Abstract
Despite a low incidence, the mortality rate of pancreatic cancer (PC) is high. Early detection and optimal treatment based on the findings of various imaging modalities are essential to improve the survival rate of patients with PC. Multi-detector (MD) row computed tomography (CT) is currently the standard imaging modality for evaluating PC worldwide. However, 10% of PC tumors can be iso-attenuated on CT and thus invisible, especially when ≤2 cm. Endoscopic ultrasonography (EUS) can detect tumors that are <10 cm at a higher rate than CT and might be useful for diagnosing high-grade pancreatic intraepithelial neoplasia (PanIN3) as well as magnetic resonance cholangiopancreatography, as they can both detect local irregular stenosis of the pancreatic duct.
Population-based screening is not feasible due to the low incidence of PC, but it is advisable for high-risk patients, in whom premalignant lesions can be detected using EUS.
The introduction of recent novel techniques such as contrast-enhanced EUS and elastography might improve the accuracy of EUS.
We reviewed the role of EUS in the detection and diagnosis of early-stage PC.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Pancreatic cancer (PC) is the deadliest of all solid malignancies. The prognosis of patients with PC is extremely poor, as vast majority of PC is diagnosed only at an advanced stage. Over 30,000 patients died of PC in Japan during 2013, and this number is expected to rise. It is projected that PC will surpass breast, prostate and colorectal cancer to become the second leading cause of cancer-related death in the USA by 2030 [1]. Therefore, PC remains one of the greatest challenges in the fight against cancer in the twenty-first century [2]. Since the poor prognosis is attributed to difficulties with diagnosis at an early stage, early detection might offer the best hope for a cure. Therefore, detecting PC at the earliest possible stage at which it is potentially curable and identifying precursor lesions have received considerable focus. PC is usually detected by computed tomography (CT) and/or magnetic resonance imaging (MRI) with magnetic resonance cholangiopancreatography (MRCP), or endoscopic ultrasound (EUS). Although multi-detector (MD) row CT is almost universally utilized in PC evaluation, its rate of detecting small pancreatic masses is low. On the other hand, EUS can detect small pancreatic masses with high sensitivity. This chapter reviews early PC diagnosis using EUS (Table 1.1).
2 EUS (Fig. 1.1)
The EUS equipment includes probes with different imaging methods: radial probes allow 360° imaging perpendicular to the long axis, and convex probes allow imaging along a plane parallel to the long axis of the instrument. The former only allows diagnostic imaging, whereas the latter was developed for fine-needle aspiration (FNA) [3, 4]. EUS uses high ultrasound frequencies, with imaging from the stomach or duodenum providing high resolution, real-time images of the pancreas. This modality therefore plays an important role in evaluating pancreatic diseases.
3 Early Diagnosis of PC Using EUS
MDCT evaluation of patients with suspected PC is the standard preoperative assessment at most medical institutions. This is because MDCT has good spatial and temporal resolution with wide anatomical coverage, and thus permits both comprehensive local and distant disease assessment during a single session [5, 6].
Among cross-sectional imaging modalities, the performance of MDCT is optimal for evaluating vascular involvement, which is the most important predictor of tumor resectability [7,8,9]. However, about 10% of PCs are iso-attenuating relative to the background pancreatic parenchyma (Fig. 1.2) [10]. CT enhancement of the PC and of pancreatic parenchyma surrounding a tumor is correlated with the degree of fibrosis. Contrast material is retained in PC with a predominant fibrous component. A similar degree of fibrosis in a tumor and surrounding pancreatic parenchyma might lead to overlapping enhancement on MDCT that could prevent the detection of PC, especially when tumors are ≤2 cm [11,12,13].
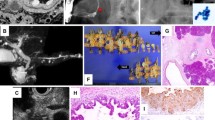
Case: a 8 mm pancreatic cancer with invasion. MRCP (a) showed short duct stenosis in pancreatic body (arrow). Contrast-enhanced CT (b) could not detect the mass in pancreatic body (arrow ahead) though main pancreatic duct (MPD) was dilated and disrupted in the body. (c) EUS could detect the low echoic mass with unclear margin. (d) Microscopic findings revealed an 8 mm tumor with invasion accompanied with 20 mm fibrosis
On the other hand, PC appears on EUS images as heterogeneous hypoechoic masses with irregular margins, which allows very high sensitivity for detecting PC [14, 15].
It is considered one of the most accurate means of detecting pancreatic focal lesions, especially when tumors are ≤2 cm [16,17,18,19].
Recent reports indicate that EUS can detect tumors <10 mm [20,21,22]. The sensitivity of EUS for detection of 25 small PC with size <10 mm was 84%, among eight Japanese high-volume centers [23]. Therefore, all patients with obstructive jaundice or unexplained pancreatic duct dilation, in whom CT or MRI do not definitively identify pancreatic lesions should be assessed using EUS [24].
However, EUS can miss a true pancreatic mass in patients with chronic pancreatitis, a diffusely infiltrating carcinoma, a prominent ventral/dorsal split, or a recent episode (<4 weeks) of acute pancreatitis [25]. The potential for suboptimal visualization of the pancreatic gland for detection of PC by EUS and other imaging modalities should be acknowledged in the setting of acute or chronic pancreatitis. Acoustic shadowing caused by an indwelling biliary or pancreatic stents, or pancreatic stones can also interfere with the visualization of small pancreatic masses.
4 Pancreatic Intraepithelial Neoplasia (PanIN)
PC develops through stepwise progression from precursor lesions comprising pancreatic intraepithelial neoplasia (PanIN), mucinous cystic neoplasm (MCN), and intraductal papillary mucinous neoplasm (IPMN). Among these, PanIN is the most common precursor of PC [26]. PanIN are noninvasive epithelial proliferations within smaller pancreatic ducts (<0.5 mm) that can be flat or papillary and classified into low (PanIN-1), intermediate (PanIN-2), and high (PanIN-3) grades according to the degree of architectural and cellular atypia [26]. Based on mutations associated with each grade, normal ductal epithelium seems to progress through low-grade PanIN, high-grade PanIN, localized adenocarcinoma, and metastatic adenocarcinoma in that order. Detection of high-grade PanIN-3 would provide an optimal opportunity to reduce mortality from PC. It has been believed that PanIN cannot be reliably visualized using clinical imaging [27] as they typically arise in the small-caliber pancreatic ducts [26].
However, it has been recently suggested that PanIN is associated with localized parenchymal changes that may be detected by EUS [28, 29]. These parenchymal changes are characterized by acinar cell loss, proliferation of small ductular structures, and fibrosis referred to as lobulocentric atrophy (LCA) [30]. Localized fibrosis and/or LCA has been pathologically identified in parenchyma around PanIN-3 [21, 25, 28, 29, 31, 32]. A slightly low echoic lesion on EUS images might suggest localized fibrosis around PanIN-3 [28]. Maire et al. [29] reported that EUS changes corresponded to PanIN lesions in 83%. EUS also detected 69% of patients with PanIN lesions and 57% of those with PanIN3 lesions. However, EUS findings for PanIN lesion were not uniformed. For instance, Maire et al. [29] defined EUS findings of PanIN lesion were microcysts or hyper-echogenic foci resulting in a heterogeneous pattern. On the other hand, Hanada et al. [28] reported slightly low echoic lesion on EUS images were the findings of PanIN. However, it should be noted that these abnormalities on EUS are not specific to PanIN or early PC, and conversely, PanIN may well occur in the absence of LCA [30, 33]. Further studies are warranted to confirm these findings.
5 Surveillance of High-Risk Individuals
Familial pancreatic cancer (FPC) kindreds are defined as families with two or more first-degree relatives (FDR) affected with PC, in the absence of other cancers or familial diseases. Klein et al. found that the risk of developing PC was 4.5- vs. 32-fold depending on whether one or at least three FDR were affected, respectively [34, 35].
A multicenter prospective cohort study (CAPS 3) implemented by Canto et al. [36] included 216 high-risk individuals (HRI) (Peutz-Jeghers syndrome, n = 2; familial breast-ovarian cancer with at least one affected first- or second-degree relative with PC, n = 19; relatives of patients with FPC with at least two FDR, n = 195). All persons were evaluated by CT, MRI, and EUS, and 92 (42%) of 216 had at least one pancreatic mass (84 cystic and 3 solid) or a dilated pancreatic duct (n = 5) according to the findings of at least one of the imaging modalities. The prevalence of these lesions increased with age of the screened persons. Pancreatic abnormalities were detected by CT, MRI, and EUS in 11%, 33.3%, and 42.6% of the patients, respectively. Among the pancreatic lesions, 82 were IPMN, and three were neuroendocrine tumors. Five patients who were surgically treated had high-grade dysplasia in IPMN <3 cm and multiple intraepithelial neoplasms. Canto et al. concluded that screening asymptomatic HRI could detect curable noninvasive high-grade and multiple cystic lesions. Both EUS and MRI were more effective diagnostic screens for HRI than CT [37]. These findings showed that screening of high-risk families can detect early precancerous changes in the pancreas [35].
6 New Screening Modality Comprising Contrast EUS and Elastography
Conventional EUS sometimes cannot detect pancreatic tumors in patients with chronic pancreatitis, diffusely infiltrating carcinoma, or a recent episode of acute pancreatitis [25]. Contrast-enhanced (CH)-EUS and EUS elastography might help to improve the diagnostic accuracy of EUS.
Parenchymal perfusion and the pancreatic microvasculature can be visualized without artifacts by CH-EUS [38], and it is useful in the differential diagnosis of PC, especially small tumors [39, 40]. Fusaroli et al. [41] reported that pancreatic tumor visualization by CH-EUS is better than that of conventional EUS. A recent meta-analysis of 1139 patients found that the sensitivity and specificity of CE-EUS for a differential diagnosis of PC were 94% and 89%, respectively [39]. That study found that hypo-enhancing lesions on CE-EUS images were a sensitive and accurate predictor of PC.
Case: a 3 mm pancreatic cancer with invasion. Contrast-enhanced CT (a) could not detect the mass in pancreatic head (arrow ahead) although main pancreatic duct (MPD) was dilated. MRCP (b) showed short duct stenosis in pancreatic body (arrow). (c) EUS showed an 7 mm low echoic mass (arrow). (d) EUS-FNA was performed from the 7 mm low echoic mass using 22G FNA needle. (e) Cytology with Papanicolaou stain showed atypical cells consistent with adenocarcinoma. (f) Macroscopic findings revealed pancreatic adenocarcinoma with invasive components of 3 mm (arrow) with 8 mm surrounding fibrosis
Because CH-EUS is more sensitive, it can be used to identify targets of EUS-FNA [41,42,43] and might also help to avoid puncturing necrotic and inflammatory areas of malignant masses or hard and scirrhous areas of inflammatory masses, thus reducing the need for repeated FNA assessments.
Another emerging technology is EUS elastography, which provides real-time visualization of tissue stiffness. It is based on the premise that compression causes less strain in hard, rather than in soft tissues [44]. The results of recent investigations using EUS elastography for diagnosing pancreatic focal lesions are promising [45,46,47]. As malignant lesions are generally harder than normal adjacent tissue, measuring strain might help to classify pancreatic masses. Two meta-analyses recently found high pooled sensitivity (95–97%) and low pooled specificity (67–76%), for a differential diagnosis of solid pancreatic masses [48, 49].
However, CH-EUS and EUS elastography are not widely available and have yet to be widely tested as screening tools for PC [37, 50].
7 Early Diagnosis of PC Using EUS-FNA (Fig. 1.3)
Although EUS has high overall sensitivity, differentiating PC from other solid lesions based only on endosonographic features remains challenging. Specimens for histopathological diagnosis can be collected using EUS-guided FNA. Since its introduction in the early 1990s, EUS-FNA has emerged as a safe and accurate means of tissue diagnosis in patients with pancreaticobiliary disorders, particularly confirmed PC. The sensitivity and specificity of EUS-FNA for diagnosing pancreatic masses is 80–95% and 75–100%, respectively [51,52,53,54,55].
Uehara et al. [56] recently reported that EUS-FNA was 96% accurate for identifying pancreatic masses <10 mm in 23 patients. Thus, EUS-FNA is useful for confirming pancreatic tumors <10 mm.
References
Rahib L, Smith BD, Aizenberg R, et al. Projecting cancer incidence and deaths to 2030: the unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res. 2014;74:2913–21.
Gungor C, Hofmann BT, Wolters-Eisfeld G, et al. Pancreatic cancer. Br J Pharmacol. 2014;171:849–58.
Inui K, Kida M, Fujita N, et al. Standard imaging techniques in the pancreatobiliary region using radial scanning endoscopic ultrasonography. Dig Endosc. 2004;16:S118–S33.
Yamao K, Irisawa A, Inoue H, et al. Standard imaging techniques of endoscopic ultrasound-guided fine-needle aspiration using a curved linear array echoendoscope. Dig Endosc. 2007;19:S180–205.
Brennan DD, Zamboni GA, Raptopoulos VD, et al. Comprehensive preoperative assessment of pancreatic adenocarcinoma with 64-section volumetric CT 1. Radiographics. 2007;27:1653–66.
Lee ES, Lee JM. Imaging diagnosis of pancreatic cancer: a state-of-the-art review. World J Gastroenterol. 2014;20:7864–77.
Li H, Zeng MS, Zhou KR, et al. Pancreatic adenocarcinoma: signs of vascular invasion determined by multi-detector row CT. Br J Radiol. 2006;79:880–7.
Manak E, Merkel S, Klein P, et al. Resectability of pancreatic adenocarcinoma: assessment using multidetector-row computed tomography with multiplanar reformations. Abdom Imaging. 2009;34:75–80.
Gusmini S, Nicoletti R, Martinenghi C, et al. Vascular involvement in periampullary tumors: MDCT, EUS, and CDU. Abdom Imaging. 2009;34:514–22.
Prokesch RW, Chow LC, Beaulieu CF, et al. Isoattenuating pancreatic adenocarcinoma at multi-detector row CT: secondary signs. Radiology. 2002;224:764–8.
Yoon SH, Lee JM, Cho JY, et al. Small (</= 20 mm) pancreatic adenocarcinomas: analysis of enhancement patterns and secondary signs with multiphasic multidetector CT. Radiology. 2011;259:442–52.
Blouhos K, Boulas KA, Tsalis K, et al. The isoattenuating pancreatic adenocarcinoma: review of the literature and critical analysis. Surg Oncol. 2015;24:322–8.
Scialpi M, Cagini L, Pierotti L, et al. Detection of small (</= 2 cm) pancreatic adenocarcinoma and surrounding parenchyma: correlations between enhancement patterns at triphasic MDCT and histologic features. BMC Gastroenterol. 2014;14:16.
Ainsworth AP, Rafaelsen SR, Wamberg PA, et al. Is there a difference in diagnostic accuracy and clinical impact between endoscopic ultrasonography and magnetic resonance cholangiopancreatography? Endoscopy. 2003;35:1029–32.
Mertz HR, Sechopoulos P, Delbeke D. EUS, PET, and CT scanning for evaluation of pancreatic adenocarcinoma. Gastrointest Endosc. 2000;52:367–71.
Tamm E, Charnsangavej C. Pancreatic cancer: current concepts in imaging for diagnosis and staging. Cancer J. 2001;7:298–311.
Dewitt J, Devereaux BM, Lehman GA, et al. Comparison of endoscopic ultrasound and computed tomography for the preoperative evaluation of pancreatic cancer: a systematic review. Clin Gastroenterol Hepatol. 2006;4:717–25. quiz 664
Agarwal B, Abu-Hamda E, Molke KL, et al. Endoscopic ultrasound-guided fine needle aspiration and multidetector spiral CT in the diagnosis of pancreatic cancer. Am J Gastroenterol. 2004;99:844–50.
Volmar KE, Vollmer RT, Jowell PS, et al. Pancreatic FNA in 1000 cases: a comparison of imaging modalities. Gastrointest Endosc. 2005;61:854–61.
Hanada K, Okazaki A, Hirano N, et al. Effective screening for early diagnosis of pancreatic cancer. Best Pract Res Clin Gastroenterol. 2015;29:929–39.
Hanada K, Okazaki A, Hirano N, et al. Diagnostic strategies for early pancreatic cancer. J Gastroenterol. 2015;50:147–54.
Greenhalf W, Neoptolemos JP. Increasing survival rates of patients with pancreatic cancer by earlier identification. Nat Clin Pract Oncol. 2006;3:346–7.
Hanada KIT, Hirano N. Role of EUS for small pancreatic cancer less than 1 cm. Tan Sui. 2009;30:343–8.
Luz LP, Al-Haddad MA, Sey MS, et al. Applications of endoscopic ultrasound in pancreatic cancer. World J Gastroenterol. 2014;20:7808–18.
Bhutani MS, Gress FG, Giovannini M, et al. The no endosonographic detection of tumor (NEST) study: a case series of pancreatic cancers missed on endoscopic ultrasonography. Endoscopy. 2004;36:385–9.
Hruban RH, Adsay NV, Albores-Saavedra J, et al. Pancreatic intraepithelial neoplasia: a new nomenclature and classification system for pancreatic duct lesions. Am J Surg Pathol. 2001;25:579–86.
Shin EJ, Canto MI. Pancreatic cancer screening. Gastroenterol Clin N Am. 2012;41:143–57.
Hanada KIT, Yamao K. Diagnostic strategies for the early diagnosis of pancreatic cancer. Gastroenterol Endosc. 2012;54:3773–82.
Maire F, Couvelard A, Palazzo L, et al. Pancreatic intraepithelial neoplasia in patients with intraductal papillary mucinous neoplasms: the interest of endoscopic ultrasonography. Pancreas. 2013;42:1262–6.
Del Chiaro M, Segersvärd R, Lohr M, et al. Early detection and prevention of pancreatic cancer: is it really possible today. World J Gastroenterol. 2014;20:12118–31.
Takaori K, Matsusue S, Fujikawa T, et al. Carcinoma in situ of the pancreas associated with localized fibrosis: a clue to early detection of neoplastic lesions arising from pancreatic ducts. Pancreas. 1998;17:102–5.
Iiboshi T, Hanada K, Fukuda T, et al. Value of cytodiagnosis using endoscopic nasopancreatic drainage for early diagnosis of pancreatic cancer: establishing a new method for the early detection of pancreatic carcinoma in situ. Pancreas. 2012;41:523–9.
Petrone MC, Arcidiacono PG. New strategies for the early detection of pancreatic cancer. Expert Rev Gastroenterol Hepatol. 2016;10:157–9.
Klein AP, Brune KA, Petersen GM, et al. Prospective risk of pancreatic cancer in familial pancreatic cancer kindreds. Cancer Res. 2004;64:2634–8.
Klein AP. Identifying people at a high risk of developing pancreatic cancer. Nat Rev Cancer. 2013;13:66–74.
Canto MI, Hruban RH, Fishman EK, et al. Frequent detection of pancreatic lesions in asymptomatic high-risk individuals. Gastroenterology. 2012;142:796–804. quiz e14–5
Bhutani MS, Koduru P, Joshi V, et al. The role of endoscopic ultrasound in pancreatic cancer screening. Endosc Ultrasound. 2016;5:8–16.
Hou X, Jin Z, Xu C, et al. Contrast-enhanced harmonic endoscopic ultrasound-guided fine-needle aspiration in the diagnosis of solid pancreatic lesions: a retrospective study. PLoS One. 2015;10:e0121236.
Gong TT, Hu DM, Zhu Q. Contrast-enhanced EUS for differential diagnosis of pancreatic mass lesions: a meta-analysis. Gastrointest Endosc. 2012;76:301–9.
Sakamoto H, Kitano M, Suetomi Y, et al. Utility of contrast-enhanced endoscopic ultrasonography for diagnosis of small pancreatic carcinomas. Ultrasound Med Biol. 2008;34:525–32.
Fusaroli P, Spada A, Mancino MG, et al. Contrast harmonic echo-endoscopic ultrasound improves accuracy in diagnosis of solid pancreatic masses. Clin Gastroenterol Hepatol. 2010;8:629–34.e1–2.
Romagnuolo J, Hoffman B, Vela S, et al. Accuracy of contrast-enhanced harmonic EUS with a second-generation perflutren lipid microsphere contrast agent (with video). Gastrointest Endosc. 2011;73:52–63.
Kitano M, Kudo M, Yamao K, et al. Characterization of small solid tumors in the pancreas: the value of contrast-enhanced harmonic endoscopic ultrasonography. Am J Gastroenterol. 2012;107:303–10.
Ophir J, Cespedes I, Ponnekanti H, et al. Elastography: a quantitative method for imaging the elasticity of biological tissues. Ultrason Imaging. 1991;13:111–34.
Giovannini M, Thomas B, Erwan B, et al. Endoscopic ultrasound elastography for evaluation of lymph nodes and pancreatic masses: a multicenter study. World J Gastroenterol. 2009;15:1587–93.
Li X, Xu W, Shi J, et al. Endoscopic ultrasound elastography for differentiating between pancreatic adenocarcinoma and inflammatory masses: a meta-analysis. World J Gastroenterol. 2013;19:6284–91.
Opacic D, Rustemovic N, Kalauz M, et al. Endoscopic ultrasound elastography strain histograms in the evaluation of patients with pancreatic masses. World J Gastroenterol. 2015;21:4014–9.
Hu DM, Gong TT, Zhu Q. Endoscopic ultrasound elastography for differential diagnosis of pancreatic masses: a meta-analysis. Dig Dis Sci. 2013;58:1125–31.
Mei M, Ni J, Liu D, et al. EUS elastography for diagnosis of solid pancreatic masses: a meta-analysis. Gastrointest Endosc. 2013;77:578–89.
Nelsen EM, Buehler D, Soni AV, et al. Endoscopic ultrasound in the evaluation of pancreatic neoplasms-solid and cystic: a review. World J Gastrointest Endosc. 2015;7:318–27.
Eloubeidi MA, Varadarajulu S, Desai S, et al. A prospective evaluation of an algorithm incorporating routine preoperative endoscopic ultrasound-guided fine needle aspiration in suspected pancreatic cancer. J Gastrointest Surg. 2007;11:813–9.
Yoshinaga S, Suzuki H, Oda I, et al. Role of endoscopic ultrasound-guided fine needle aspiration (EUS-FNA) for diagnosis of solid pancreatic masses. Dig Endosc. 2011;23(Suppl 1):29–33.
Matsubayashi H, Matsui T, Yabuuchi Y, et al. Endoscopic ultrasonography guided-fine needle aspiration for the diagnosis of solid pancreaticobiliary lesions: clinical aspects to improve the diagnosis. World J Gastroenterol. 2016;22:628–40.
Puli SR, Bechtold ML, Buxbaum JL, et al. How good is endoscopic ultrasound-guided fine-needle aspiration in diagnosing the correct etiology for a solid pancreatic mass? A meta-analysis and systematic review. Pancreas. 2013;42:20–6.
Chen J, Yang R, Lu Y, et al. Diagnostic accuracy of endoscopic ultrasound-guided fine-needle aspiration for solid pancreatic lesion: a systematic review. J Cancer Res Clin Oncol. 2012;138:1433–41.
Uehara H, Ikezawa K, Kawada N, et al. Diagnostic accuracy of endoscopic ultrasound-guided fine needle aspiration for suspected pancreatic malignancy in relation to the size of lesions. J Gastroenterol Hepatol. 2011;26:1256–61.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer Science+Business Media Singapore
About this chapter
Cite this chapter
Hijioka, S., Yamao, K., Mizuno, N., Imaoka, H., Bhatia, V., Hara, K. (2017). Early Diagnosis of Pancreatic Cancer Using Endoscopic Ultrasound. In: Yamaue, H. (eds) Innovation of Diagnosis and Treatment for Pancreatic Cancer. Springer, Singapore. https://doi.org/10.1007/978-981-10-2486-3_1
Download citation
DOI: https://doi.org/10.1007/978-981-10-2486-3_1
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-10-2485-6
Online ISBN: 978-981-10-2486-3
eBook Packages: MedicineMedicine (R0)