Abstract
Splenic artery embolization is an additional way to treat the sequel of portal hypertension like variceal hemorrhage (prevention and treatment), hypersplenism, hepatogenic ascites, and hepatic encephalopathy. Partial splenic embolization (PSE) has been widely used in selected patient for the management of sequels of portal hypertension, where other form of therapy is not useful or feasible because of either comorbid condition or deranged liver functions. Two methods are usually used for partial splenic arterial embolization; selective partial embolization and nonselective partial embolization. Proper selection of patients, intervention technique, aseptic measure, antibiotic coverage, and post-procedure care are key factors of success of PSE.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Splenic arterial interventions are alternative to surgery for the management of conditions like portal hypertension, hypersplenism, splenic arterial aneurysm, splenic trauma, and splenic neoplasm. Partial splenic embolization (PSE) is accepted for the treatment of leukocytopenia and thrombocytopenia produced by hypersplenism and is considered a decent option to splenectomy [1]. Presently PSE is used in some selected patient to treat major sequelae of portal hypertension, where other form of therapy is not useful or feasible; this includes variceal hemorrhage, hypersplenism, hepatogenic ascites, and hepatic encephalopathy. Interventional radiologist should be familiar with the indication, contraindication, and different types of splenic artery embolization techniques used. Splenic embolization is also used in combination with supplementary treatments for the moderation of portal hypertension and accompanying sequelae of portal hypertension.
-
Variceal hemorrhage (prevention and treatment)
-
Hypersplenism
-
Hepatogenic ascites
-
Hepatic encephalopathy
-
Infection (Local/systemic)
16.1 Variceal Hemorrhage
Portal hypertension in cirrhotic patients leads to the formation of varices. This along with a low platelet count increases the risk for catastrophic hemorrhage. Thrombocytopenia in these patients results due to stasis of platelet in the enlarged spleen [2]. Endoscopic obliteration of gastroesophageal varices and the creation of a transjugular intrahepatic portosystemic shunt (TIPS) are the two most widely used method to manage variceal hemorrhage. Further, liver transplantation remains the ultimate treatment for advanced liver disease with portal hypertension. However, some patients are neither suitable for TIPS nor fit for liver transplantation.
Splenic embolization was initially used to reduce the occurrence of variceal bleeding in patients with portal hypertension. First this was performed in 1973 using an autologous blood clot to treat recurrent gastrointestinal hemorrhage from esophageal varices [1]. Embolization may be some times combined with other therapeutic interventions, such as endoscopic ligation [3, 4] or balloon-occluded retrograde transvenous obliteration (BRTO) [5]. PSE along with endoscopic variceal ligation is mainly useful in subset of ill patient, where thrombocytopenia is main cause of bleeding from varices. PSE causes improvement in platelet count, this prevents esophageal variceal hemorrhage and may help in recovery of patients’ clinical status [6]. Pälsson et al. found significant increase in hemoglobin, leukocyte, and thrombocyte cell count with improvement in mean survival (50.5 months) in 26 patients treated with partial splenic embolization for esophageal varices or thrombocytopenia [6]. Similarly, in a study by Ohmoto et al. esophageal varices bleeding rate decreased from 4.3% in the pretreatment group to 1.1% after PSE in 84 cirrhotic patients with large esophageal varices and thrombocytopenia. In this study, 42 patients received endoscopic variceal ligation (EVL) and 42 received combination of EVL and PSE. They also showed improved overall survival from 31% to 50% [7]. Review of five studies by Koconis et al., which included 50 patients of PSAE, showed decrease in the yearly occurrence of variceal hemorrhage by 80% [8].
16.2 Liver Function
PSE has been used as a tool to improve liver functions in cirrhotic patients [9]. Improvements in 12 month cholinesterase, cholesterol, total protein, prothrombin time, and albumin level have been validated in few studies [ 10, 11]. PSE leads to decrease in portal pressure and thus reduces injury to the liver parenchyma.
16.3 Blood Parameters
Splenic embolization is also used to increase white blood cells and platelets in cirrhotic patients and specially indicated in symptomatic patient like patient having repeated skin rashes or frequent skin infections. Decreased splenic sequestration and increase in thrombopoietin levels lead to increase in platelet concentration [6, 12].
16.4 Hepatogenic Refractory Ascites/Hepatic Encephalopathy
Post-PSE decrease in effective splenic volume significantly reduces the venous drainage and thus, a decline in portal venous flow and hence the pressure. The reduction in portal pressure suggests a potential role for PSE in the spectrum of therapies used to treat advanced portal hypertension, especially in patients with borderline liver dysfunction and encephalopathy. PSE may be useful in patient with refractory ascites, where TIPS is not desirable because of some comorbid condition or advanced liver dysfunctions.
16.5 Technique
16.5.1 Type of Intervention—Partial Splenic Embolization
Partial splenic embolization—Two methods are usually used for PSE: Selective PSE and Nonselective PSE. In selective partial embolization, as name suggests, a few intraparenchymal splenic artery branches are super selectively catheterized, and embolized to achieve complete stasis of blood flow in 50% of splenic parenchyma (Fig. 16.1); while other branches of remaining splenic parenchyma show persistent blood flow. Initial baseline angiograms are useful to calculate the volume of the splenic tissue to be embolized. In nonselective partial embolization, the catheter tip is placed in the main splenic artery but beyond the origin of major pancreatic branches and embolic particles is injected until the parenchymal blush is reduced by around 50%.
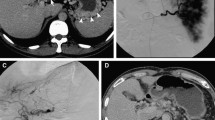
Selective partial splenic embolization—CT abdomen arterial phase coronal image (a), showing cirrhotic liver with gross splenomegaly and moderate ascites. Initial baseline splenic angiographic images (b) delineated the anatomy of splenic artery and parenchymal blush. Super selective angiogram image (c) using microcatheter and final check splenic angiographic images obtained after embolization (d) showing complete occlusion of the lower-mid segmental splenic arterial branches (~50%). Axial CT scan image (e1) and USG image (e2) shows an infarcted area in the splenic parenchyma. Follow-up coronal CT scan image (f) obtained 1 year later shows atrophic mid-lower pole of the spleen and complete resolution of ascites
Fever, local or systemic infections are contraindications for the procedure, as they are associated with increased risk of abscess formation in the infarcted tissue or aggravation of systemic infection.
Common femoral artery access is gained by Seldinger technique and 5F sheath is placed. A C2 or SIM1 catheter is usually used to cannulate the celiac axis. Main splenic artery is cannulated and angiogram is performed to evaluate the splenic arterial branches as well as the origin of pancreatic branches. A micro catheter is used for super selective cannulation of splenic artery branches for embolization (Fig. 16.1). Embolization of the peripheral branches or middle and lower pole branches of spleen (Fig. 16.1) reduce complications such as pneumonia, left upper quadrant abdominal pain, and pleural reaction/effusion. It is also beneficial while evaluating the embolized volume after the procedure [13].
Gelatin sponge pledgets/slurry, polyvinyl alcohol particles, and coils are the embolic agents of choice for PSE [ 14]. PVA particles (300 to 700-μm size) suspended in contrast medium mixed with antibiotic is most commonly used. Similarly, glue (N-butyl cyanoacrylate, NBCA) with lipiodol in concentration ranging from 1:5 to 1:7 may be used to embolize the required arteries. However, special care should be taken while using glue as an embolizing agent (optimal glue concentration, avoiding ionic solutions, and flushing of the catheter with dextrose solution) to prevent nontarget embolization and polymerization of glue over the catheter tip.
The volume of splenic infarction is the determinant for therapeutic effect versus the complication of PSE when used to treat complications of portal hypertension. Ideally, 30% to 60% of splenic volume embolization is adequate however, the volume to be embolize depends upon the clinical condition of the patient. However, opinion regarding the ideal volume to be embolized is still controversial (Fig. 16.1). Small volume reduction does not improve platelet count whereas, large volume necrosis carries high risks of complications and/or abscess formation [8]. Various studies recommend that the first embolization volume should be less than 70% of the total spleen mass, to decrease the probabilities of complications [8, 15, 16].
One study by Harned et al. [17] found that embolization leading to infraction of 30%–40% of the splenic parenchyma significantly lowers the morbidity, although there is a lesser degree of improvement in thrombocytopenia. Conservative cautious approach is advisable at initial embolization, especially in advance liver disease, and a second session of embolization, if required, may be performed subsequently.
16.6 Post-Procedure Care
Antibiotic should be started prior to the procedure and continued in perioperative period to prevent development of infection in the infracted splenic parenchyma. Routine hospital stay consists of 24 to 48 h; however the patients should be followed for a week after the procedure. Some of the patients may require continuous anti-inflammatory medications according to the symptoms.
16.7 Complications
Most of the patients will have minor side effect in the form of fever, nausea, and pain as part of post-embolization syndrome. Post-embolization syndrome is common and may be as high as 30% but generally resolve without sequelae [18]. These symptoms can be treated symptomatically with narcotics and antiemetics. Some patient may have anorexia, vomiting, pleural effusion, ascites, and ileus. Koconis et al. reported that 73% had developed a serious complication in form of abscess, pleural effusion, ascites, pneumonia, pulmonary embolus, portal vein thrombosis, liver failure, and death when the embolization volume was 70% or more [8]. Symptomatic splenic abscess may require percutaneous drainage. Large splenic infarct area and advanced liver disease (Child-Pugh class C) are risk factors for development of complications after PSE [19].
There have been few, but encouraging reports using radiofrequency ablation and microwave ablation for the treatment of hypersplenism/splenomegaly [20, 21]. In future these techniques may be used for splenic parenchyma reduction for the treatment of portal hypertension.
16.8 Conclusion
Partial splenic embolization alone or in combination with other treatments is a promising and effective alternative option in the management of portal hypertension in relatively advanced liver disease. PSE may help in decreasing the formation of ascites and esophageal variceal bleeding, and increasing hematologic indices and liver function. Patient selection, intervention technique, and post-procedure care are the key to success.
References
Maddison FE. Embolic therapy of hypersplenism. Investig Radiol. 1973;8:280–1.
Kutti J, Weinfeld A, Westin J. The relationship between splenic platelet pool and spleen size. Scand J Haematol. 1972;9:351–4.
Xu RY, Liu B, Lin N. Therapeutic effects of endoscopic variceal ligation combined with partial splenic embolization for portal hypertension. World J Gastroenterol. 2004;10:1072–4.
Taniai N, Onda M, Tajiri T, Toba M, Yoshida H. Endoscopic variceal ligation (EVL) combined with partial splenic embolization (PSE). Hepato-Gastroenterology. 1999;46:2849–53.
Chikamori F, Kuniyoshi N, Kawashima T, Shibuya S, Takase Y. Combination treatment of partial splenic embolization, endoscopic embolization and transjugular retrograde obliteration for complicated gastroesophageal varices. Hepato-Gastroenterology. 2004;51:1506–9.
Palsson B, Hallen M, Forsberg AM, Alwmark A. Partial splenic embolization: long-term outcome. Langenbeck’s Arch Surg. 2003;387:421–6.
Ohmoto K, Yoshioka N, Tomiyama Y, Shibata N, Takesue M, Yoshida K, Kuboki M, Yamamoto S. Improved prognosis of cirrhosis patients with esophageal varices and thrombocytopenia treated by endoscopic variceal ligation plus partial splenic embolization. Dig Dis Sci. 2006;51:352–8.
Koconis KG, Singh H, Soares G. Partial splenic embolization in the treatment of patients with portal hypertension: a review of the English language literature. J Vasc Interv Radiol. 2007;18:463–81.
Hirai K, Kawazoe Y, Yamashita K, et al. Transcatheter partial splenic arterial embolization in patients with hypersplenism: a clinical evaluation as supporting therapy for hepatocellular carcinoma and liver cirrhosis. Hepato-Gastroenterology. 1986;33(3):105–8.
Murata K, Shiraki K, Takase K, Nakano T, Tameda Y. Long term follow-up for patients with liver cirrhosis after partial splenic embolization. Hepato-Gastroenterology. 1996;43(11):1212–7.
Sakata K, Hirai K, Tanikawa K. A long-term investigation of transcatheter splenic arterial embolization for hypersplenism. Hepato-Gastroenterology. 1996;43(7):309–18.
Rios R, Sangro B, Herrero I, Quiroga J, Prieto J. The role of thrombopoietin in the thrombocytopenia of patients with liver cirrhosis. Am J Gastroenterol. 2005;100(6):1311–6.
Firat A, Boyvat F, Moray G, Aytekin C, Karakayali H, Haberal M. Comparison of two different percutaneous splenic artery interventions in the treatment of hypersplenism: preliminary report. Transplant Proc. 2005;37(2):1094–8. https://doi.org/10.1016/j.transproceed.2004.12.171.
Madoff DC, Denys A, Wallace MJ, et al. Splenic arterial interventions: anatomy, indications, technical considerations, and potential complications. Radiographics. 2005;25(supplement 1):S191–211.
N’Kontchou G, Seror O, Bourcier V, et al. Partial splenic embolization in patients with cirrhosis: efficacy, tolerance and long-term outcome in 32 patients. Eur J Gastroenterol Hepatol. 2005;17(2):179–84.
Obatake M, Muraji T, Kanegawa K, Satoh S, Nishijima E, Tsugawa C. A new volumetric evaluation of partial splenic embolization for hypersplenism in biliary atresia. J Pediatr Surg. 2001;36(11):1615–6.
Harned RK 2nd, Thompson HR, Kumpe DA, Narkewicz MR, Sokol RJ. Partial splenic embolization in five children with hypersplenism: effects of reduced-volume embolization on efficacy and morbidity. Radiology. 1998;209:803–6.
Piffaretti G, Tozzi M, Lomazzi C, Rivolta N, Riva F, Caronno R, Castelli P. Splenic artery aneurysms: postembolization syndrome and surgical complications. Am J Surg. 2007;193(2):166–70.
Hayashi H, Beppu T, Okabe K, Masuda T, Okabe H, Baba H. Risk factors for complications after partial splenic embolization for liver cirrhosis. Br J Surg. 2008 Jun;95(6):744–50.
Feng K, Ma K, Liu Q, Wu Q, Dong J, Bie P. Randomized clinical trial of splenic radiofrequency ablation versus splenectomy for severe hypersplenism. Br J Surg. 2011 Mar;98(3):354–61. https://doi.org/10.1002/bjs.7367.
Assal F, El Kassas M, Esmail E, Elbadry AA, Abousaif S, Mahdy R, Elfert A. Microwave ablation in the spleen versus partial splenic artery embolisation: a new technique for hypersplenism in cirrhosis. Arab J Gastroenterol. 2017 Mar;18(1):25–9. https://doi.org/10.1016/j.ajg.2017.01.001.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Patidar, Y. (2021). Interventions for Portal Hypertension: Splenic Artery Embolization. In: Mukund, A. (eds) Basics of Hepatobiliary Interventions. Springer, Singapore. https://doi.org/10.1007/978-981-15-6856-5_16
Download citation
DOI: https://doi.org/10.1007/978-981-15-6856-5_16
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-6855-8
Online ISBN: 978-981-15-6856-5
eBook Packages: MedicineMedicine (R0)