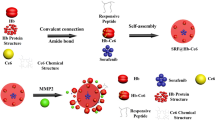
Abstract
Surgery, chemotherapy, and radiotherapy are the commonly used therapeutic interventions for cancerous tumors but they often cause deleterious side effects to the normal cells surrounding the tumor site leading to poor prognosis. In the past two decades, photodynamic therapy has emerged as one of the most investigated techniques for treating cancerous tumors due to minimum invasiveness, cancer cell selectivity, and high therapeutic effect. In photodynamic therapy, a photosensitizer is excited with a light source, resulting in a photochemical reaction within the cell’s microenvironment that generates cytotoxic free radicals. However, the overall therapeutic efficacy of photodynamic therapy depends on several factors such as tumor location and microenvironment, photosensitizer molecule, and wavelength and intensity of the activation light. Most of the photosensitizers are highly hydrophobic which often leads to aggregation in an aqueous environment resulting in decreased singlet oxygen quantum yield. By using nanomaterials as delivery agents, photosensitizers can be delivered at the target site with high load and increased aqueous solubility leading to increased therapeutic efficacy. In this chapter, we review different types of nanomaterials as delivery agents for photosensitizers. We also summarize the application of nanomaterials as down-converting and up-converting photosensitizers, and their advantages over conventional photosensitizers.
Similar content being viewed by others
References
(2001) Verteporfin therapy of subfoveal choroidal neovascularization in age-related macular degeneration: two-year results of a randomized clinical trial including lesions with occult with no classic choroidal neovascularization—verteporfin in photodynamic thera. Am J Ophthalmol 131:541–560
Applerot G et al (2009) Enhanced antibacterial activity of Nanocrystalline ZnO due to increased ROS-mediated cell injury. Adv Funct Mater 19:842–852
Arroyo-Maya IJ, McClements DJ (2015) Biopolymer nanoparticles as potential delivery systems for anthocyanins: fabrication and properties. Food Res Int 69:1–8
Baskaran R, Lee J, Yang S-G (2018) Clinical development of photodynamic agents and therapeutic applications. Biomater Res 22:25
Boyer J-C, Vetrone F, Cuccia LA, Capobianco JA (2006) Synthesis of colloidal upconverting NaYF4 nanocrystals doped with Er3+, Yb3+ and Tm3+, Yb3+ via thermal decomposition of lanthanide trifluoroacetate precursors. J Am Chem Soc 128:7444–7445
Bruns OT et al (2017) Next-generation in vivo optical imaging with short-wave infrared quantum dots. Nat Biomed Eng 1:56
Chatterjee DK, Fong LS, Zhang Y (2008) Nanoparticles in photodynamic therapy: an emerging paradigm. Adv Drug Deliv Rev 60:1627–1637
Chen Y (2009) Zhan die. (Hai tian chu ban she)
Chen J et al (2007) Using the singlet oxygen scavenging property of carotenoid in photodynamic molecular beacons to minimize photodamage to non-targeted cells. Photochem Photobiol Sci 6:1311–1317
Chen Y et al (2017) Vitamin E succinate-grafted-chitosan oligosaccharide/RGD-conjugated TPGS mixed micelles loaded with paclitaxel for U87MG tumor therapy. Mol Pharm 14:1190–1203
Chilakamarthi U, Giribabu L (2017) Photodynamic therapy: past, present and future. Chem Rec 17:775–802
Derycke ASL, De Witte PAM (2004) Liposomes for photodynamic therapy. Adv Drug Deliv Rev 56:17–30
Dougherty TJ et al (1998) Photodynamic therapy. JNCI J Natl Cancer Inst 90:889–905
Elzoghby AO, Samy WM, Elgindy NA (2012) Albumin-based nanoparticles as potential controlled release drug delivery systems. J Control Release 157:168–182
Fan J, Hu M, Zhan P, Peng X (2013) Energy transfer cassettes based on organic fluorophores: construction and applications in ratiometric sensing. Chem Soc Rev 42:29–43
Fang Y-P, Tsai Y-H, Wu P-C, Huang Y-B (2008) Comparison of 5-aminolevulinic acid-encapsulated liposome versus ethosome for skin delivery for photodynamic therapy. Int J Pharm 356:144–152
Foox M, Zilberman M (2015) Drug delivery from gelatin-based systems. Expert Opin Drug Deliv 12:1547–1563
Gomer CJ (1991) Preclinical examination of first and second generation photosensitizers used in photodynamic therapy. Photochem Photobiol 54:1093–1107
Gopi S, Balakrishnan P (2021) Liposomal nanostructures: properties and applications. in Nanoscale Processing. Elsevier. pp. 163–179
Gradishar WJ (2006) Albumin-bound paclitaxel: a next-generation taxane. Expert Opin Pharmacother 7:1041–1053
Hamblin MR (2020) Photodynamic therapy for cancer: What’s past is prologue. Photochem Photobiol 96:506–516
Hejmadi M (2014) Introduction to cancer biology. Book
Hlapisi N, Motaung TE, Linganiso LZ, Oluwafemi OS, Songca SP (2019) Encapsulation of gold nanorods with porphyrins for the potential treatment of cancer and bacterial diseases: A critical review. Bioinorg Chem Appl 2019
Huang Y et al (2018) Probing the interactions of phthalocyanine-based photosensitizers with model phospholipid bilayer by molecular dynamics simulations. J Porphyr Phthalocyanines 22:764–770
Idris NM et al (2012) In vivo photodynamic therapy using upconversion nanoparticles as remote-controlled nanotransducers. Nat Med 18:1580
Jeong H et al (2011) Photosensitizer-conjugated human serum albumin nanoparticles for effective photodynamic therapy. Theranostics 1:230–239
Jing X et al (2019) Role of hypoxia in cancer therapy by regulating the tumor microenvironment. Mol Cancer 18:157
Jones RN (2016) Development of folate directed, protein based photodynamic therapy agents. Loyola University Chicago
Josefsen LB, Boyle RW (2008) Photodynamic therapy and the development of metal-based photosensitisers. Met Based Drugs 2008:276109
Kessel D (1986) Porphyrin-lipoprotein association as a factor in porphyrin localization. Cancer Lett 33:183–188
Khadria A et al (2017) Push-pull pyropheophorbides for nonlinear optical imaging. Org Biomol Chem 15:947–956
Kim MM, Darafsheh A (2020) Light sources and dosimetry techniques for photodynamic therapy. Photochem Photobiol 96:280–294
Kirar S, Thakur NS, Laha JK, Banerjee UC (2019) Porphyrin functionalized gelatin nanoparticle-based biodegradable phototheranostics: potential tools for antimicrobial photodynamic therapy. ACS Appl Bio Mater 2:4202–4212
Kitanosono T, Masuda K, Xu P, Kobayashi S (2018) Catalytic organic reactions in water toward sustainable society. Chem Rev 118:679–746
Kou J, Dou D, Yang L (2017) Porphyrin photosensitizers in photodynamic therapy and its applications. Oncotarget 8(46)
Kwiatkowski S et al (2018) Photodynamic therapy–mechanisms, photosensitizers and combinations. Biomed Pharmacother 106:1098–1107
Lakowicz JR (2007) Principles of fluorescence spectroscopy. Springer
Lee HM et al (2013) Ursodeoxycholic acid-conjugated chitosan for photodynamic treatment of HuCC-T1 human cholangiocarcinoma cells. Int J Pharm 454:74–81
Lee SY, Kang MS, Jeong WY, Han D-W, Kim KS (2020) Hyaluronic acid-based Theranostic nanomedicines for targeted cancer therapy. Cancers 12
Lee D et al (2021) Optimized combination of photodynamic therapy and chemotherapy using gelatin nanoparticles containing Tirapazamine and Pheophorbide a. ACS Appl Mater Interfaces 13:10812–10821
Li F, Bae B, Na K (2010) Acetylated hyaluronic acid/photosensitizer conjugate for the preparation of nanogels with controllable phototoxicity: synthesis, characterization, autophotoquenching properties, and in vitro phototoxicity against HeLa cells. Bioconjug Chem 21:1312–1320
Li W et al (2016) Smart hyaluronidase-actived theranostic micelles for dual-modal imaging guided photodynamic therapy. Biomaterials 101:10–19
Lovell JF et al (2011) Porphysome nanovesicles generated by porphyrin bilayers for use as multimodal biophotonic contrast agents. Nat Mater 10:324–332
Lucky SS, Soo KC, Zhang Y (2015) Nanoparticles in photodynamic therapy. Chem Rev 115:1990–2042
Mahanta S, Paul S (2015a) Bovine α-lactalbumin functionalized graphene oxide nano-sheet exhibits enhanced biocompatibility: A rational strategy for graphene-based targeted cancer therapy. Colloids Surfaces B Biointerfaces 134:178–187
Mahanta S, Paul S (2015b) Stable self-assembly of bovine α-Lactalbumin exhibits target-specific Antiproliferative activity in multiple cancer cells. ACS Appl Mater Interfaces 7:28177–28187
Mahanta S et al (2015) Stable self-assembled nanostructured hen egg white lysozyme exhibits strong anti-proliferative activity against breast cancer cells. Colloids Surfaces B Biointerfaces 130:237–245
Mahanta S, Prathap S, Ban DK, Paul S (2017) Protein functionalization of ZnO nanostructure exhibits selective and enhanced toxicity to breast cancer cells through oxidative stress-based cell death mechanism. J Photochem Photobiol B Biol 173:376–388
Majumdar P, Nomula R, Zhao J (2014) Activatable triplet photosensitizers: magic bullets for targeted photodynamic therapy. J Mater Chem C 2:5982–5997
Maldonado-Carmona N et al (2020) Conjugating biomaterials with photosensitizers: advances and perspectives for photodynamic antimicrobial chemotherapy. Photochem Photobiol Sci 19:445–461
Managa M, Ngoy BP, Nyokong T (2017) The photophysical studies of Pluronic F127/P123 micelle mixture system loaded with metal free and Zn 5, 10, 15, 20-tetrakis [4-(benzyloxy) phenyl] porphyrins. J Photochem Photobiol A Chem 339:49–58
Marotta DE et al (2011) Evaluation of bacteriochlorophyll-reconstituted low-density lipoprotein nanoparticles for photodynamic therapy efficacy in vivo. Nanomedicine 6:475–487
Melchels FPW, Feijen J, Grijpma DW (2010) A review on stereolithography and its applications in biomedical engineering. Biomaterials 31:6121–6130
Mfouo-Tynga IS, Dias LD, Inada NM, Kurachi C (2021) Features of third generation photosensitizers used in anticancer photodynamic therapy: review. Photodiagn Photodyn Ther 34:102091
Michy T et al (2019) Verteporfin-loaded lipid nanoparticles improve ovarian cancer photodynamic therapy in vitro and in vivo. Cancers (Basel) 11:1760
Mironov AF, Zhdanova KA, Natal’ya, A. B. (2018) Nanosized vehicles for delivery of photosensitizers in photodynamic diagnosis and therapy of cancer. Russ Chem Rev 87:859
Mishra PK, Mishra H, Ekielski A, Talegaonkar S, Vaidya B (2017) Zinc oxide nanoparticles: a promising nanomaterial for biomedical applications. Drug Discov Today 22:1825–1834
Mroz P et al. (2008) Fullerenes as photosensitizers in photodynamic therapy. in Medicinal Chemistry and Pharmacological Potential of Fullerenes and Carbon Nanotubes. Springer. pp. 79–106
Ng KK, Lovell JF, Zheng G (2011) Lipoprotein-inspired nanoparticles for cancer theranostics. Acc Chem Res 44:1105–1113
Peng C-L et al (2010) Development of pH sensitive 2-(diisopropylamino) ethyl methacrylate based nanoparticles for photodynamic therapy. Nanotechnology 21:155103
Qian J et al (2012) Photosensitizer encapsulated organically modified silica nanoparticles for direct two-photon photodynamic therapy and in vivo functional imaging. Biomaterials 33:4851–4860
Quan Q et al (2011) HSA coated iron oxide nanoparticles as drug delivery vehicles for cancer therapy. Mol Pharm 8:1669–1676
Reshetov V et al (2012) Interaction of liposomal formulations of meta-tetra (hydroxyphenyl) chlorin (temoporfin) with serum proteins: protein binding and liposome destruction. Photochem Photobiol 88:1256–1264
Ricci-Júnior E, Marchetti JM (2006) Zinc (II) phthalocyanine loaded PLGA nanoparticles for photodynamic therapy use. Int J Pharm 310:187–195
Sewid FA, Skurlov ID, Kurshanov DA, Orlova AO (2021) Singlet oxygen generation by hybrid structures based on CdSe/ZnS quantum dots and tetraphenylporphyrin in organic medium. Chem Phys Lett 765:138303
Sharma SK, Chiang LY, Hamblin MR (2011) Photodynamic therapy with fullerenes in vivo: reality or a dream? Nanomedicine 6:1813–1825
Shen Z, Fisher A, Liu WK, Li Y (2018) PEGylated “stealth” nanoparticles and liposomes. in Engineering of Biomaterials for Drug Delivery Systems. Elsevier. pp. 1–26
Somu P, Paul S (2019) A biomolecule-assisted one-pot synthesis of zinc oxide nanoparticles and its bioconjugate with curcumin for potential multifaceted therapeutic applications. New J Chem 43:11934–11948
Somu P, Paul S (2021) Surface conjugation of curcumin with self-assembled lysozyme nanoparticle enhanced its bioavailability and therapeutic efficacy in multiple cancer cells. J Mol Liq 338:116623
Spring BQ et al (2016) A photoactivable multi-inhibitor nanoliposome for tumour control and simultaneous inhibition of treatment escape pathways. Nat Nanotechnol 11:378–387
Taratula O et al (2013) A multifunctional theranostic platform based on phthalocyanine-loaded dendrimer for image-guided drug delivery and photodynamic therapy. Mol Pharm 10:3946–3958
Traul DL, Sieber F (2015) Inhibitory effects of merocyanine 540-mediated photodynamic therapy on cellular immune functions: A role in the prophylaxis of graft-versus-host disease? J Photochem Photobiol B Biol 153:153–163
Turubanova VD et al (2019) Immunogenic cell death induced by a new photodynamic therapy based on photosens and photodithazine. J Immunother Cancer 7:1–13
Urano Y et al (2009) Selective molecular imaging of viable cancer cells with pH-activatable fluorescence probes. Nat Med 15:104–109
Wang S, Gao R, Zhou F, Selke M (2004) Nanomaterials and singlet oxygen photosensitizers: potential applications in photodynamic therapy. J Mater Chem 14:487–493
Wang N et al (2014) Gold nanorod-photosensitizer conjugate with extracellular pH-driven tumor targeting ability for photothermal/photodynamic therapy. Nano Res 7:1291–1301
Wieder ME et al (2006) Intracellular photodynamic therapy with photosensitizer-nanoparticle conjugates: cancer therapy using a ‘Trojan horse’. Photochem Photobiol Sci 5:727–734
Wu W, Shao X, Zhao J, Wu M (2017) Controllable photodynamic therapy implemented by regulating singlet oxygen efficiency. Adv Sci 4:1700113
Xiang G-H et al (2013) Effect of PEG-PDLLA polymeric nanovesicles loaded with doxorubicin and hematoporphyrin monomethyl ether on human hepatocellular carcinoma HepG2 cells in vitro. Int J Nanomedicine 8:4613
Xu J, Tanabe S (2019) Persistent luminescence instead of phosphorescence: history, mechanism, and perspective. J Lumin 205:581–620
Zhou L et al (2009) Water-soluble hypocrellin A nanoparticles as a photodynamic therapy delivery system. Dyes Pigments 82:90–94
Zhou A, Wei Y, Wu B, Chen Q, Xing D (2012) Pyropheophorbide A and c (RGDyK) comodified chitosan-wrapped upconversion nanoparticle for targeted near-infrared photodynamic therapy. Mol Pharm 9:1580–1589
Zhou Q, Xu L, Liu F, Zhang W (2016) Construction of reduction-responsive photosensitizers based on amphiphilic block copolymers and their application for photodynamic therapy. Polymer (Guildf) 97:323–334
Zoppellaro G (2020) Iron oxide magnetic nanoparticles (NPs) tailored for biomedical applications. in Magnetic Nanoheterostructures. Springer, 57–102
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Section Editor information
Rights and permissions
Copyright information
© 2022 Springer Nature Singapore Pte Ltd.
About this entry
Cite this entry
Shabbirahmed, A.M., Kumaravel, M., Somu, P., Paul, S., Khadria, A. (2022). Recent Advancements in Nanomaterials for Photodynamic Therapy of Cancers. In: Chakraborti, S. (eds) Handbook of Oxidative Stress in Cancer: Therapeutic Aspects. Springer, Singapore. https://doi.org/10.1007/978-981-16-1247-3_211-1
Download citation
DOI: https://doi.org/10.1007/978-981-16-1247-3_211-1
Received:
Accepted:
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-16-1247-3
Online ISBN: 978-981-16-1247-3
eBook Packages: Springer Reference Biomedicine and Life SciencesReference Module Biomedical and Life Sciences
Publish with us
Chapter history
-
Latest
Recent Advancements in Nanomaterials for Photodynamic Therapy of Cancers- Published:
- 21 April 2022
DOI: https://doi.org/10.1007/978-981-16-1247-3_211-2
-
Original
Recent Advancements in Nanomaterials for Photodynamic Therapy of Cancers- Published:
- 11 March 2022
DOI: https://doi.org/10.1007/978-981-16-1247-3_211-1