Abstract
Clean water is vital for supporting human life and the ecosystem. However, the laxity and mismanagement of water resources have endangered the availability of fresh water significantly. Water pollution and associated diseases claim around 2.1 million human lives every year. The outbreak of water-related microbial infections such as diarrhoea, typhoid, and cholera are the primary cause of the loss of lives. Though there has been remarkable progress in the control and prevention of infectious diseases, microbial risks remain a leading cause of human mortality in India, and the rest of the world and children are the worst affected. In this context, a comprehensive analysis of the source, occurrence, fate, and control of biological contaminants in dirking water is of utmost relevance. The rapid and early detection of the pathogenic organism is also of importance in mitigating the menace. This chapter elucidates the growing significance to address the issue of microbial contamination in drinking water and its associated health implications from the past to the present, recent developments in the technologies for the detection, analysis and the remediation of pathogens in the water.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
11.1 Introduction
11.1.1 Microbial Hazards: Growing Concern
Freshwater is a complex resource and is linked to almost everything in the world. Its adequate availability at the point of use is a precondition for the existence of humankind and the sustainability of the planet. Quality and quantity are the two significant attributes of this indispensable resource. Though the earth is covered with 70% of the water, only 2.53% of the water is freshwater and in which <0.1% is available for human consumption (USBR 2017).
According to a United Nations (UN) report, the water consumption rate has increased twice than the population growth (Un, n.d.). The Global monitoring body, World Health Organization (WHO) and United Nations International Children’s Emergency Fund (UNICEF) joint monitoring programme for water supply, sanitation and Hygiene (JMP) has established reports on current scenario of issues in water supply, sanitation and hygiene, since 1990. The projections to the next few decades in context to water stress and scarcity across the globe is assessed and evaluated by these organizations (Fig. 11.1).
The 2017 update of WHO/UNICEF Joint Monitoring Programme for Water Supply, Sanitation and Hygiene (JMP) reports that in 2015, 2.1 billion individuals lacked “safely managed drinking water” as shown in Fig. 11.2 (UNICEF 2017). The figure also shows that 11% of the global population (844 million people) lacked even a basic drinking water service. Globally, there are about 423 million people collect water from unprotected groundwater, and 159 million people use surface water directly. Cities and towns pose a special and unique water challenge, as they are expected to be home for 66 percent of the world’s population by 2050 (UN 2018). The impact of poor water quality on people relying on these sources not only limits the access to safe water but also increases the threat to human health.
Globally, 80% of wastewater flows back into the ecosystem without being treated, and 1.8 billion people use untreated water supply as the source of drinking water, putting them at risk of contracting waterborne diseases (UNESCO 2017). Water-related illnesses including, dysentery, typhoid, cholera, and schistosomiasis, are prevalent across developing countries, and cholera contributes top of the list outbreaks in 132 countries (WHO 2016). According to a report published in 2015, the waterborne disease remains as an increasing threat among vulnerable and disadvantaged groups across the globe, especially in low-income nations, where 1 in 25 persons is affected due to diarrhoeal diseases. The estimates shows 60% of children under the age of five are affected (UNICEF 2016). Now, waterborne diseases stand as the leading cause of disease and death and accounting 3.4 million loss of lives worldwide (WHO 2001). WHO (2016) reports that diarrhoea is one of the top ten global reasons for death and the second leading cause of death in low-income countries. Around 1.8 million deaths occurred due to diarrhoeal diseases worldwide, and the crude death rate is 58 per 100,000 populations in low-income countries (WHO 2004). Every year around 1.3 to 4 million cholera cases and 21,000 to 143,000 associated deaths are reported worldwide (WHO 2016). According to Global Estimates, there are 20 million reported cases of hepatitis E virus (HEV) infections, which also includes 3.3 million symptomatic cases of hepatitis E infection. The HEV caused approximately 44,000 mortalities in the year 2015, out of which 3.3% were due to viral hepatitis (Rein et al. 2012). As per the Environment and Health Information System, around 13,548 children (0–14 years old) dies in Europe every year due to waterborne diseases (WHO 2007). These statistical facts demonstrate that presence of biological pollutants in water, among others, is a growing concern in both developing and developed countries and it requires adequate attention to meet the safe drinking water needs of the population.
Since the inception of identifying the reasons for human health deterioration due to microbial contamination in water, there have been various theories and subsequent experimental findings to understand the occurrence, health impact, and fate of pathogens in water. For the past few decades, several efforts have been taken towards the development of robust, efficient, and affordable detection and remediation technology in curtailing the waterborne diseases caused by pathogens. Technology to mitigate the issue can be categorised into detection specific and remediation specific.
This chapter is composed primarily to enhance the knowledge on the importance of microbial contamination in drinking water by understanding the occurrences and sources of pathogens. A detailed review of literature is presented on the growing trends in the field of pathogenic detection techniques and remediation technologies from the past to the recent highlighting their principle, mechanism, applications, and limitations through illustrations and discussions thereof. The effort has also been taken to discern the real challenges in implementing these technologies, which may eventually be utilized in bridging the gap between the lab and the field.
11.1.2 Occurrences and Sources of Microbial Contaminants in Water
Presence of microbial pollutants such as bacteria, virus, protozoa, and helminths pose a severe threat to the quality of freshwater. The typical characteristics, source, and impact on human health of these organisms are summarised in Table 11.1. Their occurrences in water bodies vary depending on several factors. These include various chemical and physical characteristics of the catchment area, the intensity and extent of anthropogenic activities, and the domestic animal discharge. However, the human activities such as discharge of untreated or partially treated municipal wastewater, poor sanitation and hygiene, open defecation, industrial and agricultural wastes, and solid/semisolid refuse are the major sources of concern (Planning Commission 2002). A schematic illustration of routes of microbial contamination in water is shown in Fig. 11.3.
As per the 2002 Planning Commission report, there is a higher threat of waterborne diseases in rural areas caused by water contamination due to poorly maintained water and sewer networks, unscientific disposal of solid wastes, poor healthy sanitation and personal hygienic practices. The discharge of urban sewage is identified as a major source of contamination of Indian surface waters (Murty and Kumar 2011). The estimate shows 80% of surface water bodies in the country is polluted by domestic sewerage (Dey 2015). The Arya et al. (2019) report reveals that India produces 61,948 million litres per day (MLD) of urban sewage. The data also shows that more than 70% of the sewage is let out into the environment untreated (Arya et al. 2019).
Groundwater is a prominent source of drinking water to at least 50% of the population worldwide that also accounts for 43% of the water utilized for irrigation (Faures et al. 2001). Unlike surface water, the sub-surface water is considered less vulnerable to microbial pollution due to the barrier effects provided by the covering soil. There are higher chances of subsurface water contamination when these overlaying barriers are breached, allowing exposure to underground pollution sources, such as soak pits, toilets, and sewer lines containing municipal or commercial or industrial wastes. Though the typical presence of human enteric organism is less in groundwater, the pathogens of concern are faecal viruses, which has the potential to enter groundwater system through the porous soil matrices due to their relatively small size. The correlation of groundwater contamination to the global occurrences of the waterborne disease cannot be made typically, as there are various transmission routes. So the exposure-risk relationships are often unclear. However, it is established that several groundwater sources are contaminated with pathogenic organisms and is also responsible for waterborne diseases (Rivera-Jaimes et al. 2018). A study shows that water samples collected from the wells in proximity to the sources of untreated wastewater had higher counts of coliform and faecal coliform, making it unsuitable for both drinking and irrigation purposes (Blumenthal et al. 2000). Saha et al. (2018) and Dey et al. (2017) reported that in northwest Bangladesh, the shallow aquifers are microbiologically contaminated than deep aquifer (Saha et al. 2018; Dey et al. 2017). A study conducted in Kanpur, India documented waterborne disease at an incidence rate of 80.1 per 1000 population (Trivedi et al. 1971). Amongst the shallow wells used by the residents as a source of drinking water, 70% were found to be contaminated by pathogens.
Rural areas of the developing countries using groundwater as a source for drinking are more vulnerable to waterborne diseases than the ones using piped water supplies. The bacteriological quality analysis including Total Coliform, Faecal Coliform, and Faecal streptococci showed that the collected groundwater samples from Triffa aquifer basin, Eastern Morocco were contaminated due to unprotected septic tanks and the wastewater dumped in the upstream end of river Cheraa wadi (Yahya et al. 2017). An experimental investigation made by Venkatesan et al. (2014) to study the impact of flooding on microbial contamination in groundwater at Chennai, India after a major flood event revealed higher counts of coliform in subsurface water sources at most affected areas. The rapid escalation in the microbial growth was attributed to the contaminated storm water runoff entering into Adyar River, Tamil Nadu, India (Gowrisankar et al. 2017). The residences with on-site septic systems were likely more affected due to the infiltration of the contaminated river into the groundwater sources (Jamieson et al. 2003).
The increase in levels of pathogenic contamination in estuaries and marine environment is also poses a threat to public health. In context to marine water, the prominent reasons for microbial contamination are failures in septic systems, discharges of sewage from shoreline outfalls, farm animal wastes, and runoff from naturally vegetated areas. The storm water runoffs from urban, commercial, and industrial lands, the practice of open defecation near coastal areas are also responsible for contamination of marine waters (Pandey et al. 2014). World Ocean Network reports that around 90% of wastewater and 70% of industrial waste is being discharged into oceans by the developing countries (Vandeweerd et al. 2002). Estuaries located adjacent to residential areas, when used as a mode of transportation and for recreational activities can cause a significant impact on pathogen levels (Schriewer et al. 2010). The pathogens, including Salmonella, Vibrio cholerae, Cryptosporidium, Giardia, and Campylobacter spp. are reported in estuaries (Rhodes and Kator 1990).
The other potential source of pollution is due to the regrowth of microbes in the water distribution network (Shaheed et al. 2014). In the United States, around 10% of outbreaks are caused by contaminated water due to the improper water distribution network (Craun et al. 2010). The corrosion and poor surface finish in the water supply pipelines enables enhanced colonization of microorganisms and the formation of biofilms (Rakić 2018). These biofilms can act as a short or long-term habitats for pathogenic organisms, such as Pseudomonas aeruginosa, Escherichia coli, Legionella spp., Campylobacter spp., noroviruses, adenoviruses, rotaviruses, and parasitic protozoa (Cryptosporidium parvum) (Wingender and Flemming 2011). The emergence of new pathogens, mutants of the existing pathogens, and the presence of multi-drug resistance species are also reasons for concern.
11.1.3 Transmission of Waterborne Diseases
It is a fact that microbial hazard is a principal cause of human mortality in the developing world. There are various groups of pathogenic microorganisms, and they have different modes of transmission, as is shown in Fig. 11.4. Drinking water is observed to be the only carrier of the faecal-oral route of pathogenic transmission (WHO 2011).
The transmission characteristics of pathogens can be categorized based on latency, persistence, infectivity, and the ability to multiply. Latency is the lag time between excretion of a pathogen and the stage at which it becomes infective to a new host. Typically, protozoa, bacteria, and enteric viruses have no latent period (Feachem et al. 1983). The most of helminths require a different latent period either for eggs to progress to the transmittable stage or to pass through an intermediate form to complete their life cycles (Cotruvo et al. 2004). Persistence is determined by the span of time that a pathogenic organism exists in the environment outside a human host in viable condition. Persistent microbes can travel through a prolonged route, viz., through a sewage treatment system and can still be infectious to human living away from the original host. In general, persistence increases in the order: bacteria > protozoa > viruses > helminths, whose persistence is measured in months. The infective dose refers to the microbial concentration that can cause infection upon ingestion. Usually, the minimum infective doses for viruses and protozoa are less than that of bacteria.
A summary of various waterborne diseases linked to the protozoa and helminths, its relative infectivity, and persistence in water are discussed in Table 11.1. The pathogens that are not listed in Table 11.1 can also transmit by water, and the list is not complete.
11.1.4 Drinking Water Safety Guidelines
The purpose of the disseminated WHO standard guidelines is to enable countries and regions to develop their own standards conforming to the regulation. It suggests that immediate action must be taken if E. coli is detected in drinking water. Monitoring the levels of E. coli and faecal coliforms is a common method in the quantification of the pathogen loads in water bodies (Feachem et al. 1983). For decades, public health experts and scientists have assessed water quality in rivers, estuaries, and coastal waters in terms of faecal coliforms and E. coli (Pandey and Soupir 2013). However, E. coli cannot predict the existence of all pathogenic organisms. For example, Cryptosporidium oocysts may survive chlorine disinfection and may be present in the absence of E. coli, showing the limitation of using E. coli as a potential indicator for faecal contamination. However, E. coli is the designated WHO indicator for reliable diagnosis of microbial quality of the water (WHO 2011). The guideline values for assessing the microbial quality are given in Table 11.2.
Despite of establishing definite standard regulations for safe supply of water, the concern of detecting and monitoring these pathogens in water samples collected from various sources have been still a challenging task for both developed and developing countries. The following discussions will describe the existing methods of monitoring and removing pathogenic organisms in water.
11.2 Detection and Analysis of Pathogenic Organisms in Water
Efficient testing and fast detection of pathogenic organisms are vital in the management of water-borne illness. It is the main checkpoint in eliminating the pathogens in drinking water, food, and other biological samples. It also pays a significant role in diagnosing and preventing diseases (Vidic et al. 2017). A typical detection technique should be sensitive, rapid, and affordable. There are several routes, such as culture/growth, optical, molecular, and bio-sensing based are used to detect the pathogenic organism in these samples. The major methods under each detection technique are briefly discussed below. Table 11.3 presents a summary of the testing methods.
11.2.1 Culture/Growth Based Method
Culture or growth based technique is a traditional method employed for the detection of pathogenic organisms in the water. The majority of the testing for bacteria detection is done through this conventional approach. It involves growing and isolation of organisms on Petri-plates containing growth-media, followed by biochemical tests to confirm the presence of pathogenic microorganisms. It is a time-consuming technique, and typically take 5–7 days to obtain the results (Rajapaksha et al. 2019). It is not suitable to detect organisms which are viable but present in the non-culturable state. However, the traditional culture-dependent method is regarded as the standard method for the detection of pathogens, and it is still being used as a regulatory requirement by water treatment companies and laboratories to monitor the microbial quality of drinking water (American Public Health Association, American Water Works Association 1989). The estimation of the most probable number (MPN) or multiple fermentation is a commonly practiced growth-based method to find the concentration of viable microorganisms in the water sample. It is a statistical method that relies on the principle of extinction dilution for testing the quality of water and assesses its suitability for human consumption. The technique typically identifies the presence of an indicator organism of faecal origin to establish the existence of pathogenic microorganisms (Munoz and Silverman 1979). It works based on the principle of fermentation of lactose to produce the acid as well as gas. The presence of coliform is showed by the colour change of the medium, by a change in pH, or by the collection of gas in inverted Durham’s kept in the test medium. The total coliforms can be determined by counting the number of tubes showing both colour change and production of gas (Fung and Miller 1970). The MPN analysis is usually performed in 3 steps, including presumptive, confirmatory, and completed test. The presumptive test is the first step and is carried out to identify the presence or abscess of the coliform organism in the water sample. If this screening test is negative, the water sample is considered free from pathogens. If the test is positive, further confirmatory analysis is required to ratify the faecal origin of the coliform organisms. The completed test is performed to check and eliminate the false positive test. These steps are illustrated in Fig. 11.5.
The method is time-consuming and takes normally up to 72 h for obtaining the results. The development of Membrane Filtration (MF) technique shortens the process and reduces the completion time to 24 h. In this technique, the sample is allowed to pass through the membrane filter (pore size of 0.45 µm), and the membrane containing the trapped bacteria is transferred on to a Petri-plate containing the nutrient agar medium. The results were obtained by counting the bacterial colonies, which is grown on the incubated Petri-plates. The number of bacterial colonies grown on the agar medium is counted and is represented as CFU/ml.
The technique is highly effective for assessing the performance of chlorination as it removes the bactericidal agents through filters (Tankeshwar 2010). The process flow diagram is presented in Fig. 11.6. Apart from standard nutrient agar medium, chromogenic agar medium such as Eosin Methylene blue agar (Leininger et al. 2001) and Maccconkey agar (March and Ratnam 1986) is also used for differentiating coliforms from faecal coliforms and isolation of members of family Enterobacteriaceae, respectively. Furthermore, agar medium is available with chromogenic and fluorogenic substrates that helps in the fast and real-time detection of total coliforms and E. coli (Manafi and Kneifel 1989).
Pathogenic viruses can also be detected using the culture-based method. The method involves inoculation of virus stock aliquots onto the medium containing susceptible cell monolayers followed by incubation. The inoculated virus gets attach to the cells, and these infected cells release progeny of virus that forms the circular zone of infected cells over the medium called plaque. The result is expressed in plaque forming units (PFU) per ml (Dulbecco and Vogt 1953). Though culture based method are widely used, the technique is time-consuming and requires a lot of resources, including various laboratory equipment and a skilled workforce. The safety concern and less sensitivity of the test may limit the use of the said method in some cases. Therefore, there is a need for the development of rapid and easy to use techniques.
In drinking water treatment and distribution systems, the bio-stability of the water is assessed by assimilable organic carbon (AOC), which represents the dissolved organic carbon that is assimilated by the microbes present in the drinking water (Kooij 1992). In water distribution system, AOC can be correlated with the presence of biofilm and regrowth of microorganisms. Typically, AOC is analysed by the standard method, and it is explained in brief as follows. The AOC determination involves two steps: (i) culturing of microbes and (ii) enumeration of microbes. In the first step, the water sample is inoculated with test microorganisms such as Pseudomonas sp. (P-17) and Spirillum sp (NOX) and incubated at 15 °C for 9 days. Once the microbial growth attains the stationary phase, the cells are enumerated using the plate count on the agar medium (Tang et al. 2018; Hammes and Egli 2005). The net microbial growth is related to the growth of test organism on acetate (P-17) and oxalate (NOX). The results are represented as acetate-C equivalents. The bio-stable water should have an AOC concentration of 10 µg/L acetate-C equivalents, which depends on the available chlorine in the water (Kooij 1992).
11.2.2 Optical-Based Methods
The culture-based technique is not suitable for non-culturable organisms. The method is time intensive and laborious. On the contrary, the optical-based methods are nonculture-based technique and is simple, fast, and less costly. The optical-based microscopic techniques are used to visualize the size and morphology of the bacterial cells. However, most of the microbes lack colour and contrast which makes its visualization more difficult. In such cases, the incorporation of fluorescent dyes and stains can help in overcoming the above-said limitations (Claus 1992). The stains are made of salts containing positive or negative ion depends on the chromophore. Typically, the negatively charged bacterial cell wall sticks to the positively charged chromophores, which makes the visualization of microbes under light microscopy easier. The commonly used dyes are safranin, methylene blue, malachite green, eosin, eosin, fuchsin, rose bengal, and crystal violet (Microbiology L.l 2019). The traditional light microscope uses light (400–700 nm) for the illumination to magnify the bacterial cells in the sample. On the other hand, the fluorescence microscope uses much higher light intensity to excite the sample of interest that contains fluorescent dyes. Here, the fluorescent microscope contains the filter cube set that allows the radiation of a wavelength which matches with the fluorescing compounds (Bradburry 1996). The fluorescent microscope enables the real-time detection of dead and live bacterial cells using DEAD/LIVE bacterial viability kit, which contains the fluorescent stains (Boulos et al. 1999). The refinement in the field of microscopes paved the way for the development of differential interference and phase contrast microscope (Keevil 2003), confocal laser scanning microscopes (CLSM), and total internal reflection fluorescence microscopes (TIRF) for the visualization of microorganisms. The phase contrast microscopes are used to visualize the microbes without staining (Keevil 2003). The CLSM (Sheppard et al. 1997) and TIRF (Axelrod 2001) are mainly used to image the structural components of cells, genetic material present inside the cells, and the specific cells within major.
Recently, adenosine triphosphate (ATP), an indicator of the presence of microbial growth, based optical detection method has been developed a potential (Selan et al. 1992). The bioluminescence, a light emission due to chemical reactions in the organism, forms the basis of this detection technique. In this method, a buffering agent that lyses the cell wall of the bacteria is added to the water sample, which is concentrated by membrane filtration. This buffering agent releases the ATP, and the concentration of ATP is measured by light emission intensity (580 nm) produced via luciferin-luciferase assay. The activity of the assay is standardized against known concentrations of ATP, and the results are represented as Relative Light Unit/ml (RLU/ml) (Turner et al. 2010). This ATP based measurement was also used to check the efficiency of the treatment processes, such as ozonation, UV treatment, and chlorination with respect to bio-stability of treated water. This method also acts as the best surrogate for measuring the growth of biomass and for determining the biomass production potential (van der Wielen and van der Kooij 2010). Though the culture-based methods are convenient, simple to perform, low-cost, and rapid, the limited representation of microbial communities is a limitation.
Recently, flow cytometry (FCM), an optical based detection method, is used to identify the individual microbial cells presents in a complex microbial community (Basiji et al. 2007). The working mechanism of FCM is given in the following steps: (i) the microbes present in the suspension is allowed to pass through a laser beam, (ii) the cells present in the suspension are scattered by light, and the fluorogenic substrates are excited to produce emission. The scattered light is captured at a low angle (forward scattering) and high angle (sideward scattering). The fluorescence is detected by using the selective wavelength filters. The method can be used to find the size, shape, and the number of microorganisms present in the sample. The schematic representation is shown in Fig. 11.7. Unlike fluorescence microscopy, FCM does not produce the images, rather the characteristic feature of each cell are presented as histograms or dot plots. The FCM is highly sensitive (<100 cells/ml) and rapid technique (<3 min per sample) (Hammes and Egli 2010). Along with the bacteria, yeast cells (Díaz et al. 2010), algae (Dubelaar and Gerritzen 2000), viruses (Brussaard et al. 2000), and protozoa (Vesey et al. 1994) present in the water samples can also be detected by FCM (Ambriz-Aviña et al. 2014). The FCM is a single cell technique, and it cannot be used for analysing biofilms present in the drinking water sample. Though there are improvements in the field of optical based detection techniques for the identification and enumeration of the pathogens, the disparity in the order of magnitude of the total number of cells between microscopic counting and plate counting methods needs further attention.
11.2.3 Molecular Based Methods
The molecular-based methods are more sensitive, reliable, robust, and yield conclusive results (Derveaux et al. 2010). The technique is suitable for the detection of a broad spectrum of microorganisms, including emerging pathogens. Unlike conventional techniques, this process functions by detecting specific ribonucleic acid (RNA) or deoxyribonucleic acid (DNA) sequences in the target organism (Garibyan and Avashia 2013; Law et al. 2015). The method allows for simultaneous detection and identification of pathogenic microorganisms.
Polymerase chain reaction (PCR) is one of the popular molecular-based detection ways in practice. It was invented in the late 20th century by Kary Mullis, and he was awarded Nobel Prize in chemistry for the invention in 1993 (McPherson and Møller 2000). It is very sensitive and can detect single bacterial pathogen (Velusamy et al. 2010). It involves amplification of a primer mediated enzymatic DNA and creation of specific DNA fragments (Valones et al. 2009). The amplification typically occurs in a cyclic three-step process (Law et al. 2015). These include DNA melting, annealing, and extension. In DNA melting or DNA denaturation, a double-stranded DNA is physically separated to two-pieces of single-stranded DNA at elevated temperatures (90–97 °C). In the next step, the oligonucleotides or specific primers anneal (50–60 °C) and bind to the complementary sequences of DNA. The two DNA strands then form a template for DNA polymerase to synthesise a new DNA strand. In the finals step, the new DNA strand is used as a template to create the duplicate copies, and the original DNA template is amplified exponentially through a chain reaction.
The developments in the PCR based detection techniques include cold PCR (Milbury et al. 2011), heat pulse extension-PCR (HPE-PCR) (Orpana et al. 2013), and nanoparticle-PCR (Ma et al. 2013). But the limitations with PCR is that it cannot differentiate between live or dead cells, and it will produce false results if there is any contamination in the sample. So, the PCR technique may not be useful for the detection of pathogens present in the wastewater sample. To eliminate the lack of differentiation between live and dead cells, the reverse transcriptase PCR (RT-PCR) was developed (Cangelosi and Meschke 2014). The pathogenic viruses can also be determined by RT-PCR and real-time PCR (Mattison and Bidawid 2009). Recently, multiplex PCR assay has been developed by the researchers to detect 10 viruses in a single tube (Pham et al. 2010; Wolf et al. 2008). Furthermore, advanced molecular methods such as usage of DNA based fluorescent probes and Enzyme-Linked Immunosorbent Assay (ELISA) could be used for the identifying pathogenic species (Kittigul et al. 2001).
11.2.4 Bio-sensing Based Methods
There is an growing demand for a versatile and sensitive technique that detects pathogens in a rapid manner. Biosensors are devices that work based on the detection of signals produced by the interaction between the bio-recognition elements, such as enzymes, antibodies, aptamers, oligonucleotides probes, nucleic acids, and cell-surface molecules (Rider et al. 2003), and the target analyte species (Zourob et al. 2008).
Here, the biological response is converted to electrical signals, and it is recorded by a detector (Brindha et al. 2018). The schematic diagram showing the working principle of bio-sensors is given in Fig. 11.8. A comparative analysis of various types of biosensors is presented in Table 11.3. In addition to the existing bio-recognition elements, functional nanomaterials are also being used for the detection of pathogens (Krishnan et al. 2019). The method has a few limitations. The credibility and sensitivity of bio-sensors are affected due to the interference caused by organic/inorganic molecules and other contaminants, such as humic substances with the microbes. The natural receptors such as antibodies and enzymes that are immobilized at the transducer surface are prone to the degeneration, which results in loss of selectivity and sensitivity. These challenges may be addressed by replacing the natural bio recognition elements with bio-mimetic elements, such as aptamers, peptides, and molecular imprinted polymer that can enhance the sensitivity of the detection (Kumar et al. 2018).
Schematic representation of working mechanism of bio-sensor (courtesy of: http://agscientific.com/blog/2016/01/biosensors-and-diabetes-trending-applications)
11.3 Remediation of Microbial Contaminated Water
11.3.1 A Brief Review of Disinfection Technologies
Access to clean water is a fundamental human right and is essential for a healthy life. The relationship between the quality of water and health are well documented. One of the primary reasons for the loss of human lives in most of the countries is the consumption of contaminated water. However, providing safe water to every person is a challenging task due to the increasing presence of pollutants and the growing gap between demands and supply. There are several classes of pollutants reported in drinking water and are presented in Fig. 11.9. Among these microbial pollutants requires special attention due to their widespread occurrence and potential to cause adverse ill effects on human health. A brief review of various aspects of the treatment of pathogens is presented below.
11.3.2 Centralised Versus Point-of-Use Technologies
The goal of disinfection is to alleviate the pathogens responsible for waterborne diseases. Several treatment technologies have been employed to achieve the goal. However, several factors govern the selection of disinfection technologies. These include the ability of the disinfectant to kill a broad spectrum of pathogenic organism, the capacity to provide residual disinfection activity, affordability, the formation of disinfection by-products, and the aesthetic quality of the treated water (National Research Council (US) Safe Drinking Water Committee 1980). The scale of operation is another critical parameter that decides the success of the treatment process. Treatment may be done at a small scale in decentralized plants or can be done at community level at a centralized facility. However, establishing large community water treatment systems is challenging for developing nations. The challenges include capital investment, skilled labour, and governance, access to appropriate technologies, piped water supply networks, water scarcity, maintenance, and recontamination. The poor success in overcoming these challenges has increased the popularity of decentralized or point-of-use (POU) water treatment systems. In developing countries where only limited households have piped water (Kanungo et al. 2010), POU interventions seem to be a sustainable way of providing safe drinking water.
Though technologically advanced countries can afford to use a complex system to meet the stringent regulations, their public water system is also not completely free from pathogenic organisms. Several incidences of the pathogenic microorganism in piped water are reported from developed countries (EPA 1996). Studies show that despite keeping adequate disinfectant residual, there is a significant deterioration of water quality due to the proliferation of microbes in the bio-films attached to the distribution pipes (Machell et al. 2010; Szewzyk et al. 2000; Simoes and Simões 2013; Douterelo et al. 2014). The data further support the inefficiency of the public water supply system to contain waterborne outbreaks. In this context, there is a need for an affordable and efficient alternate disinfection system. A well designed and maintained POU disinfection seems to be an attractive option (Vagliasindi et al. 1998; Sobsey et al. 2008).
Several point-of-use water purification technologies are developed to disinfect water. Among the available technologies, chlorination with safe storage, combined coagulant-chlorine disinfection, SODIS (solar UV radiation + with thermal effects), ceramic filter, bio-sand filter are well documented and capable of reducing waterborne infectious disease (Sobsey et al. 2008; Rose et al. 2006). State-of-the-art literature reviews show that POU household interventions contribute to a 30–40% reduction in diarrheal diseases (Clasen et al. 2007; Fewtrell et al. 2005). According to a recent review, over 18 million people use POU water treatment systems, with 12.8 million using chlorination, 2.1 million using SODIS, 0.934 million using flocculation/chlorination, 0.7 million using bio-sand filtration, and 0.35 million using ceramic filtration (Sobsey et al. 2008; Clasen 2008) have compared these widely promoted and used POU systems for performance and sustainability and identified ceramic and bio-sand based systems are most effective. However, Lantagne et al. (2008) pointed out the flaws in the comparison and strongly commented that the comparison is biased. The role of participant motivation in reducing dysentery and non-dysentery diarrhoea by disinfection using SODIS among children (0.5 to 6 years) living in peri-urban communities in South Africa was studied (Du Preez et al. 2010). After comparing 383 children in 297 houses using SODIS with 335 children in 267 families with no intervention, the authors concluded that the motivation of participants is also an essential factor for measurable health gain.
Despite of obtaining promising results with the technologies validated in the laboratory and field, except boiling, the large-scale deployment of the technologies has hindered. Some of the reasons highlighted for the failure are (Sobsey et al. 2008): (i) Inability to provide adequate safe water, (ii) difficulty in operation and maintenance, (iii) large user time to treat water, (iv) affordability, (v) weak supply chain for needed replacement of units or parts, (vi) objectionable taste and odour, and (vii) bio fouling. Therefore, addressing these issues is vital for the successful implementation of POU treatment systems. Moreover, public participation, socio-economic considerations, local water quality, and consumer preference also need to be considered as sustainability criteria for developing POU water treatment systems.
11.3.3 Convectional Disinfection Technologies
The microbial quality of water can be improved through physical, chemical, or biological methods. There are several options available under each method. However, commonly practiced techniques involve chlorination, ozonation, filtration, UV irradiation, boiling, and SODIS process. A brief description of each technology is discussed below.
Disinfection with Chlorine: Chlorination is a popular disinfection processes and is achieved by introducing chlorine gas or its derivatives such as sodium hypochlorite (NaOCl) or calcium hypochlorite (Ca(OCl)2) into water. The chlorine gas was first discovered in 1774 by Karl W. Scheele. Later, Humphrey Davy recognized it as a disinfectant in 1810 (Pradeep 2009). The continuous chlorination of public water process was first introduced in 1904 by Sir Alexander Houston of the London. In 1908, the application of calcium hypochlorite to Bubbly Creek water supply of the city of Chicago was initiated in the US (Logan and Savell 1940).
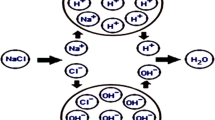
Upon addition of chlorine or its derivative to water, the chlorine agent undergoes hydrolysis and results in the formation of free-chorine species (HOCl and OCl−). These species are responsible for disinfection of water. The reactions involved in the chlorination process are given below.
Among the free chlorine species, HOCl is the more powerful oxidizing agent (Metcalf 2003). The relative concentration of these species will vary according to the pH of the water. The chlorine can also combine with ammonia present in water and form chloramines. The critical reactions involved in the formations of chloramines are given below.
The formation of chloramines is dependent on the relative concentration of ammonia and chlorine, pH, contact time, and temperature (Metcalf 2003). Though chloramines are less effective compared to free-chlorine species, they are unlikely to produce disinfecting by products (DBPs).
Disinfection with Ozone: Ozonation is the second most widely used disinfection process after chlorination. In 1906, France reported the first use of ozone as a disinfectant (Pradeep 2009). Ozone is generated on-site and is typically produced through electrical discharge method. A schematic diagram showing the generation of ozone is shown in Fig. 11.10. The freed radicals (HO and HO2) formed as a result of the decomposition are probably responsible for the defection (Metcalf 2003). Unlike chlorine and chloramines, ozone is effective against a broad spectrum of the organisms, including Giardia lamblia and Cryptosporidium parvam. It is also found to be effective against spores and cysts (Budu-Amoako et al. 2011). However, it does not maintain residual ozone concentration and less effective in preventing recontamination of water (EPA 2011).
Disinfection with UV: The first use of UV treatment in municipal water supplies was reported in 1916. Presently, the use of this technology is used in several applications. Unlike ozone and chlorine, UV light is a physical disinfecting agent, and hence, it is free from taste, odour, and harmful by-products even at high dose (EPA 2011). UV radiation at the right wavelength (255–265 nm) has shown active bactericidal and virucidal properties. The schematic diagram of the UV disinfection process is presented in Fig. 11.11b. A comprehensive review of UV based disinfection system is given elsewhere (Nyangaresi et al. 2018; Li et al. 2018).
Schematic representation of a solar disinfection (SODIS) (courtesy of www.billionbottleproject.org). b UV irradiation
Disinfection with Solar Radiation: Solar disinfection, popularly known as SODIS process, works based on the germicidal property of UV radiation and thermal heating. In the SODIS process, the water is exposed to natural sunlight instead of light from a UV lamp. The conventional UV system uses UV-C (200–280 nm) radiations, whereas the SODIS process uses UV-A (320–400 nm) radiations. The interaction of UV-A with water generates reactive oxygen species (ROS) and ROS damage the DNA and deactivate the germs in the water. However, the SODIS process is not as effective as conventional UV treatment because UV light constitutes only <5% of the total solar spectrum (McGuigan et al. 2012). The schematic diagram of SODIS process is presented in Fig. 11.11a. Some of the viruses, protozoan species exhibits resistance to chlorination. So, we are in necessity to find the alternative technology that can kill various kinds of pathogenic species.
11.3.4 Nanotechnology Enabled Disinfection Process
Nanotechnology-enabled water purifiers may hold the key for safe drinking water in the future (Shannon et al. 2010; Hossain et al. 2014). The technology uses engineered nanoscale material to improve microbial quality of the water. In comparison to conventional chemical disinfectants, nanoscale materials are not strong oxidants and hence unlikely to produce harmful DBPs. Several natural and engineered nanomaterials are available for disinfection. These include photo catalytic TiO2 (Dimitroula et al. 2012), silver nanoparticles (Sankar et al. 2013), MgO (Stoimenov et al. 2002; Ganguly et al. 2011), zero-valent iron (Crane and Scott 2012) and so on. Among these disinfectants, the silver-based system is more matured and used in the field level application. The silver nanoparticles are active disinfectant and work for a broad spectrum of bacteria and viruses (Karumuri et al. 2013; De Gusseme et al. 2010; Loo et al. 2013). Though the research and development in this area is not fully matured, the current advancement in nanotechnology may prove to be of significant interest to both developed and developing countries in addressing the problem of safe drinking water.
11.3.5 Membrane Based Pathogens Control Technologies
The use of membrane filtration system has increased significantly over the last two decades. It has become one of the popular methods of purification of water now. The process of membrane includes micro-filtration, ultra-filtration, nano-filtration, and Reverse Osmosis (RO). The classification is based on the pore sizes of the membrane. Among the membranes, RO can efficiently remove bacteria, viruses, and other suspended solids present in the water. It can control the disinfection by-products as well (Van der Bruggen et al. 2003). However, the large amount of rejects, clogging of the membrane, and high energy consumption needs further attention.
Recent years, the use of biomimetic membranes is gaining interest in water treatment. In this technology, aquaporin, a bio-inspired membrane is used. Aquaporin acts as a water channel and allows only the passage of water through it. This method may reduce the cost of filtration by 30% of the conventional membranes (Wah 2016). The schematic representation of the membrane filtration technique is given in the Fig. 11.12.
Schematic representation of membrane filtration (courtesy of: https://www.logisticon.com/en/technologies/membrane-filtration)
11.3.6 Disinfection by Advanced Oxidation Process
The advanced oxidation process (AOP) is a promising technology in the field of water purification. The process typically uses ozone (O3), UV light, hydrogen peroxide (H2O2), or combination thereof. The OH radicals formed during the processes are mainly responsible for the destruction of pathogens. The use of UV/TiO2 (Matsunaga et al. 1985), UV/H2O2 (Gassie et al. 2016), ozone-UV (Crittenden et al. 2012), and photo-Fenton (Rossi et al. 2009) are also studied as next-generation disinfectants. In photo-catalysis, a light source of a specific wavelength is used to excite the electron from valence band to conduction band. The ability of the catalyst to produce the electron-hole pairs decides the efficiency of the process. The free radicals formed during the reaction destroy the pathogens present in the water (Giannakis et al. 2018).
Equations R8–R10 explain the radical formation in the UV/TiO2 process, whereas Eqs. R11 and R12 show the radical formation in UV/H2O2 and ozone-UV process, respectively. Though AOPs are highly effective against the pathogens, the presence of natural organic matter such as fulvic acid, humic acid, and other ions affects its performance (Keane et al. 2014).
Plasma-based treatment technology is an emerging field for the removal of pathogens present in the drinking water (Rossi et al. 2009; Roth et al. 2010). In plasma technology, the Pulsed power technique (PPP) is found to be an effective method to disinfect the pathogen in short span of time (<6 min) (Singh et al. 2017). The schematic representation of UV photo-catalysis, and plasma techniques are given in Fig. 11.13a, b respectively.
11.4 Conclusion
Potable water is an absolute necessity for humankind as long as life exists. The pursuit of safe drinking water has been the highest priority for humans over centuries. The massive rise in the global population, poorly-managed water systems, and pollution have made the search more challenging. A large number of freshwater bodies across the world are contaminated and has become a major risk to human health and the ecosystem. Realizations on the link that exist between water and health, proving safely managed drinking water has become a criteria agenda in every framework of the developmental organization.
Among the contaminants identified in drinking water, the pathogen causes significant threat due to their widespread occurrence and potential to cause diseases. Though several technologies have been developed to detect and control the pathogens, the lack of reliable and affordable detection and control technologies hindered access to such interventions for a large population, especially from developing world. The emergence of disinfectant and antibiotic-resistant microorganisms also have become a cause of concern for the process of control and detection. There is a need for safe, affordable, and reliable point-of-use disinfection and detections system. There is also scope for developing hybrid systems, which facilitates both detection and removal of pathogens in drinking water. As to move forward, the technologies with abovementioned features should be made available to the public at affordable cost, thereby promoting sustainable, healthy, and productive living condition.
References
Al-Qadiri HM, Al-Alami NI, Al-Holy MA, Rasco BA (2008) Using Fourier transform infrared (FT-IR) absorbance spectroscopy and multivariate analysis to study the effect of chlorine-induced bacterial injury in water. J Agric Food Chem 56(19):8992–8997
Ambriz-Aviña V, Contreras-Garduño JA, Pedraza-Reyes M (2014) Applications of flow cytometry to characterize bacterial physiological responses. BioMed Res Int
American Public Health Association, American Water Works Association (1989) Standard methods for the examination of water and wastewater. American Public Health Association
Araya V, Maliyekkal SM, Philip L (2019) Water pollution and treatment technologies – Indian perspective. In: Mjumdar PP, Tiwari VM (eds) Water futures of India: Status of science and technology. Indian National Science Academy, pp 215–252.
Axelrod D (2001) Total internal reflection fluorescence microscopy in cell biology. Traffic 2(11):764–774
Babacan S, Pivarnik P, Letcher S, Rand A (2000) Evaluation of antibody immobilization methods for piezoelectric biosensor application. Biosens Bioelectron 15(11–12):615–621
Basiji DA, Ortyn WE, Liang L, Venkatachalam V, Morrissey P (2007) Cellular image analysis and imaging by flow cytometry. Clinics Lab Med 27(3):653–670
Blumenthal UJ, Mara DD, Peasey A, Ruiz-Palacios G, Stott R (2000) Guidelines for the microbiological quality of treated wastewater used in agriculture: recommendations for revising WHO guidelines. Bull World Health Organ 78:1104–1116
Boulos L, Prevost M, Barbeau B, Coallier J, Desjardins R (1999) LIVE/DEAD® BacLight™: application of a new rapid staining method for direct enumeration of viable and total bacteria in drinking water. J Microbiol Methods 37(1):77–86
Bradburry S (1996) Fluorescence microscopy, contrast techniques in light microscopy
Brindha J, Chanda K, Balamurali M (2018) Physical, chemical and biochemical biosensors to detect pathogens. In: Nanotechnology food security and water treatment. Springer, Berlin, pp 53–86
Brussaard CP, Marie D, Bratbak G (2000) Flow cytometric detection of viruses. J Virol Methods 85(1–2):175–182
Budu-Amoako E, Greenwood SJ, Dixon BR, Barkema HW, McClure J (2011) Foodborne illness associated with cryptosporidium and Giardia from livestock. J Food Prot 74(11):1944–1955
Cangelosi GA, Meschke JS (2014) Dead or alive: molecular assessment of microbial viability. Appl Environ Microbiol 80(19):5884–5891
Chen Q, Lin J, Gan C, Wang Y, Wang D, Xiong Y, Lai W, Li Y, Wang M (2015) A sensitive impedance biosensor based on immunomagnetic separation and urease catalysis for rapid detection of Listeria monocytogenes using an immobilization-free interdigitated array microelectrode. Biosens Bioelectron 74:504–511
Clasen TF (2008) Scaling up household water treatment: looking back, seeing forward. In: Public health and the environment, World Health Organization, Geneva
Clasen T, Schmidt W-P, Rabie T, Roberts I, Cairncross S (2007) Interventions to improve water quality for preventing diarrhoea: systematic review and meta-analysis. BMJ 334(7597):782
Claus D (1992) A standardized Gram staining procedure. World J Microbiol Biotechnol 8(4):451–452
Cotruvo J, Dufour A, Rees G, Bartram J, Carr R, Cliver DO, Craun GF, Fayer R, Gannon VP, World Health Organization (2004) What are the criteria for determining whether a disease is zoonotic and water related? In: Water borne zoonoses: identification, causes and control. IWA
Crane R, Scott T (2012) Nanoscale zero-valent iron: future prospects for an emerging water treatment technology. J Hazard Mater 211:112–125
Craun GF, Brunkard JM, Yoder JS, Roberts VA, Carpenter J, Wade T, Calderon RL, Roberts JM, Beach MJ, Roy SL (2010) Causes of outbreaks associated with drinking water in the United States from 1971 to 2006. Clin Microbiol Rev 23(3):507–528
Crittenden JC, Trussell DW, Hand KJ, Howe KJ, Tchobanoglous G (2012) MWH’s water treatment: principles and design. Wiley, New York
Das R, Sharma MK, Rao VK, Bhattacharya B, Garg I, Venkatesh V, Upadhyay S (2014) An electrochemical genosensor for Salmonella typhi on gold nanoparticles-mercaptosilane modified screen printed electrode. J Biotechnol 188:9–16
De Gusseme B, Sintubin L, Baert L, Thibo E, Hennebel T, Vermeulen G, Uyttendaele M, Verstraete W, Boon N (2010) Biogenic silver for disinfection of water contaminated with viruses. Appl Environ Microbiol 76(4):1082–1087
Derveaux S, Vandesompele J, Hellemans J (2010) How to do successful gene expression analysis using real-time PCR. Methods 50(4):227–230
Dey S (2015) 80% of India’s surface water may be polluted, report by international says. Times of India
Dey NC, Saha R, Parvez M, Bala SK, Islam AS, Paul JK, Hossain M (2017) Sustainability of groundwater use for irrigation of dry-season crops in northwest Bangladesh. Groundwater Sustain Develop 4:66–77
Díaz M, Herrero M, García LA, Quirós C (2010) Application of flow cytometry to industrial microbial bioprocesses. Biochem Eng J 48(3):385–407
Dimitroula H, Daskalaki VM, Frontistis Z, Kondarides DI, Panagiotopoulou P, Xekoukoulotakis NP, Mantzavinos D (2012) Solar photocatalysis for the abatement of emerging micro-contaminants in wastewater: synthesis, characterization and testing of various TiO2 samples. Appl Catal B 117:283–291
Dogan Ü, Kasap E, Cetin D, Suludere Z, Boyaci IH, Türkyılmaz C, Ertas N, Tamer U (2016) Rapid detection of bacteria based on homogenous immunoassay using chitosan modified quantum dots. Sens Actuators B Chem 233:369–378
Douterelo I, Husband S, Boxall J (2014) The bacteriological composition of biomass recovered by flushing an operational drinking water distribution system. Water Res 54:100–114
Du Preez M, McGuigan KG, Conroy RM (2010) Solar disinfection of drinking water in the prevention of dysentery in South African children aged under 5 years: the role of participant motivation. Environ Sci Technol 44(22):8744–8749
Dubelaar G, Gerritzen P (2000) CytoBuoy: a step forward towards using flow cytometry in operational oceanography. Scientia Marina 64(2):255–265
Dulbecco R, Vogt M (1953) Some problems of animal virology as studied by the plaque technique. In: Cold Spring Harbor symposia on quantitative biology. Cold Spring Harbor Laboratory Press
EPA (1996) National primary drinking water regulations, Monitoring requirements for public drinking water supplies. Final rule. Federal Register 61(94)
EPA (2011) Water treatment manual: disinfection, 2011
Faures J, Eliasson A, Hoogeveen J, Vallee D (2001) AQUASTAT-FAO’s information system on water and agriculture. GRID-Magazine of the IPTRID Network (FAO/United Kingdom)
Feachem R, Mara DD, Bradley DJ (1983) Sanitation and disease. Wiley, Washington DC
Fewtrell L, Kaufmann RB, Kay D, Enanoria W, Haller L, Colford JM Jr (2005) Water, sanitation, and hygiene interventions to reduce diarrhoea in less developed countries: a systematic review and meta-analysis. Lancet Infect Dis 5(1):42–52
Fung DY, Miller RD (1970) Rapid procedure for the detection of acid and gas production by bacterial cultures. Appl Microbiol 20(3):527
Ganguly A, Trinh P, Ramanujachary K, Ahmad T, Mugweru A, Ganguli AK (2011) Reverse micellar based synthesis of ultrafine MgO nanoparticles (8–10 nm): characterization and catalytic properties. J Colloid Interface Sci 353(1):137–142
Garibyan L, Avashia N (2013) Research techniques made simple: polymerase chain reaction (PCR). J Invest Dermatol 133(3):e6
Gassie LW, Englehardt JD, Wang J, Brinkman N, Garland J, Gardinali P, Guo T (2016) Mineralizing urban net-zero water treatment: Phase II field results and design recommendations. Water Res 105:496–506
Giannakis S, Voumard M, Rtimi S, Pulgarin C (2018) Bacterial disinfection by the photo-Fenton process: extracellular oxidation or intracellular photo-catalysis? Appl Catal B 227:285–295
Gowrisankar G, Chelliah R, Ramakrishnan SR, Elumalai V, Dhanamadhavan S, Brindha K, Antony U, Elango L (2017) Chemical, microbial and antibiotic susceptibility analyses of groundwater after a major flood event in Chennai. Scientific data 4:170135
Hammes FA, Egli T (2005) New method for assimilable organic carbon determination using flow-cytometric enumeration and a natural microbial consortium as inoculum. Environ Sci Technol 39(9):3289–3294
Hammes F, Egli T (2010) Cytometric methods for measuring bacteria in water: advantages, pitfalls and applications. Anal Bioanal Chem 397(3):1083–1095
Hossain F, Perales-Perez OJ, Hwang S, Roman F (2014) Antimicrobial nanomaterials as water disinfectant: applications, limitations and future perspectives. Sci Total Environ 466:1047–1059
Jamieson RC, Gordon RJ, Tattrie SC, Stratton GW (2003) Sources and persistence of fecal coliform bacteria in a rural watershed. Water Qual Res J 38(1):33–47
Kanungo S, Sah B, Lopez A, Sung J, Paisley A, Sur D, Clemens J, Nair GB (2010) Cholera in India: an analysis of reports, 1997–2006. Bull World Health Organ 88:185–191
Karumuri AK, Oswal DP, Hostetler HA, Mukhopadhyay SM (2013) Silver nanoparticles attached to porous carbon substrates: robust materials for chemical-free water disinfection. Mater Lett 109:83–87
Keane DA, McGuigan KG, Ibáñez PF, Polo-López MI, Byrne JA, Dunlop PS, O’Shea K, Dionysiou DD, Pillai SC (2014) Solar photocatalysis for water disinfection: materials and reactor design. Catal Sci Technol 4(5):1211–1226
Keevil C (2003) Rapid detection of biofilms and adherent pathogens using scanning confocal laser microscopy and episcopic differential interference contrast microscopy. Water Sci Technol 47(5):105–116
Kittigul L, Khamoun P, Sujirarat D, Utrarachkij F, Chitpirom K, Chaichantanakit N, Vathanophas K (2001) An improved method for concentrating rotavirus from water samples. Mem Inst Oswaldo Cruz 96(6):815–821
Kooij D (1992) Assimilable organic carbon as an indicator of bacterial regrowth. J Am Water Works Assoc 84(2): 57–65
Krishnan SK, Singh E, Singh P, Meyyappan M, Nalwa HS (2019) A review on graphene-based nanocomposites for electrochemical and fluorescent biosensors. RSC Adv 9(16):8778–8881
Kumar N, Hu Y, Singh S, Mizaikoff B (2018) Emerging biosensor platforms for the assessment of water-borne pathogens. Analyst 143(2):359–373
Laczka O, Skillman L, Ditcham WG, Hamdorf B, Wong DK, Bergquist P, Sunna A (2013) Application of an ELISA-type screen printed electrode-based potentiometric assay to the detection of Cryptosporidium parvum oocysts. J Microbiol Methods 95(2):182–185
Lantagne D, Meierhofer R, Allgood G, McGuigan K, Quick R (2008) Comment on “Point of use household drinking water filtration: A practical, effective solution for providing sustained access to safe drinking water in the developing world”. Environ Sci Technol 43(3):968–969
Law JWF, Ab Mutalib NS, Chan K-G, Lee L-H (2015) Rapid methods for the detection of foodborne bacterial pathogens: principles, applications, advantages and limitations. Front Microbiol 5:770
Lee-Montiel FT, Reynolds KA, Riley MR (2011) Detection and quantification of poliovirus infection using FTIR spectroscopy and cell culture. Journal of biological engineering 5(1):16
Leininger DJ, Roberson JR, Elvinger F (2001) Use of eosin methylene blue agar to differentiate Escherichia coli from other gram-negative mastitis pathogens. J Vet Diagn Invest 13(3):273–275
Li X, Cai M, Wang L, Niu F. Yang D, Zhang G (2018) Evaluation survey of microbial disinfection methods in UV-LED water treatment systems. Sci Total Environ
Logan JO, Savell WL (1940) Calcium hypochlorite in water purification. J (Am Water Works Assoc) 32(9):1517–1527
Loo S-L, Fane AG, Lim T-T, Krantz WB, Liang Y-N, Liu X, Hu X (2013) Superabsorbent cryogels decorated with silver nanoparticles as a novel water technology for point-of-use disinfection. Environ Sci Technol 47(16):9363–9371
Ma X, Cui Y, Qiu Z, Zhang B, Cui S (2013) A nanoparticle-assisted PCR assay to improve the sensitivity for rapid detection and differentiation of wild-type pseudorabies virus and gene-deleted vaccine strains. J Virol Methods 193(2):374–378
Machell J, Mounce S, Boxall J (2010) Online modelling of water distribution systems: a UK case study. Drinking Water Eng Sci 3:21–27
Manafi M, Kneifel W (1989) A combined chromogenic-fluorogenic medium for the simultaneous detection of coliform groups and E. coli in water. Int J Hygiene Environ Med 189(3): 225–234
March SB, Ratnam S (1986) Sorbitol-MacConkey medium for detection of Escherichia coli O157: H7 associated with hemorrhagic colitis. J Clin Microbiol 23(5):869–872
Matsunaga T, Tomoda R, Nakajima T, Wake H (1985) Photoelectrochemical sterilization of microbial cells by semiconductor powders. FEMS Microbiol Lett 29(1–2):211–214
Mattison K, Bidawid S (2009) Analytical methods for food and environmental viruses. Food Environ Virol 1(3–4):107–122
McGuigan KG, Conroy RM, Mosler H-J, du Preez M, Ubomba-Jaswa E, Fernandez-Ibanez P (2012) Solar water disinfection (SODIS): a review from bench-top to roof-top. J Hazard Mater 235:29–46
McPherson M, Møller S (2000) Pcr. Taylor & Francis, New York
Metcalf, L., Wastewater engineering: treatment and reuse. Metcalf & Eddy Inc. 2003, McGraw-Hill Inc., New York
Microbiology L.l (2019) Staining microscopic specimens. cited 29 May 2019. Available from: https://courses.lumenlearning.com/microbiology/chapter/staining-microscopic-specimens/
Milbury CA, Li J, Liu P, Makrigiorgos GM (2011) COLD-PCR: improving the sensitivity of molecular diagnostics assays. Expert Rev Mol Diagn 11(2):159–169
Munoz EF, Silverman MP (1979) Rapid, single-step most-probable-number method for enumerating fecal coliforms in effluents from sewage treatment plants. Appl Environ Microbiol 37(3):527–530
Murty M, Kumar S (2011) Water pollution in India: an economic appraisal. India infrastructure report. Water: policy and performance for sustainable development
National Research Council (US) Safe Drinking Water Committee (1980) Drinking water and health: the disinfection of drinking water. National Academies Press (US), New York
Nyangaresi PO, Qin Y, Chen G, Zhang B, Lu Y, Shen L (2018) Comparison of UV-LED photolytic and UV-LED/TiO2 photocatalytic disinfection for Escherichia coli in water. Catalysis Today
Orpana AK, Ho TH, Alagrund K, Ridanpää M, Aittomäki K, Stenman J (2013) Novel Heat pulse extension-PCR–based method for detection of large CTG-repeat expansions in myotonic dystrophy type 1. J Mol Diagn 15(1):110–115
Pandey PK, Soupir ML (2013) Assessing the impacts of E. coli laden streambed sediment on E. coli loads over a range of flows and sediment characteristics. JAWRA J Am Water Resour Assoc 49(6):1261–1269
Pandey PK, Kass PH, Soupir ML, Biswas S, Singh VP (2014) Contamination of water resources by pathogenic bacteria. Amb Express 4(1):51
Pham NTK, Trinh QD, Chan-It W, Khamrin P, Shimizu H, Okitsu S, Mizuguchi M, Ushijima H (2010) A novel RT-multiplex PCR for detection of Aichi virus, human parechovirus, enteroviruses, and human bocavirus among infants and children with acute gastroenteritis. J Virol Methods 169(1):193–197
Planning Commission (2002) Report of the screening commitee on drinking water supply and sanitation (Rural and Urban) for tenth five year plan. P. Commision, New Delhi, India
Pradeep T (2009) Noble metal nanoparticles for water purification: a critical review. Thin Solid Films 517(24):6441–6478
Rajapaksha P, Elbourne A, Gangadoo S, Brown R, Cozzolino D, Chapman J (2019) A review of methods for the detection of pathogenic microorganisms. Analyst 144(2):396–411
Rakić A (2018) Water quality control in the water supply system for the purpose of preventing legionnaires’ disease. In: Water challenges of an urbanizing world. IntechOpen
Rein DB, Stevens GA, Theaker J, Wittenborn JS, Wiersma ST (2012) The global burden of hepatitis E virus genotypes 1 and 2 in 2005. Hepatology 55(4):988–997
Rhodes MW, Kator HI (1990) Effects of sunlight and autochthonous microbiota on Escherichia coli survival in an estuarine environment. Curr Microbiol 21(1):65–73
Rider TH, Petrovick MS, Nargi FE, Harper JD, Schwoebel ED, Mathews RH, Blanchard DJ, Bortolin LT, Young AM, Chen J (2003) AB cell-based sensor for rapid identification of pathogens. Science 301(5630):213–215
Rivera-Jaimes JA, Postigo C, Melgoza-Alemán RM, Aceña J, Barceló D, de Alda ML (2018) Study of pharmaceuticals in surface and wastewater from Cuernavaca, Morelos, Mexico: occurrence and environmental risk assessment. Sci Total Environ 613:1263–1274
Rose A, Roy S, Abraham V, Holmgren G, George K, Balraj V, Abraham S, Muliyil J, Joseph A, Kang G (2006) Solar disinfection of water for diarrhoeal prevention in southern India. Arch Dis Child 91(2):139–141
Rossi F, Kylián O, Rauscher H, Hasiwa M, Gilliland D (2009) Low pressure plasma discharges for the sterilization and decontamination of surfaces. New J Phys 11(11):115017
Roth S, Feichtinger J, Hertel C (2010) Characterization of Bacillus subtilis spore inactivation in low-pressure, low-temperature gas plasma sterilization processes. J Appl Microbiol 108(2):521–531
Safavieh M, Ahmed MU, Sokullu E, Ng A, Braescu L, Zourob M (2014) A simple cassette as point-of-care diagnostic device for naked-eye colorimetric bacteria detection. Analyst 139(2):482–487
Saha R, Dey NC, Rahman S, Galagedara L, Bhattacharya P (2018) Exploring suitable sites for installing safe drinking water wells in coastal Bangladesh. Groundwater for Sustainable Development 7:91–100
Sankar MU, Aigal S, Maliyekkal SM, Chaudhary A, Kumar AA, Chaudhari K, Pradeep T (2013) Biopolymer-reinforced synthetic granular nanocomposites for affordable point-of-use water purification. Proc Natl Acad Sci 110(21):8459–8464
Schriewer A, Miller WA, Byrne BA, Miller MA, Oates S, Conrad PA, Hardin D, Yang H-H, Chouicha N, Melli A (2010) Presence of bacteroidales as a predictor of pathogens in surface waters of the central California coast. Appl Environ Microbiol 76(17):5802–5814
Selan L, Berlutti F, Passariello C, Thaller M, Renzini G (1992) Reliability of a bioluminescence ATP assay for detection of bacteria. J Clin Microbiol 30(7):1739–1742
Shaheed A, Orgill J, Montgomery MA, Jeuland MA, Brown J (2014) Why? improved? water sources are not always safe. Bull World Health Organ 92:283–289
Shannon MA, Bohn PW, Elimelech M, Georgiadis JG, Marinas BJ, Mayes AM (2010) Science and technology for water purification in the coming decades. In: Nanoscience and technology: a collection of reviews from nature Journals. World Scientific, pp 337–346
Sheppard C, Shotton D, Sheppard C (1997) Confocal laser scanning microscopy. BIOS Scientific Publishers, Oxford
Simoes LC, Simões M (2013) Biofilms in drinking water: problems and solutions. Rsc Adv 3(8):2520–2533
Singh RK, Babu V, Philip L, Ramanujam S (2017) Disinfection of water using pulsed power technique: effect of system parameters and kinetic study. In: Sustainability issues in civil engineering. Springe, Berlin, pp 307–336
Sobsey MD, Stauber CE, Casanova LM, Brown JM, Elliott MA (2008) Point of use household drinking water filtration: a practical, effective solution for providing sustained access to safe drinking water in the developing world. Environ Sci Technol 42(12):4261–4267
Stoimenov PK, Klinger RL, Marchin GL, Klabunde KJ (2002) Metal oxide nanoparticles as bactericidal agents. Langmuir 18(17):6679–6686
Szewzyk U, Szewzyk R, Manz W, Schleifer K-H (2000) Microbiological safety of drinking water. Ann Rev Microbiol 54(1):81–127
Tang P, Wu J, Liu H, Liu Y, Zhou X (2018) Assimilable organic carbon (AOC) determination using GFP-tagged Pseudomonas fluorescens P-17 in water by flow cytometry. PLoS ONE 13(6):e0199193
Tankeshwar (2010) Membrane filter technique for bacteriological examination of water. Cited 26 May 2019. Available from: https://microbeonline.com/membrane-filter-technique/
Thakur B, Amarnath CA, Mangoli S, Sawant SN (2015) Polyaniline nanoparticle based colorimetric sensor for monitoring bacterial growth. Sens Actuators B Chem 207:262–268
Trivedi BK, Gandhi HS, Shukla NK (1971) Bacteriological water quality and incidence of water borne diseases in a rural population. Indian J Med Sci 25(11):795–801
Turner DE, Daugherity EK, Altier C, Maurer KJ (2010) Efficacy and limitations of an ATP-based monitoring system. J Am Assoc Lab Anim Sci 49(2):190–195
UN (n.d.) Water scarcity. Available at: http://www.unwater.org/water-facts/scarcity/
UN DESA (2018) Revision of World Urbanization Prospects. Cited 27 May 2019. Available at: https://www.un.org/development/desa/publications/2018-revision-of-world-urbanization-prospects.html
UNESCO (2017) United Nations world water development report. 23 May 2019; at: http://www.unesco.org/new/en/natural-sciences/environment/water/wwap/wwdr/2017-wastewater-the-untapped-resource/
UNICEF (2016) One is too many: Ending child deaths from pneumonia and diarrhoea
UNICEF (2017) Progress on drinking water, sanitation and hygiene: 2017 update and SDG guidelines
USBR (2017) Water facts—worldwide water supply. Available at: https://www.usbr.gov/mp/arwec/water-facts-ww-water-sup.html
Vagliasindi FG, Belgiorno V, Napoli RM (1998) Water treatment in remote and rural areas: a conceptual screening protocol for appropriate POU/POE technologies. In: Environmental engineering and renewable energy. Elsevier, London, pp 329–336
Valones MAA, Guimarães RL, Brandão LAC, Souza PRE, Carvalho AAT, Crovela S (2009) Principles and applications of polymerase chain reaction in medical diagnostic fields: a review. Braz J Microbiol 40(1):1–11
Van der Bruggen B, Vandecasteele C, Van Gestel T, Doyen W, Leysen R (2003) A review of pressure-driven membrane processes in wastewater treatment and drinking water production. Environ Prog 22(1):46–56
van der Wielen PW, van der Kooij D (2010) Effect of water composition, distance and season on the adenosine triphosphate concentration in unchlorinated drinking water in the Netherlands. Water Res 44(17):4860–4867
Vandeweerd V, Bernal P, Belfiore S, Goldstein K, Cicin-Sain B (2002) A guide to oceans, coasts, and islands at the World Summit on sustainable development
Velusamy V, Arshak K, Korostynska O, Oliwa K, Adley C (2010) An overview of foodborne pathogen detection: in the perspective of biosensors. Biotechnol Adv 28(2):232–254
Venkatesan KD, Balaji M, Victor K (2014) Microbiological analysis of packaged drinking water sold in Chennai. Int J Med Sci Public Health 3(4):472–477
Vesey G, Hutton P, Champion A, Ashbolt N, Williams KL, Warton A, Veal D (1994) Application of flow cytometric methods for the routine detection of Cryptosporidium and Giardia in water. Cytometry J Int Soc Anal Cytol 16(1):1–6
Vidic J, Manzano M, Chang C-M, Jaffrezic-Renault N (2017) Advanced biosensors for detection of pathogens related to livestock and poultry. Vet Res 48(1):11
Wah TY (2016) Nature-inspired membrane set to reduce purification costs. Membr Technol 2016(5):7
Wang Y, Alocilja EC (2015) Gold nanoparticle-labeled biosensor for rapid and sensitive detection of bacterial pathogens. J Biol Eng 9(1):16
Wang L, Wei Q, Wu C, Hu Z, Ji J, Wang P (2008) The Escherichia coli O157: H7 DNA detection on a gold nanoparticle-enhanced piezoelectric biosensor. Chin Sci Bull 53(8):1175–1184
Wang W, Wang W, Liu L, Xu L, Kuang H, Zhu J, Xu C (2016a) Nanoshell-enhanced Raman spectroscopy on a microplate for staphylococcal enterotoxin B sensing. ACS Appl Mater Interfaces 8:15591–15597
Wang C, Wang J, Li M, Qu X, Zhang K, Rong Z, Xiao R, Wang S (2016b) A rapid SERS method for label-free bacteria detection using polyethylenimine-modified Au-coated magnetic microspheres and Au@ Ag nanoparticles. Analyst 141(22):6226–6238
Wingender J, Flemming H-C (2011) Biofilms in drinking water and their role as reservoir for pathogens. Int J Hyg Environ Health 214(6):417–423
Wolf S, Hewitt J, Rivera-Aban M, Greening GE (2008) Detection and characterization of F + RNA bacteriophages in water and shellfish: application of a multiplex real-time reverse transcription PCR. J Virol Methods 149(1):123–128
World Health Organization (WHO) (2001) Water for health: taking charge
World Health Organization (WHO) (2004) Water, sanitation and hygiene links to health: facts and figures
World Health Organization (WHO) (2007) Outbreak of water borne diseases. Cited 27 May 2019. Available from: http://www.euro.who.int/__data/assets/pdf_file/0006/97359/1.1.pdf?ua=1
World Health Organization (WHO) (2011) Guidelines for drinking-water quality. WHO Chron 38(4):104–108
World Health Organization (WHO) (2016) Number of reported cholera cases. Cited 23 May 2019. Available at: https://www.who.int/gho/epidemic_diseases/cholera/cases_text/en/
Xu M, Wang R, Li Y (2016) An electrochemical biosensor for rapid detection of E. coli O157: H7 with highly efficient bi-functional glucose oxidase-polydopamine nanocomposites and Prussian blue modified screen-printed interdigitated electrodes. Analyst 141(18):5441–5449
Yahya HSA, Jilali A, Mostareh MMM, Chafik Z, Chafi A (2017) Microbiological, physicochemical, and heavy metals assessment of groundwater quality in the Triffa plain (eastern Morocco). Applied Water Science 7(8):4497–4512
Zourob M, Elwary S, Turner AP (2008) Principles of bacterial detection: biosensors, recognition receptors and microsystems. Springer Science & Business Media, Berlin
Acknowledgements
Authors gratefully acknowledge the Government of India, Ministry of Science and Technology, Department of Science and Technology (DST), Technology Mission Division [Grant no: DST/TM/WTI/WIC/2K17/82(C)] for supporting the work. The author Uthradevi Kannan acknowledges DST—Innovation in Science Pursuit for Inspire Research (INSPIRE), [IF 160288] for the fellowship support to pursue PhD at IIT Tirupati. The authors also thank IIT Tirupati for the support.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Kannan, U., Krishna Prashanth, S., M. Maliyekkal, S. (2020). Measurement, Analysis, and Remediation of Biological Pollutants in Water. In: Gupta, T., Singh, S., Rajput, P., Agarwal, A. (eds) Measurement, Analysis and Remediation of Environmental Pollutants. Energy, Environment, and Sustainability. Springer, Singapore. https://doi.org/10.1007/978-981-15-0540-9_11
Download citation
DOI: https://doi.org/10.1007/978-981-15-0540-9_11
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-0539-3
Online ISBN: 978-981-15-0540-9
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)