Abstract
Dendritic cells (DCs) form a heterogeneous group of antigen-presenting cells that play different roles in skin immunology. Recent studies have revealed the existence of distinct DC populations in the skin, highlighting the complexity of the cutaneous DC network in the steady state and inflammatory conditions.
Recently, another new skin immune cell subset, innate lymphoid cells (ILCs), which are part of a heterogeneous family of innate immune cells, has emerged as an important contributor to inflammatory skin diseases, such as atopic dermatitis (AD) and psoriasis.
In this review, we will summarize the current understanding of the functions of cutaneous DCs and ILCs in the pathogenesis of AD and will discuss the potential implications of their functions in AD.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
The skin is classified into two distinct regions, the epidermis and the dermis, separated by the basement membrane. The epidermis is derived from ectoderm and presents as an epithelial layer that is composed mainly of keratinocytes. Keratinocytes constitute 90–95% of the cells in the epidermis; the remaining cells include Langerhans cells (LCs), skin-resident memory CD8+ T cells, melanocytes, and Merkel cells. In addition, murine epidermis contains approximately the same percentage of the γδ T cell subset (called dendritic epidermal T cells [DETC]), as that of LCs [1, 2]. A much more heterogeneous population of immune cells reside in the dermis, including dermal dendritic cells (dDCs), mast cells, CD4+ and CD8+ T cells, dermal γδ T cells, macrophages, natural killer (NK) cells, and innate lymphoid cells (ILCs) [2, 3] (Fig. 8.1).
Atopic dermatitis (AD) is a pruritic, chronic, retractable inflammatory skin disease that is induced by the complex interaction between susceptibility genes encoding skin barrier components and host immune responses [4,5,6,7,8,9,10]. Type 2 helper T (Th2) cells and related cytokines such as IL-4, IL-5, and IL-13 have been reported to play important roles in its pathogenesis [10,11,12,13,14,15]. Cutaneous DCs acquire protein antigens and drive the differentiation/proliferation of a distinct Th cell subset including Th2 cells and provoke antigen-specific T cell responses to external pathogens [16]. In addition to Th2 cells, several recent papers have indicated that type 2 ILCs (ILC2s) are enriched in the lesional skin of AD patients and might play a critical role in driving the Th2-type immune response in AD [17,18,19,20].
This review will define the functions of cutaneous DCs and ILCs in the pathogenesis of AD, with a focus on studies performed by Japanese researchers, and will discuss the potential implications of these functions in the context of skin immune responses in AD.
2 The Role of Cutaneous DCs in the Pathogenesis of AD
2.1 DC Subsets in the Skin
Several subsets of DCs have been identified in the skin of both mice and humans. LCs comprise a skin DC subset that resides in the epidermis. Both human and murine LCs constitutively express a lectin receptor, namely, langerin/CD207, which is capable of binding sugar moieties commonly found on a variety of microorganisms [21]. They also express the E-cadherin and epithelial cell adhesion molecules (EpCAMs), which anchor LCs to keratinocytes [22, 23], and CD205 that is implicated in antigen capture and antigen processing [24, 25]. Human LCs highly express CD1a, a member of the group 1 CD1 protein, which is capable of presenting microbial lipid antigens to T cells [26] (Table 8.1).
DCs in the dermis consist of dermal-resident DCs and migratory LCs traveling to the lymphatic vessels [27]. Recent studies have shown that murine dermal-resident DCs are further classified into two populations including langerin-positive dDCs (langerin+ dDCs) and langerin-negative dDCs (langerin− dDCs) [28,29,30]. The langerin+ dDC population represents 10–20% of the total dDC pool. In contrast to LCs, langerin+ dDCs express integrin αEβ7 (also called CD103) [31], lack the adhesion molecules E-cadherin and EpCAM, and express low levels of the integrin CD11b. Langerin+ dDCs express the same high levels of CD24 as LCs but do not express CX3CR1, F4/80, or signal-regulatory protein alpha (SIRPα); they also express low levels of CD11b and EpCAM [32] (Table 8.1). Langerin− dDCs represent the majority (up to 70%) of the dDC pool and express high levels of integrin CD11b and several macrophage markers, such as F4/80, CX3CR1, and SIRPα [32]. In human, dDCs are classified into three subsets (i.e., CD1a + dDCs, CD14+ dDCs, and CD141+ dDCs), depending on the expression pattern of surface molecules, although their functional differences remain unclear [33] (Table 8.1).
In inflamed skin, two additional subsets of DCs can be found: a DC population derived from blood monocytes (monocyte-derived DCs) [19] and plasmacytoid DCs (pDCs) [2, 34] (Table 8.1, Fig. 8.2). Another subset of the identified DC subpopulation referred to as TNF-α and inducible NO synthase (iNOS)-producing DCs (Tip-DCs) has been reported to be critical against bacterial infections [35]. In AD patients, inflammatory dendritic epidermal cells (IDECs) appear in the epidermis (Table 8.1, Fig. 8.2).
Cutaneous dendritic cells in the steady state and in the inflammatory state. TJs tight junctions, LCs Langerhans cells, dermal DCs dermal dendritic cells, IDECs inflammatory dendritic epidermal cells, Tip-DCs tumor necrosis factor and inducible nitric oxide synthase-producing dendritic cells, pDCs plasmacytoid dendritic cells. In steady state, LCs reside in the epidermis beneath the TJ barrier. Dermal DCs reside in the dermis. In inflammatory condition, such as AD, LCs elongate their dendrite through TJs, capturing external antigens. IDECs recruit into the epidermis. Tip-DCs, pDCs, and monocyte-derived DCs also recruit into the dermis from the circulation
2.2 Cutaneous DCs in the Pathogenesis of AD
Since AD is a Th2-type immune response to protein antigens, and cutaneous DCs can induce Th2 cell differentiation, it has been suggested that cutaneous DCs initiate AD [36]. It remains unclear, however, which cutaneous DC subset mediates epicutaneous sensitization to protein antigens.
Kubo et al. showed that protein antigens of large molecular size are localized above the size-selective barrier, the tight junction (TJ), and that activated LCs extend their dendrites through the TJ to take up antigens [37]. Therefore, it can be hypothesized that LCs, not dermal DCs, initiate epicutaneous sensitization with protein antigens in the development of AD.
Nakajima et al. and Ouchi et al. showed that, in a murine AD model, which is induced by epicutaneous sensitization with protein antigen, depletion of LCs led to significantly attenuated induction of IgE/IgG1 upon epicutaneous sensitization with protein antigens [38, 39]. Moreover, Nakajima et al. showed that thymic stromal lymphopoietin (TSLP) receptor (TSLPR) expression on LCs is enhanced upon protein antigen exposure to the skin and that mice lacking TSLPR on LCs exhibited significantly attenuated Th2 cell differentiation in the AD model [38].
TSLP is a cytokine that is produced mainly by non-hematopoietic cells such as fibroblasts and epithelial cells, including epidermal keratinocytes. It has been reported that TSLP plays an important role in the induction of Th2 responses through the activation of DCs via the expression of OX40L [40]. TSLP stimulation also causes LCs to express OX40L [38]. In addition, cutaneous DCs can elicit a Th2 response in a mechanical injury model, in which TSLP is produced by keratinocytes [41]. These results indicate that TSLP produced by keratinocytes acts on LCs, which induces Th2-type immune responses in the murine AD model [38] (Fig. 8.3). In keeping with this, it has been reported that TSLP is highly expressed in the lesional skin of AD patients and that it activates human LCs in vitro [42,43,44], suggesting that LCs induce Th2-type immune response in human AD as well.
In the epidermis of human AD patients, both LCs and IDECs have been detected. Both LCs and IDECs express FcεRI on the surface and have been assumed to enable specific allergen uptake through IgE-dependent antigen capture [45, 46]. Yoshida et al., however, revealed that LCs, not IDECs, extend their dendrites through the TJ with a polarized distribution of langerin but not FcεRI in human AD skin [47]. These results again suggest that LCs mediate Th2-type immune responses in a langerin-dependent manner in the pathogenesis of AD.
3 The Role of ILCs in the Pathogenesis of AD
3.1 ILCs in the Skin
ILCs are part of a family of innate immune cells that are derived from a common lymphoid progenitor, and their development is partially or completely dependent on the common γ-chain (γc or CD132), IL-7, Notch, and the transcription factor inhibitor of DNA binding 2 [48]. ILCs are predominantly tissue-resident cells that lack antigen-specific receptors, such as T and B cell receptors. Instead, ILCs rapidly respond to various environmental stimuli with cytokine production [49].
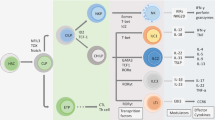
The ILC family can be subdivided into three groups based on their requirement for activating cytokines, expression of transcription factors, and production of effector cytokines (Fig. 8.4). Group 1 ILC comprises NK cells and ILC1s; both of them are activated by IL-12 depending on transcription factor T-bet and produce interferon (IFN)-γ. Groups 2 ILC contains ILC2s that are activated by epithelial cell-derived cytokines/chemokines, such as IL-25, IL-33, and TSLP. ILC2s express GATA3 and produce Th2-type cytokines, e.g., IL-4, IL-5, and IL-13 upon activation. Group 3 ILC comprises lymphoid tissue inducer (LTi) cells and ILC3s. ILC3s can be further subdivided based on their expression of natural cytotoxicity receptors. They are activated by IL-1β and IL-23, are dependent on RORγt, and produce IL-17A and/or IL-22 [50]. Intriguingly, all of the ILC-activating cytokines can be produced by skin immune cells, such as keratinocytes, LCs, and dDCs.
Regulation and function of each subset of innate lymphoid cells (ILCs) in the human skin. Human skin ILC1s express CD161 and can be activated by IL-12 and IL-15 to produce its effector cytokines TNF-α and IFN-γ. Human skin ILC2s can be activated by epithelial cell-derived cytokines/eicosanoid (e.g., IL-25, IL-33, TSLP, and PGD2) and produce its effector cytokines IL-5 and IL-13; they have thus been implicated in AD. Human skin ILC3s produce IL-17 and IL-22 in response to IL-1β and IL-23 and have been implicated in psoriasis
All three ILC subsets have been identified in healthy human adult skin, where ILC3s are the most abundant and ILC2s make up 25% of the total ILC pool [20, 51,52,53,54]. In addition to NK cells, ILC2s were the first ILCs to be identified in both healthy and AD skin [52]. Although ILCs are present in both epidermis and dermis, the majority are found in the dermis, where ILC2s constitute 5–10% of CD45+ cells [55].
3.2 ILC2s in the Pathogenesis of AD
ILC2s were originally identified in the gut and gut-associated lymphoid tissues and were found to contribute to immunity against parasitic helminth in the absence of an adaptive immune system [56,57,58,59]. Subsequent studies showed that ILC2s play key roles in airway hyperreactivity [60] and epithelial tissue repair in the lung [61]. These studies demonstrated that ILC2s were predominantly regulated by the epithelial cell-derived cytokines IL-25 and IL-33 [62] (Fig. 8.5).
Potential roles of ILC2s in AD. ILC2s are activated by epithelial cell-derived cytokines IL-33, IL-25, and TSLP, all of which are associated with barrier disruption and are upregulated in AD. ILC2 cell-derived IL-9 may increase mast cell numbers in the skin and enhance their responses to IgE-mediated activation in AD. Activated mast cell-derived PGD2 promotes ILC2 migration in the skin
When ILC2s were first identified in the skin, they were also found to be enriched in the lesional skin of AD patients [20, 52]. Type 2 cytokines, including IL-5 and IL-13, have long been suspected to play a key role in the pathogenesis of AD [63,64,65]. It was also revealed that ILC2s that produce IL-5 and IL-13 were found to be both necessary and sufficient for the development of AD-like disease in a low-dose vitamin D3-induced murine AD model [52, 55].
Mast cell numbers are elevated in chronic AD and likely drive IgE-mediated pathology in the disease [66]. Mast cells can also be directly activated by toxins derived from the skin-resident bacteria, Staphylococcus aureus, which is commonly found in the lesional skin of AD patients [67, 68]. Mast cell hyperplasia during inflammation is mediated by IL-3 and IL-9 [69, 70]. IL-9 can also augment the inflammatory cytokine response by mast cells following IgE-mediated activation [55]. ILC2s are a key source of IL-9 in vivo [71], suggesting that ILC2s potentially affect the severity of AD by regulating the number and function of mast cells in the lesional skin (Fig. 8.5). It has also been shown that prostaglandin D2 (PGD2) promotes the migration of skin ILC2s and induces type 2 cytokine production from skin ILC2s. Moreover, the supernatant of IgE/anti-IgE-activated mast cells induces migration and cytokine production by human skin ILC2s [72]. Since mast cells produce PGD2 by means of activation through FcεRI, they might play a role in migration and cytokine production by ILC2s through the effects of PGD2.
As mentioned earlier, TSLP is a crucial cytokine that causes cutaneous DCs to induce Th2 cells. ILC2s express the receptor for TSLP, and TSLP signaling in ILC2s promotes cytokine production [73]. Activation of cutaneous ILC2s was found to be dependent on TSLP signaling, suggesting that ILC2s were critical for disease pathogenesis [52] (Fig. 8.5). In addition to TSLP, Imai et al. showed that transgenic overexpression of IL-33 under a keratin 14 promoter is sufficient to induce spontaneous AD-like dermatitis in association with the accumulation of ILC2s [74]. This human and mouse evidence indicates that ILC2s are important players in the pathogenesis of AD, where their activity likely depends on the severity of AD, coinfections, and dysfunction of the skin barrier.
Conclusion
With their constitutional plasticity, LCs and ILC2s in the skin play crucial roles in initiating and modulating type 2 immune responses in AD. As many papers have demonstrated, both Th2 cells and ILC2s seem to play crucial roles in the pathogenesis of AD. The magnitude of their effect and/or their interplay, however, remains to be further elucidated.
Recent advances in our knowledge of the skin immune network and pathology in the pathogenesis of AD have opened avenues for the development of therapies that trigger skin DCs and/or ILC2s with specialized properties to control immunity.
References
Kaplan DH, Igyarto BZ, Gaspari AA. Early immune events in the induction of allergic contact dermatitis. Nat Rev Immunol. 2012;12(2):114–24. doi:10.1038/nri3150.
Nestle FO, Di Meglio P, Qin JZ, Nickoloff BJ. Skin immune sentinels in health and disease. Nat Rev Immunol. 2009;9(10):679–91. doi:10.1038/nri2622.
Otsuka A, Kabashima K. Mast cells and basophils in cutaneous immune responses. Allergy. 2015;70(2):131–40. doi:10.1111/all.12526.
Brunner PM, Silverberg JI, Guttman-Yassky E, Paller AS, Kabashima K, Amagai M, et al. Increasing comorbidities suggest that atopic dermatitis is a systemic disorder. J Invest Dermatol. 2016. doi:10.1016/j.jid.2016.08.022.
Dainichi T, Hanakawa S, Kabashima K. Classification of inflammatory skin diseases: a proposal based on the disorders of the three-layered defense systems, barrier, innate immunity and acquired immunity. J Dermatol Sci. 2014;76(2):81–9. doi:10.1016/j.jdermsci.2014.08.010.
Egawa G, Kabashima K. Multifactorial skin barrier deficiency and atopic dermatitis: essential topics to prevent the atopic march. J Allergy Clin Immunol. 2016;138(2):350–8.e1. doi:10.1016/j.jaci.2016.06.002.
Kabashima K. New concept of the pathogenesis of atopic dermatitis: interplay among the barrier, allergy, and pruritus as a trinity. J Dermatol Sci. 2013;70(1):3–11. doi:10.1016/j.jdermsci.2013.02.001.
Moniaga CS, Kabashima K. Filaggrin in atopic dermatitis: flaky tail mice as a novel model for developing drug targets in atopic dermatitis. Inflamm Allergy Drug Targets. 2011;10(6):477–85.
Saeki H, Nakahara T, Tanaka A, Kabashima K, Sugaya M, Murota H, et al. Clinical practice guidelines for the management of atopic dermatitis 2016. J Dermatol. 2016;43(10):1117–45. doi:10.1111/1346-8138.13392.
Werfel T. The role of leukocytes, keratinocytes, and allergen-specific IgE in the development of atopic dermatitis. J Invest Dermatol. 2009;129(8):1878–91. doi:10.1038/jid.2009.71.
Amano W, Nakajima S, Kunugi H, Numata Y, Kitoh A, Egawa G, et al. The Janus kinase inhibitor JTE-052 improves skin barrier function through suppressing signal transducer and activator of transcription 3 signaling. J Allergy Clin Immunol. 2015;136(3):667–677. e7. doi:10.1016/j.jaci.2015.03.051.
Ewald DA, Noda S, Oliva M, Litman T, Nakajima S, Li X et al. Major differences between human atopic dermatitis and murine models, as determined by using global transcriptomic profiling. J Allergy Clin Immunol. 2016. doi:10.1016/j.jaci.2016.08.029.
Nemoto O, Furue M, Nakagawa H, Shiramoto M, Hanada R, Matsuki S, et al. The first trial of CIM331, a humanized antihuman interleukin-31 receptor A antibody, in healthy volunteers and patients with atopic dermatitis to evaluate safety, tolerability and pharmacokinetics of a single dose in a randomized, double-blind, placebo-controlled study. Br J Dermatol. 2016;174(2):296–304. doi:10.1111/bjd.14207.
Noda S, Suarez-Farinas M, Ungar B, Kim SJ, de Guzman SC, Xu H, et al. The Asian atopic dermatitis phenotype combines features of atopic dermatitis and psoriasis with increased TH17 polarization. J Allergy Clin Immunol. 2015;136(5):1254–64. doi:10.1016/j.jaci.2015.08.015.
Nomura T, Kabashima K, Miyachi Y. The panoply of alphabetaT cells in the skin. J Dermatol Sci. 2014;76(1):3–9. doi:10.1016/j.jdermsci.2014.07.010.
Banchereau J, Briere F, Caux C, Davoust J, Lebecque S, Liu YJ, et al. Immunobiology of dendritic cells. Annu Rev Immunol. 2000;18:767–811. doi:10.1146/annurev.immunol.18.1.767.
Kim BS, Wojno ED, Artis D. Innate lymphoid cells and allergic inflammation. Curr Opin Immunol. 2013;25(6):738–44. doi:10.1016/j.coi.2013.07.013.
Kim BS, Wang K, Siracusa MC, Saenz SA, Brestoff JR, Monticelli LA, et al. Basophils promote innate lymphoid cell responses in inflamed skin. J Immunol. 2014;193(7):3717–25. doi:10.4049/jimmunol.1401307.
Leon B, Ardavin C. Monocyte-derived dendritic cells in innate and adaptive immunity. Immunol Cell Biol. 2008;86(4):320–4. doi:10.1038/icb.2008.14.
Salimi M, Barlow JL, Saunders SP, Xue L, Gutowska-Owsiak D, Wang X, et al. A role for IL-25 and IL-33-driven type-2 innate lymphoid cells in atopic dermatitis. J Exp Med. 2013;210(13):2939–50. doi:10.1084/jem.20130351.
Valladeau J, Saeland S. Cutaneous dendritic cells. Semin Immunol. 2005;17(4):273–83. doi:10.1016/j.smim.2005.05.009.
Borkowski TA, Letterio JJ, Farr AG, Udey MC. A role for endogenous transforming growth factor beta 1 in Langerhans cell biology: the skin of transforming growth factor beta 1 null mice is devoid of epidermal Langerhans cells. J Exp Med. 1996;184(6):2417–22.
Tang A, Amagai M, Granger LG, Stanley JR, Udey MC. Adhesion of epidermal Langerhans cells to keratinocytes mediated by E-cadherin. Nature. 1993;361(6407):82–5. doi:10.1038/361082a0.
Inaba K, Swiggard WJ, Inaba M, Meltzer J, Mirza A, Sasagawa T, et al. Tissue distribution of the DEC-205 protein that is detected by the monoclonal antibody NLDC-145. I. Expression on dendritic cells and other subsets of mouse leukocytes. Cell Immunol. 1995;163(1):148–56.
Jiang W, Swiggard WJ, Heufler C, Peng M, Mirza A, Steinman RM, et al. The receptor DEC-205 expressed by dendritic cells and thymic epithelial cells is involved in antigen processing. Nature. 1995;375(6527):151–5. doi:10.1038/375151a0.
Hunger RE, Sieling PA, Ochoa MT, Sugaya M, Burdick AE, Rea TH, et al. Langerhans cells utilize CD1a and langerin to efficiently present nonpeptide antigens to T cells. J Clin Invest. 2004;113(5):701–8. doi:10.1172/JCI19655.
Merad M, Ginhoux F, Collin M. Origin, homeostasis and function of Langerhans cells and other langerin-expressing dendritic cells. Nat Rev Immunol. 2008;8(12):935–47. doi:10.1038/nri2455.
Bursch LS, Wang L, Igyarto B, Kissenpfennig A, Malissen B, Kaplan DH, et al. Identification of a novel population of Langerin+ dendritic cells. J Exp Med. 2007;204(13):3147–56. doi:10.1084/jem.20071966.
Ginhoux F, Collin MP, Bogunovic M, Abel M, Leboeuf M, Helft J, et al. Blood-derived dermal langerin+ dendritic cells survey the skin in the steady state. J Exp Med. 2007;204(13):3133–46. doi:10.1084/jem.20071733.
Poulin LF, Henri S, de Bovis B, Devilard E, Kissenpfennig A, Malissen B. The dermis contains langerin+ dendritic cells that develop and function independently of epidermal Langerhans cells. J Exp Med. 2007;204(13):3119–31. doi:10.1084/jem.20071724.
Cepek KL, Shaw SK, Parker CM, Russell GJ, Morrow JS, Rimm DL, et al. Adhesion between epithelial cells and T lymphocytes mediated by E-cadherin and the alpha E beta 7 integrin. Nature. 1994;372(6502):190–3. doi:10.1038/372190a0.
Ginhoux F, Liu K, Helft J, Bogunovic M, Greter M, Hashimoto D, et al. The origin and development of nonlymphoid tissue CD103+ DCs. J Exp Med. 2009;206(13):3115–30. doi:10.1084/jem.20091756.
Patel VI, Metcalf JP. Identification and characterization of human dendritic cell subsets in the steady state: a review of our current knowledge. J Invest Med. 2016;64(4):833–47. doi:10.1136/jim-2016-000072.
Liu YJ. IPC: professional type 1 interferon-producing cells and plasmacytoid dendritic cell precursors. Annu Rev Immunol. 2005;23:275–306. doi:10.1146/annurev.immunol.23.021704.115633.
Serbina NV, Salazar-Mather TP, Biron CA, Kuziel WA, Pamer EG. TNF/iNOS-producing dendritic cells mediate innate immune defense against bacterial infection. Immunity. 2003;19(1):59–70.
Novak N, Peng W, Yu C. Network of myeloid and plasmacytoid dendritic cells in atopic dermatitis. Adv Exp Med Biol. 2007;601:97–104.
Kubo A, Nagao K, Yokouchi M, Sasaki H, Amagai M. External antigen uptake by Langerhans cells with reorganization of epidermal tight junction barriers. J Exp Med. 2009;206(13):2937–46. doi:10.1084/jem.20091527.
Nakajima S, Igyarto BZ, Honda T, Egawa G, Otsuka A, Hara-Chikuma M, et al. Langerhans cells are critical in epicutaneous sensitization with protein antigen via thymic stromal lymphopoietin receptor signaling. J Allergy Clin Immunol. 2012;129(4):1048–55.e6. doi:10.1016/j.jaci.2012.01.063.
Ouchi T, Kubo A, Yokouchi M, Adachi T, Kobayashi T, Kitashima DY, et al. Langerhans cell antigen capture through tight junctions confers preemptive immunity in experimental staphylococcal scalded skin syndrome. J Exp Med. 2011;208(13):2607–13. doi:10.1084/jem.20111718.
Ito T, Wang YH, Duramad O, Hori T, Delespesse GJ, Watanabe N, et al. TSLP-activated dendritic cells induce an inflammatory T helper type 2 cell response through OX40 ligand. J Exp Med. 2005;202(9):1213–23. doi:10.1084/jem.20051135.
Oyoshi MK, Larson RP, Ziegler SF, Geha RS. Mechanical injury polarizes skin dendritic cells to elicit a T(H)2 response by inducing cutaneous thymic stromal lymphopoietin expression. J Allergy Clin Immunol. 2010;126(5):976–84, 84.e1–5. doi:10.1016/j.jaci.2010.08.041.
Ebner S, Nguyen VA, Forstner M, Wang YH, Wolfram D, Liu YJ, et al. Thymic stromal lymphopoietin converts human epidermal Langerhans cells into antigen-presenting cells that induce proallergic T cells. J Allergy Clin Immunol. 2007;119(4):982–90. doi:10.1016/j.jaci.2007.01.003.
Liu YJ. Thymic stromal lymphopoietin: master switch for allergic inflammation. J Exp Med. 2006;203(2):269–73. doi:10.1084/jem.20051745.
Soumelis V, Reche PA, Kanzler H, Yuan W, Edward G, Homey B, et al. Human epithelial cells trigger dendritic cell mediated allergic inflammation by producing TSLP. Nat Immunol. 2002;3(7):673–80. doi:10.1038/ni805.
Bieber T. Atopic dermatitis. N Engl J Med. 2008;358(14):1483–94. doi:10.1056/NEJMra074081.
Wollenberg A, Rawer HC, Schauber J. Innate immunity in atopic dermatitis. Clin Rev Allergy Immunol. 2011;41(3):272–81. doi:10.1007/s12016-010-8227-x.
Yoshida K, Kubo A, Fujita H, Yokouchi M, Ishii K, Kawasaki H, et al. Distinct behavior of human Langerhans cells and inflammatory dendritic epidermal cells at tight junctions in patients with atopic dermatitis. J Allergy Clin Immunol. 2014;134(4):856–64. doi:10.1016/j.jaci.2014.08.001.
Mjosberg J, Eidsmo L. Update on innate lymphoid cells in atopic and non-atopic inflammation in the airways and skin. Clin Exp Allergy. 2014;44(8):1033–43. doi:10.1111/cea.12353.
Fan X, Rudensky AY. Hallmarks of tissue-resident lymphocytes. Cell. 2016;164(6):1198–211. doi:10.1016/j.cell.2016.02.048.
Sonnenberg GF, Mjosberg J, Spits H, Artis D. SnapShot: innate lymphoid cells. Immunity. 2013;39(3):622-e1. doi:10.1016/j.immuni.2013.08.021.
Dyring-Andersen B, Geisler C, Agerbeck C, Lauritsen JP, Gudjonsdottir SD, Skov L, et al. Increased number and frequency of group 3 innate lymphoid cells in nonlesional psoriatic skin. Br J Dermatol. 2014;170(3):609–16. doi:10.1111/bjd.12658.
Kim BS, Siracusa MC, Saenz SA, Noti M, Monticelli LA, Sonnenberg GF, et al. TSLP elicits IL-33-independent innate lymphoid cell responses to promote skin inflammation. Sci Transl Med. 2013;5(170):170ra16. doi:10.1126/scitranslmed.3005374.
Teunissen MB, Munneke JM, Bernink JH, Spuls PI, Res PC, Te Velde A, et al. Composition of innate lymphoid cell subsets in the human skin: enrichment of NCR(+) ILC3 in lesional skin and blood of psoriasis patients. J Invest Dermatol. 2014;134(9):2351–60. doi:10.1038/jid.2014.146.
Villanova F, Flutter B, Tosi I, Grys K, Sreeneebus H, Perera GK, et al. Characterization of innate lymphoid cells in human skin and blood demonstrates increase of NKp44+ ILC3 in psoriasis. J Invest Dermatol. 2014;134(4):984–91. doi:10.1038/jid.2013.477.
Roediger B, Kyle R, Yip KH, Sumaria N, Guy TV, Kim BS, et al. Cutaneous immunosurveillance and regulation of inflammation by group 2 innate lymphoid cells. Nat Immunol. 2013;14(6):564–73. doi:10.1038/ni.2584.
Fallon PG, Ballantyne SJ, Mangan NE, Barlow JL, Dasvarma A, Hewett DR, et al. Identification of an interleukin (IL)-25-dependent cell population that provides IL-4, IL-5, and IL-13 at the onset of helminth expulsion. J Exp Med. 2006;203(4):1105–16. doi:10.1084/jem.20051615.
Moro K, Yamada T, Tanabe M, Takeuchi T, Ikawa T, Kawamoto H, et al. Innate production of T(H)2 cytokines by adipose tissue-associated c-Kit(+)Sca-1(+) lymphoid cells. Nature. 2010;463(7280):540–4. doi:10.1038/nature08636.
Neill DR, Wong SH, Bellosi A, Flynn RJ, Daly M, Langford TK, et al. Nuocytes represent a new innate effector leukocyte that mediates type-2 immunity. Nature. 2010;464(7293):1367–70. doi:10.1038/nature08900.
Price AE, Liang HE, Sullivan BM, Reinhardt RL, Eisley CJ, Erle DJ, et al. Systemically dispersed innate IL-13-expressing cells in type 2 immunity. Proc Natl Acad Sci U S A. 2010;107(25):11489–94. doi:10.1073/pnas.1003988107.
Chang YJ, Kim HY, Albacker LA, Baumgarth N, McKenzie AN, Smith DE, et al. Innate lymphoid cells mediate influenza-induced airway hyper-reactivity independently of adaptive immunity. Nat Immunol. 2011;12(7):631–8. doi:10.1038/ni.2045.
Monticelli LA, Sonnenberg GF, Abt MC, Alenghat T, Ziegler CG, Doering TA, et al. Innate lymphoid cells promote lung-tissue homeostasis after infection with influenza virus. Nat Immunol. 2011;12(11):1045–54. doi:10.1031/ni.2131.
Monticelli LA, Sonnenberg GF, Artis D. Innate lymphoid cells: critical regulators of allergic inflammation and tissue repair in the lung. Curr Opin Immunol. 2012;24(3):284–9. doi:10.1016/j.coi.2012.03.012.
Akdis M, Akdis CA, Weigl L, Disch R, Blaser K. Skin-homing, CLA+ memory T cells are activated in atopic dermatitis and regulate IgE by an IL-13-dominated cytokine pattern: IgG4 counter-regulation by CLA- memory T cells. J Immunol. 1997;159(9):4611–9.
Bos JD, Wierenga EA, Sillevis Smitt JH, van der Heijden FL, Kapsenberg ML. Immune dysregulation in atopic eczema. Arch Dermatol. 1992;128(11):1509–12.
Plaut M. Antigen-specific lymphokine secretory patterns in atopic disease. J Immunol. 1990;144(12):4497–500.
Liu FT, Goodarzi H, Chen HY. IgE, mast cells, and eosinophils in atopic dermatitis. Clin Rev Allergy Immunol. 2011;41(3):298–310. doi:10.1007/s12016-011-8252-4.
Kong HH, Oh J, Deming C, Conlan S, Grice EA, Beatson MA, et al. Temporal shifts in the skin microbiome associated with disease flares and treatment in children with atopic dermatitis. Genome Res. 2012;22(5):850–9. doi:10.1101/gr.131029.111.
Nakamura Y, Oscherwitz J, Cease KB, Chan SM, Munoz-Planillo R, Hasegawa M, et al. Staphylococcus delta-toxin induces allergic skin disease by activating mast cells. Nature. 2013;503(7476):397–401. doi:10.1038/nature12655.
Lantz CS, Boesiger J, Song CH, Mach N, Kobayashi T, Mulligan RC, et al. Role for interleukin-3 in mast-cell and basophil development and in immunity to parasites. Nature. 1998;392(6671):90–3. doi:10.1038/32190.
Townsend JM, Fallon GP, Matthews JD, Smith P, Jolin EH, McKenzie NA. IL-9-deficient mice establish fundamental roles for IL-9 in pulmonary mastocytosis and goblet cell hyperplasia but not T cell development. Immunity. 2000;13(4):573–83.
Wilhelm C, Hirota K, Stieglitz B, Van Snick J, Tolaini M, Lahl K, et al. An IL-9 fate reporter demonstrates the induction of an innate IL-9 response in lung inflammation. Nat Immunol. 2011;12(11):1071–7. doi:10.1038/ni.2133.
Xue L, Salimi M, Panse I, Mjosberg JM, McKenzie AN, Spits H, et al. Prostaglandin D2 activates group 2 innate lymphoid cells through chemoattractant receptor-homologous molecule expressed on TH2 cells. J Allergy Clin Immunol. 2014;133(4):1184–94. doi:10.1016/j.jaci.2013.10.056.
Mjosberg J, Bernink J, Golebski K, Karrich JJ, Peters CP, Blom B, et al. The transcription factor GATA3 is essential for the function of human type 2 innate lymphoid cells. Immunity. 2012;37(4):649–59. doi:10.1016/j.immuni.2012.08.015.
Imai Y, Yasuda K, Sakaguchi Y, Haneda T, Mizutani H, Yoshimoto T, et al. Skin-specific expression of IL-33 activates group 2 innate lymphoid cells and elicits atopic dermatitis-like inflammation in mice. Proc Natl Acad Sci U S A. 2013;110(34):13921–6. doi:10.1073/pnas.1307321110.
Acknowledgments
This work was supported in part by Grants-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science, and Technology and the Ministry of Health, Labor, and Welfare of Japan.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Nakajima, S., Honda, T., Kabashima, K. (2018). Etiopathology of Atopic Dermatitis: Immunological Aspects of Dendritic Cells (DCs) and Innate Lymphoid Cells (ILCs). In: Katayama, I., Murota, H., Satoh, T. (eds) Evolution of Atopic Dermatitis in the 21st Century. Springer, Singapore. https://doi.org/10.1007/978-981-10-5541-6_8
Download citation
DOI: https://doi.org/10.1007/978-981-10-5541-6_8
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-10-5540-9
Online ISBN: 978-981-10-5541-6
eBook Packages: MedicineMedicine (R0)