical drugs, study at the genomic level to examine the role of plant secondary metabolites in diabetes, limited availability of toxicological safety data, etc. remain to be aspirated areas. Only a few medicinal plants have been studied for efficacy in humans. Thus, more efficient clinical studies are warranted for further validation.
References
Abdulazeez, S. S. (2013). Diabetes treatment: A rapid review of the current and future scope of stem cell research. Saudi Pharmaceutical Journal, 613(4), 1–8. https://doi.org/10.1016/j.jsps.2013.12.012
Acharya, C. K., Madhu, N. R., Khan, N. S., & Guha, P. (2021). Improved reproductive efficacy of Phyllanthus emblica L. (Gaertn.) on testis of male Swiss mice and a pilot study of its potential values. International Journal of Food Sciences and Nutrition, 10(4), 7–14.
Agrawal, R., Sethiya, N. K., & Mishra, S. H. (2013). Antidiabetic activity of alkaloids of Aerva lanata roots on streptozotocin-nicotinamide induced type-II diabetes in rats. Pharmaceutical Biology, 51(5), 635–642. https://doi.org/10.3109/13880209.2012.761244
Ahlem, S., Khaled, H., Wafa, M., Sofiane, B., Mohamed, D., Jean-Claude, M., & Abdelfattah el, F. (2009). Oral administration of Eucalyptus globulus extract reduces the alloxan-induced oxidative stress in rats. Chemico-Biological Interactions, 181(1), 71–76. https://doi.org/10.1016/j.cbi.2009.06.006
Alam, S., Sarker, M. M. R., Sultana, T. N., Chowdhury, M. N. R., Rashid, M. A., Chaity, N. I., Zhao, C., Xiao, J., Hafez, E. E., Khan, S. A., & Mohamed, I. N. (2022). Antidiabetic phytochemicals from medicinal plants: Prospective candidates for new drug discovery and development. Frontiers in Endocrinology, 13, 800714. https://doi.org/10.3389/fendo.2022.800714
Al-Shaqha, W. M., Khan, M., Salam, N., Azzi, A., & Chaudhary, A. A. (2015). Anti-diabetic potential of Catharanthus roseus Linn. and its effect on the glucose transport gene (GLUT-2 and GLUT-4) in streptozotocin induced diabetic Wistar rats. BMC Complementary and Alternative Medicine, 15(1), 379.
Amalraj, A., & Gopi, S. (2016). Medicinal properties of Terminalia arjuna (Roxb.) wight & amp; Arn.: A review. Journal of Traditional and Complementary Medicine, 7(1), 65–78.
Ananda, P. K., Kumarappan, C. T., Christudas, S., & Kalaichelvan, V. K. (2012). Effect of biophytum sensitivum on streptozotocin and nicotinamide-induced diabetic rats. Asian Pacific Journal of Tropical Biomedicine, 2(1), 31–35.
Atal, S., Agrawal, R. P., Vyas, S., Phadnis, P., & Rai, N. (2012). Evaluation of the effect of piperine on blood glucose level in alloxan-induced diabetic mice. Acta Poloniae Pharmaceutica, 69(5), 965–969.
Atanas, G., Waltenberger, A. B., Pferschy-Wenzig, E. M., Linder, T., Wawrosch, C., Uhrin, P., Temml, V., Wang, L., Schwaiger, S., Heiss, E. H., Rollinger, J. M., Schuster, D., Breuss, J. M., Bochkov, V., Mihovilovic, M. D., Kopp, B., Bauer, R., Dirsch, V. M., & Stuppner, H. (2015). Discovery and resupply of pharmacologically active plant-derived natural products: A review. Biotechnology Advances, 33, 1582–1614.
Balaraman, A. K., Singh, J., Dash, S., & Maity, T. K. (2010). Antihyperglycemic and hypolipidemic effects of Melothria maderaspatana and Coccinia indica in Streptozotocin induced diabetes in rats. Saudi Pharmaceutical Journal, 18(3), 173–178. https://doi.org/10.1016/j.jsps.2010.05.009
Baliga, M. S., Bhat, H. P., Baliga, B. R. V., Wilson, R., & Palatty, P. L. (2011). Phytochemistry, traditional uses and pharmacology of Eugenia jambolana lam. (black plum): A review. Food Research International, 44, 1776–1789.
Barghamdi, B., Ghorat, F., Asadollahi, K., Sayehmiri, K., Peyghambari, R., & Abangah, G. (2016). Therapeutic effects of Citrullus colocynthis fruit in patients with type II diabetes: A clinical trial study. Journal of Pharmacy & Bioallied Sciences, 8(2), 130–134.
Baset, M. E., Ali, T. I., Elshamy, H., El-Sadek, A. M., Sami, D. G., Badawy, M. T., & Abdellatif. (2020). Anti-diabetic effects of fenugreek (Trigonella foenum-graecum): A comparison between oral and intraperitoneal administration—an animal study. International Journal of Functional Nutrition, 1, 2. https://doi.org/10.3892/ijfn.2020.2
Baskaran, K., Ahamath, B. K., Shanmugasundaram, K. R., & Shanmugasundaram, E. R. B. (1990). Antidiabetic effect of a leaf extract from Gymnema sylvestre in non-insulin-dependent diabetes mellitus patients. Journal of Ethnopharmacology, 30(3), 295–305.
Bhushan, M. S., Rao, C. H. V., Ojha, S. K., Vijayakumar, M., & Verma, A. (2010). An analytical review of plants for anti-diabetic activity with their phytoconstituent & mechanism of action. International Journal of Pharmaceutical Sciences and Research, 1(1), 29–46.
Bhuyan, D. J., & Basu, A. (2017). Phenolic compounds, potential health benefits and toxicity. In Q. V. Vuong (Ed.), Utilisation of bioactive compounds from agricultural and food waste (pp. 27–59). CRC Press.
Bnouham, M., Ziyyat, A., Mekhfi, H., Tahri, A., & Legssyer, A. (2006). Medicinal plants with potential antidiabetic activity-a review of ten years of herbal medicine research (1990–2000). International Dubai Diabetes and Endocrinology Journal, 14(1), 1–25.
Cartea, M. E., Francisco, M., Soengas, P., & Velasco, P. (2011). Phenolic compounds in brassica vegetables. Molecules, 16(1), 251–280.
Costello, R. A., & Shivkumar, A. S. (2021). Stat Pearls [Internet]. Accessed June 6, 2022, from https://www.ncbi.nlm.nih.gov/books/NBK513225/
Cox-Georgian, D., Ramadoss, N., Dona, C., & Basu, C. (2019). Therapeutic and medicinal uses of terpenes.
Diabetes-Australia Insulin. (2022). Accessed June 6, 2022, from https://www.diabetesaustralia.com.au/living-with-diabetes/medicine/insulin/
Eliza, J., Daisy, P., Ignacimuthu, S., & Duraipandiyan, V. (2009). Antidiabetic and antilipidemic effect of eremanthin from Costus speciosus (Koen.) Sm., in STZ-induced diabetic rats. Chemico-Biological Interactions, 182(1), 67–72.
Eno, A. E., Ofem, O. E., Nku, C. O., Ani, E. J., & Itam, E. H. (2008). Stimulation of insulin secretion by Viscum album (mistletoe) leaf extract in streptozotocin-induced diabetic rats. African Journal of Medicine and Medical Sciences, 37(2), 141–147.
Francesca, G. M. R., Daniela, E. G. F., & Vivian, M. C. (2019). Secondary metabolites in plants: Main classes, phytochemical analysis and pharmacological activities. Bionatura, 4(4), 1000–1009. https://doi.org/10.21931/RB/2019.04.04.11
Ganogpichayagrai, A., Palanuvej, C., & Ruangrungsi, N. (2017). Antidiabetic and anticancer activities of Mangifera indica cv Okrong leaves. Journal of Advanced Pharmaceutical Technology & Research, 8(1), 19–24.
Gayathri, M., & Kannabiran, K. (2008). Antidiabetic and ameliorative potential of Ficus bengalensis bark extract in streptozotocin induced diabetic rats. Indian Journal of Clinical Biochemistry, 23(4), 394–400. https://doi.org/10.1007/s12291-008-0087-2
Geberemeskel, G. A., Debebe, Y. G., & Nguse, N. A. (2019). Antidiabetic effect of fenugreek seed powder solution (Trigonella foenum graecum L.) on hyperlipidemia in diabetic patients. Journal of Diabetes Research, 2019, 1–8. https://doi.org/10.1155/2019/8507453
Grover, J. K., Yadav, S., & Vats, V. (2002). Medicinal plants of India with anti-diabetic potential. Journal of Ethnopharmacology, 81(1), 81–100.
Guang-Kai, X. U., Xiao-Ying, Q. I., Guo-Kai, W. A., Guo-Yong, X. I., Xu-Sen, L. I., Chen-Yu, S. U., Bao-Lin, L. I., & Min-Jian, Q. I. (2017). Antihyperglycemic, antihyperlipidemic and antioxidant effects of standard ethanol extract of Bombax ceiba leaves in high-fat-diet-and streptozotocin-induced type 2 diabetic rats. Chinese Journal of Natural Medicines, 15(3), 168–177.
Gupta, R. K., Kesari, A. N., Diwakar, S., Tyagi, A., Tandon, V., Chandra, R., & Wata, G. (2008). In vivo evaluation of anti-oxidant and anti-lipidemic potential of Annona squamosa aqueous extract in type 2 diabetic models. Journal of Ethnopharmacology, 118(1), 21–25.
Harborne, J. B. (1989). General procedures and measurement of total phenolics. In J. B. Harborne (Ed.), Methods in plant biochemistry (Plant phenolics) (Vol. 1, pp. 1–28). Academic Press.
Hegazy, G. A., Alnoury, A. M., & Gad, H. G. (2013). The role of Acacia arabica extract as an antidiabetic, antihyperlipidemic, and antioxidant in streptozotocin-induced diabetic rats. Saudi Medical Journal, 34(7), 727–733.
Ho, C. T. (1992). Phenolic compounds in food. In C. T. Ho, C. Y. Lee, & M. T. Hung (Eds.), Phenolic compounds in food and their effects on health (pp. 2–7).
Hou, S. Z., Chen, S. X., Huang, S., Jiang, D. X., Zhou, C. J., Chen, C. Q., Liang, Y. M., & Lai, X. P. (2011). The hypoglycemic activity of Lithocarpus polystachyus Rehd. Leaves in the experimental hyperglycemic rats. Journal of Ethnopharmacology, 138(1), 142–149.
Howden, R. (2013). Nrf2 and cardiovascular defense. Oxidative Medicine and Cellular Longevity, 2013, 104308.
Huang, C. S., Yin, M. C., & Chiu, L. C. (2011). Antihyperglycemic and antioxidative potential of Psidium guajava fruit in streptozotocin-induced diabetic rats. Food and Chemical Toxicology, 49(9), 2189–2195.
Huseini, H. F., Kianbakht, S., Hajiaghaee, R., & Dabaghian, F. H. (2012). Anti-hyperglycemic and anti-hypercholesterolemic effects of Aloe vera leaf gel in hyperlipidemic type 2 diabetic patients: A randomized double-blind placebo-controlled clinical trial. Planta Medica, 78(04), 311–316.
Islam, M. S., & Choi, H. (2008). Dietary red chilli (Capsicum frutescens L.) is insulinotropic rather than hypoglycemic in type 2 diabetes model of rats. Phytotherapy Research, 22(8), 1025–1029.
Jacob, B., & Narendhirakannan, R. T. (2019). Role of medicinal plants in the management of diabetes mellitus: A review. Biotech, 9(1), 4. https://doi.org/10.1007/s13205-018-1528-0
Jiao, Y., Wang, X., Jiang, X., Kong, F., Wang, S., & Yan, C. (2017). Antidiabetic effects of Morus alba fruit polysaccharides on high-fat diet- and streptozotocin-induced type 2 diabetes in rats. Journal of Ethnopharmacology, 199, 119–127.
Joseph, B., & Jini, D. (2013). Antidiabetic effects of Momordica charantia (bitter melon) and its medicinal potency. Asian Pacific Journal of Tropical Disease, 3(2), 93–102.
Jung, J. Y., Lim, Y., & Moon, M. S. (2011). Onion peel extracts ameliorate hyperglycemia and insulin resistance in high fat diet/streptozotocin-induced diabetic rats. Nutrition & Metabolism, 8(1), 18. https://doi.org/10.1186/1743-7075-8-18
Kabir, A. U., Samad, M. B., Ahmed, A., Jahan, M. R., Akhter, F., Tasnim, J., Hasan, S. N., Sayfe, S. S., & Hannan, J. M. (2015). Aqueous fraction of Beta vulgaris ameliorates hyperglycemia in diabetic mice due to enhanced glucose stimulated insulin secretion, mediated by acetylcholine and GLP-1, and elevated glucose uptake via increased membrane bound GLUT4 transporters. PLoS One, 10(2), e0116546.
Kaur, R., & Arora, S. (2015). Alkaloids-important therapeutic secondary metabolites of plant origin. Journal of Critical Reviews, 2(3), 1–8.
Khan, F., Sarker, M. M. R., Ming, L. C., Mohamed, I. N., Zhao, C., Sheikh, B. Y., Tsong, H. F., & Rashid, M. A. (2019). Comprehensive review on phytochemicals, pharmacological and clinical potentials of Gymnema sylvestre. Frontiers in Pharmacology, 10, 1223. https://doi.org/10.3389/fphar.2019.01223
Kumagai, Y., Nakatani, S., Onodera, H., Nagatomo, A., Nishida, N., Matsuura, Y., Kobata, K., & Wada, M. (2015). Anti-glycation effects of pomegranate (Punica granatum L.) fruit extract and its components in vivo and in vitro. Journal of Agricultural and Food Chemistry, 63(35), 7760–7764.
Kumar, S., Kumar, V., & Prakash, O. (2011). Antidiabetic, hypolipidemic and histopathological analysis of Dillenia indica (L.) leaves extract on alloxan induced diabetic rats. Asian Pacific Journal of Tropical Medicine, 4(5), 347–352. https://doi.org/10.1016/S1995-7645(11)60101-6
Kurup, S. B., & Mini, S. (2017). Averrhoa bilimbi fruits attenuate hyperglycemia-mediated oxidative stress in streptozotocin-induced diabetic rats. Journal of Food and Drug Analysis, 25(2), 360–368.
Laha, S., & Paul, S. (2019). Gymnema sylvestre (Gurmar): A potent herb with anti-diabetic and antioxidant potential. The Pharmacogenomics Journal, 11(2), 201–206.
Lattanzio, V. (2013). Phenolic compounds: Introduction. In K. G. Ramawat & J. M. Merillon (Eds.), Natural products. https://doi.org/10.1007/978-3-642-22144-6_57
Li, P. B., Lin, W. L., Wang, Y. G., Peng, W., Cai, X. Y., & Su, W. W. (2012). Antidiabetic activities of oligosaccharides of Ophiopogonis japonicas in experimental type 2 diabetic rats. International Journal of Biological Macromolecules, 51(5), 749–755.
Liu, S., Li, D., Huang, B., Chen, Y., Lu, X., & Wang, Y. (2013). Inhibition of pancreatic lipase, α-glucosidase, α-amylase, and hypolipidemic effects of the total flavonoids from Nelumbo nucifera leaves. Journal of Ethnopharmacology, 149(1), 263–269.
Malviya, N., Jain, S., & Malviya, S. (2010). Antidiabetic potential of medicinal plants. Acta Poloniae Pharmaceutica—Drug Research, 67(2), 113–118.
Manukumar, H. M., Shiva Kumar, J., Chandrashekar, B., Raghava, S., & Umesha, S. (2016). Evidences for diabetes and insulin mimetic activity of medicinal plants: Present status and future prospects. Critical Reviews in Food Science and Nutrition, 57, 2712. https://doi.org/10.1080/10408398.2016.1143446
Mathew, L., & Babu, S. (2011). Phytotherapy in India: Transition of tradition to technology. Current Botany, 2(5), 1722.
Maulik, S. K., & Talwar, K. K. (2012). Therapeutic potential of Terminalia Arjuna in cardiovascular disorders. American Journal of Cardiovascular Drugs, 12, 157–163.
McCalla, G., Parshad, O., Brown, P. D., & Gardner, M. T. (2015). Beta cell regenerating potential of Azadirachta indica (neem) extract in diabetic rats. The West Indian Medical Journal, 65(1), 13–17. https://doi.org/10.7727/wimj.2014.224
Meliani, N., Dib, M. E. A., Allali, H., & Tabti, B. (2011). Hypoglycaemic effect of Berberis vulgaris L. in normal and streptozotocin-induced diabetic rats. Asian Pacific Journal of Tropical Biomedicine, 1(6), 468–471.
Mishra, N., Kumar, D., & Rizvi, S. I. (2016). Protective effect of Abelmoschus esculentus against alloxan-induced diabetes in Wistar strain rats. Journal of Dietary Supplements, 13(6), 634–646.
Modak, M., Dixit, P., Londhe, J., Ghaskadbi, S., Paul, A., & Devasagayam, T. (2007). Indian herbs and herbal drugs used for the treatment of diabetes. Journal of Clinical Biochemistry and Nutrition, 40(3), 163–173.
Mohanraj, R., & Sivasankar, S. (2014). Sweet potato (Ipomoea batatas [L.] lam)—A valuable medicinal food: A review. Journal of Medicinal Food, 17(7), 733–741.
Mukherjee, P. K., Rai, S., Kumar, V., Mukherjee, K., Hylands, P. J., & Hider, R. C. (2007). Plants of Indian origin in drug discovery. Expert Opinion on Drug Discovery, 2(5), 633–657.
Mukherjee, A., & Sengupta, S. (2013). Indian medicinal plants known to contain intestinal glucosidase inhibitors also inhibit pancreatic lipase activity-an ideal situation for obesity control by herbal drugs. Indian Journal of Biotechnology, 12(1), 32–39.
Ngugi, M. P., Njagi, J. M., Kibiti, C. M., & Miriti, P. M. (2012). Pharmacological Management of Diabetes Mellitus. Asian Journal of Biochemical and Pharmaceutical Research, 1(2), 375–381.
Noor, A., Bansal, V. S., & Vijayalakshmi, M. A. (2013). Current update on anti-diabetic biomolecules from key traditional Indian medicinal plants. Current Science, 104(6), 721–727.
Ojewole, J. A. (2002). Hypoglycaemic effect of Clausena anisata (Willd) hook methanolic root extract in rats. Journal of Ethnopharmacology, 81(2), 231–237.
Omóbòwálé, T. O., Oyagbemi, A. A., Ogunpolu, B. S., Ola-Davies, O. E., Olukunle, J. O., & Asenuga, E. R. (2019). Antihypertensive effect of polyphenol-rich fraction of Azadirachtaindica on Nω-Nitro-L-arginine methyl ester-induced hypertension and cardiorenal dysfunction. Drug Research, 69, 12–22.
Omóbòwálé TO, Oyagbemi, A. A., Alaba, B. A., Ola-Davies, O. E., Adejumobi, O. A., & Asenuga, E. R. (2018). Ameliorative effect of Azadirachtaindica on sodium fluoride-induced hypertension through improvement of antioxidant defence system and upregulation of extracellular signal regulated kinase 1/2 signaling. Journal of Basic and Clinical Physiology and Pharmacology, 29, 155–164.
Ozougwu, J. C. (2011). Anti-diabetic effects of Allium cepa (onions) aqueous extracts on alloxan-induced diabetic Rattus novergicus. Journal of Medicinal Plants Research, 5(7), 1134–1139. http://www.academicjournals.org/JMPR
Ozturk, M. (2018). A comparative analysis of the medicinal plants used for diabetes mellitus in the traditional medicine in Turkey, Pakistan, and Malaysia. In M. Ozturk & K. Hakeem (Eds.), Plant and human health (Vol. 1). Springer. https://doi.org/10.1007/978-3-319-93997-1_11
Parasuraman, S., Thing, G. S., & Dhanaraj, S. A. (2014). Polyherbal formulation: Concept of ayurveda. Pharmacognosy Reviews, 8(16), 73–80.
Patel, D. K., Prasad, S. K., Kumar, R., & Hemalatha, S. (2012). An overview on antidiabetic medicinal plants having insulin mimetic property. Asian Pacific Journal of Tropical Biomedicine, 2(4), 320–330.
Peer, P. A., Trivedi, P. C., Nigade, P. B., Ghaisas, M. M., & Deshpande, A. D. (2008). Cardioprotective effect of Azadirachtaindica A Juss on isoprenaline induced myocardial infarction in rats. International Journal of Cardiology, 126, 123–126.
Ponnusamy, S., Ravindran, R., Zinjarde, S., Bhargava, S., & Kumar, A. R. (2011). Evaluation of traditional Indian antidiabetic medicinal plants for human pancreatic amylase inhibitory effect in vitro. Evidence-based Complementary and Alternative Medicine, 2011, 515647.
Poongothai, K., Ponmurugan, P., Ahmed, K. S., Kumar, B. S., & Sheriff, S. A. (2011). Antihyperglycemic and antioxidant effects of solanum xanthocarpum leaves (field grown & in vitro raised) extracts on alloxan induced diabetic rats. Asian Pacific Journal of Tropical Medicine, 4(10), 778–785.
Prince, P. S. M., & Menon, V. P. (2003). Hypoglycaemic and hypolipidaemic action of alcohol extract of Tinospora cordifolia roots in chemical induced diabetes in rats. Phytotherapy Research, 17(4), 410–413.
Quideau, S., Deffieux, D., Douat-Casassus, C., & Pouyse’gu L. (2011). Plant polyphenols: Chemical properties, biological activities, and synthesis. Angewandte Chemie, International Edition, 50(3), 586–621.
Ragavan, B., & Krishnakumari, S. (2006). Antidiabetic effect of T. arjuna bark extract in alloxan induced diabetic rats. Indian Journal of Clinical Biochemistry, 21(2), 123–128.
Raman, A., & Lau, C. (1996). Anti-diabetic properties and Phytochemistry Momordica charantia L. (Cucurbitaceae). Phytomedicine, 2(4), 349–362.
Ramji, D. P., & Foka, P. (2002). CCAAT/enhancer-binding proteins: Structure, function and regulation. The Biochemical Journal, 365(Pt-3), 561–575.
Sabu, M. C., & Kuttan, R. (2004). Antidiabetic activity of aegle marmelos and its relationship with its antioxidant properties. Indian Journal of Physiology and Pharmacology, 48(1), 81–88.
Satoh, T., Igarashi, M., Yamada, S., Takahashi, N., & Watanabe, K. (2015). Inhibitory effect of black tea and its combination with acarbose on small intestinal α-glucosidase activity. Journal of Ethnopharmacology, 161, 147–155.
Sayyed, F. J., & Wadkar, G. H. (2018). Studies on in-vitro hypoglycemic effects of root bark of Caesalpinia bonducella. Annales Pharmaceutiques Françaises, 76(1), 44–49.
Shah, A. J., Gilani, A. H., & Hanif, H. M. (2014). Neem (Azadirachta indica) lowers blood pressure through a combination of Ca+ channel blocking and endothelium-dependent muscarinic receptors activation. International Journal of Pharmacology, 10, 418–428.
Sharma, A. K., Bharti, S., Goyal, S., Arora, S., Nepal, S., Kishore, K., Joshi, S., Kumari, S., & Arya, D. S. (2011). Upregulation of PPARγ by Aegle marmelos ameliorates insulin resistance and β-cell dysfunction in high fat diet fed-Streptozotocin induced type 2 diabetic rats. Phytotherapy Research, 25, 1457–1465.
Sharma, B., Salunke, R., Balomajumder, C., Daniel, S., & Roy, P. (2010). Antidiabetic potential of alkaloid rich fraction from Capparis decidua on diabetic mice. Journal of Ethnopharmacology, 127(2), 457–462. https://doi.org/10.1016/j.jep.2009.10.013
Singh, L. W. (2011). Traditional medicinal plants of Manipur as anti-diabetics. Journal of Medicinal Plant Research: Planta Medica, 5(5), 677–687.
Singh, P. K., Baxi, D., & Doshi, A. V. R. (2011). Antihyperglycaemic and renoprotective effect of Boerhaavia diffusa L. in experimental diabetic rats. Journal of Complementary and Integrative Medicine, 8(1), 1–20. https://doi.org/10.2202/1553-3840.1533
Srivastava, S., Lal, V. K., & Pant, K. K. (2012). Polyherbal formulations based on Indian medicinal plants as antidiabetic phytotherapeutics. Phytopharmacology, 2, 115.
Stefkov, G., Miova, B., Dinevska-Kjovkarovska, S., Stanoeva, J. P., Stefova, M., Petrusevska, G., & Kulevanova, S. (2014). Chemical characterization of Centaurium erythrea L. and its effects on carbohydrate and lipid metabolism in experimental diabetes. Journal of Ethnopharmacology, 152(1), 71–77.
Stohs, S. J., & Ray, S. (2015). Anti-diabetic and anti-hyperlipidemic effects and safety of Salacia reticulata and related species. Phytotherapy Research, 29(7), 986–995. https://doi.org/10.1002/ptr.5382
Sundaram, R., Naresh, R., Shanthi, P., & Sachdanandam, P. (2012). Antihyperglycemic effect of iridoid glucoside, isolated from the leaves of Vitex negundo in streptozotocin-induced diabetic rats with special reference to glycoprotein components. Phytomedicine, 19(3–4), 211–216.
Sunil, V., Shree, N., Venkataranganna, M. V., Bhonde, R. R., & Majumdar, M. (2017). The anti-diabetic and antiobesity effect of Memecylon umbellatum extract in high fat diet induced obese mice. Biomedicine & Pharmacotherapy, 89, 880–886.
Tadera, K., Minami, Y., Takamatsu, K., & Matsuoka, T. (2006). Inhibition of α-glucosidase and α- amylase by flavonoids. Journal of Nutritional Science and Vitaminology, 52(2), 149–153.
Thompson, A. T., Opeolu, O. O., Peter, R. F., & Yasser, H. A. A. W. (2014). Aqueous bark extracts of Terminalia arjuna stimulates insulin release, enhances insulin action and inhibits starch digestion and protein glycation in vitro. Austin Journal of Endocrinology and Diabetes, 1(1), 1001.
Tiong, S. H., Looi, C. Y., Hazni, H., Arya, A., Paydar, M., & Wong, W. F. (2013). Antidiabetic and antioxidant properties of alkaloids from Catharanthus roseus (L.) G.Don. Molecules, 18(8), 9770–9784.
Uchegbu, N. N., & Ishiwu, C. N. (2016). Germinated pigeon pea (Cajanus cajan): A novel diet for lowering oxidative stress and hyperglycemia. Food Science & Nutrition, 4(5), 772–777.
Umamaheswari, S., Joseph, L. D., Srikanth, J., Lavanya, R., Chamundeeswari, D., & Reddy, C. U. (2010). Antidiabetic activity of a polyherbal formulation (DIABET). International Journal of Pharmacy and Pharmaceutical Sciences, 2, 1822.
Venkatachalam, T., Kumar, V. K., Selvi, P. K., Maske, A. O., Anbarasan, V., & Kumar, P. S. (2011). Antidiabetic activity of Lantana camara Linn fruits in normal and streptozotocin-induced diabetic rats. Journal of Pharmacy Research, 4(5), 1550–1552.
Venkatesh, S., Reddy, G. D., Reddy, Y. S. R., Sathyavathy, D., & Reddy, B. M. (2004). Effect of Helicteres isora root extracts on glucose tolerance in glucose-induced hyperglycemic rats. Fitoterapia, 75(3), 364–367.
Wainstein, J., Ganz, T., Boaz, M., Bar Dayan, Y., Dolev, E., Kerem, Z., & Madar, Z. (2012). Olive leaf extract as a hypoglycemic agent in both human diabetic subjects and in rats. Journal of Medicinal Food, 15(7), 605–610.
WHO. (2022a). Global report on diabetes, Accessed July 15, 2022, from https://www.who.int/publications/i/item/9789241565257
WHO. (2022b). Health topics on diabetics, Accessed July, 15, 2022, from https://www.who.int/health-topics/diabetes#tab=tab_1
Wilcox, G. (2005). Insulin and insulin resistance. Clinical biochemist Reviews, 26(2), 19–39.
Worldwide toll of diabetes. (2020). Diabetesatlas.org. Accessed August 7, 2020, from https://www.diabetesatlas.org/en/sections/worldwide-toll-of-diabetes.html
Yarmohammadi, F., Mehri, S., Najafi, N., Salar, A. S., & Hosseinzadeh, H. (2021). The protective effect of Azadirachta indica (neem) against metabolic syndrome: A review. Iranian Journal of Basic Medical Sciences, 24(3), 280–292.
Zulcafli, A. S., Lim, C., Ling, A. P., Chye, S., & Koh, R. (2020). Antidiabetic potential of Syzygium sp.: An overview. The Yale Journal of Biology and Medicine, 93(2), 307–325.
Zuraini, A., Vadiveloo, T., Hidayat, M. T., Arifah, A., Sulaiman, M., & Somchit, M. (2006). Effects of neem (Azadirachta indica) leaf extracts on lipid and C-reactive protein concentrations in cholesterol-fed rats. Journal of Natural Remedies, 6, 109–114.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Acharya, C.K., Das, B., Madhu, N.R., Sau, S., De, M., Sarkar, B. (2023). A Comprehensive Pharmacological Appraisal of Indian Traditional Medicinal Plants with Anti-diabetic Potential. In: Noor, R. (eds) Advances in Diabetes Research and Management. Springer, Singapore. https://doi.org/10.1007/978-981-19-0027-3_8
Download citation
DOI: https://doi.org/10.1007/978-981-19-0027-3_8
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-19-0026-6
Online ISBN: 978-981-19-0027-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)