Abstract
Human activities have been severely affecting forest structure and functions in humid tropics across the globe. In present study, we estimated aboveground biomass and carbon stocks along a disturbance gradient in wet tropical forests of southern Assam, India, using non-destructive sampling method. A total of 26 forest stands were surveyed and based on a disturbance index grouped into 4 categories, viz. undisturbed (UD), mildly disturbed (MLD), moderately disturbed (MD) and highly disturbed (HD) forests. Mean aboveground carbon (AGC) stocks and basal area decreased with increased disturbance index. Though phytosociological parameters such as species richness, Shannon-Wiener diversity index, tree density, basal area and AGC stocks showed a significant negative correlation with disturbance index, tree density (693 ± 11.6 trees ha−1) and Shannon-Wiener diversity index (1.98 ± 0.07) were highest in mildly disturbed forests. Aboveground carbon stocks were positively correlated with basal area (p < 0.01) and diversity indices (p < 0.01) across disturbance regimes. Tree species such as Cynometra polyandra, Mesua ferrea, Palaquium polyanthum, Mesua floribunda, Artocarpus chama and Stereospermum personatum together contributed 41.3 ± 6.2 % and 42.4 ± 6.7% of the total AGC stocks in undisturbed and mildly disturbed forests, respectively, while Artocarpu schama, Holarrhena pubescens, Mitragyna rotundifolia, Sapium baccatum, Schima wallichii and Toona ciliata contributed 47.2 ± 3.5% in moderately and 55.4 ± 4.0% in highly disturbed forests.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
6.1 Introduction
Forests play a significant role in offsetting anthropogenic CO2 emissions and hence in climate change mitigation and adaptation (Brown et al. 1996: IPCC 2013; Brienen et al. 2015). With the widespread concern about human activities increasing level of atmospheric CO2, there is a need to assess the potential of native forests in carbon sequestration and storage (C) (Johnson and Kern 2002; Sharma et al. 2010; Borah and Garkoti 2011; Gandhi and Sundarapandian 2017). Forest biomass determines the potential amount of C that can be added to the atmosphere or sequestered in terrestrial ecosystems when they are managed for meeting emission targets (Brown et al. 1999; Brienen et al. 2015). Estimation of the existing C stocks in different forest ecosystems at national and local levels would help in appropriate decision-making on C management and country’s intended nationally determined contributions under the United Nations Framework Convention on Climate Change (UNFCC) towards atmospheric carbon reduction targets as part of its international obligations in this regard (FCCC 2014; Sahu et al. 2016). In addition, forest C stock is also a useful measure for comparing structural and functional attributes of forest ecosystems across a wide range of environmental conditions (Brown 2002; Gandhi and Sundarapandian 2017).
Tropical forests are considered as the most diverse terrestrial ecosystems and largest pool of aboveground C stock covering only about 6% of the earth surface (Beer et al. 2010; Pan et al. 2013; Brienen et al. 2015). Agricultural expansion, urbanization and industrialization have been responsible for deforestation and forest degradation in the tropics (LaFrankie et al. 2006; Brienen et al. 2015) and changes in forest structure, which in turn influence ecosystem functions (Collins and Pickett 1987; Pawar et al. 2014; Sicard and Dalstein-Richier 2015). In view of the growing threats to tropical forests, it is important to understand how natural forests and their phytosociological attributes are affected by the progressive degradation due to anthropogenic activities and functional relationship between such attributes and C storage for developing suitable management plans (Chapin et al. 2000; Tilman 1988; Srivastava and Vellend 2005; Kirby and Potvin 2007).
Being part of Indo-Burma biodiversity hotspots, tropical forests of southern Assam are rich from a biodiversity standpoint (Borah and Garkoti 2011). Though such forests occupying inaccessible areas are still rich in biodiversity, those closer to the human settlements have been facing high level of pressure mainly through extraction of timber, fuel wood, grazing and overexploitation of non-timber forest products (NTFPs) by the local people (Borah 2012; Borah et al. 2014). Such anthropogenic activities have not only accelerated biodiversity loss in the forests of the study area but also impacted other ecological functions such as productivity, carbon stocks and regeneration (Borah 2012; Borah et al. 2014; Athokpam et al. 2014; Borogayary et al. 2018). In this paper we estimate the aboveground C stocks of the tree species in some wet tropical forests of southern Assam along a disturbance gradient and establish its relationships with various phytosociological attributes.
6.2 Materials and Methods
6.2.1 Study Site
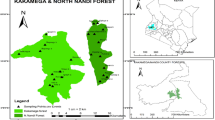
The present study was conducted in the three districts, viz. Cachar, Hailakandi and Karimganj, of southern Assam (24°08′–25°05′N, 92°15′–93°15′E) collectively known as Barak Valley (Fig. 6.1). The valley is characterized by hot and humid climate and covering 6920 km2 geographical area of which 55% is covered by forests (FSI 2017). According to Champion and Seth (1968), vegetation of the southern Assam is dominated by Cachar tropical evergreen forests and Cachar tropical semi-evergreen forests with Cynometra polyandra, Mesua ferrea, Stereospermum personatum, Artocarpus chama, Palaquium polyanthum, Mesua floribunda, Dysoxylum binectariferum, Trewia nudiflora and Pterygota alata being the most dominant tree species (Borah et al. 2014).
6.2.2 Vegetation Sampling
Based on visual reconnaissance, total 26 forest sites having different levels of disturbance (reflected by number of cut stumps, human trails and canopy cover) were selected (Fig. 6.1). The vegetation of each forest site was analysed using belt transect of 500 m × 10 m following Ganesh et al. (1996) and Borah and Garkoti (2011). Each transect was again subdivided into 50 quadrats of 10 m × 10 m size along its length. In each quadrat, all the woody plants (excluding lianas) with >10 cm circumference at breast height (CBH) were considered as tree and recorded with their CBH (Singh and Dadhwal 2009, Borah et al. 2014). The cut stumps and lopped trees in each quadrat were also counted, and their girths were measured at 10 cm from the ground for estimating the disturbance index. Plant specimens were brought to the laboratory and identified with the help of ‘Flora of Assam’ (Kanjilal 1934–1940), ‘Assam’s Flora’ (Chowdhury et al. 2005) and the herbarium of the regional centre of Botanical Survey of India located at Shillong, Meghalaya.
6.2.3 Disturbance Index
A disturbance index for each forest site was calculated following Kanzaki and Kyoji (1968), Rao et al. (1990) and Borah (2012). The disturbance index (DI) was calculated as the basal area of cut trees measured at the ground level expressed as a fraction of total basal area of all trees
Based on the disturbance index, the forest sites were classified into (1) undisturbed forest (disturbance index 0%), (2) mildly disturbed forest (disturbance index up to 20%), (3) moderately disturbed forest (disturbance index 20–40%) and (4) highly disturbed forest (disturbance index above 40%) following Bhuyan et al. (2003). Of the 26 forest sites, 5 sites were recorded as undisturbed forests, 6 forest sites were mildly disturbed, 7 sites were moderately disturbed and 8 sites were highly disturbed. Generally undisturbed and mildly disturbed forests were approximately >6 kms, and moderately and highly disturbed forests were <6 km away from the human habitation. Thus, proximity to human habitation determined the level of disturbance in the forests of the study area.
6.2.4 Phytosociological Attributes
The phytosociological data were quantitatively analysed for frequency, density, basal area, relative density, relative frequency and relative dominance (Curtis and McIntosh 1950). Distribution of tree density and basal area in different DBH classes was estimated by following Mueller-Dombois and Ellenberg (1974). Trees were categorized into ten DBH classes starting with <10 cm and ending with >90 cm class, and tree density and basal area in each DBH class were estimated.
Diversity index was calculated following Shannon and Wiener (1963) as follows:
where pi is the proportion of individuals of ith species and total number of individuals of all species.
The concentration of dominance was calculated following Simpson (1949) as follows:
where pi is the proportion of individuals of ith species and total number of individuals of all species.
6.2.5 Aboveground Biomass (AGB) and Aboveground Carbon (AGC)
Because of high species richness (132 tree species) in the studied forests, species-specific regression models were not used. Though there are several regression equations (Brown et al. 1989: Chambers et al. 2001; Chave et al. 2001; Brown and Iverson 1992) for estimating aboveground biomass (AGB), we used the following equation developed by Brown (1997) for simplicity, less prediction error and higher R2 value. The regression model is
where Y is the aboveground biomass (AGB) and D is diameter of the tree.
We assumed vegetation carbon equal to 50% of biomass for all the tree components. The estimates are based on assumption of common carbon content per biomass unit as in many other similar studies (Brown and Lugo 1982; Montagnini and Porras 1998; Borah et al. 2013).
6.2.6 Statistical Analysis
ANOVA was performed to compare the average tree density, basal area, diversity index, AGB and AGC among the forest categories with the help of SPSS 16.0. Pearson correlation coefficient was calculated to determine the functional relationships among the phytosociological attributes of the forests.
6.3 Results and Discussion
6.3.1 Forest Structure
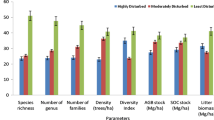
A total of 132 tree species was recorded in 26 forest stands. The highest number of species (92) was recorded in mildly disturbed forests and the lowest (47) in highly disturbed forests (Table 6.1). The Shannon diversity index of undisturbed forests ranged from 1.72 to 2.14, while it was recorded 1.78 to 2.19, 1.29 to 1.71 and 1.11 to 1.35, respectively, in mildly, moderately and highly disturbed forests. The average Shannon diversity index was highest (1.98) in mildly disturbed forests followed by undisturbed (1.94), moderately (1.51) and highly disturbed (1.23) forests (Table 6.1). The average tree density was highest in mildly disturbed forests (693 ± 12 trees ha−1) followed by undisturbed (676 ± 10 trees ha−1), moderately (675 ± 17 trees ha−1) and highly disturbed (328 ± 6 trees ha−1) forests. The average basal area was highest (42.9 ± 1.6 m2 ha−1) in undisturbed forests followed by mildly disturbed forests (39.6 ± 1.8 m2 ha−1), moderately disturbed forests (20.8 ± 1.0 m2 ha−1) and highly disturbed forests (14.4 ± 0.9 m2 ha−1). The Simpson dominance index was highest (0.075) in highly disturbed forests and lowest (0.047) in mildly disturbed forests (Table 6.1). All the phytosociological attributes (species number, tree density, diversity and basal area) were significantly different in different forest categories (Table 6.1). The forest structural analysis reveals that the species richness (47–92 species), Shannon index (1.11–2.19), tree density (328–693 trees ha−1) and tree basal area (14.37–42.91 m2 ha−1) recorded in different forest categories are comparable with earlier studies (Table 6.2) in tropical forests of north-east India by Nath et al. (2005), Deb and Sundriyal (2008), Deb et al. (2009), Borah and Garkoti (2011), Thapa et al. (2011), Borah et al. (2013, 2014) and Nandy and Das (2013).
The highest species richness, diversity and density of tree species in the mildly disturbed forests were due to the favourable conditions (such as less competition for available resources, more sunlight penetration to the forest floor through canopy gaps, etc.) for the growth and regeneration of tree species (Boarh and Garkoti 2011). Pressures such as relentless extraction of fuel wood, tree felling, and non-timber forest products including bamboo collection were found responsible for lower species richness, density, diversity and basal area in moderately and highly disturbed forests (Boarh and Garkoti 2011; Borah et al. 2014).
6.3.2 Distribution of Density and Basal Area in Different DBH Classes
Density and basal area distribution in different DBH (diameter at breast height) classes can be used as indicators of changes in population structure and species composition (Newbery and Gartlan 1996). The distribution of tree density and basal area in different DBH classes of undisturbed and mildly disturbed forests revealed a reversed J-shaped and J-shaped curve, respectively. In undisturbed and mildly disturbed forests, tree density decreased, and basal area increased with increasing DBH classes (Fig. 6.2). In both undisturbed and mildly disturbed forests, tree density was high in <10 cm DBH class, whereas tree total basal area was high in >90 cm DBH class. High tree density in lower DBH classes indicates continuous regeneration of the forest ecosystems and higher total basal area in higher DBH classes in the forests represent relatively undisturbed and old growth forests. Similar trends of density and basal area distribution were also observed for different tropical forests of north-east India by Khamyong et al. (2004), Nath et al. (2005), Deb et al. (2009), Borah and Garkoti (2011) and Borogayary et al. (2018). In moderately disturbed and highly disturbed forests, both tree density and basal area showed fluctuating distribution curves. In moderately disturbed forests, the density of younger trees (DBH <10 cm) was recorded highest, whereas trees in DBH class between 40 and 50 cm contributed more to total basal area. Lower basal area in higher DBH classes indicates the selective extraction of larger trees from these forests. In highly disturbed forests, tree density in DBH class 20–30 cm was higher, whereas trees in 80–90 cm DBH class contributed highest to total basal area. Lower density of younger trees indicates the low regeneration in the highly disturbed forests.
6.3.3 Aboveground Biomass (AGB) and Aboveground Carbon Stocks (AGC)
The mean aboveground biomass (AGB) (361.6 ± 9.0 Mg ha−1) and aboveground carbon (AGC) (180.8 ± 4.5 Mg ha−1) were estimated highest for undisturbed forests followed by mildly disturbed forests, moderately disturbed forests and highly disturbed forests. The AGB and AGC varied significantly (P < 0.001) in different forest categories (Table 6.1). The AGB in undisturbed (361.6 Mg ha−1) and mildly disturbed forests (358.0 Mg ha−1) were within the range 236.0–425.7 Mg ha−1) reported for tropical forests of India (Chaturvedi et al. 2011, Gogoi et al. 2017). Similarly, AGB of moderately (240.5 Mg ha−1) and highly disturbed forests (212.4 Mg ha−1) in the present study was comparable with disturbed tropical forests of Mizoram (116.8–278.5 Mg ha−1) as reported by Singh and Sahoo (2018).
The lower AGB and AGC recorded in the disturbed forests may be mainly due to the lower tree basal area and tree density, especially in higher size classes. Local people living in the proximity of the forests constantly extract forest resources for meeting their subsistence needs of fuel wood, construction materials, timber and NTFPs from these forests, which resulted in a decline in tree basal area as well as lower AGB and AGC in disturbed forests.
Of the total 78 species recorded in the undisturbed forests, the top 5 contributors to aboveground carbon (AGC) were Cynometra polyandra (11.5%), Palaquium polyanthum (8.8%), Artocarpus chama (7.8%), Mesua ferrea (7.4%) and Mesua floribunda (5.5%) which cumulatively contributed 41.3 ± 6.2% of the total AGC (Fig. 6.3). In mildly disturbed forests, these were C. polyandra (12.9%), M. ferrea (9.6%), S. personatum (7.3%), P. polyanthus (6.5%) and A. chama (6.5%) and together contributed 42.4 ± 6.7% of the total AGC. In moderately disturbed forests, A. chama (15.8%) was the top contributor of AGC followed by Tetrameles nudiflora, Schima wallichii, Toona ciliata and Bombax ceiba (Fig. 6.2). These five species contributed 47.2 ± 3.5% of the total AGC in moderately disturbed forests. S. wallichii, H. pubescens, S. baccatum, Mitragyna rotundifolia and T. ciliata were five major contributors to AGC and aggregately contributed 55.2 ± 4.0% of the total AGC in highly disturbed forests (Fig. 6.3). In highly disturbed forests, S. wallichii alone contributed 26.2% of the total AGC implying that species contributed higher in degraded forests were less preferred for local extraction. Removal or conservation of the top AGC contributor species greatly influenced total AGC across differently disturbed forests. Therefore, identification and conservation of such tree species are important for sustainable carbon management viewpoint in tropical forests (Kirby and Potvin 2007; Borah et al. 2013) (Table 6.3).
It was observed that the relative contribution to AGB and AGC by C. polyandra, M. ferrea, M. floribunda, P. polyanthum and S. personatum in the forest sites decreased along the disturbance gradient (Fig. 6.4). These species (C. polyandra, M. ferrea, M. floribunda, P. polyanthum and S. personatum) were dominant species in undisturbed and mildly disturbed forests (Borah 2012). However, these being the most preferred timber species in the study area, local people frequently extracted them from the forests resulting in a decrease in their presence in the disturbed forests and corresponding contribution in forest AGC. Statistical analysis revealed a significant (p < 0.1) negative correlation between disturbance and AGC of C. polyandra, M. ferrea, M. floribunda, P. polyanthum and S. personatum (Table 6.3). However, species such as H. pubescens, S. wallichii, M. rotundifolia and T. ciliata showed significantly (p < 0.0) increasing trend of AGC with the increasing levels of disturbance (Fig. 6.3; Table 6.3). Relatively higher AGC of these species in moderately and highly disturbed forests may be due to favourable conditions created by selective extraction of preferred species providing ample space for growth and regeneration and more light penetration through canopy gaps (Sahoo and Lalfakawma 2010; Asase et al. 2012). However, Bombax ceiba and Trewia nudiflora contributed higher to AGC in mildly and moderately disturbed forests (Fig. 6.4). Distribution of AGC in different DBH classes in undisturbed and mildly disturbed forests showed reversed J shaped curves while in moderately and highly disturbed forests it showed fluctuating curves (Fig. 6.5). Reversed J shaped curves in relatively undisturbed forests were due to higher total basal area in the higher DBH classes in these forests and fluctuating curves in moderately and highly disturbed forests may be due to excessive removal of certain DBH class trees from these forests.
6.3.4 Relationship of AGC with Phytosociological Attributes and Disturbance Index
AGC was positively correlated (P < 0.001) with basal area (R2 = 0.885), total number of species (R2 = 0.696) and Shannon diversity index (R2 = 0.867) (Fig. 6.6) which is similar to earlier findings of various workers for tropical forests (Terakunpisut et al. 2007; Kumar et al. 2011; Borah et al. 2013; Con et al. 2013; Thokchom and Yadava 2013; Van de Perre et al. 2018). Contrary to the present findings, studies by Enquist and Niklas (2001) and Kirby and Potvin (2007) found no correlation between AGC and diversity indices. Frivold and Frank (2002) mentioned that there may be positive or negative relationship depending on the diversity index used and relative dominance of species in mixed forests. No significant relationship was found between AGC and tree density (Fig. 6.6). The phytosociological attributes such as species richness, diversity, tree density, basal area and AGC in the present study showed significant negative correlation with disturbance index (Table 6.4). A similar relationship was also reported for different tropical forests by Bhuyan et al. (2003), Marín-Spiotta et al. (2007), Asase et al. (2012), Borah et al. (2013), Gogoi et al. (2017), and Van de Perre et al. (2018).
6.4 Conclusions
The aboveground tree carbon stocks were significantly lower in the disturbed forest stands than in undisturbed stands due to frequent removal of large trees from the formers. Locally preferred species, viz. C. polyandra, M. ferrea, M. floribunda, P. polyanthum, A. chama and S. personatum, contributed higher to the total AGC in undisturbed and mildly disturbed forests, whereas less preferred species, viz. S. wallichii, H. pubescens, S. baccatum, M. rotundifolia and T. ciliata, contributed higher in moderately and highly disturbed forests. Contribution of older trees with higher girths was higher to the total AGC stocks than the younger trees in lower girth classes irrespective of disturbance level. The phytosociological attributes, viz. tree diversity, species richness and basal area, revealed significant positive correlations with AGC stocks across disturbance gradient.
References
Athokpam FD, Garkoti SC, Borah N (2014) Periodicity of leaf growth and leaf dry mass changes in the evergreen and deciduous species of southern Assam, India. Ecol Res 29(2):153–165
Asase A, Asitoakor BK, Ekpe PK (2012) Linkages between tree diversity and carbon stocks in unlogged and logged West African tropical forests. Int J Biodivers Sci Ecosyst Serv Manag 8(3):217–230
Beer C, Reichstein M, Tomeller E et al (2010) Terrestrial gross carbon dioxide uptake: global distribution and covariation with climate. Science 329:834–838
Borah N (2012) Community structure, tree regeneration and utilization of forest resources in Cachar and Hailakandi districts of Assam, India. Ph.D. thesis, Assam University, Silchar, Assam, India
Borah N, Garkoti SC (2011) Tree Species composition, diversity, and regeneration patterns in undisturbed and disturbed forests of Barak Valley, South Assam, India. Int J Ecol Environ Sci 37(3):131–141
Borah N, Nath AJ, Das AK (2013) Aboveground biomass and carbon stocks of tree species in tropical forests of Cachar district, Assam, northeast India. Int J Ecol Environ Sci 39(2):97–106
Borah N, Athokpam FD, Garkoti SC, Das AK, Hore DK (2014) Structural and compositional variations in undisturbed and disturbed tropical forests of Bhuban hills in south Assam, India. Int J Biodivers Sci Ecosyst Serv Manage 10(1):9–19
Borogayary B, Nath AJ, Das AK (2018) Tree species composition and population structure of a secondary tropical evergreen forest in Cachar district, Assam. J Environ Biol 39:67–71
Brienen RJW, Brienen RJW, Phillips OL et al (2015) Long-term decline of the Amazon carbon sink. Nature 519:344–348
Brown S (1997) Estimating biomass and biomass change of tropical forests: a primer. FAO forestry paper 134. Food and Agriculture Organization, Rome, p 55
Brown S (2002) Measuring, monitoring and verification of carbon benefits for forest-based projects. Philos Trans R Soc A 360:1669–1683
Brown S, Iverson LR (1992) Biomass estimates for tropical forests. World Resour Rev 4:366–384
Brown S, Lugo AE (1982) The storage and production of organic matter in tropical forests and their role in the global carbon cycle. Biotropica 14:161–187
Brown S, Gillespie A, Lugo A (1989) Biomass estimation methods for tropical forests with applications to forest inventory data. For Sci 35:881–902
Brown S, Sathaye J, Cannell M, Kauppi P (1996) Mitigation of carbon estimation to the atmosphere by forest management. Commonw For Rev 75(1):80–91
Brown S, Schroeder P, Kern JS (1999) Spatial distribution of biomass in forest of the eastern USA. For Ecol Manag 123:81–90
Bhuyan P, Biodiversity and Conservation 12 (8):1753–1773
Chambers JQ, Santos JD, Ribeiro RJ, Higuchi N (2001) Tree damage, allometric relationships, and aboveground net primary production in central Amazon forest. For Ecol Manag 152:73–84
Champion HG, Seth SK (1968) A revised survey of the forest types of India. Govt. of India Publications, New Delhi
Chapin FS, Zavaleta ES, Eviner VT, Naylor RL, Vitousek PM, Reynolds HL, Hooper DU, Lavorel S, Sala OE, Hobies SE, Mack MC, Diaz S (2000) Consequences of changing biodiversity. Nature 405:234–242
Chaturvedi RK, Raghubanshi AS, Singh JS (2011) Carbon density and accumulation in woody species of tropical dry forest in India. For Ecol Manag 262:1576–1588
Chave J, Rikra B, Dubois MA (2001) Estimation of biomass in a neotropical forest of French Guiana: spatial and temporal variability. J Trop Ecol 17:79–96
Chowdhury S, Nath AK, Bora A, Das PP, Phukan U (2005) Assam’s flora. Assam Science Technology and Environment Council, Guwahati
Collins BS, Pickett STA (1987) Influence of canopy openings on the environment and herb layer in a northern hardwood forest. Vegetatio 70:3–10
Con TV, Thang NT, Ha DTT, Khiem CC, Quy TH, Lam VT, Do TV, Sato T (2013) Relationship between aboveground biomass and measures of structure and species diversity in tropical forests of Vietnam. For Ecol Manage 310:213–218
Curtis JT, McIntosh RP (1950) The interrelations of certain analytic and synthetic phytosociological characters. Ecology 31:434–455
Deb P, Sundriyal RC (2008) Tree regeneration and seedling survival patterns in old growth lowland tropical rainforest in Namdapha National Park, North-east India. For Ecol Manage 225:3995–4006
Deb P, Sundriyal RC, Shankar U (2009) Tree diversity and population structure in a lowland tropical rainforest in the Eastern Himalaya, India. Indian For 135(11):1526–1544
Enquist BJ, Niklas KJ (2001) Invariant scaling relation across tree dominated communities. Nature 410:655–660
FCCC-Framework Convention on Climate Change (2014) Report of the conference of the parties on its nineteenth session, held in Warsaw from 11 to 23 November 2013, 43pp
Frivold LH, Frank J (2002) Growth of mixed birch-coniferous stand in relation to pure coniferous stand at similar sites in south-eastern Norway. Scand J For Res 17:139–149
FSI (Forest Survey of India) (2017) State Forest report. Ministry of Environment and Forests, Dehradun, p 2017
Gandhi DS, Sundarapandian S (2017) Large-scale carbon stock assessment of woody vegetation in tropical dry deciduous forest of Sathanur reserve forest, Eastern Ghats, India. Environ Monit Assess 189:187–196
Ganesh T, Ganesan R, Devy MS, Davidar P, Bawa KS (1996) Assessment of plant biodiversity at mid-elevation evergreen forest of Kalak and Mundanthuari Tiger Reserve, Western Ghats, India. Curr Sci 71:379–392
Gogoi A, Sahoo UK, Singh SL (2017) Assessment of biomass and total carbon stock in a tropical wet evergreen rainforest of Eastern Himalaya along a disturbance gradient. J Plant Biol Soil Health 4(1):1–8
IPCC (Intergovernmental Panel on Climate Change) (2013) Climate change. The physical science basis. Working group I contribution to the IPCC fifth assessment report. Cambridge University Press, Cambridge
Johnson MG, Kern JS (2002) Quantifying the organic carbon held in forested soils of the United States and Puerto Rico. In: Kimble JM, Heath LS, Birdsey RA, Lal R (eds) The potential of U.S. forest foils to sequester carbon and mitigate the greenhouse effects. Lewis Publishers (CRC Press), Boca Raton, pp 47–72
Kanjilal UN (1934–1940) Flora of Assam, vol I–IV. Govt. of Assam, Shillong
Kanzaki M, Kyoji Y (1968) Regeneration in subalpine coniferous forests: mortality and pattern of death of canopy trees. Bot Mag Tokyo 99(1053):37–52
Khamyong S, Lykke AM, Seramethakun D, Barford AS (2004) Species composition and vegetation structure of an upper montane forest at summit of Mt. Doi Inthanon, Thailand. Nord J Bot 23:83–97
Kirby KR, Potvin C (2007) Variation in carbon storage among tree species: Implication for management of small scale carbon sink project. For Ecol Manag 246:208–221
Kumar R, Gupta SR, Singh S, Patil P, Dadhwal VK (2011) Spatial distribution of forest biomass using remote sensing and regression models in northern Haryana, India. Int J Ecol Environ Sci 37(1):37–47
LaFrankie JV, Ashton PS, Chunyong GB, Co L, Condit R, Davaries SJ, Fooster R, Hubbell SP, Kenfack D, Lagunzad D, Losos EC, Nor NSM, Tan S, Thomas DW, Valencia R, Villa G (2006) Contrasting structure and composition of the understory in species rich tropical rainforests. Ecology 87:2298–2305
Marín-Spiotta E, Ostertag R, Silver WL (2007) Long-term patterns in tropical reforestation: plant community composition and aboveground biomass accumulation. Ecol Appl 17(3):828–839
Montagnini F, Porras C (1998) Evaluating the role of plantations as carbon sink: an example of an integrative approaches the humid tropics. Environ Manag 22(3):459–470
Mueller Dombois D, Ellenberg H (1974) Aims and methods of vegetation analysis. Wiley, New York
Nandy S, Das AK (2013) Comparing tree diversity and population structure between a traditional agroforestry system and natural forests of Barak valley, Northeast India. Int J Biodivers Sci Ecosyst Serv Manage 9(2):104–113
Nath PC, Arunachalam A, Khan ML, Arunachalam K, Barbhuiya AR (2005) Vegetation analysis and tree population structure of tropical wet evergreen forest in and around Namdapha National Park, northeast India. Biol Conserv 14:2109–2136
Newbery DMC, Gartlan JS (1996) A Structural Analysis of Rain Forest at Korup and Douala-Edea, Cameroon. Proc R Soc Edinburgh 104(B):107–124
Pan Y, Birdsey RA, Phillips OL, Jackson RB (2013) The structure, distribution, and biomass of the world’s forests. Annu Rev Ecol Evol Syst 44:593–622
Pawar GV, Singh L, Jhariya MK, Sahu KP (2014) Effect of anthropogenic disturbances on biomass and carbon storage potential of a dry tropical forest in India. J Appl Nat Sci 6(2):383–392
Rao P, Barik SK, Pandey HN, Tripathi RS (1990) Community composition and tree population structure of subtropical humid forest along a disturbance gradient. Vegetatio 88:151–192
Sahoo UK, Lalfakawma (2010) Population dynamics of Schima wallichii in an undisturbed vs a disturbed tropical forest stand of North-East India. Int J Ecol Environ Sci 36(2):157–165
Sahu SC, Suresh HS, Ravindranath NH (2016) Forest structure, composition and above ground biomass of tree community in tropical dry forests of Eastern Ghats, India. Notul Sci Biologi 8(1):125–133
Shannon CE, Weaver W (1963) The Mathematical theory of communication. University of Illinois Press, Urbana, p 117
Sharma CM, Baduni NP, Gairola S, Ghildiyal SK, Suyal S (2010) Tree diversity and carbon stocks of some major forest types of Garhwal Himalaya, India. For Ecol Manag 260:2170–2179
Sicard P, Dalstein-Richier L (2015) Health and vitality assessment of two common pine species in the context of climate change in southern Europe. Environ Res 137:235–245
Simpson EH (1949) Measurement of diversity. Nature 63:688
Singh S, Dadhwal VK (2009) Manual on spatial assessment of vegetation carbon pool of India. Indian Institute of Remote Sensing (National Remote Sensing Centre), ISRO, Department of Space, Govt. of India, Dehradun
Singh SL, Sahoo UK (2018) Assessment of biomass, carbon stock and carbon sequestration potential of two major land uses of Mizoram, India. Inter J Ecol Environ Sci 44(3):293–306
Srivastava D, Vellend M (2005) Biodiversity-ecosystem function research: is it relevant to conservation? Annu Rev Ecol Syst 36:267–294
Terakunpisut J, Gajaseni N, Ruankawe N (2007) Carbon sequestration potential in aboveground biomass of Thong Pha Phun national forest, Thailand. Appl Ecol Environ Res 5:93–102
Thapa N, Upadhaya K, Baishya R, Barik SK (2011) Effect of plantation on plant diversity and soil status of tropical forest ecosystems in Meghalaya, northeast India. Int J Ecol Environ Sci 37(1):61–73
Thokchom A, Yadava PS (2013) Biomass and carbon stock assessment in the sub tropical forests of Manipur, north east India. Int J Ecol Environ Sci 39(2):107–113
Tilman D (1988) Plant strategies and the dynamics and structure of plant communities. Princeton University Press, Princeton
Van de Perre F, Willig MR, Presley SJ et al (2018) Reconciling biodiversity and carbon stock conservation in an Afrotropical forest landscape. Appl Ecol 4(3):1–9
Acknowledgements
Financial support from the Department of Biotechnology and Department of Science and Technology (PURSE Grant) is thankfully acknowledged. We also thank Prof. Uma Shankar, North Eastern Hill University, Shillong, for his support as coordinator of multi-institutional network research programme.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Borah, N., Athokpam, F.D., Das, A.K., Garkoti, S.C. (2019). Aboveground Tree Carbon Stocks Along a Disturbance Gradient in Wet Tropical Forests of South Assam, India. In: Garkoti, S., Van Bloem, S., Fulé, P., Semwal, R. (eds) Tropical Ecosystems: Structure, Functions and Challenges in the Face of Global Change. Springer, Singapore. https://doi.org/10.1007/978-981-13-8249-9_6
Download citation
DOI: https://doi.org/10.1007/978-981-13-8249-9_6
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-13-8248-2
Online ISBN: 978-981-13-8249-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)