Abstract
The coastline of Northern Sonora is dominated by hypersaline estuaries and vast rocky intertidal zones that are intermittently covered by the extreme tides characteristic of the Northern Gulf of California. Research on the spatial-temporal distribution of flora and fauna in wetland, sandy-muddy bottoms, the pelagic zone, subtidal rocky reefs and an offshore island offer an in depth characterization of the region’s habitats and allow the definition of a unique biological Corridor for the coastal zone between Punta Borrascoso and Puerto Lobos, Sonora. Trophic studies and coupled oceanographic-biological models validated by larval dispersal and population genetic studies on commercial species highlight the connectivity between marine and coastal habitats and support the Corridor as a distinct management unit, especially for fisheries. Patterns of human use along the coast (fisheries, tourism and coastal development) have been documented and currently stakeholders in six communities are engaged in fisheries monitoring and management. The wealth of information available on this Corridor supports an ecosystem-based approach for fisheries management. The traditional hurdles to successful implementation of ecosystem-based fisheries management can be overcome for the coastal fisheries of the Peñasco Corridor by defining essential habitats for important target species, identifying trophic interactions, involving fishers and coastal communities in spatial planning and decision-making, and creating a positive incentive system.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Ecosystem-based fisheries management (EBFM)
- Connectivity
- Marine spatial planning
- Northern Gulf of California
- Food web connectivity
1 Introduction
Estuarine environments are known to be among the world’s most important ecosystems for marine fisheries production (Houde and Rutherford 1993). In the Gulf of California , it has been estimated that 32 % of landings of small-scale fisheries depend on mangroves and coastal fringe environments (Aburto-Oropeza et al. 2008). The Northern Gulf, a distinct ecoregion of the Gulf of California , boasts of 2,116 km of coastline, three riparian systems, and 114,206 ha of coastal wetlands (Glenn et al. 2006). The region yields over 15 % of Mexico’s total fisheries volume (Brusca and Bryner 2004). Fishing is the primary economic activity for the region’s seventeen permanent fishing communities and 3500 small-scale fishers (Cudney-Bueno and Turk Boyer 1998; Moreno-Baez et al. 2012). Valuable shrimp, blue crab, clam, and scallop fisheries abound, with approximately 80 target species harvested by small-scale fishers (Cudney Bueno and Turk Boyer 1998; PANGAS 2012a, b, c, d, e).
Bound by the Midriff Island archipelago in the south and by the Colorado River Delta in the north, the Northern Gulf of California has a variety of topographic and bathymetric features and habitats that support the region’s biodiversity (CONABIO 2004 AICA NO-34; Thomson et al. 2000). The shallow sloping shelf of the Northern Gulf is characterized by extreme tides, temperatures and salinities (Alvarez-Borrego et al. 1975) that fuel a seasonally reversing oceanographic gyre (Marinone 2003), distributing nutrients, larvae and food, and connecting populations and communities throughout the ecosystem (Marinone et al. 2008).
High biodiversity is another hallmark of the region. Forty-six percent of the Gulf’s total ichthyofauna and a similar proportion of the Gulf’s macro-invertebrates are present in the Northern Gulf (Hastings et al. 2010; Brusca and Hendrickx 2010). Cartilaginous fish are especially well represented, with 70 % of the Gulf’s 87 species found in the region (Hastings et al. 2010). The Northern Gulf is also known for its large predatory fish of commercial and recreational importance: for example the families Serranidae and Scianidae are especially abundant (Rupnow 2008; Hernández-Velasco 2010; Aburto-Oropeza et al. 2008). Over 23 species of marine mammals (Urban 2010; Brusca et al. 2004) and five of the world’s seven sea turtle species (Seminoff and Nichols 2007) use the area for feeding, breeding and nesting. The coastal wetlands offer feeding and resting spots for migratory birds of the Pacific Flyway as well as many resident and breeding species (Hinojosa-Huerta et al. 2007) and serve many other ecosystem functions.
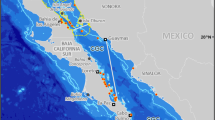
Among the most renowned species in the Northern Gulf are the endemic vaquita porpoise ( Phocoena sinus); the world’s most endangered marine cetacean (Rojas-Bracho et al. 2006; Urban 2010), and the endemic giant croaker ( Totoaba macdonaldi), one of the world’s first marine fish to be listed as endangered (Barrera-Guevara 1990; Cisneros-Mata et al. 1995). Protection of these two species has been a primary driver for biodiversity conservation and fisheries management in the entire region, but especially in the Upper Gulf of California and Colorado River Delta Biosphere Reserve (Fig. 9.1). The reserve was established in 1993 (DOF 1993) largely to protect these two species and the spawning habitat of over 22 commercially important fish species within the Colorado River Delta (Barrera-Guevara et al. 2004). A Vaquita Refuge was created in 2005 (DOF 2005) overlap-ping the Reserve, offering an additional 1,263 km2 to protect this species from entanglement in gillnets, the primary cause of incidental mortality (Rojas-Bracho et al. 2006).
The Northern Gulf of California was once a vast estuarine environment (Lavín and Sánchez 1999), fed by the mighty Colorado River and other riparian systems along the coast of Sonora, Mexico. The Colorado once contributed up to 50 % of all the freshwater entering the Gulf, with a freshwater lens extending up to 65 km south towards San Felipe, B.C. (Lavín and Sánchez 1999). Temperature and salinity profiles (Alvarez-Borrego et al. 1975), sedimentation patterns (Carriquiry and Sánchez 1999) and the perception of fishers (Cudney-Bueno and Turk-Boyer 1998) all indicate that the influence of this system was greatest along the northwestern coast of the Northern Gulf, where distinct fisheries are harvested. Today, only intermittent or subsurface flow enters the Gulf due primarily to upstream diversion for urban and agricultural use (Glenn et al. 2006; CONAGUA 2011). The impacts of the lack of freshwater from the Colorado River on the marine ecosystem have been well documented; these impacts include reductions in shrimp landings (Galindo-Bect et al. 2000), lower abundance and reduced distribution of freshwater clams (Rodríguez et al. 2001), riparian habitat loss (Glenn et al. 2001), reduction in avian breeding populations (Hinojosa-Huerta et al. 2004a, b), and alteration of reproduction and spawning habitat of the endemic totoaba population (Lercari-Bernier and Arreguín-Sánchez 2009). Incidental capture of juvenile totoaba in shrimp trawling activities (Barrera-Guevara 1990) and over-fishing (Flanagan and Hendrickson 1976) are contributing factors that led to the near extinction of this anadromous species (Rowell et al. 2008), underscoring the complexities inherent in management of coastal fisheries.
Despite the changes in freshwater input, the Upper Gulf continues to provide important ecosystem services reminiscent of estuarine conditions and primary productivity remains high (Alvarez-Borrego 2010). There is evidence, however, that fisheries populations in the region have declined (Cudney-Bueno and Turk-Boyer 1998; Sáenz-Arroyo et al. 2005; Erisman et al. 2012; Rupnow 2008). Open access, increased fishing effort, use of non-selective fishing gears, lack of management and insufficient enforcement are some of the primary causes for this decline (Cinti et al. 2010a, b). In the Northern Gulf, lack of information to advance fisheries management has been a major challenge, particularly amongst end users. In the last decade, several multi-disciplinary re-search efforts have advanced understanding of fisheries in the context of the Northern Gulf ecosystem and new strategies have emerged to solve this situation (PANGAS 2012a, b, c, d, e; Ainsworth et al. 2011).
In the Upper Gulf Reserve, where the survival of the world’s most endangered marine mammal is threatened by the region’s most important fisheries, these various management challenges are compounded and complex. Though management structures have evolved, the process has been characterized by conflict and consumed the energy of environmental and fisheries authorities. In contrast, at the southeastern corner of the Reserve a different approach to fisheries management has emerged. Here, participatory research and management processes are unfolding with the collaboration of six coastal communities that exploit distinct fisheries in a diversity of habitats. A wealth of scientific information has been generated that characterizes the Peñasco Corridor, linking coastal and marine habitats and communities from Punta Borrascoso to Puerto Lobos in Northern Sonora as a distinct eco-region. Here, important advances have been made on key components for implementation of Ecosystem-based Fisheries Management (EBFM).
This chapter explores EBFM for the Peñasco Corridor. In this Corridor, fisheries production rivals that at the mouth of the Colorado River, supported by riparian, wetland, rocky and soft-bottom habitats. Especially in areas as diverse (at the genetic, species and environmental levels) as the Peñasco Corridor, effective fisheries management can be well served by an integrated approach that recognizes the connectivity between marine and coastal systems, habitats, species and human communities. We present a characterization of the diverse habitats of this Corridor, documenting physical characteristics, overall biodiversity, presence and abundance of fisheries species, and life stages present. We describe fishing effort, the fisheries exploited in each habitat, and present an analysis of catch composition and diversity by community. We describe studies on the connectivity between habitats and species by looking at trophic structure within the estuary system and offshore environments. We also summarize research on larval dispersal and genetic structure for three important commercial species using coupled biological-oceanographic models. Finally, this work highlights an emerging participatory process for ecosystem-based fisheries management in the rich biological Corridor from Punta Borrascoso to Punta Lobos, Sonora.
The information presented here is derived from research of an ecosystem-based management research consortium studying small-scale fisheries in the Northern Gulf of California -PANGAS (http://www.pangas.arizona.edu); and from the Atlantis Ecosystem-based modeling project for the Northern Gulf ecosystem developed by the Northwest Fisheries Science Center (NWFSC), National Oceanic and Atmospheric Administration (NOAA) in collaboration with PANGAS and CEDO. Methods and results for different components of these projects are available as theses, reports, or in some cases published papers, while other publications are in process. Additional unpublished data and analyses from these studies are presented in this chapter, such as the calculation of indices of richness, trophic structure, and diversity, generation of species lists, and site-specific comparisons.
A general description of the methods employed for surveys conducted in subtidal rocky reefs of the Peñasco Corridor and Northern Gulf from 2007 to 2012 can be found in Appendix 9.1. Methods for a 2-year community catch monitoring program in the Peñasco Corridor between September 2010 to 2012 and for data analysis are outlined in Appendix 9.2.
2 Ecosystem-Based Fisheries Management
Traditional fisheries management is slowly evolving from single species stock assessment models to multi-species approaches that incorporate management of biodiversity, habitat and ecosystem processes as well as socio-economic considerations (Koen-Alonso 2009; Crowder et al. 2008; Pikitch et al. 2004). This change has resulted from the recognition of the ineffectiveness of past approaches, as well as the importance of managing not only target species, but also considering food web dynamics where both predator and prey species abundance are secured (Crowder et al. 2008) and habitats are protected for maintenance of a healthy ecosystem (Pikitch et al. 2004). Ecosystem-based fisheries management addresses many of these issues. The overall objective of EBFM is to sustain healthy marine ecosystems and the fisheries they support (Pikitch et al. 2004). Ecosystem-based fisheries management can be defined in a variety of ways, but at its core, it recognizes: (1) that the use of all resources should be sustainable for the long-term (from both ecological and socio-economic perspectives); (2) ecosystem structure and function must be preserved or restored (if possible); and (3) uncertainties, weaknesses and knowledge gaps need to be explicitly recognized and the management structure needs to address unexpected changes in system conditions (Done and Reichelt 1998).
While the Gulf of California ’s sardine fishery incorporates ecosystem approaches (Bakum et al. 2010), as yet, no integrated EBFM exists for Gulf of California fisheries (Lluch-Cota et al. 2007), although the most recent fisheries law calls for more integrated management approaches. In 2012, Mexico’s first fisheries refuges were established in Baja California Sur (DOF 2012), and a process was developed to define conditions for their establishment. Mexico has also established eleven Natural Protected Areas in the Northern Gulf (Fig. 9.1). These areas are managed by the National Commission of Natural Protected Areas (Comisión Nacional de Áreas Naturales Protegidas, CONANP), an agency of the Environmental Secretariat (Secretaría del Medio Ambiente y Recursos Naturales, SEMARNAT). A comprehensive Environmental Impact Assessment process, which is a broad environmental planning tool, is now required in all of Mexico’s protected areas (DOF 2000); to date this instrument has only been enforced for fishers in the Upper Gulf of California Reserve and Vaquita Refuge.
Lack of basic life history information on individual target species, ecological and historical data on most fisheries, and limited characterization of the diverse habitats important for fisheries has made it all but impossible to move forward with an EBFM approach in the Gulf (Lluch-Cota et al. 2007). Ecosystem modeling is a tool that can support EBFM; several ecosystem models have been developed for the Northern Gulf, where a growing body of information has made this possible (Lercari-Bernier and Arreguín-Sánchez 2009; Ainsworth et al. 2011, Díaz-Uribe et al. 2012). Nonetheless, so far there is no formal pathway to incorporate model predictions into management. Given the enormity of the task required to implement EBFM practical approaches are being sought (Cowan et al. 2012, Crowder et al. 2008). Hilborn (2011) distinguishes between core and extended EBFM and proposes that at the most basic level EBFM should: (1) manage individual species by keeping fishing mortality below Maximum Sustainable Yields and fleet capacity within the resource limits; (2) prevent bycatch of non-target species through gear modification, offering incentives to avoid bycatch, or use area and seasonal closures; and (3) avoid fishing practices that modify habitats through spatial closures in sensitive areas.
The processes necessary to implement EBFM are already present in many fisheries systems throughout the world and important advances towards implementing them are being made within the Upper Gulf of California Biosphere Reserve and the Vaquita Refuge. A permit and gear buy-out program was implemented by the Mexican government in 2008 to reduce bycatch on the vaquita porpoise (Rodríguez-Ramírez 2010). Industrial fishers in the Upper Gulf Reserve were the first group required to implement an Environmental Impact Assessment ; and in 2009 the same was required from small-scale fishers, setting an important precedent. Small-scale fishers have been active participants in conducting studies necessary for the assessment process, which includes onboard observations, fisheries monitoring, training programs, participation in decision-making processes for managing bycatch and improving compliance, based on socially accepted indicators (Pérez-Valencia et al. 2013a).
Finally, fisheries are part of a complex socio-ecological system and fishers must be incorporated into management (Gutiérrez et al. 2010; Hilborn 2011). Stakeholders need to be involved in processes to understand their fisheries, make decisions about management, and commit to comply with agreements; these aspects are as important as top-down approaches and legal tools (Cudney-Bueno et al. 2009; Espinoza-Romero et al. 2011; Cinti 2010a). Where co-management processes exist, leadership, social capital and incentives have been shown to promote success (Gutiérrez et al. 2010). The need for cross-scale forums to bring different parties together has been identified (Cudney-Bueno and Basurto 2009); many such forums are emerging in the Upper Gulf where progress is being made, such as in the Vaquita Refuge (Turk-Boyer and Barrera 2012). A balanced and comprehensive incentive system is needed to keep all parties motivated and working together towards developing functional fisheries systems (Jones et al. 2011). Socio-economic and market incentives will be the ultimate driver to maintain sustainable fisheries. Catch share systems with harvest quotas or turfs are actively being promoted in the region as one such incentive. These management tools can help prevent overfishing, promote stability and ecological stewardship (Gutiérrez et al. 2010; Fujita et al. 2012).
3 The Peñasco Biological-Fisheries Corridor
The Peñasco Biological-Fisheries Corridor, hereafter Peñasco Corridor, is located along the northeastern coast of the Northern Gulf of California between Punta Borrascoso and Puerto Lobos, Sonora (Fig. 9.2). Stakeholders from six coastal communities who engage in small-scale coastal fisheries are an integral part of this Corridor. With the exception of Puerto Peñasco, fishing is the primary economic activity for all of these communities. Puerto Peñasco has a more diversified economy with tourism, industrial (shrimp and finfish) and recreational fisheries in addition to small-scale fishing. The Corridor’s small-scale fishers engage in a variety of fisheries, predominantly using small skiffs, called “pangas”. The fisheries use a variety of gears, including commercial diving, gillnets and traps in the shallow subtidal zones up to 30 m depth, and longline and deepwater nets used in waters up to 150 m depth. Fishing for the star-studded grouper ( Hyporthodus niphobles) occurs occasionally in depths of up to 300 m using long-line, but this depth is not considered in our definition of the Corridor. An average fishing day might take a fisherman 45 km from the coast. Deeper water is closer to the shore at the southern part of the Corridor than at Puerto Peñasco.
The southern limit of this Corridor, Puerto Lobos, is the approximate latitude where the summer (May to September) counterclockwise current (Marinone 2003, 2012) turns northward along the Sonoran coast, transporting larvae along the way (Soria et al. 2012). Puerto Lobos also is the southern limit of the Puerto Peñasco regional fisheries office (Oficina de Pesca y Acuacultura, Puerto Peñasco, Sonora) which manages all fisheries issues for the communities of the Corridor as well as for the community of Golfo de Santa Clara. Given that the biological, ecological and fisheries context for Golfo de Santa Clara is distinct, we have excluded this community from the definition of the Peñasco Corridor.
4 Characterization of Habitats of the Peñasco Corridor
The variety of coastal habitats in the Peñasco Corridor, from riparian to saltwater wetlands, sandy and muddy bottom environments, intertidal and offshore rocky reefs, an island archipelago and a rich pelagic environment give sustenance to marine fisheries and a diversity of marine life.
4.1 Pelagic Environment
The offshore pelagic environment has lower salinity than other Upper Gulf sites, defining a distinct hydrogeographic zone (Sánchez-Velasco et al. 2012). Here a physical-chemical front separates the shallow sloping coastal Corridor from the deeper waters to the south, roughly corresponding to the area we describe as the “Peñasco Corridor”, up to the 40-m isobath.
Both in this pelagic zone and in adjacent wetlands of the Peñasco Corridor the abundance and richness of fish larvae are high, which underscores its relevance as nursery habitat (Iris-Maldonado 2011; Sánchez-Velasco et al. 2012). Anchoa spp., an important prey for targeted commercial species, nesting birds and sea lions populations, is the dominant species group in these waters (Iris-Maldonado 2011; Sánchez-Velasco et al. 2012). A significant portion of the nesting population of the least tern, Sternula antillarum, uses the wetlands in the Corridor (Palacios and Mellink 1996; Rosemartin 2008). Here, the terns feed and nourish their young with the abundant Anchoa spp., Leuristhes sardina, and Colpichthys regis found in the pelagic environment (Zuria and Mellink 2005; Iris-Maldonado 2011; Sánchez-Velasco et al. 2012).
4.2 Riparian Habitats
Riparian vegetation gives evidence of fresh water input at three sites along the Sonora coast (Felger 2000) (Fig. 9.3). North of Puerto Peñasco at Bahía Adair in the high intertidal zone, inside the coastal dunes, artesian springs create freshwater oases that harbor relict vegetation from the Colorado River (Felger 2000; Ezcurra et al. 1988). Riparian vegetation is also found along the now dry Sonoyta River channel (Felger 2000), which has its terminus between Estero Morúa and Estero La Pinta, once a single estuarine system (Turk-Boyer 1998). At Desemboque, the Asunción River terminates inland of the coastal dunes forming a small wetland with both mesquite trees and halophytic vegetation (Glenn et al. 2006). The marine zones offshore from these riparian areas are recognized as key spawning habitat for shrimp, blue crabs and sharks, species generally known as estuarine spawners (Calderón-Aguilera et al. 2003; Loaiza-Villanueva and Downton-Hoffmann 2011)
Coastal wetlands in the Puerto Peñasco Biological-Fisheries Corridor, Sonora, Mexico are hypersaline negative estuaries that cover 114,206 ha. (Glenn et al. 2006)
4.3 Wetland Habitats
In the Puerto Peñasco Corridor, there are eight estuaries that range in size between 235 and 98,740 ha and cover a total of 114,206 ha (Table 9.1; Fig. 9.3) (Glenn et al. 2006). These are negative estuaries with higher salinities at the head than at the mouth, resulting from high evaporation rates and lack of freshwater input (Brusca et al. 2006; Glenn et al. 2006). Environmental conditions within these estuaries are extreme and highly dependent on depth, solar heating, and tidal ex-change (Morzaria-Luna et al. 2010), with tidal fluctuations of up to 8 m (Brusca and Bryner 2004). Such extreme environmental conditions determine faunal community structure. Fish community structure is determined by tidal cycle, with higher richness and abundance during neap tide (Iris-Maldonado 2011); while fish larvae are found in zones defined by salinity and temperature gradients, with lower diversity in hypersaline waters (Sánchez-Velasco et al. 2012).
Physically, the estuaries are a complex of intertidal mudflats, tidal channels, marshes, saltpans, coastal dunes, beaches and the wetland-terrestrial ecotone (Fig. 9.4) (Ezcurra et al. 1987; Felger 2000; Johnson 1982). The Peñasco Corridor is beyond the northern limit for mangrove distribution except for a small stand of black mangroves at Puerto Lobos (Brusca et al. 2006; Glenn et al. 2006). The marsh is vegetated by halophytes and surrounded by Sonoran Desert vegetation characteristic of the Lower Colorado River Valley subdivision (Shreve and Wig-gins 1964). This is the most arid type of Sonoran Desert, because of the combination of high temperature and low precipitation (Turner and Brown 1994).
Wetland-dependent fisheries represent a large proportion of the commercial harvest in the Northern Gulf of California . For example, in 2008 shrimp landings in Sonora represented over 8000 metric t in live weight or 16 % of national production (CONAPESCA 2008). The Corridor’s most important commercial fish use these wetlands in their adult phases (Table 9.2). Fish and invertebrates of commercial importance such as shrimp and blue crab use estuaries as nursery sites. Blue shrimp ( Litopenaeus stylirostris) larvae recruit within coastal wetlands and the coastal area outside estuaries in the Corridor has the highest concentration of mature females along the coast of Sonora (Calderón-Aguilera et al. 2003). A majority of estuary fish are juveniles; an analysis of fish community structure in Estero Morúa and Estero La Salina found that 60 to ~ 80 % of fish found throughout the year were immature (Iris-Maldonado 2011).
4.4 Sandy and Muddy Bottom Habitats
The vast majority of the seafloor in the Puerto Peñasco Corridor is either sandy or muddy bottom habitat (Carriquiry and Sánchez 1999) (Fig. 9.5). The Colorado River once contributed over 160 million t of sediment per year to the Gulf (Van Andel 1964). Combined with river deposits from mainland Sonora, these sediments now cover the entire continental shelf of the Northern Gulf and fill two deep water basins with sand and mud (Carriquiry and Sanchez 1999). With no additional input today, tidal and wind driven currents continually rework these sediments (Carriquiry et al. 2011). Particle size and associated oxygen content are primary factors that determine the distribution of both infauna and epifauna in these habitats.
Substrate map showing relative distribution of major habitat types of the northern Gulf of California, Mexico: Rock ( solid black); mud ( cross-hatch); sand ( fine dots). (From Ainsworth et al. 2011)
Sandy and muddy bottom environments in the Gulf of California are known for their high diversity of invertebrate species. Considering all depths, sandy and muddy habitats account for 41.2 and 26.7 % of the total macro invertebrate species in the Gulf, respectively (Brusca and Hendrickx 2010).
Sharks and rays (Chondrichthys) are particularly diverse in soft-bottom habitats, with 52 % of the Gulf’s species present in the Upper Gulf, probably in part related to the prevalence of soft substrates (Hastings and Findley 2007). Other species groups typically found in soft bottoms in the Corridor include: anchovies (Engraulidae), herrings (Clupeidae), new world silversides (Atherinopteridae), a variety of perciform fish such as corvinas and croakers (Sciaenidae), grunts (Haemulidae), flounders and flatfishes (Pleuronectiformes) (Hastings and Findley 2007).
Sandy bottoms are the primary habitat for the region’s commercial shrimp and the valuable geoduck clam ( Panopea globosa) (Pérez-Valencia and Aragón–Noriega 2012), as well as other invertebrates of commercial importance (Appendix 9.3).
4.5 Rocky Reefs
Intertidal and subtidal rocky reefs are also found in the Northern Gulf of California . Intertidal reefs are limited to only a few sites, and three of them are in the Peñasco Corridor (Borrascoso, Puerto Peñasco, and Puerto Lobos). Subtidal rocky outcrops appear in the Corridor as a series of patch reefs that parallel the shore along the continental shelf between Bahía Adair and Bahía San Jorge, in both shallow water (2–25 m depth) and deep water (>>25 m depth) (Fig. 9.6). Composed predominantly of beach rock (limestone), the reefs appear as small exposed islands of hard substrate in a predominantly sandy and muddy bottom. These reefs were probably once shoreline intertidal habitats inundated by Pleistocene sea level rise (Thomson and Gilligan 1983). Recreational fishers identify 17 reefs they fish regularly (Pérez-Valencia et al. 2007; Rupnow 2008); while industrial trawlers have located many more rocky outcrops as targets to avoid during their trawling activities (Turk-Boyer et al. 2009).
Rocky reefs vary in size and rugosity. Some create substrate for habitat forming species, such as Sargassum spp. (Aburto-Oropeza et al. 2007) and rhodoliths (Steller et al. 2003). They offer shelter, spawning and feeding sites for large predatory fish and support coastal fisheries. Though reefs represent an essential habitat for many species, they are a patchy resource, limited in geographical coverage and physically separated from other reefs. Despite their limited abundance, rocky habitats harbor 33.4 % of the total Gulf macroinvertebrate faunal species (Brusca and Hendrickx 2010), with mollusks and echinoderms commonly associated with these habitats (Keen 1971; Brusca 1980; Maluf 1988; Cintra-Buenrostro 2001; Solís-Marín et al. 2005).
4.6 Offshore Islands
The San Jorge Island reefs are the most important offshore rocky habitats in the Peñasco Corridor in terms of diversity (Martinez 2010). San Jorge is the northern-most rocky island in the Gulf, located approximately 41 km southeast of Puerto Peñasco. With one main island and four islets, San Jorge is an island archipelago that extends 1,097 m in length. The islands are composed of granite and are arid and steep, with terrestrial and intertidal habitats that decline precipitously into subtidal rocks. San Jorge Island is a protected area under the CONANP category of Special Area for Protection of Flora and Fauna (Area Especial de Protección de Flora y Fauna: Islas del Golfo de California, Sonora), but the marine portion of this island archipelago is not included in this designation and is under no special management or protection category.
The San Jorge Island reefs represent one of the Northern Gulf’s biodiversity hot spots. Diversity indices for fish show the island to be among the highest of the Northern Gulf (Martinez 2010). Large predatory fish are abundant; the island presented the highest average trophic level for fish of all surveyed sites in the Northern Gulf in 2010 and 2011. Subtidal surveys in 2007 and 2011 show commercially important invertebrates were most abundant at San Jorge Island when compared to other sites in the Northern Gulf. Rock scallop ( Spondylus limbatus), black murex snail ( Hexaplex nigritus), pen shells, the pearl oyster ( Pinctada margaritensis), and Octopus spp. (Martinez 2010) were the most abundant at San Jorge Island. The island also had the second greatest abundance of the brown sea cucumber ( Isostichopus fuscus). Winged oyster ( Pteria sterna), an important fishery in the Corridor, was more abundant at Angel de la Guarda and Tiburon Island.
San Jorge has no terrestrial plants but supports an important seabird colony (Everett and Anderson 1991; Velarde and Anderson 1994). Reported nesting species include brown boobies ( Sula leucogaster), blue-footed boobies ( Sula sula), double-crested cormorants ( Phalacrocorax auritius), Heerman’s gulls ( Larus heermanni), and red-billed tropicbirds ( Phaethon aetherus) (Velarde and Ander-son 1994). The island is the Gulf’s second most important breeding site for the California sea lion ( Zalophus californianus californianus); sea lion censuses conducted in 2004 found 3,822 individuals with 968 pups (Szteren et al. 2006), but in July 1998, an ENSO year that number was almost double (6,717 sea lions and 793 were pups) (Mellink and Romero-Saavedra 2005).
5 Fishing Communities
The Peñasco Corridor is comprised of six fishing communities along a 200 km stretch of coast (Fig. 9.2). The largest community is Puerto Peñasco (pop. 57,342 in 2010; INEGI 2011). Puerto Peñasco also has the most diversified economy with small-scale , industrial, and recreational fisheries as well as commerce, tourism, other service industries and related activities; the municipal government is also based here. The other communities of the Corridor are much smaller and have varying degrees of infrastructure and access to basic services and education. Three of these coastal communities are fishing camps, associated with inland ejidos as follows: Bahia San Jorge (BSJ)—Ejido Rodolfo Campodónico; Santo Tomas (STO)—Ejido Álvaro Obregón; Punta Jagüey (PJA)—Ejido 15 de Septiembre. In this work we use the names of the coastal villages. The local fisheries office that services these communities is a dependency of the National Commission for Fisheries and Aquaculture (Comisión Nacional de Acuacultura y Pesca-CONAPESCA). The CONAPESCA office is located in Puerto Peñasco, where all fisheries catch is officially landed. Because of the distance, most of the catch from communities south of Puerto Peñasco was largely unreported until a community logbook program was initiated in September 2010–2012 (Downton-Hoffmann et al. 2013c). Due to its size, Puerto Peñasco has the largest number of pangas (Table 9.3). In contrast, Santo Tomás has 37 total (20 active) pangas, and no fishing permits. Bahia San Jorge, the fishing community that is most organized, has only one cooperative fishing unit and all the members of the community belong to it.
Puerto Peñasco is the community that targets the most species (46) and Bahía San Jorge the least (13). Only four target species are shared by all six communities: Gulf drum (chano), guitarfish, blue crab and flounder (Downton-Hoffmann et al. 2013c). A cluster analysis of catch composition separates Bahía San Jorge and Punta Jagüey, communities close to each other that fish in nearby sites and have similar catch (Downton-Hoffmann et al. 2013c; Appendix 9.2). The second cluster groups the two communities that are furthest apart from each other, Puerto Lobos and Puerto Peñasco. Both of these communities harvest resources in rocky habitats, which are likely connected via larval dispersal (see section on marine connectivity ). Desemboque also has catch in common with Puerto Lobos, but the latter also fishes in other types of habitats.
An analysis of the trophic level of catch by community shows in general the communities of the Corridor are impacting top predators, with average trophic level ranging between 3.5 and 3.8 (Fig. 9.7). The exceptions are the communities of Bahía San Jorge and Punta Jagüey, which fish predominantly in Bahía San Jorge and lower down the food chain.
Average trophic level of catch from the six communities of the Puerto Peñasco Corridor, Northern Gulf of California, Sonora. Based on catch data from September 2010 to September 2012. (Downton-Hoffmann et al. 2013c)
Habitat characterizations and fishery-use patterns define a distinct coastal zone and oceanographic region for the Corridor. The offshore limits for the Corridor definition are based on depth and/or distance from the shore. Fishing with traps, hooka and gillnets takes place in shallow areas. In Puerto Peñasco and Bahía San Jorge most areas used by small-scale fisheries do not exceed 30 m depth; while at Desemboque, Puerto Lobos and Santo Tomas fishing takes place in waters up to 150 m deep (Fig. 9.8), which is relatively close to shore. In the deepest areas (300 m), the star-studded grouper is caught using long line and some sharks are also captured, but most catch comes from below the 100 m isobath. At depths between 60 to 100 m longline and gillnets are used to catch angel shark, smoothhound shark and large flounders. Distance from shore might be the most realistic way to define the Corridor, as logistically and economically this distance is what limits daily trips.
Fishing zones for communities of the Peñasco Corridor in the Northern Gulf of California, Sonora. (Downton-Hoffmann et al. 2013c, Appendix 9.3)
6 Fisheries in the Peñasco Corridor
A total of 75 species are captured, either as target species or secondary catch by small-scale fishers from the six communities of the Peñasco Corridor. For communities other than Puerto Peñasco the most important commercial fish species include blue crab, winged oyster, squid (occasionally), murex snail, and guitarfish, flounder, angel shark, smoothhound shark and drum (Fig. 9.9) and at Puerto Peñasco geoduck clam, blue crab, and black and pink murex snail are the most important fisheries. Most of these are soft-bottom species or species that use both soft bottom and hard substrates.
Composition of small-scale fishing catch (in percentages) from five communities of the Peñasco Corridor, Sonora, Mexico in 2011, excluding Puerto Peñasco. (Downton-Hoffman et al. 2013a)
Though it does not register as a dominant species anymore, shrimping is an important fishery occurring in sandy and muddy habitats in the Peñasco Corridor, though it is not all backed by permits. Shrimp represented 10 % of the total catch landed in Puerto Peñasco in 2011 (OEIDRUS 2011), including landings from small-scale and industrial fishermen. Small-scale fishers use 2 ¾ inch gillnets from small boats with outboard motors called pangas, while industrial otter trawlers target over nine shrimp species. Approximately 80 % of the catch from trawlers is composed of blue shrimp ( Litopenaeus stylirostris) and most of the remaining 20 % are brown shrimp ( Farfantepenaeus californiensis) and white shrimp ( Litopenaeus vannamei), all of high economic value.
Trawling for finfish takes place after the shrimp season is closed; this fishery is directed towards big fishes such as flounder (families Paralichthyidae and Pleuronectidae), trigger fish (cochito, Balistes spp), gold-spotted sand bass (extranjero, Paralabrax spp.), corvinas ( Cynoscion spp.), Pacific hake ( Merluccius spp.) and several species of elasmobranchs (rays and sharks). Occasionally, when trawls are done close to reefs, predatory sea bass and groupers are caught as well.
The extensive subtidal sandy and muddy plains of the Bahía Adair and Bahía San Jorge Corridor are key habitats for commercially important benthic mollusks harvested by commercial divers, based mainly in Puerto Peñasco. Diving takes place in water up to 30 m, depending on the species, generally with the use of surface supplied air (hookah). Geoduck clam ( Panopea globosa), white clam ( Dosinia ponderosa), Chione spp. clams and speckled scallop ( Argopecten circularis) as well as other bivalve species; pen shells ( Pinna spp.) and winged oyster are the most important of these fisheries (Cudney Bueno and Turk Boyer 1998; Downton-Hoffman et al. 2013a). The spatial distribution of geoduck clam beds (Perez-Valencia and Aragón-Noriega 2012), and winged oyster beds (INAPESCA 2011; Downton-Hoffmann 2012) have been delineated in this region. Also the density of black murex snail (Loaiza-Villanueva et al. 2012; Cudney-Bueno 2000) and blue crab (Loaiza-Villanueva and Downton-Hoffmann 2011) is known from some areas.
The rocky reef habitats of the Peñasco Corridor are especially important sites for artisanal fisheries (Cudney-Bueno and Turk-Boyer 1998). The shallow water reefs (<25 m) near Puerto Peñasco are used mostly by commercial divers who harvest rock scallop, black murex snail and other abundant species. Shallow and deeper water reefs are used by recreational fishers who seek out large predatory fish such as groupers.
In 2009, there were 207 recreational users at Puerto Peñasco (35 small local operators, 32 local guides working aboard yachts, and 140 tourists with their own boats operating out of La Cholla, the main recreational port (Martinez et al. 2011). In 2003, recreational fishers in Bahía La Cholla mentioned that they used 27 reefs (Rupnow 2008), landing 26 species. Ninety-five percent of their catch consisted of 8 species, in order of importance: triggerfish “cochito” ( Balistes polylepis), spotted sand bass or “cabrilla arenera” ( Paralabrax maculatofasciatus), gold-spotted sandbass “extranjero” ( Paralabrax auroguttatus), red snapper “pargo”, ( Lutjanus peru), gulf grouper or “baya” ( Mycteroperca jordani), spotted cabrilla or “pinta” ( Epinephelus analogus), and leopard grouper or “cabrilla sardinera” ( Mycteroperca rosacea), Gulf corvina (Rupnow 2008) and shortfin corvina “curvina de orilla” ( Cynoscion parvipinnis) (Pérez-Valencia et al. 2007) caught by local guides. Other snappers, bass and groupers are also targeted, including the rare and endangered black sea bass, Stereolepis gigas, and other top predators.
In 2011, the jumbo squid fishery, Dosidicus gigas, appeared as an anomaly in the two most southern communities of the Corridor, Desemboque and Puerto Lobos. This fishery had never before been registered this far north, though the species has been observed in the region occasionally. This species is known to invade and colonize new areas by varying its behavior and diet to suit local environmental conditions (Bazzino et al. 2010).
7 Human Impacts
Overfishing and incidental catch are the greatest threats to the marine habitats in the Peñasco Corridor. The cumulative impacts of over 60 years of trawling on the soft bottoms of the Upper Gulf have raised the most concern (Lluch-Cota et al. 2007). Bycatch volume and ratio for shrimp trawling in Pacific waters ranges from 5 to 10.2 kg of bycatch per 1 kg of shrimp, with the ratio on the higher end for Sonora and the Gulf of California . Much of the bycatch from industrial trawling has no market value because it is composed of small individuals (García Caudillo and Gómez Palafóx 2005). Industrial trawlers from Puerto Peñasco reported 110 bycatch species in the 2004–2005 season, dominated (61.1 %) by 15 species, including big eye croaker, bronze-striped grunt, stingray, lizardfish, weak-fish, flatfish, swimming crab, robust swimming crab, sardine and shark. Small-scale fisheries that target the same species once they reach commercial size are impacted by trawler bycatch and associated diminished recruitment. Additionally, the cumulative effect of trawling on overall biodiversity and on the resilience of sandy bottoms can be high (Lluch-Cota et al. 2007; Higuera-Ciapara and Mayorga-Ríos 2006; Arreguín-Sánchez et al. 2005; Pauly et al. 1998; Rogers et al. 1999). Dive fisheries have low incidental catch and their overall environmental impact is considered low. Studies are underway to determine the environmental impact of the geoduck clam fishery, which only started in the Peñasco Corridor in 2007 (Pérez-Valencia and Aragon-Noriega 2012). Geoducks are extracted by using high powered air compressors to blow away sediments over the deeply-embedded clam.
In 2010, the community of Puerto Peñasco participated with the other communities within the Upper Gulf Biosphere Reserve in a comprehensive participatory Environmental Impact Assessment for the nine major small-scale fisheries that take place in the Reserve (Pérez-Valencia et al. 2012). Through this process, specific mitigation actions were identified for their most important fisheries, and fishers agreed to continue to develop spatial-temporal and gear solutions to reduce bycatch of the critically endangered vaquita porpoise.
Coastal areas in the Puerto Peñasco Corridor have been impacted mostly through tourism-related developments (Guido 2006). In 1972, Estero Puerto Peñasco was converted to a marina to support the large industrial fleet (Munro-Palacio 2007). Over 50 % of the original area of Estero La Cholla was converted for a residential tourist development (Glenn et al. 2006). In Estero La Pinta, 20 % of the original wetland area has been affected by dredging and filling for tourism activities (Turk-Boyer 2008). In Mexico, beaches and wetlands up to 20 m inland from the highest tide line are public domain (Federal Maritime-Terrestrial Zone) (Cortina-Segovia et al. 2007). Long-term use and exploitation of the Federal Zone requires an environmental review and permits from the Environmental Secretariat, which can define use and in some cases limit conversion of coastal areas to other uses (Morzaria-Luna et al.2013)
In 2012, the Puerto Peñasco Tourist Homeport project, proposed to receive 50,000 tourists and 48 cruise ships per year, was approved for construction along Sandy Beach, northeast of Puerto Peñasco (SEMARNAT 2012). With the construction of a 3,000 m long and 51 m wide sea wall, the project is poised to change sediment dynamics along the coast and alter key soft bottom and rocky habitats used by commercial divers for harvest of the valuable geoduck clam and rock scallop fisheries (Cudney-Bueno et al. 2009; Pérez-Valencia and Aragón–Noriega 2012).
8 Food Web Linkages Between Fisheries Species in the Peñasco Corridor
Food web considerations are important in EBFM; there is increasing emphasis on the importance of understanding food web linkages and in maintaining the diversity of such linkages (Marasco et al. 2007). In the most simplistic view, assuring an adequate food supply for fisheries species is key for sustaining fisheries populations. The first step for management is to identify the diet of the primary fisheries species and to understand trophic linkages because changes in one component of the food web can have cascading effects on other components of the food web and the ecosystem overall (Crowder et al. 2008). Overharvesting low on the food chain can have disproportionately larger impacts on the top of the food chain, and harvesting top predators leads to simplified community structure and lack of stability or resilience in the system (Pauly et al. 2002; Essington et al. 2006).
Diet composition of functional fisheries groups of the Northern Gulf was determined through stomach content analysis (Ainsworth et al. 2009). Figure 9.10 shows the relative importance of different prey to different fisheries groups identified for the Northern Gulf of California , which included samples collected from the Peñasco Corridor and in the Guaymas region.
Diet composition of functional fisheries groups of the Northern Gulf of California as defined for the Atlantis ecosystem model, derived from stomach content analysis of fish in the Peñasco Corridor (Ainsworth et al. 2009)
The abundant bivalves and mollusks that inhabit the Corridor’s rocky reefs and sandy-muddy bottoms and support Puerto Peñasco commercial dive fisheries, are part of the diet of blue crab, guitarfish, smoothhound shark and predatory murex snails (Table 9.4). Other non-commercial species, mussels for example ( Modiolus capax), are a key component of the diet of the black murex snail and other species. Small pelagic fishes sustain many of the region’s fisheries species: Gulf croaker, gold-spotted sand bass, flatfish and shrimp (Table 9.4). There is currently no formal fishery for the small pelagic species in this region of the Gulf.
Many species are opportunistic feeders and change diet as food availability varies. This shows the importance of linking food abundance to oceanographic changes and climate conditions (Marasco et al. 2007). For the Corridor, the variation in abundance and distribution of small pelagics could be an important indicator of fisheries productivity and of the health of the entire ecosystem (Velarde et al. 2004). Prey of the most important target species in the Corridor were abundant in the analysis of Ainsworth et al. (2009), highlighting the region as an important feeding habitat for fisheries species. Through identification of linkages in the food web it becomes possible to connect species and habitat use, which in turn can inform spatial planning processes.
9 Identification of Essential Fish Habitat for Different Life Stages
Essential fish habitat includes areas that are most favorable for fish populations to spawn, feed and mature (Levin and Stunz 2005). Fishery management plans in the US and the European Union take into consideration the essential fish habitat of exploited species (Bellido et al. 2008; Levin and Stunz 2005). Fishery managers are required to identify threats to essential fish habitats and design steps to ameliorate those threats (Lindeman et al. 2000). As with protected area initiatives, the essential fish habitat designation can be used to assure the sustainable use of exploited coastal resources (Lindeman et al. 2000). Although Mexico does not consider essential fish habitat within the current regulatory framework, identification of these key habitats for exploited species can nonetheless be used to support biodiversity conservation and fisheries management. Understanding which habitats and sites in the Puerto Peñasco Corridor serve as essential fish habitat could help target conservation efforts to protect a variety of species, lead to better regulatory decisions for fisheries management, and allow efficient use of limited conservation resources (Beck et al. 2001; Levin and Stunz 2005). Identification of essential fish habitats is crucial for ecosystem-based management of fisheries (Bellido et al. 2008; Levin and Stunz 2005).
Different approaches have been used to identify essential fish habitat of high conservation priority, including quantitative assessments of habitat effects at specific life history stages (Levin and Stunz 2005), spatial modeling (Valavanis et al. 2004), habitat suitability index modeling (Brown et al. 2000), and analysis of life-history characteristics including recruitment, seasonal abundance and growth rates by habitat (Gallagher and Heppell 2010). These approaches require key information for managed groups, including potential spawning aggregation sites, larval duration patterns and inshore and offshore use areas for different life stages (Lindeman et al. 2000). Often, commercial fishing activities take place over essential fish habitat (i.e. spawning aggregations); thus mapping fishing grounds can also provide relevant information on areas of biological importance (Erisman et al. 2012). Within the Puerto Peñasco coastal Corridor, emphasis has been placed in identifying essential fish habitat for fish larvae and juvenile fish (Iris-Maldonado 2011; Peguero-Icaza et al. 2008, 2011), and predatory reef fishes (Aragón-Noriega et al. 2009; Erisman et al. 2007, 2012). The spatial distribution of fishing activities has also been assessed for fisheries within the coastal Corridor (Aragón-Noriega et al. 2012; Cudney-Bueno and Turk-Boyer 1998; Erisman et al. 2012; Moreno-Báez 2010; Rodríguez-Quiroz et al. 2010) (Fig. 9.8).
Table 9.5 summarizes the primary habitats of the dominant fisheries species in the Peñasco Corridor, generated from catch data records collected during 2011 (Downton-Hoffmann et al. 2013a). For this analysis dominant fisheries species are defined as the top 75 % of the catch volume and/or species that generated 75 % of the income for that community. Presence or absence of these species in a given habitat is indicated in Table 9.5. Quantitative assessments of abundance of each of these species are available at some sites and for some life stages, but not all. In the case of some of the habitats data is available at multiple sites and for multiple seasons, which adds another dimension for assessing critical sites, key for spatially explicit management considerations.
10 Wetland Connectivity
Wetlands along the Puerto Peñasco Corridor provide a connection between the marine system and the surrounding Sonoran Desert (Glenn et al. 2006); they export organic nutrients (i.e. from bird guano), detritus (i.e. from halophyte plants), prey (i.e. invertebrates), and consumers (through foraging across habitats and migrations) (Polis et al. 1996; Rose and Polis 1998; Spackeen 2009). Thus, coastal wetlands in the Northern Gulf represent true “desert oases” that concentrate resources in the middle of an arid landscape (Glenn et al. 2006). Trophic connectivity, the transfer of food resources which can occur through the movement of either consumers or resources across habitats (Talley et al. 2006), has just begun to be studied for estuaries and surrounding environments (Alvirde-López 2012; Spackeen 2009); however linkages can be inferred from similar systems.
Estuaries and the near shore marine environment are likely linked functionally by flows of nutrients and organic matter moved through the tidal cycle (Polis et al. 1997). Salt marshes contribute to secondary productivity in nearby coastal waters through in situ consumption of marsh-derived sources, the export of dissolved and detrital organic carbon and nutrients, and the movement of fish and shellfish (Weinstein et al. 2000). The degree of contribution by salt marsh outwelling to coastal productivity is unclear, as primary production in estuaries in the coastal Corridor has not been assessed. However, stable isotope analysis of the food web in Northern Gulf estuaries suggests that phytoplankton and macrophyte detritus maintain secondary estuarine productivity (Alvirde-López 2012; Spackeen 2009).
The role of large consumers in transporting materials into the marine environment likely plays an important role for estuarine trophic connectivity. Within salt marshes, cyclic and ontogenetic movements provide trophic connectivity among habitats and with the marine environment (Rountree and Able 2007). Many fish and invertebrate species use estuaries during larval and juvenile stages, then move out as adults where they inhabit nearby subtidal habitats (Cudney-Bueno and Turk-Boyer 1998; Iris-Maldonado 2011). In this way, movements of fish and zooplankton driven by the macrotidal regime may facilitate rapid nutrient translocation (Polis et al. 1997). Adult fish, entering estuaries during spring tides (Iris-Maldonado 2011) feed and then transport materials from marshes to coastal environments. Coastal fisheries are supported by the export of juvenile fish and invertebrates from marsh channels (Calderón-Aguilera et al. 2003; Cudney-Bueno and Turk-Boyer 1998; Loaiza-Villanueva et al. 2009). Waterbirds feeding on fish and invertebrates also concentrate and transport large amounts of nutrients in their guano (Polis et al. 1997). Many migratory water birds, moving through the Pacific Flyway (Vega et al. 2006) use estuaries as wintering and stopover sites feeding on invertebrates in the mudflats (Glenn et al. 2006). Guano can then increase nutrient status and primary production in the near shore marine environment (Stapp and Polis 2003).
Marine productivity can also subsidize estuaries, through input of phytoplankton. The importance of phytoplankton can be inferred from its high palatability and the efficiency of algae-derived food webs (Deegan and Garritt 1997). The isotopic signatures of wetland dependent species from Northern Gulf estuaries, such as blue crab, suggest that marine phytoplankton is an important carbon source (Alvirde-López 2012; Spackeen 2009). Tidal exchange also deposits macroalgal wrack onshore; wrack can then be broken down by invertebrates which are food for shore birds (Polis et al. 1997). Estuaries may also provide a link between the Sonoran Desert and the marine ecosystem of the Gulf of California . For example, terrestrial reptiles and mammals, such as the coyote ( Canis latrans) subsidize their diets extensively with marine resources (Rose and Polis 1998). Trophic connectivity of estuaries should be taken into account when considering habitat protection mechanisms. However, there is little information regarding the distance offshore reached by estuarine resources or the distance that marine resources travel to subsidize estuaries. Thus, it is unclear how far is it necessary to protect marine areas in order to ensure adequate connectivity (Talley et al. 2006).
11 Marine Connectivity
Demographic connectivity between populations is characterized by relatively large amounts of propagules that have measurable effects on fisheries over ecological timescales. Genetic connectivity on the other hand usually involves fewer propagules that are key for biodiversity conservation (including genetic diversity and structure) over evolutionary time scales and affect the evolutionary potential for adaptation (Lowe and Allendorf 2010). The seasonally reversing oceanographic gyre that characterizes the predominant ocean circulation of the Northern Gulf (Marinone 2003) is the primary engine that drives marine connectivity (both demographic and genetic) in the region (Marinone et al. 2008; Marinone 2012). Since most marine organisms pass through larval dispersal phases, ocean currents are key for their transport between populations. Currents also move passive particles such as nutrients and contaminants to and from estuarine environments (Marinone 2012). By coupling biological information, such as spawning time, habitat, and larval behavior, with oceanographic models that predict ocean flow (Coupled-Biological Oceanographic models or CBOM) it is possible to begin to understand how populations are connected (Metaxas and Saunders 2009). Such information is key for understanding the flow of matter and energy within the ecosystem and for effective marine spatial planning and can significantly improve management and conservation strategies, particularly when currents are strongly asymmetric (Gaines et al. 2003; Beger et al. 2010; Kininmonth et al. 2011) as in the Northern Gulf. Cyclonic (counter-clockwise) circulation from May-September and anticyclonic circulation (clockwise) from October-April (Marinone 2012) create an asymmetrical system in the Northern Gulf. With some exceptions (e.g., geoduck clam, catarina scallop), most commercial species in the Northern Gulf spawn during May to September (Soria et al. 2014).
A coupled biological oceanographic model developed for the Northern Gulf of California (Marinone et al. 2008) was used to analyze connectivity in three fisheries species that spawn during May-July and that are of prime economic importance in the Northern Gulf ecosystem with special emphasis on the Peñasco Corridor (Soria et al. 2012; Soria et al. 2014). These three species are broadcast spawners with distinct life histories: Blue crab is a muddy bottom species with long pelagic larval duration (PLD) of 70 days, the leopard grouper is a rocky reef species with intermediate PLD (30 days) and the rock scallop is a rocky reef species of shorter larval duration (PLD = 21 days) (Soria et al. 2012). CBOM models of all species were validated using empirical data from multiple hypervariable genetic markers and, in the case of the rock scallop, data from larval spat abundance on artificial collectors at various sites where suitable habitat occurs was used as well (Soria et al. 2012) (Fig. 9.11).
Marine connectivity for three commercial species that spawn during May-July in the Northern Gulf of California, Mexico. Sites (nodes) are connected via larval dispersal ( links) according to oceanographic models validated with population genetics. Arrows show the direction of the dispersal events that follow the anti-cyclonic circulation (Soria et al. 2012; Soria et al. 2014). For clarity, some sites are not situated on their actual coastal location
Larval dispersal within the Peñasco-Lobos Corridor is analogous to a river that flows in a single direction (i.e., anti-clockwise for summer spawners, Fig. 9.11). This implies that upstream sources (e.g., Puerto Lobos) are key for sustaining fisheries and biodiversity at downstream sites (e.g., Bahia San Jorge) (Beger et al. 2010; Kininmonth et al. 2011). In such an asymmetric system, the spatial scale of connectivity scales-up with the length of the PLD. However, connectivity does not decrease with increasing distance. Instead, connectivity is low between some adjacent populations, and high between many non-adjacent localities, in a pattern described as diagonal connectivity. This points to potential challenges where local communities might not benefit from conservation and management actions within their nearby fishing sites, but may benefit from what happens at distant upstream sites.
In general, high levels of gene flow are found in the Northern Gulf of California for blue crab (Callinectes bellicosus), leopard grouper and rock scallop (Spondylus limbatus), which prevents local differentiation at most sites. However, modeling and empirical evidence indicates that Bahía San Jorge in the Upper Gulf, shows higher levels of genetic structure, larval retention and genetic relatedness, suggesting high isolation within this bay for multiple species (Soria et al. 2012). Despite large tidal fluxes, slow residual currents seem to explain why larvae get caught up in this bay. It seems that local circulation patterns within this bay likely contribute to isolating populations of other species as well, which is corroborated by the evolution in-situ of micro-endemic fish (e.g., Huang and Bernardi 2001).
Additional results for rock scallop showed connectivity (genetic and demographic) between the fished subpopulations of the Peñasco Corridor and southern sites (Soria et al. 2012). For the rock scallop, Puerto Lobos and Desemboque de Caborca which are located 150 km south of Puerto Peñasco, are potentially important upstream larval sources at the ecological scale, but Puerto Libertad, Las Cuevitas (~ 175 km away) and Desemboque de los Seris (~ 200 km away), showed weak connectivity links that imply connectivity with the Corridor at the evolutionary and genetic scales (Soria et al. 2012). On the other hand, sites as distant as Ángel de la Guarda could also be genetically linked to San Jorge Island (Soria et al. 2012). For blue crab, besides sources within the Corridor, other sites that provide significant amounts of larvae at the ecological scale include the Guaymas region south of Tiburon Island, separated by 325 km. During June, when currents reach their highest speed, marine reserves located as far away as San Pedro Martir Island (350 km distant) are key sources of larvae that could affect the demography of commercial fish populations in the Peñasco-Lobos Corridor in the case of species with a PLD of 30 days, such as leopard grouper (Soria et al. 2014). These results highlight that EBFM within the Corridor depends on multiple upstream sources that are distant and not intuitive.
San Jorge Island had the highest rock scallop spat recruitment (35–45 %) in the Corridor (Soria et al. 2012), and of all the islands surveyed throughout the Northern Gulf; it had the greatest adult densities of rock scallop (Martinez et al. 2011).
In 2002 a network of experimental marine reserves was established in the Puerto Peñasco Corridor, including a reserve around San Jorge Island, through a community-based process with a group of local divers concerned about declining populations of rock scallop and other benthic fisheries (Cudney-Bueno et al. 2009). Authors hypothesized that increases in rock scallop abundance documented at the northern-most sites of the area could be attributed to sources within the rocky reef network and that given the prevailing currents, San Jorge Island in particular, could be acting as a key source for larval export to reserves and fishing grounds on the Peñasco coast (Cudney-Bueno et al. 2009). High genetic relatedness at Bahía San Jorge further suggested that local retention was also contributing to the observed recovery at nearby sites (i.e. Sandy Beach and La Choya), established as voluntary marine reserves.
The high level of connectivity along the Corridor and the high potential for local recruitment within Bahía San Jorge have important implications for management of the Corridor. In Bahía San Jorge, local communities that participate in management decisions and actions have the potential to reap the benefits of their actions directly (Cudney-Bueno et al. 2009). On the other hand, given that fishing intensity is high in the Corridor (Moreno-Baez 2010) and increasing at the margin of the Upper Gulf Reserve where fishing activities are being restricted, it is also important to guarantee the input of larvae from more distant upstream sources within the Corridor (such as Puerto Lobos in the case of rock scallop and Desemboque for blue crab), and even in upstream locations outside of the Corridor. Protecting distant larval sources can be an important strategy for maintaining the evolutionary potential of species in the face of global climate change.
By coupling information on biological connectivity with species distribution and location of essential habitats for key species within the Corridor with patterns of use of local communities, we can begin to create management scenarios that make sense considering ecosystem trends and offer acceptable tradeoffs for the benefit of conservation, management and economic sustainability of local communities. For example, the decreasing degree of overlap of fishing zones used by communities of the Corridor from south to north (Fig. 9.8) and the grouping of Puerto Peñasco and Puerto Lobos according to the commercial species captured at each site might relate to these oceanographic dynamics.
12 Discussion: Management of the Puerto Peñasco Corridor
Until the economic boom of the mid 2000s, the Peñasco Corridor was an isolated stretch of beach, where fishers went about their business with little knowledge of or concern for regulations, except at Puerto Peñasco. As natural as it is to pick wild berries, coastal inhabitants would go out to fish. There was little local understanding of how to obtain fishing permits or why it was necessary. The end result is that even today approximately 75 % of the Corridor’s fishers are irregular; with inadequate legal access to all the resources they fish. Fishers also come from nearby towns or states into the Corridor and access fisheries intermittently with or without permits. While controlling access to resources is one way to control overexploitation, traditional fishers need to be legitimized to move forward in creating a functional fisheries system.
In general, local fisheries institutions have evolved from individual fishers to permit holders, cooperatives and then federations of cooperatives in the larger communities. These different institutions are varied in their level of organization and effectiveness for collective action. The level of organization also varies by community. For some fisheries, such as shrimp, no new permits are available, while permits for new fisheries can be obtained. This dynamic offers potential for creating good management structures, as new permits require monitoring of resources. Independent contractors are often employed for this monitoring, but when a local organization with a commitment to sustainable fisheries and conservation is employed in the process, the dynamic can be very different. Training in business management, monitoring, negotiations and many other processes contributes to improving internal operations. Participation in broader scale processes offers an important learning opportunity for local institutions. Over 15 years of work with one cooperative at Puerto Peñasco has shown impressive results in terms of their organization and collective action capacity, but also highlights the commitment required.
At its peak in the 1980s the number of industrial boats operating in the Upper Gulf reached 240 (Lluch-Cota et al. 2007; Meltzer et al. 2012; Galindo-Bect et al. 2000), the majority of these working out of Puerto Peñasco. Beginning in 2000, fishing activities with incidental catch greater than 50 % were officially prohibited in all Natural Protected Areas in Mexico, and the industrial trawlers in the Upper Gulf, with a home base at Puerto Peñasco, were the first group required to present a formal impact assessment. Fish excluders, turtle excluders, and other gear modifications are now mandatory. By 2012, only 58 boats were authorized at Puerto Peñasco, though another fisher group has a request for authorization pending to fish in the Reserve. A new impact study presented by trawlers in 2012 proposed spatial-temporal restrictions that in combination with reductions in fleet size would effectively reduce the area trawled in the Reserve by 43 % compared to previous estimates. While the season for trawling in the Reserve is only 3 months, outside the Reserve, in the Peñasco Corridor, for example, it extends through March.
In 2009, small-scale fishers of the Upper Gulf Reserve began working under mitigation measures mandated as part of the Environmental Impact Assessment (EIA) process, which includes a comprehensive monitoring program to document reduction in impacts (Pérez-Valencia et al. 2012). Through this process specific mitigation actions were identified for their most important fisheries, and they agreed to continue to develop spatial-temporal approaches to reduce impacts on the critically endangered vaquita porpoise. The EIA program requires fishers to develop training, logbook monitoring, onboard observation and participatory programs including creating a decision-making body and indicators to measure their compliance with the programs they outlined. This type of process is unprecedented in Mexico and throughout the world for traditional economic activities such as fishing. The fishers hired a local conservation NGO (Non Governmental Organization) to partner with them to develop these programs and have indicated their willingness to find the appropriate trade-offs to protect this porpoise, which is the primary driver for management in the region. It is too early to see ecosystem-wide results from these efforts. Ample evidence exists, however, to raise concerns about increased effort in areas outside the Reserve’s boundaries, which are often more heavily targeted as a result of fishing restrictions within protected areas (Kellner et al. 2007). This process is being observed at Puerto Peñasco, at the southeast margin of the Reserve, where fishers have the option to fish within or outside the Reserve, and prefer to fish outside rather than participate in the Environmental Impact process and associated costs. Some fishers from San Felipe, B.C. have expressed similar approaches to new regulations in the Reserve. At Puerto Peñasco this has the effect of increasing effort and impacts on the Corridor.
A new shrimp law (NOM-002-PESC-2013, in DOF 2013) is poised to change these dynamics even further. Through a 3 year process beginning in 2013 shrimp fishing with gillnets will be phased out in the Upper Gulf Reserve and Vaquita Refuge and replaced with a light trawl, known as RS-INP-MX to be pulled from a skiff, or other gear types as they are developed. Fishers, especially at Puerto Peñasco, are resistant to adopting this new gear, but incentives to switch are being offered by the government.
The Upper Gulf small-scale fishers’ environmental impact study is being managed at the level of communities and federations, but it is also working through cooperatives to reach individual fishermen. Decision-making committees were formalized in each community in May and June 2013 to move the impact study process forward and increase compliance. The cross-scale management groups that have formed in association with the Upper Gulf Reserve and Vaquita Refuge have mostly focused on forwarding vaquita conservation. In the Peñasco Corridor, on the other hand, collective action has taken place around individual species. Permit holders of black murex snail, for example, have gathered to discuss management options; Blue crab Subcommittees of Aquaculture and Fisheries, which link to state committees of the Secretary of Aquaculture and Fisheries, have formed to manage this resource across the Corridor. In some cases more formal integration of groups for better management of the entire chain of custody of a resource has been promoted. The Productive System is a model promoted by CONAPESCA for improving management and marketing of a product. Operated as a civil association, a Productive System organization was created for blue crab in the state of Sonora and one was initiated for the geoduck clam. These organizations are meeting with varying degrees of success depending on the leadership. An Integrator, which collects a group of producers for formalizing their commercialization of a product, is an-other type of institution and one has been created for geoduck clam, but many challenges to successful operation still exist.
A variety of management actions have been implemented on fisheries and coastal habitats of the Corridor. The Upper Gulf environment is generally known to be dynamic and unpredictable (Cudney-Bueno and Turk Boyer 1998). One year a species is abundant, the next it is not, and then new species such as the giant squid appear. The communities in the region have learned to adapt to these conditions and opportunities. The fisheries regulatory systems, however, are less adaptable. Obtaining permits for a new species is a slow process. Economic security and food security are considerations. There are few legitimate economic opportunities for communities in the region and buffers against this are needed for the most vulnerable communities (Morzaria-Luna et al. 2013). Management should consider creating sustainable development alternatives.
Management plans have been developed for rock scallop and geoduck clam), fisheries that are being managed with turfs and quotas for individual beds, while management regulations for other species are still being developed (blue crab, octopus) (PANGAS 2012c, d). Feasibility permits for the winged oyster fishery have been granted to two cooperatives at Desemboque. Permits regulate access to some of these fisheries by controlling the number of boats, gear, and sometimes dictating a quota and season.
Rock scallop is the only commercial species harvested from rocky reefs which has a management plan. This species is listed in Mexico as protected (NOM-059-ECOL), based on its population status in southern Mexico (DOF 2010). As such it cannot be exploited with commercial fishing permits, but can be harvested under a strict management program that is supervised by Mexico’s Wildlife Department (Cudney-Bueno 2007). Rock scallop has been harvested sustainably by a group of commercial divers since 2008 when they received a permit and a quota to harvest the species in an exclusively-designated area of the Peñasco Corridor. The management plan requires annual censuses of different fishing beds, estimation of biomass, and designation of site-specific quotas, season closures and size limitations (Martinez and Turk-Boyer 2011). Under this management regime, San Jorge Island was maintained as an area of high density of the species until 2011–2012, when densities began to drop. New highly valued fisheries (geoduck clam, Mexican bay scallop, Argopecten circularis, and concave scallop, Euvola vogdesi) have attracted many into diving fisheries, increasing pressure on the benthic resources (Martinez and Turk-Boyer 2011) such as rock scallop.
Commercial species such as black and pink murex snail, rock scallop and winged oyster inhabit the patch reefs that are interspersed throughout the Corridor, but the snail is also associated with the sandy bottoms where they may find prey nearby. For such benthic species, delineation of beds and management of quotas offer a good option to maintain viable densities. Such management tools are being used for geoduck clam, winged oyster and rock scallop. Use of turfs with clearly defined property rights and quotas by bed can be an important means for managing soft-bottom habitats, patch reefs and associated species. Turf rights are given in a variety of ways. The CONAPESCA permitting process gives access to a resource in a particular area and may delimit particular beds within the area for extraction by a resource user. The permit process might also designate some beds for conservation within a fisherman’s designated area, as has been done with geoduck clam. Fishermen might also be granted property rights through the Secretary of the Environment’s Wildlife Department, which gives exclusive use of an area for protected species that have management plans, such as rock scallop. Finally, long-term guarantees for the use of a resource can be obtained with a concession, which gives exclusive use of an area for extraction for an extended period of time. The process for obtaining a concession is more complicated and requires a Technical-Economic Study that evaluates the capacity of the solicitor to administer the technical, administrative and financial components of the resource adequately. As geoduck fishers obtain commercial permits, they are looking to guarantee their rights for this valuable resource through concessions.
Turfs can be effective for protecting habitats and species, only if there is compliance on the part of fishers backed by official enforcement. The number of divers that have access to diving resources in the Corridor has increased, and with this the overlap in areas of use for different species has also increased. If concessions for multiple species were possible, such that specific areas could be exclusive to a defined group of fishers, it would help resolve this issue and strengthen the sense of ownership for protection of the resources and habitat. This situation has evolved almost naturally for the San Jorge community, which has unified into one cooperative. The geographic extent of access given through fishing permits has traditionally been very extensive, however, allowing overlap between fishers and conflicts to ensue. If property rights (permits or concessions) are matched to actual fishing zones in each community the chances of management success can be increased. Implementation of such a system would be challenging, as it would limit access that fishermen currently have to larger areas, but information is available to assist in taking these steps. Local fisher knowledge has been used in the Corridor to generate a large scale perspective on spatial and temporal use of fishing zones and overlap (Cudney Bueno and Turk Boyer 1998; Moreno-Baez et al. 2012). By Coupling this information with the finer scale spatial temporal assessment obtained from 2010–2012 daily catch records (Fig. 9.8), we have a multi-dimensional perspective that can inform management decisions.
San Jorge Island stands out among the rocky habitats of the Corridor for its biodiversity value and importance for commercial species. Until recently use and impact of fishing on the island has been low. Marine connectivity studies highlight the island as a potential link with more southerly rocky habitats (Midriff islands), and as a larval source for more northerly sites, such as the rocky reefs at La Choya, Sandy Beach and Borrascoso. These studies indicate that the offshore waters at San Jorge Island represent a good site to protect, either through marine conservation tools or as fisheries refuges. This strategy could guarantee dispersal of larvae to the coastal sites along Puerto Peñasco, and could conserve the evolutionary potential of the island by maintaining connectivity with sites further south. San Jorge Island might also be an important larval source for sites as distant as Rocas Consag within the Upper Gulf Reserve, which is already a protected site and possibly a source for more southerly sites along the Baja coast. Trophic structure of catch has been calculated and related to standing trophic structure at San Jorge Island. The high trophic level of fish species at the island suggests special management for the large predatory species that live there might be in order. These are targeted by commercial fishers as well as recreational fishers, who have indicated an understanding of the importance of protecting the island (Rupnow 2008). This data on trophic level offers an important baseline for measuring changes in ecosystem health.
Mexico’s first fisheries refuges were formalized in Baja California Sur in 2012 (DOF 2012); this designation offers an alternative for protecting the important biodiversity of San Jorge Island that might be socially acceptable. The establishment of fisheries refuges in Mexico in 2012 and definition of a process for their creation sets the stage with fisheries authorities for approaching management in the Corridor from an ecosystem perspective. The new law allows for establishment of networks of refuges as well.
During 2002–2003, commercial divers voluntarily ceased fishing activities at the island, creating a no-take area around its perimeter (Cudney et al. 2009). Closed areas in coastal reefs (where new development projects have been approved) were paired with fishing areas and 2 years of data yielded important results showing the benefits of marine reserves for two dive fisheries (Cudney et al. 2009). The benefits were seen most clearly in reefs at the northern end of the study area within the Peñasco Corridor. The rapid recovery of rock scallop and black murex snail populations was attributed to local larval recruitment by dispersion from the more southerly sites, including San Jorge Island (Cudney et al. 2009). Subsequent studies of larval recruitment for rock scallop (Soria 2010; Marinone et al. 2008), one of the primary target species studied, confirmed the importance of local recruitment for this species and give a wider view of recruitment throughout the Northern Gulf (Soria et al. 2012) (see Sect. 9.11 on marine connectivity).
A time series of on-site density by bed available for rock scallop shows a clear decrease in density at San Jorge Island compared to other sites, a reflection of increased fishing pressure on diving fisheries in general (Martinez and Turk Boyer 2011). Though a good management plan exists for this species, quotas are determined annually by bed, legal access is defined, and the users are committed to complying with regulations, a widespread lack of enforcement of laws, turfs, and other regulations has caused a disintegration of good management practices. Until there is a strong enforcement process for these benthic fisheries and others, it will be difficult to maintain fisher interest in responsible fishing. Some changes have been observed in the Upper Gulf Reserve where cross-scale forums have fishers and government authorities at the table together and fishers make their pleas for enforcement clear.
Puerto Lobos represents another potential rocky site for special management. Here populations of rock scallop, winged oyster, and possibly other species (black murex snail) disperse to sites near San Francisquito and Puerto Peñasco, where they are accessed by a large number of divers. Diving at Puerto Lobos is a new occupation. There are no permits for rock scallop, but the winged oyster population has shown a resurgence here and access was opened up for two cooperatives in 2010 and 2011. Dive fisheries are increasing in this community as an economic option. New banks of the winged oyster fishery have also appeared north of San Jorge Island. When fishers try to obtain access to new fisheries, it offers an opportunity to establish limits to areas that might be important for recruitment.
Studies on marine connectivity highlight the importance of Desemboque, the mouth of the Asunción River, for recruitment of blue crab. We also know that this is a key spawning site for shrimp, shark and blue crab. Shark fishing is not permitted at the mouth of a river, and all three species have closed seasons during summer reproduction. Enforcement activities are minimal in the area. Members of this community recognize that they are extracting resources during reproduction and that resources congregate in front of their community for this purpose. The children in this community have actively promoted that fishers respect the laws; offering promise that this kind of social pressure might influence compliance. Identification of viable alternatives for this fishing community should be investigated, while also seeking a more enforceable type of closure for this key habitat through the use of fisheries refuges. The social capital that has been built in this community through education programs and other activities has potential for being harnessed in support of tangible management action such as refuges. Little is known about the opportunities that might exist to secure upstream water sources from the Asunción River for environmental purposes, but it should be considered for maintenance of this important spawning site.
Most of the region’s important fisheries species (except squid, Gulf croaker and gold-spotted sandbass) use wetland habitats as nursery and/or feeding grounds. For this reason, management of wetland habitats directly affects fishing activities. Several wetlands in the Corridor are partially protected by several tools and management processes. The Bahía Adair wetland complex and offshore sandy and muddy habitats are located within the Upper Gulf of California and Colorado River Delta Biosphere Reserve (CONANP 2007) and were declared a Ramsar Wetland of International Importance in 2009 CONANP (CONANP 2011). The Biosphere Reserve is managed through a management program and other policy instruments, such as the Environmental Impact Assessment . In 2010 the San Jorge wetland complex to the southeast of the Reserve was also designated a Ramsar Wetland of International Importance (CONANP 2011). A participatory management plan has been produced for Bahía Adair under the Ramsar designation, while processes have initiated for developing a management program for the San Jorge wetlands (see Chap. 11 in this series). At Bahía Adair federal zone transfer agreements were obtained for conservation, with 306 ha registered (DOF 2012). At Estero Morúa a federal zone transfer agreement was obtained for the municipality of Puerto Peñasco for 68,257 m2 (DOF 2012) for protection of nesting sites of the Least tern. Estero La Salina in the San Jorge wetland complex is part of Mexico’s Important Bird Areas Program (CONABIO 2004).
Federal and municipal concessions, the Reserve’s management plan, and Ramsar designations all are potential deterrents to development pressure, and help maintain the integrity of these coastal systems, but don’t directly address fisheries management. A more active strategy, however, is underway through participatory processes being promoted as part of the Ramsar sites at Bahía Adair and Bahía San Jorge. In Chap. 11 these processes are described in detail; landowners and fishers of these wetlands have engaged actively in management of resources, zoning, and development of sustainable, economic alternatives, such as monitoring and ecotourism. The social capital developed in these sessions will be leveraged for formalizing a management group for this larger concept of a Biological-Fisheries Corridor. In fact landowners in both bay regions have formalized a network for sharing experiences.
Additional evidence discussed in the section on connectivity suggests a degree of local larval retention in the Peñasco Corridor for multiple species, which lends itself to implementing local management actions in which the users will benefit directly, as was the case with the temporary reserves implemented in 2002–2003 (Cudney-Bueno and Basurto 2009; Cudney-Bueno et al. 2009). In that case, the socio-ecological feedback that resulted from fishers involvement in monitoring helped fishers maintain interest in the reserves until outsiders began to benefit from their management actions (Cudney-Bueno et al. 2009). Multiple processes are currently underway with local fishers to engage them to understand and make decisions about the management of different habitats and species of the Corridor.
Understanding of trophic interactions in the Corridor has advanced significantly. The abundance and importance of small pelagics for sustenance of the Gulf croaker, gold-spotted sand bass, fish and shrimp, all commercially important species, is rivaled only by the importance of bivalves, some of commercial value, which support crabs, snails and shark fisheries species. Several commercial species are a key part of the diet of others: shrimp is an important prey for flounder for example and crabs sustain Gulf coney, sharks, and gold-spotted sand bass. Care must be taken when managing these species for maximum sustainable yield that there is biomass available to support the diet of other targeted species. As yet there is no fishery for the small pelagic species in this region, but diving fisheries for bivalves have increased, with a potential impact on targeted populations that depend on these as prey. Whatever management scenarios and targets are proposed, they must recognize these important interactions. Controlling access to small pelagics, or mussels (which are not yet actively fished), through the permitting process for new fisheries, might help limit new pressures on these important prey resources. Especially for benthic resources, management of key habitat or areas through fisheries refuges or marine protected areas with spatial or season closures might offer some protection if clearly identified.
Though no formal integrated management initiative is yet underway in the Peñasco Corridor, many of the steps outlined by the U.S. National Marine Fisheries Service (1999) for implementing EBFM have advanced significantly. By leveraging fisher participation, government and foundation funding, collaboration with two consortiums of researchers and NGOs, and over 30 years of community experience and trust, progress has been made in collecting baseline data for forwarding EBFM in the Peñasco Corridor as discussed in this Chapter. Much of the data was collected through collaborative processes with fishers, and so the resulting co-produced data sets the stage for dialogue about potential management scenarios.
To promote an ecosystem approach to management in the Corridor will require consolidation of a management group that shares an ecosystem vision. An analysis of catch data (Downton-Hoffmann et al. 2013c) suggests that it would be appropriate to start with the communities of Puerto Peñasco and Puerto Lobos based on their similarity of catch and shared habitats. Connectivity studies also suggest the logic in promoting shared processes in resource management of these two communities as well as other groupings to initiate the collaborative integrated process. The next step for moving forward will be to survey the local communities about their objectives with fisheries management, collaborative processes and socio-economic goals. This will be followed with the generation of a specific set of management scenarios that address community interests, ecosystem processes and fisheries management needs and presenting these to the management group. The tremendous amount of data generated in this project will be overwhelming to most of the stakeholders that are involved. We will need to develop some alternative scenarios that illustrate clear tradeoffs to allow communities and other stakeholders to collectively evaluate the direction for management of the Corridor. Atlantis is a spatially explicit ecosystem model built for the Northern Gulf ecosystem that might be used to evaluate tradeoffs and operationalize decision-making, although it is unclear whether it can manage data at the scale of the Corridor.
Most of the management scenarios that emerged through analysis of this ecosystem suggest the use of spatial planning instruments. Spatial planning might incorporate fisheries refuges, clear definition of boundaries and access rights through permits or concessions, while at the same time address fisheries and ecosystem goals. Definition of access rights through the use of areas and/or quotas can be an important incentive for fishers in this region, where such basic rights as permits are lacking. In return for legal access to resources, fishers might be open to restrictions in spatial or temporal use that would otherwise be unacceptable. It will be important to evaluate stakeholders’ interest before structuring a set of viable management scenarios.
It will be necessary to assess where uncertainty lies in this system and incorporate buffers in management and conservation actions. Changes in the dynamics of the oceanographic system due to climate change are one such uncertainty that impacts larval recruitment, trophic linkages and fisheries dynamics. Connectivity studies illustrate the importance of considering both distant and local source populations to manage species in both ecological and evolutionary time frames. Though we propose the Peñasco Corridor as an ecosystem-level management unit, we also recognize the need to relate it to Northern Gulf ecosystem dynamics and the opportunity to create a network of managed areas that offer insurance against such uncertainties.
We developed a proposed set of indicators to measure ecosystem health and fisheries equilibrium, based on the data from studies on biodiversity and fisheries within the Corridor (Table 9.6). These indicators cover measures of ecosystem structure, biodiversity conservation and equilibrium of the potential resources. These indicators, if developed, monitored and evaluated through collaborative processes, can serve as a key communication tool for implementing EBFM and adapting management for the benefit of society, fishers and the ecosystem.
An important next step is to determine socio-economic management objectives and simple indicators that track economic health and well-being with input and for the use of local communities. The final set of indicators will be realized through a participatory process with the appropriate stakeholder groupings. We propose establishing a set of management scenarios, using the information generated about the ecosystem and the preliminary lists of both ecological, fisheries and socio-economic indicators as a guide.
We have described various monitoring processes that have been implemented for monitoring targeted catch, bycatch, size structure and trophic level of catch, biomass, and in situ density of various species. Through both the PANGAS initiative and the Environmental Impact Study process, procedural manuals have been developed that present standardized monitoring methods that can be used over time and throughout the ecosystem (Downton-Hoffmann et al. 2013a, b, c; Pérez-Valencia et al. 2013a, b). The ecosystem initiative supported by the David and Lucile Packard Foundation provided the funding to initiate this monitoring process. Today fishers and government agencies are sharing in this responsibility as well. To access a new species fishers must work with a research organization to conduct the needed monitoring. Fishermen also are financially responsible for implementing the programs associated with the Environmental Impact Study in the Upper Gulf Reserve, and have contracted third parties to develop the programs and conduct the needed monitoring. Within the Reserve CONANP has offered financial support to fishers through their PROCODES (Programa de Conservación para el Desarrollo Sostenible) Program and state fisheries agencies in both Baja California and Sonora have contributed together with NGOs to support fishers’ financial obligations for this process. State and national fisheries authorities are giving priority to ordering fisheries in the Upper Gulf. To insure success a clear and transparent plan of co-responsibility for financing and implementing monitoring will be needed. Involvement of fishers in monitoring has the advantage of reducing costs, but also is an important mechanism of socio-ecological feedback on the state of the resources. Precedents are already established with fishers in the region to participate in monitoring and financing such processes. In general there is a forward momentum for implementing many components of Ecosystem-based fisheries management in the Peñasco Corridor.
13 Conclusions
While there are many ways to approach managing fisheries, marine systems and, most importantly, their users are space-based. Many authors contend that place-based management and marine spatial planning can provide the most promising approach to implementing ecosystem-based fisheries management (Crowder et. al 2008; McLeod et al. 2005). Some consider this should be the second tier in implementation of EBFM, simply because of the amount of data required. Place-based management will require a hierarchy of management practices, beginning with a collective vision for ecosystem-based management and moving toward an integrated approach that contemplates multiple spatial and temporal scales. The key will lie in developing governance systems that align stakeholder incentives with management objectives.
Meaningful involvement of government agencies is also crucial. The Peñasco Corridor is not a region of high priority: the populations it serves are small communities, marginalized in many ways. Until recently the region was isolated by poor quality roads and other basic services are minimal. It is just such communities, however, that need help in sustaining their livelihoods that depend almost exclusively on resource extraction. Our experience has shown that such communities are open to dialogue, interested in connection and willing to participate in such processes, setting the stage for success.
References
Aburto-Oropeza O, Sala E, Paredes G et al (2007) Predictability of reef fish recruitment in a highly variable nursery habitat. Ecology 88(9):2220–2228
Aburto-Oropeza O, Ezcurra E, Danemann G et al (2008) Mangroves in the Gulf of California increase fishery yields. Proc Natl Acad Sci U S A 105:10456–10459. doi:10.1073/pnas.0804601105
Ainsworth CH, Kaplan IC, Levin PS et al (2009) Diet compositions of fish species in the Northern Gulf of California. In: Morzaria-Luna HN (ed) A handout of the integrative marine ecology. Ecosystem Sciences Program, Northwest Fisheries Science Center, NOAA, Seattle, p 11
Ainsworth CH, Kaplan IC, Levin PS et al (2011) Atlantis model development for the Northern Gulf of California. US Dept Commer NOAA Tech Memo NMFS-NWFSC–110
Alvarez-Borrego S (2010) Physical, chemical and biological oceanography of the Gulf of California. In: Brusca R (ed) The Gulf of California: biodiversity and conservation. University of Arizona and The Arizona-Sonora Desert Museum, Tucson, pp 24–48
Alvarez-Borrego S, Flores-Báez BP, Galindo-Bect LA (1975) Hidrología del Alto Golfo de California II. Condiciones durante invierno, primavera y verano. Cien Mar 2(1):21–36
Alvirde-López SB (2012) Estimación de la red trófica en el estero Morúa, Sonora, México, un humedal costero del Golfo de California. Universidad Autónoma de Baja California Sur
Aragón-Noriega AE, Valenzuela-Quiñones W, Esparza-Leal HM et al (2009) Analysis of management options for artisanal fishing of the Bigeye Croaker Micropogonias megalops (Gilbert 1890) in the upper Gulf of California. Int J Biodivers Sci Ecosyst Serv Manage 5:208–214. doi:10.1080/17451591003709371
Aragón-Noriega EA, Cisneros-Mata MA, Rodríguez-Quiroz G (2012) Artisanal fishery of elasmobranches within two marine protected areas in the upper Gulf of California. Indian J Fish 59:1–6
Arreguín-Sánchez F, Lercari D, Galván-Piña VH et al (2005) Modelación de ecosistemas como soporte para manejo de recursos marinos: Casos de estudio en el Pacífico Mexicano. XI Congreso Latinoamericano de Ciencias
Bakum A, Babcock EA, Lluch-Cota SE et al (2010) Issues of ecosystem-based management of forage fisheries in “open” non-stationary ecosystems: the example of the sardine fishery in the Gulf of California. Rev Fish Biol Fish 20:9–29. doi:10.1007/s11160-009-9118-1
Barrera-Guevara JC (1990) The conservation of Totoaba macdonaldi (Gilbert), (Pisces: Scianidae), in the Gulf of California, Mexico. J Fish Biol 37(Suppl. A):201–202
Barrera-Guevara JC, Parra Salazar I, Albar-Reynoso MP et al (2004) El Golfo de California: Prioridades de Conservación. World Wildlife Fund Mexico
Bazzino G, Gilly WF, Markaida U et al (2010) Horizontal movements, vertical-habitat utilization and diet of the jumbo squid ( Dosidicus gigas) in the Pacific Ocean off Baja California Sur, Mexico. Prog Oceanogr 86:59–71
Beck MW, Heck KL, Able KW et al (2001) The identification, conservation, and management of estuarine and marine nurseries for fish and invertebrates. BioScience 51(8):633–641
Beger M, Linke S, Watts M et al (2010) Incorporating asymmetric connectivity into spatial decision making for conservation. Conserv Lett 3(5):359–368. doi:10.1111/j.1755-263X.2010.00123.x
Bellido JM, Brown AM, Valavanis VD et al (2008) Identifying essential fish habitat for small pelagic species in Spanish mediterranean waters. Hydrobiologia 612:171–184. doi:10.1007/s10750-008-9481-2
Brown SK, Buja KR, Jury SH et al (2000) Habitat suitability index models for eight fish and invertebrate species in Casco and Sheepscot bays, Maine. North Am J Fish Manage 20:408–435. doi:10.1577/1548-8675(2000) 020&<0408:HSIMFE&>2.3.CO;2
Brusca RC (1980) Common intertidal invertebrates of the Gulf of California. The University of Arizona Press, Tucson
Brusca RC, Bryner GC (2004) A case study of two Mexican biosphere reserves: the upper Gulf of California and Colorado river delta and the El Pinacate and Gran Desierto de Altar biosphere reserves. In: Harrison NE, Bryner GC (eds) Science and politics in the international environment. Rowman and Littlefield Publishers, Lanham, pp 28–64
Brusca R, Hendrickx M (2010) Invertebrate biodiversity and conservation in the Gulf of California. In: Brusca R (ed) The Gulf of California: biodiversity and conservation. University of Arizona and The Arizona-Sonora Desert Museum, Tucson, pp 72–95
Brusca RC, Findley LT, Hastings PA et al (2004) Macrofaunal biodiversity in the Gulf of California. In: Cartron JLE, Ceballos G, Felger R (eds) Biodiversity, ecosystems, and conservation in Northern Mexico. Oxford University Press, New York, pp 179–203
Brusca RC, Cudney-Bueno R, Moreno-Báez M (2006) Gulf of California esteros and estuaries. Analysis, state of knowledge, and conservation and priority recommendations. Final Report to the David and Lucile Packard Foundation by the Arizona-Sonora Desert Museum
Calderón-Aguilera LE, Marinone SG, Aragon-Noriega EA (2003) Influence of oceanographic processes on the early life stages of the blue shrimp ( Litopenaeus stylirostris) in the upper Gulf of California. J Mar Syst 39:117–128
Carriquiry JD, Sánchez A (1999) Sedimentation in the Colorado river delta and upper Gulf of California after nearly century of discharge loss. Mar Geol 158:125–145
Carriquiry J, Villaescusa J, Camacho-Ibar V et al (2011) The effects of damming on the materials flux in the Colorado river delta. Environ Earth Sci 62:1407–1418. doi:10.1007/s12665-010-0626-z
Cinti A, Shaw W, Cudney-Bueno R, Rojo M (2010a) The unintended consequences of formal fisheries policies: social disparities and resource overuse in a major fishing community in the Gulf of California, Mexico. Mar Policy 34:328–339
Cinti A, Shaw W, Torre J (2010b) Insights from the users to improve fisheries performance: fishers’ knowledge and attitudes on fisheries policies in Bahia de Kino, Gulf of California, Mexico. Mar Policy 34:1322–1334
Cintra-Buenrostro CE (2001) Los Asteroideos (Echinodermata: Asteroidea) de aguas someras del Golfo de California, México. Oceánides 16:49–90
Cisneros-Mata MA, Montemayor-Lopez G, Roman-Rodriquez MJ (1995) Life history and conservation of Totoaba macdonaldi. Conserv Biol 9(4):806–814
CONABIO (2004) AICA NO-34. Bahía e Islas de San Jorge. http://conabioweb.conabio.gob.mx/aicas/doctos/NO-59.html. Accessed 29 June 2009
CONAGUA (2011) Estadísticas del agua en México. Comisión Nacional del Agua. Secretaría de Medio Ambiente y Recursos Naturales, Comisión Nacional del Agua, Mexico
CONANP (2007) Programa de conservación y manejo de la Reserva de la Biosfera Alto Golfo de California y Delta del Río Colorado. Comision Nacional de Areas Naturales Protegidas, Mexico
CONANP (2011) Listados de Sitios Ramsar. http://ramsar.conanp.gob.mx/lsr.php. Accessed 1 Oct 2011
CONAPESCA (2008) Anuario estadístico de acuacultura y pesca 2008. CONAPESCA, Mexico
Cortina-Segovia S, Brachet-Barro G, Ibañez de la Calle M, Quiñones-Valades L (2007) Océanos y costas. Análisis del marco jurídico e instrumentos de política ambiental en México. Secretaría de Medio Ambiente y Recursos Naturales. Instituto Nacional de Ecología, Mexico
Cowan JH Jr, Rice JC, Walters CJ et al (2012) Challenges for implementing an ecosystem approach to fisheries management. Mar Coast Fish 4(1):496–510
Crowder LB, Hazen EL, Avissar N et al (2008) The impacts of fisheries on marine ecosystems and the transition to ecosystem-based management. Annu Rev Ecol Evol Syst 39:259–278
Cudney-Bueno R (2000) Management and conservation of benthic resources harvested bysmall-scale hookah divers in the Northern Gulf of California, Mexico: the black murex snail fishery. MS thesis. University of Arizona
Cudney-Bueno R (2007) Plan de manejo regional para el aprovechamiento y la recuperación del callo de escarlopa, Spondylus calcifer. Documento preparado para la Dirección General de Aprovechamiento de Vida Silvestre. Centro Intercultural de Estudios de Desierto y Océanos, Puerto Peñasco
Cudney-Bueno R, Basurto X (2009) Lack of cross-scale linkages reduces robustness of community-based fisheries management. PLoS One 4(7):6253. doi:10.1371/journal.pone.0006253
Cudney-Bueno R, Turk-Boyer PJ (1998) Pescando Entre Mareas del Alto Golfo de California: Una Guía Sobre la Pesca Artesanal, Su Gente, y Sus Propuestas de Manejo. Serie Técnica # 1. Centro Intercultural de Estudios de Desiertos y Océanos, Puerto Peñasco
Cudney-Bueno R, Bourillón L, Sáenz-Arroyo A et al (2009) Governance and effects of marine reserves in the Gulf of California, Mexico. Ocean Coast Manage 52:207–218
Cudney-Bueno R, Lavin M, Marinone S, Raimondi P, and Shaw W (2009) Rapid effects of marine reserves via larval dispersal PLoS One 4:1 e4140. doi:10.1371/journal.pone.0004140
Deegan LA, Garritt RH (1997) Evidence for spatial variability in estuarine food webs. Mar Ecol Prog Ser 147:31–47. doi:10.3354/meps147031
Díaz-Uribe JG, Arreguín-Sánchez F, Lercari-Bernier D et al (2012) An integrated ecosystem trophic model for the north and central Gulf of California: an alternative view for endemic species conservation. Ecol Model 230:73–91. doi:10.1016/j.ecolmodel.2012.01.009
DOF (1993) Decreto por el que se declara área natural protegida con el carácter de Reserva de la Biosfera, la región conocida como alto Golfo de California y delta del Río Colorado, ubicada en aguas del Golfo de California y los municipios de Mexicali, BC, del Puerto Peñasco y San Luis Río Colorado, Sonora. Diario Oficial de la Federación, Secretaría de Gobernación, México
DOF (2000) Reglamento de la Ley General del Equilibrio Ecológico y la Protección al Ambiente en Materia de Evaluación del Impacto Ambiental. Artículo 2, Capítulo 5, Inciso S. Diario Oficial de la Federación, Secretaría de Gobernación, México
DOF (2005) Programa de protección de la Vaquita dentro de área de Refugio ubicada en la porción occidental del Alto Golfo de California. Diario Oficial de la Federación, Secretaría de Gobernación, México
DOF (2010) Protección ambiental-especies nativas de México flora y fauna silvestres-categorías de riesgo y especificaciones para su inclusión, exclusión o cambio-lista de especies en riesgo. Diario Oficial de la Federación, Secretaría de Medio Ambiente y Recursos Naturales, Mexico
DOF (2012) Acuerdo por el cual se establece una red de zonas de refugio en aguas marinas de jurisdicción federal frente a la costa oriental del Estado de Baja California Sur, en el corredor marino San Cosme a Punta Coyote. Diario Oficial de la Federación, Secretaría de Gobernación, Mexico
DOF (2013) Respuesta a los comentarios efectuados al Proyecto de Modificación a la Norma Oficial Mexicana NOM-002-PESC-1993, Para ordenar el aprovechamiento de las especies de camarón en aguas de jurisdicción federal de los Estados Unidos Mexicanos, publicada el 31 de diciembre de 1993 y sus modificaciones publicadas los días 30 de julio de 1997 y 28 de noviembre de 2006, publicado el 22 de febrero de 2013. Diario Oficial de la Federación, Secretaría de Gobernación, Mexico
Done TJ, Reichelt RE (1998) Integrated coastal zone and fisheries ecosystem management: generic goals and performance indices. Ecol Appl 8:110–118
Downton-Hoffmann CA (2012) Reporte final de actividades perteneciente al permiso de pesca de fomento No. DGOPA.00695.080211.0200. No. PESQ-0612-07, Centro Intercultural de Estudios de Desiertos y Océanos AC. Puerto Peñasco
Downton-Hoffmann CA, Turk-Boyer PJ, Thaxton B et al (2013a) Corredor biológico pesquero Puerto Peñasco-Puerto Lobos, Sonora. Reporte Interno CEDO. Centro Intercultural de Estudios de Desiertos y Océanos AC, Puerto Peñasco
Downton-Hoffmann CA, Loaiza-Villanueva RD, Turk-Boyer PJ et al (2013b) Protocolo de monitoreo biológico-pesquero de peces de importancia comercial en la región del norte del golfo de California. Programa de pesca sustentable. Centro Intercultural de Estudios de Desiertos y Océanos, A.C. Puerto Peñasco
Downton-Hoffmann CA, Martinez-Tovar I, Sánchez-Cruz AY et al (2013c) Protocolo de Monitoreo del Corredor Biológico Puerto Peñasco-Puerto Lobos, Sonora. Programa de Pesca Sustentable. Centro Intercultural de Estudios de Desiertos y Océanos, A.C. Puerto Peñasco
Erisman BE, Buckhorn ML, Hastings PA (2007) Spawning patterns in the leopard grouper, Mycteroperca rosacea, in comparison with other aggregating groupers. Mar Biol 151:1849–1861
Erisman BE, Aburto-Oropeza O, Gonzalez-Abraham C et al (2012) Spatio-temporal dynamics of a fish spawning aggregation and its fishery in the Gulf of California. Sci Rep 2:284. doi:10.1038/srep00284
Espinoza-Romero MJ, Chan KMA, McDaniels T, Dalmer DM (2011) Structuring decision-making for ecosystem-based management. Mar Policy 35:575–583
Essington TE, Beaudreau AH, Wiedenmann J (2006) Fishing through marine food webs. PNAS 103(9):3171–3175
Everett WT, Anderson DE (1991) Status and conservation of the breeding seabirds on offshore Pacific islands of Baja California and the Gulf of California. Seabird status and conservation: a supplement. In: Croxall JP (ed) ICBP Technical Publication No. 11. International Council for Bird Preservation, Cambridge
Ezcurra E, Equihua M, Lopez-Portillo J (1987) The desert vegetation of El-Pinacate, Sonora, Mexico. Vegetatio 71:49–60
Ezcurra E, Felger RS, Russell A, Equihua M (1988) Freshwater islands in a desert sand sea: the hydrology, flora and phytogeography of the Gran Desierto oases of Northwestern Mexico. Desert Plant 9:35–44
Felger RS (2000) Flora of the Gran Desierto and Rio Colorado of Northwestern Mexico. University of Arizona Press, Tucson
Flanagan CA, Hendrickson JR (1976) Observations on the commercial fishery and reproductive biology of the totoaba, Cynosaon macdonaldi, in the Northern Gulf of California. Fish Bull 74:531–544
Fujita R, Lynham J, Micheli F et al (2012) Ecomarkets for conservation and sustainable development in the coastal zone. Biol Rev Camb Philos Soc 88:273–286. doi:10.1111/j.1469-185X.2012.00251.x
Gaines SD, Gaylord B, Largier JL (2003) Avoiding current oversights in marine reserve design. Ecol Appl 13:32–46. doi:10.1890/1051-0761 013[0032:ACOIMR]2.0.CO;2
Galindo-Bect M, Glenn EP, Page HM et al (2000) Penaeid shrimp landings in the upper Gulf of California in relation to Colorado river freshwater discharge. Fish Bull 98(1):222–225
Gallagher MB, Heppell SS (2010) Essential habitat identification for age-0 rockfish along the central Oregon coast. Mar Coast Fish 2:60–72. doi:10.1577/C09-032.1
García-Caudillo JM, Gómez-Palafóx JV (2005) La pesca industrial de camarón en el golfo de California: Situación Económico-Financiera e Impactos Socio-ambientales Conservación Internacional México. AC, Mexico
Glenn EP, Zamora-Arroyo F, Nagler PL et al (2001) Ecology and conservation biology of the Colorado river delta, Mexico. J Arid Environ 49:5–15
Glenn EP, Nagler PL, Brusca RC, Hinojosa-Huerta O (2006) Coastal wetlands of the Northern Gulf of California: inventory and conservation status. Aquat Conserv 16:5–28
Guido S (2006) Red de monitoreo de los desarrollos turísticos del Noroeste costero. ALCOSTA-CIAD, Mazatlan
Gutiérrez NL, Hilborn R, Defeo O (2010) Leadership, social capital and incentives promote successful fisheries. Nature 470(1):386–389. doi:10.1038/nature09689
Hastings PA, Findley LT (2007) Marine fishes of the upper gulf biosphere reserve, Northern Gulf of California. Dry borders: great natural areas of the Gran Desierto and upper Gulf of California. University of Utah Press, Salt Lake City
Hastings P, Findley L, Van Der Heiden AM (2010) Fishes of the Gulf of California. In: Brusca R (ed) The Gulf of California: biodiversity and conservation. University of Arizona and The Arizona-Sonora Desert Museum, Tucson, pp 96–118
Hernández-Velasco A (2010) Efecto de la pesca artesanal en la estructura comunitaria de peces de arrecife rocosa del Noreste del Golfo de California. Undergraduate Thesis, Universidad Autónoma de Baja California Sur, La Paz
Higuera-Ciapara I, Mayorga-Ríos A (2006) Investigación científica y tecnológica: Conacyt. In: Guzmán PG, Fuentes DF (eds) Pesca, acuacultura e investigación en México, Comisión de Pesca, pp 273–284
Hilborn R (2011) Future directions in ecosystem based fisheries management: a personal perspective. Fish Res 108:235–239
Hinojosa-Huerta O, Carillo-Guerrero Y, DeStefano S et al (2004a) Waterbird communities and associated wetlands of the Colorado river delta, Mexico. Stud Avian Biol 27:52–69
Hinojosa-Huerta O, Iturribarría-Rojas H, Carrillo-Guerrero Y et al (2004b) Bird conservation plan for the Colorado river delta. Pronatura Noroeste, San Luis Río Colorado
Hinojosa-Huerta O, Garcia-Hernandez J, Carillo-Guerrero Y, Zamora-Hernandez E (2007) Hovering over the Alto Golfo: the status and conservation of birds from the Rio Colorado to the Gran Desierto. In: Felger RS, Broyles B (eds) Dry borders: great natural reserves of the Sonoran desert. The University of Utah Press, Salt Lake City, pp 383–407
Houde ED, Rutherford ES (1993) Recent trends in estuarine fisheries: predictions of fish production and yield. Estuaries 16(2):161–176
Huang D, Bernardi G (2001) Disjunct sea of cortez-pacific ocean Gillichthys mirabilis populations and the evolutionary origin of their sea of Cortez endemic relative, Gillichthys seta. Mar Biol 138:421–428
INAPESCA (2011) Evaluación de un banco de callo de árbol, El Desemboque, Sonora. Instituto Nacional de Pesca, Guaymas
INEGI (2011) Información de México para niños. http://cuentame.inegi.org.mx/monografias/informacion/son/poblacion. Accessed 4 Feb 2013
Iris-Maldonado CA (2011) Estructura de la comunidad de peces de dos esteros en el norte del Golfo de California. Dissertation, Centro de Investigación Científica y de Educación Superior de Ensenada
Johnson AF (1982) Dune vegetation along the eastern shore of the Gulf of California. J Biogeogr 9:317–330
Jones PJS, Qiu W, De Santo EM (2011) Governing marine protected areas—getting the balance right. United Nations Environment Program, Nairobi
Keen AM (1971) Sea shells of tropical west America: marine mollusks from Baja California to Peru. Stanford University Press, California
Kellner JB, Tetreault I, Gaines SD, Nisbet RM (2007) Fishing the line near marine reserves in single and multispecies fisheries. Ecol Appl 17(4):1039–1054
Kininmonth S, Beger M, Bode M et al (2011) Dispersal connectivity and reserve selection for marine conservation. Ecol Model 222:1272–1282
Koen-Alonso M (2009) Ecosystem approach to fisheries. In: Beamish RJ, Rothschild BJ (eds) The future of fisheries science in North America. Fish Fish Ser 31:185–207
Lavín MF, Sánchez S (1999) On how the Colorado river affected the hydrography of the upper Gulf of California. Cont Shelf Res 19:1545–1560
Lercari-Bernier D, Arreguín-Sánchez F (2009) An ecosystem modeling approach to deriving viable harvest strategies for multispecies management of the Northern Gulf of California. Aquat Conserv Mar Fresh Ecosyst 19:384–397
Levin PS, Stunz GW (2005) Habitat triage for exploited fishes: can we identify essential “Essential Fish Habitat?” Estuar Coast Shelf Sci 64:70–78. doi:10.1016/j.ecss.2005.02.007
Lindeman KC, Pugliese R, Waugh GT, Ault JS (2000) Developmental patterns within a multispecies reef fishery: management applications for essential fish habitats and protected areas. Bull Mar Sci 66:929–956
Lluch-Cota SE, Aragón-Noriega EA, Arreguín-Sánchez F et al (2007) The Gulf of California: review of ecosystem status and sustainability challenges. Prog Oceanogr 73:1–26
Loaiza-Villanueva R, Downton-Hoffmann CA (2011) Informe de monitoreo poblacional de Jaiba Callinectes spp. en la Costa Norte de Sonora en el Corredor Bahía Adhair-Desemboque de Caborca 2009–2011, Región Puerto Peñasco, Sonora. Centro Intercultural de Estudios de Desiertos y Océanos, A.C. Puerto Peñasco
Loaiza-Villanueva R, Castañeda-Fernández de Lara V, Pérez-Valencia S (2009) Monitoreo poblacional de jaiba Callinectes spp. en la costa norte de Sonora en el corredor Bahía Adair-Desemboque de Caborca, 2008–2009. Centro de Estudios de Desiertos y Océanos, Puerto Peñasco
Loaiza-Villanueva R, Downton-Hoffmann CA, Martínez-Tovar I et al (2012) Recomendaciones para el manejo regional de las especies de caracol chino ( Hexaplex nigritus y Phyllonotus ( Hexaplex) erythrostomus en el Norte del Golfo de California, región Puerto Peñasco, Sonora. Informe. Centro Intercultural de Estudios de Desiertos y Océanos AC. Puerto Peñasco
Lowe WH, Allendorf FW (2010) What can genetics tell us about population connectivity? Mol Ecol 19:3038–3051. doi:10.1111/j.1365-294X.2010.04688.x
Maluf LY (1988) Composition and distribution of the Central Eastern Pacific echinoderms. Nat Hist Mus Los Angeles County. Tech Rep 2:1–242
Marasco RJ, Goodman D, Grimes CB et al (2007) Ecosystem-based fisheries management: some practical suggestions. Can J Fish Aquat Sci 64:928–939
Marinone SG (2003) A three-dimensional model of the mean and seasonal circulation of the Gulf of California. J Geophys Res 108(C10). doi:10.1029/2002JC001720
Marinone SG (2012) Seasonal surface connectivity in the Gulf of California. Estuar Coast Shelf Sci 100:133–141
Marinone SG, Ulloa MJ, Parés-Sierra A et al (2008) Connectivity in the Northern Gulf of California from particle tracking in a three-dimensional numerical model. J Mar Syst 71:149–158. doi:10.1016/j.marsys.2007.06.005
Martinez I (2010) Monitoreo participativo de especies de importancia ecológica y comercial en los arrecifes rocosos de la RB Alto Golfo de California y Delta del Río Colorado y su zona de influencia en Sonora. Centro Intercultural de Estudios de Desiertos y Océanos AC, Puerto Peñasco
Martinez I, Turk-Boyer P (2011) Actualización del Plan de Manejo de callo escarlopa ( Spondylus calcifer) en el Golfo de California. Centro Intercultural de Estudios de Desiertos y Océanos, A.C. Puerto Peñasco
Martínez I, Turk-Boyer P, Sanchez A, Downton-Hoffmann CA (2011) Corredor Biológico-Pesquero Peñasco, Punta Borrascoso a Puerto Lobos, Sonora, Norte del Golfo de California. Una Propuesta de Ordenamiento Pesquero de la Pesca Ribereña. Proyecto PANGAS. Centro Intercultural de Estudios de Desiertos. Puerto Peñasco
McLeod KL, Lubchenco J, Palumbi SR, Rosenberg AA (2005) Scientific consensus statement on marine ecosystem-based management. Communication Partnership for Science and the Sea, Corvallis
Mellink BE, Romero-Saavedra AL (2005) Dieta del lobo marino de California, Zalophus californianus Lesson, 1828, en la Isla San Jorge en el norte del Golfo de California, México, 1998–1999. Cien Mar 31(2):369–377
Meltzer L, Blinick NS, Fleishman AB (2012) Management implications of the biodiversity and socio-economic impacts of shrimp trawler by-catch in Bahía de Kino, Sonora, México. PLoS One 7(6):e35609. doi:10.1371/journal.pone.0035609
Metaxas A, Saunders M (2009) Quantifying the “bio” components in biophysical models of larval transport in marine benthic invertebrates: advances and pitfalls. Biol Bull 216:257–272
Moreno-Báez M (2010) Mapping human dimensions of small-scale fisheries in the Northern Gulf of California, Mexico. Ph.D Thesis, School of Natural Resources and the Environment, University of Arizona, Tucson
Moreno-Báez M, Cudney-Bueno R, Orr B et al (2012) Integrating the spatial and temporal dimensions of fishing activities for management in the Northern Gulf of California, Mexico. Ocean Coast Manage 55:111–127. doi:10.1016/j.ocecoaman. 2011.10.001
Morzaria-Luna H, Iris-Maldonado A, Valdivia-Jiménez P (2010) Physico-chemical characteristics of negative estuaries in the Northern Gulf of California, Mexico. In: Crane JR, Solomon AE (eds.) Estuaries: Types, movement patterns and climatical impacts. Nova Science Publishers Inc. pp 201–223. ISBN: 978-1-60876-859-2
Morzaria-Luna H, Castillo-López A, Danemann GD, Turk-Boyer P (2013) Conservation strategies for coastal wetlands in the Gulf of California, Mexico Wetlands Ecol Manage doi: 10.1007/s11273-013-9328-0
Morzaria-Luna H, Turk-Boyer P, Moreno-Baez M (2013) Social indicators of vulnerability for fishing communities in the Northern Gul fof California, Mexico: Implications for climate change. Mar Policy (2013), http://dx.doi.org/10.1016/j.marpol.2013.10.013i
Munro-Palacio G (2007) Breve historia de Puerto Peñasco. De Cierto Mar Editores, Puerto Peñasco
U.S. National Marine Fisheries Service (1999) Ecosystem-based fisheries management: a report to Congress by the Ecosystem Principles Advisory Panel (online). United States Department of Commerce, National Oceanic and Atmosphereic Administration, National Marine Fisheries service, Silver Spring, MD. www.st.nmfs.gov/st7/document/epap_report.pdf. Accessed 15 Jan 2006
OEIDRUS (2011) Oficina Estatal de Información del Desarrollo Rural Sustentable del Estado de Sonora. http://www.oeidrus-sonora.gob.mx/. Accessed 26 March 2013
Palacios E, Mellink E (1996) Status of the least tern in the Gulf of California. J Field Ornithol 67:48–58
PANGAS (2012a) Callo de Escarlopa Spondylus limbatus. Ficha Informativa de la Pesca Ribereña del Norte del Golfo de California. Centro Intercultural de Estudios de Desiertos y Océanos, A.C. Puerto Peñasco
PANGAS (2012b) Caracol chino (Familia Muricidae). Ficha Informativa de la Pesca Ribereña del Norte del Golfo de California. Centro Intercultural de Estudios de Desiertos y Océanos, A.C. Puerto Peñasco
PANGAS (2012c) Pulpo Octopus bimaculatus. Ficha Informativa de la Pesca Ribereña del Norte del Golfo de California. Centro Intercultural de Estudios de Desiertos y Océanos, A.C. Puerto Peñasco
PANGAS (2012d) Jaiba Callinectes bellicosus. Ficha Informativa de la Pesca Ribereña del Norte del Golfo de California. Centro Intercultural de Estudios de Desiertos y Océanos, A.C. Puerto Peñasco
PANGAS (2012e) Baqueta Hyporthodus acanthistius. Ficha Informativa de la Pesca Ribereña del Norte del Golfo de California. Centro Intercultural de Estudios de Desiertos y Océanos, A.C. Puerto Peñasco
Pauly D, Christensen V, Dalsgaard J et al (1998) Fishing down marine food webs. Science 279:860–863. www.sciencemag.org
Pauly D, Christensen V, Walters C (2000) Ecopath, ecosim, and ecospace as tools for evaluating ecosystem impact of fisheries. ICES J Mar Sci 57:697–706
Pauly D, Christensen V, Guenette S et al (2002) Towards sustainability in world fisheries. Nature 418:689–695
Peguero-Icaza M, Sánchez-Velasco L, Lavín MF, Marinone SG (2008) Larval fish assemblages, environment and circulation in a semienclosed sea (Gulf of California, Mexico). Estuar Coast Shelf Sci 79:277–288. doi:10.1016/j.ecss.2008.04.008
Peguero-Icaza M, Sánchez-Velasco L, Lavín MF et al (2011) Seasonal changes in connectivity routes among larval fish assemblages in a semi-enclosed sea (Gulf of California, Mexico). J Plankton Res 33:517–533. doi:10.1093/plankt/fbq107
Pérez-Valencia SA, Aragón–Noriega EA (2012) Determinación de biomasa y bancos de la almeja de sifón Panopea globosa (Dall 1898) en una zona virgen del Alto Golfo de California. Interciencia 37(9):651–656
Pérez-Valencia S, Loaiza-Villanueva RD, Sánchez-Cruz AY (2007) Pesca Deportiva-Recreativa en Puerto Peñasco, Sonora; México. Informe Interno. Centro Intercultural de estudios de Desiertos y Océanos AC, Puerto Peñasco
Pérez-Valencia SA, Gorostieta-Monjaraz M, Castañeda-Fernández de Lara V et al (2012) MIA-R para la Pesca Ribereña Responsable en la Reserva de la Biosfera Alto Golfo de California y Delta del Río Colorado: Costa Oeste. Centro Intercultural de Estudios de Desiertos y Océanos, A.C. Puerto Peñasco
Pérez-Valencia SA, Polanco-Mizquez EI, Rodríguez-Uceda A et al (2013a) Programa de monitoreo abordo: manifestación de impacto ambiental regional para la pesca ribereña responsable en el Alto Golfo de California y Delta del Río Colorado, V.I. Centro Intercultural de Estudios de Desiertos y Océanos, A.C., Puerto Peñasco
Pérez-Valencia SA, Polanco-Mizquez EI, Rodríguez-Uceda A et al (2013b) Programa de monitoreo pesquero: manifestación de impacto ambiental regional para la pesca ribereña responsable en el Alto Golfo de California y Delta del Río Colorado, V.I. Centro Intercultural de Estudios de Desiertos y Océanos, A.C., Puerto Peñasco
Pikitch EK, Santora C, Babcock EA et al (2004) Ecology: ecosystem-based fishery management. Science 305:346–347
Polis GA, Holt RD, Menge BA, Winemiller K (1996) Time, space, and life history: Influences on food webs. 435–460 In: Polis GA, Winemiller K (eds) Food webs: integration of patterns and dynamics. Chapman and Hall, New York.
Polis GA, Anderson WB, Holt RD (1997) Toward an integration of landscape and food web ecology: the dynamics of spatially subsidized food webs. Annu Rev Ecol Syst 28:289–316
Rodríguez CA, Flessa KW, Dettman D (2001) Effects of upstream diversion of Colorado river water on the estuarine bivalve mollusc Mulinia coloradoensis. Conserv Biol 15:249–258
Rodríguez-Quiroz G, Aragón-Noriega A, Valenzuela-Quiñónez W, Esparza-Leal HM (2010) Pesca artesanal en las zonas de conservación del Alto Golfo de California. Rev Biol Mar y Oceanogr 45:89–98. doi:10.4067/S0718-19572010000100008
Rodríguez-Ramírez R (2010) Política ambiental, conservación y desarrollo regional sustentable: Una aproximación a través de los “Buybacks” internacionales y el Pace Vaquita en la región del Alto Golfo de California. MSc Thesis, Centro de Investigación en Alimentación y Desarrollo A.C., Hermosillo
Rogers SI, Clarke RK, Reynolds JD (1999) The taxonomic distinctness of coastal bottom-dwelling fish communities of the North-east Atlantic. J Anim Ecol 68:769–782
Rojas-Bracho L, Reeves RR, Jaramillo-Legoretta A (2006) Conservation of the vaquita Phocoena sinus. Mammal Rev 36:179–216
Rose MD, Polis GA (1998) The distribution and abundance of coyotes: the effects of allochthonous food subsidies from the sea. Ecology 79:998–1007
Rosemartin A (2008) Biological dimensions of tern management: a case study of the least tern in Sonora, Mexico and a comparative analysis of reproductive investments in terns. MSc Thesis, School of Natural Resources, University of Arizona, Tucson
Rountree R, Able K (2007) Spatial and temporal habitat use patterns for salt marsh nekton: implications for ecological functions. Aquat Ecol 41:25–45. doi:10.1007/s10452-006-9052-4
Rowell K, Flessa KW, Dettman DL et al (2008) Diverting the Colorado river leads to a dramatic life history shift in an endangered marine fish. Biol Conserv 141(4):1140–1150
Rupnow J (2008) Recreational fisheries and the ecology and management of targeted resources in the Northern Gulf of California, Mexico. MSc Thesis, Arizona State University, Tucson
Sáenz-Arroyo A, Roberts CM, Torre J, Carino-Olvera M (2005) Using list naturalists’ observations and grey literature to reassess marine species at the Gulf grouper in the Gulf of California, Mexico. Fish Fish 6:121–133
Sánchez-Velasco L, Lavín MF, Jiménez-Rosenberg SPA et al (2012) Larval fish habitats and hydrography in the biosphere reserve of the upper Gulf of California. Cont Shelf Res 33:89–99. doi:10.1016/j.csr.2011.11.009
SEMARNAT (2012) Resolutivos emitidos en el PEIA en la DGIRA. Proyecto “Home Port Turístico Puerto Peñasco” en la Gaceta Ecológica Año X Separata No. DGIRA/055/12 del 21 de noviembre de 2012. SEMARNAT/DGIRA, Mexico
Seminoff JA, Nichols WJ (2007) Sea turtles of the Alto Golfo. A struggle for survival. In: Felger RS, Broyles B (eds) Dry borders: great natural reserves of the Sonoran desert. University of Utah Press, Salt Lake City, pp 505–518
Shreve F, Wiggins IL (1964) Vegetation and flora of the Sonoran desert. Stanford University Press, Stanford
Solís-Marín FA, Laguarda-Figueras A, Durán-González A et al (2005) Equinodermos (Echinodermata) del Golfo de California, México. Rev Biol Trop 53(3):123–137
Soria G (2010) Connectivity of marine bivalve species in the Northern Gulf of California: implications for fisheries management and conservation. PhD Thesis, University of Arizona, Tucson
Soria G, Munguía-Vega A, Marinone SG et al (2012) Linking bio-oceanography and population genetics to assess larval connectivity. Mar Ecol Prog Ser 463:159–175
Soria G, Torre-Cosio J, Munguia-Vega A, Marinone SG, Lavín MF, Cinti A, Moreno-Báez M (2014) Dynamic connectivity patterns from an insular marine protected area in the Gulf of California. Journal of Marine Systems 129:248–258
Spackeen J (2009) Analysis of food web dynamics in the Northern Gulf of California using stable isotopes. Undergraduate degree, University of Miami, Miami
Stapp P, Polis GA (2003) Influence of pulsed resources and marine subsidies on insular rodent populations. Oikos 102:111–123
Steller DL, Riosmena-Rodriguez R, Foster MS, Roberts CA (2003) Rhodolith bed diversity in the Gulf of California: the importance of rhodolith structure and consequences of disturbance. Aquat Conserv Mar Fresh Ecosyst 13:S5–S20
Szteren D, Aurioles-Gamboa D, Gerber LR (2006) Population status and trends of the California sea lion (Zalophus californianus californianus) in the Gulf of California, Mexico. In: Trites A, Atkinson S, DeMaster D et al (eds) Sea lions of the world. Alaska Sea Grant College Program/University of Alaska Fairbanks, Fairbanks, pp 369–384
Talley DM, Huxel GR, Holyoak M (2006) Habitat connectivity at the land-water interface. In: Crooks KR, Sanjayan MA (eds) Connectivity conservation. Cambridge University Press, Cambridge, pp 97–129
Thomson DA, Gilligan M (1983) The rocky shore fishes. In: Case TJ, Cody ML (eds) Island biogeography in the sea of Cortez. University of California Press, Berkeley, pp 98–129
Thomson DA, Findley LT, Kerstitch AN (2000) Reef fishes of the sea of Cortez: the rocky shore fishes of the Gulf of California. University of Texas, Austin
Turk-Boyer PJ (1998) Estero Morua: the formation of an estuary. Centro Intercultural de Estudios de Desiertos y Océanos, A.C. Puerto Peñasco
Turk-Boyer PJ (2008) Integrating science and community participation: conservation and management of coastal wetlands in the Northern Gulf of California. Centro Intercultural de Estudios de Desiertos y Océanos, A.C. Puerto Peñasco
Turk-Boyer PJ, Barrera JC (2012) Indicadores y mecanismos para la evaluación de la efectividad del programa de protección de la vaquita dentro del área de refugio ubicada en la porción occidental del Alto Golfo de California. Centro Intercultural de Estudios de Desiertos y Océanos, A.C. Puerto Peñasco
Turk Boyer PJ, Castañeda-Fernández de Lara V, Morzaria-Luna HN et al (2009) Sustainable fisheries in the upper Gulf of California: characterization of fisheries and essential fish habitats, and community management interests. Final Grant Report 06-0357. Centro Intercultural de Estudios de Desiertos y Océanos, A.C. Puerto Peñasco
Turner R, Brown D (1994) Sonoran desertscrub. In: Brown D (ed) Biotic communities: Southwestern United States and Northwestern Mexico. University of Utah Press, Salt Lake City, pp 181–221
Urban J (2010) Marine mammals of the Gulf of California: an overview of diversity and conservation status. In: Brusca R (ed) The Gulf of California: biodiversity and conservation. University of Arizona and The Arizona-Sonora Desert Museum, Tucson, pp 188–209
Valavanis VD, Georgakarakos S, Kapantagakis A et al (2004) A GIS environmental modeling approach to essential fish habitat designation. Ecol Model 178:417–427. doi:10.1016/j.ecolmodel.2004.02.015
Van Andel TH (1964) Recent marine sediments of the Gulf of California. In: van Andel TJ, Shor GG (eds) Marine geology of the Gulf of California: a symposium. Am Assoc Pet Geol Mem 3:216–310
Vega X, González MA, Muñoz del Viejo A (2006) Potential new Ramsar sites in northwest Mexico: strategic importance for migratory waterbirds and threats to conservation. In: Boere GC, Galbraith CA, Stroud DA (eds) Waterbirds around the world. The Stationery Office, Edinburgh, pp 158–160
Velarde E, Anderson DW (1994) Conservation and management of seabird islands in the Gulf of California: setbacks and successes. In: Nettleship DN, Burger J, Gochfeld M (eds) Seabirds on islands: threats, case studies and action plans. Birdlife International, Cambridge, pp 229–243
Velarde E, Ezcurra E, Cisneros-Mata MA, Lavin MF (2004) Seabird ecology, El Niño anomalies, and prediction of sardine fisheries in the Gulf of California. Ecol Appl 14:607–615
Weinstein MP, Litvin SY, Bosley KL et al (2000) The role of tidal salt marsh as an energy source for marine transient and resident finfishes: a stable isotope approach. T Am Fish Soc 129:797–810. doi:10.1577/1548-8659(2000) 129<0797:TROTSM>2.3.CO;2
Zuria I, Mellink E (2005) Fish abundance and the 1995 nesting season of the least tern at Bahía San Jorge, Northern Gulf of California, Mexico. Waterbirds 28:172–180
Acknowledgments
This project would not have been possible without the vision and financial support of the David and Lucile Packard Foundation’s ecosystem-based management initiative. Too many researchers, partners and co-workers have been involved to give mention to all, but staff members that have been key to conducting this work and deserve special thanks for their dedication to different components of this long-term process, include Dr. Sergio Perez-Valencia, Rene Loaiza-Villanueva, Elia Polanco, Angeles Yazmin Sánchez Cruz, Ema Elizabeth González Plascencia, Marlene Anaid Luquin Covarrubias, David Buitrago, Verónica Castañeda, Paloma Valdivia, Hitandehui Tovar Vázquez and Alejandro Castillo. Most importantly and significantly we give thanks to the fishers of the Peñasco Corridor, who have openly shared their lives, their fisheries and aspirations to move towards creating a more functional fisheries system.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media Dordrecht
About this chapter
Cite this chapter
Turk-Boyer, P., Morzaria-Luna, H., Martinez-Tovar, I., Downton-Hoffmann, C., Munguia-Vega, A. (2014). Ecosystem-Based Fisheries Management of a Biological Corridor Along the Northern Sonora Coastline (NE Gulf of California). In: Amezcua, F., Bellgraph, B. (eds) Fisheries Management of Mexican and Central American Estuaries. Estuaries of the World. Springer, Dordrecht. https://doi.org/10.1007/978-94-017-8917-2_9
Download citation
DOI: https://doi.org/10.1007/978-94-017-8917-2_9
Published:
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-017-8916-5
Online ISBN: 978-94-017-8917-2
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)