Abstract
Crown rust, caused by Puccinia coronata f. sp. lolii is a common disease of perennial ryegrass (Lolium perenne) in Europe. To study the inheritance of resistance to crown rust, a resistant genotype of the diploid cultivar Arvella was crossed with a susceptible genotype of the cultivar Aurora to generate a F1 population. Out of this population, two parents were selected on the basis of crown rust reaction and backcrossed with a susceptible Aurora genotype to generate two sets of F2 populations. All parents, the F1 and the two F2 populations were screened for their response to three single-pustule isolates (SPI) of crown rust in a detached-leaf segment test under growth chamber conditions. Genetic analysis of resistance led to the identification of two dominant resistance genes. The first gene, LpPc4, conferred resistance to SPI A and K, but not to SPI F. The second gene, LpPc5, proved to be effective against SPI F and K, but not against SPI A.
Access provided by Autonomous University of Puebla. Download conference paper PDF
Similar content being viewed by others
Keywords
Introduction
Crown rust, caused by Puccinia coronata f. sp. lolii is a common disease of perennial ryegrass (Lolium perenne) in Europe. Resistance breeding is the most efficient method of controlling this disease. Resistance of ryegrass cultivars to crown rust has been reported in several studies (Schubiger et al. 2010) and the genetics of resistance was comprehensively reviewed by Dracatos et al. (2010). On the other hand, there is evidence of different pathotypes of crown rust which differ in their virulence on particular perennial ryegrass genotypes (Aldaoud et al. 2004). However, information about the relationship between ryegrass resistance genes and crown rust pathotypes is largely lacking.
The aim of the present study was to test the hypothesis that dominant resistance genes determine the inheritance of resistance exhibited by a genotype of the perennial ryegrass cultivar Arvella.
Materials and Methods
Single-Pustule Isolates
The three single-pustule isolates (SPI), used in this study, derived from uredinial collections from Switzerland and Belgium in 2003 and 2004, respectively (Table 34.1). Spores of each collection were sprayed separately on susceptible plants of the cultivar Aurora. Following 24 h of darkness, infection occurred in a dew chamber with a 16 h photoperiod and a day/night temperature of 18 °C. Urediniospores were collected from a single uredinial pustule (presumed to originate from a single urediniospore) and inoculated on fresh detached leaves on water agar complemented with benzimidazole. Urediniospores of individual developing rust pustules were then used to inoculate plants of Aurora to produce urediniospores of a SPI for further use.
Plant Material
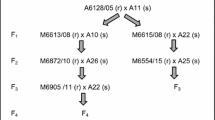
As resistant donor, the genotype Arv was used which originated from a cross between a resistant and a susceptible genotype of the diploid cultivar Arvella. Genotype Arv (resistant to the three SPI A, F and K) was crossed with a genotype A11 from the cultivar Aurora (susceptible to the three SPI A, F and K) to generate an F1 population. Out of this population, two parents were selected on the basis of crown rust reaction: Arv1 (resistant to SPI A and K and susceptible to F) and Arv2 (resistant to SPI F and K, and susceptible to A). Each of the two parents was crossed with a susceptible Aurora plant (A10) to generate two sets of F2 populations. All parents, the F1 and two F2 populations were screened for their reaction to three SPI of crown rust in a detached-leaf segment test. A total of 112 plants were tested from each cross. The plants were grown in the greenhouse for 7–8 weeks (a cut was done 5 weeks after sowing) prior to use in the detached-leaf segment test.
Detached-Leaf Segment Test
Detached-leaf segments of 2–3 week old regrowth were placed on water agar complemented with benzimidazole (35 mg l−1). SPI were applied to the detached-leaf segments using a compressed air duster. The goal of inoculation density was 400–600 urediniospores cm−2. Leaf segments were incubated for 24 h at 16 °C in the dark and then transferred to a growth chamber at 16 °C with continuous light (fluorescent lamps, 20–30 μ Mol m−2 s−1). Assessments of disease response were carried out at 14 days after inoculation, using a 0-to-4 scale (Schubiger et al. 2007). Infection types 3 and 4 were regarded as compatible (virulent SPI/susceptible host) and all others as incompatible (avirulent SPI/resistant host).
Statistical Analysis
The observed ratios of resistant to susceptible plants in the segregating populations were compared to theoretical ratios using Chi-square tests. A P-value greater than 0.05 indicates that the progeny does not deviate significantly from the presumed ratio.
Results
The F1 population derived from a cross between the genotype Arv (resistant to SPI A, F and K) and the susceptible genotype A11. The frequency of resistant and susceptible plants within the F1 population resulted in a ratio of 1R:1S for isolates A and F (Table 34.2).On the contrary, a 3R:1S segregation ratio was observed within the progeny when inoculated with the isolate K. A genotype, resistant to SPI K, was either resistant to SPI A or F as well or to both SPI: The proportion of the plants that was resistant to SPI K and A (and susceptible to SPI F), was 22 %. And 26 % of the plants were resistant to K and F but susceptible to SPI A. A quarter of the progeny (25 %) was resistant to all of the three SPI tested. Moreover, each of the genotypes susceptible to SPI K (27 %) was also susceptible to SPI A and F.
F2 families were shown to segregate for resistant versus susceptible responses to SPI depending on the parent used in the cross. Genotype Arv1, resistant to SPI A and K and susceptible to F, was crossed with the susceptible plant A10. The progeny had a good fit to the segregation ratio of 1R:1S for resistant and susceptible genotypes, when inoculated with SPI A and K but all of them were susceptible to F (Table 34.3). Each of the 60 genotypes resistant to SPI A was also resistant to SPI K.
Genotype Arv2, resistant to SPI F and K and susceptible to A, was likewise crossed with the susceptible plant A10. A segregation ratio of 1R:1S was observed within the progeny for the SPI F and K (Table 34.4). The progeny was susceptible to SPI A. The identical genotypes were resistant or susceptible, respectively, to the two SPI F and K.
Discussion
The segregation of resistant versus susceptible offspring in the F1 family suggests that two unlinked genes for crown rust resistance must have been transferred from resistance donor Arv into the F1 family: Gene LpPc4 conferred resistance to SPI A and K but not to SPI F. Gene LpPc5 conferred resistance to SPI F and K, but not to SPI A. That is to say, both genes conferred resistance to SPI K, but differed in terms of resistance response to SPI A and F, respectively. Moreover, these two genes acted dominantly and independently from each other to confer resistance.
Segregation analyses of the F2 families confirmed this suggestion. In the two F2 families, supposed to have either LpPc4 (Arv1) or LpPc5 (Arv2), the segregation fitted a 1R:1S ratio only to those SPI which were avirulent to the corresponding resistance gene.
Chances are that there are other resistance genes in the resistance donor Arv. However, these unknown genes are ineffective to the three SPI tested and the three SPI must be virulent to these genes.
Several QTL’s involved in crown rust resistance are reported in the literature Dracatos et al. (2010). The two resistance genes LpPc4 and LpPc5 have to be studied to determine whether they are different or identical to previously reported loci.
References
Aldaoud R, Anderson MW, Reed KFM, Smith KF (2004) Evidence of pathotypes among Australian isolates of crown rust infecting perennial ryegrass. Plant Breed 123:395–397
Dracatos PM, Cogan NOI, Keane PJ, Smith KF, Forster JW (2010) Biology and genetics of crown rust disease in ryegrasses. Crop Sci 50:1605–1624
Schubiger FX, Hürlimann H, Schläppi K, Streckeisen P, Boller B (2007) Virulence of crown rust isolates on genotypes of Italian and perennial ryegrass. In: Rosellini D, Veronesi F (eds) Breeding and seed production for conventional and organic agriculture. Universita’ degli Studi, Perugia, pp 301–306
Schubiger FX et al (2010) Susceptibility of European cultivars of Italian and perennial ryegrass to crown and stem rust. Euphytica 176:167–181
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media Dordrecht
About this paper
Cite this paper
Schubiger, F.X., Boller, B. (2014). Inheritance of Crown Rust Resistance in a Perennial Ryegrass Genotype of the Cultivar Arvella. In: Sokolović, D., Huyghe, C., Radović, J. (eds) Quantitative Traits Breeding for Multifunctional Grasslands and Turf. Springer, Dordrecht. https://doi.org/10.1007/978-94-017-9044-4_34
Download citation
DOI: https://doi.org/10.1007/978-94-017-9044-4_34
Published:
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-017-9043-7
Online ISBN: 978-94-017-9044-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)