Abstract
Plants produce diverse groups of secondary metabolites (SMs); terpenoids are one such large group of SMs. Terpenoids are found commonly in related and unrelated plant taxa; however, some specific terpenoids are also reported in lower and higher taxa. The pharmacological importance and commercial utilization of terpenoids are on the top among the plant SMs. A number of reviews on biosynthesis of terpenoids have been published suggesting 2-C-methyl-d-erythritol-4-phosphate (MEP) and the mevalonate (MVA) as common pathways. Today, researchers are working on target-specific production of terpenoids by altering metabolic pathways and expressing genes in microsystems. Recent high-throughput analytical techniques coupled to functional genomics approach has geared up biosynthesis and overproduction of terpenoids. In this chapter, terpenoids have been reviewed in detail for their sources, biosynthetic pathways, in vitro production technologies, scale-up techniques, and biological activities. The ecological and environmental perspectives for function of terpenoids have also been discussed. Considering commercial implications of terpenoids in therapeutic, perfumery, food, flavor, and fuel industries, a comprehensive account on their prospective future has been concentrated upon. Extraction and detection methodologies for terpenoids have been focused. Attention has also been drawn toward the need for designing possible roadmap for its sustainable utilization.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Interest in medicinal plants has increased in past few decades due to their low cost and safety. Herbal drug is a quick growing sector worldwide; it is estimated that the world trade in medicinal plants will reach US$ 5 trillion by 2050 (Anonymous 2000). Terpenoids are naturally occurring hydrocarbons of plant origin with general formula (C5H8)n. The C5H8 unit is called isoprene unit (2-methyl-1,3-butadiene). Terpenoids constitute about 30,000 identified compounds (Dzubak et al. 2006; Mufflera et al. 2011). It is reported that terpenoids are probably the largest group of phytochemicals. They include essential oils phytohormones (cytokinin and gibberellins), resins, steroids, carotenoids, and others (Lohr et al. 2012). Terpenoids or isoprenoids are a group of structurally diverse phytochemicals known for their wide range of pharmacological activities. The term terpene is used for compounds with C5 (isoprene) units. Terpenoids are classified on the basis of number of isoprene units (Table 8.1).
The isoprene units have a branched and an unbranched ends, earlier termed as head and the later as tail. Although head-tail arrangements of the isoprene units are the commonest type of linking, e.g., in mono-, sesqui-, di-, and sesterterpenoids, tail-tail arrangements are at center of tri- and tetraterpene molecules. Polyisoprenoids like rubber and gutta are two of the many other naturally occurring compounds in which non-terpenoid structural parts are linked to terpenoids. Structure and examples of terpenoids are depicted in Table 8.2.
Apart from this, lower plants like bryophytes are also reviewed for the presence of terpenoids. It is estimated that in the past 40 years, around 1600 terpenoids have been reported from this plant group (Asakawa 1982, 1995; Asakawa et al. 2013). Ludwiczuk and Asakawa (2019), in their review on bryophytes, have enlisted over 128 studied pharmacologically active terpenoids from liverworts and mosses. Many of them are species specific like dumortane type (Dumortiera hirsute), hodgsonoxanes (Lepidolaena hodgsoniae), bergamotanes, and clavigerins (L. clavigera) (Chen et al. 2018). A mini review by Pai and Joshi (2014) have enlisted about 56 plant species producing triterpenoid betulinic acid. Very recently, 7 undescribed terpenoids, alongside 26 known compounds with isolated from aerial parts of Elsholtzia rugulosa (Yang et al. 2021). In a separate study, Yu et al. (2020) reported six new terpenoids from Eclipta prostrata, thus indicating continuation of adding new terpenoids to the family. Below is the list of some plants, parts, and terpenoids reported (Table 8.3).
Isoprenoids have reported to be having functionalities in plant cells. They serve as starting material in the formation of various plant hormones. Differential expression of pathway isozymes; metabolic regulation and network, alongside regulation to light, external stimuli; and metabolic exchanges across the subcellular compartments have been well documented by Tholl (2015). Terpenoids like sterols function in fluidity of plasma membrane; polyprenyl dolichol is and sugar carrier lipid used in protein glycosylation; phytol is a part of chlorophyll, tocopherols, phylloquinones, carotenoids thus functioning in photosynthesis, and polyprenyl plastoquinone in electron transport chain (Lohr et al. 2012).
A number of genes coding enzymes and regulators in biosynthesis of terpene were studied with respect to their location, genome, and expression (Chen et al. 2011). Though much is understood about the terpene metabolism, recent findings are revealing new substrates and enzymatic reactions in its biosynthesis (Zhou and Pichersky 2020). On other hand, alternative strategies like chemical synthesis are impractical due to the complex nature of these compounds (Misawa 2011; Oksman-Caldentey and Inzé 2004). With limited commercial success, biotechnological methods give an attractive alternative to obtain these drugs. Comprehensive reviews have already been published on microbial transformation and its production using tissue culture methods (Parra et al. 2009; Malinowska et al. 2013). This chapter concentrates on in vitro production of terpenoids.
2 Biosynthesis Pathway
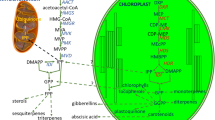
It is one of the large groups of phytochemicals in plant kingdom. They include representations in large number of plant molecules such as vitamins and hormones. Isopentenyl pyrophosphate (IPP) and dimethylallyl pyrophosphate (DMAPP) are the two precursors which significantly contribute into the biosynthesis of terpenoids. Classical studies by a number of workers have elucidated biosynthesis of terpenoids via acetyl-CoA and mevalonate (Qureshi and Porter 1981; Bloch 1992; Bach 1995; Bochar et al. 1999). Thus, mevalonate is the best studied pathway for terpenoid biosynthesis which includes mevalonic acid as an intermediate. Recent studies have revealed variations in biosynthesis of terpenoids production from the common 2-C-methyl-d-erythritol 4-phosphate (MEP) and the mevalonate (MVA) pathways. The earlier (MEP) being working in plastids and later one (MVA) in cytoplasm, endoplasmic reticulum and peroxisomes (Vranová et al. 2013).
As reported by Habtemariam (2019), acetyl-CoA a primary metabolite undergoes a series of reaction to produce a six carbon molecule mevalonic acid (Fig. 8.1). This further produces DMAPP and IPP an interchangeable 5 carbon (C) building blocks. Isopentenyl diphosphate (IPP) and dimethylallyl pyrophosphate (DMAPP) condense to produce geranyl diphosphate (GPP), which is considered as the intermediate precursor for all monoterpenes. The GPP serves precursor to monoterpene production, and further addition of a 5 carbon unit (IPP/DMAPP) produces farnesyl pyrophosphate (FPP) a precursor of sesquiterpenes (15 C) and its dimer to give triterpenes (30 carbon). The addition of another 5 carbon unit to FPP gives geranylgeranyl diphosphate (GGPP) as an originator of diterpenes (20 C). Dimerization of GGPP gives tetraterpenes (40 C). Mostly, head-to-tail arrangements by addition of 5 carbon units are observed, exceptionally head to head in tri- and tetraterpenes. Though we consider 5 carbon unit (IPP and DMAPP) for biosynthesis terpenoids, monoterpenes (10 C) are considered to be the smallest unit.
An overview of terpenoid biosynthesis (source: Habtemariam 2019)
Studies also reveal that enzymes such as Nudix hydrolases and isopentenyl phosphate kinase (IPK) affect biosynthesis of terpene in cytosol as well as plastids by regulating IPP and DMAPP ratios (Zhou and Pichersky 2020).
3 In Vitro Production Methodology
Plants are looked as inexhaustible sources of new drugs. In vitro methods as alternative strategies are applied in two ways for secondary metabolite production: (1) biomass and (2) synthesis. Biomass aggregation would rely on in vitro culturing of shoot, root, callus, etc. (Guerriero et al. 2018; Hussain et al. 2012). Terpenoids are no different, and they play vital role in the organism. Apart from pharmacological properties of terpenoids, their cosmeceuticals, antioxidant, and nutraceutical properties of terpenoids are studied vastly (Bonfill et al. 2013). In vitro culture techniques coupled with bioengineering has demonstrated great applications. Artemisinin a sesquiterpene lactone drug from Artemisia annua is known for its antimalarial property. Rationally, overexpression of farnesyl diphosphate synthase (FDS) enzyme which condenses IPP with DMAPP to form (FPP) farnesyl diphosphate yields higher artemisinin. The same was achieved by placing a 35S CaMV promoter with FPS gene using Agrobacterium mediated transformation. This study showed about fourfold increase in the content (Exposito et al. 2010). Farzaei et al. (2017) included 55 mono- and sesquiterpenoids alongside other compounds in their review on pharmacological and phytochemical properties of Mentha longifolia. They also tabulated plant part used and the extraction method. Few of the plants alongside their in vitro methods studied and terpenoids identified are enlisted below (Table 8.4).
4 Scale-up Techniques and Bioreactors
Commercially important secondary metabolites (SMs) need scale-up using bioreactors, as the traditional way of production may not suffice the need. It takes help of modern methods like genomics, proteomics, metabolomics etc. to modify the metabolic routes in an organism for scaled-up production of SMs (DellaPenna 2001). Shikimate, polyketide, and terpenoids pathways are the main routes for production of secondary metabolites. Terpenoids comprise around one-third of the total secondary metabolites (Verpoorte 2000). Scaling-up may include metabolic engineering, up- and downregulating pathways, redirecting common precursors, and targeting metabolites to specific cell compartments (Chandran et al. 2020).
Overexpressing of FaNES1 (Fragaria ananassa nerolidol synthase 1) a protein identified as sensor for production of both mono and sesquiterpene precursors. High levels of monoterpene linalool was achieved by CaMV 35S-driven promoter and engineered with wild strawberry FvNES1 (Fragaria vesca nerolidol synthase 1) plastid targeting region (Aharoni et al. 2006). It was also reported that, compared to the monoterpenes, sesquiterpene engineering in plants is difficult as precursor pool required is less. In another study, Rodríguez et al. (2014) observed that downregulating production of terpene D-limonene is inversely correlated by lowering fungal infection in transgenic oranges. Redirecting common precursor, viz., prenyl pyrophosphate (C5), to provide an adequate flux of IPP and DMAPP can be achieved by manipulation of genes regulating MVA pathway or by inserting MEP pathway in organisms having only MVA pathway (Vavitsas et al. 2018; Yang et al. 2016). Escherichia coli and Saccharomyces cerevisiae are termed as industrial workhorses and are used to produce high-valued terpenoids in bioreactors. Zhang and Hong (2020) in their mini review have well documented a list of strategies for production of terpenoids by targeting it to specific cell compartments.
Gupta et al. (2021), in an overview on pharmaceutical properties and biotechnological advancements of Withania coagulans, have reported that the plant is on the verge of extinction. Withanolides are a group of steroidal lactone triterpenoids which can also be synthesized by terpenoids forming mevalonate pathway (Kreis and Muller-Uri 2010). Seven different methods have been highlighted for increased production of bioactive compounds from the plant; they include Agrobacterium-mediated transformation, metabolic engineering, plant tissue culture, germination by seeding, abiotic/biotic elicitors, gene transfer, and nanoparticle synthesis. To infer the work of Gupta et al. (2021), in vitro system is suggested to be the attractive option to achieve the goal.
Apart from the above, Zhang et al. (2019) used Agrobacterium-mediated transformation studies to functionally expedite terpenoids biosynthesis in Tripterygium wilfordii. In a separate study, pharmaceutically important metabolites were enhanced by adding silver nitrate (linalool, estragole) and yeast extract (rutin, isoquercetin) in Ocimum basilicum suspension cultures (Açıkgöz 2020). Polzin and Rorrer (2018) reported selective production of β-myrcene (monoterpene) from Octodes secundiramea on nutrient perfusion cultivation with bromide-free medium. High levels of amorphadiene a precursor of artemisinin were produced by using Bacillus subtilis as terpenoids cell factory (Pramastya et al. 2021). Finding of new genes and computational biology has boosted production of new terpenoids using microbial methods. Expression of plant enzymes in microorganisms has helped biosynthesis of diverse class of complex terpenoid derivatives (Belcher et al. 2020).
5 Extraction and Detection Techniques
A number of extraction and detection methods are employed for determining the contents of phytochemicals from plants (Fig. 8.2). Range of methods from conventional, simple maceration to recent ultra-sonication and microwave-assisted extractions are deployed for extracting terpenoids. In a study, phenolic and terpenoids were identified using reversed-phase high-performance liquid chromatography (RP-HPLC) and head space solid phase microextraction gas chromatography coupled with mass spectrometry (HS-SPME-GC/MS), respectively (Açıkgöz 2020; Muñoz-Redondo et al. 2020)
More recently, Vaníčková et al. (2020) have identified species-specific terpenoid markers to chemo-taxonomically distinguish four species and one subspecies of Dracaena using solid-phase microextraction – coupled to a gas chromatography – ion trap tandem mass spectrometry (SPME-GC × GC-MS). Percolation and maceration extraction methods have been reported for extraction of terpenoids from stem and root material of Eurycoma longifolia. Supercritical fluid extraction and microwave-assisted extraction are some of the non-conventional methods utilized and reported for extraction in many plants including Andrographis paniculata for terpenoids andrographolide, deoxyandrographolide, and its variants (Aziz et al. 2021). Plants reported with different extraction and detection methods alongside terpenoids identified is tabulated in Table 8.5.
6 Biological Activities
The significance of terpenoids is because of their anticancer, anti-inflammatory, antiplatelet, antibacterial, hypocholesterolemic, immune adjuvant, anti-viral, antibacterial, fungicidal, and antileishmanial agents like pharmacological properties (Abdelrahman and Jogaiah 2020). Anticancer and antimicrobial properties are some of the largely studied activities of terpenoids. Apart from cancer treatment and life style disorders, withanolides as bioactive terpenoid derivative from Withania has proved antianxiety activity in albino mice model (Gupta et al. 2021). On other hand, a range of biological activities may be attributed to terpenoids from cucurbit family Cucurbitacin, and its derivatives from Cucurbitaceae have been reviewed against inflammation, cancer, and many other properties (Montesano et al. 2018). The terpenoids are not only studied from plant system but are reported from marine bacteria and fungi with similar properties. Altemicidin, marinocyanins A-F, azamerone, and napyradiomycins were some of the potent monoterpenoids identified from bacteria (Gozari et al. 2021). Chen et al. (2012) reviewed Sinularia soft coral as a source of terpenoids for potential bioactivities such as antimicrobial, anti-inflammatory, and cytotoxic activities. Terpenoids like andrographolide, glycyrrhetinic acid, ursolic acid, costunolide, β-elemene, glaucocalyxin A, and cucurbitacin B have been reviewed and reported to show anti-liver fibrosis by Ma et al. (2020).
Plant essential oils are a media of communication within and between the plants. These are chemical terpenes containing repeated units of isoprenes. Depending on the occurrence in plant parts, they may be for attracting pollinators or to repel predators. They are generally present on leaves in secretary glands (e.g., mint, sage, basil, pine), in flowers (e.g., orange, chamomile, clove), in wood (e.g., sandalwood, balsam, camphor), in fruits (e.g., star anise, fennel, apple), in rhizomes (ginger, turmeric), and in seeds (cardamom, nutmeg, pepper). Geraniol (geranium), linalool (mint), myrcene (thyme), and ß-ocimene (basil) are some of the important essential oils (and plant name) having terpenes (Böttger et al. 2018). They are termed biopesticides due to their repellent and insecticidal properties.
The sequence of outbreaks of viral infection particularly corona, starting from the year 2003, SARS-CoV; 2012, MERS-CoV; and 2019, SARS-CoV-2, raised global health concerns. Conventional drug limitations forced researchers to look into plant-based bioactive compounds. Bhattacharya et al. (2021), in their review, updated on antiviral drugs of plant origin which are potential players in counteracting these viruses. Rajan et al. (2021) in a recent commentary on promising antivirals from ayurvedic herbs against COVID-19 have mentioned 26 natural compounds along with terpenoids as a potential candidate based on in silico approach. Table 8.6 enlists some important bioactivities of terpenoids or terpenoid-derived compounds majorly reviewed by Martin-Smith and Sneader (1969).
7 Commercial Utilization and Prospects
Commercial utilization and prospects should include lucrative applications of terpenes in various industries. For example, rubber, a polyterpene with repeating units of isoprene, is extensively studied, known and one of the largest industries. Still, it is important to find and concentrate on such targets which are of real concern in the coming years. Industries like therapeutics, food, flavor, perfumery, or even fuel are highly commercially utilized, in relation to environment. However, without better environment and sustainable resources, all other industries will have poor prospects. Thus, environment and ecology being one of the three such points will be explored in this section.
7.1 Environment and Ecology
Terpenes are the most abundantly found and studied natural product. Out of the three main categories of natural products, it is estimated that terpenoid accounts for 55%, alkaloid 27%, and phenolic 18% (Croteau et al. 2000), thus overstating their importance in nature and for mankind. Cheng et al. (2007) have discussed the role of terpene in ecological point of view. Terpenes are assumed to have a role in earth’s atmosphere by interacting with solar radiations. The 500 teragrams of isoprene emission annually is being estimated which influence formation of ozone (Zwenger and Basu 2008). Beyond plant-insect interaction, terpenes also act as messengers in plant defense mechanism, which can be further utilized commercially for their insect repellent or even attractants.
7.2 Therapeutic, Perfumery, Food, Flavor
Advent of bioinformatics and progressions in molecular databases have contributed in understanding the synthesis and pathways much better. Commercially, they have contributed largely to therapeutics, perfumery, food, and flavor industries. Also their role in essential oils and cosmetics can’t be underestimated. Developing agronomic traits in medicinal plants is an important area of research explored in the past few decades. A combination of engineering to use “omics” technologies and computational and systems biology, along with synthetic chemistry, shows an imperative prospect in commercialization. Artemisinin, squalane, sandalwood, and patchouli oils are few of the examples discussed by Leavell et al. (2016) as potential terpenes in pharma and fine chemicals. The volatility of terpenoid supplies and prices has been attributed to smaller market size and the dependency on the plant source for its production. Thus, alternative strategies for stable production using fermentation are an attractive option. Amyris Inc. is a synthetic biotechnology company making its presence in all the four sectors for terpenoids, artemisinin (pharma), squalane, numerous fragrance oils (fine), isoprene, farnesene for lubricants, polymers (as commodity chemicals), and farnesene for diesel and jet fuel (fuels). Antimicrobial-resistant (AMR) bacteria or even superbug issues in present-day list of drugs can be updated by addition of phytocompounds chiefly terpenes (Mahizan et al. 2019; Zwenger and Basu 2008). Terpenes may reduce the rate of antibiotic resistance via livestock feed by substituting orthodox antibiotics.
7.3 Fuel
“Biofuel” a renewable source particularly from plant origin is another prospective application of terpenes in commercialization. Countries such as the USA, Brazil, and European Union are expressing interest and supporting research based on such alternative sources. Diesel obtained from Copaifera langsdorffii a natural biofuel largely contains terpenes. In a study, Mewalal et al. (2017) have summarized specific terpenes as appropriate standby or composite for present-day fuels. They have also made a comparison of such biofuels with much known fuels. It has been reported that terpenes could assist as standalone or blended up to 65% in diesel engines. There are more than a few challenges to leverage plant terpenes as a commercially viable source for replacing diesel or gasoline. A roadmap has been proposed by Mewalal et al. (2017) for commercial recovery of terpenes synthesized. Its tremendous potential as sustainable biofuel has been swotted and described.
The above-discussed points will raise concerns in the coming years. Thus, it was important to highlight the area of future research on terpenoids.
8 Conclusions
Terpenes are still being fully realized for their applications. Discovery of more and more number of terpenes will surely add to the ever-growing list of its utilization. Like most other bioactives, terpenoids from nature are supply limited. However, production at commercial level is driven by recent annotated technologies. Few important such terpenes are reviewed and discussed above. Differences in the challenges to produce terpenoids vary from one to another, thus making it non- comprehensive. It means that a method developed for one terpene cannot be applied to another. On the other hand, predictive engineering along with in vitro culture techniques can lead to selective production of terpenoids. With this, it is also projected that wide understanding of structure and activity liaisons of terpenes will open avenue for medicinally important phytocompounds. Further study on their activity as well as mechanism will be of great importance. Furthermore, work related to cost-effective and more contemporary way for production of biodiesels rather than the now conventional ethanol conversion from sugarcane or corn would be essential. Conclusively, owing to the ever-increasing number of terpenoids, a lot of study still can be undertaken for in vitro sustainable production, fundamental understanding of their role in biological processes, and commercial utilization.
References
Abdelrahman M, Jogaiah S (2020) Production of plant bioactive triterpenoid and steroidal Saponins. In: Bioactive molecules in plant defence. Springer, Cham, pp 5–13
Açıkgöz MA (2020) Establishment of cell suspension cultures of Ocimum basilicum L. and enhanced production of pharmaceutical active ingredients. Ind Crop Prod 148:112278
Aharoni A, Jongsma MA, Kim TY et al (2006) Metabolic engineering of terpenoid biosynthesis in plants. Phytochem Rev 5:49–58
Anonymous, Government of India, Medicinal plants introduction, Indian System of Medicine and Homoeopathy (ISMH), Department of ISMH, Ministry of Health and Family Welfare, Governmet of India (2000). http://indianmedicine.nic.in/html/plants/mimain.htm
Asakawa Y (1982) Chemical constituents of the hepaticae. In: Herz W, Grisebach H, Kirby GW (eds) Progress in the chemistry of organic natural products, vol vol. 42. Springer, Vienna, pp 1–285
Asakawa Y (1995) Chemical constituents of the bryophytes. In: Herz W, Kirby GW, Moore RE, Steglich W, Tamm C (eds) Progress in the chemistry of organic natural products, vol vol. 65. Springer, Vienna, pp 1–618
Asakawa Y, Ludwiczuk A, Nagashima F (2013) Chemical constituents of bryophytes: bio- and chemical diversity, biological activity, and chemosystematics. In: Kinghorn AD, Falk H, Kobayashi J (eds) Progress in the chemistry of organic natural products, vol vol. 95. Springer-Verlag, Vienna, pp 1–796
Aziz NAA, Hasham R, Sarmidi MR et al (2021) A review on extraction techniques and therapeutic value of polar bioactives from Asian medicinal herbs: case study on Orthosiphon aristatus, Eurycoma longifolia and Andrographis paniculata. Saudi Pharm J 29(2):143–165
Bach TJ (1995) Some aspects of isoprenoid biosynthesis in plants. A review. Lipids 30:191–202
Banthorpe DV, Bates MJ, Ireland MJ (1995) Stimulation of accumulation of terpenoids by cell suspensions of Lavandula angustifolia following pre-treatment of parent callus. Phytochemistry 40(1):83–87
Belcher MS, Mahinthakumar J, Keasling JD (2020) New frontiers: harnessing pivotal advances in microbial engineering for the biosynthesis of plant-derived terpenoids. Curr Opin Biotechnol 65:88–93
Bertoli A, Leonardi M, Krzyzanowska J et al (2011) Mentha longifolia in vitro cultures as safe source of flavouring: ingredients. Acta Biochim Pol 58(4):581–587
Bhattacharya R, Dev K, Sourirajan A (2021) Antiviral activity of bioactive phytocompounds against coronavirus: an update. J Virological Met 290:114070
Bloch K (1992) Sterol molecule: structure, biosynthesis and function. Steroids 57:378–382
Bochar DA, Friesen JA, Stauffacher CV et al (1999) Biosynthesis of mevalonic acid from acetyl-CoA. In: Cane D (ed) Comprehensive natural product chemistry, vol Vol. 2. Pergamon, Oxford, pp 15–44
Bonfill M, Malik S, Mirjalili MH et al (2013) Production and genetic engineering of Terpenoids production in plant cell and organ cultures. In: Ramawat K, Mérillon JM (eds) Natural products. Springer, Berlin, Heidelberg, pp 2761–2796
Böttger A, Vothknecht U, Bolle C et al (2018) Terpenes and terpenoids. In: Lessons on caffeine, cannabis & co. learning materials in biosciences. Springer, Cham. https://doi.org/10.1007/978-3-319-99546-5_10
Chandran H, Meena M, Barupal T et al (2020) Plant tissue culture as a perpetual source for production of industrially important bioactive compounds. Biotech Rep 26:e00450
Chen F, Tholl D, Bohlmann J et al (2011) The family of terpene synthases in plants: a mid-size family of genes for specialized metabolism that is highly diversified throughout the kingdom. Plant J 66:212–229
Chen W, Li Y, Guo Y (2012) Terpenoids of Sinularia soft corals: chemistry and bioactivity. Acta Pharm Sin B 2(3):227–237
Chen F, Ludwiczuk A, Wei G et al (2018) Terpenoid secondary metabolites in bryophytes: chemical diversity, biosynthesis and biological functions. Crit Rev Plant Sci 37:210–231
Cheng A, Lou Y, Mao Y et al (2007) Plant terpenoids: biosynthesis and ecological functions. J Integr Plant Biol 49:179–186
Croteau R, Kutchan TM, Lewis NG (2000) Natural products (secondary metabolites). In: Buchanan B, Gruissem W, Jones R (eds) Biochemistry and molecular biology of plants. American Society of Plant Physiologists, Rockville, pp 1250–1318
DellaPenna D (2001) Plant metabolic engineering. Plant Physiol 125(1):160–163
Dzubak P, Hajduch M, Vydra D et al (2006) Pharmacological activities of natural triterpenoids and their therapeutic implications. Nat Prod Rep 23:394–341
Ercioglua E, Velioglub HM, Boyaci IH (2018) Determination of terpenoid contents of aromatic plants using NIRS. Talanta 178:716–721
Exposito O, Syklowska-Baranek K, Moyano E et al (2010) Metabolic responses of Taxus media transformed cell cultures to the addition of methyl jasmonate. Biotechnol Prog 26:1145–1153
Farzaei MH, Bahramsoltani R, Ghobadi A et al (2017) Pharmacological activity of Mentha longifolia and its phytoconstituents. J Tradit Chin Med 37(5):710–720
Gozari M, Alborz M, El-Seedi HR (2021) Chemistry, biosynthesis and biological activity of terpenoids and meroterpenoids in bacteria and fungi isolated from different marine habitats. Eur J Med Chem 210:112957
Guerriero G, Berni R, Muñoz-Sanchez JA et al (2018) Production of plant secondary metabolites: examples, tips and suggestions for biotechnologists. Genes (Basel) 9(6):E309
Gupta R, Sonawane T, Pai SR (2021) An overview on pharmaceutical properties and biotechnological advancement of Withania coagulans. Adv Tradit Med. https://doi.org/10.1007/s13596-021-00558-7
Habtemariam S (2019) Introduction to plant secondary metabolites - from biosynthesis to chemistry and antidiabetic action. In: Medicinal foods as potential therapies for Type-2 diabetes and associated diseases, the chemical and pharmacological basis of their action, pp 109–132
Harman-Ware AE, Sykes R, Peter GF et al (2016) Determination of terpenoid content in pine by organic solvent extraction and fast-GC analysis. Front Energy Res 4:2. https://doi.org/10.3389/fenrg.2016.00002
Hussain MS, Fareed S, Ansari MS et al (2012) Current approaches toward production of secondary plant metabolites. J Pharm Bioallied Sci 4(1):10–20
Kreis W, Muller-Uri F (2010) Biochemistry of sterols, cardiac glycosides, brassinosteroids, phytoecdysteroids and steroid saponins. In: Michael W (ed) Annual plant reviews. Wiley, Singapore, pp 304–363
Kshirsagar PR, Pai SR, Nimbalkar MS et al (2015) Quantitative determination of three pentacyclic triterpenes from five Swertia L. species endemic to Western Ghats, India, using RP-HPLC analysis. Nat Prod Res 29(19):1783–1788
Leavell MD, McPhee DJ, Paddon CJ (2016) Developing fermentative terpenoid production for commercial usage. Curr Opin Biotechnol 37:114–119
Liu WX, Zhao JW, Zuo AX (2018) Two novel terpenoids from the cultured Perovskia atriplicifolia. Fitoterapia 130:152–155
Lohr M, Schwender J, Polle JEW (2012) Isoprenoid biosynthesis in eukaryotic phototrophs: a spotlight on algae. Plant Sci 185–186:9–22
Ludwiczuk A, Asakawa Y (2019) Bryophytes as a source of bioactive volatile terpenoids – a review. Food Chem Toxicol 132:110649
Ma X, Jiang Y, Wen J et al (2020) A comprehensive review of natural products to fight liver fibrosis: alkaloids, terpenoids, glycosides, coumarins and other compounds. Eur J Pharmacol 888:173578
Mahizan NA, Yang SK, Moo CL et al (2019) Terpene derivatives as a potential agent against antimicrobial resistance (AMR) pathogens. Molecules 24(14):2631
Malinowska M, Sikora E, Ogonowski J (2013) Production of triterpenoids with cell and tissue cultures. Acta Biochim Pol 60(4):731–735
Martin-Smith M, Sneader WE (1969) Biological activity of the terpenoids and their derivatives--recent advances. Prog Drug Res 13:11–100
Mewalal R, Rai DK, Kainer DA et al (2017) Plant-derived terpenes: a feed stock for specialty biofuels. Trends Biotechnol 35(3):227–240
Misawa N (2011) Pathway engineering for functional isoprenoid. Curr Opin Biotechnol 22:627–633
Mkaddem M, Bouajila J, Ennajar et al (2009) Chemical composition and antimicrobial and antioxidant activities of Mentha (longifolia L. and viridis) essential oils. J Food Sci 74(7):M358–M363
Monfort LEF, Bertolucci SKV, Lima AF, de Carvalho AA, Mohammed A, Blank AF, Pinto JEBP (2018) Effects of plant growth regulators, different culture media and strength MS on production of volatile fraction composition in shoot cultures of Ocimum basilicum. Ind Crop Prod 116:231–239
Montesano D, Rocchetti G, Putnik P et al (2018) Bioactive profile of pumpkin: an overview on terpenoids and their health-promoting properties. Curr Opin Food Sci 22:81–87
Motamed SM, Naghibi F (2010) Antioxidant activity of some edible plants of the Turkmen Sahra region in northern Iran. Food Chem 119(4):1637–1642
Mufflera K, Leipolda D, Schellera MC et al (2011) Biotransformation of triterpenes. Process Biochem 46:1–15
Muñoz-Redondo JM, Ruiz-Moreno MJ, Puertas B et al (2020) Multivariate optimization of headspace solid-phase microextraction coupled to gas chromatography-mass spectrometry for the analysis of terpenoids in sparkling wines. Talanta 208:120483
National Center for Biotechnology Information (2021a) PubChem compound summary for CID 3509874, Gibberellin 1. Retrieved February 6, 2021 from https://pubchem.ncbi.nlm.nih.gov/compound/Gibberellin-1
National Center for Biotechnology Information (2021b) PubChem compound summary for CID 6549, Linalool. Retrieved February 6, 2021 from https://pubchem.ncbi.nlm.nih.gov/compound/Linalool
National Center for Biotechnology Information (2021c) PubChem compound summary for CID 1254, Menthol. Retrieved February 6, 2021 from https://pubchem.ncbi.nlm.nih.gov/compound/Menthol
National Center for Biotechnology Information (2021d) PubChem compound summary for CID 5464156, (R)-Lavandulol. Retrieved February 6, 2021 from https://pubchem.ncbi.nlm.nih.gov/compound/R_-Lavandulol
National Center for Biotechnology Information (2021e) PubChem compound summary for CID 161276, Secologanin. Retrieved February 6, 2021 from https://pubchem.ncbi.nlm.nih.gov/compound/Secologanin
National Center for Biotechnology Information (2021f) PubChem compound summary for CID 92776, Zingiberene. Retrieved February 6, 2021 from https://pubchem.ncbi.nlm.nih.gov/compound/Zingiberene
National Center for Biotechnology Information (2021g) PubChem compound summary for CID 36314, Paclitaxel. Retrieved February 6, 2021 from https://pubchem.ncbi.nlm.nih.gov/compound/taxol
National Center for Biotechnology Information (2021h) PubChem compound summary for CID 64971, Betulinic acid. Retrieved February 6, 2021 from https://pubchem.ncbi.nlm.nih.gov/compound/Betulinic-acid
National Center for Biotechnology Information (2021i) PubChem compound summary for CID 5280794, Stigmasterol. Retrieved February 6, 2021 from https://pubchem.ncbi.nlm.nih.gov/compound/Stigmasterol
National Center for Biotechnology Information (2021j) PubChem compound summary for CID 99474, Diosgenin. Retrieved February 6, 2021 from https://pubchem.ncbi.nlm.nih.gov/compound/Diosgenin
National Center for Biotechnology Information (2021k) PubChem compound summary for CID 64945, Ursolic acid. Retrieved February 6, 2021 from https://pubchem.ncbi.nlm.nih.gov/compound/Ursolic-acid
National Center for Biotechnology Information (2021l) PubChem compound summary for CID 5280896, Abscisic acid. Retrieved February 6, 2021 from https://pubchem.ncbi.nlm.nih.gov/compound/S_-_-Abscisic-acid
National Center for Biotechnology Information (2021m) PubChem compound summary for CID 15102684, 5-Deoxystrigol. Retrieved February 6, 2021 from https://pubchem.ncbi.nlm.nih.gov/compound/5-Deoxystrigol
National Center for Biotechnology Information (2021n) PubChem compound summary for CID 445070, Farnesol. Retrieved February 6, 2021 from https://pubchem.ncbi.nlm.nih.gov/compound/Farnesol
National Center for Biotechnology Information (2021o) PubChem compound summary for CID 6433320, Dolichol-20. Retrieved February 6, 2021 from https://pubchem.ncbi.nlm.nih.gov/compound/Dolichol-20
National Center for Biotechnology Information (2021p) PubChem compound summary for CID 65030, Artemisin. Retrieved February 6, 2021 from https://pubchem.ncbi.nlm.nih.gov/compound/Artemisin
Oksman-Caldentey KM, Inzé D (2004) Plant cell factories in the post-genomic era: new ways to produce designer secondary metabolites. Trends Plant Sci 9:433–440
Pai SR, Joshi RK (2014) Distribution of betulinic acid in plant kingdom. Plant Sci Today 1(3):103–107
Pai SR, Joshi RK (2016) Variations in pentacyclic triterpenoids in different parts of four Ocimum species using reverse phase-high performance liquid chromatography. Proc Natl Acad Sci India Sect B 87:1153–1158
Pai SR, Joshi RK (2018) Optimized densitometric analysis for determination of triterpenoid isomers in Vitex negundo L. leaf. Natl Acad Sci Lett 41(5):323–327
Pai SR, Nimbalkar MS, Pawar NV et al (2011) Optimization of extraction techniques and quantification of Betulinic acid (BA) by RP-HPLC method from Ancistrocladus heyneanus wall. Ex Grah. Ind Crop Prod 34:1458–1464
Pai SR, Upadhya V, Hegde HV (2014) New report of triterpenoid betulinic acid along with Oleanolic acid from Achyranthes aspera by reversed- phase–ultra flow liquid chromatographic analysis and confirmation using high-performance thin-layer chromatographic and Fourier transform–infrared. J Planar Chromatogr 27:38–41
Pai SR, Upadhya V, Hegde HV et al (2016) Determination of betulinic acid, oleanolic acid and ursolic acid from Achyranthes aspera L. using RP-UFLC-DAD analysis and evaluation of various parameters for their optimum yield. Indian J Exp Biol 54:196–202
Pai SR, Upadhya V, Hegade HV et al (2018) In vitro rapid multiplication and determination of terpenoids in callus cultures of Achyranthes aspera Linn. Indian J Biotechnol 17:151–159
Parra A, Rivas F, Garcia-Granados A et al (2009) Microbial transformation of triterpenoids. Mini-Rev Org Chem 6:307–320
Polzin J, Rorrer GL (2018) Selective production of the acyclic monoterpene β-myrcene by microplantlet suspension cultures of the macrophytic marine red alga Ochtodes secundiramea under nutrient perfusion cultivation with bromide-free medium. Algal Res 36:159–166
Pramastya H, Xuea D, Abdallah II et al (2021) High level production of amorphadiene using Bacillus subtilis as an optimized terpenoid cell factory. New Biotechnol 60:159–167
Qureshi N, Porter JW (1981) Conversion of acetyl-coenzyme a to isopentenyl pyrophosphate. In: Porter JW, Spurgeon SL (eds) Biosynthesis of isoprenoid compounds, vol vol. 1. Wiley, New York, pp 47–94
Rajan M, Gupta P, Kumar A (2021) Promising antiviral molecules from Ayurvedic herbs and spices against COVID-19. Chin J Integr Med. https://doi.org/10.1007/s11655-021-3331-8
Rodríguez A, Shimada T, Cervera M et al (2014) Terpene Down-regulation triggers Defence responses in transgenic Orange leading to resistance against fungal Pathogens1[W]. Plant Physiol 164:321–339
Stojakowska A, Michalska K, Kłeczek N et al (2018) Phenolics and terpenoids from a wild edible plant Lactuca orientalis (Boiss.) Boiss.: a preliminary study. J Food Comp Anal 69:20–24
Tholl D (2015) Biosynthesis and biological functions of terpenoids in plants. Adv Biochem Eng Biotechnol 148:63–106
Upadhya V, Ankad GM, Pai SR et al (2014) Accumulation and trends in distribution of three triterpenoids in various parts of Achyranthes coynei determined using RP-UFLC analysis. Phcog Mag 10:398–401
Vaníčková L, Pompeiano A, Maděr P et al (2020) Terpenoid profiles of resin in the genus Dracaena are species specific. Phytochemistry 170:112197
Vavitsas K, Fabris M, Vickers CE (2018) Terpenoid metabolic engineering in photosynthetic microorganisms. Genes 9:520
Verpoorte R (2000) Plant secondary metabolites. In: Verpoorte R, Alfermann AW (eds) Metabolic engineering of plant secondary metabolism. Kluwer Academic Publisher, Dodrecht, Boston, London, pp 1–30
Vranová E, Coman D, Gruissem W (2013) Network analysis of the MVA and MEP pathways for isoprenoid synthesis. Annu Rev Plant Biol 64:665–700
Wang X, Jin XY, Zhou JC et al (2020) Terpenoids from the Chinese liverwort Heteroscyphus coalitus and their antivirulence activity against Candida albicans. Phytochemistry 174:112324
Yang C, Gao X, Jiang Y et al (2016) Synergy between methylerythritol phosphate pathway and mevalonate pathway for isoprene production in Escherichia coli. Metab Eng 37:79–91
Yang F, Pu HY, Yaseen A et al (2021) Terpenoid and phenolic derivatives from the aerial parts of Elsholtzia rugulosa and their anti-inflammatory activity. Phytochemistry 181:112543
Yu SJ, Yu JH, Yu ZP et al (2020) Bioactive terpenoid constituents from Eclipta prostrate. Phytochemistry 170:112192
Zhang C, Hong K (2020) Production of Terpenoids by synthetic biology approaches. Front Bioeng Biotechnol 8:347. https://doi.org/10.3389/fbioe.2020.00347
Zhang B, Huo Y, Zhang J, Zhang X et al (2019) Agrobacterium rhizogenes-mediated RNAi of Tripterygium wilfordii and application for functional study of terpenoid biosynthesis pathway genes. Ind Crops Prod 139:111509
Zhou F, Pichersky E (2020) More is better: the diversity of terpene metabolism in plants. Curr Opin Plant Biol 55:1–10
Zwenger S, Basu C (2008) Plant terpenoids: applications and future potentials. Biotech Mol Biol Rev 3(1):1–7
Acknowledgments
Author is indebted to Rayat Shikshan Sanstha, Satara, (MS) India, for providing support.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Ethics declarations
None.
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Pai, S.R. (2022). In Vitro Production of Terpenoids. In: Belwal, T., Georgiev, M.I., Al-Khayri, J.M. (eds) Nutraceuticals Production from Plant Cell Factory. Springer, Singapore. https://doi.org/10.1007/978-981-16-8858-4_8
Download citation
DOI: https://doi.org/10.1007/978-981-16-8858-4_8
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-16-8857-7
Online ISBN: 978-981-16-8858-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)