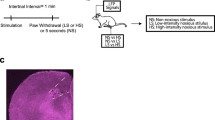
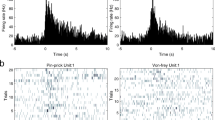
Abstract
Acute pain is triggered by noxious stimuli that induce changes in neural responses of specific neural circuits at both single neuron and population levels. Detecting acute pain signals may provide a closed-loop feedback signal for neuromodulation to achieve pain relief. Human neuroimaging studies have identified circuit changes in pain states, and brain regions identified by such methods include the primary somatosensory cortex (S1) and anterior cingulate cortex (ACC), which are thought to provide key information on sensory and affective components of pain, respectively. In animal and human studies, methods for detecting acute pain signals in real time have been developed based on invasive or noninvasive neural recordings. This chapter provides an overview of several statistical machine learning and signal processing approaches for detecting acute pain signals based on spikes and/or local field potentials (LFPs) recorded from freely behaving rats. To facilitate closed-loop neuroscience experiments or brain-machine interface (BMI), real-time computational consideration is emphasized. We present various detection strategies and illustrations using the in vivo electrophysiological recordings from the rat ACC and/or S1 and discuss important issues related to robust detection and closed-loop neuromodulation in future practice.
Similar content being viewed by others
References
Aminikhanghahi, S., Cook, D.J.: A survey of methods for time series change point detection. Knowl. Inf. Syst. 51, 339–367 (2017)
Bialek, W., Rieke, F., de Ruyter van Steveninck, R.R., Warland, D.: Reading a neural code. Science 252, 1854–1857 (1991)
Bishop, C.M.: Pattern Recognition and Machine Learning. Springer (2011)
Brodersen, K.H., Wiech, K., Lomakina, E.I., Lin, C.S., et al.: Decoding the perception of pain from fMRI using multivariate pattern analysis. Neuroimage 63, 1162–1170 (2012)
Brown, E.N., et al.: A statistical paradigm for neural spike train decoding applied to position prediction from ensemble firing patterns of rat hippocampal place cells. J. Neurosci. 18, 7411–7425 (1998)
Brown, E.N., Barbieri, R., Eden, U.T., Frank, L.M.: Likelihood methods for neural data analysis. In: Feng, J. (ed.) Computational Neuroscience: A Comprehensive Approach, pp. 253–286. CRC Press (2003)
Bushnell, M.C., Ceko, M., Low, L.A.: Cognitive and emotional control of pain and its disruption in chronic pain. Nat. Rev. Neurosci. 14, 502–511 (2013)
Buzsaki, G., Anastassiou, C.A., Koch, C.: The origin of extracellular fields and currents-EEG, ECoG, LFP and spikes. Nat. Rev. Neurosci. 13, 407–420 (2012)
Chen, Z.: Bayesian filtering: from Kalman filters to particle filters, and beyond. Technical report, McMaster University (2003)
Chen, Z., Sarma, S. (eds.): Dynamic Neuroscience. Springer (2018)
Chen, Z., Wang, J.: Statistical analysis of neuronal population codes for encoding acute pain. In: Proceedings of IEEE ICASSP, Shanghai, pp. 829–833. IEEE (2016)
Chen, Z., Ohara, S., Cao, J., Vialatte, F., Lenz, F.A., Cichocki, A.: Statistical modelling and analysis of laser-evoked potentials of electrocorticogram recordings from awake humans. Comput. Intell. Neurosci. 2007, 10479 (2007)
Chen, Z., Zhang, Q., Tong, A.P.S., Mander, T.R., Wang, J.: Deciphering neuronal population codes for acute thermal pain. J. Neural Eng. 14, 036023 (2017)
Chen, Z., Hu, S., Zhang, Q., Wang, J.: Quickest detection for abrupt changes in neuronal ensemble spiking activity using model- based and model-free approaches. In: Proceedings of IEEE Neural Engineering Conference, Shanghai, pp. 481–485. IEEE (2017)
Copits, B.A., Pullen, M.Y., Gereau, R.W. IV.: Spotlight on pain: optogenetic approaches for interrogating somatosensory circuits. Pain 157, 2424–2433 (2016)
Dale, J., Zhou, H., Zhang, Q., Martinez, E., Hu, S., Liu, K., Urien, L., Chen, Z., Wang, J.: Scaling up cortical control to inhibit chronic pain. Cell Rep. 23, 1301–1313 (2018)
Daou, I., Tuttle, A.H., Longo, G., et al.: Remote optogenetic activation and sensitization of pain pathways in freely moving mice. J. Neurosci. 33, 18631–18640 (2013)
Davis, K.D., Flor, H., Greely, H.T., Iannetti, G.D., Mackey, S., et al.: Brain imaging tests for chronic pain: medical, legal and ethical issues and recommendations. Nat. Rev. Neurol. 13, 624–638 (2017)
Doucet, A., Godsill, S., Andrieu, C.: On sequential Monte Carlo sampling methods for Bayesian filtering. Statist. Comput. 10, 197–208 (2000)
Geuter, S., Gamer, M., Onat, S., Büchel, C.: Parametric trial-by-trial prediction of pain by easily available physiological measures. Pain 155, 994–1001 (2014)
Gordon, N., Salmond, D., Smith, A.F.M.: Novel approach to nonlinear/non-Gaussian Bayesian state estimation. IEE Proc. Part F Radar Sonar Navig. 140, 107–113 (1993)
Gu, L., Uhelski, M.L., Anand, S., Romero-Ortega, M., et al.: Pain inhibition by optogenetic activation of specific anterior cingulate cortical neurons. PLoS One 10, e0117746 (2015)
Guo, X., Zhang, Q., Singh, A., Wang, J., Chen, Z.: Granger causality analysis of rat cortical functional connectivity in pain. J. Neural Eng. 17, 016050 (2020)
Hastie, T., Tibshirani, R., Friedman, J.: The Elements of Statistical Learning, 2nd edn. Springer (2008)
Hu, S., Zhang, Q., Wang, J., Chen, Z.: A real-time rodent neural interface for deciphering acute pain signals from neuronal ensemble spike activity. In: Proceedings of Asilomar Conference on Signals, Systems and Computers, pp. 93–97. IEEE (2017)
Hu, S., Zhang, Q., Wang, J., Chen, Z.: Real-time particle filtering and smoothing algorithms for detecting abrupt changes in neural ensemble spike activity. J. Neurophysiol. 149, 1394–1410 (2018)
Hu, S., Ciliberti, D., Grosmark, A.D., Michon, F., Ji, D., Penagos, H., Buzsaki, G., Wilson, M.A., Kloosterman, F., Chen, Z.: Real-time readout of large-scale unsorted neural ensemble place codes. Cell Rep. 25, 2635–2642 (2018)
Huang, G., Xiao, P., Hung, Y.S., Iannetti, G.D., Zhang, Z.G., Hu, L.: A novel approach to predict subjective pain perception from single-trial laser-evoked potentials. Neuroimage 81, 283–293 (2013)
Iyer, S.M., Montgommery, K.L., Towne, C., et al.: Virally mediated optogenetic excitation and inhibition of pain in freely moving nontransgenic mice. Nat. Biotech. 32, 274–278 (2014)
Iyer, S.M., Vesuna, S., Ramakrishnan, C., Huynh, K., et al.: Optogenetic and chemogenetic strategies for sustained inhibition of pain. Sci. Rep. 6, 30570 (2016)
Koepcke, L., Ashida, G., Kretzberg, J.: Single and multiple change point detection in spike trains: comparison of different CUSUM methods. Front. Syst. Neurosci. 10, 51 (2016)
Kuo, C.C., Yen, C.T.: Comparison of anterior cingulate and primary somatosensory neuronal responses to noxious laser-heat stimuli in conscious, behaving rats. J. Neurophysiol. 94, 1825–1836 (2005)
Lancaster, J., Mano, H., Callan, D., Kawato, M., et al.: Decoding acute pain with combined EEG and physiological data. In: Proceedings of 8th International IEEE/EMBS Conference on Neural Engineering (NER) (2017). https://doi.org/10.1109/NER.2017.8008404
Lee, M., Manders, T.R., Eberle, S.E., et al.: Activation of corticostriatal circuitry relieves chronic neuropathic pain. J. Neurosci. 35, 5247–5259 (2015)
Lotsch, J., Ultsch, A.: Machine learning in pain research. Pain 159, 623–630 (2018)
Macke, J.H., Buesing, L., Sahani, M.: Estimating state and parameters in state 809 space models of spike trains. In: Chen, Z. (ed.) Advanced State Space Methods in Neural and Clinical Data. Cambridge University Press (2015)
Malladi, R., Kalamangalam, G.P., Aazhang, B.: Online Bayesian change point detection algorithms for segmentation of epileptic activity. In: Proceedings of Asilomar Conference on Signals, Systems & Computers, pp. 1833–1837. IEEE (2013)
Mosqueiro, T., Strube-Bloss, M., Tuma, R., Pinto, R., Smith, B.H., Huerta, R.: Non-parametric change point detection for spike trains. In: Proceedings of Annual Conference on Information Sciences and Systems (2016)
Mouraux, A., Iannetti, G.D.: The search for pain biomarker in the human brain. Brain 141, 3290–3307 (2018)
Murphy, K.P.: Machine Learning: A Probabilistic Perspective. MIT Press, Cambridge, MA (2012)
Paninski, L., Ahmadian, Y., Ferreira, D.G., Koyama, S., Rad, K.R., Vidne, M., Vogelstein, J.T., Wu, W.: A new look at state-space models for neural data. J. Comput. Neurosci. 29, 107–126 (2010)
Pillow, J.W., Ahmadian, Y., Paninski, L.: Model-based decoding, information estimation, and change-point detection techniques for multineuron spike trains. Neural Comput. 23, 1–45 (2011)
Poor, H.V., Hadjiliadis, O.: Quickest Detection. Cambridge University Press (2009)
Rey, H.G., Pedreira, C., Quian Quiroga, R.: Past, present and future of spike sorting techniques. Brain Res. Bull. 119, 106–117 (2015)
Scholkopf, B., Smola, A.J.: Learning with Kernels: Support Vector Machines, Regularization, Optimization, and Beyond. MIT Press, Cambridge, MA (2001)
Schulz, E., Zherdin, A., Tiemann, L., Plant, C., Ploner, M.: Decoding an individual’s sensitivity to pain from the multivariate analysis of EEG data. Cereb. Cortex 22, 1118–1123 (2012)
Shirvalkar, P., Veuthey, T.L., Dawes, H.E., Chang, E.F.: Closed-loop deep brain stimulation for refractory chronic pain. Front. Compt. Neurosci. 12, 18 (2018)
Smith, A.C., Brown, E.N.: Estimating a state-space model from point process observations. Neural Comput. 15, 965–991 (2003)
Sun, G., Wen, Z., Ok, D., Doan, L., Wang, J., Chen, Z.: Detecting acute pain signals from human EEG. J. Neurosci. Methods 347, 108964 (2020)
Truong, C., Oudre, L., Vayatis, N.: Selective review of offline change point detection methods. Sig. Proc. 167, 107299 (2020)
Ung, H., Brown, J.E., Johnson, K.A., Younger, J., Hush, J., Mackey, S.: Multivariate classification of structural MRI data detects chronic low back pain. Cereb. Cortex 24, 1037–1044 (2012)
Urien, L., Xiao, Z., Dale, J., Bauer, E., Chen, Z., Wang, J.: Rate and temporal coding mechanisms in the anterior cingulate cortex for pain anticipation. Sci. Rep. 8, 8298 (2018)
Vierck, C.J., Whitsel, B.L., Favorov, O.V., Brown, A.W., Tommerdahl, M.: Role of primary somatosensory cortex in the coding of pain. Pain 154, 334–344 (2013)
Vijayakumar, V., Case, M., Shirinpour, S., He, B.: Quantifying and characterizing tonic thermal pain across subjects from EEG data using random forest models. IEEE Trans. Biomed. Eng. 64, 2988–2966 (2017)
Wager, T.D., Atlas, L.Y., Lindquist, M.A., Roy, M., Woo, C.-W., Kross, E.: An fMRI-based neurologic signature of physical pain. N. Engl. J. Med. 368, 1388–1397 (2013)
Wang, X., Poor, V.: Robust multiuser detection in non-Gaussian channels. IEEE Trans. Sig. Proc. 50, 289–305 (1999)
Wiech, K., Ploner, M., Tracey, I.: Neurocognitive aspects of pain perception. Trends Cogn. Sci. 12, 306–313 (2008)
Wu, W., Gao, Y., Bienenstock, E., Donoghue, J.P., Black, M.J.: Bayesian population decoding of motor cortical activity using a Kalman filter. Neural Comput. 18, 80–118 (2006)
Xiao, Z., Hu, S., Zhang, Q., Tian, X., Chen, Y., Wang, J., Chen, Z.: Ensembles of change-point detectors: implications for real-time BMI applications. J. Comput. Neurosci. 46, 107–124 (2019)
Xiao, Z., Martinez, E., Kulkarni, P., Zhang, Q., Hou, Q., Rosenberg, D., Talay, R., Shalot, L., Zhou, H., Wang, J., Chen, Z.: Cortical pain processing in the rat anterior cingulate cortex and primary somatosensory cortex. Front. Cell. Neurosci. 13, 165 (2019)
Zhang, Q., Mander, T.R., Tong, A.P.S., Yang, R., Garg, A., Martinez, E., Zhou, H., Dale, J., Goyal, A., Urien, L., Yang, G., Chen, Z., Wang, J.: Chronic pain induces generalized enhancement of aversion. eLife 6, e25302 (2017)
Zhang, Q., Xiao, Z., Huang, C., Hu, S., Kulkarni, P., Martinez, E., Tong, A.P.S., Garg, A., Zhou, H., Chen, Z., Wang, J.: Local field potential decoding of the onset and intensity of acute thermal pain in rats. Sci. Rep. 8, 8299 (2018)
Zhang, Q., Hu, S., Talay, R., Xiao, Z., Rosenberg, D., Li, A., Caravan, B.,, Liu, Y., Sun, G., Singh, A., Gould, J.D., Chen, Z., Wang, J.: A prototype closed-loop brain-machine interface for study and treatment of pain. Nat. Biomed. Eng. (2020). https://doi.org/10.1038/s41551-021-00736-7
Zhou, H., Zhang, Q., Martinez, E., et al.: Ketamine reduces aversion in rodent pain models by suppressing hyperactivity of the anterior cingulate cortex. Nat. Commun. 9, 3751 (2018)
Zhou, H., Zhang, Q., Martinez, E., Dale, J., Robinson, E., Huang, D., Wang, J.: A novel neuromodulation strategy to enhance the prefrontal control to treat pain. Mol. Pain 15, 1744806919845739 (2019)
Acknowledgements
The author likes to thank all coauthors who had contributed to previous publications. The work was supported by the NIH grant R01-NS100065 and NSF-CBET grant #1835000.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Section Editor information
Rights and permissions
Copyright information
© 2022 Springer Nature Singapore Pte Ltd.
About this entry
Cite this entry
Chen, Z.S. (2022). Real-Time Detection of Acute Pain Signals Based on Spikes/LFP. In: Thakor, N.V. (eds) Handbook of Neuroengineering. Springer, Singapore. https://doi.org/10.1007/978-981-15-2848-4_72-2
Download citation
DOI: https://doi.org/10.1007/978-981-15-2848-4_72-2
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-2848-4
Online ISBN: 978-981-15-2848-4
eBook Packages: Springer Reference EngineeringReference Module Computer Science and Engineering