Abstract
The term aquaculture is defined as a controlled cultivation and harvest of aquatic animals such as fishes, crustaceans, and molluscs along with some aquatic plants as a source of food. Fishes are often a rich source of nutritional supplement for humans. Industrial revolution has deteriorated the quality and diversity of aquatic life for a long duration. Industrial wastes consisting of by-products such as heavy metals, pesticides, herbicides, weedicides, antifouling compounds, nanoparticles, and microplastics are often known as xenobiotics, foreign substance or chemical to the body or to an ecological system. The aquatic environment has become dumping place as environmental pollution increases due to anthropogenic activities and leads to developmental and functional abnormalities of the reproductive system of fishes and often even to mortalities of aquatic organisms. Thus, the present chapter aims to review various issues related with aquatic pollution, anthropogenic activities, effects of sublethal pollutants on fish reproductive system and mechanism. The study also urges on safe elimination of household sewage and manufacturing effluents as well as laws execution legislated in order to protect the aquatic environment.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Aquatic ecosystem
- Anthropogenic activities
- Water pollution
- Heavy metals reproductive abnormalities
- Gonadal growth
Introduction
The land-based aquaculture is vital for global for fish (28.8 million tons), mollusc (13.1 million tons), and crustacean (5.0 million tons) production (John 2007; FAO 2010). International fish production in 2016 reached about 171 million tons, with percentage of 47%aquaculture performing of the total and 53%, if nonfood uses (including fish oil and fishmeal) are eliminated. With relatively stable capture fishery production since late of 1980s, aquaculture has been responsible for keeping the fundamental growth to provide humans with fishes for consumption (SWFA 2018). Recently the fish consumption has increased quickly especially with the consciousness of its therapeutic and nutritional benefits. Fishes are considered to be the source of vitamins, protein, and unsaturated fatty acids (omega3) and rich with essential minerals (El-Moselhy 2000).
Fatima and Usmani (2013) opined that the fishes are continually exposed to heavy metals and waterborne particulate concentration due to continued stream of water over food sources and gills. This leads to bioaccumulation of heavy metals in various tissues following various types of bioaccumulation factors. Detrimental materials such as paper mill waste, heavy metals, pesticides, crude oil, and polychlorinated biphenyl are usually emitted into the aquatic environment. When large amounts of these pollutants are emitted, there perhaps a severe effect as measured by large-scale unexpected mortalities of fish kills. In fish’s metabolism decreasing, gills damaged and epithelia occurs and immune suppression. Diseases related to pollution include gill disease, ulceration, hepatic damage, and fin/tail rot have been observed (Bukola et al. 2015). Gallo and Tost (2019) have reported that chemical pollution conflicts with hormone function get rises and cause endocrine disruption. Due to susceptibility of hormone receptor systems, it causes endocrine disruptors (EDs) which affect the normal reproductive functions as well as embryo evolution. For a long time, this leads to reproductive defeats and aberrations in the reproductive organs of fish, reptiles, mammals, and birds. Some evidence have been found that hormonally active substance have adverse impacts on fish populations. Field and laboratory studies reveal that a number of chemical substances hamper the reproductive performance of adult fishes, in courtship behavior and parental care. Further it results in mutilation of impaired quality of eggs and sperm (Segner 2011). However we have observed several anthropogenic activities which are accountable in the developmental abnormalities in fish health. The prime purpose of this present chapter is to estimate the factors responsible for poor reproductive development in fishes and their endocrinology.
Significance of Nutritional Value of Fish in Human Diet
Alimentary fish is an essential and opulent protein source. Fish consuming has been reported to share almost 50% of the animal protein consumed in numerous Asian countries (William and Dennis 1988). According to (FAO) report, fish is a premium nutritional importance having protein with high quality and a broad variety of minerals and vitamins, including magnesium, phosphorus, vitamins A and D, selenium, and iodine in marine fish. Fish oils are the richest source of a sort of fat that is necessary to the development of normal brain in fetuses and infants. Shellfish is a major part of worldwide seafood production. It provides bioactive peptides, digestible proteins, long-chain polyunsaturated fatty acids, essential amino acids, vitamin B12 and other vitamins, astaxanthin and other carotenoids, and minerals, including sodium, selenium, zinc, inorganic phosphate, potassium, iodine, and copper (Venugopal and Gopakumar 2017). Nutritional structure study of ordinarily found fishes in Agatti Island water of Lakshadweep Sea in India was carried out. Proximate analysis revealed that the carbohydrate and protein, as hand lipid contents, were high in Thunnus albacares (13.69%), Hyporhamphus dussumieri (6.97%), Parupeneus bifasciatus (6.12%), and T. albacares (1.65%). Major amino acids were methionine, leucine, and lysine registering 2.64–3.91%, 2.67–4.18%, and 2.84–4.56%, respectively. Fatty acid structures ranged from 31.63% to 38.97% saturated (SFA), 21.99–26.30% monounsaturated (MUFAs), 30.32–35.11% polyunsaturated acids (PUFAs), and 2.86–7.79% branched fatty acids of the total fatty acids (Dhaneesh et al. 2012). Similarly Sandhya and Smita (2013) found that R. daniconius fish has the highest protein content. Previous results reveal this fish possesses a good amount of protein so they can be used safely in food to supplement protein.
Heavy Metals Exposure and Its Impacts
Nowadays modern human society is facing environmental pollution as a major challenge (Ali and Khan 2017). Pollution caused due to heavy metals is a serious menace and key concerned to the environment (Ali et al. 2013; Hashem et al. 2017). The toxicity of heavy metals has proven to be a prime menace and causing several health risks. Toxicity of metals depends upon the route of exposure, duration of exposure, and the absorbed dose, i.e., chronic or acute (Jaishankar et al. 2014). Naturally weathering of metal-containing volcanic eruptions and rocks are the sources of its occurrence, while human sources are mining, industrial emissions, and smelting, along with agricultural activities such as use of phosphate fertilizers and pesticides. Also, fossil fuel burning adds heavy metals like cadmium (Cd) in the environment (Spiegel 2002). Heavy metal remains permanent in the environment which enters in food chain through bioaccumulation and causes various health issues due to its toxicity. Their chronic exposure in the environment now became real menace to living organisms (Wieczorek-Dąbrowska et al. 2013). Mercury is a potent heavy metal neurotoxin for living organisms. Effects and exposure of sublethal mercury on fish have been investigated. Studies reveal and suggest that the restrained effects of mercury in reproduction occur at numerous sites within the genital axis, including the gonads, hypothalamus, and pituitary. Further, ill effects including circulating reproductive steroids and reducing in gonad size in fishes have been reported (Kate and Vance 2009). In aquatic environment mercury is present in various forms, including elemental, ionic, and organic (Morel et al. 1998). Due to industrial activities highly polluted local fish population, with mercury concentrations, ranged from 8.4 to 24 μg/g wet weight in Minamata Bay, Japan (Kitamura 1968), and from 6.3 to 16 μg/g wet weight in Clay Lake, Ontario, Canada (Lockhart et al. 1972), observed. Methylmercury, one of the most lethal forms, bioaccumulates in fish mostly by dietary uptake (Spry and Wiener 1991; Hall et al. 1997). Watras and Bloom (1992) reported that the specific bioaccumulation of methyl mercury in the aqueous environment ranges to 15% in phytoplankton, 30% in zooplankton, and more than 90% in fish. Changes in reproduction, behavior, growth, biochemistry, survival, and development in fishes can occur due to exposure of methylmercury (Weiner and Spry 1996; Sorensen 1991). Rao (1989) opined that mercury may obstruct in proper functioning of sperm mitochondria, which results in decrease in energy production for sperm mobility.
Gonadotropin-releasing hormone regulates the synthesis and release of the gonadotropins (follicle-stimulating hormone [FSH] and luteinizing hormone [LH]), making it a decisive neuroendocrine arbiter of genital function. The evidence from histological analysis of neurons in the hypothalamic preoptic nuclei of the murrel (Channa punctatus) reveals that mercury as a potent inhibitory factor for neurosecretion (Ram and Joy 1988). A number of authors have reported that gonadotropic hormones (LH and FSH) released in fish from pituitary manage their yearly cycle of gonadal growth, sperm release in males, ovulation in females, and production of sex steroids in both genders (Breton et al. 1998; Weltzien et al. 2004; Kamei et al. 2005). Hence, distraction in gonadotropin excretion plays a great role on fertility. An investigation reports the decreased in atrophied seminiferous tubules, and spermatogenesis were observed in male Nile tilapia (Oreochromis niloticus) after the exposure to methylmercury within 7 months (Arnold 2000). The gonadotropic regulation of spermatogenesis and spermiogenesis in fish is mediated by androgens secreted by the interstitial cells (Yaron 1995). Exposure to methylmercury in male guppies has resulted in fibrosis and inflammation of the interstitium (Wester 1991; Wester and Canton 1992). These cytotoxic effects involve potential reverse effects on the steroidogenic potential of the interstitial cells. Androgen helps in sexual behavior, gonadal development, and secondary sexual characteristics in male fish (Kime 1998). Similarly Kime (1993) found that the secretion of testosterone and/or 11-ketotestosterone is high during gonadal recrudescence, which is rejected before spermiation. The levels of plasma testosterone drastically decline in male fathead minnows fed diets including 0.87 to 3.93 μg/g of methylmercury for 250 days (Drevnick and Sandheinrich 2003). The decreased level of 11-ketotestosterone is also observed after a 6-month exposure of methylmercury in male Nile tilapia (O. niloticus) (Arnold 2000). Now it became obvious that anthropogenic activities have great impacts on fish reproductive health.
Exposure and Impacts of Microplastics
Plastic pollution is a global issue for aquatic animals in almost marine and ocean on the earth and threat to marine life, and great economic loss for aquaculture (Eriksen et al. 2014; Derraik 2002; Thompson et al. 2009). Plastic can demean the coastal benthic habitats via smothering; this is due to formation of plastic sheet layers over the benthos and also through alterations in sediment, permeability occurs by buried plastics (Carson et al. 2011). Engulfed plastics cause gastrointestinal (GI) tract. For example, it can cause abrasions and lesions or physical disruption of the GI tract, as plastics compress in the gut (Di Bello et al. 2013). Various authors suggested that PCBs are capable to change the proper regulation and function of foremost hormones such as estrogen, testosterone, and thyroxine (Goncharov et al. 2009; Colborn et al. 1993). Moreover microplastics (MPs) are smaller pieces of plastic debris which are not visible by the naked eye termed as microplastics in aquatic medium which is a matter of concerned. Microplastics are derived from marine debris and have potential ecological impacts on marine creatures (Anthony 2011). Due to its small size and ever present microplastics found to various organisms in both pelagic and benthic aquatic habitats (Foekema et al. 2013; Mathalon and Hill 2014). During experimental study it was found that small fragments of microplastics are transferred into aquatic animals through food web, in small amounts (Lehtiniemi et al. 2018). A study carried by Critchell and Hoogenboom (2018) explains the exposure and effects of microplastics on juveniles of a planktivorous fish (Acanthochromis polyacanthus), which is prevalent and abundant on Indo-Pacific coral reefs. During study it was observed that when individual fish was exposed of plastic after a 1-week the increased numbers of plastics present in GI tract greatly get increased on decreasing the size of plastics to approximately one-fourth the size of the food particles, having maximum of 2102 small (<300 μm diameter) particles present in the gut of that fish. The result reveals negative effects on the growth of fish.
Impacts of Oil Spills
Oil spills draw a great concern, and its effects on marine biological systems cannot be ignored, due to rich biological production of harvested resources (Langangen et al. 2017).
Fish larvae and eggs are normally susceptible to toxic materials present in oil components, due to its small size, poorly developed membranes, and their positions in water column. Various experimental studies found that presence of toxic materials in oil components (such as polycyclic aromatic hydrocarbons, PAHs) at very low concentrations can cause a sublethal damage to fish larvae and eggs (Carls et al. 1999; Hicken et al. 2011; Meier et al. 2010; Scott and Sloman 2004; Sørhus et al. 2015). The oil spills in the aquatic medium cause a sublethal damage to fish eggs as well as their larval stage; for example, morphological deformities, reduced feeding, and growth rates increase susceptibility to predators, and starvation occurs (Sørhus et al. 2016; Hicken et al. 2011). It spoil their habitat, loss of hatching ability of eggs, impaired reproduction, growth, development, fouling of gill structures, feeding, respiration (Blackburn et al. 2014). Mostly various species of fishes utilized most of their time living in groups, along with group coordination which plays an important role in the emergent benefits of group-living. But these group cohesions can be faint due to several factors in which exposure to toxic environmental contaminants is one of them. Experimental study reveals that oil spill components and its exposure have negative effects on fish behavior which may lead to reduce their ecological success (Armstrong et al. 2019). Osuagwu and Olaifa (2018) suggested that presence of crude oil spills over coastal water increases the risk of contamination to fish hatcheries of valuable fishes. Their findings show that oil spills have negatively affected fish production. However another study reveals that oil spills contain polycyclic aromatic hydrocarbons (PAHs) which have endocrine-disrupting properties. This shows a negative effect on the polar cod (Boreogadus saida), an Arctic keystone species which possesses an extensive and energy-intensive reproductive development systems. Study reveals the considerable alterations in sperm mobility due to crude oil exposure in males, and data were compared to the controls. During investigation somatic indices (gonad and hepatic cells), germ cell development along with plasma steroid levels (estradiol-17 [females], testosterone [males and females], and 11-ketotestosterone [males]) was found not drastically distorted by constant dietary exposure to crude oil (Bender et al. 2016).
Impacts of Pesticides and Insecticides
Pesticides are broadly used in the advanced agricultural practices, in diverse quantities, over the world. However it is very helpful for crop yielding, there are some severe issues in the environment, such as health- and safety-related concerns for aquatic as well as terrestrial living organisms which include humans, animals, and plants (Sana Ullah et al. 2018). Pollution due to pesticides is a major menace to freshwater ecosystems throughout the world (Madeleine et al. 2015). Freshwater ecosystems are essential collections of almost all ecosystems, like potable water, irrigation water for agriculture, industrial water, water storage, water recreation, and an environment for organisms which helps to grow fish and other important foods. An invertebrate which is part of food chains provides transfer of energy and nutrients from primary producers to higher tropic levels (Madeleine et al. 2015).
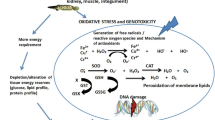
Fishes get exposed to pesticides and their residues through various routes such as runoffs or spray coming directly from agricultural fields and gardens. In aquatic ecosystem fish are predictably one of most affected organisms by pollutant present in aquatic medium. Pesticides present in aquatic bodies deteriorate its quality through alteration in physicochemical parameters and threat aquatic organisms (Sarwar et al. 2007; Sabae et al. 2014). A number of insecticides have been documented by the UK-based Pesticides Action Network (UK PAN 2009). Globally, pesticides are found in various compositions which consist of herbicides (15%), fungicides (1.46%), and insecticides (80%) (Marigoudar 2012). Cypermethrin (CYP), a pyrethroid, derived from pyrethrin, extracted from Chrysanthemum cinerariaefolium, is a broadly used, capable, and readily available insecticide. It is almost widely used insecticide found in various kinds of agricultural practices, gardens, lawn, buildings, and forestry in order to protect from insects and cotton and soya beans from pests as well as repel and control mosquitoes which are malaria parasite carriers. Furthermore it is the expansively used pesticide in agriculture and households, and also its concentration in aquatic ecosystem has been reported. Its existence in our environment is posing a brutal threat to humans as well as other nontarget terrestrial and aquatic organisms (Sana Ullah et al. 2018). The negative effects of cypermethrin and toxicity in fish have been illustrated in Fig. 20.1. Similarly the developmental abnormalities and effect of cypermethrin (CYP) in various fish breed have been tabulated in Table 20.1.
The toxic effects through cypermethrin induced in fish (adapted from Sana Ullah et al., 2018; image of Labeo rohita uploaded by S M Majharul Islam and Zebra fish (https://www.google.com)
Conclusion and Future Direction
Anthropogenic activities result in accumulations of heavy metals, oil spills, pesticides, insecticides, and microplastics in aquatic ecosystem which renders several reproductive abnormalities in fish. Contaminations of pesticide toxicity in natural resources may impose significant risks on environment and untargeted organisms of different verities from helpful soil microorganisms to insects, aquatic animals, birds, humans, and plants. Metabolism due to such toxicity of heavy metals and other noxious constituents mostly affects the rate of survival and growth of fishes and alters their various body functions inducing neurotoxicity, hematotoxicity, toxic histopathological effects, immunotoxicity, genotoxicity, and disruptions of endocrine systems. It also distorts the neuroendocrine system, which plays a vital role in reproductive function of LH and FSH hormones by its mechanisms through hypothalamus which maintains homeostasis, proper functioning of reproductive system, metabolic process, energy utilization, osmoregulations, and blood pressure. Therefore the present study concludes that unsystematic disposal of any kind of pollutants without proper pretreatment should not be allowed, in order to minimize the negative effects of water pollution on fish health. Safe and sound disposals of any domestic sewage and industrial effluents should be practiced through the enforcement of enacted laws in order to protect aquatic environment.
References
Ali, H., & Khan, E. (2017). Environmental chemistry in the twenty first century. Environmental Chemistry Letters, 15(2), 329–346.
Ali, H., Khan, E., & Sajad, M. A. (2013). Phytoremediation of heavy metals-concepts and applications. Chemosphere, 91(7), 869–881.
Anthony, L. A. (2011). Microplastics in the marine environment. Marine Pollution Bulletin, 62, 1596–1605.
Armstrong, T., Khursigara, A. J., Killen, S. S., Fearnley, H., Parsons, K. J., & Esbaugh, A. J. (2019). Oil exposure alters social group cohesion in fish. Scientific Reports, 9, 13520.
Arnold, B. S. (2000). Disruption of mercury within different trophic levels of the Okefenokee swamp, within tissues of top level predators, and reproductive effects of methylmercury in the Nile tilapia (Oreochromis niloticus). PhD thesis, University of Georgia, Athens, GA, USA.
Begum, G. (2009). Enzymes as biomarkers of cypermethrin toxicity: response of Clarias batrachus tissues ATPase and glycogen phosphorylase as a function of exposure and recovery at sublethal level. Toxicology Mechanisms and Methods, 19(1), 29–39.
Bender, M. L., Frantzen, M., Vieweg, I., Falk-Petersen, I.-B., Johnsen, H. K., Rudolfsen, G., et al. (2016). Effects of chronic dietary petroleum exposure on reproductive development in polar cod (Boreogadus saida). Aquatic Toxicology, 180, 196–208.
Blackburn, M., Mazzacano, C. A. S., Fallon, C., & Black, S. H. (2014). Oil in our oceans: A review of the impacts of oil spills on marine invertebrates (p. 152). Portland, OR: The Xerces Society for Invertebrate Conservation.
Breton, B., Govoroun, M., & Mikolajczyk, T. (1998). GTH I and GTH II secretion profiles during the reproductive cycle in female rainbow trout: Relationship with pituitary responsiveness to GnRH-A stimulation. General and Comparative Endocrinology, 111, 38–50.
Bukola, D., Aderolu, Z., Elegbede, I. O., & Adekunbi, F. (2015). Consequences of anthropogenic activities on fish and the aquatic environment. Poultry, Fisheries and Wildlife Sciences, 3(2), 46–47.
Carls, M. G., Rice, S. D., & Hose, J. E. (1999). Sensitivity of fish embryos to weathered crude oil: Part I. Low-level exposure during incubation causes malformations, genetic damage, and mortality in larval pacific herring (Clupea pallasi). Environmental Toxicology and Chemistry, 18, 481–493.
Carriquiriborde, P., Díaz, J., Lopez, G. C., Ronco, A. E., & Somoza, G. M. (2009). Effects of cypermethrin (10% EC) induced stress in biochemical and hematological parameters of Indian major carp Catla catla (Hamilton, 1822). World Journal of Pharmaceutical Research, 3(4), 1976–1996.
Carson, H. S., Colbert, S. L., Kaylor, M. J., & McDermid, K. J. (2011). Small plastic debris changes water movement and heat transfer through beach sediments. Marine Pollution Bulletin, 62(8), 1708–1713.
Colborn, T., vom Saal, F. S., & Soto, A. M. (1993). Developmental effects of endocrine-disrupting chemicals in wildlife and humans. Environmental Health Perspectives, 101(5), 378–384.
Critchell, K., & Hoogenboom, M. O. (2018). Effects of microplastic exposure on the body condition and behaviour of planktivorous reef fish (Acanthochromis polyacanthus). PLoS One, 13(3), e0193308.
Dawar, F. U., Zuberi, A., Azizullah, A., & Khattak, M. N. K. (2016). Effects of cypermethrin on survival, morphological and biochemical aspects of rohu (Labeo rohita) during early development. Chemosphere, 144, 697–705.
Derraik, J. G. B. (2002). The pollution of the marine environment by plastic debris: A review. Marine Pollution Bulletin, 44, 842–852.
Dhaneesh, K. V., Noushad, K. M., & Ajith Kumar, T. T. (2012). Nutritional evaluation of commercially important fish species of Lakshadweep Archipelago, India. PLoS One, 7(9), e45439.
Di Bello, A., Valastro, C., Freggi, D., Lai, O. R., Crescenzo, G., & Franchini, D. (2013). Surgical treatment of injuries caused by fishing gear in the intracoelomic digestive tract of sea turtles. Diseases of Aquatic Organisms, 106(2), 93–102.
Drevnick, P. E., & Sandheinrich, M. B. (2003). Effects of dietary methylmercury on reproductive endocrinology of fathead minnows. Environmental Science & Technology, 37, 4390–4396.
El-Moselhy, K. M. (2000). Accumulation of copper, cadmium and lead in some fish from the Gulf of Suez. Egyptian Journal of Aquatic Biology and Fisheries, 4(3), 235–249.
Eriksen, M., Lebreton, L. C. M., Carson, H. S., Thiel, M., Moore, C. J., Borerro, J. C., et al. (2014). Plastic pollution in the world’s oceans: more than 5 trillion plastic pieces weighing over 250,000 tons afloat at sea. PLoS One, 9(12), e111913.
Fatima, M., & Usmani, N. (2013). Histopathology and bioaccumulation of heavy metals (Cr, Ni and Pb) in fish (Channa striatus and Heteropneustes fossilis) tissue: A study for toxicity and ecological impacts. Pakistan Journal of Biological Sciences, 16, 412–420.
Foekema, E. M., De Gruijter, C., Mergia, M. T., van Franeker, J. A., Murk, A. J., & Koelmans, A. A. (2013). Plastic in North Sea fish. Environmental Science & Technology, 47(15), 8818–8824.
Food and agriculture organization of United Nations (FAO). (2010). The State of World Fisheries and Aquaculture (p. 197). Rome.
Gallo, A., & Tost, E. (2019). Effects of ecosystem stress on reproduction and development. Molecular Reproduction and Development, 86, 1269–1272.
Goncharov, A., Rej, R., Negoita, S., Schymura, M., Santiago-Rivera, A., & Morse, G. (2009). Lower serum testosterone associated with elevated polychlorinated biphenyl concentrations in native American Men. Environmental Health Perspectives, 117, 1454–1460.
Hall, B. D., Bodaly, R. A., Fudge, R. J. P., Rudd, J. W. M., & Rosenberg, D. M. (1997). Food as the dominant pathway of methylmercury uptake by fish. Water, Air, and Soil Pollution, 100, 13–24.
Hashem, M. A., Nur-A-Tomal, M. S., Mondal, N. R., & Rahman, M. A. (2017). Hair burning and liming in tanneries is source of pollution by arsenic, lead, zinc, manganese and iron. Environmental Chemistry Letters, 15(3), 501–506.
Hicken, C. E., Linbo, T. L., Baldwin, D. H., Willis, M. L., Myers, M. S., et al. (2011). Sublethal exposure to crude oil during embryonic development alters cardiac morphology and reduces aerobic capacity in adult fish. Proceedings of the National Academy of Sciences of the United States of America, 108, 7086–7090.
Jaishankar, M., Tseten, T., Anbalagan, N., Mathew, B. B., & Beeregowda, K. N. (2014). Toxicity, mechanism and health effects of some heavy metals. Interdisciplinary Toxicology, 7(2), 60–72.
Jin, Y., Zheng, S., & Fu, Z. (2011). Embryonic exposure to cypermethrin induces apoptosis and immunotoxicity in zebrafish (Danio rerio). Fish & Shellfish Immunology, 30(4), 1049–1054.
John, M. (2007). Introduction to aquaculture, Department of agriculture, environment and rural affairs. Retrieved from https://www.daera-ni.gov.uk/articles/introduction-aquaculture.
Kamei, H., Kawazoe, I., Kaneko, T., & Aida, K. (2005). Purification of follicle-stimulating hormone from immature Japanese eel, Anguilla japonica, and its biochemical properties and steroidogenic activities. General and Comparative Endocrinology, 143, 257–266.
Kannan, M., Muthusamy, P., & Venkatachalam, U. (2014). Response of synthetic pyrethroid cypermethrin (10% EC) induced stress in biochemical and haematological parameters of Indian major carp Catla catla (Hamilton, 1822). World Journal of Pharmaceutical Research, 3(4), 1976–1996.
Kate, L. C., & Vance, L. T. (2009). Mercury induced reproductive impairment in fish. Environmental Toxicology and Chemistry, 28(5), 895–907.
Kime, D. E. (1993). Classical and non-classical reproductive steroids in fish. Reviews in Fish Biology and Fisheries, 3, 160–180.
Kime, D. E. (1998). Endocrine disruption in fish. Boston, MA: Kluwer Academic.
Kitamura, S. (1968). Determination on mercury content in bodies of inhabitants, cats, fishes and shells in Minamata District and in the mud of Minamata Bay. In M. Kutsuna (Ed.), Minamata disease (pp. 257–266). Kumamoto: Kumamoto University Press.
Kutluyer, F., Benzer, F., Eris, M., Ogretmen, F., & Inanan, B. E. (2016). The in vitro effect of cypermethrin on quality and oxidative stress indices of rainbow trout Oncorhynchus mykiss spermatozoa. Pesticide Biochemistry and Physiology, 128, 63–67.
Langangen, Ø., Olsen, E., Stige, L. C., Ohlberger, J., Yaragina, N. A., Vikebø, F. B., et al. (2017). The effects of oil spills on marine fish: Implications of spatial variation in natural mortality. Marine Pollution Bulletin, 119, 102–109.
Lehtiniemi, M., Hartikainen, S., Näkki, P., Engström-Öst, J., Koistinen, A., & Setälä, O. (2018). Size matters more than shape: Ingestion of primary and secondary microplastics by small predators. Food Webs, 17, e00097.
Lockhart, W. L., Uthe, J. F., Kenney, A. R., & Mehrle, P. M. (1972). Methylmercury in northern pike (Esox lucius)—Distribution, elimination, and some biochemical characteristics of contaminated fish. Journal of the Fisheries Research Board of Canada, 29, 1519–1523.
Loteste, A., Scagnetti, J., Simoniello, M. F., Campana, M., & Parma, M. J. (2013). Hepatic enzymes activity in the fish Prochilodus lineatus (Valenciennes, 1836) after sublethal cypermethrin exposure. Bulletin of Environmental Contamination and Toxicology, 90(5), 601–604.
Madeleine, C., David, K., Edward, A. D. M., Christy, A. M., Dominique, A. N., & Van der Sluijs, J. P. (2015). Risks of large-scale use of systemic insecticides to ecosystem functioning and services. Environmental Science and Pollution Research, 22, 119–134.
Marigoudar, S. R. (2012). Cypermethrin induced pathophysiological and some biochemical changes in the freshwater teleost, Labeo Rohita (Hamilton). PhD thesis, Department of Zoology, Karnatak University, Dharwad-580 003, India.
Masud, S., & Singh, I. J. (2013). Effect of Cypermethrin on some hematological parameters and prediction of their recovery in a freshwater Teleost, Cyprinus carpio. African Journal of Environmental Science and Technology, 7(9), 852–856.
Mathalon, A., & Hill, P. (2014). Microplastic fibers in the intertidal ecosystem surrounding Halifax Harbor, Nova Scotia. Marine Pollution Bulletin, 81(1), 69–79.
Meier, S., Craig Morton, H., Nyhammer, G., Grøsvik, B. E., Makhotin, V., Geffen, A., et al. (2010). Development of Atlantic cod (Gadus morhua) exposed to produced water during early life stages: Effects on embryos, larvae, and juvenile fish. Marine Environmental Research, 70, 383–394.
Monir, M. S., Ashaf-Ud-Doulah, M., Rahman, M. K., Akhter, J. N., Hossain, M. R., & Sultana, S. (2016). Histoarchitecture changes in the ovary of Stinging catfish, Shing (Heteropneustes fossilis) under cypermethrin toxicity. Asian-Australasian Journal of Bioscience and Biotechnology, 1(1), 47–53.
Morel, F. M. M., Kraepiel, A. M. L., & Amyot, M. (1998). The chemical cycle and bioaccumulation of mercury. Annual Review of Ecology and Systematics, 29, 543–566.
Osuagwu, E. S., & Olaifa, E. (2018). Effects of oil spills on fish production in the Niger Delta. PLoS One, 13(10), e0205114.
Poletta, G. L., Gigena, F., Loteste, A., Parma, M. J., Kleinsorge, E. C., & Simoniello, M. F. (2013). Comet assay in gill cells of Prochilodus lineatus exposed in vivo to cypermethrin. Pesticide Biochemistry and Physiology, 107(3), 385–390.
Ram, R. N., & Joy, K. P. (1988). Mercurial induced changes in the hypothalamo-neurohypophysical complex in relation to reproduction in the teleostean fish, Channa punctatus (Bloch). Bulletin of Environmental Contamination and Toxicology, 41, 329–336.
Rao, M. V. (1989). Toxic effects of methylmercury on spermatozoa in vitro. Experientia, 45, 985–987.
Sabae, S. Z., El-Sheekh, M. M., Khalil, M. A., Elshouny, W. A. E., & Badr, H. M. (2014). Seasonal and regional variation of physicochemical and bacteriological parameters of surface water in El-Bahr El-Pherony, Menoufia, Egypt. World Journal of Fish and Marine Sciences, 6(4), 328–335.
Sandhya, M. P., & Smita, R. S. (2013). Fish muscle protein highest source of energy. International Journal of Biodiversity and Conservation, 5(7), 433–435.
Sarwar, S., Ahmad, F., & Khan, J. (2007). Assessment of the quality of Jehlum river water for irrigation and drinking at district Muzaffarabad Azad Kashmir. Sarhad Journal of Agriculture, 23(4), 1041.
Scott, G. R., & Sloman, K. A. (2004). The effects of environmental pollutants on complex fish behaviour: Integrating behavioural and physiological indicators of toxicity. Aquatic Toxicology, 68, 369–392.
Segner, H. (2011). Reproductive and developmental toxicity in fishes. In Book: Reproductive and developmental toxicology (pp. 1145–1166). Elsevier.
Sorensen, E. M. B. (1991). Metal poisoning in fish. Boca Raton, FL: CRC.
Sørhus, E., Edvardsen, R. B., Karlsen, Ø., Nordtug, T., van der Meeren, T., Thorsen, A., et al. (2015). Unexpected interaction with dispersed crude oil droplets drives severe toxicity in Atlantic haddock embryos. PLoS One, 10(4), e0124376.
Sørhus, E., Incardona, J. P., Karlsen, Ø., Linbo, T., Sørensen, L., et al. (2016). Crude oil exposures reveal roles for intracellular calcium cycling in Haddock craniofacial and cardiac development. Scientific Reports, 6, 31058.
Spiegel, H. (2002). Trace element accumulation in selected bio-indicators exposed to emissions along the industrial facilities of Danube Lowland. Turkish Journal of Chemistry, 6(26), 815–823.
Spry, D. J., & Wiener, J. G. (1991). Metal bioavailability and toxicity to fish in low-alkalinity lakes-A critical review. Environmental Pollution, 71, 243–304.
SWFA (The State of World Fisheries and Aquaculture). (2018). Present FAO’s official world fishery and aquaculture statistics. Rome: FAO
Thompson, R. C., Moore, C. J., von Saal, F. S., & Swan, S. H. (2009). Plastics, the environment and human health: Current consensus and future trends. Philosophical Transactions of the Royal Society Biological Sciences, 1, 1–14.
UK PAN (Pesticides Action Network). (2009). List of lists: A catalogue of lists of pesticides identifying those associated with particularly harmful health or environmental impacts. P.A. Network.
Ullah, S., Zuberi, A., Alagawany, M., Farag, M. R., Dadar, M., Karthik, K., et al. (2018). Cypermethrin induced toxicities in fish and adverse health outcomes: Its prevention and control measure adaptation. Journal of Environmental Management, 206, 863–871.
Venugopal, V., & Gopakumar, K. (2017). Shellfish: Nutritive value, health benefits, and consumer safety. Comprehensive Reviews in Food Science and Food Safety, 16, 1219–1241.
Watras, C. J., & Bloom, N. S. (1992). Mercury and methylmercury in individual zooplankton-Implications for bioaccumulation. Limnology and Oceanography, 37, 1313–1318.
Weiner, J. G., & Spry, D. J. (1996). Toxicological significance of mercury in freshwater fish. In W. N. Beyer, G. H. Heinz, & A. W. Redmon-Norwood (Eds.), Environmental contaminants in wildlife: Interpreting tissue concentrations (pp. 297–339). Boca Raton, FL: Lewis.
Weltzien, F. A., Andersson, E., Andersen, O., Shalchian-Tabrizi, K., & Norberg, B. (2004). The brain-pituitary-gonad axis in male teleosts, with special emphasis on flatfish (Pleuronectiformes). Comparative Biochemistry and Physiology. Part A, Molecular & Integrative Physiology, 137, 447–477.
Wester, P. W. (1991). Histopathological effects of environmental pollutants beta-HCH and methylmercury on reproductive organs in fresh water fish. Comparative Biochemistry and Physiology Part C: Pharmacology, Toxicology and Endocrinology, 100, 237–239.
Wester, P. W., & Canton, H. H. (1992). Histopathological effects in Poecilia reticulata (guppy) exposed to methylmercury chloride. Toxicologic Pathology, 20, 81–92.
Wieczorek-Dąbrowska, M., Tomza-Marciniak, A., Pilarczyk, B., & Balicka-Ramisz, A. (2013). Roe and red deer as bio indicators of heavy metals contamination in north-western Poland. Chemistry and Ecology, 2(29), 100–110.
William, C., & Dennis, C. (1988). Food microbiology (4th ed.). Singapore: McGraw-Hill Book Company.
Xu, C., Tu, W., Lou, C., Hong, Y., & Zhao, M. (2010). Enation selective separation and zebra fish embryo toxicity of insecticide beta-cypermethrin. Journal of Environmental Sciences, 22(5), 738–743.
Yaron, Z. (1995). Endocrine control of gametogenesis and spawning induction in the carp. Aquaculture, 129, 49–73.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Ansari, I., Maiti, D., Sundararajan, M., Ashar, M.S. (2021). Anthropogenic Exposure and Its Impact on Reproductive System of Fishes. In: Sundaray, J.K., Rather, M.A., Kumar, S., Agarwal, D. (eds) Recent updates in molecular Endocrinology and Reproductive Physiology of Fish. Springer, Singapore. https://doi.org/10.1007/978-981-15-8369-8_20
Download citation
DOI: https://doi.org/10.1007/978-981-15-8369-8_20
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-8368-1
Online ISBN: 978-981-15-8369-8
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)