Abstract
The borer, Conogethes punctiferalis species infests a wide variety of crops and plant parts, is difficult to manage with insecticides. However, newer compounds, spinetoram and cyazypyr, have shown some promise which can be selectively applied at appropriate time and dose. Timely harvests, clean cultivation, encouragement of pollinators and natural enemies, fruit thinning and bagging and balanced nutrition can greatly help in managing the Conogethes populations. Mass trapping of adult moths by pheromone traps and botanical formulations can lead to realistic management of the pest resulting in sustainable crop yields. Such a set of management tools can be practical, cheap and environmentally sound.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
22.1 Introduction
Conogethes sahyadriensis has been identified on zingibers from India (Shashank et al. 2018). Conogethes (= Dichocrocis) punctiferalis (Guenee) is presently considered as one of the most important and widely spread lepidopterous pests causing economical losses to cultivated crops as well as the wild plant species. Larvae of this pest are polyphagous, feeding more than 120 plant species belonging to 16 families (Sekiguchi 1974). C. punctiferalis has been a species complex (Solis 1999) and difficult to identify at the species level. There has been no taxonomic revision or change to separate the species within genus (Robinson et al. 1994). Armstrong (2010) described Conogethes spp. and mentioned as species complex. This pest is commonly called as yellow peach moth, but there are several other common names. This pest has been considered a potential hazard (CPC 2007). Feeding behaviour of this pest is different from host to host, and there is varied life table (Chakravarthy et al. 2012; Thyagaraj 2003).This pest breeds throughout the year in India and parts of Australia (USDA 1957). Most adults are active at night, and they remain on the back of host leaves and bark (CPC 2007) during the day. Both male and female moths feed on the nectar of the host and surrounding plants (CPC 2007). Till date, 15 species have been identified by DNA bar codes and deposited in the Barcode of Life Data Systems (BOLD).
The advantage of IPM tools for the efficient management of C. punctiferalis has been tried by several workers throughout the world on crops this pest attacks. An attempt is being made to present in this chapter an overview on management of the pest. Other chapters in this book also cover management aspect, but briefly. C. punctiferalis is clearly potential and hazardous pest throughout its geographical range of occurrence. The methods, in practice, are far from satisfactory for the pest.
22.2 Cultural Practices
If simple, practicable management tools are taken at appropriate time, then substantial yield losses can be prevented. Cultural practices followed in different crops play an important role in reducing pest damage. In cardamom plantations, some of the practices reduced the pest incidence effectively. Following clean cultivation, i.e. removal of alternate host plants, maintaining proper shade, proper weeding and removal of affected plant parts significantly C. punctiferalis, currently identified as C. sahyadriensis on zingiber crop plants from South India (Shashank et al. 2018) damage to panicle, shoot and capsules. The use of recommended N fertilizer reduces the incidence of pest attack wherein increased dose increases the pest damage. Plant population also contribute to the pest incidence, i.e. the higher the population, the higher the pest damage has been recorded. Therefore, plant density played a role in increasing or decreasing the borer infestation (Thyagaraj 2003) (Tables 22.1and 22.2). Sloan (1945) suggested that harvesting of sorghum grains as soon as possible and drying and threshing immediately aid in reducing infestation. Appropriate cultural practices coupled with clean management of farm and orchards prevent pest incidence to some extent (Chakravarthy and Khan 1987). Chakravarthy and Thyagaraj (1999) reported that increased levels of nitrogen fertilizers significantly increased pest damage on cardamom. Sridharan et al. (1990) reported the higher pest damage on cardamom in closer planting plantations. Phosphorus fertilization has been known to reduce the incidence of pest, and low potassium supply frequently favours pest damage. Application of biofertilizers makes the plants hardier to attack. Excessive use of nitrogenous fertilizers attracts pests, and cultivation of trap crops is a basic strategy in which major population of pest infesting main crop can be reduced by trapping. Crop rotation is an effective cultural tool for reducing pest population (Singh et al. 2009), as also in Conogethes. Crop rotation is difficult to accomplish in perennial crops like plantation and spice crops. Here, as in cardamom plantations, mixed cropping can be adopted.
22.3 Host Plant Resistance and Transgenic
Plant resistance is a natural and useful strategy that can be exploited in the field of pest management. It constitutes a cheap and practically feasible input in the IPM system. Sharma et al. (1995) evaluated select cultivars of castor against yield potential and resistant once to C. punctiferalis and found positive results. The delta toxin of the Bt is expressed in many genetically modified crops for control of major pests in several countries (Singh et al. 2009). Devasahayam et al. (2010) tested some of the resistant varieties in ginger germplasm against Conogethes sp. and reported that none of them showed resistance. Wang Jiang et al. (2013) studied the molecular bar coding for differentiating species of Conogethes in order to induce effective management practices. Hence, the same tool can be adopted in the management of C. punctiferalis elsewhere. There is a scope to produce GM crops in different plant species which are attacked by C. punctiferalis. Li De Yu et al. (2015) showed the effect of different host plants on the development and reproduction of yellow peach moth, C. punctiferalis, and suggested this could be well exploited in the pest management programmes. Oviposition was induced by plant olfactory stimuli, but also that many non-host plants potentially stimulate oviposition. Even in host plants, biophysical plant features can restrict oviposition, suggesting that the real host range for oviposition may be narrower than that expected from laboratory assessments based on olfactory responses of female moths (Luo and Honda 2015). Li De Yu et al. (2015) reported the effects of different host plants on the development and the reproduction of C. punctiferalis; this would help in understanding the population dynamics on different host plants. Screening of new isolates of Bt for cry1 genes and testing of toxicity against C. punctiferalis proved effective. New isolates of Bt, T27, showed 100% larval mortality on the fifth day after treatment (Manikandan et al. 2016). Joseph Rajkumar et al. (2002) conducted a study on luminal protease in C. punctiferalis and identified potential targets for proteinaceous biopesticides, such as protease inhibitors. They have explained the possible incorporation of protease inhibitors into transgenic plants. Shashank et al. (2014) conducted behavioural studies on C. punctiferalis and revealed that there are host-associated populations with occurrence of cryptic species. Shetty et al. (2015) studied the correlation coefficient and occurrence of pest and diseases in turmeric crop in hill zone of Karnataka and found that varietal characters significantly influenced the pest damage. This study would certainly be useful in developing resistant varieties through genetic engineering against the borer damage.
22.4 Mechanical Methods
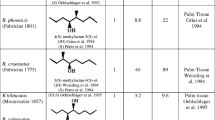
Trapping adult moths using components of pheromone is an effective tool for the management. Walsh (1867) reported that insect speciation could be helped by shifting and adapting to new host plants. Larvae of C. punctiferalis can feed upon more than hundred host plants such as fruit trees, annual cultivated crops, spices crops, vegetables and wild plant species as well (Lu et al. 1995). Thyagaraj (2003) showed that the use of pheromone substances to trap C. punctiferalis adult moths in the field conditions is an effective tool (Table 22.3). Boo (1998) showed variations in sex pheromone composition of a few lepidopteran species as it is species specific. Konno et al. (1980) extracted pheromone compounds from female moths of C. punctiferalis and field evaluated and good response in trapping adult moths. Konno et al. (1981) studied the mating behaviour in the laboratory and found that artificial pheromone compounds can be successfully used to trap adult moths. Kondo et al. (2008) evaluated different sex pheromones and reported that there are differences in species based on location. Accordingly, the traps are to be designed. Mori et al. (1990) synthesized two biological pheromone of C. punctiferalis to use as mechanical methods of trapping adult moths and found promising. Cai and Mu (1993) designed cup traps for attraction of moths as one of the IPM tools. Liu et al. (1994) suggested sexual attraction as effective mechanical methods for pest management. Tomomatsu et al. (1995) suggested the mechanical eradication of C. punctiferalis larvae and eggs using methyl bromide fumigation and reported 100% motility. High degree of response uniformity between the species and the sexes was observed (Jyothi et al. 1996). Chakravarthy and Thyagaraj (1997 and 1998) showed synthetic pheromone compounds are effective in trapping adult moths in cardamom plantations. Kimura and Honda (1999) trapped C. punctiferalis moths in the peach plantation using pheromone traps.
Luo and Honda (2015) suggested segmentation method for touching pest image could improve the segmentation performance and had a remarkable significance for the future extraction, identification and management. From these reports it could be that mechanical methods of collection of C. punctiferalis adult moths is wonderful tool successfully employed in the pest management. Wang NianFeng et al. (2016) showed the efficacy of trap crops in peach orchards as a potential tool for the management of C. punctiferalis. Gaur (2014) and Akashe et al. (2015) showed that meteorological parameters affect the growth and development; this key factor can well be exploited in the management of C. punctiferalis. Disinfecting chestnuts by validation of radio frequency treatments as an alternative non-chemical method proved effective against C. punctiferalis (Hou LiXia et al. 2015). Hou LiXia et al. (2015) showed that thermal death kinetics of C. punctiferalis at different life stages, heating rate and temperature are essential for developing postharvest treatments to control pests in chestnuts. This information is useful and effective in developing protocols for postharvest pest management. Jia XiaoJian et al. (2015) showed that the cDNA cloning, expression profiling and binding affinity assay of the pheromone-binding protein Cpun-PBP1 in C. punctiferalis help in developing artificial traps for the Conogethes management.
22.5 Use of Botanicals/Biorationals
Several plant species possess insecticidal properties, but only few are exploited for pest management. None of the tested botanicals are effective against C. punctiferalis (Thyagaraj et al. 2002). Particularly, the use of neem- and tobacco-based insecticides is less effective. Thyagaraj (2003) tested few botanicals to suppress C. punctiferalis in cardamom plantations and proved ineffective (Table 22.4). Eapan (1994) reported that tested botanicals failed to suppress the Conogethes population. Gopakumar et al. reported that neem insecticides failed to reduce pests on cardamom. Varadarasan (2001) evaluated botanicals to control cardamom pests and reduced the cost of cultivation in Kerala, South India. Joseph Rajkumar et al. (2002) tested biorationals against cardamom pests and found that none of them were effective except fish oil insecticidal soaps (FOIS) 2.5 % + tobacco extract 2.5 %, but they found that lowest number of honey bees visited; this again affected the capsule yield. Bhat (2000) also reported adverse effects of neem on areca nut production with phytotoxicity on inflorescence and button shedding as well. Rajabaskar and Regupathy (2013) found that none of the botanicals were effective against pests with chemical molecules. They also found that the use of any individual components of IPM was less effective suppressing in controlling C. punctiferalis population than harmonious blend of components.
22.6 Biological Agents
Numbers of biopesticides are also available commercially for better management of C. punctiferalis. Devasahayam (2000) evaluated biopesticides against cardamom pests and reported some of the promising ones that can be used in the pest management. Biopesticides meet the needs of the export rules where the importation laws are straight and stringent. But they have certain limitations also as their efficacy under field conditions always includes depending on several factors like inactivation by sunlight, low persistence and slow in action. Feeding behaviour, stage of the crops, plant species and ecological factors govern the efficacy of biopesticides and bio-agents. Certain fungal spores when sprayed on buds and young developing fruits attract ovipositing Conogethes moths that deposit eggs on fruits/buds, but later succumb to the action of the fungus. Ali et al. (2014) reported for the first time larval parasitoids on C. punctiferalis from Tamil Nadu Agricultural University (TNAU), Coimbatore, India. They have not mentioned any impact assessment on the extent of pest control. Rashid Pervez et al. (2014) isolated and identified positive strains of entomopathogenic nematodes (EPNs) and reported as they are of great potential for biological control of C. punctiferalis. Ingle et al. (2016) tested host range assay of entomopathogenic fungi Nomuraea rileyi and Metarhizium anisopliae on major insect pest of common occurrence in Vidarbha (Maharashtra) in select crop ecosystems. They proved that effective on C. punctiferalis. Golden-backed woodpecker Dinopium benghalensis L. was found feeding on larvae of C. sahyadriensis in cardamom plantations at Mudigere and surrounding areas as reported by Chakravarthy (1988) from South India. Patel and Gangrade (1971) found Microbracon hebtor parasitizes larvae of C. punctiferalis. Jacob (1981) reported that Myosoma spp., Xanthopimpla australis and a nematode parasitized larval stages of C. punctiferalis. Details on biological control are also found in a separate chapter in this book.
22.7 Chemical Methods
Since C. punctiferalis exhibits highly diverse feeding behaviour, several workers evaluated different molecules and reported their efficacy. Thyagaraj (2003) evaluated few chemical molecules and reported their efficacy and importance in cardamom ecosystems, but their ill-effects on useful insects have not been assessed (Table 22.5). Wilson et al. (1978) evaluated few chemical molecules and reported their efficacy on the cardamom pests. Mandal et al. (1978), Nari et al. (1979), Kumaresan (1982, 1983), Krishnamurthy et al. (1989), He (1997), Chakravarthy and Thyagaraj (1998) and Sharma et al. (1992) tested different chemical molecules in different crops against C. punctiferalis and reported the most effective one. Chethan et al. (2016) evaluated some of the chemical molecules against C. sahyadriensis and found effective, but no information on pollinators was provided. This is because pollinators play an important role in cardamom pollination as it is 100 % cross-pollinated crop. Pollination is affected particularly by a large number of hymenopterans. From the reports made available in the publications, it is clear that select chemical molecules might have given effective control, but their effect on the ecological systems and useful entities have not been mentioned anywhere. This aspect should be considered as many of the crops solely depend on the activity of pollinators for the pollination. The acute and selective toxicity of profenofos against the Conogethes borer on small cardamom was much higher compared to endosulfan which is now banned in India (Renuka and Regupathy 2008).
Spinetoram is new class of chemicals in the spinosyn group. It is a semi-synthetic insecticide recovered from fermenting substances of Saccharopolyspora spinosa. Spinetoram is a broad-spectrum insecticide with effectiveness against a broad range of pests (Table 22.1). The formulation (01ANA) has good insecticidal properties with rapid action and short preharvest interval (Report, Sumitomo Chemical Co. Ltd. 2012). This formulation has proved effective against whiteflies, aphids, thrips, leaf miners and lepidopteran pests. In field trials at GKVK farm and IIHR farm, Bengaluru, during 2016–2017, spinetoram @50 mg a.i./litre solution or 47 ppm has proved effective against C. punctiferalis on castor (Chakravarthy AK, 2015, Pers observ). Spinetoram interferes with nicotinic acetylcholine receptors and gamma-aminobutyric acid (GABA) receptors. One of the important properties of spinetoram is that it kills pests through direct contacts but also those that feed on treated plant parts. Spinetoram is a multicomponent compound but comprising mainly of major and minor components (Fig. 22.1).
Major and minor components of spinetoram. (Source: Sumitomo Chemical Co. Ltd. 2012)
Spinetoram has also translaminar activity and is effective against soil-dwelling pests. Spinetoram has superior efficacy against Hellula undalis (Fabr.) over emamectin benzoate, flubendiamide and chlorantraniliprole (Fig. 22.2). It has also shown high activity against Thrips tabaci L. (Fig. 22.3).
Efficacy against H. undalis on cabbage by foliar spray. (Source: Sumitomo Chemical Co. Ltd. 2012)
Efficacy against T. tabaci on cabbage by foliar spray. (Source: Sumitomo Chemical Co. Ltd. 2012)
22.8 Integrated Pest Management
Dependence on chemical pesticides has led to the problems such as insect pest resistance, resurgence and escalating cost of cultivation. Considering the ill-effects of chemical pesticides and the growing preference for chemical-free bio-products, efforts should be made to develop and popularize IPM technologies/strategies. Hence, the integration of all possible management strategies is thought and tested in several crops attacking different pests. Similarly, to control C. punctiferalis, many workers evaluated some of the available methods and reported the possibilities of using the same techniques in future as holistic approaches. Thyagaraj (2003) evaluated some of the strategies, viz. cultural, mechanical, botanicals and chemicals on C. sahyadriensis in cardamom plantations and found that individual methods failed to reduce the pest damage and recorded good control in combined strategies (Table 22.6). Chakravarthy and Khan (1987) reported the effectiveness of IPM tools in the management of C. sahyadriensis, and they found satisfactory reduction in pest population in combined and integrated manner. Rajagopal et al. (1987) evaluated some of the IPM tools for the management and found effective than individual methods. It has been reported that IPM tools are effective against cardamom thrips and shoot and fruit borer in Karnataka (Anonymous, 1990). Koya et al. (1991) suggested the similar pest management approaches for the management of ginger pests in Kerala. Hata et al. (1992) reported the IPM approaches as a novel method of pest management in red ginger in Hawaii. Devasahayam and Koya (1999) found IPM modules to be the most effective for cardamom pests in Kerala. Sarkar et al. (2016) evaluated bio-efficacy and non-target toxicity of an IPM compatible thiourea compound diafenthiuron against cardamom thrips and shoot and fruit borer and found effective and suggested that this can be included in the IPM schedule in other areas. Thrips and the borer are the major pests in all the cardamom cultivated areas in South India.
Decision-making in pest management requires a thorough analysis of agroecosystems. With increasing availability of information and understanding on how pests cause damage, new strategies are being devised to enhance protection that is possible. Plant breeding and biotechnology tools in combination are already providing new materials for better plant management. The pest management tools that have deployed have had a positive impact on the environment by reducing the amount of chemical pesticides applied to these crops. Farmers/growers’ awareness in understanding the agroecosystems plays a very important role in practising IPM strategies. So, emphasis is now often laid on non-chemicals. An environmentally sustainable agricultural practice is recognized worldwide due to ecological imbalance caused by intensive agricultural practices. In order to address the adverse impact, integrated pest management has been evolved on ETL-based approach. Ecological engineering for pest management, a new paradigm, is gaining importance as a strategy for promoting biointensive integrated pest management. Since, there is rising public concern about the potential adverse effects of chemical pesticides on the human health, environment and biodiversity, IPM strategies are gaining importance. Crop protection practices, however, are advanced for the management of Conogethes spp. across different crops and locations.
Simple strategies are:
-
Cultivate variety resistant/tolerant to Conogethes spp.
-
Select healthy seeds/seedlings/planting materials for cultivation.
-
Treat the seed/seedlings/planting materials with recommended soil pesticides.
-
Follow appropriate spacing.
-
Maintain good soil health, not too acidic/alkaline.
-
Maintain required nutrients in the soil, balanced nutrition with biorational materials.
-
Adopt proper irrigation systems like drip irrigation.
-
Follow crop rotation, mixed cropping and intercropping.
-
Use selective/recommended chemical molecules with need-based applications.
-
Encourage natural enemies and inimical factors that suppress the borer populations.
C. punctiferalis management is really hard and tricky; the farmers/growers are facing difficulty. Unless they are made to know the weak stages or vulnerable conditions of this pest in different cropping situations, it is risky to grow crops. As it is already mentioned that C. punctiferalis is a potential and hazardous difficult-to-manage pest, large groups of workers throughout the world have tried their best by adopting multiple strategies to manage this pest. Such useful strategies have been discussed in this chapter in order to achieve sustainable crop yields. Information on the IPM of Conogethes is also discussed in other chapters of this book.
References
Akashe VB, Indi DV, Patil SR, Jadhav JD, Pawar PB (2015) Incidence of insect pest damage in castor in relation to meteorological parameters in the scarcity zone of Maharastra. J Agrometeorology 17(1):139–141
Ali MAA, Manoharan T, Kuttalam S (2014) Trends in Bioscience 7(22):3771–3773
Anonymous (1990) Improved cultivation practices for horticultural crops- hill region. UAS, Bangalore, pp 10–11
Armstrong K (2010) DNA bar-coding: a new module in New Zealand’s plant biosecurity diagnostic toolbox. Bull OEPP/EPPO 40:91–100
Bhat VR (2000) Gameticidal effect of plant based pesticides in aercanut. Indian Farming 50(7):33–36
Boo KS (1998) Variation in sex pheromone composition of a few selected lepidopteran. J Asia Pac Entomol 1(1):17–23
CAB International (2007) Crop protection compendium. CAB International, Wallingford
Cai RX, Mu XL (1993) Trap trials for Conogethes punctiferalis with sex pheromones in citrus orchards. China Citrus 22(1):33
Chakravarthy AK (1988) Predation by golden backed wood pecker Dinopium bengalensis on cardamom shoot borer Conogethes punctiferalis. J Bombay Nat Hist Soc 85(2):427
Chakravarthy AK, Khan MM (1987) Innovative tools for protecting cardamom from herbivores- plea for crop management. Q News Lett Asia Pac Plant Prot Comm 30(34):12–17
Chakravarthy AK, Thyagaraj NE (1997) Response of cardamom shoot and fruit borer to different pheromone compounds. Insect Environ 2(4):127–128
Chakravarthy AK, Thyagaraj NE (1998) Evaluation of certain synthetic pheromones of the cardamom shoot and fruit borer Conogethes punctiferalis in Karnataka. Pest Manag Hortic Eco-syst 4(2):78–82
Chakravarthy AK, Thyagaraj NE (1999) Effect of different NPK fertilizer levels on the incidence of cardamom pests. Insect Environ 4(4):139–140
Chakravarthy AK, Shashank PR, Doddabasappa B, Thyagaraj NE (2012) Status of shoot and fruit borer, Conogethes spp (Lepidoptera: Crambidae) in the orient: biosystematics, biology and management. J Insect Sci 25(2):107–117
Chethan KS, Swamy BCH, Sowmya DK, Nagaraja A, Sathish R (2016) Evaluation of insecticide molecules against Shoot borer Conogethes punctiferalis. Environ Ecol 34(1A):225–228
Devasahayam S (2000) Evaluation of biopesticides for the management of Conogethes punctiferalis on ginger. In: Proceedings of the Centennial conference on spice and aromatic plants. Indian Society for Spices, Calicut, India, pp 276–277
Devasahayam S, Koya KMA (1999) Integrated management of insect pests in spice. Indian J Areca Nut, Spice Med Plants 1(1):19–23
Devasahayam S, Jacob TK, Abdul Koya KM, Sashikumar B (2010) Screening of ginger germplasm for resistance to Conogethes punctiferalis. J Med Aromat Plant Sci 32(2):137–138
Eapan SJ (1994) Effect of three granular insecticides on damage by thrips and borer in small cardamom. J Entomol Res 18(2):181–183
Gaur RK (2014) Diversity of insect pests of castor, Ricinus communis and their ecological interaction in South-West Haryana. Int J Farm Sci 4(4):147–152
Hata TY, Hara AH, Jang EB, Imaino AS, Hu BKS, Tenbrink VL (1992) Pest management before harvest and insecticidal dip after harvest as a system approach to quarantine security for red ginger. J Econ Entomol 85(6):2310–2316
He RQ (1997) Study on the control of yellow peach moths China. South –China-Fruits 26(4):33
Hou LX, Hou JC, Li ZX, Jonson JA, Wang SJ (2015) Validation of radio frequency treatments as alternative non-chemical methods for disinfecting chestnuts. J Stored Prod Res 63:75–79
Ingle YV, Vadaskar RM, Burgoni EB (2016) Host spectrum and relative virulence of entomological fungi Nomuraea rileyi and Metarhizium anisopliae. Curr Biotica 9(4):348–355
Jacob SA (1981) Biology of Conogethes punctiferalis on turmeric. J Plantation Crops 9(2):119–123
Jia XJ, Hao SD, Du YL, Zhang MZ, Quin ZC, Wang JZ, Wang HX, Ji WR (2015) cDNA cloning ,expression profiling and binding affinity assay of the pheromone binding protein Cpun – PBP1 in the yellow peach moth Conogethes punctiferalis. Acta Entomol Sin 58(11):1167–1176
Joseph Rajkumar A, Sainamole Kurian P, Backiyarani S, Murugan M (2002) Evaluation of botanicals against cardamom pests in cardamom plantations. J Spice Aromat Crops 11(2):132–134
Jyothi KN, Prasanna AL, Sighamony S, Prasad A, RandYadav JS (1996) Comparative electro-antenogram study of Parallelia algiria and Conogethes punctiferalis to genera plant volatiles. J Entomol Res 20(3):189–195
Kimura T, Honda H (1999) Identification and possible functions of hair pencil scent of the yellow peach moth Conogethes punctiferalis in Japan. Appl Entomol Zool 34(1):147–153
Kondo A, Nagata K, Mochizuki (2008) Geographical difference in pheromone trap performance of the yellow peach moth Conogethes punctiferalis, occurring in Japanese peach orchards. Jpn Appl Entomol Zool Chugoku Branch 50:35–38
Konno Y, Honda H, Matsumoto Y (1980) Observation on the mating behavior and biology for the sex pheromones of yellow peach moth Conogethes punctiferalis. Appl Entomol Zool 15(3):321–327
Konno Y, Honda N, Matsumoto Y (1981) Mechanism of reproductive isolation between the fruit feeding and pin ace feeding type of yellow peach moth, Dichocrosis punctiferalis. Jpn J Appl Entomol Zool 25(4):253–258
Koya KMA, Devasahayam S, Kumar TP (1991) Insect pests of ginger and turmeric in India. J Plant Crop 19(1):1–3
Krishnamurthy KK, Avdani KK, Venkatesh J, Siddaramaih AL, Chakravathy AK, Gurumurthy BR (1989) Three decades of cardamom research, Regional Research Station, Mudigere (1958–1988). Station Technical Bulletin, pp 44–48
Kumaresan D (1982) Efficacy of modern synthetic insecticides for control of cardamom pests. Appl Entomol Zool 50(2):183–189
Kumaresan D (1983) Field evaluation of insecticides for control of cardamom pests. South-Indian Horticulture 2-3151-152
Li DY, Ali PP, Du YL, Sun SL, Zang MZ (2015) Effect of different host plants on the development and reproduction of yellow peach moth Conogethes punctiferalis. Aust Entomol 54(2):149–153
Liu MY, Tian Y, Li XY (1994) Identification of minor components of the sex pheromones of yellow peach moth Conogethes punctiferalis. Entomol Sin 1(2):150–155
Luo H, Honda H (2015) Olfactory and bio physical assessment of the oviposition stimulating potential of the host and non-host plants for the yellow peach moth Conogethes punctiferalis. Appl Entomol Zool 50(2):183–189
Mandal SC, Ghosh SN, Sinha RP (1978) Chemiocal control of castor capsule borer Conogethes punctiferalis. Indian J Entomol 40(4):460–462
Manikandan R, Muthukumar C, Ramalaxmi A, Balasubramani V, Udayasuriyan (2016) Screening of new isolates of Bacillus thuringiensis for cry1genes and testing of toxicity against Conogethes punctiferalis. Microbiol (Moscow) 85(2):191–197
Mori K, Watanable H, Fugiwahara M, Kuwahara S (1990) (E)-and(Z)-tetradecenyl formate, potent sex pheromone mimics against yellow peach moth. Pheromone Synth 122:1257–1259
Nari C, Kunikrishan, Zacharia PK, George KV, Nair MRGK, Kunhikrishan Nair C (1979) Field evaluation of newer insecticides for control of cardamom pests. Pesticides 13(8):49–50
Patel RK, Gangrade GA (1971) Note on the biology of castor capsule borer Conogethes punctiferalis. Indian J Agric Sci 41(5):443–444
Pervez R, Eapan SJ, Devasahayam S, Jacob TK (2014) Natural occurrence of Entomopathogenic nematodes associated with ginger eco-systems in India. Indian J Nematol 44(2):238–246
Rajabhaskar D, Regupathy R (2013) Plantation Journal of Biological Sciences, pp 1–8. ISSN:1028-8880. https://doi.org/10.3023/pjbs.2013
Rajagopal D, Chakravarthy AK, Gangappa E (1987) Role of biotic and abiotic factors in regulating the pests of cardamom. J Coffee Res 7(1):103–104
Renuka S, Regupathy A (2008) Acute and Selective toxicity of Profenofos against the shoot and capsule borer of the small cardamom. Resistant Pest Manag Newsl 17(2):33–35
Robinson GS, Tuck KR, Shaffer M (1994) A field guide to the smaller moths of South-East Asia. Malayan Nature Society and the Natural History Museum. Art Printing Works Sdn. Dhd., Kualalumpur
Sarkar PK, Roy D, Chakraraborthy G (2016) Bio-effectiveness and non-target toxicity of an IPM compatible thiourea compound diafenthiuron against cardamom pests under hill zone of the West Bengal 40(2):177–185
Sekiguchi K (1974) Morphology, biology and control of yellow peach moth Conogethes punctiferalis. Bulletin of the Ibaraki Horticultural Experiment Station, Special Issue, p 89
Sharma YR, Devasayam S, Anandaraj M (1992) Black pepper and cardamom: problems and prospects. In: Proceedings of national seminar on black pepper and cardamom, 17–18 May 1992, Calicut, Kerala
Sharma ML, Shukla A, Reddy RK (1995) Field testing of castor cultivars for field yield potential and resistance against shoot and fruit borer Conogethes punctiferalis. Crop Res Hissar 10(1):54–58
Shashank PR, Chakravarthy AK, Chandrashekaraiah R, Banu KRM (2014) Behavioural studies on shoot and fruit borer Conogethes punctiferalis host associated populations reveal occurrence of cryptic species. Entomol Generalis 35(2):103–115
Shashank PR, Kammar V, Mally R, Chakravarthy AK (2018) A new Indian species of shoot and capsule borer of the genus Conogethes (Lepidoptera: Crambidae), feeding on cardamom. Zootaxa 4374(2):215–234
Shetty GR, Hanchinamani V, Venkatesha J (2015) Correlation coefficients and occurrence of pests and diseases in turmeric cultivars under hill zone of Karnataka. J Environ Ecol 33(3):1201–1204
Singh SS, Rai AB, Mayank K, Kamal S (2009) Status, constraints and strategies of pest management in vegetable crops. Progress Hortic 41(1):46–53
Slon WJS (1945) Insect pests of grain sorghum. Queens Land Agric J 61(4):221–239
Solis MA (1999) Key to selected pyraloidea larvae intercepted at US ports of entry: revision of pyraloidea in keys to some frequently intercepted lepidopterous larvae by Weisman 1986. Proc Entomol Soc Wash 101(3):645–666
Sridharan S, Nagarajn N, Thamuraj S, Mohidin MK (1990) Effect of plating density on the capsule dame by cardamom major pests. South Indian Hortic 38(2):120–121
Sumitomo Chemical Co Ltd (2012) Development of the Novel Insecticide Spinetoram (DIANA®). A report
Thyagaraj NE (2003) Integrated management of some important cardamom pests in hill region of Karnataka, South India. Ph.D. thesis submitted to Dr. B.R. Ambedkar University, Agra, UP, p 226
Thyagaraj NE, Singh PK, Chakravarthy AK (2002) Effect of plat based insecticides on cardamom major pests infestation. Insect Environ 7(4):179–180
Tomomatsu S, Sekaguchi T, Ogino T, Misumi T, Kawakami F (1995) Methyl bromide fumigation for quarantine control of persimmon fruit moth and yellow peach moth on Japanese Persimon. Res Bull Plant Prot Serv 31:67–73
USDA (1957) Insects not known to in the United States. Yellow peach moth Conogethes punctiferalis, pp 37–38
Varadarasan S (2001) Insect pest management in cardamom –key to reduce cost of production. Spice India 14(7):19–22
Walsh BJ (1867) The apple worm and maggot. J Hortic 2:338–343
Wan NF, Zhang Y, Huang KH, Ji XY, Jiang JX (2016) Ecological engineering of trap cropping promotes biocontrol services in peach orchard ecosystems. Ecol Eng 90:427–430
Wang J, Zhang T-T, Wang Z-Y, He K-L, Liu Y, Jing L (2013) Molecular taxonomy of Conogethes punctiferalis, and Conogethes pinicolalis (Lepidoptera: Crambidae) based on mitochondrial DNA sequences. J Integr Agric 13(9). https://doi.org/10.1016/S2095-3119(3)60678-4
Wilson KI, Josseph D, Rehim MA, Nair MRGK (1978) Use of newer insecticides for control of cardamom pests. Agric Res J Kerala 15(2):192–194
Acknowledgement
Authors are thankful to the authorities of the University of Agricultural Sciences, GKVK, Bengaluru, Dr BR Ambedkar University, Agra and Dow Agri-Sci, Bombay, for the encouragement and the material used in this chapter.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Thyagaraj, N.E., Jagadish, K.S., Kumar, N. (2018). Novel Tools for the Management of Conogethes punctiferalis Guenée (Crambidae: Lepidoptera). In: Chakravarthy, A. (eds) The Black spotted, Yellow Borer, Conogethes punctiferalis Guenée and Allied Species. Springer, Singapore. https://doi.org/10.1007/978-981-13-0390-6_22
Download citation
DOI: https://doi.org/10.1007/978-981-13-0390-6_22
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-13-0389-0
Online ISBN: 978-981-13-0390-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)