Abstract
Upon attacked by insect herbivores, plants can activate a range of defenses that result in direct and/or indirect resistance to subsequent challenge by a herbivore. Significant development in understanding of the physiological and molecular basis of these herbivore-induced plant defense responses has been achieved over the past decade. Our understanding of these defense responses has led to new ideas of herbivore management methods that can be environmental friendly and safer. We studied the responses of rice, one of the most important food crops of the world, to infestation by insect herbivores, including rice planthoppers. In this review, we first briefly summarize the fundamentals and molecular basis of herbivore-induced rice defense responses. We then introduce the methods of planthopper management in rice that could be exploited. These methods include using herbivore-induced plant volatiles (HIPVs) as attractants of the natural enemies, the application of chemical elicitors, and the genetic modification of crop variety. Finally, some insights are given about the directions of future research and how to tap this opportunity of herbivore-induced plant defense response in rice pest management.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Rice planthoppers
- Herbivore-induced plant defense response
- Herbivore-induced plant volatiles
- Chemical elicitors
- Tritrophic interactions
5.1 Introduction
In nature, plants suffer from various biotic stresses, such as herbivores and pathogens. In order to protect themselves, plants have evolved a series of defense mechanisms (Wu and Baldwin 2010; Arimura et al. 2011; Bonaventure et al. 2011; Hilker and Meiners 2011; Erb et al. 2012). These strategies include constitutive defenses , which exist in plants before pest attack, and induced defenses , which are activated following attack.
Plant-induced defenses, which can be elicited by feeding or oviposition of herbivores, are known to be triggered by elicitors derived from herbivores and/or the interaction between plants and herbivores (Mithöfer and Boland 2008; Heil 2009; Wu and Baldwin 2010; Arimura et al. 2011; Bonaventure et al. 2011; Hilker and Meiners 2011; Erb et al. 2012). Induced plant defenses may positively or negatively influence the performance of subsequent herbivores directly or indirectly by influencing the effectiveness of the natural enemies of the herbivores (Bostock 2005; Howe and Jander 2008; Baldwin 2010; Heil and Karban 2010; War et al. 2011; Mithofer and Boland 2012; Clavijo McCormick et al. 2012). For example, herbivore infestation induces some plants to release volatiles and/or produce extra-floral nectar , both of which can influence the behavior and performance of the predators and parasitoids of herbivores (Baldwin 2010; Erb et al. 2012; Peñaflor and Bento 2013). Moreover, these plant-mediated interactions may occur not only among above-ground herbivores but also between above-ground herbivores and below-ground herbivores (Erb et al. 2012). Thus, herbivore-induced plant defense responses play an important role in modulating the composition of the arthropod community and structure in an ecosystem (Kessler et al. 2007; Zheng and Dicke 2008). Some of these plant defense traits, such as volatiles attracting natural enemies of herbivores and elicitors inducing plant defense, can enhance the efficiency of natural enemies and/or reducing the performance of herbivores; these could be exploited for new measures for managing herbivores (War et al. 2011; Peñaflor and Bento 2013).
Rice is one of the most important food crops of the world. It suffers from many insect pests. In China, the main rice insect pests include rice planthoppers—brown planthopper (BPH) Nilaparvata lugens (Stål), white-backed planthopper (WBPH) Sogatella furcifera (Horváth), and small brown planthopper (SBPH) Laodelphax striatellus (Fallen); rice borers—striped stem borer (SSB) Chilo suppressalis (Walker) and Scircophaga incertulas (Walker); and rice leaf folder (LF) Cnaphalocrocis medinalis Guenee. In some regions, the water weevil Lissorhoptrus oryzophilus Kuschel, the gall midge Orseoia oryzae (Wood-Mason), and the thrip Chloethrips oryzae (Wil.) also heavily infest rice. To prevent rice plants from herbivore infestation , some control methods have been tried. The major control methods include resistant varieties, cultural controls, biological controls , and chemical controls (Lou et al. 2013). However, since the number of rice varieties resistant to herbivores is small, and cultural and biological controls are labor consuming and less efficient, the major control measure for rice insect pests is the use of insecticides. These not only cause severe environmental pollution but also lead eventually to the resurgence of herbivores and reduce populations of the natural enemies. Therefore, developing safe and effective methods for managing rice insect pests is essential.
Rice plants have also been reported to produce defense responses following attack by insect pests, including rice planthoppers (Lou et al. 2005a, b, 2006; Zhou et al. 2009, 2011; Lu et al. 2011; Qi et al. 2011). These defense responses may influence the performance of the subsequent conspecific and non-conspecific herbivores directly and/or indirectly by regulating the third trophic level (Lou et al. 2005a, b, 2006; Zhao et al. 2001, 2009, 2011; Lu et al. 2011; Qi et al. 2011). Moreover, a few studies have revealed that induced rice defense responses are able to modulate the arthropod community composition and the population densities of insect pests in the field (Xiao et al. 2012). Therefore, appropriately inducing rice defense traits may decrease the population densities of rice insect pests, in turn reducing the amount of chemical insecticides.
In this review, we first summarize the fundamentals and molecular basis of herbivore-induced rice defense responses. Then, we introduce methods of managing rice insect pests, especially rice planthoppers, that could be exploited based on these defense responses. Finally, future research directions are proposed.
5.2 Herbivore-Induced Rice Defenses
Like many other plant species, such as Arabidopsis, tomato, and tobacco, rice produces defense responses when attacked by insect herbivores; these responses can influence the behavior and performance of subsequent herbivores directly and/or indirectly by attracting the natural enemies of herbivores (Chen et al. 2002; Lou et al. 2005a, b, 2006). Striped stem borer larvae fed on SSB larvae-infested plants grow slowly compared to those fed on non-manipulated plants (Zhou et al. 2009). In response to BPH infestation , rice plants release volatiles that attracts egg parasitoid Anugrus nilaparvatae (Lou et al. 2005a) and enhance the susceptibility of rice to WBPH (Zhao et al. 2001). Prior feeding by Spodoptera frugiperda or jasmonic acid (JA) treatment increases resistance in rice to the water weevil, Lissorhoptrus oryzophilus, a root-damaging herbivore (Hamm et al. 2010). Moreover, plants treated with JA significantly reduce number of immature L. oryzophilus relative to untreated plants. Like the other plant species, herbivore-induced rice defense responses also change with rice variety (Lou and Cheng 2003, 2006) and plant growth stage (Ma et al. 2004; Wang et al. 2011), herbivore species (Zhou et al. 2009; Lu et al. 2011), density, and infestation time (Ma et al. 2004; Xiang et al. 2008), and with abiotic factors, such as nitrogen levels (Lou and Cheng 2003). The information suggests that herbivore-induced rice defenses have strong plasticity and play an important role in shaping the composition of the arthropod community and structure in rice ecosystem.
The process by which herbivore attack induces a plant’s defensive response is complicated. First, the plant has to recognize herbivore-associated molecular patterns (HAMPs); subsequently, these activate early events and multiple signaling pathways , such as a change in the plasma transmembrane potential (Vm), calcium flux, calcium-dependent protein kinase (CDPK), and mitogen-activated protein kinase (MAPK) cascades, and JA -, salicylic acid (SA)-, ethylene -, and H2O2-mediated pathways (Wu and Baldwin 2010; Arimura et al. 2011; Erb et al. 2012). The activated signals and pathways increase the levels of defense-related gene transcripts and defense-related compounds, and this increase causes plants to become resistant to subsequently attacking herbivores (Howe and Jander 2008; Wu and Baldwin 2010; Arimura et al. 2011; Bonaventure et al. 2011; Erb et al. 2012).
Thus far, several HAMPs, such as fatty acid–amino acid conjugates (FACs) (Alborn et al. 1997; Halitschke et al. 2001; Yoshinaga et al. 2007), β-glucosidase (Mattiacci et al. 1995), inceptin (Schmelz et al. 2007), caeliferins (Alborn et al. 2007), bruchins (Doss et al. 2000), and benzyl cyanide (Fatouros et al. 2008), have been identified. Like herbivore infestation , some of these HAMPs have been found to induce Vm change and calcium flux (Maffei et al. 2004; Arimura et al. 2011), as well as the activation of MAPKs and JA , and ethylene biosynthesis and signaling (Halitschke et al. 2001; Giri et al. 2006; Wu et al. 2007; Skibbe et al. 2008). Moreover, calcium flux, protein kinase (such as CDPK and MAPK) cascades, and JA-, SA -, ethylene-, and H2O2-mediated signaling pathways have been confirmed to play a central role in shaping the specificity of herbivore-induced plant defense responses (Wu et al. 2007; Howe and Jander 2008; Wu and Baldwin 2010; Heinrich et al. 2011; Meldau et al. 2012; Mithofer and Boland 2012). However, nothing was known about plant receptors that perceive HAMPs . So far, only two possible receptors, lectin receptor kinase 1 (Gilardoni et al. 2011) and BAK1 (Yang et al. 2011), have been reported.
Studies on the mechanism underlying herbivore-induced rice defense also revealed that the process by which rice responds defensively is complex and involves changes in the transcript levels of many genes that belong to 18 functional groups and the reconfiguration of a wide variety of metabolic, physiological, and biochemical processes (Zhang et al. 2004; Hua et al. 2007; Zhou et al. 2011). An integrated signaling network consisting of phytohormones, especially JA , SA , and ethylene , and secondary signal transduction components, such as Ca2+ signaling, reactive oxygen species, G protein signaling, and protein kinases, underlies the entire process (Lu et al. 2006; Wang et al. 2008; Zhou et al. 2009, 2011; Lu et al. 2011; Qi et al. 2011). This signaling network alters the transcript levels of many genes by activating many transcription factors; the activation of these factors has many biological consequences, including the accumulation of defense chemicals and decreases in photosynthetic activity (Zhou et al. 2011).
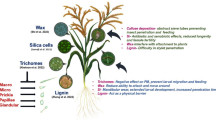
Recently, several early components that regulate signaling pathways in rice have been identified. OsERF3, for example, has been found to function upstream of OsMPK3, JA , SA , ethylene , and H2O2 pathways, and to positively regulate the biosynthesis of JA, SA, and ethylene but negatively modulate H2O2 production (Lu et al. 2011). OsPLDα4 and α5 were reported to influence the production of JA and green leaf volatiles (GLVs) , the products of the hydroperoxide lyase (HPL) branch of the oxylipin pathway (Qi et al. 2011). Bph14 , a coiled-coil, nucleotide-binding, and leucine-rich repeat (CC-NB-LRR) protein predominantly expressed in vascular bundles, was found to confer resistance to BPH by activating an SA signaling pathway , inducing callose deposition in phloem cells and enhancing the activity of trypsin protease inhibitors (TrypPIs) after planthopper infestation (Du et al. 2009). OsMPK3 positively mediates the production of elicited JA , which subsequently affects levels of herbivore-induced TrypPIs and decreases the performance of SSB larvae (Wang et al. 2013). These findings suggest that the herbivore-elicited rice defense responses are regulated by a set of signaling networks (Fig. 5.1).
Current information on mechanisms responsible for herbivore-induced defense responses in rice. Rice plants recognize herbivore-associated molecular patterns (HAMPs) derived from herbivores or interaction with the plant and then activate components, such as calcium influx, OsERF3 and OsPLDs. These activated early components will elicit MAPK cascades, jasmonic acid (JA) -, salicylic acid (SA)-, ethylene (ET)-, and H2O2-mediated signaling pathways , which in turn enhance levels of defense-related gene transcripts and defense compounds. This increases resistance to herbivores. Some herbivores may secrete effectors (E) which suppress the plant’s defenses. However, for resistant varieties, plants possess resistance genes (R), such as BPH14 , that will recognize the herbivore’s effectors and activate the second layer of defense responses; this is known as effector-triggered immunity. Among these compounds, herbivore-induced plant volatiles (HIPVs) , including green leaf volatiles (GLVs), which are mainly regulated by JA and ET pathways, can influence the behavior and performance of both chewing herbivores and piercing and sucking herbivores, such as striped stem borers (SSBs) and brown planthoppers (BPHs), directly and indirectly by the natural enemies of the herbivores. Trypsin proteinase inhibitors (TrypPIs) are also positively modulated by both JA and ET signaling pathways and negatively affect the performance of chewing herbivores, such as SSBs. Both callose deposition that may be mediated by H2O2 and SA pathways, and H2O2 itself, have a negative effect on BPH performance. JA- and ET-mediated pathways negatively modulate resistance in rice to BPHs
Plants’ defense responses to different herbivores are modulated by the different signaling pathways that they elicit (Howe and Jander 2008; Wu and Baldwin 2010; Meldau et al. 2012; Mithofer and Boland 2012). Various signaling pathways in rice were found to play different roles in modulating resistance to herbivores with different feeding habits. The resistance of rice to lepidopteran caterpillars, such as the larvae of SSB and LF, is mainly positively mediated by JA and ethylene signaling pathways , whereas resistance to piercing and sucking herbivores, such as BPHs, is modulated negatively by JA but positively by H2O2 and SA pathways (Zhou et al. 2009; Lu et al. 2011) (Fig. 5.1).
Research with numerous plant species has revealed that a great variety of constitutive and inducible chemicals , such as terpenoids, phenolics, alkaloids, glucosinolates, and cyanogenic glycosides, and defensive proteins, such as proteinase inhibitors (PIs), cysteine proteases, lectins, chitinases, and polyphenol oxidases (PPOs), have toxic or antifeedant effects on insect herbivores (Ryan 1990; Peumans et al. 1995; Wang and Constable 2004; Aharoni et al. 2005; Mohan et al. 2006; Howe and Jander 2008; Gill et al. 2010). Moreover, herbivore-induced plant volatiles (HIPVs) and GLVs have been reported to influence the behavior and/or performance of the subsequent herbivores directly and/or indirectly by attracting the predators and parasitoids (Kessler and Baldwin 2001; Degenhardt et al. 2003; Dicke and Baldwin 2010; Snoeren et al. 2010). In rice, the levels of some defense-related compounds, including PIs, phytoalexins, pathogenesis-related proteins, and terpenoids, were observed to be enhanced when plants were infested by herbivores or treated with defense-related signals, such as JA (Rakwal and Komatsu 2000; Zhou et al. 2009). In addition, the TrypPI activity, which was positively regulated by JA and ethylene signaling pathways (Zhou et al. 2009; Lu et al. 2011), negatively affected the performance of lepidopteran caterpillars, such as the larvae of SSB and LF (Zhou et al. 2009; Lu et al. 2011). Volatiles emitted from rice plants infested by herbivores or treated with JA can attract parasitoids and enhance the parasitism of herbivores (Lou et al. 2005a, b, 2006). Some volatiles , such as linalool and (E)-β-caryophyllene (Xiao et al. 2012), as well as GLVs (Qi et al. 2011; Tong et al. 2012), were found to not only influence the efficiency of the natural enemies but also to affect the performance of herbivores, including BPHs, SSBs, and LFs (Fig. 5.1). Callose deposition in phloem cells, which could be induced by BPH infestation , was regarded as one of the resistance mechanisms of rice plants to BPH (Hao et al. 2008). Moreover, benzyl benzoate, a chemical that is elicited by WBPH egg deposition has been shown to increase ovicidal activity against WBPH (Seino et al. 1996). In general, defense compounds used against insect herbivores in rice were less well understood (Horgan 2009).
5.3 Potential of Herbivore-Induced Defense in Rice Planthopper Management
Many studies have demonstrated that herbivore-induced plant direct and indirect defenses can influence the population dynamics of herbivores in the field (Karban and Baldwin 1997; Baldwin 1998; Halitschke et al. 2008; Degenhardt et al. 2009; Allmann and Baldwin 2010; Xiao et al. 2012). Moreover, these defenses can enhance plant fitness (Baldwin 1998; Steppuhn et al. 2004; Schuman et al. 2012). For example, Baldwin (1998) found that MeJA treatment increases resistance in tobacco (Nicotiana attenuate) to herbivores and enhances the plant’s fitness in nature. Field studies revealed that HIPVs emitted from tobacco and maize can reduce herbivore populations from 24 % to more than 90 %, via enhancing the predation and parasitism of herbivores (Kessler and Baldwin 2001; Rasmann et al. 2005; Halitschke et al. 2008; Degenhardt et al. 2009; Allmann and Baldwin 2010) and deterring herbivores from egg deposition (Kessler and Baldwin 2001). Thus, appropriately using and/or regulating these defense traits might reduce densities of herbivores and hence decrease the amount of pesticides applied.
5.3.1 Influence of Synthetic HIPVs in Attracting Natural Enemies
Based on the biosynthesis pathways or their known within-plant functions HIPVs mainly include three chemical groups (Holopainen and Gershenzon 2010). The first group, the terpenoids, is generally the dominant group of HIPVs in many plant species and is produced by two separate pathways, one active in plastids (MEP) and the other one (MVA) in cytosol (Loreto and Schnitzler 2010; Maffei 2010). The second group includes fatty acid derivatives, including the C6 lipoxygenase products, so-called GLVs . The third group is the volatile aromatic compounds, such as methyl salicylate and indole. In addition, there are a multitude of other volatile compounds, such as the volatile plant hormone ethylene and volatile amino acid derivatives (Dicke and Baldwin 2010; Holopainen and Blande 2013; Peñaflor and Bento 2013).
HIPVs are known to have multiple functions: Some signal within a plant to activate systemic defenses, priming and activating defenses in neighboring plants; some influence the behavior or performance of herbivores and pollinators; and some attract natural enemies of herbivores (Dicke and Baldwin 2010; Holopainen and Blande 2013; Peñaflor and Bento 2013). These functions may be helpful for controlling insect pests, as more than 20 single compounds or mixtures, including GLVs , terpenoids, and aromatics, have been found to attract the natural enemies of herbivores in the field (James 2003a, 2005a, b; Yu et al. 2008; Lee 2010; Orre et al. 2010); methyl salicylate and GLVs are strong candidate chemicals for this purpose (Table 5.1). Methyl salicylate (MeSA), for example, has been reported to attract many species of the natural enemies of herbivores, such as Stethorus punctum picipes, Orius tristicolor, Erigonidium graminicolum, Orius similis, Chrysopa nigricornis, Deraeocoris brevis, and Anagrus spp. (Table 5.1). In addition, MeSA was found to actually enhance the efficiency of the natural enemies and thus to dramatically reduce the population densities of the main pests, spider mites, and aphids (James and Price 2004). However, HIPVs may also attract herbivores and the fourth trophic level, the natural enemies of parasitoids and predators, or repel the natural enemies of the herbivores (Dicke and Baldwin 2010; Holopainen and Blande 2013; Peñaflor and Bento 2013). For example, the parasitoid Diadegma semiclausum is repelled by MeSA (Snoeren et al. 2010; Braasch et al. 2012). Thus, the synthetic HIPVs that attract natural enemies but not herbivores and/or hyperparasitoids should be screened (Kaplan 2012).
In rice, previous studies have shown that herbivore infestation or jasmonic acid (JA) treatment alters the volatile profiles of rice plants (Lou et al. 2005a, b, 2006; Yan et al. 2010; Zhou et al. 2011). These volatiles mainly include fatty acid derivatives and terpenoids (Lou et al. 2006; Zhou et al. 2011; Xiao et al. 2012). N. lugens-induced or JA-elicited rice volatiles are strongly attractive to the egg parasitoid A. nilaparvatae, a major natural enemy of the rice planthopper (Lou et al. 2005a, b). Moreover, the parasitism of N. lugens eggs by A. nilaparvatae on plants that were surrounded by JA -treated plants is more than twofold higher than on control plants in the greenhouse and in the field (Lou et al. 2005a, 2006), implying that augmenting the release of rice-produced attractants has the potential to enhance the effectiveness of the parasitoid in the control of N. lugens. Recently, Wang and Lou (2013) found that five compounds—Z-3-hexenyl acetate, 1-penten-3-ol, Z-3-hexenal, linalool, and MeSA—attract the parasitoid. Moreover, A. nilapareatae were strongly attracted by the mixtures of MeSA plus Z-3-hexenal, one containing Z-3-hexenal, Z-3-hexenyl acetate and linalool, and one containing MeSA, Z-3-hexenal, Z-3-hexenyl acetate, and linalool. Field experiments demonstrated that the parasitism of BPH eggs was significantly increased on plants that received a septa containing one of the three chemicals (Z-3-hexenal, Z-3-hexenyl acetate, and linalool) or the mixture containing MeSA, Z-3-hexenal, Z-3-hexenyl acetate, and linalool. The findings may contribute to improving or may help improve the biological control of N. lugens in the future.
5.3.2 Effects of Exogenous Application of Chemical Elicitors
Chemical elicitors are defined as chemicals that trigger resistance or enhance a plant’s ability to mobilize induced defense responses (priming agents) to pests and are themselves not directly toxic to the pest (Kessmann et al. 1994; Pare et al. 2005; Beckers and Conrath 2007; Kim and Felton 2013). Thus far, many such natural and synthetic elicitors, especially elicitors that induce plant defense responses to pathogens, have been identified and/or synthesized. These elicitors include microbe-associated molecular patterns (MAMPs), such as bacterial flagellin, lipopolysaccharides (LPSs), and peptidoglycan (PGN) as well as fungi chitin and β-glucan, HAMPs , phytohormones and their analogs, such as JA and its analogs, SA and its analogs, and other chemicals, such as terpenoids and BABA (Pare et al. 2005; Silipo et al. 2010; Hilker and Meiners 2011; Newman et al. 2013). These elicitors can activate defense-related signaling pathways (Schmelz et al. 2009; Silipo et al. 2010; Newman et al. 2013) or increase the sensitivity in triggering defense responses (Pare et al. 2005; Kim and Felton 2013), which subsequently makes plants able to resist pathogens or insect herbivores. Up to now, several of these chemical elicitors, such as benzo-(1,2,3) thiadiazole-7-carbothioic acid S-methyl ester (BTH), 2,6-dichloroisonicotinic acid (INA), and β-aminobutyric acid (BABA), have been commercialized for plant disease control (Vallad and Goodman 2004; Beckers and Conrath 2007; Knoth et al. 2009).
There are more than 20 chemical elicitors , mainly including HAMPs, phytohormones and their analogs, that induce or prime plant defense against insect herbivores (Table 5.2). For example, HAMPs and JA have been reported to activate defense-related signaling pathways and thus increase direct and indirect resistance in plants to herbivores (Table 5.2). Terpenoids, GLVs , and two inorganic chemicals, copper and silicon, can prime plants and enhance their ability to defend themselves against subsequent invaders (Table 5.2). In general, JA and elicitors that induce the JA signaling pathway mainly trigger resistance to necrotrophic pathogens and chewing herbivores, whereas SA and SA-inducing elicitors mainly elicit resistance in plants to biotrophic pathogens and piercing/sucking insects (Thaler et al. 2002; Arimura et al. 2005). However, Turlings and his research group recently found that BTH and laminarin, both of which elicit the SA signaling pathway , reduced the emission of herbivore-induced volatiles but increased direct and indirect resistance (attraction of the parasitoids) to the herbivores (Rostas and Turlings 2008; Sobhy et al. 2012), suggesting the complexity of induced plant defense responses.
Like elicitors that induce plant defense to pathogens, some of elicitors that elicit defenses to insect herbivores have also been proven to enhance the resistance of plants to herbivores in the field. For example, Baldwin (1998) found that MeJA treatment increases resistance in N. attenuata to herbivores and enhances its fitness in nature. The exogenous application of JA can elicit direct and indirect defense responses of plants to herbivores in the field (Thaler 1999; Thaler et al. 2002). Wheat plants in the field sprayed with low levels of cis-jasmone as an aqueous emulsion have been found to have lower aphid infestations (Moraes et al. 2008). Recently, Bingham et al. (2013) found that the application of micro-encapsulated cis-jasmone combined with piperonyl butoxide, a synergistic agent of pesticide, on tomato plants in the field resulted in a nearly 90 % reduction in the number of the tobacco whitefly, Bemisia tabaci. Moreover, plant seeds treated with elicitors have been found to enhance plant defense responses to herbivores. For example, tomato plants whose seeds were treated with JA showed enhanced responsiveness of defense-related genes and increased resistance to the tobacco hornworm Manduca sexta, green peach aphids Myzus persicae, and spider mites Tetranychus urticae (Worrall et al. 2012). Sobhy et al. (2014) reported that treating maize seeds with either BTH or laminarin increased the attractiveness of herbivore-infested plants to all three tested parasitoid species, Microplitis rufiventris Kok., Cotesia marginiventris (Cresson), and Campoletis sonorensis (Cameron). These works suggest that the appropriate application of chemical elicitors could effectively reduce the population densities of herbivores.
In rice, it has been reported that N. lugens-induced or JA-elicited rice volatiles are strongly attractive to the parasitoid A. nilaparvatae, and the parasitism of N. lugens eggs by A. nilaparvatae on plants that were surrounded by JA -treated plants is more than twofold higher than on control plants in the greenhouse and field (Lou et al. 2005a, 2006). Recently, by developing a high-throughput chemical genetics screening system, Xin et al. (2012) found that 2,4-dicholorophenoxyacetic acid (2,4-D) induced a strong defensive reaction and a significant increase in volatile production. Induced plants were more resistant to SSB but became highly attractive to BPH , as well as its parasitoid, A. nilaparvatae. In a subsequent field experiment, 2,4-D application were able to draw away BPH from non-treated plants and turn the treated plants into deadly traps by also attracting large numbers of parasitoids. Such a chemical elicitor, combined with push-pull management strategy (Cook et al. 2007), may improve our ability to manage BPH. When rice plants at the edge of a field, for example, are sprayed with 2,4-D, a trapping zone is created that reduces pest damage in the inner parts of the field. This may also lead to higher numbers of egg parasitoids in these zones, keeping pest reproduction to a minimum and creating a source of parasitoids that can attack the next generation of the pest in the untreated parts of the fields (Xin et al. 2012).
5.3.3 Genetic Modification of Rice Variety
As our understanding of the molecular mechanisms responsible for herbivore-induced plant defense responses increases, the genetic engineering of plants may offer a means to enhance resistance in plants to herbivores (Degenhardt et al. 2003; War et al. 2011; Peñaflor and Bento 2013). By overexpressing or knocking down one or more genes, including those responsible for defense-related signaling pathways , volatile emission, and defense compound synthesis, genetically modified plants have been made in many plant species, such as Arabidopsis, maize, rice, tomato, and tobacco, and some have been proven to enhance the direct and indirect resistance of plants to herbivorous insects in the field (Degenhardt et al. 2009; Schuman et al. 2012; Xiao et al. 2012). For example, when the ability of American corn varieties to emit (E)-β-caryophyllene (an important signal used by entomopathogenic nematodes to find hosts) was restored (expressing a (E)-β-caryophyllene synthase gene in plants), transgenic corn plants were damaged much less by western corn rootworm than were corn plants lacking the enzyme (Degenhardt et al. 2009). The larvae of Manduca sexta grew faster on and preferred plants of N. attenuata whose nicotine levels had been reduced by 95 % via genetic engineering (Steppuhn et al. 2004). When planted in their native habitat, plants whose nicotine production had been impaired were attacked more frequently and, compared to wild-type plants, lost threefold more leaf area from a variety of native herbivores, suggesting that nicotine functions as an efficient defense in nature and that plants with high levels of nicotine will resist herbivore damage. Recently, a 2-year field study by Schuman et al. (2012) has shown that plants that emit GLVs produce twice as many buds and flowers—a measure of fitness—as plants that have been genetically engineered not to emit GLVs; GLVs only increased plants’ fitness when various species of Geocoris (a bug that preys on Manduca sexta) reduced the number of herbivores.
Xiao et al. (2012) found that rice lines impaired in emissions of S-linalool, a compound that is strongly induced by BPH infestation, were less attractive to the egg parasitoid of rice planthoppers, A. nilaparvatae, as well as to predatory spiders but more attractive to BPH than were wild-type plants. On the other hand, lines with low levels of (E)-β-caryophyllene, a constitutively emitted volatile that is not inducible by BPH, attracted both BPH and its natural enemies less often than did WT plants. Both of the lines resulted in an increase in the BPH population in the field (Xiao et al. 2012). Thus, such transgenic lines may be used in combination with control BPH: Rice plants that produce (E)-β-caryophyllene but not S-linalool could be sown at the edges of the field to attract both BPH and its natural enemies, and other plants could be engineered to produce high amounts of S-linalool but not (E)-β-caryophyllene in order to attract egg parasitoids and avoid BPH colonization (Xiao et al. 2012).
Although there are some successful cases of reducing population densities of herbivores by using transgenic plants, developing a transgenic variety that constitutively expresses some defense-related genes should be avoided. The continuous production of defense chemicals may decrease plant fitness as the production of defense compounds is expensive (Steppuhn et al. 2004; Zavala et al. 2004), and herbivores adapt quickly to plants because selection pressure is high. Moreover, if the defense chemicals that constitutively produced are volatiles , such a variety will lose its attractiveness to natural enemies because the attracted natural enemies have no rewards (neither hosts nor prey) (Turlings and Ton 2006; Khan et al. 2008), and may attract pathogens and herbivores (Carroll et al. 2006; Halitschke et al. 2008). Therefore, a better approach to enhancing resistance in plants would be to develop lines whose defenses could be stronger and more rapidly induced by herbivores (Degenhardt et al. 2003; Turlings and Ton 2006).
5.4 Conclusions and Perspectives
In this review, we summarize recent advances in the understanding of herbivore-induced plant defense response and their application in pest management. With the accumulation of knowledge about genomics, proteomics, and metabolomes, much progress has been made over the past 10 years in understanding the molecular basis of herbivore-induced defense responses in rice. Moreover, based on this knowledge, some methods for insect pest control, mainly including HIPV-based attractants for the natural enemies of herbivores, the application of chemical elicitors and the genetic manipulation of crop variety , are being exploited. These methods have been shown to increase not only the foraging efficiency of the natural enemies of herbivores but also the resistance in plants to herbivores, and their use often decreases the population densities of and the damage by herbivores; in the end, fewer pesticides are applied. More importantly, in crops, especially in annual cropping systems , natural enemies are always one step behind the pest, which generally reduces the foraging efficiency of the natural enemies. Using methods such as HIPV-based attractants or elicitors, natural enemies may be attracted to crops earlier. In rice, such methods also show great potential, especially in the management of planthoppers (Lou et al. 2005a, b, 2006; Xiao et al. 2012; Xin et al. 2012; Wang and Lou 2013). Because these methods are environmentally friendly and can redistribute herbivores and their natural enemies in space and time, they are promising for pest management.
Like all measures for pest management, such as pesticides, however, the herbivore-induced plant defense-based methods also have weaknesses. First, HIPVs have been proven to mediate multiple functions in ecosystems. In addition to repelling herbivores (De Moraes et al. 2001; Bruinsma et al. 2007; Szendrei and Rodriguez-Saona 2010) and attracting their natural enemies, HIPVs can also provide signals with which parasitic plants (Runyon et al. 2006) or herbivores (Bolter et al. 1997; Kalberer et al. 2001; Carroll et al. 2006; Halitschke et al. 2008) can locate their hosts. Therefore, the inappropriate application of synthetic HIPVs may enhance the probability of infestation by other herbivores. Second, some parasitoid species use HIPVs to locate their hosts, relying on associative learning (Meiners et al. 2003). The diffuse application of synthetic HIPVs might attract the natural enemies but no “rewards” (hosts or prey), which will thus decrease the foraging efficiency of the parasitoids (Turlings and Ton 2006; Khan et al. 2008). Third, a chemical that is an elicitor for some pests may become a suppressor for others. For example, treatment with 2,4-D induces resistance in rice to SSB but induces susceptibility to the brown planthopper , Nilarpavata lugens (Xin et al. 2012). The application of JA makes plants resistant to herbivores, but at the same time, the SA -mediated pathway might be inhibited and plants could become more susceptible to pathogens (Stout et al. 1998; Thaler et al. 1999). Moreover, some elicitors, especially those analogous to plant hormones, may influence the growth and reproduction of plants. In addition, plants that constitutively enhance levels of defense-related compounds may decrease the efficiency in controlling herbivores as stated above. Therefore, it is important to investigate the main species of herbivores and their natural enemies in different locations of each crop system, as well as the interactions mediated by the induced defenses in the context of agroecosystem. Moreover, appropriate defense-related compounds that repress herbivore populations directly or indirectly and adapt to specific herbivores and areas of each crop system, various elicitors that induce different resistance mechanisms of plants to herbivores and have no harmful effect on plants, and crop varieties in which defense responses could be rapidly and strongly elicited by herbivore infestation should be exploited. Based on these results, effective methods of herbivore management that adapt to various areas and crop systems may be developed. Furthermore, various elicitors that induce different resistance mechanisms in plants could be used in combination so that herbivores cannot adapt to resistance traits induced by elicitors.
These herbivore management methods could also be combined with other management strategies, such as push-pull (Cook et al. 2007) and an ecological engineering program (Gurr et al. 2011), which may greatly reduce the damage by herbivores. For example, if rice plants that produce (E)-β-caryophyllene but not S-linalool are sown at the edges of the field, both BPH and its natural enemies could be strongly attracted. If the rest of the plants are engineered to produce high amounts of S-linalool but not (E)-β-caryophyllene, they will likely remain pest free (Xiao et al. 2012). Combined with an ecological engineering program, such methods, for example, HIPV-based attractants for natural enemies and the application of chemical elicitors , will further enhance the effect of natural enemies by helping them to locate their hosts/prey, an effect that neither of the two measures can achieve alone (Gurr et al. 2011).
In rice, defense compounds, especially non-volatile defense compounds against insect herbivores, including rice planthoppers, are largely unknown. Thus, to effectively exploit herbivore-induced defense responses, we should first identify the main defense chemicals in rice. Once the genes that regulate these chemicals as molecular markers are found, appropriate chemical elicitors and rice varieties could be exploited. Recently, an ecological engineering program that focuses on vegetation diversity and aims to enhance the population density and efficiency of natural enemies of insect pests has been established, and the results have shown that this program can effectively augment the effect of natural enemies, decreasing the frequency of outbreaks of insect pests, including planthoppers, and the need for pesticides in rice (Gurr et al. 2011). Therefore, the use of management methods developed from herbivore-induced rice defense responses and an ecological engineering program will further enhance population densities and the effectiveness of employing the natural enemies of insect pests (Gurr et al. 2011). By decreasing the population levels of rice insect pests, we can reduce the amount of insecticides and environmental pollution.
References
Aharoni A, Jongsma MA, Bouwmeester HJ. Volatile science? metabolic engineering of terpenoids in plants. Trends Plant Sci. 2005;10:594–602.
Alborn HT, Turlings T, Jones TH, Stenhagen G, Loughrin JH, Tumlinson JH. An elicitor of plant volatiles from beet armyworm oral secretion. Science. 1997;276:945–9.
Alborn HT, Hansen TV, Jones TH, Bennett DC. Novel disulfoxy fatty acids from the American bird grasshopper Shistocerca americana elicitors of plant volatiles. Proc Natl Acad Sci USA. 2007;104:12976–81.
Allmann S, Baldwin IT. Insects betray themselves in nature to predators by rapid isomerization of green leaf volatiles. Science. 2010;329:1075–8.
Arimura G, Kost C, Boland W. Herbivore-induced, indirect plant defences. BBA-Mol Cell Biol Lipids. 2005;1734:91–111.
Arimura G, Shiojiri K, Karban R. Acquired immunity to herbivory and allelopathy caused by airborne plant emissions. Phytochemistry. 2010;71:1642–9.
Arimura G, Ozawa R, Maffei ME. Recent advances in plant early signaling in response to herbivory. Int J Mol Sci. 2011;12:3723–39.
Arimura G, Muroi A, Nishihara M. Plant-plant-plant communications, mediated by (E)-β-ocimene emitted from transgenic tobacco plants, prime indirect defense responses of lima beans. J Plant Interact. 2012;7:193–6.
Baldwin IT. Jasmonate-induced responses are costly but benefit plants under attack in native populations. Proc Natl Acad Sci USA. 1998;95:8113–8.
Baldwin IT. Plant volatiles. Curr Biol. 2010;20:392–7.
Beckers GJ, Conrath U. Priming for stress resistance: from the lab to the field. Curr Opin Plant Biol. 2007;10:425–31.
Bingham G, Alptekin S, Delogu G, Gurkan O, Moores G. Synergistic manipulations of plant and insect defences. Pest Manag Sci. 2013;70:566–71.
Bolter CJ, Dicke M, Van Loon JJ, Visser JH, Posthumus MA. Attraction of Colorado potato beetle to herbivore-damaged plants during herbivory and after its termination. J Chem Ecol. 1997;23:1003–23.
Bonaventure G, Vandoorn A, Baldwin IT. Herbivore-associated elicitors: FAC signaling and metabolism. Trends Plant Sci. 2011;16:294–9.
Bostock RM. Signal crosstalk and induced resistance: straddling the line between cost and benefit. Annu Rev Phytopathol. 2005;43:545–80.
Braasch J, Wimp GM, Kaplan I. Testing for phytochemical synergism: arthropod community responses to induced plant volatile blends across crops. J Chem Ecol. 2012;38:1264–75.
Bruce TJ, Matthes MC, Chamberlain K, Woodcock CM, Mohib A, Webster B, Smart LE, Birkett MA, Pickett JA, Napier JA. cis-Jasmone induces Arabidopsis genes that affect the chemical ecology of multitrophic interactions with aphids and their parasitoids. Proc Natl Acad Sci USA. 2008;105:4553–8.
Bruinsma M, Van Dam NM, Van Loon JJ, Dicke M. Jasmonic acid-induced changes in Brassica oleracea affect oviposition preference of two specialist herbivores. J Chem Ecol. 2007;33:655–68.
Carroll MJ, Schmelz EA, Meagher RL, Teal PEA. Attraction of Spodoptera frugiperda larvae to volatiles from herbivore-damaged maize seedlings. J Chem Ecol. 2006;32:1911–24.
Chen HC, Lou YG, Cheng JA. Selection responses of Cotesia chilons, a larval parasitoid of the rice striped-stem-borer Chilo suppressalis, to volatile compounds from its host and host-plants. Acta Entomol Sinica. 2002;45:617–22.
Clavijo Mccormick A, Unsicker SB, Gershenzon J. The specificity of herbivore-induced plant volatiles in attracting herbivore enemies. Trends Plant Sci. 2012;17:303–10.
Cook SM, Khan ZR, Pickett JA. The use of push-pull strategies in integrated pest management. Annu Rev Entomol. 2007;52:375–400.
Cooper LD, Doss RP, Price R, Peterson K, Oliver JE. Application of Bruchin B to pea pods results in the up-regulation of CYP93C18, a putative isoflavone synthase gene, and an increase in the level of pisatin, an isoflavone phytoalexin. J Exp Bot. 2005;56:1229–37.
De Moraes CM, Mescher MC, Tumlinson JH. Caterpillar-induced nocturnal plant volatiles repel conspecific females. Nature. 2001;410:577–80.
Degenhardt J, Gershenzon J, Baldwin IT, Kessler A. Attracting friends to feast on foes: engineering terpene emission to make crop plants more attractive to herbivore enemies. Curr Opin Biotechnol. 2003;14:169–76.
Degenhardt J, Hiltpold I, Kollner TG, Frey M, Gierl A, Gershenzon J, Hibbard BE, Ellersieck MR, Turlings TCJ. Restoring a maize root signal that attracts insect-killing nematodes to control a major pest. Proc Natl Acad Sci USA. 2009;106:13213–8.
Dicke M, Baldwin IT. The evolutionary context for herbivore-induced plant volatiles: beyond the ‘cry for help’. Trends Plant Sci. 2010;15:167–75.
Doss RP, Oliver JE, Proebsting WM, Potter SW, Kuy SR, Clement SL, Williamson RT, Carney JR, DeVilbiss ED. Bruchins: insect-derived plant regulators that stimulate neoplasm formation. Proc Natl Acad Sci USA. 2000;97:6218–23.
Du B, Zhang WL, Liu BF, Hu J, Wei Z, Shi ZY, He RF, Zhu LL, Chen RZ, Han B, He GC. Identification and characterization of Bph14, a gene conferring resistance to brown planthopper in rice. Proc Natl Acad Sci USA. 2009;106:22163–8.
Engelberth J, Alborn HT, Schmelz EA, Tumlinson JH. Airborne signals prime plants against insect herbivore attack. Proc Natl Acad Sci USA. 2004;101:1781–5.
Erb M, Meldau S, Howe GA. Role of phytohormones in insect-specific plant reactions. Trends Plant Sci. 2012;17:250–9.
Fatouros NE, Huigens ME, van Loon JJ, Dicke M, Hilker M. Chemical communication: butterfly anti-aphrodisiac lures parasitic wasps. Nature. 2005;433:704.
Fatouros NE, Broekgaarden C, Bukovinszkine’Kiss G, Loon JJA, Mumm R, Huigens ME, Dicke M, Hilker M. Male-derived butterfly anti-aphrodisiac mediates induced indirect plant defense. Proc Natl Acad Sci USA. 2008;105:10033–8.
Ferry A, Dugravot S, Delattre T, Christides JP, Auger J, Bagneres AG, Poinsot D, Cortesero AM. Identification of a widespread monomolecular odor differentially attractive to several Delia radicum ground-dwelling predators in the field. J Chem Ecol. 2007;33:2064–77.
Frost CJ, Mescher MC, Carlson JE, De Moraes CM. Plant defense priming against herbivores: getting ready for a different battle. Plant Physiol. 2008;146:818–24.
Gilardoni PA, Hettenhausen C, Baldwin IT, Bonaventure G. Nicotiana attenuata LECTIN RECEPTOR KINASE1 suppresses the insect-mediated inhibition of induced defense responses during Manduca sexta herbivory. Plant Cell. 2011;23:3512–32.
Gill RS, Gupta AK, Taggar GK, Taggar MS. Review article: Role of oxidative enzymes in plant defenses against insect herbivory. Acta Phytopathol Entomol Hung. 2010;45:277–90.
Giri AP, Wunsche H, Mitra S, Zavala JA, Muck A, Svatoš A, Baldwin IT. Molecular interactions between the specialist herbivore Manduca sexta (Lepidoptera, Sphingidae) and its natural host Nicotiana attenuata. VII. Changes in the plant’s proteome. Plant Physiol. 2006;142:1621–41.
Godard K, White R, Bohlmann J. Monoterpene-induced molecular responses in Arabidopsis thaliana. Phytochemistry. 2008;69:1838–49.
Gurr GM, Liu J, Read D, Catindig JL, Chen JA, Lan LP, Heong KL. Parasitoids of Asian rice planthopper (Hemiptera: Delphacidae) pests and prospects for enhancing biological control by ecological engineering. Ann Appl Biol. 2011;158:149–76.
Halitschke R, Schittko U, Pohnert G, Boland W, Baldwin IT. Molecular interactions between the specialist herbivore Manduca sexta (Lepidoptera, Sphingidae) and its natural host Nicotiana attenuata. III. Fatty acid-amino acid conjugates in herbivore oral secretions are necessary and sufficient for herbivore-specific plant responses. Plant Physiol. 2001;125:711–7.
Halitschke R, Stenberg JA, Kessler D, Kessler A, Baldwin IT. Shared signals-‘alarm calls’ from plants increase apparency to herbivores and their enemies in nature. Ecol Lett. 2008;11:24–34.
Hamm JC, Stout MJ, Riggio RM. Herbivore- and elicitor-induced resistance in rice to the rice water weevil (Lissorhoptrus oryzophilus Kuschel) in the laboratory and field. J Chem Ecol. 2010;36:192–9.
Hao PY, Liu CX, Wang YY, Chen RZ, Tang M, Du B, Zhu LL, He GC. Herbivore-induced callose deposition on the sieve plates of rice: An important mechanism for host resistance. Plant Physiol. 2008;146:1810–20.
Heil M. Damaged-self recognition in plant herbivore defence. Trends Plant Sci. 2009;14:356–63.
Heil M, Karban R. Explaining evolution of plant communication by airborne signals. Trends Ecol Evol. 2010;25:137–44.
Heinrich M, Baldwin IT, Wu JQ. Two mitogen-activated protein kinase kinases, MKK1 and MEK2, are involved in wounding- and specialist lepidopteran herbivore Manduca sexta-induced responses in Nicotiana attenuata. J Exp Bot. 2011;62:4355–65.
Hilker M, Meiners T. Plants and insect eggs: how do they affect each other? Phytochemistry. 2011;72:1612–23.
Hilker M, Stein C, Schröder R, Varama M, Mumm R. Insect egg deposition induces defence responses in Pinus sylvestris: characterisation of the elicitor. J Exp Biol. 2005;208:1849–54.
Hirao T, Okazawa A, Harada K, Kobayashi A, Muranaka T, Hirata K. Green leaf volatiles enhance methyl jasmonate response in Arabidopsis. J Biosci Bioeng. 2012;114:540–5.
Holopainen JK, Blande JD. Where do herbivore-induced plant volatiles go? Front Plant Sci. 2013;4:185.
Holopainen JK, Gershenzon J. Multiple stress factors and the emission of plant VOCs. Trends Plant Sci. 2010;15:176–84.
Hopke J, Donath J, Blechert S, Boland W. Herbivore-induced volatiles: The emission of acyclic homoterpenes from leaves of Phaseolus lunatus and Zea mays can be triggered by a β-glucosidase and jasmonic acid. FEBS Lett. 1994;352:146–50.
Horgan D. Suicide prevention. Australas Psychiat. 2009;17:509.
Howe GA, Jander G. Plant immunity to insect herbivores. Annu Rev Plant Biol. 2008;59:41–66.
Hua HX, Lu Q, Cai M, Xu CG, Zhou DX, Li XH, Zhang QF. Analysis of rice genes induced by striped stemborer (Chilo suppressalis) attack identified a promoter fragment highly specifically responsive to insect feeding. Plant Mol Biol. 2007;65:519–30.
James DG. Field evaluation of herbivore-induced plant volatiles as attractants for beneficial insects: methyl salicylate and the green lacewing, Chrysopa nigricornis. J Chem Ecol. 2003a;29:1601–9.
James DG. Synthetic herbivore-induced plant volatiles as attractants for beneficial insects. Environ Entomol. 2003b;32:977–82.
James DG. Further field evaluation of synthetic herbivore-induced plant volatiles as attractants for beneficial insects. J Chem Ecol. 2005a;31:481–95.
James DG, Grasswitz TR. Synthetic herbivore-induced plant volatiles increase field captures of parasitic wasps. Biocontrol. 2005b;50:871–80.
James DG, Price TS. Field-testing of methyl salicylate for recruitment and retention of beneficial insects in grapes and hops. J Chem Ecol. 2004;30:1613–28.
Jones VP, Steffan SA, Wiman NG, Horton DR, Miliczky E, Zhang QH, Baker CC. Evaluation of herbivore-induced plant volatiles for monitoring green lacewings in Washington apple orchards. Biol Control. 2011;56:98–105.
Kalberer NM, Turlings TCJ, Rahier M. Attraction of a leaf beetle (Oreina cacaliae) to damaged host plants. J Chem Ecol. 2001;27:647–61.
Kaplan I. Attracting carnivorous arthropods with plant volatiles: The future of biocontrol or playing with fire? Biol Control. 2012;60:77–89.
Karban R, Baldwin IT. 1997. Induced responses to herbivory. Plant Pathol. Chicago (USA): Univ Chicago Pr. p. 294.
Kessler A, Baldwin IT. Defensive function of herbivore-induced plant volatile emissions in nature. Science. 2001;291:2141–4.
Kessler A, Halitschke R. Specificity and complexity: the impact of herbivore-induced plant responses on arthropod community structure. Curr Opin Plant Biol. 2007;10:409–14.
Kessler A, Halitschke R, Diezel C, Baldwin IT. Priming of plant defense responses in nature by airborne signaling between Artemisia tridentata and Nicotiana attenuata. Oecologia. 2006;148:280–92.
Kessmann H, Staub T, Hofmann C, Maetzke T, Herzog J, Ward E, Uknes S, Ryals J. Induction of systemic acquired disease resistance in plants by chemicals. Annu Rev Phytopathol. 1994;32:439–59.
Khan ZR, James DG, Midega CA, Pickett JA. Chemical ecology and conservation biological control. Biol Control. 2008;45:210–24.
Kim J, Felton GW. Priming of antiherbivore defensive responses in plants. Insect Sci. 2013;20:273–85.
Knoth C, Salus MS, Girke T, Eulgem T. The synthetic elicitor 3,5-dichloroanthranilic acid induces NPR1-dependent and NPR1-independent mechanisms of disease resistance in Arabidopsis. Plant Physiol. 2009;150:333–47.
Kopke D, Schroder R, Fischer HM, Gershenzon J, Hilker M, Schmidt A. Does egg deposition by herbivorous pine sawflies affect transcription of sesquiterpene synthases in pine? Planta. 2008;228:427–38.
Landolt PJ. New chemical attractants for trapping Lacanobia subjuncta, Mamestra configurata, and Xestia c-nigrum (Lepidoptera: Noctuidae). J Econ Entom. 2000;93:101–6.
Lee JC. Effect of methyl salicylate-based lures on beneficial and pest arthropods in strawberry. Environ Entomol. 2010;39:653–60.
Little D, Darimont CG, Bruessow F, Reymond P. Oviposition by pierid butterflies triggers defense responses in Arabidopsis. Plant Physiol. 2007;143(2):784–800.
Loreto F, Schnitzler JP. Abiotic stresses and induced BVOCs. Trends Plant Sci. 2010;15:154–66.
Lou YG, Cheng JA. Role of rice volatiles in the foraging behaviour of the predator Cyrtorhinus lividipennis for the rice brown planthopper Nilaparvata lugens (Stål). Biocontrol. 2003;48:73–86.
Lou YG, Du MH, Turlings TCJ, Cheng JA, Shan WF. Exogenous application of jasmonic acid induces volatile emissions in rice and enhances parasitism of Nilaparvata lugens eggs by the parasitoid Anagrus nilaparvatae. J Chem Ecol. 2005a;31:1985–2002.
Lou YG, Ma B, Cheng JA. Attraction of the parasitoid Anagrus nilaparvatae to rice volatiles induced by the rice brown planthopper Nilaparvata lugens. J Chem Ecol. 2005b;31:2357–72.
Lou YG, Hua XY, Turlings TCJ, Cheng JA, Chen XX, Ye GY. Differences in induced volatile emissions among rice varieties result in differential attraction and parasitism of Nilaparvata lugens eggs by the parasitoid Anagrus nilaparvatae in the field. J Chem Ecol. 2006;32:2375–87.
Lou YG, Zhang GR, Zhang WQ, Hu Y, Zhang J. Biological control of rice insect pests in China. Biol Control. 2013;67:8–20.
Lu YJ, Wang X, Lou YG, Cheng JA. Role of ethylene signaling in the production of rice volatiles induced by the rice brown planthopper Nilaparvata lugens. Chin Sci Bull. 2006;51:2457–65.
Lu J, Ju HP, Zhou GX, Zhu CS, Erb M, Wang XP, Wang P, Lou YG. An EAR-motif-containing ERF transcription factor affects herbivore-induced signaling, defense and resistance in rice. Plant J. 2011;68:583–96.
Ma B, Lou YG, Cheng JA. Effects of some biotic factors on activities of the volatiles emitted from rice plants infested by the rice brown planthopper, Nilaparvata lugens (Stål). J Zhejiang Univ (Agri Life Sci). 2004;30:589–95.
Maffei ME. Sites of synthesis, biochemistry and functional role of plant volatiles. S Afr J Bot. 2010;76(4):612–31.
Maffei M, Bossi S, Spiteller D, Mithofer A, Boland W. Effects of feeding Spodoptera littoralis on lima bean leaves. I. Membrane potentials, intracellular calcium variations, oral secretions, and regurgitate components. Plant Physiol. 2004;134:1752–62.
Mattiacci L, Dicke M, Posthumus MA. Beta-glucosidase: an elicitor of herbivore-induced plant odor that attracts host-searching parasitic wasps. Proc Natl Acad Sci USA. 1995;92:2036–40.
Meiners T, Wackers F, Lewis WJ. Associative learning of complex odours in parasitoid host location. Chem Senses. 2003;28:231–6.
Meldau S, Erb M, Baldwin IT. Defence on demand: mechanisms behind optimal defence patterns. Ann Bot. 2012;110:1503–14.
Mithofer A, Boland W. Recognition of herbivory-associated molecular patterns. Plant Physiol. 2008;146:825–31.
Mithofer A, Boland W. Plant defense against herbivores: chemical aspects. Annu Rev Plant Biol. 2012;63:431–50.
Mohan S, Ma PW, Pechan T, Bassford ER, Williams WP, Luthe DS. Degradation of the S. frugiperda peritrophic matrix by an inducible maize cysteine protease. J Insect Physiol. 2006;52:21–8.
Moraes MCB, Birkett MA, Gordon-Weeks R, Smart LE, Martin JL, Pye BJ, Bromilow R, Pickett JA. cis-Jasmone induces accumulation of defence compounds in wheat, Triticum aestivum. Phytochemistry. 2008;69:9–17.
Murai T, Imai T, Maekawa M. Methyl anthranilate as an attractant for two thrips species and the thrips parasitoid Ceranisus menes. J Chem Ecol. 2000;11:2557–65.
Newman MA, Sundelin T, Nielsen JT, Erbs G. MAMP (microbe-associated molecular pattern) triggered immunity in plants. Front Plant Sci. 2013;4:139.
Oluwafemi S, Dewhirst SY, Veyrat N, Powers S, Bruce TJ, Caulfield JC, Pickett JA, Birkett MA. Priming of production in maize of volatile organic defence compounds by the natural plant activator cis-Jasmone. PLoS ONE. 2013;8:e62299.
Orre GUS, Wratten SD, Jonsson M, Hale RJ. Effects of an herbivore-induced plant volatile on arthropods from three trophic levels in brassicas. Biol Control. 2010;53:62–7.
Pare PW, Farag MA, Krishnamachari V, Zhang HM, Ryu CM, Kloepper JW. Elicitors and priming agents initiate plant defense responses. Photosynth Res. 2005;85:149–59.
Peñaflor MF, Bento JM. Herbivore-induced plant volatiles to enhance biological control in agriculture. Neotrop Entomol. 2013;42:331–43.
Peumans WJ, Van Damme EJ. Lectins as plant defense proteins. Plant Physiol. 1995;109:347.
Pickett JA, Poppy GM. Switching on plant genes by external chemical signals. Trends Plant Sci. 2001;6:137–9.
Qi JF, Zhou GX, Yang LJ, Erb M, Lu YH, Sun XL, Cheng JA, Lou YG. The chloroplast-localized phospholipases D alpha4 and alpha5 regulate herbivore-induced direct and indirect defenses in rice. Plant Physiol. 2011;157:1987–99.
Rakwal R, Komatsu S. Role of jasmonate in the rice (Oryza sativa L.) self-defense mechanism using proteome analysis. Electrophor. 2000;21:2492–500.
Rasmann S, Kollner TG, Degenhardt J, Hiltpold I, Toepfer S, Kuhlmann U, Gershenzon J, Turlings TCJ. Recruitment of entomopathogenic nematodes by insect-damaged maize roots. Nature. 2005;434:732–7.
Rostás M, Turlings TCJ. Induction of systemic acquired resistance in Zea mays also enhances the plant’s attractiveness to parasitoids. Biol Control. 2008;46:178–86.
Runyon JB, Mescher MC, De Moraes CM. Volatile chemical cues guide host location and host selection by parasitic plants. Science. 2006;313:1964–7.
Ryan CA. Protease inhibitors in plants: genes for improving defenses against insects and pathogens. Annu Rev Phytopathol. 1990;28:425–49.
Schmelz EA, Leclere S, Carroll MJ, Alborn HT, Teal PEA. Cowpea chloroplastic ATP synthase is the source of multiple plant defense elicitors during insect herbivory. Plant Physiol. 2007;144:793–805.
Schmelz EA, Engelberth J, Alborn HT, Tumlinson JH III, Teal PEA. Phytohormone-based activity mapping of insect herbivore-produced elicitors. Proc Natl Acad Sci USA. 2009;106:653–7.
Schmelz EA, Huffaker A, Carroll MJ, Alborn HT, Ali JG, Teal PEA. An amino acid substitution inhibits specialist herbivore production of an antagonist effector and recovers insect-induced plant defenses. Plant Physiol. 2012;160:1468–78.
Schröder R, Forstreuter M, Hilker M. A plant notices insect egg deposition and changes its rate of photosynthesis. Plant Physiol. 2005;138:470–7.
Schröder R, Cristescu SM, Harren FJM, Hilker M. Reduction of ethylene emission from Scots pine elicited by insect egg secretion. J Exp Bot. 2007;58:1835–42.
Schüler G, Mithöfer A, Baldwin IT, Berger S, Ebel J, Santos JG, Herrmann G, Hölscher D, Kramell R, Kutchan TM, Maucher M, Schneider B, Stenzel I, Wasternack C, Boland W. Coronalon: a powerful tool in plant stress physiology. FEBS Lett. 2004;563:17–22.
Schuman MC, Barthel K, Baldwin IT. Herbivory-induced volatiles function as defenses increasing fitness of the native plant Nicotiana attenuata in nature. Elife. 2012;1:e00007.
Seino Y, Suzuki Y, Sogawa K. An ovicidal substance produced by rice plants in response to oviposition by the whitebacked planthopper, Sogatella furcifera (Horváth) (Homoptera: Delphacidae). Appl Entomol Zool. 1996;31:467–73.
Silipo A, Erbs G, Shinya T, Dow JM, Parrilli M, Lanzetta R, Shibuya N, Newman MA, Molinaro A. Glycoconjugates as elicitors or suppressors of plant innate immunity. Glycobiology. 2010;20:406–19.
Skibbe M, Qu N, Galis I, Baldwin IT. Induced plant defenses in the natural environment: Nicotiana attenuata WRKY3 and WRKY6 coordinate responses to herbivory. Plant Cell. 2008;20:1984–2000.
Snoeren TAL, Mumm R, Poelman EH, Yang Y, Pichersky E, Dicke M. The herbivore-induced plant volatile methyl salicylate negatively affects attraction of the parasitoid Diadegma semiclausum. J Chem Ecol. 2010;36:479–89.
Sobhy IS, Erb M, Sarhan AA, El-Husseini MM, Mandour NS, Turlings TCJ. Less is more: treatment with BTH and laminarin reduces herbivore-induced volatile emissions in maize but increases parasitoid attraction. J Chem Ecol. 2012;38:348–60.
Sobhy IS, Erb M, Lou YG, Turlings TCJ. The prospect of applying chemical elicitors and plant strengtheners to enhance the biological control of crop pests. Philos Trans R Soc B. 2014;369:20120283.
Steppuhn A, Gase K, Krock B, Halitschke R, Baldwin IT. Nicotine’s defensive function in nature. PLoS Biol. 2004;2:1074–80.
Stout MJ, Workman KV, Bostock RM, Duffey SS. Stimulation and attenuation of induced resistance by elicitors and inhibitors of chemical induction in tomato (Lycopersicon esculentum) foliage. Entomol Exp Appl. 1998;86:267–79.
Svoboda J, Boland W. Plant defense elicitors: Analogues of jasmonoyl–isoleucine conjugate. Phytochemistry. 2010;71:1445–9.
Szendrei Z, Rodriguez-Saona C. A meta–analysis of insect pest behavioral manipulation with plant volatiles. Entomol Exp Appl. 2010;134:201–10.
Thaler JS. Jasmonate-inducible plant defences cause increased parasitism of herbivores. Nature. 1999;399:686–8.
Thaler JS, Fidantsef AL, Duffey SS, Bostock RM. Trade-offs in plant defense against pathogens and herbivores: a field demonstration of chemical elicitors of induced resistance. J Chem Ecol. 1999;25:1597–609.
Thaler JS, Farag MA, Pare PW, Dicke M. Jasmonate-deficient plants have reduced direct and indirect defences against herbivores. Ecol Lett. 2002;5:764–74.
Tong XH, Qi JF, Zhu XD, Mao BZ, Zeng LJ, Wang BH, Li Q, Zhou GX, Xu XJ, Lou YG, He ZH. The rice hydroperoxide lyase OsHPL3 functions in defense responses by modulating the oxylipin pathway. Plant J. 2012;71:763–75.
Turlings TCJ, Ton J. Exploiting scents of distress: the prospect of manipulating herbivore-induced plant odours to enhance the control of agricultural pests. Curr Opin Plant Biol. 2006;9:421–7.
Vallad GE, Goodman RM. Systemic acquired resistance and induced systemic resistance in conventional agriculture. Crop Sci. 2004;44:1920–34.
Wang JH, Constable CP. Polyphenol oxidase overexpression in transgenic Populus enhances resistance to herbivory by forest tent caterpillar (Malacosoma disstria). Planta. 2004;220:87–96.
Wang P, Lou YG. Screening and field evaluation of synthetic plant volatiles as attractants for Anagrus nilaparvatae (Pang et Wang), an egg parasitoid of rice planthoppers. Chinese Bull Entomolo. 2013;50:431–40.
Wang X, Zhou GX, Xiang CY, Du MH, Cheng JA, Liu SS, Lou YG. β-Glucosidase treatment and infestation by the rice brown planthopper Nilaparvata lugens elicit similar signaling pathways in rice plants. Chin Sci Bull. 2008;53:53–7.
Wang X, Hu LC, Zhou GX, Cheng JA, Lou YG. Salicylic acid and ethylene signaling pathways are involved in the production of the rice trypsin proteinase inhibitors induced by the leaf folder Cnaphalocrocis medinalis (Guenée). Chin Sci Bull. 2011;56:2351–8.
Wang Q, Li JC, Hu LF, Zhang TF, Zhang GR, Lou YG. OsMPK3 positively regulates the JA signaling pathway and plant resistance to a chewing herbivore in rice. Plant Cell Rep. 2013;32:1075–84.
War AR, Sharma HC, Paulraj MG, War MY, Ignacimuthu S. Herbivore induced plant volatiles: their role in plant defense for pest management. Plant Signal Behav. 2011;6:1973–8.
Williams III L, Rodriguez-Saona C, Castle SC, Zhu S. EAG-active herbivore-induced plant volatiles modify behavioral responses and host attack by an egg parasitoid. J Chem Ecol. 2008;34:1190–201.
Winter TR, Borkowski L, Zeier J, Rostas M. Heavy metal stress can prime for herbivore-induced plant volatile emission. Plant, Cell Environ. 2012;35:1287–98.
Worrall D, Holroyd GH, Moore JP, et al. Treating seeds with activators of plant defence generates long-lasting priming of resistance to pests and pathogens. New Phytol. 2012;193:770–8.
Wu JQ, Baldwin IT. New insights into plant responses to the attack from insect herbivores. Annu Rev Genet. 2010;44:1–24.
Wu JQ, Hettenhausen C, Meldau S, Baldwin IT. Herbivory rapidly activates MAPK signaling in attacked and unattacked leaf regions but not between leaves of Nicotiana attenuata. Plant Cell. 2007;19:1096–122.
Xiang CY, Ren N, Wang X, Sumera A, Cheng JA, Lou YG. Preference and performance of Anagrus nilaparvatae (Hymenoptera: Mymaridae): effect of infestation duration and density by Nilaparvata lugens (Homoptera: Delphacidae). Environ Entomol. 2008;37:748–54.
Xiao YT, Wang Q, Erb M, Turlings TCJ, Ge LQ, Hu LF, Li JC, Han X, Zhang TF, Lu J, Zhang GR, Lou YG. Specific herbivore-induced volatiles defend plants and determine insect community composition in the field. Ecol Lett. 2012;15:1130–9.
Xin ZJ, Yu ZN, Erb M, Turlings TCJ, Wang BH, Qi JF, Liu SN, Lou YG. The broad-leaf herbicide 2,4-dichlorophenoxyacetic acid turns rice into a living trap for a major insect pest and a parasitic wasp. New Phytol. 2012;194:498–510.
Yan F, Wang X, Lu J, Pang BP, Lou YG. Comparison of the volatiles from rice plants infested by rice striped stem borer, Chilo suppressalis and rice leaf folder, Cnapholocrocis medinalis. Chin Bull Entomolo. 2010;47:43–7.
Yang DH, Hettenhausen C, Baldwin IT, Wu JQ. BAK1 regulates the accumulation of jasmonic acid and the levels of trypsin proteinase inhibitors in Nicotiana attenuata’s responses to herbivory. J Exp Bot. 2011;62:641–52.
Ye M, Song Y, Long J, Wang RL, Baerson SR, Pan ZQ, Zhu-Salzman KY, Xie JF, Cai KZ, Luo SM, Zeng RS. Priming of jasmonate-mediated antiherbivore defense responses in rice by silicon. Proc Natl Acad Sci USA. 2013;110:E3631–9.
Yoshinaga N, Aboshi T, Ishikawa C, Fukui M, Shimoda M, Nishida R, Lait CG, Tumlinson JH, Mori N. Fatty acid amides, previously identified in caterpillars, found in the cricket Teleogryllus taiwanemma and fruit fly Drosophila melanogaster larvae. J Chem Ecol. 2007;33:1376–81.
Yoshinaga N, Alborn HT, Nakanishi T, Suckling DM, Nishida R, Tumlinson JH, Mori N. Fatty acid-amino acid conjugates diversification in lepidopteran caterpillars. J Chem Ecol. 2010;36:319–25.
Yu H, Zhang Y, Wu K, Gao XW, Guo YY. Field-testing of synthetic herbivore-induced plant volatiles as attractants for beneficial insects. Environ Entomol. 2008;37:1410–5.
Zavala JA, Patankar AG, Gase K, Baldwin IT. Constitutive and inducible trypsin proteinase inhibitor production incurs large fitness costs in Nicotiana attenuata. Proc Natl Acad Sci USA. 2004;101:1607–12.
Zhang F, Zhu L, He GC. Differential gene expression in response to brown planthopper feeding in rice. J Plant Physiol. 2004;161:53–62.
Zhao WC, Lou YG, Cheng JA, Zhu ZR. Intra-and interspecific relationship of Nilaparvata lugens (Stål) and Sogatella furcifera (Horváth) on various rice varieties. Acta Ecol Sinica. 2001;21:629–38.
Zheng SJ, Dicke M. Ecological genomics of plant-insect interactions: from gene to community. Plant Physiol. 2008;146:812–7.
Zhou GX, Qi JF, Ren N, Cheng JA, Erb M, Mao BZ, Lou YG. Silencing OsHI-LOX makes rice more susceptible to chewing herbivores, but enhances resistance to a phloem feeder. Plant J. 2009;60:638–48.
Zhou GX, Wang X, Yan F, Li R, Cheng JA, Lou YG. Genome-wide transcriptional changes and defence-related chemical profiling of rice in response to infestation by the rice striped stem borer Chilo suppressalis. Physiol Plant. 2011;143:21–40.
Zhu J, Park K. Methyl salicylate, a soybean aphid-induced plant volatiles attractive to the predator Coccinella septempunctata. J Chem Ecol. 2005;31:1733–44.
Acknowledgments
We thank Emily Wheeler for editorial assistance. The study was jointly sponsored by the National Basic Research Program of China (2010CB126200), the National Natural Science Foundation of China (31272052), and the China Agriculture Research System (CARS-01-21).
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Zhejiang University Press, Hangzhou and Springer Science+Business Media Dordrecht
About this chapter
Cite this chapter
Lou, Y., Hu, L., Li, J. (2015). Herbivore-Induced Defenses in Rice and Their Potential Application in Rice Planthopper Management. In: Heong, K., Cheng, J., Escalada, M. (eds) Rice Planthoppers. Springer, Dordrecht. https://doi.org/10.1007/978-94-017-9535-7_5
Download citation
DOI: https://doi.org/10.1007/978-94-017-9535-7_5
Published:
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-017-9534-0
Online ISBN: 978-94-017-9535-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)