Abstract
Biofilms are colonies of microorganisms that adhere to a surface, often in a flowing system. They convey advantages to the community of cells within, such as access to nutrients, protection from physicochemical stress, intercellular communication and horizontal gene transfer. In many instances, biofilms are viewed as problematic, for example, causing difficult-to-treat infections or corroding metal surfaces. However, the properties that make biofilms a challenge in some circumstances are beneficial in others. As biofilms have mechanical stability, are resistant to the toxic effects of xenobiotics and are stable for extended time periods, they have potential applications in the continuous production of chemicals and the biodegradation of pollutants. In this chapter, we will present examples of the research that has been conducted on examining single-species biofilms of bacteria and fungi in the biosynthesis of commodity and fine chemicals and the biodegradation of xenobiotics. The merits and disadvantages of biofilms in different applications are discussed.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
5.1 Introduction
Biofilms exist as a natural form of immobilisation, in which the microbial cells produce an extracellular polymeric substance (EPS) that adheres them to a suitable surface, provides mechanical stability and a barrier against toxic compounds. Extensive work has been conducted with various microorganisms in relation to their growth as biofilms and common stages of biofilm development are recognised (Fig. 5.1). Biofilms are most frequently associated with surfaces submerged in an aqueous solution to which free-floating, or planktonic, cells might adhere. Following initial attachment, the cells start to produce EPS, which adheres the colony to the surface and allows further development to form a mature biofilm that contains channels that allow the distribution of nutrients and signalling molecules. Eventually, parts of the biofilm may detach (dispersal). The nature of the surface is a key factor in biofilm development and in particular the degree of hydrophobicity. This is reflected in the protocols used to cultivate biofilms in the laboratory which often employ polystyrene microtitre plates, glass slides and silicone as surfaces to promote attachment (Azeredo et al. 2017). The EPS is typically comprised of polysaccharide, protein and/or extracellular DNA (eDNA). In nature, multispecies biofilms are normal, and there is genetic exchange between the cells as well as syntrophy, facilitating a more complex suite of metabolic reactions. This is in contrast to most studies on biofilms conducted in the laboratory, which often focus on a single species. The importance of biofilms in secondary wastewater treatment, for example, in trickling filters, to decrease the biochemical oxygen demand has been well established; however, in recent years, the attention of researchers has been on how to treat biofilms that cause infection or foul industrial pipes and metal surfaces; thus, there is a widespread perception that biofilms are ‘bad’. In this chapter, we will illustrate the advantage of biofilms (mainly single culture) in the areas of fermentation, bioremediation and biocatalysis and highlight the importance of understanding and optimising/controlling their growth so that novel, sustainable methods of pollution removal and biochemical production can be developed.
5.1.1 Useful Biofilms
Suspended culture is the main form of microbial growth that is used in the application of microorganisms in the production of valuable compounds, either by fermentation or biotransformation. However, these cultures can suffer from relatively short periods of activity and are sensitive to the toxic effects of accumulating metabolites or the starting substrate; thus, they may not be sufficiently productive to replace an effective chemical method of production (van Beilen et al. 2003; Garzón-Posse et al. 2018). At the other end of the pipeline, microorganisms are important for the biodegradation of xenobiotic pollutants, but although there are many successful examples, the introduction of a new strain directly into a polluted environment to promote bioremediation (bioaugmentation) often results in the strain being out-grown by indigenous organisms (Mrozik and Piotrowska-Seget 2010). Furthermore, as the number of specialised chemicals that we use increases, traditional wastewater treatment facilities are unable to completely eliminate them from contaminated influents, thus they become persistent in aquatic environments.
It has been known for several decades that artificially immobilising microbial cells and enzymes, e.g. by encapsulating in a hydrogel, has proven to alleviate the problems associated with using suspended or planktonic cultures (Es et al. 2015). By extension, naturally immobilised cells would be expected to have similar features, with the advantage that no additional manipulation is required.
5.1.2 Bioreactor Configuration
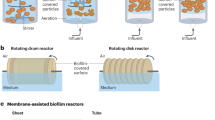
Small-scale cultivation of biofilms is mainly done in multiwell-plates, but to operate biofilms for longer time periods and study their characteristics in relation to productivity, other bioreactors are employed, which enable operation for longer periods of time with controlled parameters. In addition to high reaction rates, biofilm reactors also have some other advantages like improved mixing, improved mass transfer, high shear stress, controlled biofilm growth, high product yield, providing resistance to toxic compounds, high product recovery, reduced fermentation time, high operational stability, compact technology, etc. Biofilm reactors have various configurations the most common of which are shown in Fig. 5.2. Different biofilm reactors have different applications depending upon their configuration and size. Biofilms grow attached to a range of materials that are incorporated into the reactor, including glass, metal, plastic and ceramics; the surfaces can be continuous, such as in a membrane or tubular reactor, or particles of the biofilm carrier that might be found in a moving or fluidised bed reactor. The nature of both the surface and reactor type depends on the microorganism and the application.
5.2 Biofilms Applied to Fermentations
Microorganisms that are used industrially in the production of valuable compounds and enzymes are most commonly cultivated in batch or fed-batch fermentors as suspended cultures. While this approach is effective, one major drawback is the repeated stopping of the reactor followed by a re-establishment of the culture, resulting in time delays and additional consumption of resources. One potential application of biofilms is in the continuous production of small molecules and enzymes, and several examples of research in this area are given below.
5.2.1 Saccharomyces cerevisiae
This unicellular yeast is widely studied as a eukaryotic model organism for cell and molecular biology and has been instrumental for the production of beverages and biofuels as it can convert sugars to ethanol at a high rate (Dombek and Ingram 1987).
Saccharomyces cerevisiae has been extensively studied for biofilm formation on numerous surfaces like plastic, mat and ore (Reynolds and Fink 2001). To unveil the mechanism of biofilm regulation requires transcriptomic and proteomic analyses of S. cerevisiae, since biofilm enhances fermentation rates and ethanol resistance which has industrial relevance for full-scale production. The switch from planktonic to biofilm growth of S. cerevisiae is regulated by Cyc8p, Tup1p and Flo11. The latter is a surface glycoprotein involved in cell adhesion and Tup1p regulates Cyc8p-mediated FLO11 repression and prevents Flo11p degradation (Van Nguyen et al. 2020). Additionally, a FLO11 deletion mutant abated ethanol tolerance of yeast cells in the biofilm fermentation (Gu et al. 2018). Along with FLO11 the other FLO genes like FLO1, FLO5, FLO9 and FLO10 encode glycoproteins, which are responsible for cell-cell adherence, and their deletion mutants decrease biofilm formation during fermentation (Halme et al. 2004). Another study showed a transcription factor, MIG1 expresses more in S. cerevisiae biofilms than in their planktonic cells and it induces yeast morphology as filamentous growth, which is confirmed primarily under the control of FLO genes (Yang et al. 2018). A low concentration of nitric oxide can also induce S. cerevisiae biofilm growth by activation of a transcriptional factor Mac1p that regulates a transmembrane protein Ctr1 which is independent of FLO11 genetic system. Furthermore, MAC1 and CTR1 contribute to the intracellular tolerance to ethanol (Yang et al. 2019a).
In earlier work, Demirci et al. (1997) used S. cerevisiae ATCC 24859 as biofilms in a continuous and batch-culture biofilm reactor to compare the ethanol production on a polypropylene support (PPS) and plastic composite-supports (PCS; containing soybean hull, flour, yeast extract-salt and polypropylene). Their study showed immobilised yeast on the PCS produces 2–10 times more ethanol (30 g L−1 with low-cost, low-nutrient ammonium sulphate medium) than PPS (5 g L−1), as PCS supplies nutrients to the immobilised cells. In contrast Liu et al. (2015) showed a maximum of 2.90 g L−1 ethanol using suspended cells of S. cerevisiae. More recently, Izmirlioglu and Demirci (2016) optimised growth parameters of S. cerevisiae like pH, temperature and agitation theoretically using Box-Behnken design of response surface methods (RSM) for a similar kind of PCS-biofilm reactor, then reported further improvement in the bioethanol production from potato waste hydrolysate to 37.05 g L−1 at pH 4.2 and incubation temperature of34 °C and agitation of 100 rpm. Kana et al. (1989) used very cheap and abundant mineral ores of kissiris for immobilisation of S. cerevisiae cells and found improved ethanol production (53 g L−1; 115 g L−1 d−1) when immobilised cells inoculated in the glucose-containing media for up to 29 repeated cycles of batch fermentations. Roukas and Kotzekidou (2020) used a rotary biofilm reactor (RBR) to enhance bioethanol production using agro-industrial waste (non-sterilised beet molasses). They achieved an ethanol concentration of 52.3 g L−1 and productivity of 1.45 g L−1 h−1 in a repeated-batch reactor for 60 days with 84.8% energy recovery.
5.2.2 Zymomonas mobilis
This bacterium ferments glucose to ethanol and has potential application in the production of biofuel. Early work by Kunduru and Pometto (1996a, b) demonstrated that biofilms of this bacterium grown on polypropylene support in a packed bed arrangement, continuously operated for 60 days, dramatically outperformed planktonic cultures with respect to ethanol production (536 g L−1 h−1 compared with 5 g L−1 h−1). Li et al. (2006) subsequently demonstrated that Z. mobilis biofilms were more resistant to the toxic effects of benzaldehyde than planktonic cells, retaining 45% metabolic activity after exposure to 50 mM of the solvent, whereas the planktonic cells were inactivated. Interest in Z. mobilis biofilms for the production of ethanol has continued, and researchers have investigated lignocellulose hydrolysates as fermentable substrates on different support surfaces such as DEAE cellulose and corn silk (Todhanakasem et al. 2015; Todhanakasem et al. 2016). Furthermore, it has been shown that the biofilm is more resistant than planktonic cells to toxic compounds that are typically found in the hydrolysates, such as furfural (Todhanakasem et al. 2018; Todhanakasem et al. 2014).
5.2.3 Bacillus subtilis
Bacillus spp. are industrially important bacteria; B. subtilis is particularly relevant owing to its GRAS status and is applied in the production of enzymes and pharmaceutically important compounds (Gu et al. 2018). For example, menaquinone-7 (MK-7), or vitamin K2, is produced industrially by Bacillus subtilis natto in static fermentation, where it forms pellicles (Mahdinia et al. 2019). To investigate if biofilm growth might improve productivity and alleviate problems associated with heat and mass transfer, Mahdinia et al. (2017) developed a biofilm rector using plastic composite supports arranged in a grid formation (Fig. 5.3), but the MK-7 concentration was low compared to the static fermentation in a glycerol-based culture medium, with concentrations of approx. 1 mg L−1 in biofilm compared with over 30 mg L−1 in static fermentations. However, further medium optimisations using a glucose-based medium resulted in improvement of productivity to 20 mg L−1 (Mahdinia et al. 2018).
Bacillus spp. also produce the lipopeptides surfactin, iturin A and fengycin (amongst others) that have significant biological activity. They are also biosurfactants, and their production in suspended cultures can result in foaming, which is detrimental to the fermentation, and one potential solution to this problem is to employ biofilms. Several bioreactor configurations have been investigated to promote biofilm growth and production of lipopeptides, for example, Chtioui et al. (2012) used a rotating disc bioreactor to cultivate B. subtilis ATCC 21332, which produces surfactin and fengycin. In this bioreactor, the discs were partially submerged in the culture medium enabling surface aeration, thus avoiding foam formation and favouring the production of fengycin over surfactin. Using 14 discs and an airflow of 100 L h−1, fengycin productivity reached 838 mg L−1 after 72 h, which was greater than that of planktonic cultures and an improvement on other bubbleless reactors studied up to that point. A further alteration in the design to include agitators to improve oxygen transfer further improved the selectivity of the reactor for fengycin production (Chtioui et al. 2014).
Bruck et al. (2019) investigated the genetic changes required for B. subtilis 168 to grow effectively as a biofilm and produce surfactin. This is a domesticated strain that is widely studied but has key mutations that both prevent surfactin production and impede biofilm growth. These researchers repaired the sfp and epsC genes, which enabled the cells to produce surfactin and EPS; they also deleted sepF, which codes for a protein involved in septation, resulting in cell filamentation. The resulting mutants had much improved biofilm-forming capabilities, which was mainly down to the production of EPS, and the biofilms demonstrated ten-fold enhanced surfactin production compared with strains that could only grow planktonically but had a functional sfp.
The same strain could be manipulated to produce another lipopeptide, iturin A, by horizontal transfer of the itu operon and introduction of the sfp gene plus the pleiotropic regulator degQ (Tsuge et al. 2005). Rahman et al. (2007) investigated this strain for its biofilm-forming capability and found that it formed thick biofilm in a multiwell plate; furthermore, it produced approx. Double the concentration of iturin A of planktonic cultures (~800 mg L−1). Productivity of iturin A in planktonic and biofilm cultures is also impacted by the medium used: Zohora et al. (2013) found that by using a medium containing 12% maltose and 5% fish protein, B. subtilis RB14 biofilms grown in Erlenmeyer flasks could produce over 5 g L−1 of the lipopeptide.
5.2.4 Propionibacteria
Propionibacterium spp. are anaerobic, Gram-positive bacilli that are found in milk, dairy products, skin, soil, plants or digestive tracts of ruminants (Zárate 2012). Probiotics and cheese industries employ Propionibacterium to produce vitamin B12, tetrapyrrole containing compounds and propionic acid (Kiatpapan and Murooka 2002). These bacteria are capable of catabolizing various polyols (like glycerol), carbohydrates (like glucose, trehalose, etc.), lactic acid and pyruvic acid into succinic acid, propionic acid, acetic acid and CO2; however, propionic acid is a major product in the fermentation (Roy 2019). Various species of Propionibacteria (P. freudenreichii, P. acnes, P. acidipropionici, P. acidifaciens, P. namnetense, P. propionicus, etc.) can form a biofilm, so have potential application in large-scale production.
Propionibacteria acnes can cause blood infections in immunocompromised patients (like pneumonia and chronic granulomatous disease) and endocarditis patients and like infections in ventriculoperitoneal shunts, pacemaker devices, prosthetic joints, catheters, etc. (Bayston et al. 2007; Holmberg et al. 2009; Tyner and Patel 2016). Coenye et al. (2008) cultured in vitro biofilms from planktonic P. acnes obtained from skin infections. The biofilms hydrolyzed triglycerides yielding free fatty acids that were subsequently catabolised to short-chain fatty acids (like propionic acid and butyric acid) depending upon the bacterial subtype and free fatty acid length (Linfante et al. 2018).
Propionibacteria freudenreichii, P. acidipropionici and P. arabinosum can immobilise as biofilms and have been exploited for propionic acid and acetic acid production (Xu et al. 2011). Dishisha et al. (2012) used polyethylenimine-treated Poraver® (glass beads) matrix for P. acidipropionici DSM 4900 immobilisation and obtained propionic acid productivity of 0.35 g L−1 h−1 in batch or fed-batch fermentation and 1.40 g L−1 h−1 in continuous mode. In another experiment, a maximum propionic acid productivity of 1.63 g L−1 h−1 was obtained from 90 g L−1 glycerol in sequential batch fermentation (Dishisha et al. 2015). Chen et al. (2013) assembled a plant fibrous-bed bioreactor for immobilisation of P. freudenreichii CCTCC M207015 for effective production of propionic acid. Up to 41.20 ± 2.03 g L−1 propionic acid was obtained from 80 g L−1 glucose in 108 h, which was 21.07% more than that produced in suspended cell fermentation. Furthermore, they optimised the fermentation by a constant fed-batch process which led to 136.23 ± 6.77 g L−1 propionic acid in 108 h. Recently, Cavero-Olguin et al. (2019) used biofilms of P. acidipropionici in recycle batch reactors (Poraver®) for improved fermentation of carbohydrates and glycerol for propionic acid production. Citric acid and NaCl were used to induce bacteria to form biofilm as these increased the biofilm-forming capacity (BFC) index, EPS production and trehalose production with increased expression treY gene. They obtained the highest productivity of 0.7 g L−1 h−1with citric acid and 0.78 g L−1 h−1 with NaCl.
5.2.5 Actinobacillus succinogenes
This rod-shaped, facultatively anaerobic bacterium, isolated from the bovine rumen, produces succinic acid from pentose and hexose sugars and is promising for its wide utilisation of carbon (Jiang et al. 2019; Pateraki et al. 2016). It can also yield other organic acids (e.g. pyruvic and lactic acids) under microaerobic conditions (Li et al. 2010; Wang et al. 2016). Recently, Yang et al. (2019b) reviewed metabolic engineering strategies to improve succinic acid synthesis with reduced accumulation of the by-product, via elimination of the metabolic pathways involved in by-product formation. However, there are many key factors (such as media components, trace substances, neutralizing agents, CO2, redox potential, temperature, pH, stirring) that can affect bacterial growth and significantly improve the succinic acid accumulation without the formation of any by-product. For instance, Zhu et al. (2012) employed Box-Behnken design method of RSM to optimise the fermentation media compositions for the production of maximal succinic acid, and they achieved 52.7 ± 0.8 g L−1.
Actinobacillus succinogenes is well studied for biofilm formation via self-immobilisation to the support surfaces under prolonged operation (Bradfield and Nicol 2014). Actinobacillus succinogenes biofilm has immense potential for improved yield of succinic acid over free-cell fermentations since both cell growth and maintenance rates in free-cell fermentation are drastically decreased with increased succinic acid titre. In contrast to suspended cells in a chemostat, biofilm was capable of achieving higher cell densities and improved succinic acid yield (Brink and Nicol 2014). Mokwatlo and Nicol (2017) characterised the cellular structure and viability of A. succinogenes biofilms as it exhibits a heterogeneous structure: The top layer cells have increased cell viability, and biofilm sessile cells have different morphology compared to planktonic cells and extensive connection fibres. In earlier experiments, Urbance et al. (2003, 2004) evaluated succinic acid production by A. succinogenes in both continuous and repeat-batch in a plastic composite support bioreactor for biofilm formation. As in continuous fermentation, repeat-batch can operate in the biofilm mode as it retained biomass after every batch cycle. More recently, Longanesi et al. (2018) used Glaxstone®, which is a sintered glass, as the support material in a 1 L packed-bed reactor for succinic acid production with grown A. succinogenes biofilms. The attached cells were fed with cheese whey (lactose-rich by-product of cheese processing), and maximum productivity of 0.72 g L−1 h−1 was achieved. Similarly, Ferone et al. (2018) also used a packed-bed biofilm reactor for continuous succinic acid production for over 5 months and evaluated different dilution rates and carbon sources. The bioreactor showed maximum productivity of 43 g L−1 h−1 at a dilution rate of 0.5 h−1 with glucose as the substrate.
5.2.6 Lactobacillus
Lactobacilli are anaerobic (aerotolerant), non-spore-forming, Gram-positive bacteria, present in the human mouth, associated with dental caries, gastrointestinal tract and genital tract of females (Makarova et al. 2006). Currently, the use of Lactobacillus spp. is expanding towards other dairy products such as energy bars, chocolate, juices, shakes, etc. This extensive use of Lactobacillus spp. requires reduction of foodborne pathogenic contaminations and their biofilm formation in the food industries. These bacteria are mostly employed for the production of lactic acid in the food, cosmetics and pharmaceutical industries (Taskila and Ojamo 2013). One other major use of lactic acid is in the production of biodegradable and renewable raw material such as poly lactic acid (PLA) for household applications (Krishna et al. 2018) and in the synthesis of environmentally friendly solvents, such as butyl lactate (Wee et al. 2006).
The biofilms of Lactobacillus are more resistant to acetic acid/vinegar and ethanol (used as a food preservative) than planktonic cultures. Kubota et al. (2008) investigated biofilm formation by 40 different species of lactic acid bacteria on the glass coverslips and found longer cells than in their planktonic cultures. Among these species, L. plantarum M606 have maximum survival percentage in 10% acetic acid and 30% ethanol. In earlier work, Demirci et al. (1993) compared pure- and mixed-cultures of Streptomyces viridosporus T7A and Lactobacillus casei subsp. rhamnosus for biofilm formation and continuous production of lactic acid through fermentation in a biofilm reactor with plastic composite support (PCS) chips. Their work showed two to five times faster lactic acid production in the pure- and mixed-culture bioreactors than those of their suspension culture (control). Similarly, Cotton et al. (2001) designed a biofilm reactor with six PCS tubes composed of agricultural products (50% w/w) and polypropylene (50% w/w) for continuous lactic acid fermentation using Lactobacillus casei subsp. rhamnosus. The study found that biofilm growth on the PCS tubes can be controlled by the speed of agitation and reported the PCS biofilm reactor improved the yield of lactic acid by 70%. The PCS biofilm reactor achieved a 9 g L−1 h−1 optimal average production rate, whereas the control reactor without PCS tubes achieved only 5.8 g L−1 h−1 at 125 rpm agitation and 0.4 per hour dilution rate. Cuny et al. (2019) investigated a strain of L. delbrueckii that was a high producer of lactic acid, for its ability to form biofilms in a tubular reactor and reported higher cell densities and productivity of 9 g L−1 h−1. The biofilm could be maintained for more than 2 weeks.
5.2.7 Aspergillus niger
Lignocellulolytic enzymes are of intense interest for the digestion of lignocellulose biomass, which is comprised of lignin, cellulose and hemicellulose, to simple sugars that can be used in the production of biofuel. Aspergillus niger elaborates a number of these enzymes and their activities have been investigated in biofilm cultures of the fungus. Villena and Gutierrez-Correa (2006) cultivated A. niger biofilms on polyester cloth with lactose as a growth substrate, either in flasks or in microbioreactors, showed that the biofilms produced up to three times more cellulase than the free mycelium cultures. Similarly, when biofilm was grown on perlite, the production of cellulase, endoglucanase and xylanase was greater than that in either submerged culture or solid-state fermentation (Gamarra et al. 2010). Comparison of cellulase gene expression in A. niger grown as biofilm and submerged fermentation demonstrated a higher level of expression in biofilm, which is reflective of the enzyme activity measurements (Mahmoud et al. 2016). Izmirlioglu and Demirci (2017) established a dual biofilm with A. niger and S. cerevisiae to couple saccharification and fermentation, generating bioethanol directly from food waste. Biofilms were established on plastic composite support and under optimised conditions almost 38 g L−1 ethanol was produced after 72 h.
A. niger is also known for the industrial production of citric acid for the food and pharmaceutical industries. The current method involves submerged fermentation, but efforts have been made to investigate the possibility of using biofilms to enable continuous production of this important organic acid. Earlier work examined the A. niger biofilms growing in rotating biological contactors, for example, Wang (2000) used polyurethane foam-covered discs to grow the fungus and observed citric acid productivity of 0.896 g L−1 h−1, compared with 0.33 g L−1 h−1 in submerged culture. More recently, Yu et al. (2018) employed a porous foam carrier, comprised of polyurethane and carbon black, in repeated fed-batch fermentation. Stable citric acid production was achieved over 600 h, with 8 fed-batch cycles, yielding 2.26 g L−1 h−1.
5.3 Biofilms in Biotransformation
Biocatalysis describes the use of an enzyme or whole cell to afford the biotransformation of a substrate to a valuable intermediate or product and is increasingly important in the synthesis of fine chemicals, since the reactions are more environmentally-friendly than those of classical organic chemistry. Industrially important chemicals such as acrylamide, synthons such as R-2-chloropropionic acid and drugs such as L-DOPA rely on biocatalysis for their production (Murphy 2012); increasing applications are inevitable as manufacturing processes require sustainability. Bacterial biofilms have been studied for their potential use in biocatalytic processes since they typically have a longer effective half-life compared with suspended cells, have a higher tolerance to the often unnatural substrates that are required, can be easily reused and require no separation step from the product.
5.3.1 Pseudomonas taiwanensis VLB120ΔC
The metabolic diversity in Pseudomonas spp. has resulted in these bacteria being used in numerous biocatalytic applications. Pseudomonas taiwanensis VLB120ΔC biofilms have been extensively investigated for the continuous production of valuable compounds in a variety of bioreactors. The bacterium was originally identified as a potentially useful biofilm biocatalyst following a screen of strains, including those isolated from biofilters and contaminated soils, in a well-plate assay (Gross et al. 2007). As it demonstrated comparatively good biofilm growth and had been already investigated for its ability to stereospecifically transform styrene to (S)-styrene oxide, it was selected for further investigation in a tubular bioreactor. The biofilm was established on the inner wall of the silicone tubing that was partially submerged in liquid styrene; the substrate diffused into the biofilm and was biotransformed continuously over a 55-day period. As with Z. mobilis, exposure to the solvent (styrene) impacted planktonic cells more than biofilm cells, which rapidly adapted to solvent exposure by producing more extracellular polymeric substances (Halan et al. 2011). Further experiments demonstrated that oxygen was a major limiting factor of styrene oxide productivity in the biofilm, which was affected by membrane area and tube wall thickness (Gross et al. 2010); biofilm growth on microporous ceramic material was shown to enable efficient oxygen transfer (Halan et al. 2010), demonstrating the impact of the support material when cultivating biofilms. Additional iteration of the bioreactor design employed an aqueous-air segmented flow arrangement (Fig. 5.4) (Karande et al. 2014), whereby the biofilm was allowed to develop in a tube in single-phase flow for 72 h; air segments were introduced, which caused biofilm detachment, then regrowth of a stable second generation biofilm. When the biofilm was exposed to styrene, a volumetric productivity of (S)-styrene oxide of 46 g Ltube−1 day−1 was achieved, which was 21-fold greater than the initial single-phase flow used in the original experiments. Further improvement of the biofilm biocatalyst was achieved by using a mutant strain of the bacterium, designated VLB120ΔCeGFP 04710, in which a gene responsible for the degradation of the second messenger c-di-GMP was deleted (Schmutzler et al. 2017). This strain adhered more strongly owing to higher EPS production and increased hydrophobicity, resulting in higher cell numbers in the initial phase.
Schematic set-up of the aqueous-air segmented flow biofilm reactor. (1) Medium reservoir; (2) 1.5 mm silicone tubing; (3) peristaltic pump; (4) silicone tubing as growth surface (2 mm inner diameter and 2000 mm long); (5) styrene phase; (6) magnified sketch of aqueous-air segments describing the mass transfer scheme of substrate and products in the reactor. S—styrene; P—(S)-styrene oxide. (Copyright © 2014 WILEY_VCH Verlag GmbH & Co. KgaA, Weinheim. Used with permission from Rohan Karande et al., “Segmented flow is controlling growth of catalytic biofilms in continuous multiphase microreactors”, Biotechnology and Bioengineering, John Wiley and Sons)
In addition to the biotransformation of styrene, VLB120 has been metabolically engineered to produce other important chemicals, either via biotransformation or from the growth substrate/carbon source. For example, Lang et al. (2015) engineered the bacterium to produce (S)-3-hydroxybutyric acid fermentatively from glucose by eliminating 3-hydroxyisobutyric acid dehydrogenase activity through random mutagenesis, heterologously expressing kivD from Lactococcus lactis, which codes for 2-ketoacid dehydrogenase, and homologously expressing genes in the pathway for 2-ketoisovalerate synthesis. The bacterium was engineered to express other enzymes to expand the capabilities of the biofilm in the continuous production of other important chemicals such as n-octanol (Gross et al. 2013).
Finally, the strain was employed in a dual-species biofilm alongside the cyanobacterium Synechocystis sp. PCC6803 to alleviate oxidative stress and allow the high cell density growth of the photoautotroph (Hoschek et al. 2019). Both strains expressed a cyclohexane monooxygenase, and the biofilm was applied in the production of cyclohexanol and was active for at least 1 month.
5.3.2 Escherichia coli PHL644
This strain of E. coli is a mutant strain of a K-12 strain (MC4100) that overproduces curli adhesin, which enables it to readily form biofilm. Tsoligkas et al. (2011) engineered the strain to express a tryptophan synthetase from Salmonella enterica sv Typhimurium so that it could biocatalytically produce 5-halotryptophans from 5-haloindoles. After growing the bacterium as a biofilm on poly-L-lysine-coated glass slides, using a spin-coating method to improve biofilm growth, the biotransformation of halotryptophans was compared with that in free cells and immobilised enzyme, and found to be superior. Furthermore, the biofilm lost little activity through three 12 h biocatalytic cycles. The longevity of the enzyme activity was explored by Tong et al. (2016) who used stable isotopic labelled amino acids in cell cultures (SILAC) to follow the fate of the recombinant tryptophan synthetase. Surprisingly, the reason for the extended enzyme activity was not due to the stabilising influence of the extracellular matrix, as might have been expected, but because the enzyme was continually replenished.
5.3.3 Acinetobacter ST-550
The biocatalytic production of the dye indigo from indole has been the subject of numerous investigations but is complicated by the toxicity of the substrate. As has been noted already, one advantageous characteristic of biofilms is their improved resistance to the toxic effects of certain compounds. Acinetobacter ST-550 is a bacterium originally isolated from soil and demonstrated efficient indigo production in organic solvent (Doukyu et al. 2002). However, the bacterium does not readily form biofilms under conventional conditions, so they would not be a candidate for such investigations. However, Ishikawa et al. (2014) employed a novel approach to improve the biofilm-forming characteristics of ST-550 by expressing the gene coding for the adhesin AtaA from another Acinetobacter strain (tol 5). This protein makes the tol 5 strain extremely sticky and enables it to adhere to numerous abiotic surfaces (Furuichi et al. 2018). The transformed ST-550 was subsequently able to adhere to polyurethane and the resulting biofilm displayed an improved tolerance for the starting material and catalysed the production of indigo at a rate that was five-fold faster than planktonic cells. The strategy of expressing the ataA gene in bacteria already known to catalyse industrially important reactions could speed up the development of biofilm reactors for continuous processes. Indeed, the same approach was taken with the hydrogen-producing bacterium Enterobacter aerogenes (Nakatani et al. 2018), which was immobilised on polyurethane foam and could continually produce hydrogen from glucose. Most recently, a truncated AtaA was employed to adhere an artificial cell (liposome) to polystyrene and glass (Noba et al. 2019); the liposomes also had β-glucuronidase encapsulated, the activity of which could be measured after adherence. Such artificial biofilms might be useful in circumstances where genetically engineered organisms are prohibited.
5.3.4 Pseudomonas diminuta
Glycolic acid can be used as skincare agent in pharmaceutical products, as a preservative and flavouring agent in food processing, as a dyeing/tanning agent in textile processing and as an additive in adhesives, polymers, paints, plastics, inks, etc. (Hua et al. 2018). Pseudomonas diminuta (or Brevundimonas diminuta) can biotransform ethylene glycol to glycolic acid without the formation of by-products (Li et al. 2007); thus, it is of industrial interest. Furthermore, it can grow efficiently as a biofilm and the ability of the bacterium to continually produce glycolic acid has been examined by Li et al. (2013). These researchers evaluated glycolic acid production in a trickle-bed biofilm reactor fitted with Montz-pak A3–500 stainless steel structured packing for improved catalyst stability (up to 60 days) and efficient gas-liquid exchange. A steady state productivity was achieved up to 1.6 g L−1 h−1 which compares favourably to that of planktonic cultures, for example, Kataoka et al. (2001) examined glycolic acid production in planktonic cultures of different organisms and observed up to 0.92 g L−1 h−1.
5.3.5 Cunninghamella elegans
The recognition that filamentous fungi can grow as biofilms is relatively recent. Harding et al. (2009) proposed criteria for defining these biofilms, which are based on differences in structural features (complex aggregated growth of hyphae, surface-associated growth, production of extracellular matrix) and physiology (enhanced tolerance to biocides, changes in enzyme and metabolite production). One fungus capable of biofilm growth is the zygomycete Cunninghamella elegans. This fungus is a model of mammalian drug metabolism, and many examples exist in the literature reporting the production of mammalian-equivalent phase I (oxidative) and phase II (conjugative) metabolites upon incubation of the fungus with different drugs (Asha and Vidyavathi 2009). Recently, the complement of cytochromes P450, which are responsible for the oxidation of drugs, were identified in the fungus (Palmer-Brown et al. 2019a). Most studies focus on using planktonically-grown cells to biotransform xenobiotics, but Amadio et al. (2013) and Mitra et al. (2013) described how the fungus can be grown as a biofilm, either on a spring included in an Erlenmeyer flask (Fig. 5.5) or on polymethylmethacrylate. Under these conditions, biotransformation reactions are enhanced; for example, Mitra et al. (2013) observed that fluoranthrene biotransformation was enhanced by 22-fold in biofilm compared with planktonic cultures. Amadio et al. (2013) demonstrated the convenient reusability of the biofilm by using it to catalyse multiple rounds of biotransformation of the drug flurbiprofen. Furthermore, the biotransformations could be performed in water rather than culture medium, and by shortening the incubation time with the drug and introducing rejuvenation steps with a fresh culture medium, Quinn et al. (2015) improved the productivity of phase I metabolites in biofilms from 1 mg L−1 h−1 to over 4 mg l−1 h−1.
C. elegans grown as biofilm in an Erlenmeyer flask with a spring included. The spring must be touching the glass for the biofilm to properly form. (Reprinted by permission from Springer: Applied Microbiology and Biotechnology (Filamentous fungal biofilm for production of human drug metabolites, Amadio et al.), © 2013)
5.4 Biofilms in Biodegradation
For several decades, bacteria and fungi have been studied for their ability to biotransform a wide range of xenobiotics. They are able to do this since many xenobiotics are similar in structure to naturally occurring compounds and microbial enzymes have a relaxed substrate specificity. For example, some Pseudomonas bacteria have evolved pathways to catabolise biphenyl, which occurs naturally from the pyrolysis of lignin; the same bacteria are able to biotransform anthropogenic polychlorinated biphenyl (Field and Sierra-Alvarez 2008). Since microbes are environmentally benign, this gives them an advantage over chemical and physical approaches to remediation of contaminated sites (bioremediation) and waste streams. The advantages that immobilisation conveys are particularly relevant to biodegradation, and in this section, examples of how biofilms can remove pollutants from toxic waste streams are presented.
5.4.1 Pseudomonas knackmussii B13
Many drugs and xenobiotics contain fluorine, which makes them particularly resistant to biodegradation owing to the strength of the carbon-fluorine bond. P. knackmussii was studied several decades ago for its ability to biodegrade fluorobenzoate. Schreiber et al. (1980) demonstrated that the bacterium could use 4-fluorobenzoate as a sole carbon and energy source, yielding fluoride ion. This characteristic was later exploited by Misiak et al. (2011) who grew B13 in a membrane-aerated biofilm reactor (MABR, Fig. 5.6) for up to 600 h. Two growth phases were observed: an initial period of fast growth up to 200 h, followed by a period of much slower growth. By measuring the concentration of fluoride ion and 4-fluorobenzoate concentration in the effluent it was clear that approx. 15% of the available fluorine remained in the biomass. Subsequent experiments using a tubular bioreactor showed that fluoride ion accumulated in the biofilm and inhibited growth. These experiments revealed that while biofilms can be advantageous for the continuous degradation of persistent chemicals, the toxic intermediate may accumulate and impact on performance.
Schematic diagram of the MABR used to grow P. knackmussii B13 biofilm on 4-fluorobenzoate. The biofilm grew on the outer surface of the silicone tubing. (Reprinted from Water Research, Volume 45, Misiak et al., Factors influencing 4-fluorobenzoate degradation in biofilm cultures of Pseudomonas knackmussii B13, Pages 3512–3520, Copyright (2011), with permission from Elsevier)
5.4.2 Pseudomonas stutzeri T102
This bacterium was isolated from the sludge in an oil tank in Okinawa, Japan and degrades PAHs including naphthalene (Hirano et al. 2004) and was selected for further investigation by Shimada et al. (2012) as it readily formed biofilms on a number of surfaces. These researchers compared naphthalene degradation in biofilm and planktonic cultures of the bacterium and found that although initially degradation was faster in planktonic cultures, after 4 h the biofilm culture degraded naphthalene quicker. Sloughing of cells was noted in the biofilm cultures, which is common, and Shimada and colleagues found that the expression of nahAc, which codes for the large subunit of naphthalene dioxygenase, was much higher in these cells than in planktonic cells. Thus, it was concluded that the rapid degradation of naphthalene in biofilms was due to the detached cells rather than the cells remaining in the biofilm.
5.4.3 Cunninghamella elegans
In addition to the application of the fungal biofilm to biocatalytic production of drug metabolites, its usefulness in the biodegradation of pollutants has been investigated. Hussain et al. (2017) investigated the bioremediation of water containing the dye malachite green and the metal Cr (VI). An earlier study had shown that C. elegans could degrade malachite green via leucomalachite green and N-demethylated and N-oxidised products (Cha et al. 2001). The biofilm rapidly (15 min) absorbed the dye from the water, which was followed by a slower decolourisation of the biomass; the biofilm could be used repeatedly for dye decolourisation. Furthermore, when Cr(VI) was also included in the water, this was simultaneously removed, even in the presence of high salt concentrations, demonstrating that the biofilm was robust and could be applied to the removal of several pollutants under harsh environmental conditions.
Planktonically-grown C. elegans is able to biotransform agrochemicals such as the insecticides fenitrothion and diazinon (Zhao et al. 2020; Zhu et al. 2017), and the fungicide mepaniprim (Zhu et al. 2010). Palmer-Brown et al. (2019b) compared planktonic and biofilm cultures of the fungus in the biodegradation of the pyrethroid insecticide cyhalothrin and found that similar to malachite green, the compound was initially biosorbed in the biofilm before being more slowly degraded. Interestingly, planktonic cells were more effective at degrading the pesticide as only 6% of the initial material remaining after 120 h, whereas, in the biofilm, 22% of the starting substrate remained after the same incubation period. Repeated additions of pesticides were not investigated in this study.
5.4.4 Bacillus subtilis N4
Acetonitrile is a toxic chemical that is biotransformed to hydrogen cyanide and acetaldehyde. It can be degraded by some microorganisms, such as Pseudomonas aeroginousa, Mesorhizobium sp. and Rhodococcus rhodochrous (Feng and Lee 2009; Chapatwala et al. 1990; Li et al. 2012), via the actions of nitrile hydratase and amidase. The genes coding for these enzymes in R. rhodochrous BX2 were cloned into B. subtilis N4, which grows well as biofilm. The strain was grown on amine-modified polypropylene carriers, which were added to a movable bed biofilm reactor (MBBR) that had been seeded with activated sludge (Li et al. 2018). The biofilm was able to withstand repeated additions of synthetic wastewater containing acetonitrile and community analysis of the biofilm reactor revealed that the recombinant strain was the dominant organism after 50 days’ operation, demonstrating the applicability of the organism to a wastewater treatment system.
5.5 Future Prospects
The research on productive biofilms is limited compared with the efforts that have been made to investigate other methods of immobilisation. Nevertheless, the studies consistently demonstrate that compared with suspended cultures, biofilms are more robust, are resistant to toxic effects, are more productive and are stable over much longer timeframes. However, there are obvious challenges to applying biofilms at larger scales, notably sloughing, which is difficult to predict and control and might lead to blockages in tubing or contamination of the product. Additionally, the biofilm bioreactors are quite specialised; thus, considerable investment would be required from industry if they were to be adapted. Nevertheless, there is sufficient evidence that research on biofilms that might be applied in industry should be accelerated.
In relation to biodegradation, although single-species biofilms might play an important role in the decolourisation of dyes (Cerron et al. 2015; Malachova et al. 2013; Mawad et al. 2016; Munck et al. 2018; Novotny et al. 2012), the structural complexity of many xenobiotics means that a single species is unlikely to have the catabolic capacity to fully degrade a particular pollutant. Thus, mixed-species biofilms are more useful in this regard. For instance, the degradation of isomeric dinitrotoluene (DNT), i.e. 2,4-dinitrotoluene (2,4-DNT) and 2,6-dinitrotoluene (2,6-DNT), was studied by Lendenmann et al. (1998) using mixed culture bacteria that are capable of growing on these isomers as the sole carbon and nitrogen source. In an aerobic fluidised-bed biofilm reactor, the mixed culture had a degradation efficiency of 98% for 2,4-DNT and 94% for 2,6-DNT. Similarly, Oh and Tuovinen (1994) studied biodegradation of the phenoxy herbicides methylchlorophenoxypropionic acid (MCPP) and 2,4-dichlorophenoxyacetic acid (2,4-D) using a mixed culture of herbicide-degrading bacterial biofilm in fixed-film column reactors and found overall efficiency of degradation of MCPP was partial and 2,4-D was complete. A major problem with current waste-water treatment facilities is that pharmaceuticals and personal care products are not effectively removed and consequently are released back into waterways where they are a potential hazard. Bioaugmentation of mixed-species biofilms that are found in wastewater treatment plants with strains developed for the biodegradation of specific xenobiotics, such as that described in Sect. 5.4.4, is therefore an important strategy.
References
Amadio J, Casey E, Murphy CD (2013) Filamentous fungal biofilm for production of human drug metabolites. Appl Microbiol Biotechnol 97(13):5955–5963. https://doi.org/10.1007/s00253-013-4833-x
Asha S, Vidyavathi M (2009) Cunninghamella – a microbial model for drug metabolism studies – a review. Biotechnol Adv 27(1):16–29. https://doi.org/10.1016/j.biotechadv.2008.07.005
Azeredo J, Azevedo NF, Briandet R, Cerca N, Coenye T, Costa AR, Desvaux M, Di Bonaventura G, Hébraud M, Jaglic Z, Kačániová M, Knøchel S, Lourenço A, Mergulhão F, Meyer RL, Nychas G, Simões M, Tresse O, Sternberg C (2017) Critical review on biofilm methods. Crit Rev Microbiol 43(3):313–351. https://doi.org/10.1080/1040841X.2016.1208146
Bayston R, Ashraf W, Barker-Davies R, Tucker E, Clement R, Clayton J, Freeman BJC, Nuradeen B (2007) Biofilm formation by Propionibacterium acnes on biomaterials in vitro and in vivo: impact on diagnosis and treatment. J Biomed Mater Res A 81A(3):705–709. https://doi.org/10.1002/jbm.a.31145
Bradfield MFA, Nicol W (2014) Continuous succinic acid production by Actinobacillus succinogenes in a biofilm reactor: steady-state metabolic flux variation. Biochem Eng J 85:1–7
Brink HG, Nicol W (2014) Succinic acid production with Actinobacillus succinogenes: rate and yield analysis of chemostat and biofilm cultures. Microb Cell Factories 13(1):111
Bruck HL, Delvigne F, Dhulster P, Jacques P, Coutte F (2019) Molecular strategies for adapting Bacillus subtilis 168 biosurfactant production to biofilm cultivation mode. Bioresour Technol 293. https://doi.org/10.1016/j.biortech.2019.122090
Cavero-Olguin VH, Hatti-Kaul R, Cardenas-Alegria OV, Gutierrez-Valverde M, Alfaro-Flores A, Romero-Calle DX, Alvarez-Aliaga MT (2019) Stress induced biofilm formation in Propionibacterium acidipropionici and use in propionic acid production. World J Microbiol Biotechnol 35(7):101
Cerron LM, Romero-Suarez D, Vera N, Ludena Y, Villena GK, Gutierrez-Correa M (2015) Decolorization of textile reactive dyes and effluents by biofilms of Trametes polyzona LMB-TM5 and Ceriporia sp LMB-TM1 isolated from the Peruvian rainforest. Water Air and Soil Pollution 226(8). https://doi.org/10.1007/s11270-015-2505-4
Cha CJ, Doerge DR, Cerniglia CE (2001) Biotransformation of malachite green by the fungus Cunninghamella elegans. Appl Environ Microbiol 67(9):4358–4360. https://doi.org/10.1128/aem.67.9.4358-4360.2001
Chapatwala KD, Nawaz MS, Richardson JD, Wolfram JH (1990) Isolation and characterization of acetonitrile utilizing bacteria. J Ind Microbiol 5(2–3):65–70. https://doi.org/10.1007/bf01573854
Chen F, Feng X, Xu H, Zhang D, Ouyang P (2013) Propionic acid production in a plant fibrous-bed bioreactor with immobilized Propionibacterium freudenreichii CCTCC M207015. J Biotechnol 164(2):202–210
Chtioui O, Dimitrov K, Gancel F, Dhulster P, Nikov I (2012) Rotating discs bioreactor, a new tool for lipopeptides production. Process Biochem 47(12):2020–2024. https://doi.org/10.1016/j.procbio.2012.07.013
Chtioui O, Dimitrov K, Gancel F, Dhulster P, Nikov I (2014) Selective fengycin production in a modified rotating discs bioreactor. Bioprocess Biosyst Eng 37(2):107–114. https://doi.org/10.1007/s00449-013-0964-9
Coenye T, Honraet K, Rossel B, Nelis H (2008) Biofilms in skin infections: Propionibacterium acnes and acne vulgaris. Infect Disord Drug Targets 8(3):156–159
Cotton JC, Pometto AL, Gvozdenovic-Jeremic J (2001) Continuous lactic acid fermentation using a plastic composite support biofilm reactor. Appl Microbiol Biotechnol 57(5–6):626–630
Cuny L, Pfaff D, Luther J, Ranzinger F, Ödman P, Gescher J, Guthausen G, Horn H, Hille-Reichel A (2019) Evaluation of productive biofilms for continuous lactic acid production. Biotechnol Bioeng 116(10):2687–2697. https://doi.org/10.1002/bit.27080
Demirci A, Pometto AL, Johnson KE (1993) Lactic-acid production in a mixed-culture biofilm reactor. Appl Environ Microbiol 59(1):203–207. https://doi.org/10.1128/aem.59.1.203-207.1993
Demirci A, Pometto A III, Ho KG (1997) Ethanol production by Saccharomyces cerevisiae in biofilm reactors. J Ind Microbiol Biotechnol 19(4):299–304
Dishisha T, Alvarez MT, Hatti-Kaul R (2012) Batch-and continuous propionic acid production from glycerol using free and immobilized cells of Propionibacterium acidipropionici. Bioresour Technol 118:553–562
Dishisha T, Ibrahim MH, Cavero VH, Alvarez MT, Hatti-Kaul R (2015) Improved propionic acid production from glycerol: combining cyclic batch-and sequential batch fermentations with optimal nutrient composition. Bioresour Technol 176:80–87
Dombek K, Ingram L (1987) Ethanol production during batch fermentation with Saccharomyces cerevisiae: changes in glycolytic enzymes and internal pH. Appl Environ Microbiol 53(6):1286–1291
Doukyu N, Nakano T, Okuyama Y, Aono R (2002) Isolation of an Acinetobacter sp. ST-550 which produces a high level of indigo in a water-organic solvent two-phase system containing high levels of indole. Appl Microbiol Biotechnol 58(4):543–546. https://doi.org/10.1007/s00253-001-0919-y
Es I, Vieira JDG, Amaral AC (2015) Principles, techniques, and applications of biocatalyst immobilization for industrial application. Appl Microbiol Biotechnol 99(5):2065–2082. https://doi.org/10.1007/s00253-015-6390-y
Feng Y-S, Lee C-M (2009) The potential of the acetonitrile biodegradation by Mesorhizobium sp F28. J Hazard Mater 164(2–3):646–650. https://doi.org/10.1016/j.jhazmat.2008.08.039
Ferone M, Raganati F, Ercole A, Olivieri G, Salatino P, Marzocchella A (2018) Continuous succinic acid fermentation by Actinobacillus succinogenes in a packed-bed biofilm reactor. Biotechnol Biofuels 11(1):1–11
Field JA, Sierra-Alvarez R (2008) Microbial transformation and degradation of polychlorinated biphenyls. Environ Pollut 155(1):1–12. https://doi.org/10.1016/j.envpol.2007.10.016
Furuichi Y, Iwasaki K, Hori K (2018) Cell behavior of the highly sticky bacterium Acinetobacter sp Tol 5 during adhesion in laminar flows. Sci Rep 8. https://doi.org/10.1038/s41598-018-26699-5
Gamarra NN, Villena GK, Gutierrez-Correa M (2010) Cellulase production by Aspergillus niger in biofilm, solid-state, and submerged fermentations. Appl Microbiol Biotechnol 87(2):545–551. https://doi.org/10.1007/s00253-010-2540-4
Garzón-Posse F, Becerra-Figueroa L, Hernández-Arias J, Gamba-Sánchez D (2018) Whole cells as biocatalysts in organic transformations. Molecules 23(6). https://doi.org/10.3390/molecules23061265
Gross R, Hauer B, Otto K, Schmid A (2007) Microbial biofilms: new catalysts for maximizing productivity of long-term biotransformations. Biotechnol Bioeng 98(6):1123–1134. https://doi.org/10.1002/bit.21547
Gross R, Lang K, Buhler K, Schmid A (2010) Characterization of a biofilm membrane reactor and its prospects for fine chemical synthesis. Biotechnol Bioeng 105(4):705–717. https://doi.org/10.1002/bit.22584
Gross R, Buehler K, Schmid A (2013) Engineered catalytic biofilms for continuous large scale production of n-octanol and (S)-styrene oxide. Biotechnol Bioeng 110(2):424–436. https://doi.org/10.1002/bit.24629
Gu Y, Xu X, Wu Y, Niu T, Liu Y, Li J, Du G, Liu L (2018) Advances and prospects of Bacillus subtilis cellular factories: from rational design to industrial applications. Metab Eng 50:109–121. https://doi.org/10.1016/j.ymben.2018.05.006
Halan B, Schmid A, Buchler K (2010) Maximizing the productivity of catalytic biofilms on solid supports in membrane aerated reactors. Biotechnol Bioeng 106(4):516–527. https://doi.org/10.1002/bit.22732
Halan B, Schmid A, Buehler K (2011) Real-time solvent tolerance analysis of Pseudomonas sp strain LB120 Delta C catalytic biofilms. Appl Environ Microbiol 77(5):1563–1571. https://doi.org/10.1128/aem.02498-10
Halme A, Bumgarner S, Styles C, Fink GR (2004) Genetic and epigenetic regulation of the FLO gene family generates cell-surface variation in yeast. Cell 116(3):405–415
Harding MW, Marques LLR, Howard RJ, Olson ME (2009) Can filamentous fungi form biofilms? Trends Microbiol 17(11):475–480. https://doi.org/10.1016/j.tim.2009.08.007
Hirano SI, Kitauchi F, Haruki M, Imanaka T, Morikawa M, Kanaya S (2004) Isolation and characterization of Xanthobacter polyaromaticivorans sp nov 127W that degrades polycyclic and heterocyclic aromatic compounds under extremely low oxygen conditions. Biosci Biotech Bioch 68(3):557–564. https://doi.org/10.1271/bbb.68.557
Holmberg A, Lood R, Morgelin M, Soderquist B, Holst E, Collin M, Christensson B, Rasmussen M (2009) Biofilm formation by Propionibacterium acnes is a characteristic of invasive isolates. Clin Microbiol Infect 15(8):787–795. https://doi.org/10.1111/j.1469-0691.2009.02747.x
Hoschek A, Heuschkel I, Schmid A, Buhler B, Karande R, Buhler K (2019) Mixed-species biofilms for high-cell-density application of Synechocystis sp. PCC 6803 in capillary reactors for continuous cyclohexane oxidation to cyclohexanol. Bioresour Technol 282:171–178. https://doi.org/10.1016/j.biortech.2019.02.093
Hua X, Cao R, Zhou X, Xu Y (2018) Integrated process for scalable bioproduction of glycolic acid from cell catalysis of ethylene glycol. Bioresour Technol 268:402–407
Hussain S, Quinn L, Li JJ, Casey E, Murphy CD (2017) Simultaneous removal of malachite green and hexavalent chromium by Cunninghamella elegans biofilm in a semi-continuous system. Int Biodeterior Biodegradation 125:142–149. https://doi.org/10.1016/j.ibiod.2017.09.003
Ishikawa M, Shigemori K, Hori K (2014) Application of the adhesive bacterionanofiber ataa to a novel microbial immobilization method for the production of indigo as a model chemical. Biotechnol Bioeng 111 (1):16–24. doi:https://doi.org/10.1002/bit.25012
Izmirlioglu G, Demirci A (2016) Ethanol production in biofilm reactors from potato waste hydrolysate and optimization of growth parameters for Saccharomyces cerevisiae. Fuel 181:643–651
Izmirlioglu G, Demirci A (2017) Simultaneous saccharification and fermentation of ethanol from potato waste by co-cultures of Aspergillus niger and Saccharomyces cerevisiae in biofilm reactors. Fuel 202:260–270. https://doi.org/10.1016/j.fuel.2017.04.047
Jiang M, Zhang W, Yang Q, Wu M, Liu H, Zhou J, Dong W, Ma J, Xin F (2019) Metabolic regulation of organic acid biosynthesis in Actinobacillus succinogenes. Front Bioeng Biotechnol 7:216
Kana K, Kanellaki M, Psarianos C, Koutinas A (1989) Ethanol production by Saccharomyces cerevisiae immobilized on mineral kissiris. J Ferment Bioeng 68(2):144–147
Karande R, Halan B, Schmid A, Buehler K (2014) Segmented flow is controlling growth of catalytic biofilms in continuous multiphase microreactors. Biotechnol Bioeng 111(9):1831–1840. https://doi.org/10.1002/bit.25256
Kataoka M, Sasaki M, Hidalgo A, Nakano M, Shimizu S (2001) Glycolic acid production using ethylene glycol-oxidizing microorganisms. Biosci Biotech Bioch 65(10):2265–2270. https://doi.org/10.1271/bbb.65.2265
Kiatpapan P, Murooka Y (2002) Genetic manipulation system in propionibacteria. J Biosci Bioeng 93(1):1–8
Krishna BS, Sarva G, Nikhilesh S, Tarun B, Saibaba K, Narayana GR (2018) Industrial production of lactic acid and its applications. Int J Biotech Res 1(1):42–54
Kubota H, Senda S, Nomura N, Tokuda H, Uchiyama H (2008) Biofilm formation by lactic acid bacteria and resistance to environmental stress. J Biosci Bioeng 106(4):381–386. https://doi.org/10.1263/jbb.106.381
Kunduru MR, Pometto AL (1996a) Continuous ethanol production by Zymomonas mobilis and Saccharomyces cerevisiae in biofilm reactors. J Ind Microbiol 16(4):249–256. https://doi.org/10.1007/bf01570029
Kunduru MR, Pometto AL (1996b) Evaluation of plastic composite-supports for enhanced ethanol production in biofilm reactors. J Ind Microbiol 16(4):241–248. https://doi.org/10.1007/bf01570028
Lang K, Buehler K, Schmid A (2015) Multistep synthesis of (S)-3-Hydroxyisobutyric acid from glucose using Pseudomonas taiwanensis VLB120 B83 T7 catalytic biofilms. Adv Synthes Catal 357(8):1919–1927. https://doi.org/10.1002/adsc.201500205
Lendenmann U, Spain JC, Smets BF (1998) Simultaneous biodegradation of 2, 4-dinitrotoluene and 2, 6-dinitrotoluene in an aerobic fluidized-bed biofilm reactor. Environ Sci Technol 32(1):82–87
Li XZ, Webb JS, Kjelleberg S, Rosche B (2006) Enhanced benzaldehyde tolerance in Zymomonas mobilis biofilms and the potential of biofilm applications in fine-chemical production. Appl Environ Microbiol 72(2):1639–1644. https://doi.org/10.1128/aem.72.2.1639-1644.2006
Li XZ, Hauer B, Rosche B (2007) Single-species microbial biofilm screening for industrial applications. Appl Microbiol Biotechnol 76(6):1255–1262
Li Q, Wang D, Song Z, Zhou W, Wu Y, Xing J, Su Z (2010) Dual-phase fermentation enables Actinobacillus succinogenes 130ZT to be a potential role for high-level lactate production from the bioresource. Bioresour Technol 101(19):7665–7667
Li Y, Xiong M, Cheng X, Sun J, Zang H, Pan J, Li C (2012) Identification and biological characterization of an acetonitrile degrading strain. Weishengwu Xuebao 52(2):250–255
Li XZ, Hauer B, Rosche B (2013) Catalytic biolfilms on structured packing for the production of glycolic acid. J Microbiol Biotechnol 23:195–204
Li CY, Sun YL, Yue ZL, Huang MY, Wang JM, Chen X, An XJ, Zang HL, Li DP, Hou N (2018) Combination of a recombinant bacterium with organonitrile-degrading and biofilm-forming capability and a positively charged carrier for organonitriles removal. J Hazard Mater 353:372–380. https://doi.org/10.1016/j.jhazmat.2018.03.058
Linfante A, Allawh R, Allen H (2018) The role of Propionibacterium acnes biofilm in acne vulgaris. J Clin Exp Dermatol Res 9:1–4
Liu Z, Inokuma K, Ho SH, den Haan R, Hasunuma T, van Zyl WH, Kondo A (2015) Combined cell-surface display- and secretion-based strategies for production of cellulosic ethanol with Saccharomyces cerevisiae. Biotechnol Biofuels 8. https://doi.org/10.1186/s13068-015-0344-6
Longanesi L, Frascari D, Spagni C, DeWever H, Pinelli D (2018) Succinic acid production from cheese whey by biofilms of Actinobacillus succinogenes: packed bed bioreactor tests. J Chem Technol Biotechnol 93(1):246–256
Mahdinia E, Demirci A, Berenjian A (2017) Strain and plastic composite support (PCS) selection for vitamin K (Menaquinone-7) production in biofilm reactors. Bioprocess Biosyst Eng 40(10):1507–1517. https://doi.org/10.1007/s00449-017-1807-x
Mahdinia E, Demirci A, Berenjian A (2018) Enhanced vitamin K (Menaquinone-7) production by Bacillus subtilis natto in biofilm reactors by optimization of glucose-based medium. Curr Pharm Biotechnol 19(11):917–924. https://doi.org/10.2174/1389201020666181126120401
Mahdinia E, Demirci A, Berenjian A (2019) Biofilm reactors as a promising method for vitamin K (menaquinone-7) production. Appl Microbiol Biotechnol 103(14):5583–5592. https://doi.org/10.1007/s00253-019-09913-w
Mahmoud MA, Al-Othman MR, Abd-El-Aziz ARM, Metwaly HA, Mohamed HA (2016) Expression of genes encoding cellulolytic enzymes in some Aspergillus species. Genet Mol Res 15(4). https://doi.org/10.4238/gmr15048913
Makarova K, Slesarev A, Wolf Y, Sorokin A, Mirkin B, Koonin E, Pavlov A, Pavlova N, Karamychev V, Polouchine N (2006) Comparative genomics of the lactic acid bacteria. Proc Natl Acad Sci 103(42):15611–15616
Malachova K, Rybkova Z, Sezimova H, Cerven J, Novotny C (2013) Biodegradation and detoxification potential of rotating biological contactor (RBC) with Irpex lacteus for remediation of dye-containing wastewater. Water Res 47(19):7143–7148. https://doi.org/10.1016/j.watres.2013.07.050
Mawad AMM, Yousef NMH, Shoreit AAM (2016) Robust Aspergillus terreus biofilm supported on graphene oxide/hematitenanocomposites for adsorption of anthraquinone dye. Desalin Water Treat 57(1):24341–24351. https://doi.org/10.1080/19443994.2016.1138885
Misiak K, Casey E, Murphy CD (2011) Factors influencing 4-fluorobenzoate degradation in biofilm cultures of Pseudomonas knackmussii B13. Water Res 45(11):3512–3520. https://doi.org/10.1016/j.watres.2011.04.020
Mitra S, Pramanik A, Banerjee S, Haldar S, Gachhui R, Mukherjee J (2013) Enhanced biotransformation of fluoranthene by intertidally derived Cunninghamella elegans under biofilm-based and niche-mimicking conditions. Appl Environ Microbiol 79(24):7922–7930. https://doi.org/10.1128/aem.02129-13
Mokwatlo SC, Nicol W (2017) Structure and cell viability analysis of Actinobacillus succinogenes biofilms as biocatalysts for succinic acid production. Biochem Eng J 128:134–140
Mrozik A, Piotrowska-Seget Z (2010) Bioaugmentation as a strategy for cleaning up of soils contaminated with aromatic compounds. Microbiol Res 165(5):363–375. https://doi.org/10.1016/j.micres.2009.08.001
Munck C, Thierry E, Grassle S, Chen SH, Ting ASY (2018) Biofilm formation of filamentous fungi Coriolopsis sp on simple muslin cloth to enhance removal of triphenylmethane dyes. J Environ Manag 214:261–266. https://doi.org/10.1016/j.jenvman.2018.03.025
Murphy CD (2012) The microbial cell factory. Org Biomol Chem 10(10):1949–1957. https://doi.org/10.1039/c2ob06903b
Nakatani H, Ding N, Ohara Y, Hori K (2018) Immobilization of Enterobacter aerogenes by a trimeric autotransporter adhesin, AtaA, and its application to biohydrogen production. Catalysts 8(4). https://doi.org/10.3390/catal8040159
Noba K, Ishikawa M, Uyeda A, Watanabe T, Hohsaka T, Yoshimoto S, Matsuura T, Hori K (2019) Bottom-up creation of an artificial cell covered with the adhesive bacterionanofiber protein AtaA. J Am Chem Soc 141(48):19058–19066. https://doi.org/10.1021/jacs.9b09340
Novotny C, Trost N, Susla M, Svobodova K, Mikeskova H, Valkova H, Malachova K, Pavko A (2012) The use of the fungus Dichomitus squalens for degradation in rotating biological contactor conditions. Bioresour Technol 114:241–246. https://doi.org/10.1016/j.biortech.2012.03.080
Oh KH, Tuovinen OH (1994) Microbiological treatment of fertilizer solid waste material containing phenoxyalkanoic herbicides 2, 4-D and MCPP. J Chem Technol Biotechnol 61(4):299–305
Palmer-Brown W, Miranda-CasoLuengo R, Wolfe KH, Byrne KP, Murphy CD (2019a) The CYPome of the model xenobiotic-biotransforming fungus Cunninghamella elegans. Sci Rep 9. https://doi.org/10.1038/s41598-019-45706-x
Palmer-Brown W, Souza PLD, Murphy CD (2019b) Cyhalothrin biodegradation in Cunninghamella elegans. Environ Sci Pollut Res 26(2):1414–1421. https://doi.org/10.1007/s11356-018-3689-0
Pateraki C, Patsalou M, Vlysidis A, Kopsahelis N, Webb C, Koutinas AA, Koutinas M (2016) Actinobacillus succinogenes: advances on succinic acid production and prospects for development of integrated biorefineries. Biochem Eng J 112:285–303
Quinn L, Dempsey R, Casey E, Kane A, Murphy CD (2015) Production of drug metabolites by immobilised Cunninghamella elegans: from screening to scale up. J Ind Microbiol Biotechnol 42(5):799–806. https://doi.org/10.1007/s10295-015-1594-9
Rahman MS, Ano T, Shoda M (2007) Biofilm fermentation of iturin a by a recombinant strain of Bacillus subtilis 168. J Biotechnol 127(3):503–507. https://doi.org/10.1016/j.jbiotec.2006.07.013
Reynolds TB, Fink GR (2001) Bakers' yeast, a model for fungal biofilm formation. Science 291(5505):878–881
Roukas T, Kotzekidou P (2020) Rotary biofilm reactor: a new tool for long-term bioethanol production from non-sterilized beet molasses by Saccharomyces cerevisiae in repeated-batch fermentation. J Clean Prod 257:120519
Roy D (2019) Probiotics. In: Moo-Young M (ed) Comprehensive biotechnology, 2nd edn. Academic Press, pp 591–602
Schmutzler K, Kupitz K, Schmid A, Buehler K (2017) Hyperadherence of Pseudomonas taiwanensis VLB120C increases productivity of (S)-styrene oxide formation. Microb Biotechnol 10(4):735–744. https://doi.org/10.1111/1751-7915.12378
Schreiber A, Hellwig M, Dorn E, Reineke W, Knackmuss HJ (1980) Critical reactions in fluorobenzoic acid degradation by Pseudomonas sp B-13. Appl Environ Microbiol 39(1):58–67. https://doi.org/10.1128/aem.39.1.58-67.1980
Shimada K, Itoh Y, Washio K, Morikawa M (2012) Efficacy of forming biofilms by naphthalene degrading Pseudomonas stutzeri T102 toward bioremediation technology and its molecular mechanisms. Chemosphere 87(3):226–233. https://doi.org/10.1016/j.chemosphere.2011.12.078
Taskila S, Ojamo H (2013) The current status and future expectations in industrial production of lactic acid by lactic acid bacteria. In: Kongo M (ed) Lactic acid bacteria-R & D for food, health and livestock purposes. IntechOpen. https://doi.org/10.5772/51282
Todhanakasem T, Sangsutthiseree A, Areerat K, Young GM, Thanonkeo P (2014) Biofilm production by Zymomonas mobilis enhances ethanol production and tolerance to toxic inhibitors from rice bran hydrolysate. New Biotechnol 31(5):451–459. https://doi.org/10.1016/j.nbt.2014.06.002
Todhanakasem T, Narkmit T, Areerat K, Thanonkeo P (2015) Fermentation of rice bran hydrolysate to ethanol using Zymomonas mobilis biofilm immobilization on DEAE-cellulose. Electron J Biotechnol 18(3):196–201. https://doi.org/10.1016/j.ejbt.2015.03.007
Todhanakasem T, Tiwari R, Thanonkeo P (2016) Development of corn silk as a biocarrier for Zymomonas mobilis biofilms in ethanol production from rice straw. J Gen Appl Microbiol 62(2):68–74. https://doi.org/10.2323/jgam.62.68
Todhanakasem T, Yodsanga S, Sowatad A, Kanokratana P, Thanonkeo P, Champreda V (2018) Inhibition analysis of inhibitors derived from lignocellulose pretreatment on the metabolic activity of Zymomonas mobilis biofilm and planktonic cells and the proteomic responses. Biotechnol Bioeng 115(1):70–81. https://doi.org/10.1002/bit.26449
Tong XX, Barberi TT, Botting CH, Sharma SV, Simmons MJH, Overton TW, Goss RJM (2016) Rapid enzyme regeneration results in the striking catalytic longevity of an engineered, single species, biocatalytic biofilm. Microb Cell Factories 15. https://doi.org/10.1186/s12934-016-0579-3
Tsoligkas AN, Winn M, Bowen J, Overton TW, Simmons MJH, Goss RJM (2011) Engineering biofilms for biocatalysis. Chembiochem 12(9):1391–1395. https://doi.org/10.1002/cbic.201100200
Tsuge K, Inoue S, Ano T, Itaya M, Shoda M (2005) Horizontal transfer of iturin a operon, Itu, to Bacillus subtilis 168 and conversion into an iturin a producer. Antimicrob Agents Chemother 49(11):4641–4648. https://doi.org/10.1128/aac.49.11.4641-4648.2005
Tyner H, Patel R (2016) Propionibacterium acnes biofilm A sanctuary for Staphylococcus aureus? Anaerobe 40:63–67. https://doi.org/10.1016/j.anaerobe.2016.05.014
Urbance SE, Pometto AL III, DiSpirito AA, Demirci A (2003) Medium evaluation and plastic composite support ingredient selection for biofilm formation and succinic acid production by Actinobacillus succinogenes. Food Biotechnol 17(1):53–65
Urbance SE, Pometto AL, DiSpirito AA, Denli Y (2004) Evaluation of succinic acid continuous and repeat-batch biofilm fermentation by Actinobacillus succinogenes using plastic composite support bioreactors. Appl Microbiol Biotechnol 65(6):664–670
van Beilen JB, Duetz WA, Schmid A, Witholt B (2003) Practical issues in the application of oxygenases. Trends Biotechnol 21(4):170–177. https://doi.org/10.1016/s0167-7799(03)00032-5
Van Nguyen P, Plocek V, Váchová L, Palková Z (2020) Glucose, Cyc8p and Tup1p regulate biofilm formation and dispersal in wild Saccharomyces cerevisiae. NPJ Biofilms Microbiomes 6(1):1–10
Villena GK, Gutierrez-Correa M (2006) Production of cellulase by Aspergillus niger biofilms developed on polyester cloth. Lett Appl Microbiol 43(3):262–268. https://doi.org/10.1111/j.1472-765X.2006.01960.x
Wang JL (2000) Production of citric acid by immobilized Aspergillus niger using a rotating biological contactor (RBC). Bioresour Technol 75(3):245–247
Wang Z, Xiao W, Zhang A, Ying H, Chen K, Ouyang P (2016) Potential industrial application of Actinobacillus succinogenes NJ113 for pyruvic acid production by microaerobic fermentation. Korean J Chem Eng 33(10):2908–2914
Wee YJ, Kim JN, Ryu HW (2006) Biotechnological production of lactic acid and its recent applications. Food Technol Biotechnol 44(2):163–172
Xu Z, Shi Z, Jiang L (2011) Acetic and propionic acids. In: Comprehensive biotechnology 2nd edition (M Moo-Young) Academic Press.189-199
Yang L, Zheng C, Chen Y, Ying H (2018) FLO genes family and transcription factor MIG1 regulate Saccharomyces cerevisiae biofilm formation during immobilized fermentation. Front Microbiol 9:1860
Yang L, Zheng C, Chen Y, Shi X, Ying Z, Ying H (2019a) Nitric oxide increases biofilm formation in Saccharomyces cerevisiae by activating the transcriptional factor Mac1p and thereby regulating the transmembrane protein Ctr1. Biotechnol Biofuels 12(1):30
Yang Q, Wu M, Dai Z, Xin F, Zhou J, Dong W, Ma J, Jiang M, Zhang W (2019b) Comprehensive investigation of succinic acid production by Actinobacillus succinogenes: a promising native succinic acid producer. Biofuels Bioprod Biorefining
Yu B, Zhang X, Sun WJ, Xi X, Zhao N, Huang ZC, Ying ZJ, Liu L, Liu D, Niu HQ, Wu JL, Zhuang W, Zhu CJ, Chen Y, Ying HJ (2018) Continuous citric acid production in repeated-fed batch fermentation by Aspergillus niger immobilized on a new porous foam. J Biotechnol 276:1–9. https://doi.org/10.1016/j.jbiotec.2018.03.015
Zárate G (2012) Dairy Propionibacteria: less conventional probiotics to improve the human and animal health. Prob Anim:153–202
Zhao MA, Gu H, Zhang CJ, Jeong H, Kim EOH, Zhu YZ (2020) Metabolism of insecticide diazinon by Cunninghamella elegans ATCC36112. RSC Adv 10(33):19659–19668. https://doi.org/10.1039/d0ra02253e
Zhu Y-Z, Keum YS, Yang L, Lee H, Park H, Kim J-H (2010) Metabolism of a fungicide mepanipyrim by soil fungus Cunninghamella elegans ATCC36112. J Agric Food Chem 58(23):12379–12384. https://doi.org/10.1021/jf102980y
Zhu L-W, Wang C-C, Liu R-S, Li H-M, Wan D-J, Tang Y-J (2012) Actinobacillus succinogenes ATCC 55618 fermentation medium optimization for the production of succinic acid by response surface methodology. J Biomed Biotechnol
Zhu Y-Z, Fu M, Jeong I-H, Kim J-H, Zhang C-J (2017) Metabolism of an insecticide Fenitrothion by Cunninghamella elegans ATCC36112. J Agric Food Chem 65(49):10711–10718. https://doi.org/10.1021/acs.jafc.7b04273
Zohora US, Rahman MS, Khan AW, Okanami M, Ano T (2013) Improvement of production of lipopeptide antibiotic iturin a using fish protein. J Environ Sci (China) 25(Suppl 1):S2–S7. https://doi.org/10.1016/s1001-0742(14)60616-1
Acknowledgement
One of the authors (MFK) is a recipient of a Government of Ireland Postdoctoral Fellowship (IRC/GOI-PD/1064).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Khan, M.F., Murphy, C.D. (2022). Application of Microbial Biofilms in Biocatalysis and Biodegradation. In: Mulla, S.I., Bharagava, R.N. (eds) Enzymes for Pollutant Degradation . Microorganisms for Sustainability, vol 30. Springer, Singapore. https://doi.org/10.1007/978-981-16-4574-7_5
Download citation
DOI: https://doi.org/10.1007/978-981-16-4574-7_5
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-16-4573-0
Online ISBN: 978-981-16-4574-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)