Abstract
Pulmonary hypertension (PH) is a chronic, debilitating condition which gravely affects exercise tolerance and quality of life. Though most therapies focus purely on medical intervention, there is a growing body of evidence to suggest the role and benefits of exercise training. This chapter discusses the various physiological basis for exercise intolerance observed in PH and highlights the rationale for exercise training. Recent evidence related to exercise training is summarized and potential pathways to suggest adaptations to exercise training are put forward. While keeping the paper applicable to clinicians, details on evaluating exercise intolerance, prescribing exercise and setting up rehabilitation centers for PH are discussed.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Pulmonary hypertension (PH) is a condition that is gaining global attention with the rise in prevalence, thanks to the rise in other diseases such as heart failure (HF) and chronic obstructive pulmonary disease (COPD) [1]. The recent World Symposium on Pulmonary Hypertension at France, has classified PH into five distinct etiological groups which are clinically relevant [2]. However, based on haemodynamic parameters, PH can also be classified as pre-capillary, post-capillary, isolated post-capillary and combined post- and pre-capillary PH [3, 4]. The changes in haemodynamics are related to pulmonary vascular remodeling which occurs due to a dysfunction of the pulmonary endothelial cells and vascular smooth muscles [5]. All these changes result in gross limitation to perform exercise as a result of various physiological mechanisms causing symptoms of dyspnea, fatigue and syncope [6,7,8].
Current therapies have focused on targeting the three main pathways of pulmonary vascular remodeling (i.e., endothelin pathway, nitric oxide pathway and prostacyclin pathway) through either sequential combination therapy or through an initial double or triple combination therapy [9]. With these advancements in medical therapies, survival years have only improved which have resulted in greater survivors of PH having poor functional capacity and quality of life [10, 11]. Thus, there is a need to work towards improving their function and quality of life through various rehabilitative interventions such as exercise training.
This chapter will focus on the various physiological mechanisms limiting exercise performance, the rationale for exercise training, evaluation of exercise capacity and an updated review of recent literature on exercise training in PH. In addition, key information on setting up a PH rehabilitation center will also be described.
2 Exercise Limitations in Pulmonary Hypertension
Exercise intolerance is a major finding across all forms of PH. A complex interaction between the pulmonary, cardiovascular and musculoskeletal systems are responsible for exercise intolerance seen in PH. Early studies by Sun et al., identified various central and peripheral mechanisms in idiopathic PH from cardiopulmonary exercise testing (CPX) [12]. A recent review, described the various limiting factors for various etiologies of PH and postulated possible mechanisms through which the various systems involving the right and left ventricles, pulmonary circulation, respiratory system and skeletal muscles all contribute to poor exercise tolerance in PH [6].
2.1 Haemodynamics and Exercise Limitation
Altered haemodynamics in the pulmonary circulation is a key finding in PH. Changes in pulmonary vascular resistance (PVR) and pulmonary artery pressures (PAP) under normal circumstances, allow for adequate decrease and maintenance to ensure homeostasis within the pulmonary circulation [7]. In a normal healthy individual, the response to exercise causes a drop in PVR secondary to recruitment of the vascular bed. In addition, minimal alteration in the radius of the vessel results in an almost fourfold increase in PVR [7]. These are similar to changes in pulmonary vascular distensibility in which a 2% per mmHg decrease of mean PAP occurs during high cardiac output [13]. However, changes in distensibility by even a meager 0.1% per mmHg, greatly increases the mean PAP resulting in a limitation of exercise. The impact of the raised PVR is felt on the right ventricle (RV) and this results in uncoupling of the RV and the pulmonary vasculature [14]. A recent study further identified altered resting ventriculo-arterial coupling ratio which failed to increase during exercise thereby advancing the RV dysfunction and limiting exercise [15].
The altered size of the RV in turn compresses the left ventricle (LV) which subsequently affects CO by affecting the distensibility of the LV [16, 17]. This cycle continues with the progression of the uncoupling and consequently affects the RV contractile reserve [18]. In conditions like HF with reduced ejection fraction (HFrEF), there is a gradual increase in PAP due to the prolonged elevation of the PCWP [19]. HF with preserved ejection fraction (HFpEF), however, increases in left atrial pressure leads to remodeling and a decrease in compliance of the pulmonary arterial bed, thereby increasing the oscillatory load on the RV [20].
2.2 Cardiorespiratory Function and Exercise Limitation
Cardiorespiratory function as evaluated from cardiopulmonary exercise testing (CPX), which has now received a Level B, Class IIa recommendation for diagnostic evaluation [21], was initially used to describe two potential pathways resulting in exercise intolerance in PAH, i.e., an increase in ventilatory demand and impaired muscle contraction (which will be described in Sect. 2.3) [12].
These changes are more profound in chronic lung disease and chronic thromboembolic PH (i.e., Group 3 and group 4). Altered diffusion in chronic lung disease occurs as the result of altered biomechanics of the thorax and the hypoxia induced vasoconstriction causing the raised PVR [22] [23]. In addition, CTEPH, as a result of increased dead space ventilation, shows an abnormal decrease in end tidal carbon dioxide (PETCO2) and abnormal rise in the minute ventilation – carbon dioxide production (V̇E/V̇CO2) relationship [24]. This has also been observed in other forms of PH like idiopathic PH, PH due to congenital heart disease and connective tissue disorders [25].
2.3 Muscles and Exercise Limitation
In addition to the altered pulmonary haemodynamics and cardiorespiratory system, muscle dysfunctions (both peripheral and respiratory) further contribute to exercise limitations. Poor oxygen delivery at the periphery due to circulatory changes result in lactic acid build up which has been shown to limit exercise [26]. In addition, limited cardiac output could result in overactivation of the sympathetic systems similar to that seen in HF [27]. Both these factors could contribute to the “generalized myopathy” observed [28]. The involvement of the diaphragm, along with the peripheral muscles, has been shown to further contribute to the exercise limitations seen in PH [29]. Changes in respiratory muscle strength have recently been shown to moderately strong relationships with functional capacity (r = 0.40) and physical activity (r = 0,38–0.61 for vigorous and moderate physical activity respectively) [30].
Thus, the contributions of the various systems to exercise limitations in PH are summarized. Figure 10.1 provides a pictorial representation of the complex interplay of the various physiological systems.
3 Rationale for Exercise Training
Exercise training has been found to have numerous benefits on the cardiovascular system [31, 32] and skeletal muscle systems. Patients with HF and COPD have been shown to respond favorably to exercise training with marked improvements in exercise capacity, quality of life and longevity [33]. There exists similarity between limitation to exercise performance in both PH and HF. Both conditions have contributions from central factors, vascular function, respiratory system and peripheral muscles [6, 34]. Studies in HF have suggested potential mechanisms for improvements from the changes in the neurohumoral systems, endothelial function, anti-inflammatory effects, cardiovascular effects and skeletal muscle changes [35]. Considering these effects observed in HF, it is only logical to assume that these same effects in a patient with PH would no doubt improve outcomes with exercise training. Indeed it was the anti-oxidant effects and improved vascular function of studies in HF that drove the hypothesis for exercise training in PH [36].
Since the publication of the first randomized controlled trial of exercise training study in humans [36], studies have continued to demonstrate significant benefits with exercise training on cardiorespiratory fitness (peak V̇O2), functional capacity (6 min walk distance-6MWD) and peripheral and respiratory muscle function in individuals with PH. In the first review on this topic Desai and Channick (2008) highlighted the rationale for exercise training in 2008 [37]. These authors also hypothesized the benefits of exercise in chronic obstructive pulmonary disease (COPD) and their mechanisms as potential reasons to advocate exercise training. Since then, the recent systematic reviews of the existing literature have supported the benefits of exercise training on various functional outcomes [38,39,40]. Thus, it appears that exercise training through various mechanisms, which still need to be elucidated, have an impact on clinical and functional outcomes in PH.
4 Evidence on Exercise Training in Pulmonary Hypertension
Evidence on the effects of exercise training in PH were limited till early 2000. However, there has been a steady increase in the number of trails registered in various clinical trial registries across the world [41]. Recently, there have been excellent reviews and meta-analysis on this topic and each one has focused on various aspects [38,39,40]. Moreover, the results of a Cochrane review on randomized controlled trials, has also recently been published [42].
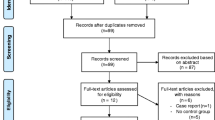
This section compiles information from all these reviews and further contributes to articles available since the publication of these reviews. Among these three recent reviews, only one has included all forms of study designs [40] while the other two have focused only on randomized controlled trials [38, 39]. From the 15 trials included in the systematic review, there have been an additional four articles that have been published since the last search performed in the review [43,44,45,46], thus making a total of 19 observational and randomized controlled trial of exercise training published till date. A summary of these studies are included in Table 10.1.
As observed, most of the studies have observed changes in functional capacity and quality of life. Recent meta-analysis and systematic reviews have reiterated the benefits seen with regard to exercise capacity, functional class and quality of life [38,39,40, 47]. At present, conclusions from the randomized clinical trials [36, 44, 45, 48,49,50] suggest significant benefits in terms of exercise capacity (peak V̇O2), 6 min walk distance (6MWD), right ventricular systolic pressure (RVSP), mean PAP, PVR and quality of life. In addition to these studies, a recent paper assessed the use of home based exercises in children with PH [51]. However, in spite of the growing literature, the evidence base for exercise training remains narrow and the quality of the evidence remains low [42]. Furthermore, much of our evidence comes from the same group of authors that used an inpatient training program followed by a home based program. This highly supervised, expensive model of exercise training would be difficult to incorporate into most standard rehabilitation programs around the world [52, 53].
5 Adaptations to Exercise Training
With the evidence supporting the use of exercise training to improve functional capacity and quality of life in PH, there are certain adaptations that could occur as a result of the exercise training programs. Most of the studies have utilized a combination of aerobic and resistance training interventions [36, 45, 54,55,56,57,58,59,60,61] with only a few relying solely on aerobic [62,63,64], peripheral [65] and respiratory muscle strengthening [44] and home-based exercise training [43].
The effects reported from all the studies have reflected to a certain extent changes in the RV and muscle strength. Few have also reported haemodynamic changes. Single groups studies have not reported significant changes, though there is a minimal decrease by 2–4 mmHg in mPAP at rest when assessed by either echocardiography or right heart catheterisation [36, 45] while some have reported no change at all [43]. Recent meta-analysis have also reported changes in peak systolic pulmonary artery pressure with exercise training (−3.66 mm Hg; 95% CI: -5.45, −1.87; p = 0.694) [39]. Recently, an abstract presented at the recent American Thoracic Society conference (2016) found that supervised exercise training improve RV function (with respect to RV stroke volume and ejection fraction) when evaluated with cardiac MRI [66]. RV function determined from tricuspid annular plane systolic excursion (TAPSE) was also seen to improve from 23 ± 10 mm to 21 ± 3 mm in a single group observational study. This is the only study till date to report improvements in TAPSE following exercise training [46]. Another cardiac MRI study also identified that 3 weeks of exercise training produced a reduction pulmonary artery flow resistance along with an increase in pulmonary perfusion [49]. Figure 10.2 summarizes the various adaptations reported from all the studied till date.
Adaptations to exercise training programs in pulmonary hypertension form published literature
Abbreviations: RV – Right ventricle; EF – Ejection fraction; TAPSE – Tricuspid annulus planar systolic excursion; PVR – Pulmonary Vascular resistance; mPAP – mean pulmonary artery pressure; CO – Cardiac output; Pul – Pulmonary; FEV1 – Forced expiratory volume in 1st second; FVC – Forced vital capacity
6 Assessing and Prescribing Exercise in Pulmonary Hypertension
Assessment of exercise capacity in PH has always played an important role. All through the years, the 6 min walk test (6MWT) has been used extensively due to its ease of administration. The importance of evaluating exercise capacity with CPX and 6MWT has been reiterated in the recent ESC-ERS guidelines as well [3]. The use of CPX or other functional tests like the 6 min walk test (6MWT), shuttle walk test, functional walk test or bag and carry test are dependent on setting and expertise [67]. However, CPX and 6MWT are the most commonly used methods for evaluating exercise capacity.
6.1 Cardiopulmonary Exercise Testing
CPX is the gold standard for the evaluation of exercise capacity and currently receives evidence based recommendations for diagnosis, prognostication and evaluation of therapeutic efficacy [21]. CPX is a non-invasive method that estimates the expired ventilation and concentration of oxygen and carbon dioxide through a breath-by-breath gas analyser while the patient exercises on a bicycle or treadmill. Various outcomes like peak V̇O2, peak respiratory exchange ratio (RER), anaerobic/lactate threshold, ventilatory efficiency (V̇E/V̇CO2 slope), end tidal CO2 (PETCO2) and dead space to tidal volume ratio (Vd/Vt) [68, 69]. In addition to these variables, heart rate, blood pressure, oxygen saturation, Borg’s rating of perceived exertion and symptoms are further determined during the test. For the evaluation of PH however, peak V̇O2, V̇E/V̇CO2 slope, PETCO2, anaerobic threshold and Vd/Vt are the primary variables of interest.
Many of these variables have also been shown to have prognostic importance [21, 67]. High V̇E/V̇CO2 slope (>45), low peak V̇O2 (<10 ml/kg/min) and low PETCO2 (no value established as yet) were consistently seen to have poor prognosis in PH with level IIb recommendation for Level B evidence [21]. In addition, CPX has also been found to have a role in determining therapeutic efficacy having a IIb level of recommendation with Level C evidence. This may change in the current years, as to date, nearly all the existing PH research has relied very heavily on the popular 6 min walk test (6MWT).
As with standard exercise testing procedures, pre-testing screening and calibration of equipment is important. Adequate infrastructure and space for the exercise testing lab are crucial considering the amount of equipment that will need to be stored. Presence of emergency resuscitation equipment and personnel trained in both basic and/or advanced resuscitation need to be available. In addition, to life saving skills, competencies in ECG recording and interpretations are highly important [70]. Finally, the level of experience in the individuals is important for CPX testing, with more experienced centres showing greater reliability [71].
6.2 Six Minutes Walk Test
The 6MWT has been widely used in PH studies as a measure of functional outcome and has been used as recommended by the American Thoracic Society and European Respiratory Societies [72, 73]. The recent guidelines continue to emphasis the need for measurement of the 6MWD as an outcome for risk stratification [3]. The 6MWT has been used as an outcome measure in a number of clinical trials and the minimally important difference (MID) well characterized in the PH population [72, 74]. The 6MWT has been found to elicit a maximal cardiovascular response among patients with PH [75]. The test has been found to predict peak VO2 in both children and adults with PH with varying levels of accuracy (r = 0.87; p < 0.001 and r = 0.68; p < 0.001 respectively) [76]. In adults however, ventilatory efficiency was found to be lower during the 6MWT with only a moderate correlation between 6MWD and peak VO2 from CPX (r = 0.49) [75]. Yet, the worsening of 6MWD has been shown to be associated with poor prognosis, though improvements in 6MWD have not been found to be reflective of mortality benefits as yet [77]. However, this is now a topic of controversy as a recent study did not find any changes in 6MWD and between patients on monotherapy and triple therapy despite differences being observed in their V̇E/V̇CO2 and PETCO2 [25].
Despite the fair amount of inaccuracy of the 6MWT to predict peak V̇O2, the fact that the test elicits a maximal cardiovascular response makes it an ideal alternative to test patients with PH [78]. This holds good in low resource settings where the cost for setting up a CPX lab can go higher than USD50000 [67].
The 6MWT though used frequently is not without any risks. There are a few studies that have reported adverse events with the 6MWT which further increases the need for close monitoring during the test [40, 79, 80]. The use of telemetric monitoring systems (where available) during the test or having the supervisor of the test walk behind the individual being tested could be an appropriate safeguard to this group of patients who can experience sudden arrhythmias resulting in cardiac arrest even during a sub-maximal test [72].
7 Setting Up a Pulmonary Hypertension Rehabilitation Center
Rehabilitation centers focusing on PH are limited. Currently, these patients are enrolled along with HF cardiac rehabilitation programs or pulmonary rehabilitation programs. However, there are no dedicated programs for PH per se. The Pulmonary Hypertension Association, USA has initiated a program to establish PH care centers (PHCC) across the various parts of the US (accessed from: http://www.phassociation.org/PHCareCenters). This program accredits centers with expertise in the evaluation and management of PH to improve outcomes of patients with PH [81]. The goal of these centers are to provide evidence based team care to patients with PH, improve access of specialized health care, promote adherence to guidelines to optimize research and clinical services, and promote awareness [82].
Development of centers for comprehensive care require expertise in staff and support services, facilitates and research. Despite these guidelines for PH specific centers, there is limited mention for the need of physiotherapists/exercise physiologists as part of the comprehensive healthcare team. Though referral to cardiac or pulmonary rehabilitation programs are described, this is not considered as a part of the center requirements. This section, expert driven, will provide a framework for the development of a PH rehabilitation center which has been developed and modified from both the cardiac and pulmonary rehabilitation programs (Tables 10.2 and 10.3).
The PH rehabilitation center working along with PH care centers or as a part of them can be developed on the model of both cardiac and pulmonary rehabilitation centers. Table 10.2 provides a list a staff, facilities and services that should be made available at a PH rehabilitation center. Though these are components that would be desirable to have, it should also be kept in mind that even the use of simple cost effective methods are acceptable. The recent model for and consensus statement on low cost cardiac rehabilitation could be used as a source to guide setting exercise training programs in low resource settings [83, 84] even for PH; though this will need to be evaluated 8.
8 Future Recommendations
The future for exercise in PH is bright. There is scope numerous avenues of research in this area. At present, greater good quality studies are required to further systematically assess the effects of exercise training through various models, intensities and modes on cardiovascular and haemodynamic outcomes. In addition, long term studies assessing time to clinical worsening and hard outcomes like mortality need to be addressed. If PH rehabilitation centers can be developed, they will offer valuable evidence through prospective databases and registries on the effects of exercise.
9 Conclusion
Exercise intolerance is a major hindrance to function and poor quality of life among patients with PH. Evaluation of the mechanisms of intolerance are important prior to initiation of rehabilitation. Cardiopulmonary exercise testing and the use of functional tests are useful in the assessment and prognosis of these patients. Exercise training interventions are showing promising results, however, there is a need for more generalizable results and feasible exercise training protocols for patients with PH. Nevertheless, exercise training will have an impact on the various physiological systems of the body and will result in numerous adaptations which will help improve function and quality of life (Table 10.3).
References
Hoeper MM, Humbert M, Souza R et al (2016) A global view of pulmonary hypertension. Lancet Respir Med 4(4):306–322
Simonneau G, Gatzoulis MA, Adatia I et al (2013) Updated clinical classification of pulmonary hypertension. J Am Coll Cardiol 62(25 Suppl):D34–D41
Galie N, Humbert M, Vachiery JL et al (2016) 2015 ESC/ERS guidelines for the diagnosis and treatment of pulmonary hypertension: the joint task force for the diagnosis and treatment of pulmonary hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS): endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC), International Society for Heart and Lung Transplantation (ISHLT). Eur Heart J 37(1):67–119
Lau EM, Manes A, Celermajer DS et al (2011) Early detection of pulmonary vascular disease in pulmonary arterial hypertension: time to move forward. Eur Heart J 32(20):2489–2498
Rabinovitch M (2008) Molecular pathogenesis of pulmonary arterial hypertension. J Clin Invest 118(7):2372–2379
Babu AS, Arena R, Myers J et al (2016) Exercise intolerance in pulmonary hypertension: mechanism, evaluation and clinical implications. Expert Rev Respir Med 10(9):1–12
Waxman AB (2012) Exercise physiology and pulmonary arterial hypertension. Prog Cardiovasc Dis 55(2):172–179
Fowler RM, Gain KR, Gabbay E (2012) Exercise intolerance in pulmonary arterial hypertension. Pulm Med 2012:359204
Kanwar MK, Thenappan T, Vachiery JL (2016) Update in treatment options in pulmonary hypertension. J Heart Lung Transplant 35(6):695–703
Mathai SC, Suber T, Khair RM et al (2016) Health-related quality of life and survival in pulmonary arterial hypertension. Ann Am Thorac Soc 13(1):31–39
Halank M, Einsle F, Lehman S et al (2013) Exercise capacity affects quality of life in patients with pulmonary hypertension. Lung 191(4):337–343
Sun XG, Hansen JE, Oudiz RJ et al (2001) Exercise pathophysiology in patients with primary pulmonary hypertension. Circulation 104(4):429–435
Naeije R, Vanderpool R, Dhakal BP et al (2013) Exercise-induced pulmonary hypertension: physiological basis and methodological concerns. Am J Respir Crit Care Med 187(6):576–583
Roberts JD, Forfia PR (2011) Diagnosis and assessment of pulmonary vascular disease by Doppler echocardiography. Pulm Circ 1(2):160–181
Lin AC, Strugnell WE, Seale H et al (2016) Exercise cardiac MRI-derived right ventriculo-arterial coupling ratio detects early right ventricular maladaptation in PAH. Eur Respir J 48(6):1797–1800
Lai YC, Potoka KC, Champion HC et al (2014) Pulmonary arterial hypertension: the clinical syndrome. Circ Res 115(1):115–130
Kasner M, Westermann D, Steendijk P et al (2012) Left ventricular dysfunction induced by nonsevere idiopathic pulmonary arterial hypertension: a pressure-volume relationship study. Am J Respir Crit Care Med 186(2):181–189
Spruijt OA, de Man FS, Groepenhoff H et al (2015) The effects of exercise on right ventricular contractility and right ventricular-arterial coupling in pulmonary hypertension. Am J Respir Crit Care Med 191(9):1050–1057
Guazzi M, Arena R (2010) Pulmonary hypertension with left-sided heart disease. Nat Rev Cardiol 7(11):648–659
Borlaug BA (2014) Mechanisms of exercise intolerance in heart failure with preserved ejection fraction. Circ J 78(1):20–32
Pinkstaff SO, Burger CD, Daugherty J et al (2016) Cardiopulmonary exercise testing in patients with pulmonary hypertension: clinical recommendations based on a review of the evidence. Expert Rev Respir Med 10(3):279–295
Ferrazza AM, Martolini D, Valli G et al (2009) Cardiopulmonary exercise testing in the functional and prognostic evaluation of patients with pulmonary diseases. Respiration 77(1):3–17
Weitzenblum E, Chaouat A, Kessler R (2013) Pulmonary hypertension in chronic obstructive pulmonary disease. Pneumonol Alergol Pol 81(4):390–398
Yasunobu Y, Oudiz RJ, Sun XG et al (2005) End-tidal PCO2 abnormality and exercise limitation in patients with primary pulmonary hypertension. Chest 127(5):1637–1646
Morris NR, Seale H, Harris J et al (2017) Gas exchange responses during 6-min walk test in patients with pulmonary arterial hypertension. Respirology 22(1):165–171
Tolle J, Waxman A, Systrom D (2008) Impaired systemic oxygen extraction at maximum exercise in pulmonary hypertension. Med Sci Sports Exerc 40(1):3–8
Velez-Roa S, Ciarka A, Najem B et al (2004) Increased sympathetic nerve activity in pulmonary artery hypertension. Circulation 110(10):1308–1312
Bauer R, Dehnert C, Schoene P et al (2007) Skeletal muscle dysfunction in patients with idiopathic pulmonary arterial hypertension. Respir Med 101(11):2366–2369
de Man FS, van Hees HW, Handoko ML et al (2011) Diaphragm muscle fiber weakness in pulmonary hypertension. Am J Respir Crit Care Med 183(10):1411–1418
Kuran Aslan G, Akinci B, Yeldan I et al (2016) Respiratory muscle strength in patients with pulmonary hypertension: the relationship with exercise capacity, physical activity level and quality of life. Clin Respir J. doi:10.1111/crj.12582
Swift DL, Lavie CJ, Johannsen NM et al (2013) Physical activity, cardiorespiratory fitness, and exercise training in primary and secondary coronary prevention. Circ J 77(2):281–292
Lavie CJ, Arena R, Swift DL et al (2015) Exercise and the cardiovascular system: clinical science and cardiovascular outcomes. Circ Res 117(2):207–219
Rochester CL, Vogiatzis I, Holland AE et al (2015) An official American Thoracic Society/European Respiratory Society policy statement: enhancing implementation, use, and delivery of pulmonary rehabilitation. Am J Respir Crit Care Med 192(11):1373–1386
Phillips SA, Vuckovic K, Cahalin LP et al (2015) Defining the system: contributors to exercise limitations in heart failure. Heart Fail Clin 11(1):1–16
Piepoli MF (2013) Exercise training in chronic heart failure: mechanisms and therapies. Neth Hear J 21(2):85–90
Mereles D, Ehlken N, Kreuscher S et al (2006) Exercise and respiratory training improve exercise capacity and quality of life in patients with severe chronic pulmonary hypertension. Circulation 114(14):1482–1489
Desai SA, Channick RN (2008) Exercise in patients with pulmonary arterial hypertension. J Cardiopulm Rehabil Prev 28(1):12–16
Yuan P, Yuan XT, Sun XY et al (2015) Exercise training for pulmonary hypertension: a systematic review and meta-analysis. Int J Cardiol 178:142–146
Pandey A, Garg S, Khunger M et al (2015) Efficacy and safety of exercise training in chronic pulmonary hypertension: systematic review and meta-analysis. Circ Heart Fail 8(6):1032–1043
Babu AS, Padmakumar R, Maiya AG et al (2016) Effects of exercise training on exercise capacity in pulmonary arterial hypertension: a systematic review of clinical trials. Heart Lung Circ 25(4):333–341
Babu AS, Padmakumar R, Maiya AG (2013) A review of ongoing trials in exercise based rehabilitation for pulmonary arterial hypertension. Indian J Med Res 137(5):900–906
Morris NR, Kermeen F, Holland AE (2017) Exercise-based rehabilitation programmes for pulmonary hypertension. Cochrane Database Syst Rev 1:Cd011285
Inagaki T, Terada J, Tanabe N et al (2014) Home-based pulmonary rehabilitation in patients with inoperable or residual chronic thromboembolic pulmonary hypertension: a preliminary study. Respir Invest 52(6):357–364
Saglam M, Arikan H, Vardar-Yagli N et al (2015) Inspiratory muscle training in pulmonary arterial hypertension. J Cardiopulm Rehabil Prev 35(3):198–206
Ehlken N, Lichtblau M, Klose H et al (2016) Exercise training improves peak oxygen consumption and haemodynamics in patients with severe pulmonary arterial hypertension and inoperable chronic thrombo-embolic pulmonary hypertension: a prospective, randomized, controlled trial. Eur Heart J 37(1):35–44
Bussotti M, Gremigni P, Pedretti RF et al (2016) Effects of an outpatient service rehabilitation programme in patients affected by pulmonary arterial hypertension: an observational study. Cardiovasc Hematol Disord Drug Targets 16(3)
Buys R, Avila A, Cornelissen VA (2015) Exercise training improves physical fitness in patients with pulmonary arterial hypertension: a systematic review and meta-analysis of controlled trials. BMC Pulm Med 15:40
Chan L, Chin LM, Kennedy M et al (2013) Benefits of intensive treadmill exercise training on cardiorespiratory function and quality of life in patients with pulmonary hypertension. Chest 143(2):333–343
Ley S, Fink C, Risse F et al (2013) Magnetic resonance imaging to assess the effect of exercise training on pulmonary perfusion and blood flow in patients with pulmonary hypertension. Eur Radiol 23(2):324–331
Weinstein AA, Chin LM, Keyser RE et al (2013) Effect of aerobic exercise training on fatigue and physical activity in patients with pulmonary arterial hypertension. Respir Med 107(5):778–784
Zoller D, Siaplaouras J, Apitz A et al (2016) Home exercise training in children and adolescents with pulmonary arterial hypertension: a pilot study. Pediatr Cardiol 38(1):191–198
Babu AS, Padmakumar R, Maiya AG (2015) Letter by Babu et al regarding article, “advances in therapeutic interventions for patients with pulmonary arterial hypertension”. Circulation 132(12):e153
Lau EM, Montani D, Jais X et al (2015) Response to letter regarding article, “advances in therapeutic interventions for patients with pulmonary arterial hypertension”. Circulation 132(12):e154
Grunig E, Ehlken N, Ghofrani A et al (2011) Effect of exercise and respiratory training on clinical progression and survival in patients with severe chronic pulmonary hypertension. Respiration 81(5):394–401
Nagel C, Prange F, Guth S et al (2012) Exercise training improves exercise capacity and quality of life in patients with inoperable or residual chronic thromboembolic pulmonary hypertension. PLoS One 7(7):e41603
Grunig E, Lichtblau M, Ehlken N et al (2012) Safety and efficacy of exercise training in various forms of pulmonary hypertension. Eur Respir J 40(1):84–92
Grunig E, Maier F, Ehlken N et al (2012) Exercise training in pulmonary arterial hypertension associated with connective tissue diseases. Arthritis Res Ther 14(3):R148
Becker-Grunig T, Klose H, Ehlken N et al (2013) Efficacy of exercise training in pulmonary arterial hypertension associated with congenital heart disease. Int J Cardiol 168(1):375–381
Grunig E, Tiede H, Enyimayew EO et al (2013) Assessment and prognostic relevance of right ventricular contractile reserve in patients with severe pulmonary hypertension. Circulation 128(18):2005–2015
Ehlken N, Verduyn C, Tiede H et al (2014) Economic evaluation of exercise training in patients with pulmonary hypertension. Lung 192(3):359–366
Kabitz HJ, Bremer HC, Schwoerer A et al (2014) The combination of exercise and respiratory training improves respiratory muscle function in pulmonary hypertension. Lung 192(2):321–328
Shoemaker MJ, Wilt JL, Dasgupta R et al (2009) Exercise training in patients with pulmonary arterial hypertension: a case report. Cardiopulm Phys Ther J 20(4):12–18
Mainguy V, Maltais F, Saey D et al (2010) Effects of a rehabilitation program on skeletal muscle function in idiopathic pulmonary arterial hypertension. J Cardiopulm Rehabil Prev 30(5):319–323
Fox BD, Kassirer M, Weiss I et al (2011) Ambulatory rehabilitation improves exercise capacity in patients with pulmonary hypertension. J Card Fail 17(3):196–200
de Man FS, Handoko ML, Groepenhoff H et al (2009) Effects of exercise training in patients with idiopathic pulmonary arterial hypertension. Eur Respir J 34(3):669–675
Morris N, Strugnell W, Seale H et al (2016) Outpatient-based rehabilitation program improves right ventricular function in patients with pulmonary hypertension: results from a pilot study. In: A69 PULMONARY REHABILITATION IN PULMONARY HYPERTENSION, INTERSTITIAL LUNG DISEASE, AND OTHER CONDITIONS: WHAT’S NEW? Am Thorac Soc 193:A2319–A2319
Babu AS, Myers J, Arena R et al (2013) Evaluating exercise capacity in patients with pulmonary arterial hypertension. Expert Rev Cardiovasc Ther 11(6):729–737
Guazzi M, Adams V, Conraads V et al (2012) EACPR/AHA joint scientific statement. Clinical recommendations for cardiopulmonary exercise testing data assessment in specific patient populations. Eur Heart J 33(23):2917–2927
Paolillo S, Farina S, Bussotti M et al (2012) Exercise testing in the clinical management of patients affected by pulmonary arterial hypertension. Eur J Prev Cardiol 19(5):960–971
Colquhoun D, Freedman B, Cross D et al (2015) Clinical exercise stress testing in adults (2014). Heart Lung Circ 24(8):831–837
Oudiz RJ, Barst RJ, Hansen JE et al (2006) Cardiopulmonary exercise testing and six-minute walk correlations in pulmonary arterial hypertension. Am J Cardiol 97(1):123–126
Holland AE, Spruit MA, Troosters T et al (2014) An official European Respiratory Society/American Thoracic Society technical standard: field walking tests in chronic respiratory disease. Eur Respir J 44(6):1428–1446
American Thoracic Society (2002) ATS statement: Guidelines for the six-minute walk test. Am J Respir Crit Care Med 166(1):111–117
Mathai SC, Puhan MA, Lam D et al (2012) The minimal important difference in the 6-minute walk test for patients with pulmonary arterial hypertension. Am J Respir Crit Care Med 186(5):428–433
Deboeck G, Niset G, Vachiery JL et al (2005) Physiological response to the six-minute walk test in pulmonary arterial hypertension. Eur Respir J 26(4):667–672
Zapico AG, Fuentes D, Rojo-Tirado MA et al (2016) Predicting peak oxygen uptake from the 6-minute walk test in patients with pulmonary hypertension. J Cardiopulm Rehabil Prev 36(3):203–208
Farber HW, Miller DP, McGoon MD et al (2015) Predicting outcomes in pulmonary arterial hypertension based on the 6-minute walk distance. J Heart Lung Transplant 34(3):362–368
De Carvalho ET, Arakelian V, Costa IP et al (2016) Functional performance tests elicit a maximal cardiovascular response in patients with pulmonary hypertension. Am J Respir Crit Care Med 193:A2315
Seale H, Harris J, Hall K et al (2011) The hidden risk of a 6-minute walk test in pulmonary arterial hypertension. Pulm Circ 1:S37
Morris NR, Seale H, Harris J et al (2015) Serious adverse events during a 6-min walk test in patients with pulmonary hypertension. Eur Respir J 45(4):1179–1182
Sahay S, Melendres-Groves L, Pawar L et al (2016) Pulmonary hypertension care center network improving care and outcomes in PH. Chest 151(4):749–754
Chakinala MM, Duncan M, Wirth J (2016) Managing the patient with pulmonary hypertension: specialty care centers, coordinated care, and patient support. Cardiol Clin 34(3):489–500
Grace SL, Turk-Adawi KI, Contractor A et al (2016) Cardiac rehabilitation delivery model for low-resource settings. Heart 102(18):1449–1455
Grace SL, Turk-Adawi KI, Contractor A et al (2016) Cardiac rehabilitation delivery model for low-resource settings: an International Council of Cardiovascular Prevention and Rehabilitation Consensus Statement. Prog Cardiovasc Dis 59(3):303–322
Martinez-Quintana E, Miranda-Calderin G, Ugarte-Lopetegui A et al (2010) Rehabilitation program in adult congenital heart disease patients with pulmonary hypertension. Congenit Heart Dis 5(1):44–50
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Babu, A.S., Arena, R., Morris, N.R. (2017). Evidence on Exercise Training in Pulmonary Hypertension. In: Xiao, J. (eds) Exercise for Cardiovascular Disease Prevention and Treatment. Advances in Experimental Medicine and Biology, vol 1000. Springer, Singapore. https://doi.org/10.1007/978-981-10-4304-8_10
Download citation
DOI: https://doi.org/10.1007/978-981-10-4304-8_10
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-10-4303-1
Online ISBN: 978-981-10-4304-8
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)