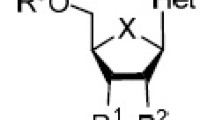Abstract
One of the most common classes of drugs is nucleoside analogues, which have long served as a cornerstone for antiviral, antiparasitic, and anticancer treatments. This is due to their close resemblance to the naturally occurring nucleosides found in many biological processes. In terms of antiviral drug design, typically viral proteins or cellular proteins are targeted. Each has its pros and cons; however, both carry the risk of the development of drug resistance, making it imperative that novel and more effective antivirals are developed. This chapter will focus on a specific class of shaped-modified nucleoside analogues called fleximers. These flexible nucleoside analogues possess a split purine ring system, which endows flexibility to the nucleobase scaffold. This flexibility has been shown to be beneficial for increased antiviral activity but also results in broad-spectrum antiviral activity as well as the potential to overcome point mutations related to viral drug resistance. For over two decades now, the fleximer technology has been applied to numerous nucleoside analogues and has led to potent, broad-spectrum activity against a wide array of viruses including flaviviruses, filoviruses, and coronaviruses, among others. Their history and development, the various synthetic routes to realize them, and some of the biological data obtained to date for the fleximers will be discussed within this chapter.
Similar content being viewed by others
References
Andersson J, Sköldenberg B, Ernberg I, Britton S, Henle W, Andersson U (1985) Acyclovir treatment in primary Epstein-Barr virus infection. A double-blind placebo-controlled study. Scand J Infect Dis Suppl 47:107–115
Barkholt L, Lewensohn-Fuchs I, Ericzon BG, Tydén G, Andersson J (1999) High-dose acyclovir prophylaxis reduces cytomegalovirus disease in liver transplant patients. Transpl Infect Dis 1(2):89–97
Chiang PK (1998) Biological effects of inhibitors of S-adenosylhomocysteine hydrolase. Pharmacol Ther 77(2):115–134
Chudinov MV (2020) Nucleoside Analogs with Fleximer nucleobase. Chem Heterocycl Compd (NY, US) 56(6):636–643
Clercq ED, Holý A (2005) Acyclic nucleoside phosphonates: a key class of antiviral drugs. Nat Rev Drug Discov 4(11):928–940
De Clercq E (1994) Antiviral activity Spectrum and target of action of different classes of nucleoside analogues. Nucleosides Nucleotides 13(6–7):1271–1295
De Clercq E (2002) Strategies in the design of antiviral drugs. Natl Rev Drug Discov 1(1):13–25
De Clercq E (2005a) Antiviral drug discovery and development: where chemistry meets with biomedicine. Antivir Res 67(2):56–75
De Clercq E (2005b) Recent highlights in the development of new antiviral drugs. Curr Opin Microbiol 8(5):552–560
De Clercq E, Neyts J (2009) Antiviral agents acting as DNA or RNA chain terminators. Handb Exp Pharmacol 189:53–84
De Winter H, Herdewijn P (1996) Understanding the binding of 5-substituted 2′-deoxyuridine substrates to thymidine kinase of herpes simplex virus type-1. J Med Chem 22(39(24)):4727–4737
Deval J (2009) Antimicrobial strategies: inhibition of viral polymerases by 3′-hydroxyl nucleosides. Drugs 69(2):151–166
Deval J, Powdrill MH, D'Abramo CM, Cellai L, Götte M (2007) Pyrophosphorolytic excision of nonobligate chain terminators by hepatitis C virus NS5B polymerase. Antimicrob Agents Chemother 51(8):2920–2928
Docea A, Tsatsakis A, Albulescu D, Cristea O, Zlatian O, Vinceti M, Moschos S, Tsoukalas D, Goumenou M, Drakoulis N, Dumanov J, Tutelyan V, Onischenko G, Aschner M, Spandidos D, Calina D (2020) A new threat from an old enemy: re-emergence of coronavirus (review). Int J Mol Med 45
Fischer E (1894) Einfluss der Configuration auf die Wirkung der Enzyme. Ber Dtsch Chem Ges 27(3):2985–2993
Greco NJ, Tor Y (2005) Simple fluorescent pyrimidine analogues detect the presence of DNA Abasic sites. J Am Chem Soc 127(31):10784–10785
Khandazhinskaya A, Eletskaya B, Fateev I, Kharitonova M, Konstantinova I, Barai V, Azhayev A, Hyvonen MT, Keinanen TA, Kochetkov S, Seley-Radtke K, Khomutov A, Matyugina E (2021) Novel fleximer pyrazole-containing adenosine analogues: chemical, enzymatic and highly efficient biotechnological synthesis. Org Biomol Chem 19
Koshland DE (1958) Application of a theory of enzyme specificity to protein synthesis. Proc Natl Acad Sci U S A 44(2):98–104
Ku T, Lopresti N, Shirley M, Mori M, Marchant J, Heng X, Botta M, Summers MF, Seley-Radtke KL (2019) Synthesis of distal and proximal fleximer base analogues and evaluation in the nucleocapsid protein of HIV-1. Bioorg Med Chem 27(13):2883–2892
Leonard NJ, Hiremath SP (1986) Dimensional probes of binding and activity. Tetrahedron 42(7):1917–1961
Leonard NJ, Morrice AG, Sprecker MA (1975) Linear benzoadenine. Stretched-out analog of adenine. J Org Chem 40(3):356–363
Mattelaer H-P, Van Hool A-S, de Jong F, Van der Auweraer M, Van Meervelt L, Dehaen W, Herdewijn P (2020) New metal-free route towards imidazole substituted uridine. Eur J Org Chem 2020
Matyugina ES, Khandazhinskaya AL, Kochetkov SN, Seley-Radtke KL (2020) Synthesis of 3-hetarylpyrroles by Suzuki-Miyaura cross-coupling. Mendeleev Commun 30(2):231–232
McGuigan C, Cahard D, Sheeka HM, De Clercq E, Balzarini J (1996) Aryl phosphoramidate derivatives of d4T have improved anti-HIV efficacy in tissue culture and may act by the generation of a novel intracellular metabolite. J Med Chem 39(8):1748–1753
McGuigan C, Hassan-Abdallah A, Srinivasan S, Wang Y, Siddiqui A, Daluge SM, Gudmundsson KS, Zhou H, McLean EW, Peckham JP, Burnette TC, Marr H, Hazen R, Condreay LD, Johnson L, Balzarini J (2006) Application of phosphoramidate ProTide technology significantly improves antiviral potency of carbocyclic adenosine derivatives. J Med Chem 49(24):7215–7226
Mcguigan C, Madela K, Aljarah M, Gilles A, Brancale A, Zonta N, Chamberlain S, Vernachio J, Hutchins J, Hall A, Ames B, Gorovits E, Ganguly B, Kolykhalov A, Wang J, Muhammad J, Patti JM, Henson G (2010) Design, synthesis and evaluation of a novel double pro-drug: INX-08189. A new clinical candidate for hepatitis C virus. Bioorg Med Chem Lett 20(16):4850–4854
Mehellou Y, Rattan HS, Balzarini J (2018) The ProTide prodrug technology: from the concept to the clinic. J Med Chem 61(6):2211–2226
Mori M, Dietrich U, Manetti F, Botta M (2010) Molecular dynamics and DFT study on HIV-1 Nucleocapsid Protein-7 in complex with viral genome. J Chem Inf Model 50(4):638–650
Mori M, Manetti F, Botta M (2011) Predicting the binding mode of known NCp7 inhibitors to facilitate the Design of Novel Modulators. J Chem Inf Model 51(2):446–454
Morrice AG, Sprecker MA, Leonard NJ (1975) The angular benzoadenines. 9-Aminoimidazo[4,5-f]quinazoline and 6-aminoimidazo [4,5-h]quinazoline. J Org Chem 40(3):363–366
Najjar A, Karaman R (2019) The prodrug approach in the era of drug design. Expert Opin Drug Deliv 16(1):1–5
Nguyen Van T, Hospital A, Lionne C, Jordheim LP, Dumontet C, Périgaud C, Chaloin L, Peyrottes S (2016) Beta-hydroxyphosphonate ribonucleoside analogues derived from 4-substituted-1,2,3-triazoles as IMP/GMP mimics: synthesis and biological evaluation. Beilstein J Organ Chem 12:1476–1486
OʼBrien JJ, Campoli-Richards DM (1989) Acyclovir. Drugs 37(3):233–309
Ouellette R, Rawn JD (2018) Nucleosides, nucleotides, and nucleic acid. Organic chemistry: Structure, mechanism, synthesis. Academic Press, London, pp 973–1000
Périgaud C, Gosselin G, Imbach JL (1992) Nucleoside analogues as chemotherapeutic agents: a review. Nucleosides Nucleotides 11(2–4):903–945
Peters, H. L. (2015). The design, synthesis, and biological evaluation of a series of acyclic Fleximer nucleoside antivirals. Ph.D, University of Maryland, Baltimore County
Peters HL, Jochmans D, de Wilde AH, Posthuma CC, Snijder EJ, Neyts J, Seley-Radtke KL (2015) Design, synthesis and evaluation of a series of acyclic fleximer nucleoside analogues with anti-coronavirus activity. Bioorg Med Chem Lett 25(15):2923–2926
Pierson TC, Diamond MS (2020) The continued threat of emerging flaviviruses. Nat Microbiol 5(6):796–812
Polak M, Seley KL, Plavec J (2004) Conformational properties of shape modified nucleosides - Fleximers. J Am Chem Soc 126(26):8159–8166
Quirk S, Seley KL (2005) Identification of catalytic amino acids in the human GTP fucose pyrophosphorylase active site. Biochemistry 44(39):13172–13178
Ross BS, Ganapati Reddy P, Zhang H-R, Rachakonda S, Sofia MJ (2011) Synthesis of Diastereomerically pure nucleotide Phosphoramidates. J Org Chem 76(20):8311–8319
Sadler JM, Ojewoye O, Seley-Radtke KL (2008) "reverse fleximers": introduction of a series of 5-substituted carbocyclic uridine analogues. Nucleic Acids Symp Ser (Oxf) (52):571–572
Seley KL, Zhang L, Hagos A (2001) "Fleximers". Design and synthesis of two novel Split nucleosides. Org Lett 3(20):3209–3210
Seley KL, Zhang L, Hagos A, Quirk S (2002) "Fleximers". Design and synthesis of a new class of novel shape-modified nucleosides. J Org Chem 67(10):3365–3373
Seley KL, Quirk S, Salim S, Zhang L, Hagos A (2003) Unexpected inhibition of S-adenosyl-L-homocysteine hydrolase by a guanosine nucleoside. Bioorg Med Chem Lett 13(12):1985–1988
Seley KL, Salim S, Zhang L (2005) "molecular chameleons". Design and synthesis of C-4-substituted imidazole Fleximers. Org Lett 7(1):63–66
Seley-Radtke K (2018) Flexibility-not just for yoga anymore! Antivir Chem Chemother 26:2040206618756788
Seley-Radtke KL, Sunkara NK (2009) Carbocyclic thymidine analogues for use as potential therapeutic agents. Nucleosides Nucleotides Nucleic Acids 28(5–7):633–641
Seley-Radtke KL, Yates MK (2018) The evolution of nucleoside analogue antivirals: a review for chemists and non-chemists. Part 1: early structural modifications to the nucleoside scaffold. Antivir Res 154:66–86
Slusarczyk M, Serpi M, Pertusati F (2018) Phosphoramidates and phosphonamidates (ProTides) with antiviral activity. Antivir Chem Chemother 26:204020661877524
St. Amant AH, Bean LA, Guthrie JP, Hudson RHE (2012) Click fleximers: a modular approach to purine base-expanded ribonucleoside analogs. Org Biomol Chem 10(32):6521–6525
Tan ELC, Ooi EE, Lin C-Y, Tan HC, Ling AE, Lim B, Stanton LW (2004) Inhibition of SARS coronavirus infection in vitro with clinically approved antiviral drugs. Emerg Infect Dis 10(4):581–586
Thames JE, Waters CD, Valle C, Bassetto M, Aouadi W, Martin B, Selisko B, Falat A, Coutard B, Brancale A, Canard B, Decroly E, Seley-Radtke KL (2020) Synthesis and biological evaluation of novel flexible nucleoside analogues that inhibit flavivirus replication in vitro. Bioorg Med Chem 28(22):115713
Tripathi A, Bankaitis V (2017) Molecular docking: from lock and key to combination lock. J Mol Med Clin Appl 2(1)
Vichier-Guerre S, Dugué L, Pochet S (2014) A convenient synthesis of 4(5)-(hetero)aryl-1H-imidazoles via microwave-assisted Suzuki–Miyaura cross-coupling reaction. 55(46):6347–6350
Vichier-Guerre S, Ku TC, Pochet S, Seley-Radtke KL (2020) An expedient synthesis of flexible nucleosides through enzymatic glycosylation of proximal and distal Fleximer bases. Chembiochem 21(10):1412–1417
Wauchope OR, Velasquez M, Seley-Radtke K (2012) Synthetic routes to a series of proximal and distal 2′-deoxy fleximers. Synthesis 44(22):3496–3504
Wigerinck P, Pannecouque C, Snoeck R, Claes P, De Clercq E, Herdewijn P (1991) 5-(5-Bromothien-2-yl)-2′-deoxyuridine and 5-(5-chlorothien-2-yl)-2′-deoxyuridine are equipotent to (E)-5-(2-bromovinyl)-2′-deoxyuridine in the inhibition of herpes simplex virus type I replication. J Med Chem 34(8):2383–2389
Yates MK, Seley-Radtke KL (2019) The evolution of antiviral nucleoside analogues: a review for chemists and non-chemists. Part II: complex modifications to the nucleoside scaffold. Antivir Res 162:5–21
Yates MK, Raje MR, Chatterjee P, Spiropoulou CF, Bavari S, Flint M, Soloveva V, Seley-Radtke KL (2017) Flex-nucleoside analogues - novel therapeutics against filoviruses. Bioorg Med Chem Lett 27(12):2800–2802
Yates MK, Chatterjee P, Flint M, Arefeayne Y, Makuc D, Plavec J, Spiropoulou CF, Seley-Radtke KL (2019) Probing the effects of pyrimidine functional group switches on acyclic fleximer analogues for antiviral activity. Molecules 24(17):3184
Yin D, Yang X, Hu Y, Kuczera K, Schowen RL, Borchardt RT, Squier TC (2000) Substrate binding stabilizes S-adenosylhomocysteine hydrolase in a closed conformation. Biochemistry 39(32):9811–9818
Zimmermann SC, Sadler JM, Andrei G, Snoeck R, Balzarini J, Seley-Radtke KL (2011) Carbocyclic 5′-nor "reverse" fleximers. Design, synthesis, and preliminary biological activity. Medchemcomm 2(7)
Zimmermann SC, Sadler JM, O'Daniel PI, Kim NT, Seley-Radtke KL (2013) “Reverse” carbocyclic Fleximers: synthesis of a new class of adenosine deaminase inhibitors. Nucleosides Nucleotides Nucleic Acids 32(3):137–154
Zimmermann SC, O'Neill E, Ebiloma GU, Wallace LJM, De Koning HP, Seley-Radtke KL (2014) Design and synthesis of a series of truncated neplanocin fleximers. Molecules 19(12):21200–21214. 21215 pp
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Section Editor information
Rights and permissions
Copyright information
© 2022 Springer Nature Singapore Pte Ltd.
About this entry
Cite this entry
Seley-Radtke, K.L., Kutz, C.H.M., Thames, J.E. (2022). Flex-Nucleosides – A Strategic Approach to Antiviral Therapeutics. In: Sugimoto, N. (eds) Handbook of Chemical Biology of Nucleic Acids. Springer, Singapore. https://doi.org/10.1007/978-981-16-1313-5_67-1
Download citation
DOI: https://doi.org/10.1007/978-981-16-1313-5_67-1
Received:
Accepted:
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-16-1313-5
Online ISBN: 978-981-16-1313-5
eBook Packages: Springer Reference Chemistry and Mat. ScienceReference Module Physical and Materials ScienceReference Module Chemistry, Materials and Physics