Abstract
More and more metabolic diseases, chronic inflammatory diseases, and cancers are being linked with the alteration in gut microbiota. Host genetics and environment are some of the factors that are thought to contribute in shaping gut flora. Recent research has suggested that development of gut microbial consortia and thereby host-microbe interaction is essentially guided by the early colonizers and the diet. Important metabolites derived from the gut bacteria and diet help in the development and maintenance of a healthy gut and consequently the immune system. In the present review, we have examined the impact of diet on evolution, stabilisation and dysbiosis of human gut microbiota and how diet induced subtle changes in the microbiota lead to the disease state. Diet, an important environmental factor, plays crucial role in tilting this fine balance in either directions. The importance of dietary pattern in regulating growth of beneficial bacteria has been discussed. We have also examined different intervention strategies that affect microbiota and consequently the metabolite profiles resulting in a diseased state. Various high-throughput techniques that are used in the studies described here have also been discussed.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
21.1 Gut Flora: From Oblivion to Limelight
We have witnessed a gigantic leap in the past one and half decade in the field of microbiome research that has helped us to realize the critical role of gut bacteria in shaping our health through manipulation of overall human physiology. The dynamic changes in the gut microbiome (GM) can now be evaluated with new technologies that allow us to interrogate nucleic acid sequence, metabolome, and proteome at an unprecedented depth. This helps in analyzing complex microbial communities, such as GM of healthy and diseased individuals. As early as 2001, Hooper and Gordon had described that human lives are affected by the microbes, both positively and negatively from birth to death, and human body surfaces are colonized by vast, diverse, and dynamic microbial consortia. The structure, variability, and functionality of the microbial consortium vary depending on the body environment. The human gut is densely populated in a nutrient-rich environment housing trillions of microbes of diverse microbial species. The entire gut from oral cavity to rectum is populated by bacteria, but the majority lives in our colon with maximum densities reaching to 1011–1012 cells/mL (Whitman et al. 1998). Each individual harbors about 1000 or more phylotypes of gut bacteria, and the combined gene content of GM exceeds that of human by about 100-fold (Qin et al. 2010). Moreover, there is tremendous amount of diversity in terms of different bacteria within and between different persons and can fluctuate at different time points especially during disease (Lozupone et al. 2012). The gut bacteria broadly include the members of phyla Bacteroides, Firmicutes, Actinobacteria, Proteobacteria, Verrucomicrobia, Cyanobacteria (Melainabacteria), and Archaea (Eckburg et al. 2005).
One of the most important metabolic functions carried out by gut microbiota (GMt) is fermentation of non-digested food components in the large intestine and extraction of extra energy in the form of short-chain fatty acids (SCFAs), i.e., acetate, propionate, and butyrate. GMt also produces vitamins and helps in absorption of minerals by the host gut. Gut flora maintains the equilibrium between the enteric nervous system and gut immune system of the host. The alterations in configuration of gut flora may be potentially harmful to the host. Several recent research reports have indicated the involvement of GMt in the progression of several chronic diseases, such as obesity, diabetes mellitus, rheumatoid arthritis, inflammatory bowel disease (IBD), heart diseases, allergy, and cancer. Alterations in GMt can be attributed to the use of antibiotics, age, disease, stress, dietary habits, socioeconomic status, and lifestyle (Flint et al. 2012). The diet has been considered to be a universal external factor that can influence the gut microbial composition of the host. Preliminary evidence suggests that the dietary patterns are associated with specific composition of the microbiota, and it is now possible to link various constituents of food with respect to growth of specific bacteria. In this chapter, we have focused on summarizing current understanding on various effects of diet on gut flora, optimizing gut microbiota composition by dietary means and various challenges faced in designing and interpreting the results from human studies.
21.2 Factors Affecting the Composition of Gut Microbiota
The composition of gut bacterial communities is generally stable in adult individuals and yet quite dynamic as it varies from person to person. In the uterus, fetus encounters very few microbes and microbial components maintaining near sterility in the gut. The colonization of infant gut begins during the birth and continues after the birth. The diversity, richness and the stability of gut microbiota are influenced by several factors, such as mode of delivery, age, diet, host genetics, hygiene, sanitation and pharmacological exposure to antibiotics (Fig. 21.1). Broadly the factors can be defined as follows.
21.2.1 Age
The development of human fetus takes place in almost sterile ambience, and the colonization of gut begins immediately after the birth of an infant. Firstly, the mode of delivery (vaginal birth vs. cesarean section) affects the early colonization of gut. Infants born through vaginal route predominantly harbor Lactobacillus and Prevotella (bacteria found in vagina), whereas infants delivered through cesarean section are likely to harbor bacteria found on mother’s skin and in the hospital environment (Dominguez-Bello et al. 2010). Babies born through cesarean section are exposed to antibiotics early in life which may influence the gut microbiota. The gut flora of infants is initially shaped by mother’s milk or formula milk and later by solid food at the postweaning stage. In about 2–5 years, the gut flora of child matures and resembles with that of the adult in terms of richness and diversity (Yatsunenko et al. 2012). The gut flora of elderly persons depends on the lifestyle, diet, health status, and exposure to antibiotics (Claesson et al. 2012; Jeffery et al. 2016). The diversity and stability of gut microbiota vary with age from infancy to adulthood and depend on interaction with host genetics and environmental factors as depicted in Fig. 21.2.
21.2.2 Diet
The word “diet” originated from the Greek word “diaita” which means balanced way of life. Diet plays very critical role in configuring the gut microbial composition and the effect of diet on gut flora begins very early in the life with very first meal of the infant in the form of mother’s milk. Gut flora of breast-fed infants is different from that of the formula-fed infants (Harmsen et al. 2000). Dietary interventions with high-fat and high-sugar (HFHS) diet and plant polysaccharide-based diet in mice of different genetic background have shown that the effect of diet dominates the genetic background in configuring the gut microbiota (Kashyap et al. 2013; Carmody et al. 2015). Diet not only affects the composition of gut bacterial flora of host but it can also affect gene expression of gut microbes. The gut bacteria Bacteroides thetaiotaomicron changes gene expression depending on the availability of dietary fiber (plant glycan) in food. If diet is low in dietary fiber, the expression of gene for fermentation of host mucus glycans is upregulated, and reverse phenomenon is seen when dietary fiber is high in food (Kashyap et al. 2013). The diet of an individual depends on several factors, such as geographical location, climate, socioeconomic status, ethnicity, mode of subsistence and health status (Fig. 21.3). The composition of major food components (proportion of carbohydrate, protein and fat), source/type (vegan, vegetarian, nonvegetarian, or omnivorous), and pattern (long term or short term) of diet affects gut microbiota of individuals.
21.2.3 Host Genetics
The composition of gut bacterial flora is the result of coevolution between host and gut bacteria (Ley et al. 2006). The gut microbiota of twins is more similar than that of non-twins and parents depending on relatedness (Zoetendal et al. 2001). The host genotype can affect the gut microbial diversity in number of ways. For example, the absence of α-1-2-fucosyltransferase activity in non-secretor (FUT2 mutants) individuals led to altered mucus structures in the host intestine and eventually deceased α-diversity of gut microbiota (Kashyap et al. 2013). Humans harboring mutation in FUT2 gene are more susceptible to gut microbiota-associated chronic inflammatory diseases, such as Crohn’s disease (McGovern et al. 2010) and primary sclerosing cholangitis (Folseraas et al. 2012). The host genotype is responsible for lactose digestion in the small intestine of individuals. Two SNPs (C/T13,910 and G/A22,018) located in the 14 and 22 kb upstream of the 5′-end of lactase gene have been associated with lactase persistence. Lactose-intolerant individuals are unable to digest it in the small intestine and slowly move to the colon, and subsequently colonic flora metabolizes them into unwanted by-products like large quantity of H2 and methane gas (Enattah et al. 2002).
Even, the presence of some microbial taxa has been linked with host genetics. The members of the family Christensenellaceae, methanogenic archaea, and few other undefined bacteria coexist in human gut in the form of a network that is inherited. The consortium of Christensenellaceae along with the partners is associated with low body mass index (BMI) individuals indicating the role of host genetics in shaping the gut microbial composition and consequently the host metabolism (Goodrich et al. 2014).
21.2.4 Hygiene
Several animal studies indicate that gut microbial richness and diversity play an important role in the development and progression of noncommunicable diseases like IBD, diabetes, autoimmune disorders, colon cancer, and cardiovascular diseases. Due to poor hygienic practices, infectious diseases are highly prevalent whereas noncommunicable diseases are very less prevalent in nonindustrialized countries such as Papua New Guinea, Burkina Faso, Malawi, and Tanzania. Various studies on the fecal samples of children and adults of non-industrialized countries (Burkina Faso (De Filippo et al. 2010), children and adults in Malawi and Amazonian Amerindians (Yatsunenko et al. 2012), and adult Hadza hunter-gatherers in Tanzania (Schnorr et al. 2014), adults in Papua New Guinea) have shown high α-diversity and lower β-diversity in gut microbiota in comparison to US and European population (Clemente et al. 2015; Martinez et al. 2015). Progressive shrinkage in gut microbial diversity is a cumulative result of changes in lifestyle, diet, hygienic practices, and exposure to antibiotics over the years in Western countries. According to microflora hypothesis, “the low microbial exposure of individuals following western lifestyle affects colonization of the infant’s gut and disturbs the immune system development leading to chronic inflammatory diseases” (Wold 1998). Several reports do indicate and validate the microflora hypothesis in humans. Recently Zhou et al. reported that gut microbial diversity and serum IgE levels in mice vary with exposure to antigen or bacteria from housing environment such as soil, house dust, and decaying plants (Zhou et al. 2016). Excessive hygienic and sanitation practices affect the diversity of gut microbiota and lead to poor development of immune system.
21.2.5 Antibiotics
The use of antibiotics in control, management, and treatment of bacterial infections has been a milestone in public health and has saved millions of lives worldwide. Though antibiotics have saved the lives, the collateral damage they cause to indigenous host flora is now being evaluated to fine-tune its use in medicine (Modi et al. 2014). Exposure to antibiotics may start very early in life of C-section babies and other children depending on health status. In mice, pulsed antibiotic treatment in early life has resulted in perturbed gut flora with altered metabolic functions, altered gene expression in intestinal epithelial cells, and recruitment of different T-cells in the lamina propria. These non-obese diabetic mice developed type 1 diabetes early in life than controls (Livanos et al. 2016). The use of broad spectrum antibiotic results in depletion of commensal gut flora diversity, and colonization and proliferation of antibiotic-associated diarrhea (AAD) associated pathogens such as Salmonella typhimurium and Clostridium difficile (Hogenauer et al. 1998). AAD is prevalent in 5–25% of antibiotic-treated patients (Bergogne-Berezin 2000).
The growth of S. typhimurium and Clostridium difficile in lumen of antibiotic-treated patients is facilitated by microbiota-generated mucosal carbohydrates, fucose and sialic acid. S. typhimurium catabolizes fucose and sialic acid, while C. difficile uses sialic acid in vivo. The impact of antibiotic treatment on human gut flora can be detected as early as 3–4 days of treatment initiation and may persist for more than 2 years. Frequent antibiotic courses may result in alternative and stable microbial configuration enriching antibiotic resistance gene in gut metagenome. The recovery to pre-antibiotic treatment state varies across individuals and remains often incomplete (Dethlefsen and Relman 2011). The unnecessary, excessive, incomplete dose compliance has resulted in multidrug resistance in pathogens. Interestingly, functional antibiotic resistance genes have been reported in gut flora of Yanomami tribe, Venezuela, which has never been exposed to commercial antibiotics. However, the source of antibiotic resistance genes for Yanomami tribe may be from antibiotic-producing soil bacteria or human commensal ancestors found in soil (Clemente et al. 2015). Antibiotic exposure leads to enrichment of antibiotic resistance genes and transfers of these genes to other bacteria via horizontal gene transfer. Antibiotic exposure along with other environmental factors, such as high-fat diet, enhances the propensity of development of a number of metabolic diseases mainly due to alteration in GM (Mahana et al. 2016).
21.2.6 Parasitism
It has been hypothesized that the gut bacterial components from the intestinal microbiota function as a stimulatory factor in the pathogenicity of the parasite giardia (Torres et al. 2000). Entamoeba histolytica, an intestinal protozoan parasite infection, has been shown to alter the gut flora (Verma et al. 2012). A recent study showed that colonization of gut by commensal segmented filamentous bacteria protects from E. histolytica infection in a murine model (Burgess et al. 2014). The higher parasitic load (E. histolytica) and significantly increased population of Prevotella copri in symptomatic cases have been found to be associated with diarrhea in Bangladesh children below 2 years of age (Gilchrist et al. 2016). Further, a recently conducted metagenomic study in African population stated that colonization by E. histolytica can be estimated with ~80% accuracy on the basis of gut microflora of an individual. Individuals with entamoeba infection show high alpha diversity and low interindividual variation. However, it is important to note that these subjects were asymptomatic at the time of this study (Morton et al. 2015). Furtther studies are required to understand if the observed relationship of parasitic infection and gut flora is cause or effect.
21.3 Diet as a Modulating Factor of Gut Bacterial Flora
Diet is considered a crucial factor that shapes gut flora, and the population of each microbial community can also be linked with diet. Diet-induced modifications in gut microbiota have been implicated in high incidence/prevalence of diseases of affluence such as obesity, cardiovascular disease, diabetes and IBD in industrialized USA and other Western countries (Hou et al. 2011; Moschen et al. 2012). Population can be separated from each other on the basis of characteristic differences in their gut flora associated with dietary pattern. The ratio of dominant gut flora like Bacteroides and Prevotella shows good correlation with overall diversity and dietary pattern. High concentration of Prevotella has been associated with consumption of high-fiber diet in children of African villages (agrarian diet), whereas high concentration of Bacteroides has been linked with diet rich in animal protein, saturated fat, and refined carbohydrates typical of Western diet (Wu et al. 2011; Yatsunenko et al. 2012).
The categorization of microbial configuration of individuals into distinct groups is called enterotyping, and a particular discrete microbial configuration is called enterotype. The concept of enterotypes links gut microbial configurations to diet. It is clear from multiple studies that evolution of specific enterotypes happens due to long-term dietary pattern. Specific genera that constitute an enterotype remain stable unaffected by age, sex, BMI, and nationality of an individual (Arumugam et al. 2011). The predominant enterotypes can be categorized as Bacteroides, Prevotella, and Ruminococcus. Bacteroides enterotype dominated by members of phylum Bacteroidetes (especially by Alistipes and Parabacteroides) is associated with the consumption of high animal fat and high-protein diet. Similarly Prevotella enterotype dominated by Paraprevotella (phylum Bacteroidetes) and Catenibacterium (phylum Firmicutes) is associated with high-fiber/high-carbohydrate diet. The Ruminococcus enterotype partially overlaps with that of Bacteroides enterotype and cannot be classified as a separate enterotype (Wu et al. 2011; Tremaroli and Backhed 2012; Graf et al. 2015).
21.4 Effect of Dietary Constituents on Gut Flora
Relative composition of major diet constituents, such as lipids, proteins, and carbohydrates, affects gut microbiota and human health differently. Gut microbes use different mechanisms to collect energy from lipids, proteins, and carbohydrates.
21.4.1 Carbohydrates
A large fraction of our diet is made up of different types of carbohydrates, a main source of energy and nutrient for both host and microbes. Many components of diet including digestible carbohydrates like sucrose, lactose, starch, etc. are digested and assimilated in the small intestine by human enzymes except undigested dietary complex carbohydrates. The undigested products are subsequently passed into the large intestine for fermentation by gut bacteria. Human gut microbiome (a model microbiome of 177sp.) is very rich in glycoside hydrolases (>9000) and polysaccharide lyases (~300) in comparison to human genome which barely encodes ~17 glycoside hydrolases and no polysaccharide lyases (Cantarel et al. 2012; El Kaoutari et al. 2013). According to the “American Society of Cereal Chemists” (2001), dietary fiber can be defined as “the edible parts of plants or analogous carbohydrates that are resistant to digestion and absorption in human small intestine with complete or partial fermentation in the large intestine.” The carbohydrates which are metabolically available for fermentation by gut bacteria are collectively referred as microbiota-accessible carbohydrates (MACs) (Sonnenburg and Sonnenburg 2014). The main sources of dietary MACs are plant glycans (e.g., dietary fiber), glycans of animal origin, host-produced glycans (e.g., mucin), and polysaccharides produced by food-associated microbes. The quantity of MAC present in a food source depends on members of gut microbiota of an individual. In Japan, a popular food sushi is prepared using a nutritional seaweed Porphyra spp. (nori). The gut bacterium Bacteroides plebeius which can digest complex algal polysaccharides porphyran and agarose is found only in Japanese individuals but not in Westerners. Thus, algal polysaccharide porphyran is MAC for Japanese but not for North Americans and Europeans (Hehemann et al. 2010). However, it is important to note that the genes for digestion of algal polysaccharides have been acquired by gut bacterium Bacteroides plebeius from marine bacteria associated with seafood. This indicates the source of novel genes and how gut flora evolves with diet in individuals of a geographic location.
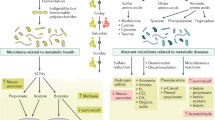
Consumption of fiber-rich diet has been associated with high microbial diversity with predominance of Prevotella enterotype. The microbial fermentation of dietary fibers produces acetate, propionate, and butyrate commonly known as short-chain fatty acids (SCFAs). Acetate, propionate, and butyrate are found at a molar ratio of 60:20:20 in colonic lumen. About 80–90% of SCFAs are absorbed in the colon and have profound impact on gut and host physiology. The acetate and propionate are sources of energy in peripheral tissues whereas butyrate for colonocytes. Both of these SCFAs are used as substrate for lipogenesis and gluconeogenesis by the liver, respectively (Rombeau and Kripke 1990). Propionate is reported to enhance intestinal gluconeogenesis via afferent nervous system (De Vadder et al. 2014). Acetate is used as substrate for production of butyrate by Faecalibacterium prausnitzii and Roseburia spp. (Duncan et al. 2004) and protects from enteric infections (Fukuda et al. 2011). Butyrate (Davie 2003) and acetate (Thorburn et al. 2015) can inhibit histone deacetylase inhibitors and consequently affect gene expression in colonic epithelial cells and peripheral tissues, respectively. Butyrate has shown anti-inflammatory and anticarcinogenic activity in cell lines, but studies on human subjects are limited (Hamer et al. 2008). Butyrate has also been associated with strong antitumor activity. The cellular effect of short-chain fatty acids is mediated through G-protein-coupled receptors GPR41/FFAR3 (Samuel et al. 2008) and GPR43/FFAR2 affecting inflammation (Maslowski et al. 2009). It also helps in the regulation of antidiabetic hormone GLP-1 secretion (Tolhurst et al. 2012). SCFAs are anti-inflammatory, regulate glucose homeostasis, promote integrity of intestinal epithelium and gut homeostasis, and regulate the number and function of the regulatory T-cells in the colon (Thorburn et al. 2015). Two predominant phyla Bacteroidetes and Firmicutes are main producers of SCFAs in human gut. Acetate and propionate are mainly produced by several members of Bacteroidetes, whereas butyrate is produced in high amounts by the Firmicutes (Macfarlane and Macfarlane 2003). Human milk is rich in milk oligosaccharides (MOS), most of which are indigestible glycans and possess prebiotic property (Stark and Lee 1982). MOS, phosphate, and milk proteins are known to encourage the growth of Bifidobacteria spp. and Lactobacillus spp. (Yoshioka et al. 1983). Breast-fed babies harbor more Bifidobacteria and Lactobacillus sp. but less of Bacteroides spp., Enterobacteria, and Clostridium spp. in comparison to formula-fed babies (Harmsen et al. 2000; Fallani et al. 2011).
The fermentation of different types of cellulose in human colon is influenced by the presence or absence of methanogens which directs the configuration of cellulolytic microbial consortium in human gut. The cellulose fermenters belong mainly to Bacteroidetes in non-methane excreting individuals, whereas members of Firmicutes degrade cellulose in methane excreting individuals. This diversity of cellulose degraders enables the fermentation of different kinds of cellulose (Chassard et al. 2010). Dietary modifications can influence host-microbe interaction, the immune system, and subsequently inflammation in different tissues. Experiments on mice have shown that mice fed on high-fiber diet had higher concentration of circulating SCFAs and were protected from lung allergy in comparison to mice fed on low-fiber diet. Influence of diets in the well-being of individuals is likely to be through microbes, microbial metabolites, and hematopoiesis, though we still do not understand detailed pathways linking diet to disease processes (Trompette et al. 2014).
Arabinogalactan and inulin have the potential to promote a gut bacterial profile found in a lean individual. Propionate and acetate are the two SCFAs predominantly generated by arabinogalactan fermentation. Fermentation of arabinogalactan by microbiota of an obese individual showed a higher production of propionate in comparison to n-butyrate. The fermentation of both substrates by lean microbiota produces more n-butyrate in comparison to that in obese microbiota (Aguirre et al. 2016). Arabinogalactan fermentation has been shown to be associated with a statistically significant increase in the population of bacteria particularly Bacteroidetes and Faecalibacterium prausnitzii with a simultaneous decrease in pathogenic bacteria Clostridium perfringens (Terpend et al. 2013). The effect of dietary glycans on gut microbiota and overall metabolic functions may vary depending on initial gut microbial configuration (Walker et al. 2011).
21.4.2 Effect of Noncaloric/Nonnutritive Sweeteners on Health and Gut Microbiota
Noncaloric/nonnutritive sweeteners (synthetic and natural) are regularly being used as additives in food and beverages across the globe in order to control negative impact of high-caloric sugar on human health that includes weight gain and obesity, among other diseases (Swithers 2013; Suez et al. 2014). Aspartame (l-aspartyl-l-phenylalanine methyl ester), sucralose (trichlorogalactosucrose), saccharin (1,1-dioxo-1,2-benzothiazol-3-one), acesulfame-K (5,6-dimethyl-1,2,3-oxathiazine-4(3H)-one 2,2-dioxide), stevia (steviol glycosides, rebaudioside A, stevioside), neotame (N-[N-(3,3-dimethylbutyl)-l-α-aspartyl]-l-phenylalanine-1-methyl ester), and luo han guo extract (cucurbitane glycosides, mogrosides II, III, IV, V, VI) are the artificial sweeteners approved by FDA, USA (Fitch and Keim 2012). All of these noncaloric/nonnutritive sweeteners have also been approved by FSSAI, India, for use as an additive in food and beverages except luo han guo extract. Nonnutritive sweeteners are generally considered healthy alternative to caloric sugars, but recent animal studies suggest that replacing caloric sugars with synthetic sweeteners leads to metabolic dysregulation. Saccharin and aspartame induce greater weight gain than sucrose in rats with similar caloric consumption (Feijo et al. 2013). Artificial sweeteners such as acesulfame-K and saccharin and natural nonnutritive sweeteners such as stevioside (sweeteners extracted from the leaves of the Stevia rebaudiana plant) both increase food intake, weight gain, and accumulation of body fat in Wistar rats in comparison to glucose (Swithers and Davidson 2008; Swithers et al. 2010; Swithers 2013). Chronic dietary consumption of sucralose induces reversible chronic sweet/energy imbalance and results in activation of neuronal starvation response leading to increased food intake, perturbed glucose homeostasis, insomnia, and hyperactivity in fruit flies and mammals (Wang et al. 2016). As most of the noncaloric sweeteners are not digested in human intestine, chronic ingestion of some of the noncaloric sweeteners, such as saccharin sucralose and aspartame, disturbs glucose homeostasis and gut flora in C57B1/6 mice. Metagenomic analysis of gut flora of saccharin-consuming mice showed alteration in more than 40 OTU with relative enrichment of several taxa belonging to Bacteroides genus and Clostridiales order. Saccharin-consuming mice produced high level of SCFAs (acetate and propionate) than glucose-consuming mice, and glycan-degrading pathways are overrepresented in their GM. Saccharin induced changes in GM, and consequently glucose intolerance was also observed in humans (Suez et al. 2014).
21.4.3 Fat
Fat is an important constituent of our diet. It enhances the flavor and palatability of food and is an important component of cooking across the world. Dietary fat provides energy, helps in absorption of fat-soluble vitamins, and is considered as the main source of essential fatty acids such as ω-6 linoleic acid and ω-3 linolenic acid (Schmid 2011). Fat is an essential structural component of cell wall, a substrate for hormones and participates as cell-signaling molecule. Fat plays a very crucial role in the development of neurological and brain function in young children (Milner and Allison 1999). It is synthesized in our body and only a small amount of fat is needed for our daily needs. Dietary fat mainly contains three types of fats, viz., saturated, unsaturated, and trans fat. Most of the food items contain mixed types of fat. Unsaturated fats contain mono- and polyunsaturated fatty acids mostly generated from plant products such as seeds, nuts, and vegetable oils. On the other hand, saturated fat is generated mainly from animal-derived food, such as meat, fish, and dairy products. However, few plant products such as palm oil and coconut oil are also sources of saturated fat. The main source of trans fats is hydrogenated vegetable oil, but it is also present in small amount in beef and dairy fat. The quality and quantity of dietary fat affect human health, and excess consumption of fat has been associated with obesity and cardiovascular diseases. The nature of dietary fat consumed can induce significant changes in microbiota and gut barrier function as shown in mice (Lam et al. 2015). Diets enriched in saturated lipids have been found to be linked with white adipose tissue (WAT) inflammation, obesity, and insulin resistance in mice. When two groups of mice were fed with diet differing only in fat composition (lard or fish oil), the mice on saturated fat diet (lard) gained more weight, consumed more food, and had higher level of fasting glucose and insulin level compared to mice fed on fish oil. After 11 weeks, mice on lard diet showed increased TLR activation, high WAT inflammation, and decreased insulin sensitivity than that of fish oil-fed mice (Caesar et al. 2015). On analyzing the gut flora, it was observed that fish oil promotes the growth of Lactobacillus which has earlier been shown to reduce inflammation in IBD models in mice (Guarner et al. 2005). Fish oil also promotes the enrichment of Akkermansia mucinophila in gut flora, and this is thought to be associated with decrease in weight gain, infiltration of WAT by macrophages, and improvement of gut barrier function and glucose metabolism (Everard et al. 2013). Intake of high-fat diet leads to more secretion of bile acids (Reddy 1981). Higher concentration of bile acids creates conducive environment for growth of a number of bile-resistant gastrointestinal pathogens such as B. wadsworthia, Helicobacter hepaticus, and Listeria monocytogenes and protozoan parasites such as giardia, microsporidia, and cryptosporidia. Bacterial antigens and metabolic products of these bacteria such as H2S or secondary bile acids damage the gut mucosal barrier resulting in increased immune cell infiltration and damaged tissues. High levels of bile acids change microbial assemblage by promoting the growth of Bilophila wadsworthia, a sulfite-reducing pathobiont, known for exacerbating colitis by inducing Th-1 immune response in genetically susceptible mice IL10−/− (Devkota et al. 2012). The quantity and quality of dietary fat affect the composition of gut flora. The consumption of low-fat diet promotes the growth of Firmicutes and reduces the abundance of others. Intake of polyunsaturated (safflower oil) fat (PUFA) and saturated (milk-derived) fat diets (MF) increases the population of Bacteroidetes and decreases abundance of Firmicutes. MF uniquely promotes the growth of B. wadsworthia otherwise hard to detect in healthy individuals. The abundance of B. wadsworthia has been associated with appendicitis and other intestinal inflammatory disorders. This kind of alteration in gut flora may shift the balanced immune state toward chronic diseases in genetically susceptible hosts. High-fat and high-sugar Western diets had been linked with obesity, imbalanced glucose homeostasis, IBD, and other chronic inflammatory diseases in genetically susceptible hosts.
Cheese, seafood, eggs, and meat are considered to be rich sources of phospholipid, phosphatidylcholine, choline, and carnitine. Choline is an important constituent of cell membrane and is considered as a substrate for synthesis of acetylcholine and provides a pool of methyl groups during metabolism of amino acids methionine and homocysteine (Zeisel 2006). Choline is involved in the formation of very-low-density lipoprotein (VLDL) and lipid metabolism in the liver. Though choline is partially synthesized by humans, insufficient dietary consumption may lead to disturbed gut flora and nonalcoholic fatty liver disease (NAFLD) in mice (Henao-Mejia et al. 2012). Microbial biotransformation of choline into TMA in human gut decreases bioavailability of choline for the host. Choline deficiency has been linked with a number of liver diseases such as NAFLD, steatosis, and hepatocarcinomas. Hepatic steatosis phenotype is influenced by GM and can be characterized using metabolomic profiling (Corbin and Zeisel 2012). Changes in the relative levels of Gammaproteobacteria and Erysipelotrichia have been reported to be associated with NAFLD in humans (Spencer et al. 2011). In excessive phosphatidylcholine-rich diet, choline gets biotransformed at a higher rate because of bacterial TMA lyase activity into TMA in gut which further gets converted into TMAO in the liver by hepatic flavin monooxygenase 3 (FMNO3), thereby increasing the risk of atherosclerosis and cardiovascular diseases (Tang et al. 2013). Recently, it has been shown that DMB (3,3-dimethyl-1-butanol), a structural analog of choline, can act as an inhibitor of bacterial TMA lyase activity and thus can control the biotransformation of choline into TMA without any side effects, thereby reducing the risk of atherosclerosis (Wang et al. 2015).
21.4.4 Protein-Rich Diet
Proteins are a major constituent of a balanced diet. Our system is unable to synthesize a number of amino acids and thus need to be taken from dietary sources for sustenance. Absorption of fat and protein normally occurs in the small intestine, while gut bacteria are mainly located in the large intestine/colon. As undigested carbohydrates reach the large intestine, some proteins also reach into the colon where fermentation of protein by the gut microflora takes place. Some of main dominant proteolytic bacteria observed in stool samples are Bacteroides spp., Propionibacterium spp., Streptococcus, Clostridium, Bacillus, and Staphylococcus (Macfarlane et al. 1986). Association of Bacteroides enterotype has been reported in individuals consuming diet high in animal protein (De Filippo et al. 2010). Residual undigested proteins that enter the large intestine are fermented by Bacteroides and Clostridium into varied by-products based on the amino acid composition of the proteins (Macfarlane et al. 1992).
Microbial fermentation of proteins leads to formation of different gases as well as other metabolites increasing the nitrogenous substrate for the gut microbiota and consequently putrefactive fermentation products (Silvester and Cummings 1995). This process largely takes place in the colon region. Putrefactive fermentation has been identified as one of the main causes of common bowel diseases such as CRC and IBD. Among the beneficial properties of the gut bacteria, colonic health is the major outcome. When the diet consists of fermentable carbohydrate substrates, colonic bacteria grow optimally and contribute amino acids and proteins. It has been estimated that a substantial amount (1 and 20%) of amino acids, especially lysine and threonine derived from the gut flora, have been observed in circulating plasma of adults (Laparra and Sanz 2010; Compare et al. 2012). When the gut bacteria undergo proteolytic fermentation, it produces some beneficial compounds such as polyphenols that induce anti-inflammatory, anti-oxidative, and antiaging effects and promotes generation of a reasonable concentration of SCFAs. However, anaerobic metabolism of proteins known as putrefaction leads to the production of toxic substances including ammonia, hydrogen sulfide, amines, phenols, thiols, and indoles. These compounds are recognized as cytotoxins, genotoxins, and carcinogens (Hughes et al. 2000) and are considered to be detrimental for the host’s health (Cummings et al. 1979; Macfarlane et al. 1986; Compare et al. 2010; Lopez-Legarrea et al. 2014; Conlon and Bird 2015). Putrefactive fermentation is suggested to play a critical role in the development and progression of diseases like CRC and IBD in the distal colon (Macfarlane and Macfarlane 2012) (Toden et al. 2005). Western diets rich in red meat, milk, and eggs deliver higher amounts of sulfur compounds to the colon and hence favor sulfidogenic hydrogen disposal by GM (Conlon and Bird 2015). High level of sulfides in fecal samples is positively associated with high dietary protein consumption by humans. It has been shown in a mouse model that higher intake of red meat is associated with damage to colonic mucosa during shortage of fermentable dietary carbohydrates (Toden et al. 2007). Red meat is a rich source of amino acid l-carnitine. Recently it was shown that GM metabolizes dietary l-carnitine into TMA (trimethylamine) that later gets converted to trimethylamine-N-oxide (TMAO) by flavin monooxygenases in the liver. Increased TMAO levels have been associated with increase in atherosclerosis in mice and risk of cardiovascular diseases in humans. Microbiota of omnivorous human was found to be more efficient in producing TMAO from dietary carnitine than that of vegans or vegetarians (Koeth et al. 2013). Dietary intake of proteins may also affect drug bioavailability. Bioavailability of digoxin, a glycoside widely used as a cardiac drug in humans, is reduced in some patients due to excretion of inactive digoxin metabolite, dihydrodigoxin. It has been observed that the colonization by different strains of Eggerthella lenta and host diet acts together and affect bioavailability of digoxins. Pharmacokinetic studies in gnotobiotic mice have shown that dietary protein reduces in vivo microbial metabolism of digoxin by E. lenta, resulting in significant changes in the drug concentration both in the serum and urine (Haiser et al. 2013).
Large epidemiological studies have indicated an association of colorectal cancer (CRC) with the high consumption of red and processed meat (Norat et al. 2005). In animal models, significant changes were observed in the composition of the GMt with increase in age and high-fat-containing diet. However, the effect can be nullified if the diet is supplemented with high protein to sucrose ratio (Kiilerich et al. 2016). Similar findings were observed in another study where in rodents fed with high-fat diet, obesity was prevented with a supplement diet high in protein to sucrose ratio (Pichon et al. 2006; Freudenberg et al. 2013). It was also observed that a diet high in protein and low in carbohydrates affected the GMt and fatty acid profiles in obese individuals. After consumption of high-protein diet for about 4 weeks, there was an increase in branched-chain fatty acids with a simultaneous decrease in butyrate concentration. This was further confirmed by a decrease in Roseburia/Eubacterium numbers. Further, it was also noted that due to high intake of proteins and low carbohydrate, concentration of fiber-derived antioxidant phenolic acids decreased (Russell et al. 2011). The decrease in Roseburia/E. rectale population decreases the butyrate level in fecal SCFAs (Lopez-Legarrea et al. 2014). Controlled dietary studies in obese men on high-protein/low-carbohydrate diet for 3–4 weeks have shown reduction in population of Collinsella aerofaciens, E. rectale, Roseburia, and Bifidobacterium spp. (Duncan et al. 2007; Russell et al. 2011).
Zonulin is a marker of intestinal permeability. The plasma level of zonulin is reported to be higher in celiac disease and type 1 diabetes. The plasma level of zonulin has been reported to be inversely proportional to the amount of protein taken up through diet and is associated with diet composition (Zak-Golab et al. 2013). Bacteria and gluten cause zonulin levels to rise in all individuals, suggesting that gluten-free diet may prevent an individual from leaky gut condition.
Gluten proteins are mainly found in barley, wheat, and rye, and consumption of these causes celiac disease in genetically predisposed subjects. Gluten-degrading microorganisms in the GI tract have been identified as different species of Rothia possessing gliadin enzyme. This enzyme helps in the digestion of dietary gluten. Since this organism and its enzyme is capable of neutralizing the harmful effects of gluten in celiac disease patients, therefore, development of a novel therapy based on different species of Rothia is being considered (Zamakhchari et al. 2011). A gluten-free diet (GFD) reduces beneficial gut bacteria populations (Bifidobacterium, Lactobacillus, and B. longum) and the ability to stimulate host’s immunity in celiac disease patients. So, the impact of GFD on gut health needs to be taken under consideration in treatment of celiac disease patients (De Palma et al. 2009).
21.5 Effect of Different Dietary Patterns on the Microbiota
21.5.1 Vegetarian vs. Nonvegetarian Diet
The consumption of plant-based food increases taxonomic and bacterial gene diversity, leads to higher levels of short-chain fatty acid, and is linked with higher Prevotella/Bacteroides ratio. Refined foods from plant sources such as cereals and potatoes are not considered healthy food choice and are often linked with increased risk of metabolic and cardiovascular diseases (Fung et al. 2001; Willett et al. 2002). Adventist Health Studies (AHS-2), conducted by researchers at the Loma Linda University School of Public Health, provides an opportunity to examine the profile of microbiota of individuals consuming vegan, vegetarian, or nonvegetarian diets and also those who are omnivorous. Vegetarian diets in general were found to be more beneficial compared to omnivore diets; however, vegan diets confer an additional advantage in bringing down the odds ratio for developing type 2 diabetes (Tonstad et al. 2013). Bacteroidetes phylum was more abundant in vegans and vegetarians compared with omnivores (p < 0.05), and higher Firmicutes/Bacteroidetes ratio was predominantly observed in omnivores (De Filippis et al. 2015). Lachnospira and Prevotella were significantly associated to plant-based diets, whereas L-Ruminococcus (Ruminococcus genus assigned to Lachnospiraceae family) was positively associated to omnivore diets.
Further, individuals consuming vegan diets exhibited reduced inflammations and were less prone to female-specific cancer risks. However, therapeutic effect of vegan diet that can be prescribed for long-term health benefits needs to be established (Glick-Bauer and Yeh 2014). Examination of fecal samples of lacto-vegetarian and omnivore South Indian population showed enrichment of clostridium and butyrate-producing taxa in omnivores (Kabeerdoss et al. 2012). Consumption of animal-based diet is linked to higher deoxycholic acid (DCA) formation in gut. DCA, a secondary bile acid produced by the microbial action on bile in gut, promotes liver cancer. DCA is reported to decrease the growth of members of the Bacteroidetes and Firmicutes phyla while promoting the growth of a bile-resistant B. wadsworthia, a sulfite-reducing bacterium known to cause gut inflammation in mouse models (Yoshimoto et al. 2013).
21.5.2 Western Diet
Classical Western diet generally contains high amount of fat, animal proteins (red meat), and refined sugars/carbohydrates but low amount of fresh fruit, vegetables, and whole grain cereals (Hou et al. 2011). The intake of high-fat and high-sugar diet leads to reduction in microbial richness, taxonomic diversity, and metabolic activity of gut bacterial flora in comparison to low-fat and plant-based diet of agrarian societies (De Filippo et al. 2010). Such diet-induced changes in GMt due to consumption of Western diet along with sedentary lifestyle are thought to be responsible for high incidence of inflammatory diseases such as IBD and other metabolic diseases such as obesity, heart disease, and diabetes (Law 2000). Intake of dietary saturated fats is associated with progressive increase in population of pro-inflammatory gut microbes such as B. wadsworthia, Staphylococcus, E. coli, and members of Enterobacteriaceae. Recovery of healthy diverse GM may require long-term dietary intervention in individuals on Western diet.
21.5.3 Mediterranean Diet
The Mediterranean-style diet (MD) is an ensemble of traditional dietary habits of people living in different countries of the Mediterranean region (Cyprus, Croatia, Spain, Greece, Italy, Morocco, Portugal on the coast of the Mediterranean Sea) (Sofi et al. 2010). It is one of the healthiest dietary styles and has been listed in Intangible Cultural Heritage of Humanity of UNESCO (http://www.unesco.org/culture/ich/index.php?lg5en&pg50001). According to the European Food Safety Authority,
“Mediterranean diet is generally characterized by high consumption of vegetables, fruits, cereals (unprocessed, whole grain), legumes, nuts, and seeds; moderate consumption of dairy products (mostly in the form of cheese or yogurt), fish, poultry, eggs, and unsaturated fats, such as olive oil as the primary source of monounsaturated fat for cooking and dressing; low to moderate intake of wine during meals; and little intake of red, processed meats and saturated fats” (Trichopoulou et al. 2003).
Metagenomic studies conducted on individuals adhering to the MD pattern of diet exhibited increased number of genes associated with polysaccharide degradation and SCFA metabolism (De Filippis et al. 2015). A relationship between MD diet and TMAO level was also established in this study providing a valuable insight for a possible modulation of TMAO levels through diet. A study using Greek population showed that the Mediterranean diet significantly reduced overall mortality due to coronary heart disease and cancer, even after taking care of confounders like age, sex, body mass index, and physical activity level (Trichopoulou et al. 2003; Martinez-Gonzalez et al. 2012).
21.6 Longitudinal Effect of Diet: Effect of Short-Term and Long-Term Dietary Intervention
Short-term dietary interventions have been found to affect gut microbial composition and gene expression within 24 hours. The changes in gut microbial composition and gene expression were correlated with the changes in diet composition (plant- or animal-based diet). This dynamic and quick response of gut flora to diet is probably due to survival pressure on GMt because of day-to-day variation in food intake of individuals. However, it is also probable that there may be other reasons as short-term dietary intervention (10 days) failed to achieve any changes in enterotype (David et al. 2014). Despite rapid response of gut flora to the consumed diet, it is the long-term consumption that shapes microbial configuration and functionality of gut flora in an individual. Long-term consumption of diet strongly correlates with formation of gut microbial enterotypes, and some of the human health problems may be a consequence of variations in diets (Wu et al. 2011). The GMt of patients with inflammatory bowel disease (IBD), obese individuals, and elderly patients with inflammation is less diverse in comparison to that of healthy people (Le Chatelier et al. 2013). Though short-term dietary intervention improves gut microbial gene richness and clinical parameters in low gene content (LGC) individuals, complete recovery of gene richness is not achieved. However, permanent changes in gut microbial consortium can be achieved by long-term dietary intervention with appropriate diet (Wu et al. 2011; Cotillard et al. 2013). It is important to note here that due to individualized nature of GM, there is substantial variation in the gut flora in response to a particular diet and people can be categorized as responders and nonresponders. Responders show better metabolic functions than non-responders. So, any therapy based on dietary intervention designed to improve gut flora should identify the potential beneficiary first for optimum benefit (Kovatcheva-Datchary et al. 2015). It appears that human genotype may have a role in the type of GM and the non-responsiveness to dietary intervention phenotype seen in some individuals.
21.7 Tools being Used to Elucidate the Microbial Metabolites
Our quest to study various metabolic syndromes has significantly advanced, as more and more metagenomic studies have suggested that microbial metabolites significantly vary during disease conditions. LC-MS and nuclear magnetic resonance (NMR) are two important and complimentary molecular techniques that are being used to assess the potential microbial metabolites involved in health and disease (Wikoff et al. 2009). These techniques are suitable for analysis of complex samples due to their reproducibility and their wide dynamic range. Untargeted MS-based metabolomics can be employed in animal models to successfully assess the impact of GM on blood chemistry.
Tools such as CASINO (Community And Systems-level Interactive Optimization) are now available to quantify the metabolic changes due to alterations in the diet in the human gut. For example, with the help of CASINO, one can predict the responses of specific metabolites such as SCFAs to individual food. This is a comprehensive computational platform for understanding and analyzing GM through metabolic modeling and gives a precise information regarding intolerance of a person to a specific dietary component. This information can subsequently be used to design functional foods that can increase colonic levels of SCFAs. This model can also be used to assess changes in the level of amino acids in stool and serum in response to dietary intervention (Shoaie et al. 2015).
21.8 Limitation and Opportunities of Gut Flora Research
Despite of enormous progress made in gut flora research gaps exist in our understanding of the GM and its interaction with diet. Here we present some of the issues that have emerged from this review and need to be addressed. Advancement made in the detection and analysis of hundreds of microorganisms from the human gut using metagenomic analysis has enabled us to enumerate bacterial population and extrapolate functional features including metabolism being carried out in the gut. However, to get a clear insight into their role in disease conditions, we need to understand the role of the bacteria that are poorly represented in healthy condition but can amplify in a disease state leading to major changes in the harmful metabolic products released by them.
It is very important to understand the impact of diet in the maintenance of fine dynamic equilibrium between different phylotypes. It has been observed that diet low in carbohydrate but high in protein tilts the balance in favor of Bacteroidetes, whereas a diet rich in fiber but low in protein shows more abundance of Prevotella. Metagenomic analysis has shown that this leads to low gene content (LGC) in the former case with low diversity of microbiota, whereas high gene content (HGC) has been recorded in the latter case showing higher diversity of microbiota (Cotillard et al. 2013). This study needs to be further extended in order to understand the fluctuations in some useful bacteria, such as butyrate-producing bacteria.
The consumption of low-fiber diet increases the expression of host glycan (mucin) utilizing genes and the number of glycan utilizing generalists such as Bacteroides thetaiotaomicron (Sonnenburg et al. 2005). It also causes slow bowel movement, low production of SCFAs, and more calorie intake from other diet components such as fat, protein, and refined sugar and selects a microbial consortium enriched in mucus-utilizing gut bacteria. The subsequent metabolic changes have been associated with several diseases of Western world (Sonnenburg and Sonnenburg 2014). The long-term impact of low-fiber diet on the integrity of the gut mucosa, inflammation, erosion of mucus layer, and pathobiology of associated diseases calls for further investigation by detailed studies for better management of diseases through diet.
The gut mucosal integrity is of vital importance to human health; a better understanding of the role of GM in shaping our immune system is required. It is also known that dietary components help in the maintenance of barrier integrity by lowering the concentrations of some microbes that secrete various microbial products including toxins. These compounds function as stimulant causing damage to the tissue and result in inflammation. Further knowledge in this area would help to tailor specific diets that can reduce access of these toxins to the tissue.
Studies integrating the factors like environmental, host genetics, and diet in the development of microbial profile are limited. Research should focus on the impact of diet and the environmental factors on children with different genetic background so as to achieve an optimal diet for maintaining a healthy gut profile at later period of life. Variations in macro and micronutrients of diet have demonstrated high potential in the pathogenesis of a disease; however, their role in modulating gut flora is not yet well understood. Studies on the impact of macronutrients like carbohydrate, protein, and lipids on the GM have been initiated, and preliminary results have just started emerging on the sources of these macronutrients (animal or plant source) and their role modulating the gut flora. Information is lacking on the impact of micronutrients, such as specific vitamins in changing the gut profile.
So far most of the studies concentrated on profiling of gut flora of the large intestine due to more abundance of bacteria in this region. However, detail profiling of the bacterial flora of the small intestine is crucial so as to understand the mechanisms that allow digestion process in the small intestine. Higher rate of digestion in the small intestine will pass on the lesser level of undigested products to the large intestine increasing chances of complete digestion with minimal generation of harmful by-products. There is a need to conduct case-controlled studies supplemented with metabolic approaches to assess the physiological impact of identified microbial metabolites and decipher its specific mechanism of action.
Recently the role of gut-brain axis (GBA) has been proposed to have an important role in health and disease. Evidence of GBA-microbiota relationship has come to the surface since dysbiosis in GMt has been linked with diseases such as autism, multiple sclerosis, and IBS. Understanding the specific microbial products that reach the brain and how dietary manipulations of microbiota can impact our health is yet not convincingly established.
So far the knowledge in this field has clearly demonstrated that the predominant bacteria are crucial for maintaining a healthy gut. Now it is important to culture those bacteria and maintain a library of these isolates that can be delivered to diseased individuals in the form of prebiotics/probiotics as and when required for stimulating the growth of beneficial flora lost due to pathogenesis. This exercise also calls for a tailored diet in the form of prebiotics to maintain the healthy gut. High-throughput techniques must be employed for identification of a minimal set of biomarkers screened from microbial metabolites that are associated with the disease state.
Conclusion
Due to influence of diet, host genetics, and various environmental factors, healthy microbiota of an individual remains undefined. Changes in gut flora affect the development of several diseases such as obesity, diabetes, IBD, atherosclerosis, airway allergy, and multiple sclerosis as evident from animal studies; however, any interventions should be designed with a pinch of salt as alteration reported in animal studies may not be equally strong and reproducible in human subjects. However, it is noteworthy that several of these associations have shown promising results in IBD patients after fecal transplant. The human genome is relatively static, whereas gut microbiome is dynamic. This plasticity of genomic asymmetry between the human genome and gut microbiome gives an opportunity to take advantage of the plasticity of our gut residents for pragmatic and logical manipulation of gut flora to fortify human health and better management or prevention of various diseases. Remarkable progress in our understanding on diet-microbiota-host interactions has opened avenues to explore therapeutic approaches to manipulate the microbiota selectively. This can produce beneficial metabolite to promote health and prevent disease condition. However, whether changes in lifestyle, diet, and genetic predisposition all together or individually affect microbial profile in a disease state needs to be further investigated.
References
Aguirre M, Bussolo de Souza C, Venema K (2016) The gut microbiota from lean and obese subjects contribute differently to the fermentation of arabinogalactan and inulin. PLoS One 11. doi:10.1371/journal.pone.0159236
Arumugam M, Raes J, Pelletier E, Le Paslier D, Yamada T, Mende DR, Fernandes GR, Tap J, Bruls T, Batto JM, Bertalan M, Borruel N, Casellas F, Fernandez L, Gautier L, Hansen T, Hattori M, Hayashi T, Kleerebezem M, Kurokawa K, Leclerc M, Levenez F, Manichanh C, Nielsen HB, Nielsen T, Pons N, Poulain J, Qin J, Sicheritz-Ponten T, Tims S, Torrents D, Ugarte E, Zoetendal EG, Wang J, Guarner F, Pedersen O, de Vos WM, Brunak S, Doré J, MetaHIT Consortium, Antolín M, Artiguenave F, Blottiere HM, Almeida M, Brechot C, Cara C, Chervaux C, Cultrone A, Delorme C, Denariaz G, Dervyn R, Foerstner KU, Friss C, van de Guchte M, Guedon E, Haimet F, Huber W, van Hylckama-Vlieg J, Jamet A, Juste C, Kaci G, Knol J, Lakhdari O, Layec S, Le Roux K, Maguin E, Mérieux A, Melo Minardi R, M’rini C, Muller J, Oozeer R, Parkhill J, Renault P, Rescigno M, Sanchez N, Sunagawa S, Torrejon A, Turner K, Vandemeulebrouck G, Varela E, Winogradsky Y, Zeller G, Weissenbach J, Ehrlich SD, Bork P (2011) Enterotypes of the human gut microbiome. Nature 473:174–180. doi:10.1038/nature09944
Bergogne-Berezin E (2000) Treatment and prevention of antibiotic associated diarrhea. Int J Antimicrob Agents 16:521–526. doi:10.1016/S0924-8579(00)00293-4
Burgess SL, Buonomo E, Carey M, Cowardin C, Naylor C, Noor Z, Wills-Karp M, Petri WA Jr (2014) Bone marrow dendritic cells from mice with an altered microbiota provide interleukin 17A-dependent protection against Entamoeba histolytica colitis. MBio 5:e01817. doi:10.1128/mBio.01817-14
Caesar R, Tremaroli V, Kovatcheva-Datchary P, Cani PD, Backhed F (2015) Crosstalk between gut microbiota and dietary lipids aggravates WAT inflammation through TLR signaling. Cell Metab 22:658–668. doi:10.1016/j.cmet.2015.07.026
Cantarel BL, Lombard V, Henrissat B (2012) Complex carbohydrate utilization by the healthy human microbiome. PLoS One 7:e28742. doi:10.1371/journal.pone.0028742
Carmody RN, Gerber GK, Luevano JM Jr, Gatti DM, Somes L, Svenson KL, Turnbaugh PJ (2015) Diet dominates host genotype in shaping the murine gut microbiota. Cell Host Microbe 17:72–84. doi:10.1016/j.chom.2014.11.010
Chassard C, Delmas E, Robert C, Bernalier-Donadille A (2010) The cellulose-degrading microbial community of the human gut varies according to the presence or absence of methanogens. FEMS Microbiol Ecol 74:205–213. doi:10.1111/j.1574-6941.2010.00941.x
Claesson MJ, Jeffery IB, Conde S, Power SE, O’Connor EM, Cusack S, Harris HM, Coakley M, Lakshminarayanan B, O’Sullivan O, Fitzgerald GF, Deane J, O’Connor M, Harnedy N, O’Connor K, O’Mahony D, van Sinderen D, Wallace M, Brennan L, Stanton C, Marchesi JR, Fitzgerald AP, Shanahan F, Hill C, Ross RP, O’Toole PW (2012) Gut microbiota composition correlates with diet and health in the elderly. Nature 488:178–184. doi:10.1038/nature11319
Clemente JC, Pehrsson EC, Blaser MJ, Sandhu K, Gao Z, Wang B, Magris M, Hidalgo G, Contreras M, Noya-Alarcon O, Lander O, McDonald J, Cox M, Walter J, Oh PL, Ruiz JF, Rodriguez S, Shen N, Song SJ, Metcalf J, Knight R, Dantas G, Dominguez-Bello MG (2015) The microbiome of uncontacted Amerindians. Sci Adv 1:e1500183. doi:10.1126/sciadv.1500183
Compare D, Rocco A, Nardone G (2010) Risk factors in gastric cancer. Eur Rev Med Pharmacol Sci 14:302–308
Compare D, Coccoli P, Rocco A, Nardone OM, De Maria S, Carteni M, Nardone G (2012) Gut-liver axis: the impact of gut microbiota on non alcoholic fatty liver disease. Nutr Metab Cardiovasc Dis 22:471–476. doi:10.1016/j.numecd.2012.02.007
Conlon MA, Bird AR (2015) The impact of diet and lifestyle on gut microbiota and human health. Nutrients 7:17–44. doi:10.3390/nu7010017
Corbin KD, Zeisel SH (2012) Choline metabolism provides novel insights into nonalcoholic fatty liver disease and its progression. Curr Opin Gastroenterol 28:159–165. doi:10.1097/MOG.0b013e32834e7b4b
Cotillard A, Kennedy SP, Kong LC, Prifti E, Pons N, Le Chatelier E, Almeida M, Quinquis B, Levenez F, Galleron N, Gougis S, Rizkalla S, Batto JM, Renault P, ANR MicroObes Consortium, Doré J, Zucker JD, Clément K, Ehrlich SD (2013) Dietary intervention impact on gut microbial gene richness. Nature 500:585–588. doi:10.1038/nature12480
Cummings JH, Hill MJ, Bone ES, Branch WJ, Jenkins DJ (1979) The effect of meat protein and dietary fiber on colonic function and metabolism. II. Bacterial metabolites in feces and urine. Am J Clin Nutr 32:2094–2101
David LA, Maurice CF, Carmody RN, Gootenberg DB, Button JE, Wolfe BE, Ling AV, Devlin AS, Varma Y, Fischbach MA, Biddinger SB, Dutton RJ, Turnbaugh PJ (2014) Diet rapidly and reproducibly alters the human gut microbiome. Nature 505:559–563. doi:10.1038/nature12820
Davie JR (2003) Inhibition of histone deacetylase activity by butyrate. J Nutr 133:2485s–2493s
De Filippis F, Pellegrini N, Vannini L, Jeffery IB, La Storia A, Laghi L, Serrazanetti DI, Di Cagno R, Ferrocino I, Lazzi C, Turroni S, Cocolin L, Brigidi P, Neviani E, Gobbetti M, O’Toole PW, Ercolini D (2015) High-level adherence to a Mediterranean diet beneficially impacts the gut microbiota and associated metabolome. Gut. doi:10.1136/gutjnl-2015-309957
De Filippo C, Cavalieri D, Di Paola M, Ramazzotti M, Poullet JB, Massart S, Collini S, Pieraccini G, Lionetti P (2010) Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc Natl Acad Sci U S A 107:14691–14696. doi:10.1073/pnas.1005963107
De Palma G, Nadal I, Collado MC, Sanz Y (2009) Effects of a gluten-free diet on gut microbiota and immune function in healthy adult human subjects. Br J Nutr 102:1154–1160. doi:10.1017/S0007114509371767
De Vadder F, Kovatcheva-Datchary P, Goncalves D, Vinera J, Zitoun C, Duchampt A, Backhed F, Mithieux G (2014) Microbiota-generated metabolites promote metabolic benefits via gut-brain neural circuits. Cell 156:84–96. doi:10.1016/j.cell.2013.12.016
Dethlefsen L, Relman DA (2011) Incomplete recovery and individualized responses of the human distal gut microbiota to repeated antibiotic perturbation. Proc Natl Acad Sci U S A 108:4554–4561. doi:10.1073/pnas.1000087107
Devkota S, Wang Y, Musch MW, Leone V, Fehlner-Peach H, Nadimpalli A, Antonopoulos DA, Jabri B, Chang EB (2012) Dietary-fat-induced taurocholic acid promotes pathobiont expansion and colitis in Il10-/- mice. Nature 487:104–108. doi:10.1038/nature11225
Dominguez-Bello MG, Costello EK, Contreras M, Magris M, Hidalgo G, Fierer N, Knight R (2010) Delivery mode shapes the acquisition and structure of the initial microbiota across multiple body habitats in newborns. Proc Natl Acad Sci U S A 107:11971–11975. doi:10.1073/pnas.1002601107
Duncan SH, Holtrop G, Lobley GE, Calder AG, Stewart CS, Flint HJ (2004) Contribution of acetate to butyrate formation by human faecal bacteria. Br J Nutr 91:915–923. doi:10.1079/BJN20041150
Duncan SH, Belenguer A, Holtrop G, Johnstone AM, Flint HJ, Lobley GE (2007) Reduced dietary intake of carbohydrates by obese subjects results in decreased concentrations of butyrate and butyrate-producing bacteria in feces. Appl Environ Microbiol 73:1073–1078. doi:10.1128/AEM.02340-06
Eckburg PB, Bik EM, Bernstein CN, Purdom E, Dethlefsen L, Sargent M, Gill SR, Nelson KE, Relman DA (2005) Diversity of the human intestinal microbial flora. Science 308:1635–1638. doi:10.1126/science.1110591
El Kaoutari A, Armougom F, Gordon JI, Raoult D, Henrissat B (2013) The abundance and variety of carbohydrate-active enzymes in the human gut microbiota. Nat Rev Microbiol 11:497–504. doi:10.1038/nrmicro3050
Enattah NS, Sahi T, Savilahti E, Terwilliger JD, Peltonen L, Jarvela I (2002) Identification of a variant associated with adult-type hypolactasia. Nat Genet 30:233–237. doi:10.1038/ng826
Everard A, Belzer C, Geurts L, Ouwerkerk JP, Druart C, Bindels LB, Guiot Y, Derrien M, Muccioli GG, Delzenne NM, de Vos WM, Cani PD (2013) Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity. Proc Natl Acad Sci U S A 110:9066–9071. doi:10.1073/pnas.1219451110
Fallani M, Amarri S, Uusijarvi A, Adam R, Khanna S, Aguilera M, Gil A, Vieites JM, Norin E, Young D, Scott JA, Doré J, Edwards CA, INFABIO Team (2011) Determinants of the human infant intestinal microbiota after the introduction of first complementary foods in infant samples from five European centres. Microbiology 157:1385–1392. doi:10.1099/mic.0.042143-0
Feijo FD, Ballard CR, Foletto KC, Batista BAM, Neves AM, Ribeiro MFM, Bertoluci MC (2013) Saccharin and aspartame, compared with sucrose, induce greater weight gain in adult Wistar rats, at similar total caloric intake levels. Appetite 60:203–207. doi:10.1016/j.appet.2012.10.009
Fitch C, Keim KS (2012) Position of the academy of nutrition and dietetics: use of nutritive and nonnutritive sweeteners. J Acad Nutr Diet 112:739–758. doi:10.1016/j.jand.2012.03.009
Flint HJ, Scott KP, Louis P, Duncan SH (2012) The role of the gut microbiota in nutrition and health. Nat Rev Gastroenterol Hepatol 9:577–589. doi:10.1038/nrgastro.2012.156
Folseraas T, Melum E, Rausch P, Juran BD, Ellinghaus E, Shiryaev A, Laerdahl JK, Ellinghaus D, Schramm C, Weismuller TJ, Gotthardt DN, Hov JR, Clausen OP, Weersma RK, Janse M, Boberg KM, Björnsson E, Marschall HU, Cleynen I, Rosenstiel P, Holm K, Teufel A, Rust C, Gieger C, Wichmann HE, Bergquist A, Ryu E, Ponsioen CY, Runz H, Sterneck M, Vermeire S, Beuers U, Wijmenga C, Schrumpf E, Manns MP, Lazaridis KN, Schreiber S, Baines JF, Franke A, Karlsen TH (2012) Extended analysis of a genome-wide association study in primary sclerosing cholangitis detects multiple novel risk loci. J Hepatol 57:366–375. doi:10.1016/j.jhep.2012.03.031
Freudenberg A, Petzke KJ, Klaus S (2013) Dietary L-leucine and L-alanine supplementation have similar acute effects in the prevention of high-fat diet-induced obesity. Amino Acids 44:519–528. doi:10.1007/s00726-012-1363-2
Fukuda S, Toh H, Hase K, Oshima K, Nakanishi Y, Yoshimura K, Tobe T, Clarke JM, Topping DL, Suzuki T, Taylor TD, Itoh K, Kikuchi J, Morita H, Hattori M, Ohno H (2011) Bifidobacteria can protect from enteropathogenic infection through production of acetate. Nature 469:543–U791. doi:10.1038/nature09646
Fung TT, Willett WC, Stampfer MJ, Manson JE, Hu FB (2001) Dietary patterns and the risk of coronary heart disease in women. Arch Intern Med 161:1857–1862. doi:10.1001/archinte.161.15.1857
Gilchrist CA, Petri SE, Schneider BN, Reichman DJ, Jiang N, Begum S, Watanabe K, Jansen CS, Elliott KP, Burgess SL, Ma JZ, Alam M, Kabir M, Haque R, Petri WA Jr (2016) Role of the gut microbiota of children in diarrhea due to the protozoan parasite Entamoeba histolytica. J Infect Dis 213:1579–1585. doi:10.1093/infdis/jiv772
Glick-Bauer M, Yeh MC (2014) The health advantage of a vegan diet: exploring the gut microbiota connection. Nutrients 6:4822–4838. doi:10.3390/nu6114822
Goodrich JK, Waters JL, Poole AC, Sutter JL, Koren O, Blekhman R, Beaumont M, Van Treuren W, Knight R, Bell JT, Spector TD, Clark AG, Ley RE (2014) Human genetics shape the gut microbiome. Cell 159:789–799. doi:10.1016/j.cell.2014.09.053
Graf D, Di Cagno R, Fak F, Flint HJ, Nyman M, Saarela M, Watzl B (2015) Contribution of diet to the composition of the human gut microbiota. Microb Ecol Health Dis 26:26164. doi:10.3402/mehd.v26.26164
Guarner F, Perdigon G, Corthier G, Salminen S, Koletzko B, Morelli L (2005) Should yoghurt cultures be considered probiotic? Br J Nutr 93:783–786. doi:10.1079/BJN20051428
Haiser HJ, Gootenberg DB, Chatman K, Sirasani G, Balskus EP, Turnbaugh PJ (2013) Predicting and manipulating cardiac drug inactivation by the human gut bacterium Eggerthella lenta. Science 341:295–298. doi:10.1126/science.1235872
Hamer HM, Jonkers D, Venema K, Vanhoutvin S, Troost FJ, Brummer RJ (2008) Review article: the role of butyrate on colonic function. Aliment Pharmacol Ther 27:104–119. doi:10.1111/j.1365-2036.2007.03562.x
Harmsen HJ, Wildeboer-Veloo AC, Raangs GC, Wagendorp AA, Klijn N, Bindels JG, Welling GW (2000) Analysis of intestinal flora development in breast-fed and formula-fed infants by using molecular identification and detection methods. J Pediatr Gastroenterol Nutr 30:61–67
Hehemann JH, Correc G, Barbeyron T, Helbert W, Czjzek M, Michel G (2010) Transfer of carbohydrate-active enzymes from marine bacteria to Japanese gut microbiota. Nature 464:908–U123. doi:10.1038/nature08937
Henao-Mejia J, Elinav E, Jin C, Hao L, Mehal WZ, Strowig T, Thaiss CA, Kau AL, Eisenbarth SC, Jurczak MJ, Camporez JP, Shulman GI, Gordon JI, Hoffman HM, Flavell RA (2012) Inflammasome-mediated dysbiosis regulates progression of NAFLD and obesity. Nature 482:179–185. doi:10.1038/nature10809
Hogenauer C, Hammer HF, Krejs GJ, Reisinger EC (1998) Mechanisms and management of antibiotic-associated diarrhea. Clin Infect Dis 27:702–710. doi:10.1086/514958
Hou JK, Abraham B, El-Serag H (2011) Dietary intake and risk of developing inflammatory bowel disease: a systematic review of the literature. Am J Gastroenterol 106:563–573. doi:10.1038/ajg.2011.44
Hughes R, Magee EA, Bingham S (2000) Protein degradation in the large intestine: relevance to colorectal cancer. Curr Issues Intest Microbiol 1:51–58
Jeffery IB, Lynch DB, O’Toole PW (2016) Composition and temporal stability of the gut microbiota in older persons. ISME J 10:170–182. doi:10.1038/ismej.2015.88
Kabeerdoss J, Devi RS, Mary RR, Ramakrishna BS (2012) Faecal microbiota composition in vegetarians: comparison with omnivores in a cohort of young women in southern India. Br J Nutr 108:953–957. doi:10.1017/S0007114511006362
Kashyap PC, Marcobal A, Ursell LK, Smits SA, Sonnenburg ED, Costello EK, Higginbottom SK, Domino SE, Holmes SP, Relman DA, Knight R, Gordon JI, Sonnenburg JL (2013) Genetically dictated change in host mucus carbohydrate landscape exerts a diet-dependent effect on the gut microbiota. Proc Natl Acad Sci U S A 110:17059–17064. doi:10.1073/pnas.1306070110
Kiilerich P, Myrmel LS, Fjaere E, Hao Q, Hugenholtz F, Sonne SB, Derrien M, Pedersen LM, Petersen RK, Mortensen A, Licht TR, Rømer MU, Vogel UB, Waagbø LJ, Giallourou N, Feng Q, Xiao L, Liu C, Liaset B, Kleerebezem M, Wang J, Madsen L, Kristiansen K (2016) Effect of a long-term high-protein diet on survival, obesity development, and gut microbiota in mice. Am J Physiol Endocrinol Metab 310:E886–E899. doi:10.1152/ajpendo.00363.2015
Koeth RA, Wang Z, Levison BS, Buffa JA, Org E, Sheehy BT, Britt EB, Fu X, Wu Y, Li L, Smith JD, DiDonato JA, Chen J, Li H, Wu GD, Lewis JD, Warrier M, Brown JM, Krauss RM, Tang WH, Bushman FD, Lusis AJ, Hazen SL (2013) Intestinal microbiota metabolism of L-carnitine, a nutrient in red meat, promotes atherosclerosis. Nat Med 19:576–585. doi:10.1038/nm.3145
Kovatcheva-Datchary P, Nilsson A, Akrami R, Lee YS, De Vadder F, Arora T, Hallen A, Martens E, Bjorck I, Backhed F (2015) Dietary fiber-induced improvement in glucose metabolism is associated with increased abundance of Prevotella. Cell Metab 22:971–982. doi:10.1016/j.cmet.2015.10.001
Lam YY, Ha CWY, Hoffmann JMA, Oscarsson J, Dinudom A, Mather TJ, Cook DI, Hunt NH, Caterson ID, Holmes AJ, Storlien LH (2015) Effects of dietary fat profile on gut permeability and microbiota and their relationships with metabolic changes in mice. Obesity 23:1429–1439. doi:10.1002/oby.21122
Laparra JM, Sanz Y (2010) Interactions of gut microbiota with functional food components and nutraceuticals. Pharmacol Res 61:219–225. doi:10.1016/j.phrs.2009.11.001
Law M (2000) Dietary fat and adult diseases and the implications for childhood nutrition: an epidemiologic approach. Am J Clin Nutr 72:1291s–1296s
Le Chatelier E, Nielsen T, Qin J, Prifti E, Hildebrand F, Falony G, Almeida M, Arumugam M, Batto JM, Kennedy S, Leonard P, Li J, Burgdorf K, Grarup N, Jørgensen T, Brandslund I, Nielsen HB, Juncker AS, Bertalan M, Levenez F, Pons N, Rasmussen S, Sunagawa S, Tap J, Tims S, Zoetendal EG, Brunak S, Clément K, Doré J, Kleerebezem M, Kristiansen K, Renault P, Sicheritz-Ponten T, de Vos WM, Zucker JD, Raes J, Hansen T, MetaHIT Consortium, Bork P, Wang J, Ehrlich SD, Pedersen O (2013) Richness of human gut microbiome correlates with metabolic markers. Nature 500:541–546. doi:10.1038/nature12506
Ley RE, Peterson DA, Gordon JI (2006) Ecological and evolutionary forces shaping microbial diversity in the human intestine. Cell 124:837–848. doi:10.1016/j.cell.2006.02.017
Livanos AE, Greiner TU, Vangay P, Pathmasiri W, Stewart D, McRitchie S, Li H, Chung J, Sohn J, Kim S, Gao Z, Barber C, Kim J, Ng S, Rogers AB, Sumner S, Zhang XS, Cadwell K, Knights D, Alekseyenko A, Bäckhed F, Blaser MJ (2016) Antibiotic-mediated gut microbiome perturbation accelerates development of type 1 diabetes in mice. Nat Microbiol 1:16140. doi:10.1038/nmicrobiol.2016.140
Lopez-Legarrea P, Fuller NR, Zulet MA, Martinez JA, Caterson ID (2014) The influence of Mediterranean, carbohydrate and high protein diets on gut microbiota composition in the treatment of obesity and associated inflammatory state. Asia Pac J Clin Nutr 23:360–368. doi:10.6133/apjcn.2014.23.3.16
Lozupone CA, Stombaugh JI, Gordon JI, Jansson JK, Knight R (2012) Diversity, stability and resilience of the human gut microbiota. Nature 489:220–230. doi:10.1038/nature11550
Macfarlane S, Macfarlane GT (2003) Regulation of short-chain fatty acid production. Proc Nutr Soc 62:67–72. doi:10.1079/PNS2002207
Macfarlane GT, Macfarlane S (2012) Bacteria, colonic fermentation, and gastrointestinal health. J AOAC Int 95:50–60. doi:10.5740/jaoacint.SGE_Macfarlane
Macfarlane GT, Cummings JH, Allison C (1986) Protein degradation by human intestinal bacteria. J Gen Microbiol 132:1647–1656. doi:10.1099/00221287-132-6-1647
Macfarlane GT, Gibson GR, Cummings JH (1992) Comparison of fermentation reactions in different regions of the human colon. J Appl Bacteriol 72:57–64. doi:10.1111/j.1365-2672.1992.tb04882.x
Mahana D, Trent CM, Kurtz ZD, Bokulich NA, Battaglia T, Chung J, Muller CL, Li H, Bonneau RA, Blaser MJ (2016) Antibiotic perturbation of the murine gut microbiome enhances the adiposity, insulin resistance, and liver disease associated with high-fat diet. Genome Med 8:48. doi:10.1186/s13073-016-0297-9
Martinez I, Stegen JC, Maldonado-Gomez MX, Eren AM, Siba PM, Greenhill AR, Walter J (2015) The gut microbiota of rural papua new guineans: composition, diversity patterns, and ecological processes. Cell Rep 11:527–538. doi:10.1016/j.celrep.2015.03.049
Martinez-Gonzalez MA, Guillen-Grima F, De Irala J, Ruiz-Canela M, Bes-Rastrollo M, Beunza JJ, del Burgo CL, Toledo E, Carlos S, Sanchez-Villegas A (2012) The Mediterranean diet is associated with a reduction in premature mortality among middle-aged adults. J Nutr 142:1672–1678. doi:10.3945/jn.112.162891
Maslowski KM, Vieira AT, Ng A, Kranich J, Sierro F, Yu D, Schilter HC, Rolph MS, Mackay F, Artis D, Xavier RJ, Teixeira MM, Mackay CR (2009) Regulation of inflammatory responses by gut microbiota and chemoattractant receptor GPR43. Nature 461:1282–U1119. doi:10.1038/nature08530
McGovern DPB, Jones MR, Taylor KD, Marciante K, Yan XF, Dubinsky M, Ippoliti A, Vasiliauskas E, Berel D, Derkowski C, Dutridge D, Fleshner P, Shih DQ, Melmed G, Mengesha E, King L, Pressman S, Haritunians T, Guo X, Targan SR, Rotter JI, International IBD Genetics Consortium (2010) Fucosyltransferase 2 (FUT2) non-secretor status is associated with Crohn’s disease. Hum Mol Genet 19:3468–3476. doi:10.1093/hmg/ddq248
Milner JA, Allison RG (1999) The role of dietary fat in child nutrition and development: summary of an ASNS workshop. American Society for Nutritional Sciences. J Nutr 129:2094–2105
Modi SR, Collins JJ, Relman DA (2014) Antibiotics and the gut microbiota. J Clin Invest 124:4212–4218. doi:10.1172/JCI72333
Morton ER, Lynch J, Froment A, Lafosse S, Heyer E, Przeworski M, Blekhman R, Segurel L (2015) Variation in rural African gut microbiota is strongly correlated with colonization by Entamoeba and subsistence. PLoS Genet 11:e1005658. doi:10.1371/journal.pgen.1005658
Moschen AR, Wieser V, Tilg H (2012) Dietary factors: major regulators of the Gut’s microbiota. Gut Liver 6:411–416. doi:10.5009/gnl.2012.6.4.411
Norat T, Bingham S, Ferrari P, Slimani N, Jenab M, Mazuir M, Overvad K, Olsen A, Tjonneland A, Clavel F, Boutron-Ruault MC, Kesse E, Boeing H, Bergmann MM, Nieters A, Linseisen J, Trichopoulou A, Trichopoulos D, Tountas Y, Berrino F, Palli D, Panico S, Tumino R, Vineis P, Bueno-de-Mesquita HB, Peeters PH, Engeset D, Lund E, Skeie G, Ardanaz E, González C, Navarro C, Quirós JR, Sanchez MJ, Berglund G, Mattisson I, Hallmans G, Palmqvist R, Day NE, Khaw KT, Key TJ, San Joaquin M, Hémon B, Saracci R, Kaaks R, Riboli E (2005) Meat, fish, and colorectal cancer risk: the European Prospective Investigation into cancer and nutrition. J Natl Cancer Inst 97:906–916. doi:10.1093/jnci/dji164
Pichon L, Huneau JF, Fromentin G, Tome D (2006) A high-protein, high-fat, carbohydrate-free diet reduces energy intake, hepatic lipogenesis, and adiposity in rats. J Nutr 136:1256–1260
Qin J, Li R, Raes J, Arumugam M, Burgdorf KS, Manichanh C, Nielsen T, Pons N, Levenez F, Yamada T, Mende DR, Li J, Xu J, Li S, Li D, Cao J, Wang B, Liang H, Zheng H, Xie Y, Tap J, Lepage P, Bertalan M, Batto JM, Hansen T, Le Paslier D, Linneberg A, Nielsen HB, Pelletier E, Renault P, Sicheritz-Ponten T, Turner K, Zhu H, Yu C, Li S, Jian M, Zhou Y, Li Y, Zhang X, Li S, Qin N, Yang H, Wang J, Brunak S, Doré J, Guarner F, Kristiansen K, Pedersen O, Parkhill J, Weissenbach J, MetaHIT Consortium, Bork P, Ehrlich SD, Wang J (2010) A human gut microbial gene catalogue established by metagenomic sequencing. Nature 464:59–65. doi:10.1038/nature08821
Reddy BS (1981) Diet and excretion of bile acids. Cancer Res 41:3766–3768
Rombeau JL, Kripke SA (1990) Metabolic and intestinal effects of short-chain fatty-acids. JPEN J Parenter Enteral Nutr 14:S181–S185. doi:10.1177/014860719001400507
Russell WR, Gratz SW, Duncan SH, Holtrop G, Ince J, Scobbie L, Duncan G, Johnstone AM, Lobley GE, Wallace RJ, Duthie GG, Flint HJ (2011) High-protein, reduced-carbohydrate weight-loss diets promote metabolite profiles likely to be detrimental to colonic health. Am J Clin Nutr 93:1062–1072. doi:10.3945/ajcn.110.002188
Samuel BS, Shaito A, Motoike T, Rey FE, Backhed F, Manchester JK, Hammer RE, Williams SC, Crowley J, Yanagisawa M, Gordon JI (2008) Effects of the gut microbiota on host adiposity are modulated by the short-chain fatty-acid binding G protein-coupled receptor, Gpr41. Proc Natl Acad Sci U S A 105:16767–16772. doi:10.1073/pnas.0808567105
Schmid A (2011) The role of meat fat in the human diet. Crit Rev Food Sci Nutr 51:50–66. doi:10.1080/10408390903044636
Schnorr SL, Candela M, Rampelli S, Centanni M, Consolandi C, Basaglia G, Turroni S, Biagi E, Peano C, Severgnini M, Fiori J, Gotti R, De Bellis G, Luiselli D, Brigidi P, Mabulla A, Marlowe F, Henry AG, Crittenden AN (2014) Gut microbiome of the Hadza hunter-gatherers. Nat Commun 5:3654. doi:10.1038/ncomms4654
Shoaie S, Ghaffari P, Kovatcheva-Datchary P, Mardinoglu A, Sen P, Pujos-Guillot E, de Wouters T, Juste C, Rizkalla S, Chilloux J, Hoyles L, Nicholson JK, MICRO-Obes Consortium, Dore J, Dumas ME, Clement K, Bäckhed F, Nielsen J (2015) Quantifying diet-induced metabolic changes of the human gut microbiome. Cell Metab 22:320–331. doi:10.1016/j.cmet.2015.07.001
Silvester KR, Cummings JH (1995) Does digestibility of meat protein help explain large-bowel cancer risk. Nutr Cancer 24:279–288. doi:10.1080/01635589509514417
Sofi F, Abbate R, Gensini GF, Casini A (2010) Accruing evidence on benefits of adherence to the Mediterranean diet on health: an updated systematic review and meta-analysis. Am J Clin Nutr 92:1189–1196. doi:10.3945/ajcn.2010.29673
Sonnenburg ED, Sonnenburg JL (2014) Starving our microbial self: the deleterious consequences of a diet deficient in microbiota-accessible carbohydrates. Cell Metab 20:779–786. doi:10.1016/j.cmet.2014.07.003
Sonnenburg JL, Xu J, Leip DD, Chen CH, Westover BP, Weatherford J, Buhler JD, Gordon JI (2005) Glycan foraging in vivo by an intestine-adapted bacterial symbiont. Science 307:1955–1959. doi:10.1126/science.1109051
Spencer MD, Hamp TJ, Reid RW, Fischer LM, Zeisel SH, Fodor AA (2011) Association between composition of the human gastrointestinal microbiome and development of fatty liver with choline deficiency. Gastroenterology 140:976–986. doi:10.1053/j.gastro.2010.11.049
Stark PL, Lee A (1982) The microbial ecology of the large bowel of breast-fed and formula-fed infants during the first year of life. J Med Microbiol 15:189–203. doi:10.1099/00222615-15-2-189
Suez J, Korem T, Zeevi D, Zilberman-Schapira G, Thaiss CA, Maza O, Israeli D, Zmora N, Gilad S, Weinberger A, Kuperman Y, Harmelin A, Kolodkin-Gal I, Shapiro H, Halpern Z, Segal E, Elinav E (2014) Artificial sweeteners induce glucose intolerance by altering the gut microbiota. Nature 514:181–186. doi:10.1038/nature13793
Swithers SE (2013) Artificial sweeteners produce the counterintuitive effect of inducing metabolic derangements. Trends Endocrinol Metab 24:431–441. doi:10.1016/j.tem.2013.05.005
Swithers SE, Davidson TL (2008) A role for sweet taste: calorie predictive relations in energy regulation by rats. Behav Neurosci 122:161–173. doi:10.1037/0735-7044.122.1.161
Swithers SE, Martin AA, Davidson TL (2010) High-intensity sweeteners and energy balance. Physiol Behav 100:55–62. doi:10.1016/j.physbeh.2009.12.021
Tang WHW, Wang ZE, Levison BS, Koeth RA, Britt EB, Fu XM, Wu YP, Hazen SL (2013) Intestinal microbial metabolism of phosphatidylcholine and cardiovascular risk. N Engl J Med 368:1575–1584. doi:10.1056/NEJMoa1109400
Terpend K, Possemiers S, Daguet D, Marzorati M (2013) Arabinogalactan and fructo-oligosaccharides have a different fermentation profile in the Simulator of the Human Intestinal Microbial Ecosystem (SHIME (R)). Environ Microbiol Rep 5:595–603. doi:10.1111/1758-2229.12056
Thorburn AN, McKenzie CI, Shen S, Stanley D, Macia L, Mason LJ, Roberts LK, Wong CHY, Shim R, Robert R, Chevalier N, Tan JK, Mariño E, Moore RJ, Wong L, McConville MJ, Tull DL, Wood LG, Murphy VE, Mattes J, Gibson PG, Mackay CR (2015) Evidence that asthma is a developmental origin disease influenced by maternal diet and bacterial metabolites. Nat Commun 6:7320. doi:10.1038/ncomms8320
Toden S, Bird AR, Topping DL, Conlon MA (2005) Resistant starch attenuates colonic DNA damage induced by higher dietary protein in rats. Nutr Cancer 51:45–51. doi:10.1207/s15327914nc5101_7
Toden S, Bird AR, Topping DL, Conlon MA (2007) High red meat diets induce greater numbers of colonic DNA double-strand breaks than white meat in rats: attenuation by high-amylose maize starch. Carcinogenesis 28:2355–2362. doi:10.1093/carcin/bgm216
Tolhurst G, Heffron H, Lam YS, Parker HE, Habib AM, Diakogiannaki E, Cameron J, Grosse J, Reimann F, Gribble FM (2012) Short-chain fatty acids stimulate glucagon-like peptide-1 secretion via the G-protein-coupled receptor FFAR2. Diabetes 61:364–371. doi:10.2337/db11-1019
Tonstad S, Stewart K, Oda K, Batech M, Herring RP, Fraser GE (2013) Vegetarian diets and incidence of diabetes in the Adventist Health Study-2. Nutr Metab Cardiovasc Dis 23:292–299. doi:10.3945/ajcn.113.071233
Torres MF, Uetanabaro AP, Costa AF, Alves CA, Farias LM, Bambirra EA, Penna FJ, Vieira EC, Nicoli JR (2000) Influence of bacteria from the duodenal microbiota of patients with symptomatic giardiasis on the pathogenicity of Giardia duodenalis in gnotoxenic mice. J Med Microbiol 49:209–215. doi:10.1099/0022-1317-49-3-209
Tremaroli V, Backhed F (2012) Functional interactions between the gut microbiota and host metabolism. Nature 489:242–249. doi:10.1038/nature11552
Trichopoulou A, Costacou T, Bamia C, Trichopoulos D (2003) Adherence to a Mediterranean diet and survival in a Greek population. N Engl J Med 348:2599–2608. doi:10.1056/NEJMoa025039
Trompette A, Gollwitzer ES, Yadava K, Sichelstiel AK, Sprenger N, Ngom-Bru C, Blanchard C, Junt T, Nicod LP, Harris NL, Marsland BJ (2014) Gut microbiota metabolism of dietary fiber influences allergic airway disease and hematopoiesis. Nat Med 20:159–166. doi:10.1038/nm.3444
Verma AK, Verma R, Ahuja V, Paul J (2012) Real-time analysis of gut flora in Entamoeba histolytica infected patients of Northern India. BMC Microbiol 12:183. doi:10.1186/1471-2180-12-183
Walker AW, Ince J, Duncan SH, Webster LM, Holtrop G, Ze XL, Brown D, Stares MD, Scott P, Bergerat A, Louis P, McIntosh F, Johnstone AM, Lobley GE, Parkhill J, Flint HJ (2011) Dominant and diet-responsive groups of bacteria within the human colonic microbiota. ISME J 5:220–230. doi:10.1038/ismej.2010.118
Wang Z, Roberts AB, Buffa JA, Levison BS, Zhu W, Org E, Gu X, Huang Y, Zamanian-Daryoush M, Culley MK, DiDonato AJ, Fu X, Hazen JE, Krajcik D, DiDonato JA, Lusis AJ, Hazen SL (2015) Non-lethal inhibition of gut microbial trimethylamine production for the treatment of atherosclerosis. Cell 163:1585–1595. doi:10.1016/j.cell.2015.11.055
Wang QP, Lin YQ, Zhang L, Wilson YA, Oyston LJ, Cotterell J, Qi Y, Khuong TM, Bakhshi N, Planchenault Y, Browman DT, Lau MT, Cole TA, Wong AC, Simpson SJ, Cole AR, Penninger JM, Herzog H, Neely GG (2016) Sucralose promotes food intake through NPY and a neuronal fasting response. Cell Metab 24:75–90. doi:10.1016/j.cmet.2016.06.010
Whitman WB, Coleman DC, Wiebe WJ (1998) Prokaryotes: the unseen majority. Proc Natl Acad Sci U S A 95:6578–6583
Wikoff WR, Anfora AT, Liu J, Schultz PG, Lesley SA, Peters EC, Siuzdak G (2009) Metabolomics analysis reveals large effects of gut microflora on mammalian blood metabolites. Proc Natl Acad Sci U S A 106:3698–3703. doi:10.1073/pnas.0812874106
Willett W, Manson J, Liu SM (2002) Glycemic index, glycemic load, and risk of type 2 diabetes. Am J Clin Nutr 76:274s–280s
Wold AE (1998) The hygiene hypothesis revised: is the rising frequency of allergy due to changes in the intestinal flora? Allergy 53:20–25
Wu GD, Chen J, Hoffmann C, Bittinger K, Chen YY, Keilbaugh SA, Bewtra M, Knights D, Walters WA, Knight R, Sinha R, Gilroy E, Gupta K, Baldassano R, Nessel L, Li H, Bushman FD, Lewis JD (2011) Linking long-term dietary patterns with gut microbial enterotypes. Science 334:105–108. doi:10.1126/science.1208344
Yatsunenko T, Rey FE, Manary MJ, Trehan I, Dominguez-Bello MG, Contreras M, Magris M, Hidalgo G, Baldassano RN, Anokhin AP, Heath AC, Warner B, Reeder J, Kuczynski J, Caporaso JG, Lozupone CA, Lauber C, Clemente JC, Knights D, Knight R, Gordon JI (2012) Human gut microbiome viewed across age and geography. Nature 486:222–227. doi:10.1038/nature11053
Yoshimoto S, Loo TM, Atarashi K, Kanda H, Sato S, Oyadomari S, Iwakura Y, Oshima K, Morita H, Hattori M, Honda K, Ishikawa Y, Hara E, Ohtani N (2013) Obesity-induced gut microbial metabolite promotes liver cancer through senescence secretome. Nature 499:97–101. doi:10.1038/nature12347
Yoshioka H, Iseki K, Fujita K (1983) Development and differences of intestinal flora in the neonatal period in breast-fed and bottle-fed infants. Pediatrics 72:317–321
Zak-Golab A, Kocelak P, Aptekorz M, Zientara M, Juszczyk L, Martirosian G, Chudek J, Olszanecka-Glinianowicz M (2013) Gut microbiota, microinflammation, metabolic profile, and zonulin concentration in obese and normal weight subjects. Int J Endocrinol 2013:674106. doi:10.1155/2013/674106
Zamakhchari M, Wei GX, Dewhirst F, Lee J, Schuppan D, Oppenheim FG, Helmerhorst EJ (2011) Identification of Rothia bacteria as gluten-degrading natural colonizers of the upper gastro-intestinal tract. PLoS One 6:e24455. doi:10.1371/journal.pone.0024455
Zeisel SH (2006) Choline: critical role during fetal development and dietary requirements in adults. Annu Rev Nutr 26:229–250. doi:10.1146/annurev.nutr.26.061505.111156
Zhou D, Zhang H, Bai Z, Zhang A, Bai F, Luo X, Hou Y, Ding X, Sun B, Sun X, Ma N, Wang C, Dai X, Lu Z (2016) Exposure to soil, house dust and decaying plants increases gut microbial diversity and decreases serum immunoglobulin E levels in BALB/c mice. Environ Microbiol 18:1326–1337. doi:10.1111/1462-2920.12895
Zoetendal EG, Akkermans ADL, Akkermans-van Vliet WM, de Visser JAGM, de Vos WM (2001) The host genotype affects the bacterial community in the human gastrointestinal tract. Microb Ecol Health Dis 13:129–134
Acknowledgments
The authors wish to acknowledge the financial support received from the DBT-European Union Grant (BT/IN/EU/Infect-Era/01/AB/2015). AB acknowledges support from the Department of Science & Technology for J.C. Bose fellowship.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Verma, A.K., Kumari, R., Bhattacharya, A., Paul, J. (2017). Dietary Impacts on the Composition of Microbiota in Human Health and Disease. In: Kalia, V., Shouche, Y., Purohit, H., Rahi, P. (eds) Mining of Microbial Wealth and MetaGenomics. Springer, Singapore. https://doi.org/10.1007/978-981-10-5708-3_21
Download citation
DOI: https://doi.org/10.1007/978-981-10-5708-3_21
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-10-5707-6
Online ISBN: 978-981-10-5708-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)