Abstract
The prefrontal cortex (PFC) is the brain area that distinguishes us as uniquely human. It guides decision making based on past experience, allowing us to break away from reflexively responding to the sensory conditions on hand at any moment. Sequential planning, cognitive flexibility and rational thinking all rely on the PFC. When Patricia Goldman-Rakic began her research career in the 1970s, the PFC was a terra incognita. Her work dispelled the notion that higher cognitive function is beyond the reach of the scientific method and revealed the basic neurobiology governing prefrontal executive function. Goldman-Rakic started her journey by probing the behavior disrupted by lesions in the PFC. An early foray into anatomic tract tracing led to the discovery of columns of afferent terminal labeling in the PFC. From this foothold, she began a relentless quest to understand the modular organization of prefrontal architecture and how the vertical arrangement of functionally related neurons translates into the mechanistic underpinnings of working memory. She characterized neuronal activity in the PFC that forms the essence of spatial mnemonic capacity. Her work teased apart the intricacies of local neurocircuitry in the PFC and examined modulation of this circuitry by monoaminergic neurotransmitters, especially dopamine. Goldman-Rakic’s studies of executive function extended from the regional level in distinguishing the dorsal and ventral domains that process spatial and object information, respectively, to the subcellular level in mapping the precise distribution of dopamine receptors. Her life’s work stands today as the foundation for our contemporary understanding of prefrontal cortical function.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Patricia Goldman-Rakic’s career spanned decades and encompassed a wide range of research topics. Her studies touched on nearly every corner of the brain from the cerebral mantle to the brain stem and delved into the workings of many different transmitter systems, including glutamate, the monoamines, and neuropeptides. She investigated how basic brain mechanisms might be altered in diseased states like schizophrenia and examined the neurophysiologic action of antipsychotic drugs used to treat schizophrenia. However, there was never any question about where her real passion lay, that is in understanding the basic neurobiology of working memory function in the prefrontal cortex (PFC). Therefore, this review focuses on the centerpiece of her research: elucidating the neuroanatomic and neurophysiologic substrates of working memory function, their columnar organization, the manner in which dopamine (DA) afferent input modulates cognitive function in the PFC, and parcellation of the PFC into separate processing domains. The narrow focus by no means diminishes the importance or impact of her many satellite studies but rather underscores Dr. Goldman-Rakic’s conviction that characterizing the basic neurobiology of the PFC is essential for progress in understanding altered states induced by drugs or disease.
Goldman-Rakic was at the pinnacle of her career when her life tragically was cut short more than a decade ago in July, 2003. Her untimely passing is an enormous loss to the field of neurobiology. She left a number of studies in midstream. As a result, publication of her work continued for 5 years after her passing, allowing us a tantalizing glimpse of the direction it was taking, though we will never know what how much farther she could have advanced our understanding of the PFC. This review represents a retrospective look at Patricia Goldman-Rakic’s seminal findings in the PFC that is intended to refresh her work in the minds of her contemporaries and to introduce her findings to a new generation of neurobiologists and clinicians.
3.1 Columns in the Dorsolateral Prefrontal Cortex
Patricia Goldman-Rakic’s interest in the dorsolateral PFC (dlPFC) began with studies of the behavioral consequences of prefrontal lobectomy in nonhuman primates. Building on work by her mentors, H. Enger Rosvold and Mortimer Mishkin, Goldman-Rakic further distinguished the functional domains of the orbitofrontal cortex and the dlPFC and the capacity for recovery of function following early brain lesions (Goldman and Rosvold 1970; Goldman et al. 1970a, b, 1971, 1974; Goldman 1971, 1976). She found that the dlPFC, and in particular the cortex surrounding the principal sulcus, plays a critical role in mediating the working memory component of the spatial Delayed Response (DR) task (Goldman and Rosvold 1970). Working memory, the ability to hold information in the mental sketchpad is the essence of executive functioning of the PFC because this processing mechanism allows internalized representations of the world, rather than immediate sensory stimulation, to guide behavior (Baddeley and Hitch 1974; Goldman-Rakic 1987).
In the mid-1970s, Goldman-Rakic trained in Walle Nauta’s laboratory in order to master the then new neuroanatomic tracing method utilizing tritiated amino acids anterogradely transported from the site of injection to label terminal fields throughout the brain. They made a rather astonishing finding, that cortical terminal fields in diverse cortical areas including the dlPFC exhibit a columnar patterning (Goldman and Nauta 1977). For example, injection in the cortex surrounding the principal sulcus produces wide bands of terminal labeling in the contralateral principal sulcal cortex that extend throughout all six cortical layers with unlabeled bands interspersed (Fig. 3.1). Prior to this discovery, cortico-cortical columnar terminal labeling had been described in the sensory cortex (Jones et al. 1975; Shanks et al. 1975), and patterned thalamo-cortical labeling had been found in the visual cortex, corresponding to the vertically orientated ocular dominance columns identified by physiologic recordings (Hubel and Wiesel 1972). From these early anatomic studies, Goldman-Rakic recognized that the PFC exhibits the same modular organization as sensory cortices and therefore that higher cognitive functioning could be interrogated with neurophysiologic and neuroanatomic methods comparable to those used to discern the basis of perception. With this insight, Goldman-Rakic began what would become a life-long pursuit to eludicate the cellular basis of higher cognitive function as governed by the PFC. She chose to study the PFC in the rhesus macaque monkey because development, expansion, and specification of the cortical mantle in monkeys more closely parallels that of humans compared to other species (Rakic 1988).
Darkfield photomicrograph illustrating columns of terminal labeling in the principal sulcal cortex following an injection of tritiated amino acids in the principal sulcal cortex of the opposite hemisphere (From Goldman and Nauta 1977. Reproduced with permission of Elsevier BV in the format reuse in a book/textbook via Copyright Clearance Center)
Goldman-Rakic and her collaborators established that columnar patterning represents an overriding organizational principal of the PFC. Columnar terminal labeling is a feature of both callosal and long tract intrahemispheric connections to the PFC, as for example those from the posterior parietal cortex (Schwartz and Goldman-Rakic 1984; Cavada and Goldman-Rakic 1989). Moreover, columnar labeling in cortical areas and patchy, patterned terminal distributions in subcortical areas characterize a widespread network of projections originating in the PFC (Goldman-Rakic et al. 1984; Selemon and Goldman-Rakic 1985, 1988), as well as subcortical afferent terminal labeling in the PFC from the mediodorsal thalamus (Giguere and Goldman-Rakic 1988). Similarly, intrinsic circuitry of the PFC exhibits a distinct modular organization. Small injections of a retrogradely transported tracer into each of the six cortical layers label a vertically oriented column of neurons that extend from the site of injection to the pial surface above and to the white matter below (Kritzer and Goldman-Rakic 1995). Of particular interest, injections in layer IIIc produce a tangential pattern that extends as far as 7 mm laterally and consists of clusters of distant layer IIIc neurons, predominantly pyramidal neurons, interspersed with unlabeled territories (Fig. 3.2; Kritzer and Goldman-Rakic 1995). This patterning indicates that a single column in the PFC receives input from multiple distant columns that are interdigitated with columns that do not send input to that same column. In parallel with these findings, Lewis and colleagues established that pyramidal neurons give rise to a disjunctive intralaminar projection and that these distant pyramidal terminals synapse on the dendritic spines of other pyramidal neurons forming excitatory contacts (Levitt et al. 1993; Pucak et al. 1996; Melchitzky et al. 1998). Thus, recurrent excitatory input from one pyramidal cell to another in layer III is an important feature of interconnectivity between distant columns.
Schematic diagram of labeled neurons in the PFC following an injection into layer IIIc of the retrogradely transported tracer, cholera toxin (Β-subunit) conjugated to colloidal gold. Labeled neurons exhibit a disjunctive patterning that includes a vertical column surrounding the injection site and multiple clusters of neurons located at spaced intervals from the injection site (Figure generously provided by Dr. Mary F. Kritzer)
3.2 Prefrontal Cortical Activity During a Spatial Working Memory Task
Single unit recording had established that neurons in the prefrontal cortex are active during spatial working memory tasks and that a substantial proportion of active neurons fire during the delay period, i.e. when the cue is no longer present but before a response is required (Fuster and Alexander 1971; Kubota and Niki 1971; Fuster 1973; Niki 1974a, b; Fuster et al. 1982; Joseph and Barone 1987). Goldman-Rakic and colleagues re-examined neuronal activity in the dlPFC using an oculomotor version of the spatial DR task. These studies revealed several properties that had not been recognized, among them the precise spatial tuning of many neurons in the principal sulcal cortex of the dlPFC (Fig. 3.3; Funahashi et al. 1989, 1990). A large proportion of neurons exhibit enhanced firing in the cue, delay, or response phases of the task, with some active in more than one phase (Funahashi et al. 1989, 1990). Interestingly, the majority of cue (97 %) and delay (79 %) period neurons show directionality such that activity is either significantly enhanced or inhibited when the cue appears in one location of visual space while unaltered when stimuli appears in other quadrants of the visual axis (Funahashi et al. 1989, 1990). In some instances, units that are fire more frequently in response to a particular direction of space are inhibited by a stimulus appearing in the opposite direction, i.e. when the two stimuli are 180° apart (Funahashi et al. 1989). Moreover, most of the neurons are tuned to the visual space of the contralateral hemisphere such that lesions of the cortex in around the principal sulcus result in failure to perform oculomotor DR when cues are located in the contralateral hemifield (Funahashi et al. 1989, 1990, 1993). Importantly, spatially discriminative neurons in the dlPFC are selectively activated when memory of the spatial location is required, thus distinguishing them from neurons in visual cortex that are activated by stimuli in specific retinotopic locations but only when the stimulus is present (Kojima and Goldman-Rakic 1984). In sum, these findings established that sustained firing of neurons in the dlPFC during the delay period represents the electrophysiologic basis for retaining a mental trace of spatial information until the proper response is required and that neurons in the dlPFC have preferred retinotopic locations for the memory trace, i.e., “memory fields.”
Neuronal activity of a single unit during the oculomotor DR task in control conditions and after treatment with D1R acting drugs. (a) A schematic diagram (left) shows the sequence of events occurring during the task. Note that rasters and histograms directly below are aligned to these task epochs. During the task, the monkey fixates on the center position (0) of the screen (right) while one of the eight target positions is lit briefly (cue period). The target light is extinguished for a period of 3–6 s (delay period) during which time the monkey must remember the location of the previously lit target. The central fixation point is then extinguished as a signal for the monkey to make a saccade in the direction of the remembered target location (response period) to receive a juice reward. (b) The control condition shows that this unit is weakly activated during the delay period by a target in position 2 and not responsive to a target in position 7. Iontophoresis of the D1R antagonist SCH 39166 enhances the delay period activation of this unit to the preferred location (2) while suppressing activation to the non-preferred location (7). These effects are reversed by the partial D1 agonist SKF 38393. (From Goldman-Rakic 1999a. Reproduced with permission of New York Academy of Sciences in the format reprint in a book via Copyright Clearance Center)
3.3 Intrinsic Neurocircuitry of the Dorsolateral Prefrontal Cortex
Goldman-Rakic and colleagues learned more about how persistent firing of delay period neurons is related to cellular networks and the modular organization of prefrontal cortex by recording simultaneously from pairs of neurons in the dlPFC. One focus of their studies was elucidating the interaction between pyramidal cells, identified electrophysiologically by their regular spiking (RS) pattern, and populations of interneurons characterized by a fast spiking (FS) pattern of activity. FS neurons have been identified as the parvalbumin-positive basket and chandelier subtypes of interneurons (Kawaguchi 1995). They found that FS neurons are also spatially tuned to a restrictive area of visual space. Pairs of RS-RS neurons in close proximity (<400 μm) are not only spatially tuned to the same visual quadrant but also respond alike with increased or decreased firing rate to the preferred visual location (Wilson et al. 1994). In contrast, RS-FS pairs have inverted patterns of activity such that excitation of activity in a RS neuron is accompanied by inhibition in the FS pair, or vice versa (Wilson et al. 1994). The opposite patterns of activation and inhibition in FS-RS pairs suggested that interneurons inhibit pyramidal neurons with opponent tuning. Further study confirmed that closely aligned neurons are isodirectionally tuned and showed that shared responsiveness to a particular spatial location of neurons within a column is due to activation by the same afferent stimulation (Rao et al. 1999). Such neurons with shared spatial directionality form “microcolumns” within the larger column. Notably, in this 1999 study, the majority of FS-RS pairs, which were in even closer proximity to each other than in the earlier study, showed isodirectional tuning rather than opponent tuning. Together these studies indicate that interneurons may be involved in two inhibitory processes: iso-directional inhibition and cross-directional inhibition (Fig. 3.4; Rao et al. 1999). Iso-directional tuning functions locally to allow sharpening of tuning to a specific spatial direction via inhibition between neighboring microcolumns that are tuned to similar, though not identical, locations of visual space. Cross-directional inhibition suppresses activity between adjacent columns that are tuned to opposite quadrants of space.
Schematic illustration of iso-directional (top) and cross-directional (bottom) inhibition. (Top left) Interneurons (round somas) make short distance connections with pyramidal cells (triangular somas) in neighboring microcolumns that are tuned to nearly the same region of visual space. (Top right) 5-HT2A-like immunoreactivity in the PFC showing prominent bundles of apical dendrites presumably from pyramidal neurons in single microcolumns. (Bottom) Interneurons make long distance connections with pyramidal cells that reside further away and are tuned to the opposite visual quadrant (From Rao et al. 1999. Reproduced with permission of American Physiological Society [etc.] in the format reuse in a book/textbook via Copyright Clearance Center)
Additional features of columnar organization in the PFC were revealed by paired recordings of pyramidal neurons located in supragranular layers. For example, neurons that share directional tuning to a particular quadrant of visual space and are active during the same task epoch, i.e. cue, delay, or response, show the highest level of cross correlation activity, essentially a measure of time-linked co-activation (Constantinidis et al. 2001). Co-activation of neurons thus is a key component that defines local microcircuits of neurons that code a specific spatial location during a particular temporal epoch of oculomotor DR. These studies also revealed that neurons tuned to all angles of visual space are encountered in an expanse of 200–300 μm, suggesting that this width constitutes the limits of a single column comprised of microcolumns representing each individual direction in space. Perhaps surprisingly, cross correlation activity is even higher for pairs of interneurons identified by their fast spiking properties, indicating that tightly correlated activity is a particularly important property of local networks (Constantinidis and Goldman-Rakic 2001).
3.4 Multiple Roles for Interneurons in Local Networks
The aforementioned findings show that interneuron populations play a critical role in spatial mnemonic function. Indeed, application of a γ-aminobutyric acid subtype A (GABAA) receptor inhibitor alters spatial tuning of PFC neurons by eroding and broadening the tuning of some units and unmasking tuning in others (Rao et al. 2000). Interneurons are a heterogeneous population with respect to morphology (Lund and Lewis 1993) and electrophysiologic properties (Kawaguchi 1995); therefore, it stands to reason that distinct populations of interneurons play different roles in integrating information in the PFC. In slice preparations of the ferret PFC, Krimer and Goldman-Rakic (2001) found evidence for this diversity of function in interneuron populations. They used dual whole-cell voltage recording to examine the relationship between pyramidal cells and closely aligned interneurons of three categories (local arbor, medium arbor, and wide arbor) and intracellular filling to examine the synaptic contacts between recorded pairs of pyramidal neurons and interneurons. Previous studies had established that local and wide arbor neurons are parvalbumin-positive GABA neurons whereas medium arbor interneurons use cholecystokinin as a transmitter (Lund and Lewis 1993). Pyramidal neurons reliably excite local arbor interneurons, including chandelier neurons, via a single synaptic contact whereas progressively greater numbers of synaptic contacts link pyramidal neurons with medium and wide arbor interneurons, and activation at multiple synapses is required for depolarization of these interneurons subtypes (Krimer and Goldman-Rakic 2001). Notably, axon length is also longer in wider arbor cells, as local arbor interneurons have axons extending only 300 μm tangentially whereas medium and wide arbor interneurons extend 600 and 900 μm lateral to the cell soma (Krimer and Goldman-Rakic 2001). Because interneurons are thought to give rise to the interposed inhibitory synapse between pyramidal cells in the PFC, different classes of interneurons are in prime position to mediate intracolumnar and intercolumnar inhibition. Local arbor interneurons, receiving excitatory input from nearby pyramidal cells, may suppress activity in pyramidal cells belonging to adjacent microcolumns with spatial field tuning that is similar but not exactly the same as that of the original pyramidal cell, i.e. iso-directional inhibition. In contrast, medium and wide arbor neurons, receiving input from nearby pyramidal cells, could mediate suppression at more distant pyramidal cells that have intermediate and opponent spatial tuning. In particular, wide arbor interneurons with the longest axons are capable of integration between columns, i.e. cross-directional inhibition.
Similar analysis of the reverse connectivity between adjacent interneurons and pyramidal neurons also uncovered cell type specificity. FS neurons with basket cell morphology predominantly innervate the soma and proximal dendrites of pyramidal cells whereas non-FS interneurons exhibiting bitufted, double bouquet, or Martinotti cell morphologies contact the distal dendrites of pyramidal cells (Gao et al. 2003). The importance of this connectional distinction will be discussed in the context of dopaminergic modulation of neural circuitry in the next section.
3.5 Dopaminergic Modulation of Prefrontal Network Activity
Early studies by Goldman-Rakic and colleagues established that dopamine is a key player in modulating neuronal activity in the PFC. Dopamine is present in higher levels in the PFC than in more posterior cortices of the primate brain (Brown and Goldman 1977; Brown et al. 1979), and depletion of dopamine in the PFC produces a cognitive deficit nearly as severe as ablation of the same region (Brozoski et al. 1979). Deciphering how dopamine exerts this powerful control over PFC activity became a major focus of Goldman-Rakic’s studies and one that has profound implications for understanding cognitive dysfunction in diseases like schizophrenia and Parkinson’s disease (Goldman-Rakic et al. 2000, 2004).
Neuroanatomic studies from the Goldman-Rakic laboratory established the precise patterning of dopamine innervation of the prefrontal cortex, the distribution of receptor subtypes and synaptic contacts. Dopamine-immunoreactive fibers in frontal cortex innervate all cortical areas although differences in density of fibers across medio-lateral and rostro-caudal axes are apparent (Williams and Goldman-Rakic 1993). In most of the frontal cortex, including the dlPFC, dopamine fibers are densely packed in two broad bands encompassing (1) layer I, II, and upper III and (2) layer V-VI, with the middle layers less densely innervated (Williams and Goldman-Rakic 1993). The origin of the mesocortical dopamine projection in the nonhuman primate, arising from all three dopaminergic midbrain groups A8-A10, is more widespread than that of rodent species, underscoring parallels between the phylogenetic expansion of the dopaminergic system and the enlargement and differentiation of the primate prefrontal cortex (Williams and Goldman-Rakic 1998).
Ultrastructural analysis of dopaminergic terminals revealed that dopaminergic boutons form symmetric (inhibitory) contacts with the soma, shafts, and dendritic spines of pyramidal cells (Goldman-Rakic et al. 1989). Most notably, dopamine terminations on dendritic spines constitute one third of a triadic arrangement with the other components identified as the postsynaptic spine and an afferent asymmetric (excitatory) terminal contacting the same postsynaptic spine (Fig. 3.5; Goldman-Rakic et al. 1989). This triadic arrangement suggests that dopamine is poised to modulate excitatory input although the exact origin of this afferent excitatory stimulation is not known; it could emanate from distant cortical areas, the thalamus, or perhaps recurrent axons terminals of local pyramidal neurons. Further analysis revealed that dopaminergic appositions are concentrated on distal dendrites and, in concordance with earlier findings of dopamine fiber distributions, that pyramidal neurons in layer II/superficial III are most heavily laden with dopamine contacts (Krimer et al. 1997). However, dopamine innervation is not confined to pyramidal neurons as dopaminergic boutons also form symmetric contacts with interneurons in the PFC (Sesack et al. 1995; Krimer et al. 1997).
Drawings illustrating the triadic arrangement of dopamine afferents (DA) with dendritic spines and unidentified excitatory afferents (UA). (a) Pyramidal neurons receive dopaminergic afferents predominantly on distal dendritic spines. (b) Dopamine-containing afferents contact a dendritic spine that is also contacted by an afferent that lacks dopamine immunoreactivity. (c) At the ultrastructural level, the dopamine-positive terminal forms a symmetrical (inhibitory) synapse with the spine. In contrast, the dopamine-negative terminal is excitatory as evidenced by the presence of an asymmetric synapse on the spine. (From Goldman-Rakic 1995. Reproduced with permission of Cell Press in the format reuse in a book/textbook via Copyright Clearance Center)
The dopamine D1 receptor (D1R) is the preponderant receptor subtype in the PFC, with a density 10–20 times greater than that of the D2R (Lidow et al. 1991). D1R is distributed most densely in superficial layers (I, II and superficial III) and predominantly on spines of pyramidal cells, suggesting that this is the receptor subtype that mediates dopaminergic modulation of excitatory transmission (Goldman-Rakic et al. 1990; Lidow et al. 1991; Smiley et al. 1994; Bergson et al. 1995). In contrast, the D2R is largely concentrated in layer V (Goldman-Rakic et al. 1990). Notably, D1R is found in perisynaptic and extrasynaptic regions of the membrane and, despite the triadic arrangement described previously, D1R-positive spines rarely are opposed by axons having features of dopamine boutons (Smiley et al. 1994). Both of these characteristics suggest that volumetric neurotransmission of dopamine at sites distant to the synapse may represent an alternative mode of action for dopamine to impact neural circuitry in the PFC. Although D1R is most abundantly found on the dendritic spines and shafts of pyramidal neurons, this receptor subtype has also been localized to excitatory axon terminals (Smiley et al. 1994; Paspalas and Goldman-Rakic 2005). Moreover, D1R is found in parvalbumin-positive (FS type) interneurons, both in the dendritic and terminal compartments (Muly et al. 1998). Interestingly, D1R-positive axon terminals only contact pyramidal cell spines; they do not synapse with parvalbumin-positive interneurons (Paspalas and Goldman-Rakic 2005). Considered together, these findings suggest that D1R is present in multiple elements of the local neuronal network that governs spatial mnemonic behavior in the PFC, yet there is an elegant specificity to the targeting of dopamine that preferentially modulates excitatory input onto pyramidal cell spines.
One of Goldman-Rakic’s most influential findings emanated from studies of the action of dopaminergic agonists on neuronal activity in the PFC. Williams and Goldman-Rakic (1995) discovered that in vivo iontophoresis of a selective D1R antagonist onto pyramidal cell neurons selectively accentuates firing for targets located in the cells preferred memory field during the delay period. D1R antagonists do not have similar effects on neurons active during cue or response period, and the selective D2R antagonist raclopride suppresses activity of neurons non-selectively across all task epochs (Williams and Goldman-Rakic 1995). These findings suggest that delay period neurons are preferentially targeted by dopamine acting via the D1R. In addition, enhancement of delay period activity is dose dependent with higher doses of the D1R antagonist diminishing the delay activity (Fig. 3.3; Williams and Goldman-Rakic 1995). This dose sensitive relationship is best described as an inverted “u” such that too little or too much dopamine D1R stimulation has detrimental effects on delay period firing of neurons during the oculomotor DR. In concert with these findings, local injection of D1R antagonists into the PFC induces errors and increases latency in performance on the oculomotor DR in a dose dependent fashion (Sawaguchi and Goldman-Rakic 1991), and systemic injection of D1R acting compounds modulate working memory performance of monkeys on a manual DR task (Arnsten et al. 1994).
Exploration of the mechanisms by which dopamine may exert control over delay firing in PFC neurons revealed multiple roles for D1R in network activity. For example, in deep layer V, dopamine acts via presynaptic mechanisms to decrease the lateral excitation of recurrent excitatory contacts between local pyramidal cells thought to be important in maintaining delay period firing (Gao et al. 2001). In addition, findings by the Pittsburgh group suggest that in layer III dopamine increases excitability of pyramids through the D1R and acts via a combined D1R and D2R mechanism to suppress excitatory input from distant afferent sources, e.g. the thalamus or other cortical areas (Henze et al. 2000; Urban et al. 2002). These properties further support a role for dopamine in modulating excitatory input onto pyramidal dendritic spines in the PFC. Although decreased excitatory neurotransmission at the pyramidal spine seems at odds with activation and maintenance of sustained firing during the delay period, it must be remembered that not all spines and not all excitatory terminals are D1R-containing and therefore specificity in the modulation of excitatory input onto pyramidal cells may curtail input from irrelevant stimuli and consequently accentuate the signal to noise ratio for relevant stimuli during a memory related task.
Dopamine also influences network activity by acting on interneurons. In contrast to its modulatory role at the pyramidal to pyramidal synapse, dopamine does not decrease excitatory potency at excitatory contacts between pyramidal cells and interneurons (Gao and Goldman-Rakic 2003). Instead, dopamine directly excites the majority of interneurons (Zhou and Hablitz 1999; Gorelova et al. 2002; Gao and Goldman-Rakic 2003). In this regard, dopamine’s selectivity in presynaptic site of action corresponds with the anatomic specificity of D1R distribution, i.e. located in excitatory terminals in apposition to pyramidal cells spines but not those contacting interneurons (Paspalas and Goldman-Rakic 2005). The exquisite specificity of dopamine’s action also extends to modulation of interneuron inhibition of pyramidal cells. Dopamine acts via the D1R to facilitate inhibition at FS interneuron (basket cell) synapses that are known to innervate the perisomatic region of pyramidal cells while diminishing inhibitory neurotransmission of non-FS interneuron (double bouquet, tufted and Martinotti cells) that contact the distal dendrites of pyramids (Gao et al. 2003). The multitude of dopamine actions in the PFC, including dampening excitatory transmission between pyramidal cells and both facilitating and curtailing interneuron inhibition of pyramidal cells, attests to the complexity of dopaminergic modulation of the mnemonic-related network activity in the PFC. Certainly, a full understanding of the modulatory role of dopamine on PFC network circuitry remains beyond our grasp. However, what is clear is that optimal dopamine stimulation enhances relevant input for a PFC neuron’s memory field and optimizes delay period firing for the target location.
3.6 Spatial and Object Working Memory Domains
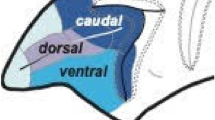
Goldman-Rakic’s work centered on the dlPFC, specifically the cortex surrounding the principal sulcus; however, studies of the connections and the physiologic properties of more ventrally located cortex in the PFC led to a remarkable discovery: the ventral PFC is specialized for the memory of “what” in contrast to the more dorsal cortex that remembers “where.” Goldman-Rakic and colleagues found that cortical neurons located on the ventral convexity of the PFC are responsive to stimuli hitting the foveal region of the retina and that they are tuned to the color and form of objects (Wilson et al. 1993). Some neurons in the ventral PFC exhibit even greater specificity by responding only to faces (Wilson et al. 1993; O’Scalaidhe et al. 1997, 1999). Importantly, during the spatial oculomotor DR, a task in which the location of the target must be remembered, neurons in the ventral PFC are not active during the delay period; however, when an object must be remembered as a signal for the correct saccadic response, these same neurons in the ventral PFC exhibit sustained firing to bridge the gap between cue and response periods (Fig. 3.6, Wilson et al. 1993).
Schematic representation of separate visual streams that feed into spatial and object domains in the dorsal and ventral PFC. (Top) Rasters and histograms showing enhanced activity of a delay period neuron in the dlPFC to the preferred target location. (Bottom) Rasters and histograms illustrating the response of a neuron to a monkey face in the ventral PFC. (From Goldman-Rakic 1999b. Reproduced with permission of Elsevier Inc. in the format reuse in a book/textbook via Copyright Clearance Center)
The segregation of working memory function into dorsal and ventral domains that respectively mediate memory for spatial location and object features fits perfectly with the known connectivity of dorsal and ventral PFC. A wealth of data have established that discrete features of visual information coming from the retina are processed separately in the visual system (Livingston and Hubel 1988). Moreover, visual information leaving the primary visual cortex to reach areas of higher cortical processing are also segregated. Visual attributes related to form and color are channeled from the visual cortex into a “ventral stream” that relays information through the inferotemporal cortex to the ventral PFC while spatial location is conveyed through the “dorsal stream” via the posterior parietal cortex to the dlPFC (Macko et al. 1982; Barbas 1988; Cavada and Goldman-Rakic 1989; Morel and Bullier 1990; Baizer et al. 1991; Webster et al. 1994). The partitioning of cortex into spatial and object domains is also supported by lesion studies in the non-human primate that show a dissociation of spatial and non-spatial deficits by dorsal and ventral site (e.g., Mishkin and Manning 1978; Passingham 1975; Levy and Goldman-Rakic 2000). It should be noted that this division of labor for the PFC has not been universally accepted with some arguing that different regions of the PFC mediate higher versus lower levels of cognitive processing (Owen et al. 1996; Petrides et al. 2002). To some degree discrepancies in location of data collection may account for conflicting evidence on this topic (for a review, see Romanski 2004).
Parcellation of the PFC with respect to the location or identity of the memorandum is not limited to the visual modality. Auditory afferent input conveying location and feature identity are also segregated in the PFC: the rostral principal sulcal cortex and surrounding cortices, including the inferior convexity, receive information about auditory features while the caudal principal sulcal and dorsal periarcuate cortices are innervated by auditory projections that convey spatial location (Romanski et al. 1999a, b). The electrophysiologic specificity in responding to stimuli again mirrors the anatomic duality. Neurons in the dlPFC have auditory responsive properties and have been shown to be sensitive to sound source location (Azuma and Suzuki 1984; Kikuchi-Yorioka and Sawaguchi 2000) whereas neurons that respond to complex features of sound, e.g., species-specific vocalizations, are located in the ventral PFC adjacent to the visual object domain (Romanski and Goldman-Rakic 2002; Romanski et al. 2005). Some evidence also suggests that somatosensory information may contribute to memory for objects in the ventral PFC (Romo et al. 1999). These data further support Goldman-Rakic’s (1996, 1999b) concept of separate domains for memory of “what” and “where” that may be even further modularized into subdomains that process information from diverse sensory streams.
3.7 Summary
Perhaps the most extraordinary aspect of Goldman-Rakic’s work is the breadth and depth of her studies on the PFC. She examined the entire hierarchy of PFC organization extending from the “macromodular” level of organization represented by these large, functionally distinct domains down to the microcolumnar structure devoted to representation of a single aspect of a memorandum, e.g. spatial location in the dlPFC. Her theories of prefrontal function represent the synthesis of work from many different methodologic approaches, encompassing the fields of neuroanatomy, neurophysiology, neuropharmacology, and behavior. Patricia Goldman-Rakic’s diverse studies laid the foundation for contemporary understanding of prefrontal structure and function. Although she is clearly missed, the many young scientists whom she mentored continue her legacy as they pursue a multitude of avenues of research relating to the PFC.
References
Arnsten AF, Cai JX, Murphy BL, Goldman-Rakic PS (1994) Dopamine D1 receptor mechanisms in the cognitive performance of young adult and aged monkeys. Psychopharmacology 116:143–151
Azuma M, Suzuki H (1984) Properties and distribution of auditory neurons in the dorsolateral prefrontal cortex of the alert monkey. Brain Res 298:343–346
Baddeley AD, Hitch GJ (1974) Working memory. In: Bower GA (ed) Recent advances in learning and motivation, vol 8. Academic, New York, pp 47–90
Baizer JS, Ungerleider LG, Desimone R (1991) Organization of visual inputs to the inferior temporal and posterior parietal cortex in macaques. J Neurosci 11:168–190
Barbas H (1988) Anatomic organization of basoventral and mediodorsal visual recipient prefrontal regions in the rhesus monkey. J Comp Neurol 276:313–342
Bergson C, Mrzljak L, Smiley JF, Pappy M, Levenson R, Goldman-Rakic PS (1995) Regional, cellular, and subcellular variations in the distribution of D1 and D5 dopamine receptors in primate brain. J Neurosci 15:7821–7836
Brown RM, Goldman PS (1977) Catecholamines in neocortex of rhesus monkeys: regional distribution and ontogenetic development. Brain Res 124:576–580
Brown RM, Crane AM, Goldman PS (1979) Regional distribution of monoamines in the cerebral cortex and subcortical structures of the rhesus monkey: concentrations and in vivo synthesis rates. Brain Res 168:133–150
Brozoski TJ, Brown RM, Rosvold HE, Goldman PS (1979) Cognitive deficit caused by regional depletion of dopamine in prefrontal cortex of rhesus monkey. Science 205:929–932
Cavada C, Goldman-Rakic PS (1989) Posterior parietal cortex in rhesus monkey: II. Evidence for segregated corticocortical networks linking sensory and limbic areas with the frontal lobe. J Comp Neurol 287:422–445
Constantinidis C, Goldman-Rakic PS (2001) Correlated discharges among putative pyramidal neurons and interneurons in the primate prefrontal cortex. J Neurophysiol 88:3487–3497
Constantinidis C, Franowicz MN, Goldman-Rakic PS (2001) Coding specificity in cortical microcircuits: a multiple-electrode analysis of primate prefrontal cortex. J Neurosci 21:3646–3655
Funahashi S, Bruce CJ, Goldman-Rakic PS (1989) Mnemonic coding of visual space in the monkey’s dorsolateral prefrontal cortex. J Neurophysiol 61:331–349
Funahashi S, Bruce CJ, Goldman-Rakic PS (1990) Visuospatial coding in primate prefrontal neurons revealed by oculomotor paradigms. J Neurophysiol 63:814–831
Funahashi S, Bruce CJ, Goldman-Rakic PS (1993) Dorsolateral prefrontal lesions and oculomotor delayed response performance: evidence for mnemonic “scotomas”. J Neurosci 13:1479–1497
Fuster JM (1973) Unit activity in prefrontal cortex during delayed-response performance: neuronal correlates of transient memory. J Neurophysiol 36:61–78
Fuster JM, Alexander GE (1971) Neuron activity related to short-term memory. Science 173:652–654
Fuster JM, Bauer RH, Jervey JP (1982) Cellular discharge in the dorsolateral prefrontal cortex of the monkey in cognitive tasks. Exp Neurol 77:679–694
Gao W-J, Goldman-Rakic PS (2003) Selective modulation of excitatory and inhibitory microcircuits by dopamine. Proc Natl Acad Sci 100:2836–2841
Gao W-J, Krimer LS, Goldman-Rakic PS (2001) Presynaptic regulation of recurrent excitation by D1 receptors in prefrontal circuits. Proc Natl Acad Sci U S A 98:295–300
Gao W-J, Wang Y, Goldman-Rakic PS (2003) Dopamine modulation of perisomatic and peridendritic inhibition in prefrontal cortex. J Neurosci 23:1622–1630
Giguere M, Goldman-Rakic PS (1988) Mediodorsal nucleus: areal, laminar, and tangential distribution of afferents and efferents in the frontal lobe of rhesus monkeys. J Comp Neurol 277:195–213
Goldman PS (1971) Functional development of the prefrontal cortex in early life and the problem of neuronal plasticity. Exp Neurol 32:366–387
Goldman PS (1976) The role of experience in recovery of function following orbital prefrontal lesions in infant monkeys. Neuropsychologia 14:401–412
Goldman PS, Nauta WJH (1977) Columnar distribution of cortico-cortical fibers in the frontal association, limbic, and motor cortex of the developing rhesus monkey. Brain Res 122:393–413
Goldman PS, Rosvold HE (1970) Localization of function within the dorsolateral prefrontal cortex of the rhesus monkey. Exp Neurol 27:291–304
Goldman PS, Rosvold HE, Mishkin M (1970a) Evidence for behavioral impairment following prefrontal lobectomy in the infant monkey. J Comp Physiol Psychol 70:454–463
Goldman PS, Rosvold HE, Mishkin M (1970b) Selective sparing of function following prefrontal lobectomy in infant monkeys. Selective sparing of function following prefrontal lobectomy in infant monkeys. Exp Neurol 29:221–226
Goldman PS, Rosvold HE, Vest B, Galkin TW (1971) Analysis of the delayed-alternation deficit produced by dorsolateral prefrontal lesions in the rhesus monkey. J Comp Physiol Psychol 77:212–220
Goldman PS, Crawford HT, Stokes LP, Galkin TW, Rosvold HE (1974) Sex-dependent behavioral effects of cerebral cortical lesions in the developing rhesus monkey. Science 186:540–542
Goldman-Rakic PS (1987) Circuitry of the prefrontal cortex and the regulation of behavior by representational knowledge. In: Plum F, Mountcastle V (eds) Handbook of physiology, vol 5. American Physiological Society, Bethesda, pp 373–417
Goldman-Rakic PS (1995) Cellular basis of working memory. Neuron 14:477–485
Goldman-Rakic PS (1996) Regional and cellular fractionation of working memory. Proc Natl Acad Sci U S A 93:13473–13480
Goldman-Rakic PS (1999a) The “psychic” neuron of the cerebral cortex. Ann N Y Acad Sci 868:13–26
Goldman-Rakic PS (1999b) The physiological approach: functional architecture of working memory and disordered cognition in schizophrenia. Biol Psychiatry 46:650–661
Goldman-Rakic PS, Selemon LD, Schwartz ML (1984) Dual pathways connecting the dorsolateral prefrontal cortex with the hippocampal formation and parahippocampal cortex in the rhesus monkey. Neuroscience 12:719–743
Goldman-Rakic PS, Leranth C, Williams SM, Mons N, Geffard M (1989) Dopamine synaptic complex with pyramidal neurons in primate cerebral cortex. Proc Natl Acad Sci U S A 86:9015–9019
Goldman-Rakic PS, Lidow MS, Gallager DW (1990) Overlap of dopaminergic, adrenergic, and serotoninergic receptors and complementarity of their subtypes in primate prefrontal cortex. J Neurosci 10:2125–2138
Goldman-Rakic PS, Muly EC III, Williams GV (2000) D1 receptors in prefrontal cells and circuits. Brain Res Rev 31:295–301
Goldman-Rakic PS, Castner SA, Svensson TH, Siever LJ, Williams GV (2004) Targeting the dopamine D1 receptor in schizophrenia: insights for cognitive dysfunction. Psychopharmacology 174:3–16
Gorelova N, Seamans JK, Yang CR (2002) Mechanisms of dopamine activation of fast-spiking interneurons that exert inhibition in rat prefrontal cortex. J Neurophysiol 88:3150–3166
Henze DA, Gonzalez-Burgos GR, Urban NN, Lewis DA, Barrionuevo G (2000) Dopamine increases excitability of pyramidal neuron in primate prefrontal cortex. J Neurophysiol 84:2799–2809
Hubel DH, Wiesel TN (1972) Laminar and columnar distribution of geniculo-cortical fibers in the macaque monkey. J Comp Neurol 146:421–450
Jones EG, Burton H, Porter R (1975) Commissural and cortico-cortical “columns” in the somatic sensory cortex of primates. Science 190:572–574
Joseph JP, Barone P (1987) Prefrontal unit activity during a delayed oculomotor task in the monkey. Exp Brain Res 67:460–468
Kawaguchi Y (1995) Physiologic subgroups of nonpyramidal cells with specific morphological characteristics in layer II/III of the rat frontal cortex. J Neurosci 15:2638–2655
Kikuchi-Yorioka Y, Sawaguchi T (2000) Parallel visuospatial and audiospatial working memory processes in the monkey dorsolateral prefrontal cortex. Nat Neurosci 3:1075–1076
Kojima S, Goldman-Rakic PS (1984) Functional analysis of spatially discriminative neurons in prefrontal cortex of rhesus monkey. Brain Res 291:229–240
Krimer LS, Goldman-Rakic PS (2001) Prefrontal microcircuits: membrane properties and excitatory input of local, medium, and wide arbor interneurons. J Neurosci 21:3788–3796
Krimer LS, Jakab RL, Goldman-Rakic PS (1997) Quantitative three-dimensional analysis of the catecholaminergic innervation of identified neurons in the macaque prefrontal cortex. J Neurosci 17:7450–7461
Kritzer MF, Goldman-Rakic PS (1995) Intrinsic circuit organization of the major layers and sublayers of the dorsolateral prefrontal cortex in the rhesus monkey. J Comp Neurol 359:131–143
Kubota K, Niki H (1971) Prefrontal cortical unit activity and delayed alternation performance in monkeys. J Neurophysiol 34:337–347
Levitt JB, Lewis DA, Yoshioka T, Lund JS (1993) Topography of pyramidal neuron intrinsic connections in macaque monkey prefrontal cortex (areas 9 and 46). J Comp Neurol 338:360–376
Levy R, Goldman-Rakic PS (2000) Segregation of working memory functions within the dorsolateral prefrontal cortex. Exp Brain Res 133:23–32
Lidow MS, Goldman-Rakic PS, Gallager DW, Rakic P (1991) Distribution of dopaminergic receptors in the primate cerebral cortex: quantitative autoradiographic analysis using [3H]raclopride, [3H]spiperone and [3H]SCH23390. Neuroscience 40:657–671
Livingston M, Hubel D (1988) Segregation of form, color, movement, and depth: anatomy, physiology, and perception. Science 240:740–749
Lund JS, Lewis DA (1993) Local circuit neurons of developing and mature macaque prefrontal cortex. Golgi and immunocytochemical characteristics. J Comp Neurol 328:282–312
Macko KA, Jarvis CD, Kennedy C, Miyaoka M, Shinohara M, Sokoloff L, Mishkin M (1982) Mapping the primate visual system with 2-14C-deoxyglucose. Science 218:394–397
Melchitzky DS, Sesack SR, Pucak ML, Lewis DA (1998) Synaptic targets of pyramidal neurons providing intrinsic horizontal connections in monkey prefrontal cortex. J Comp Neurol 390:211–224
Mishkin M, Manning FJ (1978) Non-spatial memory after selective prefrontal lesions in monkeys. Brain Res 143:313–323
Morel A, Bullier J (1990) Anatomical segregation of two cortical visual pathways in the macaque monkey. Vis Neurosci 4:555–578
Muly EC III, Szigeti K, Goldman-Rakic PS (1998) D1 receptor in interneurons of macaque prefrontal cortex: distribution and subcellular localization. J Neurosci 18:10553–10565
Niki H (1974a) Differential activity of prefrontal units during right and left delayed response trials. Brain Res 70:346–349
Niki H (1974b) Prefrontal unit activity during delayed alternation in the monkey. I. Relation to direction of response. Brain Res 68:185–196
O’Scalaidhe SP, Wilson FAW, Goldman-Rakic PS (1997) Areal segregation of face-processing neurons in prefrontal cortex. Science 278:1135–1138
O’Scalaidhe SP, Wilson FAW, Goldman-Rakic PS (1999) Face-selective neurons during passive viewing and working memory performance of rhesus monkeys: evidence for intrinsic specialization of neuronal coding. Cereb Cortex 9:459–475
Owen AM, Evans AC, Petrides M (1996) Evidence for a two-stage model of spatial working memory processing within the lateral frontal cortex: a positron emission tomography study. Cereb Cortex 6:31–38
Paspalas CD, Goldman-Rakic PS (2005) Presynaptic D1 dopamine receptors in primate prefrontal cortex: target-specific expression in the glutamatergic synapse. J Neurosci 25:1260–1267
Passingham R (1975) Delayed matching after selective prefrontal lesions in monkeys (Macaca mulatta). Brain Res 92:89–102
Petrides M, Alivisatos B, Frey S (2002) Differential activation of the human orbital, mid-ventrolateral, and mid-dorsolateral prefrontal cortex during the processing of visual stimuli. Proc Natl Acad Sci U S A 99:5649–5654
Pucak ML, Levitt JB, Lund JS, Lewis DA (1996) Patterns of intrinsic and associational circuitry in monkey prefrontal cortex. J Comp Neurol 376:614–630
Rakic P (1988) Specification of cerebral cortical areas. Science 241:170–176
Rao SG, Williams GV, Goldman-Rakic PS (1999) Isodirectional tuning of adjacent interneurons and pyramidal cells during working memory: evidence for microcolumnar organization in PFC. J Neurophysiol 81:1903–1916
Rao SG, Williams GV, Goldman-Rakic PS (2000) Destruction and creation of spatial tuning by disinhibition: GABAA blockade of prefrontal cortical neurons engaged by working memory. J Neurosci 20:485–494
Romanski LM (2004) Domain specificity in the primate prefrontal cortex. Cogn Affect Behav Neurosci 4:421–429
Romanski LM, Goldman-Rakic PS (2002) An auditory domain in the primate prefrontal cortex. Nat Neurosci 5:15–16
Romanski LM, Bates JF, Goldman-Rakic PS (1999a) Auditory belt and parabelt projections to the prefrontal cortex in the rhesus monkey. J Comp Neurol 403:141–157
Romanski LM, Tian B, Fritz J, Mishkin M, Goldman-Rakic PS, Rauschecker JP (1999b) Dual streams of auditory afferents target multiple domains in the primate prefrontal cortex. Nat Neurosci 2:1131–1136
Romanski LM, Averbeck BB, Diltz M (2005) Neural representation of vocalizations in the primate ventrolateral prefrontal cortex. J Neurophysiol 93:734–747
Romo R, Brody CD, Hernandez A, Lemus L (1999) Neuronal correlates of parametric working memory in the prefrontal cortex. Nature 399:470–473
Sawaguchi T, Goldman-Rakic PS (1991) D1 dopamine receptors in prefrontal cortex: involvement in working memory. Science 251:947–950
Schwartz ML, Goldman-Rakic PS (1984) Callosal and intrahemispheric connectivity of the prefrontal association cortex in rhesus monkey: relation between intraparietal and principal sulcal cortex. J Comp Neurol 226:403–420
Selemon LD, Goldman-Rakic PS (1985) Longitudinal topography and interdigitation of corticostriatal projections in the rhesus monkey. J Neurosci 5:776–794
Selemon LD, Goldman-Rakic PS (1988) Common cortical and subcortical targets of the dorsolateral prefrontal and posterior parietal cortices in the rhesus monkey: evidence for a distributed neural network subserving spatially guided behavior. J Neurosci 8:4049–4068
Sesack SR, Snyder CL, Lewis DA (1995) Axon terminals immunolabeled for dopamine or tyrosine hydroxylase synapse on GABA-immunoreactive dendrites in rat and monkey cortex. J Comp Neurol 363:264–280
Shanks MF, Rockel AJ, Powell TPS (1975) The commissural fiber connections of the primary somatic sensory cortex. Brain Res 98:166–171
Smiley JF, Levey AI, Ciliax BJ, Goldman-Rakic PS (1994) D1 dopamine receptor immunoreactivity in human and monkey cerebral cortex: predominant and extrasynaptic localization in dendritic spines. Proc Natl Acad Sci U S A 91:5720–5724
Urban NN, Gonzalez-Burgos G, Henze DA, Lewis DA, Barrionuevo G (2002) Selective reduction by dopamine of excitatory synaptic inputs to pyramidal neurons in primate prefrontal cortex. J Physiol 593:707–712
Webster MJ, Bachevalier J, Ungerleider LG (1994) Connections of inferior temporal areas TEO and TE with parietal and frontal cortex in macaque monkeys. Cereb Cortex 4:470–483
Williams GV, Goldman-Rakic PS (1995) Modulation of memory fields by dopamine D1 receptors in prefrontal cortex. Nature 376:572–575
Williams SM, Goldman-Rakic PS (1993) Characterization of the dopaminergic innervation of the primate frontal cortex using a dopamine-specific antibody. Cereb Cortex 3:199–222
Williams SM, Goldman-Rakic PS (1998) Widespread origin of the primate mesofrontal dopamine system. Cereb Cortex 8:321–345
Wilson FAW, O’Scalaidhe SP, Goldman-Rakic PS (1993) Dissociation of object and spatial processing domains in primate prefrontal cortex. Science 260:1955–1958
Wilson FAW, O’Scalaidhe SP, Goldman-Rakic PS (1994) Functional synergism between putative γ-aminobutyrate-containing neurons and pyramidal neurons in prefrontal cortex. Proc Natl Acad Sci U S A 91:4009–4013
Zhou F-M, Hablitz JJ (1999) Dopamine modulation of membrane and synaptic properties of interneurons in rat cerebral cortex. J Neurophysiol 81:967–976
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer Science+Business Media Dordrecht
About this chapter
Cite this chapter
Selemon, L.D. (2015). Modular Organization of the Prefrontal Cortex: The Legacy of Patricia Goldman-Rakic. In: Casanova, M., Opris, I. (eds) Recent Advances on the Modular Organization of the Cortex. Springer, Dordrecht. https://doi.org/10.1007/978-94-017-9900-3_3
Download citation
DOI: https://doi.org/10.1007/978-94-017-9900-3_3
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-017-9899-0
Online ISBN: 978-94-017-9900-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)