Abstract
Studies in Xenopus laevis have greatly contributed to understanding the roles that the Zic family of zinc finger transcription factors play as essential drivers of early development. Explant systems that are not readily available in other organisms give Xenopus embryos a unique place in these studies, facilitated by the recent sequencing of the Xenopus laevis genome. A number of upstream regulators of zic gene expression have been identified, such as inhibition of BMP signaling, as well as calcium, FGF, and canonical Wnt signaling. Screens using induced ectodermal explants have identified genes that are direct targets of Zic proteins during early neural development and neural crest specification. These direct targets include Xfeb (also called glipr2; hindbrain development), aqp3b (dorsal marginal zone in gastrula embryos and neural folds), snail family members (premigratory neural crest), genes that play roles in retinoic acid signaling, noncanonical Wnt signaling, and mesoderm development, in addition to a variety of genes some with and many without known roles during neural or neural crest development. Functional experiments in Xenopus embryos demonstrated the involvement of Zic family members in left-right determination, early neural patterning, formation of the midbrain-hindbrain boundary, and neural crest specification. The role of zic genes in cell proliferation vs. differentiation remains unclear, and the activities of Zic factors as inhibitors or activators of canonical Wnt signaling may be dependent on developmental context. Overall, Xenopus has contributed much to our understanding of how Zic transcriptional activities shape the development of the embryo and contribute to disease.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
The Zic family of zinc finger proteins plays multiple roles during early development. In this chapter, we will examine how studies with Xenopus laevis embryos have contributed to our understanding of zic genes and their activities. Although complete gene knockout in early developmental stages of Xenopus is difficult, partly because maternal mRNAs can persist past MBT (Blum et al. 2015), gene expression levels can easily be altered in Xenopus embryos using morpholino oligonucleotides or injection of mRNAs. Further, Xenopus embryos readily lend themselves to physical manipulation. Therefore, studies in Xenopus laevis have contributed much to our understanding of the functional roles of zic genes during neural induction, early neural patterning, and formation of the neural crest. In addition, microarray screens have identified a number of direct targets of Zic proteins, prompting a number of new and ongoing studies. Due to years of study, a large body of knowledge has been amassed on Xenopus embryo development, gene regulation, and cell fate mapping, which helps put the roles of zic genes into context.
1.1 Experimental Approaches Unique to Xenopus
1.1.1 Ectodermal Explants (Animal Caps)
Ectodermal explants (animal caps) allow researchers to study gene expression in cells that are competent to respond to neural induction. At the same time, these explants allow the study of gene regulation free from the variety of inductive signals that characterize gastrulation and neural induction. For animal cap experiments, two-cell embryos are typically injected with mRNAs or other molecules into the animal hemisphere of both blastomeres. After maturing to late blastula (stage 9), ectodermal explants are harvested from the animal hemisphere of the embryos. The explants form characteristic balls, which can be aged to gastrula and neurula stages (using intact sibling embryos for staging), at which point they are processed in assays to determine gene expression (Sive et al. 2007). Ectodermal explants have been used extensively to identify gene regulatory relationships between zic and other genes, which can then be tested in whole embryos. In addition, ectodermal explants make Xenopus embryos uniquely suited to identify or confirm genes that are direct targets of transcription factors active during early development. A hormone-inducible transcription factor is constructed by fusing the glucocorticoid receptor domain (hGR) to the transcription factor, and mRNA for this inducible construct is injected into the embryos. The hGR domain forms a complex with endogenous HSP90, thus retaining the transcription factor in the cytoplasm (Kolm and Sive 1995; Mattioni et al. 1994). Treatment with the hormone dexamethasone allows the hGR-bound transcription factor to detach and enter the nucleus. In order to identify direct transcriptional targets, the hormone-inducible transcription factor is activated in the presence of protein synthesis inhibitors. More detail is provided below in the description of two screens for direct targets of Zic1 (Cornish et al. 2009; Plouhinec et al. 2014).
1.1.2 Keller Explants
Keller open-faced explants are derived from the dorsal marginal zone of early gastrula embryos (Keller and Danilchik 1988). They comprise prospective mesoderm and ectoderm and allow powerful studies of the genes involved in regulating convergent extension movements (Keller et al. 1992). With regard to zic genes, this system is being used to study the role of aqp3b, a direct target of Zic1, in convergent extension (See and Merzdorf unpublished). Keller explants have also been used to study neural induction free from vertical signals, since the signals that pass from the mesoderm to the ectoderm portion of the explant are limited to planar signals. This system demonstrated that calcium transients are required for induction of zic3 expression (Leclerc et al. 2003). With the identification of direct targets of Zic1 that play roles in noncanonical Wnt signaling (Cornish et al. 2009), Keller explants may help understand the roles that these genes play in convergent extension.
2 Zic Family Genes and Their Expression in Xenopus Embryos
2.1 Comparison of Zic Genes in the Allotetraploid Genome of Xenopus laevis
The genomes of both Xenopus species are nearly complete (Hellsten et al. 2010; Session et al. 2016). Xenopus zic genes show the same chromosomal arrangement as Zic genes in mouse and humans, with zic genes clustered on the same chromosome in a head-to-head orientation: zic1 with zic4, zic2 with zic5, and zic3 on a different chromosome (Grinberg and Millen 2005; Aruga et al. 2006). In addition to the zinc finger (ZF) DNA-binding domain, the Zic-Opa (ZOC) and zinc finger-nucleocapsid (ZF-NC) domains (both N-terminal to the zinc fingers) are conserved between Xenopus and mammalian zic genes (ZOC is present only in zic1-3) (Houtmeyers et al. 2013).
Xenopus laevis and Xenopus tropicalis both have five zic genes, but due to the allotetraploid nature of X. laevis, its genome possesses two versions of each zic gene, one on either a longer or shorter chromosome. The zic genes are therefore named zic.S and zic.L. Table 7.1 shows the results of comparing nucleotide sequences of S and L zic gene-coding regions and the amino acid sequences of S and L Zic proteins. The S and L variants were also compared to the X. tropicalis versions of each Zic protein (Table 7.1) (Ricker et al. unpublished). The amino acid sequence identities indicate that the S and L versions of X. laevis Zic proteins are about equally divergent from each other as they are from the Zic proteins in X. tropicalis. The differences include substitutions and small gaps spanning up to three consecutive amino acids. Outside of the coding region, in the 5′UTR and 3′UTR, the sequences are more divergent between the S and L versions. The untranslated regions of the X. tropicalis zic genes are significantly different from the UTRs of the X. laevis zic genes.
The presence of two versions for each zic gene in X. laevis allowed each gene to diverge and possibly even perform different functions. For example, the Zic1 direct target gene aqp3b is the L version of the X. laevis aqp3 gene. It is expressed at gastrula and neurula stages, while aqp3a, the S version of the gene, is not expressed during early development (Cornish et al. 2009). In adult frogs, the tissues that express the two aqp3 genes vary, although the composite of the expression patterns is similar to the overall expression pattern of the single Aqp3 gene in mice (Cornish et al. 2009; King et al. 2004). Thus, the individual roles of the S and L copies of each zic gene may vary but, taken together, may perform similar functions as a single copy in other species. Finally, while X. tropicalis is a useful model, to date it has not been used to study the roles of zic genes.
2.2 Expression of Zic Genes in Xenopus Embryos
The gene expression patterns of zic genes in Xenopus embryos overlap extensively (Fujimi et al. 2006, 2012), which is also the case for zic genes in other vertebrates, for example, mouse and chick embryos (Nagai et al. 1997; Furushima et al. 2000; Gaston-Massuet et al. 2005; McMahon and Merzdorf 2010). Despite the overlap, there are significant differences in the expression domains of zic genes.
2.2.1 Blastula Embryos
The zic2 gene is the only maternally expressed zic gene in Xenopus embryos (Nakata et al. 1998). The expression of zic1, zic3, and zic4 begins at stage 9, after midblastula transition, although the expression of zic4 is initially very low (Fig. 7.1a). The zic5 gene is not expressed in blastula embryos. In situ hybridization shows that in late blastula embryos (stage 9.5), zic1, zic2, and zic3 are expressed in the dorsal marginal zone in both ectoderm and mesoderm (Fig. 7.1b). There does not appear to be significant expression of these zic genes in the roof of the blastocoel.
Expression of zic genes in Xenopus embryos. (a) Expression of zic1-5 determined by RT-PCR in unfertilized eggs (e) and Xenopus embryos at different developmental stages, including pre-MBT blastula (stage 6), post-MBT blastula (stage 9), late gastrula (stage 12), mid-neurula (stage 15), and tailbud stages (stage 19 and older). zic2 is expressed both maternally and throughout early development. zic1 and zic3 are first detected at stage 9. The expression of zic3 peaks in late gastrula/early neurula, while zic1 expression remains strong. Weak expression of zic4 is first detected at stage 9 and continues until tailbud (stage 22 and later stages), when it is more strongly expressed. Weak expression of zic5 is detected by late gastrula (stage 12), and it is strongly expressed in neurula stages and beyond. (b) Expression of zic1-3 by in situ hybridization in whole embryos. Dorsal is to the right. zic2 mRNAs are more extensively present at stage 9.5, likely due to residual maternal mRNA. During late blastula (stage 9.5) and throughout gastrula (stages 10.5-12), the zic1-3 genes are expressed in the dorsal ectoderm and in the involuting mesoderm. zic3 is also expressed in the ventral and lateral involuting mesoderm (Fujimi et al. 2012; Kitaguchi et al. 2000). During neurula (stage 14) zic1-3 are expressed in the neural plate and to some extent in the notochord (Reproduced from Fujimi et al. 2012 with permission of the publisher)
2.2.2 Gastrula Embryos
As gastrulation begins, the zic1-3 genes are strongly expressed in the prospective neural ectoderm and moderately expressed in the mesoderm (stages 10.5 and 11; Fig. 7.1b). zic4 expression is quite low, and zic5 expression begins in late gastrula embryos (Figs 7.1a and 7.2). In late gastrula embryos (stage 11.5), zic1, zic2, zic3, and zic5 are expressed to varying degrees in a broad region of the prospective neural ectoderm (Fig. 7.2), while zic4 expression is extremely weak. These expression patterns are consistent with the significant roles that the zic1-3 genes play during early stages of development and show that zic genes are among the earliest genes expressed in response to neural induction.
Expression of zic genes in gastrula, neurula, and tailbud Xenopus embryos. The expression of the zic1-5 genes was determined by in situ hybridization in whole embryos. During gastrulation (a: stage 11.5), all zic genes with the exception of zic4 are expressed in the presumptive neural plate. During neurula (stages 15 and 19) and tailbud (stages 23 and later stages), all zic genes show expression in the dorsal neural tube. Other tissues also show zic gene expression, including the hyoid and branchial crest (black arrow), eye (red arrowheads), somites (green arrowheads), lateral mesoderm (blue arrowheads), and olfactory placode (white arrowheads) (Reproduced from Fujimi et al. 2006 with permission of the publisher)
2.2.3 Neurula Embryos
During neurula stages, all five zic genes are expressed in the neural plate border. Only zic2 and zic3 are expressed within the neural plate. zic3 is found in the midbrain-hindbrain region, and zic2 is expressed at the midline of the neural plate (Fujimi et al. 2006) and in the progenitor cells located between the stripes of primary neurons (Brewster et al. 1998). At the neural plate border, zic1-3 are strongly expressed in wide regions, while zic4 and zic5 are more restricted to the regions of the neural folds (Fig. 7.2b). zic1-3 and zic5 are expressed strongly in anterior regions. After closure of the neural tube, all zic family members continue to be expressed in the dorsal neural tube (Fig. 7.2c). Thus, zic genes are expressed in areas required for neural patterning, neural crest specification, and neural tube closure.
2.2.4 Tailbud Stage Embryos
During tailbud and later stages, zic1 is strongly and zic4 is weakly expressed in the dorsal neural tube along the entire embryo (Fig. 7.2A, D d–g). zic2, zic3, and zic5 are expressed more strongly in the anterior and posterior regions of the dorsal neural tube (Fig. 7.2B, C, E d–g). In tailbud stage embryos, zic1, zic2, and zic5 show some expression in the region of the eye, both zic1 and zic4 are expressed in the somites (Nakata et al. 2000), and zic3 is uniquely expressed in caudal lateral plate mesoderm (Fig. 7.2). Thus, zic gene expression patterns overlap extensively but also show unique aspects. Some of these correlate to known differences in zic gene function, although many of these differences in expression are not yet understood.
3 Upstream Regulators of Zic Gene Expression
Xenopus zic genes are expressed extensively during early development (Figs. 7.1 and 7.2), and a number of mechanisms are known to regulate zic gene expression.
3.1 Inhibition of BMP Signaling
The inhibition of bone morphogenetic protein (BMP) signaling is critical for neural induction (Sasai et al. 1996; Sasai and De Robertis 1997) and plays an early role in regulating zic gene expression. Signaling by BMP specifies ventral, non-neural fates and represses neural genes in Xenopus and other vertebrates. Thus, BMP signaling represses the expression of the zic1, zic2, and zic3 genes (Gamse and Sive 2001; Nakata et al. 1997). Conversely, the inhibition of BMP signaling, often mediated by Noggin and Chordin, is essential for dorsal determination and specification of neural fate. Accordingly, misexpression of noggin (Mizuseki et al. 1998; Gamse and Sive 2001) or FRL-1 (Yabe et al. 2003), which also represses BMP signaling, results in an increase in zic gene expression. Indeed, the promoter of zic1 contains a 215 bp BMP inhibitory response module (BIRM) (−2.7 to −2.5 kb 5′ to the transcription start site). The BIRM is required for transcription of the zic1 gene in the absence of BMP signaling in animal cap-based reporter assays (Tropepe et al. 2006). The BIRM contains consensus binding sites for several transcription factors, including one Smad binding site and binding sites for the Ets, Oct, Lef/Tcf, and Sox transcription factors. Mutations in most of these putative transcription factor binding sites eliminate the ability of the BIRM to respond to Noggin, suggesting that multiple signals must cooperate to mediate zic1 transcription in response to BMP inhibition. A dominant interfering Smad is able to induce expression of zic1 in the absence of translation (Marchal et al. 2009), indicating direct regulation. However, mutation of the putative Smad binding site within the BIRM does not activate reporter gene expression (Tropepe et al. 2006). Thus, the inhibition of BMP signaling is required for zic gene expression, but which region of the zic gene directly responds to lack of BMP signaling, or exactly how the BIRM is responsive to suppression of BMP signaling remains to be answered.
3.2 Siamois and Twin
Organizer-specific transcription factors, such as Siamois and Twin, are responsible for the expression of BMP antagonists, including noggin and chordin. These BMP antagonists are secreted from the organizer (dorsal mesoderm) and block BMP in the neural ectoderm, which results in the upregulation of zic genes, as described above. Accordingly, zic genes are expressed in a wide domain in the neural ectoderm during gastrula stages (Fig. 7.2). Klein and Moody (2015) examined whether the expression of neural genes could be induced directly by organizer transcription factors, in addition to the indirect induction by BMP inhibitors. They found that in late blastula embryos, ectopic expression of the organizer genes siamois and twin induced ectopic zic2 expression directly, in the absence of translation. Later in development, as gastrulation begins, zic2 is present in the involuting dorsal mesoderm at moderate levels, with stronger expression in the neural ectoderm (Fujimi et al. 2012), while Siamois and Twin are limited to the dorsal mesoderm. This lack of overlap suggests that zic2 expression in neural ectoderm is now regulated indirectly through induction of BMP inhibitors by Siamois and Twin. The significance for this bimodal regulation needs to be explored further. However, the direct induction of zic2 by Siamois and Twin may serve to bias the dorsal region of the late blastula/early gastrula toward neural induction, and further studies demonstrated that maternal zic2 is able to exert this bias as well (Gaur et al. 2016). This is supported by the finding that Zic1 is able to sensitize the future neural ectoderm for neural induction (Kuo et al. 1998).
3.3 Calcium Signaling
Calcium signaling helps mediate the activity of BMP inhibitors during neural induction. Noggin causes an increase in calcium transients in the prospective neural ectoderm, and experimentally increasing calcium demonstrated that it is a potent inducer of early neural genes and a repressor of epidermal genes (Moreau et al. 2008; Leclerc et al. 2006). Thus, calcium signaling is required for neural ectoderm formation and is required for zic3 gene expression. Blocking L-type calcium channels with specific antagonists in gastrula stage embryos and in Keller open-faced explants results in a reduction of zic3 expression (Leclerc et al. 2000, 2003). The xPRMT1b gene, which codes for an arginine methyltransferase, is upregulated by Noggin in a calcium-dependent manner, and xPRMT1b can induce the expression of zic3 (Batut et al. 2005). Thus, xPRMT1b appears to be a link between early calcium transients resulting from BMP inhibition and the expression of neural genes during neural induction, including zic genes.
3.4 FGF Signaling
FGF signaling is required for neural ectoderm formation in a variety of vertebrates (Patthey and Gunhaga 2014; Aruga and Mikoshiba 2011). In Xenopus embryos, FGF signaling in conjunction with Noggin activates zic1 gene expression in ectodermal explants (Gamse and Sive 2001). In embryos with blocked BMP signaling, inhibition of FGF signaling only slightly reduced the induction of zic1 expression but completely abolished the induction of zic3 expression (Marchal et al. 2009). This suggests that FGF signaling increases the expression of zic1 from the level established by BMP inhibition, while both FGF signaling and BMP inhibition are required for zic3 gene expression. Further, zic3, but not zic1, is upregulated by FGF in the presence of cycloheximide, suggesting a direct mechanism for zic3 expression. Conversely, zic1, but not zic3, expression is activated by Noggin in the presence of cycloheximide (Marchal et al. 2009). This suggests different regulatory mechanisms for the induction of these two zic genes, and it shows an important involvement for FGF signaling in their regulation.
3.5 Wnt Signaling
Wnt signaling contributes to early patterning of the neural ectoderm and promotes the expression of zic genes. During early anterior to posterior patterning in Xenopus embryos, wnt expression (in conjunction with Noggin) activates zic1 expression in the posterior portion of the presumptive neural plate (Gamse and Sive 2001). Consistent with this finding, the BIRM regulatory element upstream of zic1 contains a Lef/Tcf binding site. Mutation of this Lef/Tcf site eliminates the ability of the BIRM to respond to Noggin in reporter assays (Tropepe et al. 2006), supporting a requirement for Wnt signaling in zic gene regulation in posterior regions of Xenopus embryos.
3.6 FoxD4 and Other Factors
A regulator of zic gene expression in the early neural ectoderm is the forkhead transcription factor FoxD4 (also called FoxD5), which is expressed in tissue destined to become the neural ectoderm (Yan et al. 2009). During early Xenopus neural ectoderm formation, inhibiting foxD4 expression causes a reduction of zic2 expression but expands the expression domains of both zic1 and zic3. Testing an activator construct of FoxD4 (FoxD4 fused to the VP16-activating domain) and a repressor construct of FoxD4 (FoxD4 fused to the EnR repressor domain) in whole embryos showed that FoxD4 acts as activator to induce zic2 expression but as repressor to repress zic1 and zic3 expression (Yan et al. 2009). The acidic blob region in the N-terminal domain of FoxD proteins is required for induction activity, while interaction with a co-repressor at a site in the C-terminal domain is required for repressive activity (Pohl and Knochel 2005). Structure-function experiments indicate that the activating function of FoxD4 is a direct process, while its ability to inhibit genes requires intermediate factors. Further studies showed that the upregulation of zic1 and zic3 expression as a result of inhibiting foxD4 expression can be rescued by zic2 mRNA injections (Neilson et al. 2012). Thus, direct induction of zic2 may contribute to the inhibition of zic1 and zic3 expression during the formation of the neural plate. Interestingly, this interaction is different in gastrula embryos, when FoxD4 has an activating effect on zic1 and zic3 expression (Yan et al. 2009). Thus, zic genes are regulated differently at different times during development, and individual zic genes are regulated by independent mechanisms.
Following the broad induction of zic gene expression by the inhibition of BMP signaling in conjunction with FGF and Wnt signaling, other factors help refine and limit the expression pattern of zic genes. A 5 kb region upstream from the transcription start site of zic1 (a region containing the BIRM) encompasses additional binding elements that restrict zic1 expression, since loss of this region caused an expansion of zic1 expression (Tropepe et al. 2006). Candidate transcriptional repressors that limit the expression of zic genes are the Msx1 and Dlx1 transcription factors. Both are direct targets of intermediate levels of BMP signaling and are expressed in the epidermal-neural boundary region. Both repress zic gene expression in ectodermal explants, and Dlx1 was shown to repress zic3 expression in Xenopus embryos (Tribulo et al. 2003; Feledy et al. 1999; Yamamoto et al. 2000; Monsoro-Burq et al. 2005). Thus, blocking BMP signaling creates a permissive environment for zic gene expression in the presumptive neural plate, while Dlx3 and Msx1 may prevent the expression of zic genes beyond the neural plate border region.
The TALE-family homeodomain proteins Pbx1 and Meis1 are important in early neural patterning, and their misexpression causes an increase in zic3 expression (Maeda et al. 2001, 2002; Kelly et al. 2006). Further analysis showed that Pbx1 and Meis1 synergistically interact with a 3.1 kb region directly upstream of the zic3 transcription start site (Kelly et al. 2006). In the anterior portion of the neural plate, the Six1, Six3, and Xrx1 transcription factors may promote expression of zic genes, since these transcription factors increase the transcription of zic2 (Brugmann et al. 2004; Gestri et al. 2005; Andreazzoli et al. 2003). These transcription factors help refine zic gene expression patterns.
In summary, zic genes are expressed in the prospective neural ectoderm during gastrula stages and are among the first genes expressed in the early neural plate. Studies in Xenopus have greatly contributed to our understanding of the mechanisms that regulate zic gene expression. Inhibition of BMP, the resulting calcium transients, in conjunction with FGF and Wnt signaling are responsible for early zic gene expression. Nodal also counts among the upstream regulators of zic gene expression, which has mostly been explored in mouse (Houtmeyers et al. 2016). After initial induction of zic genes, their expression patterns are limited and refined by a number of other transcription factors and signaling mechanisms. Among these factors is expression of shh in the ventral neural tube, which represses zic gene transcription and therefore limits zic expression to the dorsal neural tube (Aruga et al. 2002). Overall, the mechanisms that are responsible for regulating zic genes individually at different times during development remain to be explored in greater detail.
4 Direct Transcriptional Targets of Zic Proteins
The DNA-binding domain of Zic transcription factors consists of five C2H2 zinc fingers. While the three-dimensional structure of this domain has not been determined for any of the Zic proteins, the significant similarity between the zinc fingers in Zic and Gli proteins allows the assumption that zinc fingers 2–5 interact with the major groove of the target gene, while zinc finger 1 engages in protein-protein interactions (Pavletich and Pabo 1993). Interestingly, Zic proteins have not been reported to act as homodimers (Brown et al. 2005).
4.1 Screens for Zic1 Direct Targets in Xenopus
Zic proteins are involved in the downstream regulation of a wide variety of genes. In Xenopus, two screens were conducted for direct target genes that are relevant during early neural development (Cornish et al. 2009) and during neural crest specification (Plouhinec et al. 2014). The unique ability to use ectodermal explants from Xenopus embryos makes the identification of direct targets more readily feasible than in other organisms. In these screens, an inducible zic construct (zic1GR) was used. Zic1GR is a fusion of Zic1 to the ligand-binding domain of the human glucocorticoid receptor, which renders Zic1GR inducible with dexamethasone (Kuo et al. 1998). In order to identify direct targets of Zic1, animal caps injected with zic1GR are aged to the desired stage and then first treated with cycloheximide to prevent protein synthesis, followed by treatment with dexamethasone to activate Zic1GR (Fig. 7.3). The animal caps are harvested and assayed for the transcription of new mRNAs, which are direct targets of Zic1. The Cornish et al. (2009) screen aimed to identify early neural genes. Therefore, the animal caps were neuralized with a low dose of co-injected noggin mRNA. Plouhinec et al. (2014) set out to identify neural crest specifiers. Therefore, the animal caps were co-injected with hormone-inducible zic1 and pax3. Both screens identified a number of genes, which are summarized in Table 7.2. Although both screens used a zic1GR construct to induce transcription of direct targets of Zic1, it is likely that the identified genes include direct targets of other Zic proteins, since the zinc finger domains of the Zic1-3 proteins are highly similar (Fujimi et al. 2006).
Experimental design for microarray screens to identify direct transcriptional targets of Zic1. Embryos were injected at the two-cell stage into both cells with mRNAs for either zic1GR/noggin to induce early neural genes or zic1GR/pax3GR to induce neural crest genes. Control embryos were injected with mRNAs for noggin only or pax3GR only, respectively (Cornish et al. 2009; Plouhinec et al. 2014). Animal caps were dissected at stage 9. At the desired age, the isolated animal caps were treated first with cycloheximide (CHX) to prevent protein synthesis and later with dexamethasone (DEX) to induce the GR-conjugated transcription factors. The caps were then cultured to the correct stage and RNA isolated for microarray analysis and RT-PCR
4.2 Direct Targets of Zic1 During Early Neurula Stages
A large number of genes were identified in the screen for direct targets of Zic1 during neural plate development (Cornish et al. 2009). The genes included in Table 7.2 are limited to direct targets that were confirmed by RT-PCR, and many were additionally shown to be regulated by Zic1 in whole embryos by in situ hybridization (Fig. 7.4). The screen was conducted at the equivalent of early neurula stages, and most of these genes are expressed in parts of the neural plate or in the neural plate border, overlapping with the expression patterns of Zic1 (Fig. 7.2).
Zic1 regulates the expression of direct target genes in neurula embryos (Cornish et al. 2009). Gene names are listed along the left with the original names or identifiers in parentheses. Shown are in situ hybridization expression patterns for neurula (stage 15–18) embryos that were uninjected (first column), injected with the dominant interfering construct zic1ΔN (dnzic1; second column), or injected with zic1 mRNA (third column). Interfering with zic1 activity reduced the expression levels of all direct target genes shown, indicating that Zic1 is required for their expression. Misexpressing zic1 resulted in expansion of aqp-3b, ptgds, and CRABP-2 expression. Arrowheads mark the injected sides (Reproduced from Cornish et al. 2009 with permission from publisher)
4.2.1 Xfeb (Glipr2)
Among the direct target genes of Zic1, the putative metalloprotease Xfeb (Glipr2) was identified in both screens (Cornish et al. 2009; Plouhinec et al. 2014) and in an earlier spotted array (Li et al. 2006). It is expressed in the hindbrain and represses the expression of both the hindbrain gene hoxB1 and the otx2 gene, which is expressed anterior to the midbrain-hindbrain boundary (Li et al. 2006). This suggests that Xfeb contributes to patterning the neural plate and may be part of the regulatory mechanism that prevents expression of the otx2 gene posterior to the midbrain-hindbrain boundary (Fig. 7.4). The identification of Xefb (Glipr2) in the Plouhinec et al. (2014) screen suggests that it also plays a role during neural crest specification. Xfeb and gbx2 are both expressed in the hindbrain (Li et al. 2006; Rhinn and Brand 2001), and Gbx2 has neural crest specifier activity, which is dependent on the presence of Zic1 activity in ectodermal explants (Li et al. 2009). Thus, the induction of Xfeb by Zic1 may be required for the neural crest induction activity by Gbx2.
4.2.2 aqp3b
The aqp3b gene codes for an aquaporin, specifically an aquaglyceroporin. Aquaporins are channel proteins that allow passage of water and other small molecules (like glycerol) across cell membranes along their concentration gradients (Verkman 2005). In Xenopus neurula embryos, aqp3b is expressed in cells at the tips of the rising neural folds during neural tube closure (Fig. 7.4; Cornish et al. 2009). These cells, called “IS” cells (Schroeder 1970), separate the epidermal ectoderm and the neural ectoderm. During neurulation, the cells of the neural plate apically constrict, which allows the neural folds to rise and the neural tube to close (reviewed in Wallingford 2005). Compromising aqp3b expression in Xenopus embryos results in loss of apical constriction in neural plate cells and defective neural tube closure (Forecki and Merzdorf unpublished). Neural tube closure defects have been observed with mutations in human or mouse zic2, zic3, and zic5 genes (Grinberg and Millen 2005). Thus, Aqp3b may be part of the mechanism that allows zic genes to control neural tube closure.
In gastrula embryos, aqp3b is expressed in the marginal zones and in the sensorial layer of the blastocoel roof (Forecki et al. 2018). Thus, aqp3b expression overlaps with zic1-3 expression, which are expressed in the epithelial and sensorial layers of the dorsal marginal zone (Nakata et al. 1998; Fig. 7.1). Disrupting aqp3b expression in the dorsal marginal zone of whole embryos results in compromised border integrity between involuted mesendoderm and noninvoluted ectoderm and defective deposition of fibril fibronectin matrix at this boundary (Forecki et al. 2018). Further, inhibiting aqp3b expression in explants of the dorsal marginal zone region (Keller explants) interfered with their convergent extension, which was rescued with players in noncanonical Wnt signaling (See and Merzdorf unpublished). Although Zic proteins have not yet been examined for their roles in maintaining border integrities in gastrula embryos between involuted and noninvoluted cells, their expression patterns are consistent with this possibility. Further, involvement of Zic proteins in noncanonical Wnt signaling has not been demonstrated to date. However, identification in this screen of several genes that are involved in noncanonical Wnt signaling pathways suggests that Zic proteins may play such a role.
4.2.3 pkdcc2 and Prickle Act in Noncanonical Wnt Signaling
The pkdcc2 gene encodes a protein kinase, which regulates JNK-dependent Wnt/PCP signaling. It is important in both blastopore and neural tube closure (Vitorino et al. 2015). Prickle is a cytoplasmic protein that plays a key role in Wnt/PCP signaling as one of the six core components of Wnt/PCP signaling (reviewed in, e.g., Davey and Moens 2017). Accordingly, it is important for cell movements during Xenopus gastrulation and neural tube closure (Takeuchi et al. 2003). Thus, these direct targets strongly suggest a new role for Zic proteins as regulators of noncanonical Wnt signaling.
4.2.4 crabp2, ptgds, ncoa3, and cyp26cl Are Genes Related to Retinoic Acid Signaling
The expression domains of the crabp2 and ptgds (also called cpl-1 or lpgds) genes overlap with the zic1 expression domain (Fig. 7.4; Cornish et al. 2009). Both proteins function in regulating the cellular availability of retinoic acid during development. CRABP2 (cellular retinoic acid-binding protein 2) binds retinoic acid intracellularly and delivers it to the nucleus (Dong et al. 1999; Lepperdinger 2000). PTGDS acts dually as prostaglandin D2 synthase and as a lipocalin carrier for retinoic acid (Urade and Hayaishi 2000). Mutation analysis demonstrated that Zic1 acts only through the lipocalin function of PTGDS (Jaurena et al. 2015). The transcriptional coactivator Ncoa3 (also called SRC-3) activates the RAR/RXR nuclear receptor in response to retinoid binding in Xenopus (Kim et al. 1998). The direct target gene cyp26cl codes for a retinoic acid metabolizing enzyme, which is involved in anterior/posterior patterning of Xenopus embryos (Tanibe et al. 2008). Interestingly, in the pre-placodal ectoderm, Zic1 upregulates both the cyp26cl gene and the raldh2 gene, which codes for a retinoic acid-synthesizing enzyme, although raldh2 most likely is not a direct target of Zic1 (Jaurena et al. 2015). The authors hypothesize that retinoic acid synthesized by Raldh2 in zic1-expressing cells diffuses to and elicits signaling in surrounding cells, while the zic1-expressing cells themselves are not subject to signaling by the retinoic acid they produce due to the presence of Cyp26cl. Thus, a sharp boundary of retinoic acid-induced gene expression is created (Jaurena et al. 2015). Therefore, it appears that Zic1 regulates the expression of genes that control multiple aspects of retinoic acid signaling, which includes the synthesis and degradation of retinoic acid and aspects of its transport and availability.
4.2.5 VegT, Eomesodermin, and myoD Are Transcription Factors Important for Mesoderm Development
Eomesodermin acts very early in mesoderm development and regulates the expression of the t-box transcription factor VegT (Fukuda et al. 2010). VegT helps organize the paraxial mesoderm in Xenopus embryos (Fukuda et al. 2010). Experiments in chick embryos suggest that Zic1 may induce but not maintain myoD expression during somite development (Sun Rhodes and Merzdorf 2006). zic genes are known to play roles in mesoderm development, which have mostly been studied in other organisms.
4.2.6 Other direct targets of Zic1
Additional direct targets of Zic1 include celf3, sall1, and hesx1, which are associated with regulating gene expression in the developing nervous system. The celf3 gene (also called brunol1) is broadly and strongly expressed in the neural plate border region. It codes for an RNA-binding protein with roles in regulating splicing events in the nucleus (Wu et al. 2010). The sall1 transcription factor is expressed in the midbrain and in posterior regions of the neural plate (Hollemann et al. 1996). Sall1 is required for neural tube closure in mice (Böhm et al. 2008). The homeobox transcription factor Hesx1 is expressed in the anterior neural plate, where it promotes differentiation of the neural ectoderm and acts as a repressor of the xbf-1, otx2, and pax6 genes (Ermakova et al. 1999).
Interestingly, no genes were identified in this screen, which are directly related to cell cycle control, and the gene most related to cell proliferation or cell differentiation is the hesx1 gene, described above. Overall, the identified direct targets point to known and new activities for Zic transcription factors during early neural development.
4.3 Neural Crest-Specific Direct Targets of Zic1
The screen by Plouhinec et al. (2014) was a multi-step screen designed to limit identification of direct targets to only genes that act during neural crest specification. To this end, inducible zic1GR RNA was co-expressed with pax3GR RNA in animal caps (Fig. 7.3), and targets of Pax3GR alone were subtracted from the results. A variety of genes were identified, and those that were confirmed by an additional method are included in Table 7.2. Among these targets is the Xfeb gene (also called glipr2), which was identified in both screens and is discussed above.
The Plouhinec et al. (2014) screen identified the snail1 and snail2 (slug) genes as direct Zic1 targets, which are known to be expressed in the neural plate border region prior to neural crest migration. Snail1 has also been shown to induce snail2 and other neural crest markers, including zic5 and ets1 (Aybar et al. 2003). Further, there has been indication that Zic1 induces snail2 acting as a repressor, indicating an indirect regulatory mechanism (a zic1-EnR construct activated snail2 expression; Merzdorf unpublished). Thus, there may be more than one way in which Zic proteins can induce snail2 expression.
Additional genes identified by Plouhinec et al. (2014) include the ets1, dusp5, and pdgfra genes. The gene for the Ets1 transcription factor is expressed in Xenopus premigratory neural crest cells destined to become cardiac tissues and has functions similar to Snail proteins (Nie and Bronner 2015). Dusp5 is a MAP kinase phosphatase and an important regulator of MAPK signaling (Caunt and Keyse 2013). MAPK signaling is essential for neural crest induction (Stuhlmiller and Garcia-Castro 2012a). Pdgfra is a receptor tyrosine kinase for PDGF. It is important for directed migration of cells in Xenopus gastrula embryos (Van Stry et al. 2005). During Wnt-induced cell proliferation of osteoblasts, Pdgfra is activated in a disheveled-dependent manner (Caverzaiso et al. 2013).
The number of direct target genes for Zics identified in humans and mouse is relatively small. These targets include ApoE, Math1, αCaM kinase II, dopamine receptor 1, and Pax3 (Salero et al. 2001; Yang et al. 2000; Ebert et al. 2003; Sakurada et al. 2005; Sanchez-Ferras et al. 2014). A ChIP-seq screen for direct targets of zebrafish Zic3 has yielded a large number of regulatory regions that drive a variety of genes involved in early development (Winata et al. 2013).
4.4 Interaction with Other Proteins
Zic proteins are transcription factors that bind DNA using C2H2 zinc finger domains, as stated earlier. There is some evidence that, like most transcription factors, their activity is regulated by interacting proteins. Xenopus Gli proteins, which are also C2H2 zinc finger transcription factors, interact with Zic proteins. Zic1, Zic2, and Zic3 and the Gli1, Gli2, and Gli3 proteins interact physically (through zinc fingers 3–5 of both Zic and Gli proteins) (Koyabu et al. 2001). In these Zic/Gli heterodimers, zinc fingers 3–5 would be occupied by binding to each other, thus preventing DNA binding by either protein. Therefore, in cases of co-expression, Zic and Gli proteins may regulate each other’s activity as transcription factors. Indeed, in Xenopus embryos and in cell culture reporter assays, Zic and Gli proteins are able to reduce each others’ activities as transcriptional activators (Brewster et al. 1998; Koyabu et al. 2001; Mizugishi et al. 2001). Zic2 has also been shown to interact with TCF1 and, via its zinc fingers, with TCF4, thereby interfering with Wnt/β-catenin signaling (Fujimi et al. 2012; Pourebrahim et al. 2011). In other organisms, there are not many proteins known to interact with Zic proteins. A yeast two-hybrid screen identified Imfa as a direct binding partner of Zic1, Zic2, and Zic3 in mouse (Mizugishi et al. 2004). In order to understand the activities of Zic factors, it will be important to learn more about proteins that modulate Zic activity by direct protein-protein interactions.
5 Biological Roles of Zic Transcription Factors
The Xenopus model lends itself to functional studies of genes. Loss and gain of function experiments combine to illustrate the activities of Zic transcription factors during embryonic development, particularly during gastrulation and early neural development.
5.1 Role of Maternally Expressed Zic2
Among the Xenopus zic genes, only zic2 maternally expressed (Nakata et al. 1998). The role of maternally expressed zic2 was studied using the host transfer method, where maternal zic2 mRNA was depleted in oocytes that were then transferred back into Xenopus females for ovulation (Houston and Wylie 2005). After fertilization and during development, this depletion resulted in exogastrulation, anterior truncations, thickened notochord, and axial abnormalities due to an overall increase in Nodal signaling (Houston and Wylie 2005). Similarly, double zic2/zic3 morphants had a shortened body axis, smaller heads, and thicker, wider notochords (Fujimi et al. 2012). Thus, maternal expression of zic2 is essential for early patterning of the embryo.
5.2 Zic Genes During Gastrulation and Early Patterning of Xenopus Embryos
Zygotic expression of the zic1-4 genes begins shortly after midblastula transition, and all five zic genes are expressed during gastrulation, most strongly in the area of the presumptive neural plate (Fig. 7.1). As described above, the expression of zic genes appears to bias the ectoderm toward a neural fate in early embryos, since expression of zic1 in animal cap ectoderm (from late blastula embryos) amplifies the neural inducing effects of Noggin (Kuo et al. 1998). In addition, maternal zic2 and early zygotically expressed zic2, which is induced by the organizer transcription factors Siamois and Twin, also bias the presumptive ectoderm toward neural fate (Klein and Moody 2015; Gaur et al. 2016). The mechanism by which early zic gene expression is able to confer this predisposition for neural fate on the future neural ectoderm prior to gastrulation is currently not understood.
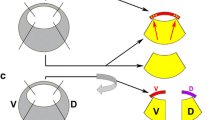
Zic3-null mice and Xenopus zic3 morphants exhibit left-right (L-R) asymmetry defects (Purandare et al. 2002; Ware et al. 2006a; Cast et al. 2012). Of the Xenopus zic genes, zic3 is most widely expressed in gastrula embryos (Fig. 7.2), and it is the only one among the zic genes that is involved in L-R asymmetry establishment. There are two prevailing models for the establishment of left-right (L-R) asymmetry in Xenopus. Evidence indicates asymmetry establishment either during early cleavage stages via ion flux or during gastrulation by cilia-driven flow (Blum et al. 2014). The result of breaking the symmetry by either mechanism is the asymmetric expression of the TGFβ-type growth factor nodal on the left side of the embryo. The zic3 gene is a direct target of Nodal signaling, most likely via the activin response element found in the first intron of zic3 (Weber and Sokol 2003). Zic3 then transmits this signal to downstream factors that determine left-sidedness (Kitaguchi et al. 2000). Zic3 has also been shown to regulate nodal expression in mice (Ware et al. 2006b). Thus, Zic3 may act upstream and downstream of the Nodal signaling that is required for L-R asymmetry formation.
5.3 Zic Proteins and Wnt Signaling
Zic proteins interact with canonical Wnt signaling, although the effects of these interactions appear to be dependent on Xenopus developmental stage. In late blastula embryos (stage 9.5), misexpression of zic3 reduces the expression of the direct Wnt/β-catenin targets goosecoid and siamois, which are genes expressed in the organizer, resulting in impaired notochord development. Zic3 is hypothesized to act as an early tuner of Wnt/β-catenin signaling in organizer mesoderm, where it is expressed at moderate levels (Fig. 7.1). It is likely that several Zic family members are able to affect Wnt/β-catenin signaling, since all five zic genes are able to reduce Wnt/β-catenin transcriptional activity in a luciferase reporter assay in Xenopus gastrula embryos and Zic3 was shown to physically interact with TCF1 (Fuijmi et al. 2012). Similarly, Zic2 binds directly to TCF4 and inhibits the ability of the β-catenin/TCF4 complex to activate transcription, thereby reducing the ability of β-catenin to induce Wnt targets in Xenopus animal caps (Pourebrahim et al. 2011). Further, the direct Zic1 targets Sp7 (also called Osterix) and Hesx1 (Cornish et al. 2009), both transcription factors, repress Wnt/β-catenin activity, and Hesx1 is expressed during late gastrula stage in the neural ectoderm (Andoniadou et al. 2011; Ermakova et al. 1999). These lines of evidence suggest that during gastrula stages and neural induction, Zic proteins inhibit canonical Wnt signaling.
Later in development, as Zic proteins contribute to patterning the neural plate, the effect of Zic1 on Wnt activity shifts. In neurula embryos, Zic1 acts as an activator of wnt8b expression, and it is able to activate wnt1 and wnt4 expression in neuralized animal caps. Further, Zic1 requires Wnt signaling to induce expression of the engrailed-2 gene in ectodermal explants (Merzdorf and Sive 2006), indicating a role for Zic1 in promoting canonical Wnt signaling. Finally, the direct Zic1 targets pkdcc2 and prickle (Cornish et al. 2009) suggest an unexplored role for Zic proteins in noncanonical Wnt signaling.
5.4 Zic Genes During Patterning of the Neural Plate
During Xenopus neurula stages, all five zic genes are expressed in overlapping yet distinct domains in the lateral neural plate and in the dorsal region of the closed neural tube (Fig. 7.2). Misexpression of each member of the Xenopus zic family expands the neural plate (zic1: Kuo et al. 1998; Mizuseki et al. 1998; Nakata et al. 1998), (zic2: Brewster et al. 1998; Nakata et al. 1998), (zic3: Nakata et al. 1997), (zic4: Fujimi et al. 2006), (zic5: Nakata et al. 2000). zic genes are expressed in relatively broad domains, as are other factors that pattern the neural plate. Combinations of these transcription factors, together with secreted factors, activate the expression of genes that are expressed in more limited domains. These include the wnt genes mentioned above (Merzdorf and Sive 2006): wnt1, which is expressed at the midbrain-hindbrain boundary, and wnt4 and wnt8b, which are expressed at the forebrain/midbrain boundary and in the midbrain. Additional genes induced by the expression of zic1 include the dorsal neural marker pax3, the hindbrain markers krox20, hoxD1 (Kuo et al. 1998), and Xfeb (glipr2) (Li et al. 2006). All zic genes induce the midbrain-hindbrain boundary marker en-2 (Nakata et al. 1997, 1998, 2000; Kuo et al. 1998; Fujimi et al. 2006). zic1-3 induce the forebrain and midbrain marker otx2 and the cement gland markers XAG-1 or XCG, while zic4 and zic5 are not able to induce these anterior genes (Kuo et al. 1998; Fujimi et al. 2006; Nakata et al. 2000). None of the zic genes are able to induce the posterior gene hoxB9. Most of these results were obtained in animal cap explants, although the regulation of the pax3, en-2, wnt8b, and krox20 genes was confirmed in whole embryos (Kuo et al. 1998; Merzdorf and Sive 2006; Gutkovich et al. 2010). Thus, Zic proteins regulate genes in the neural plate regions that give rise to the brain but so far do not appear to be involved in regulation of genes important for spinal cord development.
The zic1 gene is likely to play a role in the development of the midbrain-hindbrain boundary (MHB). Zic1 is required for expression of the MHB genes en-2 and wnt1. Since Wnt signaling is required for activation of en-2 expression by Zic1, Zic1 most likely induces wnt1 transcription, which in turn induces expression of the en-2 gene (Merzdorf and Sive 2006). Zic1 may also help maintain the MHB through its direct target gene Xfeb (glipr2). The Xfeb gene codes for a putative protease, which represses otx2 expression (Li et al. 2006). Xfeb is expressed in the hindbrain up to the MHB. The transcription factors Otx2 and Gbx2 maintain the MHB by mutual repression (Rhinn and Brand 2001). Xfeb activity may help maintain a posterior limit to otx2 expression during MHB formation. Thus, Zic1 may play a role in establishing and maintaining the midbrain-hindbrain boundary.
Zic family members appear to be essential for the formation of the hindbrain. Interfering with either zic1 or zic5 expression results in the loss of hindbrain cell fates (Gutkovich et al. 2010). Similar defects are observed when the transcription factor Xmeis is knocked down. In fact, defects in zic1 and zic5 morphants could be rescued with co-injection of xmeis RNA (Gutkovich et al. 2010). hoxD1, a gene that contributes to patterning the hindbrain, is a direct target of Xmeis and is known to be upregulated by zic1 (Kuo et al. 1998). This indicates that zic genes work upstream of xmeis and hoxD1 to promote formation of the hindbrain in Xenopus embryos. In addition, interfering with the expression of the Zic1 direct target gene Xfeb (glipr2) resulted in loss of hoxD1 expression (Li et al. 2006). While it is not known if Xfeb may lie upstream of xmeis1 or be part of a separate pathway, zic genes play an important upstream role during hindbrain development.
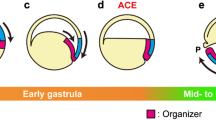
5.5 Zic Genes and the Neural Crest
zic genes act as neural crest specifiers, which has been shown in multiple organisms (reviewed in Merzdorf 2007; Houtmeyers et al. 2013). Neural crest cells are a migratory population of cells that originate from the neural plate border region. Multiple signaling pathways work together to specify the neural crest in two phases. During phase one, BMP, Wnt, and FGF signaling induces the expression of transcription factors like pax, msx, and zic family members, which are neural border specifiers. During phase two, these neural border specifiers induce the expression of neural crest specifiers, including snail1, snail2, ets1, and FoxD3 (Stuhlmiller and García-Castro 2012b). Accordingly, mutations in the mouse Zic2 or Zic5 genes result in a reduction in neural crest cells and deformities in neural crest-derived structures (Inoue et al. 2004; Elms et al. 2003). In Xenopus, all zic family members are expressed in the neural plate border region (Fig. 7.2; Fujimi et al. 2006) and are important for the formation of neural crest cells. Misexpression of zic1, zic2, or zic3 increases the extent of neural crest cell fate in whole embryos, and expression in animal cap explants results in the induction of neural crest markers (Nakata et al. 1997, 1998; Kuo et al. 1998). Similarly, misexpression of zic4 in Xenopus embryos generates ectopic pigment cells, a neural crest-derived cell type (Fujimi et al. 2006). Misexpression of zic5 in whole embryos causes strong induction of neural crest genes, but, unlike other zic family members, zic5 is not as efficient at inducing neural genes (Nakata et al. 2000). Conversely, interfering with the expression of zic genes results in a reduction in the expression of neural crest genes (Hong and Saint-Jeannet 2007; Fujimi et al. 2006; Nakata et al. 2000; Gutkovich et al. 2010). Thus, while having slightly different roles, all members of the zic family contribute to induction of the neural crest.
The Zic1 and Pax3 transcription factors work jointly to induce neural crest cell fate in the developing embryo. The expression of zic1 and pax3 overlaps in the presumptive neural crest region (Sato et al. 2005; Hong and Saint-Jeannet 2007). Misexpression of either zic1 or pax3 alone increases neural crest marker expression only in the ectoderm bordering the neural crest field, while overexpression of both genes together induces ectopic neural crest formation in the ventral ectoderm (Sato et al. 2005) in a Wnt-dependent manner (Monsoro-Burq et al. 2005). When ectopically induced neural crest cells (by activating zic1 and pax3 in animal cap explants) are transplanted into embryos, they are able to migrate correctly and form differentiated cell types characteristic of neural crest cell fates. Interestingly, the cooperation between Zic1 and Pax3 is required for these fates, since transplanting cells in which zic1 alone is activated results in the formation of neural tissue only (Milet et al. 2013). Thus, Zic1 and Pax3 can work together to induce a complete neural crest fate. While physical interaction between the Zic1 and Pax3 proteins was originally elusive (Sato et al. 2005), such an interaction was suggested by expressing these proteins in cultured cells (Himeda et al. 2013).
Since Zic1 and Pax3 together are able to induce a neural crest program in ectodermal explants, this synergy was employed to identify downstream neural crest genes (Plouhinec et al. 2014; Bae et al. 2014). The Plouhinec et al. (2014) screen focused on the identification of direct targets of Zic1/Pax3 and is described above. Bae et al. (2014) used a similar approach but did not limit their screen to direct targets. Both screens identified the snail1 and snail2 (slug) genes. The latter screen identified a variety of additional neural crest genes that may or may not be direct targets. Overall, a variety of familiar and new genes were identified that are activated by Zic1 and Pax3 acting together.
Among the neural crest specifiers, the snail1, snail2 (slug), and ets1 genes were identified as direct target genes of the interaction between Zic1 and Pax3 (Plouhinec et al. 2014). Further, Zic1 is required for snail1, snail2, and foxD3 expression (Plouhinec et al. 2014; Sasai et al. 2001; Gutkovich et al. 2010). FoxD3, which is not known as a direct target of Zic1 at this time, restricts cells to a neural crest fate and also aids in the migration of neural crest cells (Sasai et al. 2001). The Snail family and Ets1 are among the transcription factors that facilitate the delamination and migration of neural crest cells (Nie and Bronner 2015; Aybar et al. 2003). Both screens also identified the pdgfra gene, which codes for the alpha subunit of a platelet-derived growth factor (PDGF) receptor. PDGF receptor is important for migration of neural crest cells in mouse embryos (Soriano 1997), and in Xenopus it has been implicated in cell migration during gastrulation (Nagel et al. 2004; Van Stry et al. 2005). Thus, Zic1 is required for stabilizing neural crest fate and for the expression of genes that prepare neural crest cells for delamination and migration. While zic1 is expressed in premigratory neural crest cells and is essential for the expression of genes required for the transition of neural crest cells to emigrate, studies in chick show that it ceases to be expressed as soon as neural crest cells become migratory (Sun Rhodes and Merzdorf 2006). Since zic genes can repress neural differentiation genes, their role may include keeping the premigratory neural crest population in an undifferentiated state until the time of cell migration. Overall, the two screens confirmed that the neural plate border specifier Zic1 acts to induce neural crest specifier genes, with some of these interactions identified as direct. This adds further detail to the role of the Zic transcription factors in the gene regulatory landscape that governs neural crest specification.
5.6 Zic Genes and the Proliferation and Differentiation of Cells in the Nervous System
Xenopus embryos undergo primary neurogenesis, during which six discrete stripes of N-tubulin-positive primary neurons differentiate in the early neural plate, while the remainder of the neural plate remains as undifferentiated progenitors. zic2 is expressed in these undifferentiated progenitors between the stripes of primary neurons (Brewster et al. 1998). Misexpression of zic2 in the regions of primary neuron differentiation resulted in a significant decrease in the number of N-tubulin-positive primary neurons, indicating a role for Zic2 in preventing the differentiation of primary neurons. Consistent with this finding, Zic2 has a repressive effect on transcription of the bHLH gene neurogenin (ngnr-1), a gene that promotes neural differentiation (Brewster et al. 1998). Similarly, Sonic Hedgehog (Shh) signaling upregulates zic2 expression, and overexpression of Xenopus shh during primary neurogenesis causes expanded expression of zic2 and reduced N-tubulin-positive stripes (Franco et al. 1999). This indicates that zic2 acts in maintaining progenitors and preventing neurogenesis in certain areas of the neural plate, possibly under the regulation of Shh.
While Zic2 represses transcription of the neural differentiation factor ngnr-1 (Brewster et al. 1998), Zic1 and Zic3 have inductive effects on the expression of the proneural genes ngnr-1 and neuroD in animal cap explants (Nakata et al. 1997; Mizuseki et al. 1998). Further, hesx1, which promotes differentiation, is a direct target of Zic1 (Cornish et al. 2009). However, in mouse and chick embryos, Zic1 represses proneural gene expression (Ebert et al. 2003), misexpression of Zic1 blocks neuronal differentiation, and mutations in zic genes cause a decrease in cell proliferation in the dorsal neural tube (Ebert et al. 2003; Aruga et al. 2002; Nyholm et al. 2007). In addition, Zic1 promotes proliferation in the cerebellum and Zic1 and Zic3 in retinal precursors (Blank et al. 2011; Watabe et al. 2011). Consistent with these results from other organisms, interfering with btg2 expression in Xenopus embryos (Btg2 reduces proliferation and promotes neuronal differentiation) results in increased zic3 expression (Sugimoto et al. 2007), indicating that Btg2 downregulates zic3 gene expression to allow neurogenesis to begin. Thus, it appears that Zic2 has a role in maintaining undifferentiated progenitors in the neural ectoderm, while the role of Zic1 and Zic3 in proliferation and differentiation is not completely clear and may be context dependent. Interestingly, the early neural transcription factor FoxD4 regulates these zic genes differently. It induces zic2 transcription directly while indirectly repressing zic1 and zic3 expression (Neilson et al. 2012; Yan et al. 2009), which has been interpreted as FoxD4 keeping the neural ectoderm in a proliferative state by promoting zic2 and repressing zic1 and zic3 expression. Since the expression domains of the zic1, zic2, and zic3 genes overlap in the neural plate, it will be interesting and important to sort out the potentially opposite and context-dependent influences that these genes exert on neural differentiation.
6 Xenopus Studies Contribute to Our Understanding of Human Diseases
Xenopus embryos are increasingly employed as a model system in functional studies of human diseases (Kofent and Spagnoli 2016; Lienkamp 2016; Hardwick and Philpott 2015). With the near completion of the Xenopus genome and the advent of the TALEN and CRISPR-Cas9 systems of genome editing, such studies have become feasible (Tandon et al. 2016). With regard to diseases caused by mutations in human ZIC genes, Xenopus embryos were used to examine gene regulatory interactions in human craniosynostosis caused by mutations in the ZIC1 gene. Craniosynostosis is the premature fusion of skull sutures that leads to abnormalities in brain development and brain function in human patients. Five independent families with a history of coronal craniosynostosis showed four different mutations in the third exon of ZIC1, C-terminal to the zinc finger region (Twigg et al. 2015). These mutations include one point mutation and three nonsense mutations that result in truncations of the ZIC1 protein. The ZIC1 and engrailed (EN1) gene expression domains overlap in the developing sutures (Twigg et al. 2015). Using the regulatory relationship between Zic1 and the engrailed (en-2) gene as a model, misexpression of wild-type Xenopus zic1 or human ZIC1 does not change the engrailed (en-2) expression domain at the midbrain-hindbrain boundary in Xenopus embryos (Merzdorf and Sive 2006; Twigg et al. 2015). In contrast, the human mutant ZIC1 genes elicit increased and/or abnormal en-2 expression in Xenopus embryos, indicating that the mechanism by which these C-terminal ZIC1 mutations cause craniosynostosis may lie in dysregulation of the EN1 gene in the developing sutures (Twigg et al. 2015). En1 has been shown to regulate osteogenic differentiation and induction of Osterix (Sp7) during the formation of mouse skull sutures (Deckelbaum et al. 2006). Interestingly, Osterix (Sp7) is a direct target of zic1 (Table 7.2). Thus, ZIC1 appears to participate in a gene regulatory network, which is disturbed by mutations in the C-terminal domain of ZIC1, resulting in abnormal bone development in the coronal sutures and craniosynostosis in human patients.
Xenopus embryos were used to study the mechanism by which a mutation in the first zinc finger of the human ZIC3 gene causes TGA (transposition of the great arteries), which is a complex heart defect (Chhin et al. 2007). Zic3 plays a role in left-right axis formation and induction of the neural crest (Cast et al. 2012; Kitaguchi et al. 2000; Nakata et al. 1997, 1998), which are processes that may underlie the defects seen in the human patients. Injection of wild-type human ZIC3 into Xenopus embryos induced misexpression of the left lateral plate mesoderm marker pitx2 and the neural crest marker snail2. This induction activity was diminished when Xenopus embryos were injected with the mutant ZIC3 gene (Chhin et al. 2007). Thus, it appears that the mutation in the first zinc finger (which does not bind to DNA but engages in protein-protein interactions) diminishes the overall activity of ZIC3 in both left-right axis formation and neural crest induction. Thus, Xenopus embryos have proven useful in studying the interactions of mutant forms of human ZIC genes with developmental mechanisms to identify a molecular basis for human disease.
7 Conclusion
Work with Xenopus embryos has greatly contributed to understanding the role of Zic transcription factors during development. While zic gene family members are important players in many developmental processes, much remains to be understood about the molecular mechanisms that govern zic gene expression and Zic activities. The screens for direct and indirect targets of Zic transcription factors have yielded a variety of genes that are supporting ongoing and new research and are giving rise to new insights. Important are the advent of new genetic tools, such as new methods for genome editing, and the sequencing of the Xenopus laevis genome. Thus, previous studies can now be combined with genomic studies that have long been the strengths of other model organisms to form a more complete understanding of how Zic proteins drive development. Zic gene expression overlaps and their activities are partially redundant. Thus, it will be important to discover how individual zic genes are regulated and what distinguishes their functions. These studies will help with understanding the basis for human diseases. Indeed, Xenopus embryos have already been used to examine the molecular mechanisms underlying two human diseases caused by mutations in ZIC genes.
References
Andoniadou CL, Signore M, Young RM, Gaston-Massuet C, Wilson SW, Fuchs E, Martinez-Barbera JP (2011) HESX1- and TCF3-mediated repression of Wnt/β-catenin targets is required for normal development of the anterior forebrain. Development 138(22):4931–4942. https://doi.org/10.1242/dev.066597
Andreazzoli M, Gestri G, Cremisi F, Casarosa S, Dawid IB, Barsacchi G (2003) Xrx1 controls proliferation and neurogenesis in Xenopus anterior neural plate. Development 130(21):5143–5154. https://doi.org/10.1242/dev.00665
Aruga J, Mikoshiba K (2011) Role of BMP, FGF, calcium signaling, and Zic proteins in vertebrate neuroectodermal differentiation. Neurochem Res 36(7):1286–1292. https://doi.org/10.1007/s11064-011-0422-5
Aruga J, Tohmonda T, Homma S, Mikoshiba K (2002) Zic1 promotes the expansion of dorsal neural progenitors in spinal cord by inhibiting neuronal differentiation. Dev Biol 244(2):329–341. https://doi.org/10.1006/dbio.2002.0598
Aruga J, Kamiya A, Takahashi H, Fujimi TJ, Shimizu Y, Ohkawa K, Yazawa S, Umesono Y, Noguchi H, Shimizu T, Saitou N, Mikoshiba K, Sakaki Y, Agata K, Toyoda A (2006) A wide-range phylogenetic analysis of Zic proteins: implications for correlations between protein structure conservation and body plan complexity. Genomics 87(6):783–792. https://doi.org/10.1016/j.ygeno.2006.02.011
Aybar MJ, Nieto MA, Mayor R (2003) Snail precedes slug in the genetic cascade required for the specification and migration of the Xenopus neural crest. Development 130(3):483–494
Bae CJ, Park BY, Lee YH, Tobias JW, Hong CS, Saint-Jeannet JP (2014) Identification of Pax3 and Zic1 targets in the developing neural crest. Dev Biol 386(2):473–483. https://doi.org/10.1016/j.ydbio.2013.12.011
Batut J, Vandel L, Leclerc C, Daguzan C, Moreau M, Néant I (2005) The Ca2+−induced methyltransferase xPRMT1b controls neural fate in amphibian embryo. Proc Natl Acad Sci U S A 102(42):15128–15133. https://doi.org/10.1073/pnas.0502483102
Blank MC, Grinberg I, Aryee E, Laliberte C, Chizhikov VV, Henkelman RM, Millen KJ (2011) Multiple developmental programs are altered by loss of Zic1 and Zic4 to cause Dandy-Walker malformation cerebellar pathogenesis. Development 138(6):1207–1216. https://doi.org/10.1242/dev.054114
Blum M, Schweickert A, Vick P, Wright CV, Danilchik MV (2014) Symmetry breakage in the vertebrate embryo: when does it happen and how does it work? Dev Biol 393(1):109–123. https://doi.org/10.1016/j.ydbio.2014.06.014
Blum M, De Robertis EM, Wallingford JB, Niehrs C (2015) Morpholinos: antisense and sensibility. Dev Cell 35(2):145–149. https://doi.org/10.1016/j.devcel.2015.09.017
Böhm J, Buck A, Borozdin W, Mannan AU, Matysiak-Scholze U, Adham I, Schulz-Schaeffer W, Floss T, Wurst W, Kohlhase J, Barrionuevo F (2008) Sall1, sall2, and sall4 are required for neural tube closure in mice. Am J Pathol 173(5):1455–1463. https://doi.org/10.2353/ajpath.2008.071039
Brewster R, Lee J, Ruiz i Altaba A (1998) Gli/Zic factors pattern the neural plate by defining domains of cell differentiation. Nature 393(6685):579–583. https://doi.org/10.1038/31242
Brown L, Paraso M, Arkell R, Brown S (2005) In vitro analysis of partial loss-of-function ZIC2 mutations in holoprosencephaly: alanine tract expansion modulates DNA binding and transactivation. Hum Mol Genet 14(3):411–420. https://doi.org/10.1093/hmg/ddi037
Brugmann SA, Pandur PD, Kenyon KL, Pignoni F, Moody SA (2004) Six1 promotes a placodal fate within the lateral neurogenic ectoderm by functioning as both a transcriptional activator and repressor. Development 131(23):5871–5881. https://doi.org/10.1242/dev.01516
Cast AE, Gao C, Amack JD, Ware SM (2012) An essential and highly conserved role for Zic3 in left-right patterning, gastrulation and convergent extension morphogenesis. Dev Biol 364(1):22–31. https://doi.org/10.1016/j.ydbio.2012.01.011
Caunt CJ, Keyse SM (2013) Dual-specificity MAP kinase phosphatases (MKPs): shaping the outcome of MAP kinase signalling. FEBS J 280(2):489–504. https://doi.org/10.1111/j.1742-4658.2012.08716.x
Caverzasio J, Biver E, Thouverey C (2013) Predominant role of PDGF receptor transactivation in Wnt3a-induced osteoblastic cell proliferation. J Bone Miner Res 28(2):260–270. https://doi.org/10.1002/jbmr.1748
Chhin B, Hatayama M, Bozon D, Ogawa M, Schön P, Tohmonda T, Sassolas F, Aruga J, Valard AG, Chen SC, Bouvagnet P (2007) Elucidation of penetrance variability of a ZIC3 mutation in a family with complex heart defects and functional analysis of ZIC3 mutations in the first zinc finger domain. Hum Mutat 28(6):563–570. https://doi.org/10.1002/humu.20480
Cornish EJ, Hassan SM, Martin JD, Li S, Merzdorf CS (2009) A microarray screen for direct targets of Zic1 identifies an aquaporin gene, aqp-3b, expressed in the neural folds. Dev Dyn 238(5):1179–1194. https://doi.org/10.1002/dvdy.21953
Davey CF, Moens CB (2017) Planar cell polarity in moving cells: think globally, act locally. Development 144(2):187–200. https://doi.org/10.1242/dev.122804
Deckelbaum RA, Majithia A, Booker T, Henderson JE, Loomis CA (2006) The homeoprotein engrailed 1 has pleiotropic functions in calvarial intramembranous bone formation and remodeling. Development 133(1):63–74. https://doi.org/10.1242/dev.02171
Dong D, Ruuska SE, Levinthal DJ, Noy N (1999) Distinct roles for cellular retinoic acid-binding proteins I and II in regulating signaling by retinoic acid. J Biol Chem 274(34):23695–23698
Ebert PJ, Timmer JR, Nakada Y, Helms AW, Parab PB, Liu Y, Hunsaker TL, Johnson JE (2003) Zic1 represses Math1 expression via interactions with the Math1 enhancer and modulation of Math1 autoregulation. Development 130(9):1949–1959
Elms P, Siggers P, Napper D, Greenfield A, Arkell R (2003) Zic2 is required for neural crest formation and hindbrain patterning during mouse development. Dev Biol 264(2):391–406
Ermakova GV, Alexandrova EM, Kazanskaya OV, Vasiliev OL, Smith MW, Zaraisky AG (1999) The homeobox gene, Xanf-1, can control both neural differentiation and patterning in the presumptive anterior neurectoderm of the Xenopus laevis embryo. Development 126(20):4513–4523
Feledy JA, Beanan MJ, Sandoval JJ, Goodrich JS, Lim JH, Matsuo-Takasaki M, Sato SM, Sargent TD (1999) Inhibitory patterning of the anterior neural plate in Xenopus by homeodomain factors Dlx3 and Msx1. Dev Biol 212(2):455–464. https://doi.org/10.1006/dbio.1999.9374
Forecki J, Van Antwerp D, Lujan S, Merzdorf C, Antwerp V (2018) Roles for Xenopus aquaporin-3b (aqp3.L) during gastrulation: fibrillar fibronectin and tissue boundary establishment in the dorsal margin. Dev Biol 433(1):3–16. https://doi: 10.1016/j.ydbio.2017.11.001
Franco PG, Paganelli AR, López SL, Carrasco AE (1999) Functional association of retinoic acid and hedgehog signaling in Xenopus primary neurogenesis. Development 126(19):4257–4265
Fujimi TJ, Mikoshiba K, Aruga J (2006) Xenopus Zic4: conservation and diversification of expression profiles and protein function among the Xenopus Zic family. Dev Dyn 235(12):3379–3386. https://doi.org/10.1002/dvdy.20906
Fujimi TJ, Hatayama M, Aruga J (2012) Xenopus Zic3 controls notochord and organizer development through suppression of the Wnt/β-catenin signaling pathway. Dev Biol 361(2):220–231. https://doi.org/10.1016/j.ydbio.2011.10.026
Fukuda M, Takahashi S, Haramoto Y, Onuma Y, Kim YJ, Yeo CY, Ishiura S, Asashima M (2010) Zygotic VegT is required for Xenopus paraxial mesoderm formation and is regulated by nodal signaling and Eomesodermin. Int J Dev Biol 54(1):81–92. https://doi.org/10.1387/ijdb.082837mf
Furushima K, Murata T, Matsuo I, Aizawa S (2000) A new murine zinc finger gene. Opr Mech Dev 98(1–2):161–164
Gamse JT, Sive H (2001) Early anteroposterior division of the presumptive neurectoderm in Xenopus. Mech Dev 104(1–2):21–36
Gaston-Massuet C, Henderson DJ, Greene ND, Copp AJ (2005) Zic4, a zinc-finger transcription factor, is expressed in the developing mouse nervous system. Dev Dyn 233(3):1110–1115. https://doi.org/10.1002/dvdy.20417
Gaur S, Mandelbaum M, Herold M, Majumdar HD, Neilson KM, Maynard TM, Mood K, Daar IO, Moody SA (2016) Neural transcription factors bias cleavage stage blastomeres to give rise to neural ectoderm. Genesis 54(6):334–349. https://doi.org/10.1002/dvg.22943
Gestri G, Carl M, Appolloni I, Wilson SW, Barsacchi G, Andreazzoli M (2005) Six3 functions in anterior neural plate specification by promoting cell proliferation and inhibiting Bmp4 expression. Development 132(10):2401–2413. https://doi.org/10.1242/dev.01814
Grinberg I, Millen KJ (2005) The ZIC gene family in development and disease. Clin Genet 67(4):290–296. https://doi.org/10.1111/j.1399-0004.2005.00418.x
Gutkovich YE, Ofir R, Elkouby YM, Dibner C, Gefen A, Elias S, Frank D (2010) Xenopus Meis3 protein lies at a nexus downstream to Zic1 and Pax3 proteins, regulating multiple cell-fates during early nervous system development. Dev Biol 338(1):50–62. https://doi.org/10.1016/j.ydbio.2009.11.024
Hardwick LJ, Philpott A (2015) An oncologist’s friend: how Xenopus contributes to cancer research. Dev Biol 408(2):180–187. https://doi.org/10.1016/j.ydbio.2015.02.003
Hellsten U, Harland RM, Gilchrist MJ, Hendrix D, Jurka J, Kapitonov V, Ovcharenko I, Putnam NH, Shu S, Taher L, Blitz IL, Blumberg B, Dichmann DS, Dubchak I, Amaya E, Detter JC, Fletcher R, Gerhard DS, Goodstein D, Graves T, Grigoriev IV, Grimwood J, Kawashima T, Lindquist E, Lucas SM, Mead PE, Mitros T, Ogino H, Ohta Y, Poliakov AV, Pollet N, Robert J, Salamov A, Sater AK, Schmutz J, Terry A, Vize PD, Warren WC, Wells D, Wills A, Wilson RK, Zimmerman LB, Zorn AM, Grainger R, Grammer T, Khokha MK, Richardson PM, Rokhsar DS (2010) The genome of the western clawed frog Xenopus tropicalis. Science 328(5978):633–636. https://doi.org/10.1126/science.1183670
Himeda CL, Barro MV, Emerson CP (2013) Pax3 synergizes with Gli2 and Zic1 in transactivating the Myf5 epaxial somite enhancer. Dev Biol 383(1):7–14. https://doi.org/10.1016/j.ydbio.2013.09.006
Hollemann T, Schuh R, Pieler T, Stick R (1996) Xenopus Xsal-1, a vertebrate homolog of the region specific homeotic gene spalt of Drosophila. Mech Dev 55(1):19–32
Hong CS, Saint-Jeannet JP (2007) The activity of Pax3 and Zic1 regulates three distinct cell fates at the neural plate border. Mol Biol Cell 18(6):2192–2202. https://doi.org/10.1091/mbc.E06-11-1047
Houston DW, Wylie C (2005) Maternal Xenopus Zic2 negatively regulates nodal-related gene expression during anteroposterior patterning. Development 132(21):4845–4855. https://doi.org/10.1242/dev.02066
Houtmeyers R, Souopgui J, Tejpar S, Arkell R (2013) The ZIC gene family encodes multi-functional proteins essential for patterning and morphogenesis. Cell Mol Life Sci 70(20):3791–3811. https://doi.org/10.1007/s00018-013-1285-5
Houtmeyers R, Tchouate Gainkam O, Glanville-Jones HA, Van den Bosch B, Chappell A, Barratt KS, Souopgui J, Tejpar S, Arkell RM (2016) Zic2 mutation causes holoprosencephaly via disruption of NODAL signalling. Hum Mol Genet 25(18):3946–3959. https://doi.org/10.1093/hmg/ddw235
Inoue T, Hatayama M, Tohmonda T, Itohara S, Aruga J, Mikoshiba K (2004) Mouse Zic5 deficiency results in neural tube defects and hypoplasia of cephalic neural crest derivatives. Dev Biol 270(1):146–162. https://doi.org/10.1016/j.ydbio.2004.02.017
Jaurena MB, Juraver-Geslin H, Devotta A, Saint-Jeannet JP (2015) Zic1 controls placode progenitor formation non-cell autonomously by regulating retinoic acid production and transport. Nat Commun 6:7476. https://doi.org/10.1038/ncomms8476
Keller R, Danilchik M (1988) Regional expression, pattern and timing of convergence and extension during gastrulation of Xenopus laevis. Development 103(1):193–209
Keller R, Shih J, Sater AK, Moreno C (1992) Planar induction of convergence and extension of the neural plate by the organizer of Xenopus. Dev Dyn 193(3):218–234. https://doi.org/10.1002/aja.1001930303
Kelly LE, Carrel TL, Herman GE, El-Hodiri HM (2006) Pbx1 and Meis1 regulate activity of the Xenopus laevis Zic3 promoter through a highly conserved region. Biochem Biophys Res Commun 344(3):1031–1037. https://doi.org/10.1016/j.bbrc.2006.03.235
Kim HJ, Lee SK, Na SY, Choi HS, Lee JW (1998) Molecular cloning of xSRC-3, a novel transcription coactivator from Xenopus, that is related to AIB1, p/CIP, and TIF2. Mol Endocrinol 12(7):1038–1047. https://doi.org/10.1210/mend.12.7.0139
King LS, Kozono D, Agre P (2004) From structure to disease: the evolving tale of aquaporin biology. Nat Rev Mol Cell Biol 5(9):687–698. https://doi.org/10.1038/nrm1469
Kitaguchi T, Nagai T, Nakata K, Aruga J, Mikoshiba K (2000) Zic3 is involved in the left-right specification of the Xenopus embryo. Development 127(22):4787–4795
Klein SL, Moody SA (2015) Early neural ectodermal genes are activated by Siamois and Twin during blastula stages. Genesis 53(5):308–320. https://doi.org/10.1002/dvg.22854
Kofent J, Spagnoli FM (2016) Xenopus as a model system for studying pancreatic development and diabetes. Semin Cell Dev Biol 51:106–116. https://doi.org/10.1016/j.semcdb.2016.01.005
Kolm PJ, Sive HL (1995) Efficient hormone-inducible protein function in Xenopus laevis. Dev Biol 171(1):267–272. https://doi.org/10.1006/dbio.1995.1279
Koyabu Y, Nakata K, Mizugishi K, Aruga J, Mikoshiba K (2001) Physical and functional interactions between Zic and Gli proteins. J Biol Chem 276(10):6889–6892. https://doi.org/10.1074/jbc.C000773200
Kuo JS, Patel M, Gamse J, Merzdorf C, Liu X, Apekin V, Sive H (1998) Opl: a zinc finger protein that regulates neural determination and patterning in Xenopus. Development 125(15):2867–2882
Leclerc C, Webb SE, Daguzan C, Moreau M, Miller AL (2000) Imaging patterns of calcium transients during neural induction in Xenopus laevis embryos. J Cell Sci 113(Pt 19):3519–3529
Leclerc C, Lee M, Webb SE, Moreau M, Miller AL (2003) Calcium transients triggered by planar signals induce the expression of ZIC3 gene during neural induction in Xenopus. Dev Biol 261(2):381–390
Leclerc C, Néant I, Webb SE, Miller AL, Moreau M (2006) Calcium transients and calcium signaling during early neurogenesis in the amphibian embryo Xenopus laevis. Biochim Biophys Acta 1763(11):1184–1191. https://doi.org/10.1016/j.bbamcr.2006.08.005
Lepperdinger G (2000) Amphibian choroid plexus lipocalin, Cpl1. Biochim Biophys Acta 1482(1–2):119–126
Li S, Shin Y, Cho KW, Merzdorf CS (2006) The Xfeb gene is directly upregulated by Zic1 during early neural development. Dev Dyn 235(10):2817–2827. https://doi.org/10.1002/dvdy.20896
Li B, Kuriyama S, Moreno M, Mayor R (2009) The posteriorizing gene Gbx2 is a direct target of Wnt signalling and the earliest factor in neural crest induction. Development 136(19):3267–3278. https://doi.org/10.1242/dev.036954
Lienkamp SS (2016) Using Xenopus to study genetic kidney diseases. Semin Cell Dev Biol 51:117–124. https://doi.org/10.1016/j.semcdb.2016.02.002
Maeda R, Mood K, Jones TL, Aruga J, Buchberg AM, Daar IO (2001) Xmeis1, a protooncogene involved in specifying neural crest cell fate in Xenopus embryos. Oncogene 20(11):1329–1342. https://doi.org/10.1038/sj.onc.1204250
Maeda R, Ishimura A, Mood K, Park EK, Buchberg AM, Daar IO (2002) Xpbx1b and Xmeis1b play a collaborative role in hindbrain and neural crest gene expression in Xenopus embryos. Proc Natl Acad Sci U S A 99(8):5448–5453. https://doi.org/10.1073/pnas.082654899
Marchal L, Luxardi G, Thomé V, Kodjabachian L (2009) BMP inhibition initiates neural induction via FGF signaling and Zic genes. Proc Natl Acad Sci U S A 106(41):17437–17442. https://doi.org/10.1073/pnas.0906352106
Mattioni T, Louvion JF, Picard D (1994) Regulation of protein activities by fusion to steroid binding domains. Methods Cell Biol 43(Pt A):335–352
McMahon AR, Merzdorf CS (2010) Expression of the zic1, zic2, zic3, and zic4 genes in early chick embryos. BMC Res Notes 3:167. https://doi.org/10.1186/1756-0500-3-167
Merzdorf CS (2007) Emerging roles for zic genes in early development. Dev Dyn 236(4):922–940. https://doi.org/10.1002/dvdy.21098
Merzdorf CS, Sive HL (2006) The zic1 gene is an activator of Wnt signaling. Int J Dev Biol 50(7):611–617. https://doi.org/10.1387/ijdb.052110cm
Milet C, Maczkowiak F, Roche DD, Monsoro-Burq AH (2013) Pax3 and Zic1 drive induction and differentiation of multipotent, migratory, and functional neural crest in Xenopus embryos. Proc Natl Acad Sci U S A 110(14):5528–5533. https://doi.org/10.1073/pnas.1219124110
Mizugishi K, Aruga J, Nakata K, Mikoshiba K (2001) Molecular properties of Zic proteins as transcriptional regulators and their relationship to GLI proteins. J Biol Chem 276(3):2180–2188. https://doi.org/10.1074/jbc.M004430200
Mizugishi K, Hatayama M, Tohmonda T, Ogawa M, Inoue T, Mikoshiba K, Aruga J (2004) Myogenic repressor I-mfa interferes with the function of Zic family proteins. Biochem Biophys Res Commun 320(1):233–240. https://doi.org/10.1016/j.bbrc.2004.05.158
Mizuseki K, Kishi M, Matsui M, Nakanishi S, Sasai Y (1998) Xenopus Zic-related-1 and Sox-2, two factors induced by chordin, have distinct activities in the initiation of neural induction. Development 125(4):579–587
Monsoro-Burq AH, Wang E, Harland R (2005) Msx1 and Pax3 cooperate to mediate FGF8 and WNT signals during Xenopus neural crest induction. Dev Cell 8(2):167–178. https://doi.org/10.1016/j.devcel.2004.12.017
Moreau M, Néant I, Webb SE, Miller AL, Leclerc C (2008) Calcium signalling during neural induction in Xenopus laevis embryos. Philos Trans R Soc Lond Ser B Biol Sci 363(1495):1371–1375. https://doi.org/10.1098/rstb.2007.2254
Nagai T, Aruga J, Takada S, Günther T, Spörle R, Schughart K, Mikoshiba K (1997) The expression of the mouse Zic1, Zic2, and Zic3 gene suggests an essential role for Zic genes in body pattern formation. Dev Biol 182(2):299–313. https://doi.org/10.1006/dbio.1996.8449
Nagel M, Tahinci E, Symes K, Winklbauer R (2004) Guidance of mesoderm cell migration in the Xenopus gastrula requires PDGF signaling. Development 131(11):2727–2736. https://doi.org/10.1242/dev.01141
Nakata K, Nagai T, Aruga J, Mikoshiba K (1997) Xenopus Zic3, a primary regulator both in neural and neural crest development. Proc Natl Acad Sci U S A 94(22):11980–11985
Nakata K, Nagai T, Aruga J, Mikoshiba K (1998) Xenopus Zic family and its role in neural and neural crest development. Mech Dev 75(1–2):43–51
Nakata K, Koyabu Y, Aruga J, Mikoshiba K (2000) A novel member of the Xenopus Zic family, Zic5, mediates neural crest development. Mech Dev 99(1–2):83–91
Neilson KM, Klein SL, Mhaske P, Mood K, Daar IO, Moody SA (2012) Specific domains of FoxD4/5 activate and repress neural transcription factor genes to control the progression of immature neural ectoderm to differentiating neural plate. Dev Biol 365(2):363–375. https://doi.org/10.1016/j.ydbio.2012.03.004
Nie S, Bronner ME (2015) Dual developmental role of transcriptional regulator Ets1 in Xenopus cardiac neural crest vs. heart mesoderm. Cardiovasc Res 106(1):67–75. https://doi.org/10.1093/cvr/cvv043
Nyholm MK, Wu SF, Dorsky RI, Grinblat Y (2007) The zebrafish zic2a-zic5 gene pair acts downstream of canonical Wnt signaling to control cell proliferation in the developing tectum. Development 134(4):735–746. https://doi.org/10.1242/dev.02756
Patthey C, Gunhaga L (2014) Signaling pathways regulating ectodermal cell fate choices. Exp Cell Res 321(1):11–16. https://doi.org/10.1016/j.yexcr.2013.08.002
Pavletich NP, Pabo CO (1993) Crystal structure of a five-finger GLI-DNA complex: new perspectives on zinc fingers. Science 261(5129):1701–1707
Plouhinec JL, Roche DD, Pegoraro C, Figueiredo AL, Maczkowiak F, Brunet LJ, Milet C, Vert JP, Pollet N, Harland RM, Monsoro-Burq AH (2014) Pax3 and Zic1 trigger the early neural crest gene regulatory network by the direct activation of multiple key neural crest specifiers. Dev Biol 386(2):461–472. https://doi.org/10.1016/j.ydbio.2013.12.010
Pohl BS, Knochel W (2005) Of fox and frogs: fox (fork head/winged helix) transcription factors in Xenopus development. Gene 344:21–32. https://doi.org/10.1016/j.gene.2004.09.037
Pourebrahim R, Houtmeyers R, Ghogomu S, Janssens S, Thelie A, Tran HT, Langenberg T, Vleminckx K, Bellefroid E, Cassiman JJ, Tejpar S (2011) Transcription factor Zic2 inhibits Wnt/β-catenin protein signaling. J Biol Chem 286(43):37732–37740. https://doi.org/10.1074/jbc.M111.242826
Purandare SM, Ware SM, Kwan KM, Gebbia M, Bassi MT, Deng JM, Vogel H, Behringer RR, Belmont JW, Casey B (2002) A complex syndrome of left-right axis, central nervous system and axial skeleton defects in Zic3 mutant mice. Development 129(9):2293–2302
Rhinn M, Brand M (2001) The midbrain – hindbrain boundary organizer. Curr Opin Neurobiol 11(1):34–42
Sakurada T, Mima K, Kurisaki A, Sugino H, Yamauchi T (2005) Neuronal cell type-specific promoter of the alpha CaM kinase II gene is activated by Zic2, a Zic family zinc finger protein. Neurosci Res 53(3):323–330. https://doi.org/10.1016/j.neures.2005.08.001
Salero E, Pérez-Sen R, Aruga J, Giménez C, Zafra F (2001) Transcription factors Zic1 and Zic2 bind and transactivate the apolipoprotein E gene promoter. J Biol Chem 276(3):1881–1888. https://doi.org/10.1074/jbc.M007008200
Sanchez-Ferras O, Bernas G, Laberge-Perrault E, Pilon N (2014) Induction and dorsal restriction of paired-box 3 (Pax3) gene expression in the caudal neuroectoderm is mediated by integration of multiple pathways on a short neural crest enhancer. Biochim Biophys Acta 1839(7):546–558. https://doi.org/10.1016/j.bbagrm.2014.04.023
Sasai Y, De Robertis EM (1997) Ectodermal patterning in vertebrate embryos. Dev Biol 182(1):5–20. https://doi.org/10.1006/dbio.1996.8445
Sasai Y, Lu B, Piccolo S, De Robertis EM (1996) Endoderm induction by the organizer-secreted factors chordin and noggin in Xenopus animal caps. EMBO J 15(17):4547–4555
Sasai N, Mizuseki K, Sasai Y (2001) Requirement of FoxD3-class signaling for neural crest determination in Xenopus. Development 128(13):2525–2536
Sato T, Sasai N, Sasai Y (2005) Neural crest determination by co-activation of Pax3 and Zic1 genes in Xenopus ectoderm. Development 132(10):2355–2363. https://doi.org/10.1242/dev.01823
Schroeder TE (1970) Neurulation in Xenopus laevis. An analysis and model based upon light and electron microscopy. J Embryol Exp Morpholog 23(2):427–462
Session AM, Uno Y, Kwon T, Chapman JA, Toyoda A, Takahashi S, Fukui A, Hikosaka A, Suzuki A, Kondo M, van Heeringen SJ, Quigley I, Heinz S, Ogino H, Ochi H, Hellsten U, Lyons JB, Simakov O, Putnam N, Stites J, Kuroki Y, Tanaka T, Michiue T, Watanabe M, Bogdanovic O, Lister R, Georgiou G, Paranjpe SS, van Kruijsbergen I, Shu S, Carlson J, Kinoshita T, Ohta Y, Mawaribuchi S, Jenkins J, Grimwood J, Schmutz J, Mitros T, Mozaffari SV, Suzuki Y, Haramoto Y, Yamamoto TS, Takagi C, Heald R, Miller K, Haudenschild C, Kitzman J, Nakayama T, Izutsu Y, Robert J, Fortriede J, Burns K, Lotay V, Karimi K, Yasuoka Y, Dichmann DS, Flajnik MF, Houston DW, Shendure J, DuPasquier L, Vize PD, Zorn AM, Ito M, Marcotte EM, Wallingford JB, Ito Y, Asashima M, Ueno N, Matsuda Y, Veenstra GJ, Fujiyama A, Harland RM, Taira M, Rokhsar DS (2016) Genome evolution in the allotetraploid frog Xenopus laevis. Nature 538(7625):336–343. https://doi.org/10.1038/nature19840
Sive HL, Grainger RM, Harland RM (2007) Animal cap isolation from Xenopus laevis. CSH Protoc 2007:pdb.prot4744
Soriano P (1997) The PDGF alpha receptor is required for neural crest cell development and for normal patterning of the somites. Development 124(14):2691–2700
Stuhlmiller TJ, García-Castro MI (2012a) FGF/MAPK signaling is required in the gastrula epiblast for avian neural crest induction. Development 139(2):289–300. https://doi.org/10.1242/dev.070276
Stuhlmiller TJ, García-Castro MI (2012b) Current perspectives of the signaling pathways directing neural crest induction. Cell Mol Life Sci 69(22):3715–3737. https://doi.org/10.1007/s00018-012-0991-8
Sugimoto K, Okabayashi K, Sedohara A, Hayata T, Asashima M (2007) The role of XBtg2 in Xenopus neural development. Dev Neurosci 29(6):468–479. https://doi.org/10.1159/000097320
Sun Rhodes LS, Merzdorf CS (2006) The zic1 gene is expressed in chick somites but not in migratory neural crest. Gene Expr Patterns 6(5):539–545. https://doi.org/10.1016/j.modgep.2005.10.006
Takeuchi M, Nakabayashi J, Sakaguchi T, Yamamoto TS, Takahashi H, Takeda H, Ueno N (2003) The prickle-related gene in vertebrates is essential for gastrulation cell movements. Curr Biol 13(8):674–679
Tandon P, Conlon F, Furlow JD, Horb ME (2016) Expanding the genetic toolkit in Xenopus: approaches and opportunities for human disease modeling. Dev Biol. https://doi.org/10.1016/j.ydbio.2016.04.009
Tanibe M, Michiue T, Yukita A, Danno H, Ikuzawa M, Ishiura S, Asashima M (2008) Retinoic acid metabolizing factor xCyp26c is specifically expressed in neuroectoderm and regulates anterior neural patterning in Xenopus laevis. Int J Dev Biol 52(7):893–901. https://doi.org/10.1387/ijdb.082683mt
Tribulo C, Aybar MJ, Nguyen VH, Mullins MC, Mayor R (2003) Regulation of Msx genes by a Bmp gradient is essential for neural crest specification. Development 130(26):6441–6452. https://doi.org/10.1242/dev.00878
Tropepe V, Li S, Dickinson A, Gamse JT, Sive HL (2006) Identification of a BMP inhibitor-responsive promoter module required for expression of the early neural gene zic1. Dev Biol 289(2):517–529. https://doi.org/10.1016/j.ydbio.2005.10.004
Twigg SR, Forecki J, Goos JA, Richardson IC, Hoogeboom AJ, van den Ouweland AM, Swagemakers SM, Lequin MH, Van Antwerp D, SJ MG, Westbury I, Miller KA, Wall SA, van der Spek PJ, Mathijssen IM, Pauws E, Merzdorf CS, Wilkie AO, Consortium W (2015) Gain-of-function mutations in ZIC1 are associated with coronal craniosynostosis and learning disability. Am J Hum Genet 97(3):378–388. https://doi.org/10.1016/j.ajhg.2015.07.007
Urade Y, Hayaishi O (2000) Biochemical, structural, genetic, physiological, and pathophysiological features of lipocalin-type prostaglandin D synthase. Biochim Biophys Acta 1482(1–2):259–271
Van Stry M, Kazlauskas A, Schreiber SL, Symes K (2005) Distinct effectors of platelet-derived growth factor receptor-alpha signaling are required for cell survival during embryogenesis. Proc Natl Acad Sci U S A 102(23):8233–8238. https://doi.org/10.1073/pnas.0502885102
Verkman AS (2005) More than just water channels: unexpected cellular roles of aquaporins. J Cell Sci 118(Pt 15):3225–3232. https://doi.org/10.1242/jcs.02519
Vitorino M, Silva AC, Inácio JM, Ramalho JS, Gur M, Fainsod A, Steinbeisser H, Belo JA (2015) Xenopus Pkdcc1 and Pkdcc2 are two new tyrosine kinases involved in the regulation of JNK dependent Wnt/PCP signaling pathway. PLoS One 10(8):e0135504. https://doi.org/10.1371/journal.pone.0135504
Wallingford JB (2005) Neural tube closure and neural tube defects: studies in animal models reveal known knowns and known unknowns. Am J Med Genet C: Semin Med Genet 135C(1):59–68. https://doi.org/10.1002/ajmg.c.30054
Ware SM, Harutyunyan KG, Belmont JW (2006a) Zic3 is critical for early embryonic patterning during gastrulation. Dev Dyn 235(3):776–785. https://doi.org/10.1002/dvdy.20668
Ware SM, Harutyunyan KG, Belmont JW (2006b) Heart defects in X-linked heterotaxy: evidence for a genetic interaction of Zic3 with the nodal signaling pathway. Dev Dyn 235(6):1631–1637. https://doi.org/10.1002/dvdy.20719
Watabe Y, Baba Y, Nakauchi H, Mizota A, Watanabe S (2011) The role of Zic family zinc finger transcription factors in the proliferation and differentiation of retinal progenitor cells. Biochem Biophys Res Commun 415(1):42–47. https://doi.org/10.1016/j.bbrc.2011.10.007
Weber JR, Sokol SY (2003) Identification of a phylogenetically conserved activin-responsive enhancer in the Zic3 gene. Mech Dev 120(8):955–964
Winata CL, Kondrychyn I, Kumar V, Srinivasan KG, Orlov Y, Ravishankar A, Prabhakar S, Stanton LW, Korzh V, Mathavan S (2013) Genome wide analysis reveals Zic3 interaction with distal regulatory elements of stage specific developmental genes in zebrafish. PLoS Genet 9(10):e1003852. https://doi.org/10.1371/journal.pgen.1003852
Wu J, Li C, Zhao S, Mao B (2010) Differential expression of the Brunol/CELF family genes during Xenopus laevis early development. Int J Dev Biol 54(1):209–214. https://doi.org/10.1387/ijdb.082685jw
Yabe S, Tanegashima K, Haramoto Y, Takahashi S, Fujii T, Kozuma S, Taketani Y, Asashima M (2003) FRL-1, a member of the EGF-CFC family, is essential for neural differentiation in Xenopus early development. Development 130(10):2071–2081
Yamamoto TS, Takagi C, Ueno N (2000) Requirement of Xmsx-1 in the BMP-triggered ventralization of Xenopus embryos. Mech Dev 91(1–2):131–141
Yan B, Neilson KM, Moody SA (2009) FoxD5 plays a critical upstream role in regulating neural ectodermal fate and the onset of neural differentiation. Dev Biol 329(1):80–95. https://doi.org/10.1016/j.ydbio.2009.02.019
Yang Y, Hwang CK, Junn E, Lee G, Mouradian MM (2000) ZIC2 and Sp3 repress Sp1-induced activation of the human D1A dopamine receptor gene. J Biol Chem 275(49):38863–38869. https://doi.org/10.1074/jbc.M007906200
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Merzdorf, C., Forecki, J. (2018). Amphibian Zic Genes. In: Aruga, J. (eds) Zic family. Advances in Experimental Medicine and Biology, vol 1046. Springer, Singapore. https://doi.org/10.1007/978-981-10-7311-3_7
Download citation
DOI: https://doi.org/10.1007/978-981-10-7311-3_7
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-10-7310-6
Online ISBN: 978-981-10-7311-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)