Abstract
The quality and quantity of rotifer production in a hatchery primarily determine the yield of larval production; therefore, considerable studies geared toward mass culture and preservation of rotifers. Rotifer mass production, attaining ultrahigh density and greater culture stability, was attained when condensed microalgal pastes (Chlorella and Nannochloropsis) were introduced and used in a closed recirculation system. The rotifer culture health status can be determined by measuring water quality, rotifer physiological conditions such as egg ratio, swimming speed, ingestion rate, and in vivo enzyme that takes 1–2 days to finish. Addition of chemicals and hormones such as γ-aminobutyric acid (GABA), porcine growth hormone, serotonin, and human chorionic gonadotropin is known to improve the health status of cultured rotifers. Mass-produced rotifers can be preserved at low temperature; these rotifers can either be directly used as feed for the larvae or as starters of rotifer culture. For long-term storage, collection and storage of resting eggs are recommended. For species which do not produce resting eggs, cryopreservation of amictic eggs is recommended.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
1 Introduction
The rotifer Brachionus plicatilis species complex is being used worldwide as a live food for the initial stages of larval rearing of fish and shellfish because of their suitable size and shape. Based on morphometric characteristics, rotiferologists have classified B. plicatilis into three morphotypes (Fu et al. 1991): L-type (130–340 μm), S-type (100–210 μm), and SS-type (90–150 μm; see review Hagiwara et al. 1995, 2001). This classification is supported by biochemical and ecological differences (Snell 1998), including mating behavior (based on 29-kDa glycoprotein sex pheromone; see Snell et al. 1995), hence, assigned a name to distinguish these different species as B. plicatilis for L-type, B. ibericus for S-type, and B. rotundiformis for SS-type (Kotani et al. 2005). However, this physiology-based classification is considered a difficult approach with weak boundary species (Kotani et al. 1997); thus, molecular phylogeny was adapted to classify rotifer species complex. Several cryptic species were found within B. plicatilis sp. complex using ITS1and COI sequences (Gómez et al. 2002), and to date, 15 different species are classified (Mills et al. 2016). Among rotifers, this taxon is well studied because of its ease in culture and high reproductive performance and thus commonly used in fish and crustacean hatcheries (Hagiwara et al. 2007). The quality and quantity of rotifer production in a hatchery primarily determine the yield of larval production; therefore, considerable attention and researches are being done in the mass culture and preservation of this species complex (Hagiwara 1994; Lubzens and Zmora 2003).
Mass production of B. plicatilis sp. complex was initiated in the early 1960s with the use of Nannochloropsis oculata as diet (Hirata 1964). Thereafter, rotifer mass culture trials were conducted using various diets such as baker’s yeast, marine yeast, bacteria, omega yeast, and Tetraselmis tetrathele (reviewed by Nagata and Hirata 1986). Rotifer mass production has been systematized with the combination of improved diet formulation (Yoshimatsu and Hossain 2014), and rotifer densities reached to 2.3 × 104 ind/mL (Dhert et al. 2001). However, in spite of these advancements, fresh phytoplankton and/or baker’s yeast is still generally used for the mass culture of rotifers worldwide. The use of baker’s yeast is beneficial for aquaculturists since it lowers the cost for rotifer production. However, rotifer cultures fed yeast are less stable owing to the rapid decline of water quality. Moreover, rotifers fed yeast need further nutritional enrichment before being fed to fish larvae; therefore, phytoplankton remains the ideal food for rotifer cultures (Lubzens et al. 1995a). However, culturing of phytoplankton requires considerable space in a hatchery. Condensed microalgal paste was found to be the ideal food for ultrahigh production of rotifers. During the late 1980s, a phytoplankton industry has developed new products such as condensed microalgal paste (Chlorella and Nannochloropsis), which enabled culturists to mass culture rotifers at a high density as much as 1.6 × 105 ind/mL (Lubzens et al. 1995a; Yoshimura et al. 1996, 2003; Hagiwara and Kuwada 2004) with higher stability of culture. Furthermore, it was the most convenient, since it could be stored at low temperature (refrigerator or freezer) for at least 2 months or even longer without significant loss of its essential fatty acid content and maintaining its nutrient composition in cultured rotifers (Lubzens et al. 1995a; Welladsen et al. 2014). However, condensed microalgal pastes are not commercially available in developing countries because of higher price. Recently, detritus from macroalga, Ulva pertusa, was shown to sustain high population growth and enhance the nutritional value of B. plicatilis (Yin et al. 2013). Detrital macroalgae can also be stored and collected from the wild; thus, it is considered economical in large-scale production of rotifers.
In this chapter, various culture methods commonly used for mass culture of clonal rotifer B. plicatilis sp. complex are discussed. Since the major problem encountered by aquaculturists is the unpredictable culture collapse, some of the techniques used to diagnose and treat rotifer culture are presented. To address the inadequate supply of larval food, some preservation methods that have been proposed by many researchers are also discussed.
2 Culture Methods
2.1 Batch Culture
Batch culture is a common method for rotifer mass production in marine fish hatcheries (Fig. 3.1a). The culture maintains a constant volume with an increasing rotifer density or a constant rotifer density by increasing the culture volume (Dhert et al. 2001). When the desired density is achieved, the culture is entirely harvested at once by draining the culture medium and collecting the rotifers in a net. The harvested rotifers are used to feed the larvae, and the remaining is used as the inoculum for the next culture (Lubzens 1987; Hino 1993; Dhert et al. 2001). The size of culture vessels is flexible: 500–1000 L for plastic tanks or up to 10 ton for concrete tanks. In case of S-type rotifers, the obtainable densities at harvest time are over 1000 ind/mL when fed with Nannochloropsis oculata and/or backer’s yeast (Hino 1993) and 600 ind/mL when fed with artificial diet (Culture Selco®) (Suantika et al. 2000). This method is used in many countries; however, it has many disadvantages, including low efficiency in terms of labor and utilization of infrastructure, low production yield, unstable and unpredictable culture, and high costs of operation (Dhert et al. 2001).
Various mass culture systems of rotifer Brachionus plicatilis sp. complex. (a) Batch culture, (b) semicontinuous culture, and high-density culture including (c) automatic continuous and (d) closed recirculation systems. Normal, dot, and double head arrows indicate major inflows of materials, fresh medium, and rotifer harvest line, respectively
2.2 Semicontinuous Culture
The semicontinuous culture method is also known as “thinning out” culture, because the rotifer density is maintained constant by periodic harvesting (Hino 1993). The size of the culture tank is usually larger (usually between 20 and 480 tons) than that used in batch culture. The initial density of rotifers inoculated into the system varies between 50 and 200 ind/mL and might reach 300 to over 1000 ind/mL in 3–7 days when fed fresh microalgae and/or baker’s yeast (Dhert et al. 2001). Unlike in the batch culture method, in this system, a fixed fraction of culture water (which contains rotifers and residual food) is harvested at regular intervals and replaced by an equal quantity of fresh culture medium (Fig.3.1b). The dilution rate determines the rotifer population dynamics. Monod kinetics and related mathematical concepts are used to model the quasi-steady state of periodically diluted cultures used in this system (Schlüter et al. 1987). Navarro and Yufera (1998) used the quasi-steady state method and freeze-dried microalgae and used dilution rates of 0.3/day and 0.2/day, which resulted in the best production (mg rotifer/day) and food conversion efficiency (mg rotifer developed/mg microalgae consumed) for L- and S-type rotifers, respectively.
2.3 High-Density Culture
An intensive mass culture system for rotifers was developed by Japanese scientists in the late 1990s. In the culture trial with the use of concentrated freshwater Chlorella diet, an ultrahigh S-type rotifer density ranging from 10,000 to 30,000 ind/mL was obtained (Yoshimura et al. 1996). Under this batch, culture method, culture instability due to ammonia accumulation, presence of bacteria/protozoa, and food shortage or oxygen decline, however, are often encountered (Hagiwara and Kuwada 2004). To solve these problems, Yoshimura et al. (2003) employed several remedies including oxygen gas supplementation, regulation of pH (adjusted to 7), and a filtering equipment which prevents particulate organic matter, debris, and bacteria from clogging the collection net during harvest. In addition, Fu et al. (1997) introduced an automatic continuous culture system as the solution to maintain culture stability, as well as to reduce labor and create more space in the hatchery (Fig. 3.1c). This system consists of a filtration unit, culture unit, and harvest unit. Filtered water and food are continuously supplied into the rotifer culture tank at a predetermined rate, and the same amount of culture water is transferred into the harvest tank to obtain a significant rotifer biomass. By using this system, Fu et al. (1997) successfully mass produced about 2.1 billion rotifers/day for S-type in a 1-m3 tank with densities ranging from 3000 to 6000 ind/mL and about 0.17 billion rotifers/day for L-type in a 500-L tank with densities ranging from 1100 to 2200 ind/mL. The longest duration of their culture was over 110 days. Hagiwara and Kuwada (2004) described continuous rotifer culture system using a large tank (20 ton), which uses facilities in old hatcheries. Commercially available concentrated freshwater Chlorella vulgaris was used as feed for both species. In order to achieve high-density rotifers, a closed recirculation culture system (Fig. 3.1d) was lastly performed. This system comprised of protein skimmers, novel filters, ozone, and an addition of sodium hydroxymethanesulfonate to neutralize ammonia in the tank. With these modifications, ultrahigh rotifer densities (over 5000 ind/mL) were maintained for longer culture period (up to 30 days) without compromising the water quality (Suantika et al. 2000, 2001; Bentley et al. 2008). The development of a high-density rotifer culture system significantly reduced the space needed at the hatchery for live food production, allowing the production of sufficient amount of rotifers even in small tanks, improved the water quality, and decreased the harmful bacterial load of the culture; thus, this system was significantly economical (Suantika et al. 2003).
3 Culture Diagnosis and Treatment
Rotifers have been successfully mass produced; however, maintenance of the culture for a long time remains a critical unresolved problem. Several factors, including decreased feeding activity, protozoan contamination, and poor water quality, caused rotifer cultures to collapse (Yu and Hirayama 1986; Jung et al. 1997). The water quality in the rotifer culture can be assessed by measuring pH, ammonia level, and viscosity (Snell et al. 1987; Hagiwara et al. 1998). The reported critical pH and unionize ammonia concentrations are at 7 (Yoshimura et al. 1996) and at less than 2.1 ppm (Yu and Hirayama 1986), respectively. The viscosity of rotifer culture medium increases with the accumulation of dissolve organic substances, and the higher viscosity causes a decline in the rotifer population (Araujo et al. 2001). It is therefore necessary to continuously monitor rotifer cultures in order to determine their health status. Several methods are recommended to assess the physiological status of cultured rotifers, as well as to predict culture collapse (for review see Hagiwara and Kuwada 2004). Egg ratio (the number of parthenogenetic eggs per female) was used by Snell et al. (1987); however, this method required 1–2 days for the assessment and was not a sensitive end point for culture diagnosis. Swimming speed (Snell et al. 1987; Janssen et al. 1994) and ingestion rate (Ferrando et al. 1993; Juchelka and Snell 1994) were also found to be sensitive indicators to detect stress in rotifer culture. These two characteristics can be detected easily and rapidly (require around 1–2 h). Furthermore, in vivo enzyme activity test, which is considered to be the most sensitive method (Araujo et al. 2001), is another way to assess the health of rotifer cultures; however, it requires instruments such as a computer, fluorometer, and an image analyzer, which increases the cost for operation.
Several treatment methods have been suggested to improve rotifer culture and prevent collapse (Table 3.1). Araujo and Hagiwara (2005) found that the addition of γ-aminobutyric acid (GABA) can improve the health condition of rotifers when they are exposed to stressful environment (e.g., increase of unionized ammonia and protozoa contamination). Addition of GABA during nutritional enrichment culture is also shown to improve survival and swimming activity of rotifers (Gallardo et al. 2001). In addition, the supplementation of porcine growth hormone significantly enhances the rotifer population growth when environmental stressors are low, such as under optimal food conditions with a low concentration of free ammonia (Gallardo et al. 1999). Serotonin and human chorionic gonadotropin are also effective to increase the rotifer population growth. For the improvement of rotifer sexual reproduction, in addition to GABA, serotonin can also be applied under low food conditions. On the other hand, juvenile hormone increases mixis production under optimal and suboptimal food conditions. To apply the mentioned chemicals into rotifer mass cultures, the following issues have to be dealt with: (1) several chemicals also affect body size of rotifers (Gallardo et al. 1997) and unknown influences remain, (2) chemical-treated rotifers would influence fish larvae, and (3) effluents containing these hormones would have effects on indigenous species in receiving waters (Hagiwara et al. 2001).
4 Preservation
Despite the progress of successful establishment of mass culture of rotifers, diagnosis of culture status, treatment of culture for recovery, and methods to preserve rotifers for either feeding fish stocks or starting a new culture are necessary. Mass preservation is essential because of the usual problem of culture collapse. Rotifers can be preserved for a short time (few days to 4 weeks) at low temperatures (between −2 and 8 °C) and for long term (years) by producing and collecting resting eggs.
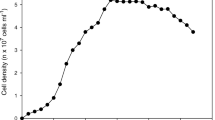
Rotifers preserved at low temperature can either be directly used as feed for the larvae or as starters of rotifer culture (Assavaaree et al. 2001; reviewed by Hagiwara et al. (2001)). However, B. plicatilis sp. complex respond differently to various conditions during preservation. Assavaaree et al. (2001) found that B. plicatilis s. s. were more resistant to low-temperature (4 °C) preservation than B. rotundiformis (Fig. 3.2). They also found that exchange of culture media during the incubation at low temperature is necessary to maintain the viability of the rotifers. The rotifer survival at low temperature is also significantly related to the rotifer culture conditions, e.g., food and salinity before exposure to low temperature (Lubzens et al. 1990), where syntheses of specific proteins, including 94 kD and HSP 60, are being synthesized (Lubzens et al. 1995b). The recovery rate of preserved B. plicatilis for 14 days at density of 20,000 ind/mL was about 50%. The S- and SS-type B. rotundiformis strains were comparatively less resistant to low-temperature preservation but can be improved by GABA treatment (Assavaaree and Hagiwara 2011).
Percent survival after preservation at 4 °C. (a) Survival of 6 L-type strains on day 30 and (b) survival of 8 S- and SS-type strains on day 5. Columns and vertical bars indicate mean rotifer survival of three replicates and standard deviation, respectively. Results of Tukey test were presented (a>b>c, P < 0.05) (Adopted from Hagiwara et al. 2001)
For long-term storage, resting eggs of rotifers can be produced and hatched when needed. The resting egg is a notable feature of rotifer life cycle and is the end product of sexual reproduction which is resistant to harsh environment. The advantages of rotifer resting eggs are highlighted for preservation (reviewed by Hagiwara and Hirayama 1993; Hagiwara et al. 1997). These eggs can be stored for more than 20 years in sterilized seawater under complete darkness at 5 °C. The hatching rates of dried resting egg gradually decrease, but those preserved by canning at an atmospheric pressure of 48–61 kPa after lyophilization (at −30 °C) can be maintained for up to 6 months (Hagiwara et al. 1997). The resting egg production, however, is an obstacle for mass production of rotifers because of the appearance of mictic females which cannot contribute to population growth. Resting eggs, however, could be a by-product during hatchery off-season operation and then can be hatched with the same manner as Artemia cysts when needed. However, even with these merits, not all species of B. plicatilis sp. complex produce resting eggs, and hatchlings from resting eggs would have different reproductive characteristics from their parent because of gene recombination during resting egg formation. As solution to these issues, other methods such as cryopreservation of amictic eggs were tested (Toledo and Kurokura 1990; Toledo et al. 1991; Lubzens et al. 2001). With this method, rotifers showed around 50% of survival rate. This method is recommended and is useful for the maintenance of a certain strain with desired morphological and genetic characteristics. Nevertheless, there are several drawbacks to this technique such as practical only in small-scale and low population growth after thawing (Toledo et al. 1991).
References
Assavaaree M, Hagiwara A (2011) Effect of gamma-aminobutyric acid and porcine growth hormone on survival of the euryhaline rotifers Brachionus plicatilis sp. complex preserved at low temperature. Fish Sci 77:599–605
Assavaaree M, Hagiwara A, Ide K, Maruyama K, Lubzens E (2001) Low-temperature preservation (at 4°C) of marine rotifer Brachionus. Aquac Res 32:29–39
Bentley CD, Carroll PM, Watanabe WO (2008) Intensive rotifer production in a pilot-scale continuous culture recirculating system using nonviable microalgae and an ammonia neutralizer. J World Aquacult Soc 39:625–635
de Araujo AB, Hagiwara A (2005) Screening methods for improving rotifer culture quality. Hydrobiologia 546:553–558
de Araujo AB, Hagiwara A, Snell TW (2001) Effect of unionized ammonia, viscosity and protozoan contamination on reproduction and enzyme activity of the rotifer Brachionus rotundiformis. Hydrobiologia 446(447):363–368
Dhert P, Rombaut G, Suantika G, Sorgeloos P (2001) Advancement of rotifer culture and manipulation techniques in Europe. Aquaculture 200:129–146
Ferrando MD, Janssen CR, Persoone G (1993) Ecotoxicological studies with the freshwater rotifer Brachionus calyciflorus. III. The effects of chemicals on feeding behavior. Ecotoxicol Environ Saf 26:1–9
Fu Y, Hirayama K, Natsukari Y (1991) Genetic divergence between S and L type strains of the rotifer Brachionus plicatilis O. F. Müller. J Exp Mar Biol Ecol 151:43–56
Fu Y, Hada A, Yamashita T, Yoshida Y, Hino A (1997) Development of a continuous culture system for stable mass production of the marine rotifer Brachionus. Hydrobiologia 358:145–151
Gallardo WG, Hagiwara A, Tomita Y, Soyano K, Snell TW (1997) Effect of some vertebrate and invertebrate hormones on the population growth, mictic female production, and body size of the marine rotifer Brachionus plicatilis Müller. Hydrobiologia 358:113–120
Gallardo WG, Hagiwara A, Tomita Y, Snell TW (1999) Effect of growth hormone and gamma-aminobutyric acid on Brachionus plicatilis (Rotifera) reproduction at low food or high ammonia levels. J Exp Mar Biol Ecol 240:179–191
Gallardo WG, Hagiwara A, Snell TW (2001) Use of GABA to enhance rotifer reproduction in enrichment culture. Aquac Res 32:243–246
Gómez A, Serra M, Carvalho GR, Lunt DH (2002) Speciation in ancient cryptic species complexes: evidence from the molecular phylogeny of Brachionus plicatilis (Rotifera). Evolution 56:1431–1444
Hagiwara A (1994) Practical use of rotifer cysts. Israel J Aquac Bamidgeh 46:13–21
Hagiwara A, Hirayama K (1993) Preservation of rotifers and its application in the finfish hatchery. In: Lee CS, Su MS, Liao IC (Eds.) Finfish Hatchery in Asia ‘91. TML Conference Proceedings 3:61–71. Tunkang Marine Laboratory, Taiwan Fisheries Research Institute, Tungkang, Pingtung, Taiwan
Hagiwara A, Kuwada H (2004) Current status of live food culture in Japan. The Second Hatchery Feeds and Technology Workshop. Sydney, Australia, pp. 24–33
Hagiwara A, Kotani T, Snell TW, Assavaaree M, Hirayama K (1995) Morphology, reproduction, genetics and mating behavior of small, tropical marine rotifer Brachionus strains (Rotifera). J Exp Mar Biol Ecol 194:25–37
Hagiwara A, Balompapueng MD, Munuswamy N, Hirayama K (1997) Mass production and preservation of the resting eggs of the euryhaline rotifer Brachionus plicatilis and B. rotundiformis. Aquaculture 155:223–230
Hagiwara A, Yamamiya N, de Araujo AB (1998) Effect of water viscosity of the population growth of the rotifer Brachionus plicatilis Muller. Hydrobiologia 387(388):489–494
Hagiwara A, Gallardo WG, Assavaaree M, Kotani T, Araujo AB (2001) Live food production in Japan: recent progress and future aspects. Aquaculture 200:111–127
Hagiwara A, Suga K, Akazawa A, Kotani T, Sakakura Y (2007) Development of rotifer strains with useful traits for rearing fish larvae. Aquaculture 268:44–52
Hino A (1993) Present culture systems of the rotifer (Brachionus plicatilis) and the function of micro-organisms. TML Conf Proc 3:51–59
Hirata H (1964) Culture of live food at Yashima station. Saibai Gyogyo News 2:4
Janssen RC, Ferrando DM, Persoone G (1994) Ecotoxicological studies with the freshwater rotifer Brachionus calyciflorus IV. Rotifer behavior as a sensitive and rapid sublethal test criterion. Ecotoxicol Environ Saf 28:244–255
Juchelka CM, Snell TW (1994) Rapid toxicity assessment using rotifer ingestion rate. Arch Environ Contam Toxicol 26:549–554
Jung M-M, Hagiwara A, Hirayama K (1997) Interspecific relations in the marine rotifer microcosm. Hydrobiologia 358:121–126
Kotani T, Hagiwara A, Snell TW (1997) Genetic variations among marine rotifer strains and function of mate recognition pheromone (MRP). Hydrobiologia 358:105–112
Kotani T, Hagiwara A, Snell TW, Serra M (2005) Euryhaline Brachionus strains (Rotifera) from tropical habitats: morphology and allozyme patterns. Hydrobiologia 546:161–167
Lubzens E (1987) Raising rotifers for use in aquaculture. Hydrobiologia 147:245–255
Lubzens E, Zmora O (2003) Production and nutritional value of rotifers, 17–64 pp, 300–303 pp. In; Live feeds in marine aquaculture. Eds: JG Støttrup and LA McEvoy, Balckwell Science Ltd, 318 pp
Lubzens E, Kolodny G, Perry B, Galai N, Sheshinski R, Wax Y (1990) Factors affecting survival of rotifers (Brachionus plicatilis O. F. Müller) at 4°C. Aquaculture 91:23–47
Lubzens E, Gibson O, Zmora O, Sukenik A (1995a) Potential advantages of frozen algae (Nannochloropsis sp.) for rotifer (Brachionus plicatilis) culture. Aquaculture 133:295–309
Lubzens E, Rankevich D, Kolodny G, Gibson O, Cohen A, Khayat M (1995b) Physiological adaptations in the survival of rotifers (Brachionus plicatilis, O.F. Müller) at low temperatures. Hydrobiologia 313:175–183
Lubzens E, Zmora O, Barr Y (2001) Biotechnology and aquaculture of rotifers. Hydrobiologia 446:337–353
Mills S, Alcántara-Rodríguez A, Ciros-Pérez J, Gómez A, Hagiwara A, Galindo KH, Jersabek CD, Malekzadeh-Viayeh R, Leasi F, Lee J-S, Welch DBM, Papakostas S, Riss S, Segers H, Serra M, Shiel R, Smolak R, Snell TW, Stelzer CP, Tang CQ, Wallace RL, Fontaneto D, Walsh EJ (2016) Fifteen species in one: deciphering the Brachionus plicatilis species complex (Rotifera, Monogononta) through DNA taxonomy. Hydrobiologia. doi:10.1007/s10750-016-2725-7
Nagata WD, Hirata H (1986) Mariculture in Japan: past, present, and future prospective. Mini Rev Data File Res 4:1–38
Navarro N, Yufera M (1998) Population dynamics of rotifers Brachionus plicatilis and Brachionus rotundiformis in semi-continuous culture fed freeze-dried microalgae: influence of dilution rate. Aquaculture 166:297–309
Schlüter M, Soeder CJ, Groeneweg J (1987) Growth and food conversion of Brachionus rubens in continuous culture. J Plankton Res 9:761–783
Snell TW (1998) Chemical ecology of rotifers. Hydrobiologia 387:267–276
Snell TW, Childress MJ, Boyer EM, Hoff FH (1987) Assessing the status of rotifer mass cultures. J World Aquacult Soc 18:270–277
Snell TW, Rico-Martinez R, Kelly LN, Battle TE (1995) Identification of a sex pheromone from a rotifer. Mar Biol 123:347–353
Suantika G, Dhert P, Nurhudah M, Sorgeloos P (2000) High-density production of the rotifer Brachionus plicatilis in a recirculation system: consideration of water quality, zootechnical and nutritional aspects. Aquac Eng 21:201–213
Suantika G, Dhert P, Rombaut G, Vandenberghe J, de Wolf T, Sorgeloos P (2001) The use of ozone in a high density recirculation system for rotifers. Aquaculture 201:35–49
Suantika G, Dhert P, Sweetman E, O’Brien E, Sorgeloos P (2003) Technical and economical feasibility of a rotifer recirculation system. Aquaculture 227:173–189
Toledo JD, Kurokura H (1990) Cryopreservation of the euryhaline rotifer Brachionus plicatilis embryos. Aquaculture 91:385–394
Toledo JD, Kurokura H, Nakagawa H (1991) Cryopreservation of different strains of the euryhaline rotifer Brachionus plicatilis embryos. Nippon Suisan Gakkaishi 57:1347–1350
Welladsen H, Kent M, Mangott A, Li Y (2014) Shelf-life assessment of microalgae concentrates: effect of cold preservation on microalgal nutrition profiles. Aquaculture 430:241–247
Yin XW, Min WW, Lin HJ, Chen W (2013) Population dynamics, protein content, and lipid composition of Brachionus plicatilis fed artificial macroalgal detritus and Nannochloropsis sp. diets. Aquaculture 380-383:62–69
Yoshimatsu T, Hossain MA (2014) Recent advances in the high-density rotifer culture in Japan. Aquac Int 22:1587–1603
Yoshimura K, Hagiwara A, Yoshimatsu T, Kitajima C (1996) Culture technology of marine rotifers and the implications for intensive culture of marine fish in Japan. Mar Freshw Res 47:217–222
Yoshimura K, Tanaka K, Yoshimatsu T (2003) A novel culture system for the ultra-high-density production of the rotifer, Brachionus rotundiformis—a preliminary report. Aquaculture 227:165–172
Yu J-P, Hirayama K (1986) The effect of un-ionized ammonia on the population growth of the rotifer in mass culture. Nippon Suisan Gakkaishi 52:1509–1513
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer Nature Singapore Pte Ltd. and the Japanese Society of Fisheries Science
About this chapter
Cite this chapter
Hagiwara, A., Kim, HJ., Marcial, H. (2017). Mass Culture and Preservation of Brachionus plicatilis sp. Complex. In: Hagiwara, A., Yoshinaga, T. (eds) Rotifers. Fisheries Science Series. Springer, Singapore. https://doi.org/10.1007/978-981-10-5635-2_3
Download citation
DOI: https://doi.org/10.1007/978-981-10-5635-2_3
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-10-5633-8
Online ISBN: 978-981-10-5635-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)