Abstract
The main mechanisms of nitrogen (N) deposition impacts on terrestrial biodiversity, mainly from studies in Europe, are identified as: direct foliar impacts; eutrophication; acidification; negative effects of reduced N; and increased susceptibility to secondary stress and disturbance factors such as drought, frost, pathogens or herbivores. The relation of several of these mechanisms to aquatic ecosystems is also described, as is the relative lack of N impact studies on faunal species/communities compared to floral ones. The factors that moderate N impacts on ecosystems are also considered and are categorized as: (1) the duration and total amount of the N inputs; (2) the chemical and physical form of the airborne N input; (3) the intrinsic sensitivity to the changes in N availability of the plant and animal species present; (4) the abiotic conditions (such as the ability of soils and waters to neutralize acidification effects); and (5) the past and present land use or management. The increased susceptibility of plants (or animal) species to stresses and disturbances, induced by enhanced atmospheric N loads, is highly dependent of the large differences in the physiological functioning of individual species. Therefore, the generalization of the effects of N deposition over a range of ecosystems is hardly, if at all, possible, although these impacts have been demonstrated to be of major importance in some ecosystems.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
The emissions of ammonia (NH3) and nitrogen oxides (NOx) strongly increased in the second half of the twentieth century (e.g., Sutton et al. 2008) . Ammonia is volatilized from agricultural systems, such as dairy farming and intensive animal husbandry, whereas NOx originates mainly from burning of fossil fuel by traffic, the power generation sector and industry. Because of short- and long-range transport of these nitrogenous compounds, atmospheric nitrogen (N) deposition has clearly increased in many natural and semi-natural ecosystems across the world. Areas with high atmospheric N deposition are nowadays found in central and western Europe, eastern USA and, since the 1990s, Eastern Asia (e.g. Dentener et al. 2006). For more details on N deposition see Chap. 2 (Dentener et al. 2014; this volume). In this background paper we firstly give a short overview of the “mechanisms” which lead to change in species performance, composition and diversity after N enrichment. Secondly, factors that affect the extent or severity of the N deposition impacts on biodiversity are identified . Sensitivities of different major ecosystems with respect to biodiversity effects and their effect thresholds, will not treated in this paper, but for details on this topic, see Bobbink et al. (2010). Finally, we present a list of major questions, which have to be solved in the future.
2 Impacts on Plant Biodiversity
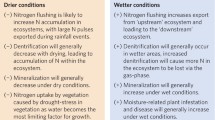
The series of events which occurs when N inputs increase in a region with originally low background N deposition rates is highly complex. Many ecological processes interact and operate at different temporal and spatial scales. Despite this highly diverse sequence of events, the following main impact “categories” can be recognised. A schematic overview of the possible sequence of events is given in Fig. 14.1.
Schematic of the main impacts of enhanced N deposition on ecosystems as demonstrated by studies on temperate ecosystems in Europe and North America. Stress is considered to occur when external constraints limit the rate of dry matter production of the vegetation, whereas disturbance consists of mechanisms which affect plant biomass by causing its partial or total destruction
2.1 Direct Foliar Impacts
An important effect of N gasses, aerosols and dissolved compounds can be direct toxicity to the above-ground parts of individual plants . Nitrogen dioxide (NO2), NH3 and ammonium (NH4) in particular are phytotoxic . The impacts have been mostly studied for crops, and young trees (saplings), but studies with native herbaceous or dwarf-shrub species in open top chambers (OTCs) have also demonstrated leaf injury, changes in physiology and reductions in growth at (very) high air concentrations of airborne N pollutants (e.g. Pearson and Stewart 1993; Krupa 2003). These impacts were observed in parts of Europe and Northern America in 1980s but are nowadays very rare in these regions because of pollutant control measures. However, increasing air N pollutant concentrations are now found in Asia (China and India), potentially causing direct foliar impacts . In addition, lichens are clearly the most sensitive organisms with respect to direct toxicity of dry deposited NH3 (Van Herk et al. 2003) whereas direct toxic effects of wet deposited N have been reported for bryophytes and lichens at rather low deposition rates (Bates 2002) .
2.2 Eutrophication
Nitrogen is the limiting nutrient for plant growth in many natural and semi-natural terrestrial ecosystems, especially of oligotrophic and mesotrophic conditions. Research in the temperate ecosystems of Europe and North America has shown that enhanced N deposition results in an increase in the availability of inorganic N in the topsoil in the short-term. This gradually leads to an increase in plant productivity in N-limited vegetation and thus to higher annual litter production. Because of this, N mineralisation will gradually increase, which may cause an extra increase in plant productivity. This is a positive feedback, because higher N mineralisation gives higher N uptake, etc. Above a certain level of primary productivity, local species diversity declines as production increases. Observational studies across N deposition gradients and many N-addition experiments demonstrated this effect in the longer-term. Competitive exclusion (“overshading”) of characteristic species of oligotrophic or mesotrophic habitats by relatively fast-growing nitrophilic species is to be expected and especially rare species with low abundances or a low stature are at risk (e.g. Bobbink et al. 1998, 2003; Suding et al. 2005). The rate of N cycling in the ecosystem is clearly enhanced in this situation, although the response time to enhanced N inputs can be long in this respect in highly organic soils with a high C:N ratio, or, perhaps in any soil with large potential N sinks. When the N deficiencies in the ecosystem are no longer limiting, plant growth becomes restricted by other resources, such as phosphorus (P) or water. In this situation, the productivity of the vegetation will not increase further. Nitrogen concentrations in the plants will, however, tend to increase because N availability still increases, which may affect the palatability of the vegetation for herbivores or the sensitivity to pathogens (see later). In addition, it is to be expected that the shift from N to P limitation and the abnormal ratio between them, high N and low P, will gradually lead to changes in plant species composition.
Productivity of aquatic ecosystems is generally considered to be limited by phosphorus. However, there is clear evidence that N is also a very important limiting factor in (sub)alpine lakes, high-latitude lakes, (shallow) soft water bodies and shallow coastal seas. In these situations, N enrichment in originally pristine areas can lead to significant changes in algal communities, or even algal blooms . This may also lead to related foodweb-based shifts in these aquatic systems. In shallow N-limited lakes, macrophyte composition can also be highly affected by eutrophication via N inputs (e.g. Roelofs 1983) .
2.3 Acidification
Soil or water acidification is characterized by a wide variety of long-term effects. It is defined as the loss of buffering capacity (Acid Neutralizing Capacity (ANC) or alkalinity in water) and may lead to a decrease in pH. Decreases in pH are dependent on the buffering capacity of the soil or water layer (e.g. Ulrich 1983, 1991) .
Acidifying inputs (as N and sulphur (S)), deposited on calcareous soils, will at first not give a change in acidity. In these soils HCO3 − and Ca2 + ions leach from the system, but the pH remains the same until almost all calcium carbonate has been depleted. In soils dominated by silicate minerals (pH 6.5–4.5) buffering is carried out by cation exchange processes of the soil adsorption complexes. In this situation, protons are exchanged with Ca2 + and Mg2 +, and these cations are leached from the soil together with anions (mostly nitrate or sulphate). Because of the restricted capacity of this buffering system, soil pH will soon start to decrease. In mineral soils with a large cation exchange capacity and high base saturation, this buffering may, however, hold for several decades, even at relatively high acidic inputs . At low pH (< 5.0), clay minerals are broken down and hydrous oxides of several metals are dissolved. This causes a strong increase of the concentration of toxic Al3 + and other metals in the soil solution. As a consequence of the decrease in pH, nitrification is strongly hampered or even completely absent in most soils. This may lead to accumulation of ammonium, whereas nitrate decreases to almost zero at these low pH values (e.g. Roelofs et al. 1985). In addition, the decomposition rate of organic material in the soil is lower in these acidified soils, which leads to increased accumulation of litter (e.g. Van Breemen et al. 1982; Ulrich 1983, 1991). As a result of this complex of changes, growth of plant species and the species composition of the vegetation can be seriously affected; acid-resistant plant species will gradually become dominant, and several species typical of intermediate and higher pHs disappear.
The buffering of the water layer in aquatic systems is highly dependent on the bicarbonate concentrations in the water. Input of acidity (protons) will change the carbon dioxide-bicarbonate equilibrium, finally resulting in lowering of the alkalinity and the pH in the water layer. Aquatic systems with low bicarbonate concentrations or low inputs of bicarbonate from the catchment, as in many softwater lakes, are among the most sensitive with respect to acidification of all ecosystems across the globe .
2.4 Negative Effect of Reduced Nitrogen
In many regions with a relatively high rate of N deposition, a (very) high proportion of the deposited N originates from ammonia and ammonium (e.g. Asman et al. 1998; Fowler 2002). This may cause a change in the dominant N form in the soil from nitrate to ammonium, especially in habitats with low nitrification rates (pH < 4.5). The response of sensitive plant species can be significantly affected by this change. Species of calcareous or somewhat acidic soils are able to use nitrate, or a combination of nitrate and ammonium, as the N source, whereas early studies showed that species of acidic habitats generally use ammonium (e.g. Gigon and Rorison 1972; Kinzel 1982), because at least some of these plants do not have the nitrate reductase enzyme (Ellenberg 1996). Laboratory and field studies demonstrated that most forest understory species are favoured when both ammonium and nitrate can be taken up (Falkengren-Grerup 1998; Olsson and Falkengren-Grerup 2000). One of the impacts of increased ammonium uptake is the reduced uptake of base cations and exchange of these cations (K +, Ca2 + and Mg2 +) to the rhizospere. Ultimately this can lead to severe nutritional imbalances, which are considered to be important in the decline in tree growth in areas with high ammonia/ammonium deposition (Nihlgård 1985; Van Dijk et al. 1990). High ammonium concentrations in the soil or water layer are also toxic to many sensitive plant species, causing disturbed cell physiology, cell acidification, accumulation of N-rich amino acids, very poor root development, and finally, inhibition of shoot growth. Strong evidence exists that several endangered vascular plants of grasslands and heathlands, and fen bryophytes are (very) intolerant to increased concentrations and high NH4 +/NO3 − ratios (De Graaf et al. 1998; Paulissen et al. 2004; Kleijn et al. 2008; Van den Berg et al. 2008). This phenomenon is also clearly demonstrated for macrophyte communities of soft-water lakes: increased high NH4 +/NO3 − ratios clearly inhibited the growth of the typical macrophytes of this system, but stimulated the growth of some resistant species, leading to strong monocultures of species .
2.5 Increased Susceptibility to Secondary Stress and Disturbance Factors such as Drought, Frost, Pathogens or Herbivores
The sensitivity of plants to stress, i.e. external constraints that limit dry matter production rate, or disturbance factors, i.e. mechanisms which affect plant biomass by causing its partial or complete destruction, may be significantly affected by N deposition. With increasing N deposition, the susceptibility to fungal pathogens and attacks by insects can be enhanced (e.g. Flückiger et al. 2002) . This is probably due to altered concentrations of phenolic compounds (lower resistance) and soluble N compounds such as free amino acids, together with the lower vitality of individual plants as a consequence of air pollution. Negative impacts of pathogenic fungi have been found in N-addition studies and correlative field studies for several tree species, but for most ecosystems data are lacking and the influence on diversity is still unclear .
In general, herbivory is affected by the palatability of the plant material, which is strongly determined by the N content (Throop 2004). Increased organic N contents of plants, caused by N deposition, can thus result in increased (insect) herbivory. Data on this process are scarce, but it has been demonstrated for attacks by heather beetle in Calluna heathlands in the Netherlands (Brunsting and Heil 1985; Berdowski 1993) . Outbreaks of heather beetle (Lochmaea suturalis), a chrysomelid beetle, can occur in dry lowland heaths. It forages exclusively on the green parts of C. vulgaris. Outbreaks of the beetle lead to the opening of closed C. vulgaris canopy, greatly reducing light interception and leading to enhanced growth of understorey grasses, such as Deschampsia flexuosa or Molinia caerulea. The frequency and intensity of these outbreaks were clearly related with N inputs and N concentrations in the heather, although the exact controlling processes need further quantification.
Furthermore, N-related changes in plant physiology, biomass allocation (root/shoot ratios) and mycorrhizal infection can also differentially influence the sensitivity of plant species to drought or frost stress, leading to reduced growth of some species and possible changes in plant interactions (e.g. Pearson and Stewart 1993; Bobbink et al. 2003) .
3 Impacts of Nitrogen Deposition on Fauna
Until recently, research on the impacts of N deposition has mainly focused on plants/vegetation and abiotic processes . Experimental research on fauna is complicated, as different species use the landscape at different spatial scales and animal species richness is much higher than that of plants. Consequently, research on the effects of increased N inputs on faunal diversity in semi-natural and natural ecosystems is largely lacking. There is however a clear impact as N impacts can affect food and environmental conditions, including micro-climate, but also the vegetation structure and heterogeneity of the landscape, needed by animal species to complete their life-cycles.
Elevated N deposition causes changes in nutrient content of plant organic matter and plant species composition and thereby alters the micro-climate (temperature and moisture regimes) experienced by animals. As an example, increased N deposition has consequences for herbivorous animals like caterpillars, as their host plants may decrease or increase in abundance, or because of changes in food quality. It is likely that for caterpillars the species density declines in N-affected and less diverse vegetation, although direct experimental evidence is scarce (Weiss 1999; Ockinger et al. 2006). However, some caterpillar species may indirectly profit from N deposition if their preferred plant species becomes the dominant species as a consequence of the N inputs. Changes in the nutrient content of dead organic matter also has consequences for detritivores, as shown for aquatic invertebrates (e.g. Smith and Schindler 2009) and micro/mesofauna in forest soils (e.g. Bobbink et al. 2003).
Because of elevated N deposition, vegetation and landscape heterogeneity has also often declined due to e.g. extensive grass encroachment or the development of vegetation with low diversity (see Sect. 14.2, this chapter). The occurrence of animal species is related to landscape heterogeneity by at least three mechanisms. First, species may depend on specific conditions, which are only present in transitions between different biotopes . Second, many animal species require different parts (biotopes) of the landscape for reproduction, resting, foraging, etc. Third, heterogeneity creates the possibility of risk spreading, leading to a higher persistence of species. Thus, N deposition affects faunal diversity not only directly (e.g., changes in food quality and micro-climate), but also indirectly through changes in vegetation and landscape configuration and heterogeneity. To illustrate this, two examples are given of the impacts of N deposition on faunal diversity.
Ground beetle (Carabidae) assemblages on dry open coastal dune grasslands are characterised by species preferring drought and higher temperatures. N deposition, however, results in grass encroachment (e.g. Bobbink et al. 2003). Consequently, the characteristic micro-climate of coastal dune grasslands (very warm during day time, but fairly cold at night and continuously dry) changes to a buffered micro-climate (continuously cool and moist). Comparison of the ground beetle assemblage between 15 coastal dune grasslands on the Waddensea Isles Ameland and Terschelling showed that encroachment with the grasses Calamagrostis epigejos and, to a lesser extent, Ammophila arenaria results in a change in the relative numbers of drought vs. moisture preferring species. Thus, it is likely that because of N inputs the beetle assemblage is dominated by moisture preferring species, instead of the warmth and drought preferring species dominating in intact dune grasslands in low N-input regions (Nijssen et al. 2001).
The decline of the red-backed shrike (Lanius collurio) illustrates how the effects of elevated N deposition can have repercussions across an entire food web (Beusink et al. 2003). This bird species strongly declined from 1950 onwards throughout Western Europe. It has currently disappeared from the coastal dunes of the Netherlands and it is disappearing from the coastal dunes of northern Germany and southern Denmark. Only in the coastal dunes of northern Denmark is the population of red-backed shrikes still stable. This pattern in population trends is clearly correlated to atmospheric N deposition levels, although the occurrence of this bird species can of course not directly be related to higher N availability. Red-backed shrikes feed on large insects and small vertebrates (e.g. lizards) and carry only a single prey item to the nest at a time . Prey demands of the nestlings have to be met during the day under different weather conditions and also during the whole breeding period. To ensure a constant and sufficient energy supply, the red-backed shrikes require a high diversity of large prey species which in turn depends on landscape heterogeneity. In Dutch coastal dunes, increased N deposition led to the encroachment by tall grasses and bushes, a decrease of open sandy areas and a loss of succession stages rich in species. The decline in landscape heterogeneity seriously affected the prey availability for red-backed shrikes. In particular, the lack of sufficient large prey species because of grass encroachment in the areas with high N deposition (Netherlands) has been shown to be the main factor in the decline of the population of this bird species (Esselink et al. 2007) .
4 Factors Affecting the Severity of Nitrogen Deposition Impacts
The severity of the different impacts of atmospheric N deposition (see Sect. 14.2, this chapter) depends on a complex number of factors, of which the most important are: (1) the duration and total amount of the N inputs; (2) the chemical and physical form of the airborne N input; (3) the intrinsic sensitivity to the changes of the plant and animal species present; (4) the abiotic conditions; and (5) the past and present land use or management. Buffer capacity (ANC; alkalinity), original soil nutrient availability (N, P), and soil factors which influence decomposition, nitrification, N immobilisation and denitrification rates, are especially important. As a consequence, high variations in sensitivity to atmospheric N deposition have been observed between different ecosystems across the globe (e.g. Bobbink et al. 1998, 2010).
Direct foliar effects by N pollutants are most strongly influenced by the intrinsic sensitivity of the physiology of the different species (groups) to this stress type. Eutrophication effects of atmospheric N enrichment are strongly related to several processes in the N cycling in ecosystems, such as mineralisation, nitrification, N immobilisation and denitrification rates . These processes can affect the rate of removal from the ecosystem (e.g. leaching of nitrate; N2 or N2O output via denitrification), the form of N (nitrification), but also the availability of N compounds. The original nutrient status of the system is also very important in this respect, whereas other limiting factors, such as P, K or water can highly influence the outcome of all the processes .
The amount of buffering capacity and the buffering rate in terrestrial or aquatic ecosystems are particularly related with their sensitivity to the acidifying consequences of atmospheric N deposition . Nitrification of deposited ammonium can be an important part in this process, because two protons are produced per N molecule in this process. Acidification can also reduce nitrification rates in soils or waters, leading to dominance of reduced N in regions with high reduced N loads and to increased risks of long-term negative effects of this N form. In addition, the effects of reduced N are observed particularly in formerly weakly buffered ecosystems (pH between 4.5 and 6.5), because many characteristic plants (and animals?) are adapted to nitrate as the dominant N form, and high ammonium availability leads to severe toxicity effects .
The increased susceptibility of plants (or animal) species to stresses and disturbances, induced by enhanced atmospheric N loads, is highly dependent on the large differences in the physiological functioning of individual species. Therefore, the generalization of the effects of N deposition over a range of ecosystems is hardly, if at all, possible, although these impacts have been demonstrated to be of major importance in some ecosystems. Further information on the sensitivity of European ecosystems to N deposition can be found in Nordin et al. (2011) and Bobbink and Hettelingh (2011).
5 Questions for Discussion
-
a.
Which factors about N deposition actually affect the biodiversity?
-
is it soil chemistry?
-
is it a competitive response?
-
is it a combination of factors, such as climate change AND N deposition, sulphuric acid AND N deposition?
-
-
b.
How confident are you in your results? How confident are you in extrapolating your results?
-
c.
What are the major unknowns yet related to your area of speciality? How can they be addressed?
These questions and others were considered by the working group on effects of N deposition (see Baron et al. 2014; Chap. 49, this volume).
References
Asman, W. A. H., Sutton, M. A., & Schjorring, J. K. (1998). Ammonia: Emission, atmospheric transport and deposition. New Phytologist, 139, 27–48.
Baron, J. S., Barber, M., Adams, M., Agboola, J. I., Allen, E. B., Bealey, W. J., Bobbink, R., Bobrovsky, M. V., Bowman, W. D., Branquinho, C., Bustamente, M. M. C., Clark, C. M., Cocking, E. C., Cruz, C., Davidson, E., Denmead, O. T., Dias, T., Dise, N. B., Feest, A., Galloway, J. N., Geiser, L. H., Gilliam, F. S., Harrison, I., Khanina, L. G., Lu, X., Manrique, E., Ochoa-Hueso, R., Ometto, J. P. H. P., Payne, R., Scheuschner, T., Sheppard, L. J., Simpson, G. L., Singh, Y. V., Stevens, C. J., Strachan, I., Sverdrup, H., Tokuchi, N., van Dobben, H., & Woodin, S. (2014). The effects of atmospheric nitrogen deposition on terrestrial and freshwater biodiversity. In M. A. Sutton, K. E. Mason, L. J. Sheppard, H. Sverdrup, R. Haeuber, & W. K. Hicks (Eds.), Nitrogen deposition, critical loads and biodiversity. Proceedings of the International Nitrogen Initiative workshop, linking experts of the Convention on Long-range Transboundary Air Pollution and the Convention on Biological Diversity. (Chap. 49 this volume). Springer.
Bates, J. W. (2002). Effects on bryophytes and lichens. In J. N. B. Bell & M. Treshow (Eds.), Air pollution and plant life (2nd ed., pp. 309–342). Chichester: Wiley.
Berdowski, J. J. M. (1993). The effect of external stress and disturbance factors on Calluna-dominated heathland vegetation. In R. Aerts & G. W. Heil (Eds.), Heathlands: Patterns and processes in a changing environment (pp. 85–124). Dordrecht: Kluwer.
Beusink, P., Nijssen, M., Van Duinen, G.-J., & Esselink, H. (2003). Broed- en voedselecologie van Grauwe klauwieren in intacte kustduinen bij Skagen, Denemarken. Nijmegen: Stichting Bargerveen, Afdeling Dierecologie, K.U.
Bobbink, R., Hornung, M., & Roelofs, J. G. M. (1998). The effects of air-borne nitrogen pollutants on species diversity in natural and semi-natural European vegetation. Journal of Ecology, 86, 717–738.
Bobbink, R., Ashmore, M., Braun, S., Flückiger, W., & van den Wyngaert, I. J. J. (2003). Empirical critical loads for natural and semi-natural ecosystems: 2002 update. In B. Achermann & R. Bobbink (Eds.), Empirical Critical Loads of Nitrogen (SAEFL Report 164., pp. 43–169). Bern: Swiss Agency for Environment Forests and Landscape.
Bobbink, R., Hicks, K., Galloway, J., Spranger, T., Alkemade, R., Ashmore, M., Bustamante, M., Cinderby, S., Davidson, E., Dentener, F., Emmett, B., Erisman, J. W., Fenn, M., Gilliam, F., Nordin, A., Pardo, L., & de Vries, W. (2010). Global assessment of nitrogen deposition effects on terrestrial plant diversity: A synthesis. Ecological Applications, 20, 30–59.
Bobbink, R., & Hettelingh, J. P. (2011). Review and revision of empirical critical loads and dose-response relationships. National Institute for Public Health and the Environment (RIVM), RIVM report 680359002/2011 (p. 244).
Brunsting, A. M. H., & Heil, G. W. (1985). The role of nutrients in the interactions between a herbivorous beetle and some competing plant species in heathlands. Oikos, 44, 23–26.
De Graaf, M. C. C., Bobbink, R., Roelofs, J. G. M., & Verbeek, P. J. M. (1998). Differential effects of ammonium and nitrate on three heathland species. Plant Ecology, 135, 185–196.
Dentener, F., Drevet, J., Lamarque, J. F., Bey, I., Eickhout, B., Fiore, A. M., Hauglustaine, D., Horowitz, L. W., Krol, M., Kulshrestha, U. C., Lawrence, M., Galy-Lacaux, C., Rast, S., Shindell, D., Stevenson, D., Noije, T. V., Atherton, C., Bell, N., Bergman, D., Butler, T., Cofala, J., Collins, B., Doherty, R., Ellingsen, K., Galloway, J., Gauss, M., Montanaro, V., Müller, J. F., Pitari, G., Rodriguez, J., Sanderson, M., Solmon, F., Strahan, S., Schultz, M., Sudo, K., Szopa, S., & Wild, O. (2006). Nitrogen and sulfur deposition on regional and global scales: A multimodel evaluation. Global Biogeochemical Cycles, 20, 21.
Dentener, F., Vet, B., Dennis, R. L., Enzai, D., Kulshrestha, U. C., & Galy-Lacaux, C. (2014). Progress in monitoring and modeling estimates of nitrogen deposition at local, regional and global scales. In M. A. Sutton, K. E. Mason, L. J. Sheppard, H. Sverdrup, R. Haeuber, & W. K. Hicks (Eds.), Nitrogen Deposition, Critical Loads and Biodiversity. Proceedings of the International Nitrogen Initiative workshop, linking experts of the Convention on Long-range Transboundary Air Pollution and the Convention on Biological Diversity. (Chap. 2 this volume). Springer.
Ellenberg, H. (1996). Vegetation Mitteleuropas mit den Alpen (5th edn). Stuttgart: Verlag Eugen Ulmer.
Esselink, H., Van Duinen, G.-J., Nijssen. M., Geertsma, M., Beusink, P., & Van den Burg, A. (2007). De grauwe klauwier mist kevers door verruigende duinen. Vakblad Natuur, Bos & Landschap, 2007–2004, 22–24.
Falkengren-Grerup, U. (1998). Nitrogen response of herbs and graminoids in experiments with simulated acid soil solution. Environmental Pollution, 102, 93–99.
Flückiger, W., Braun, S., & Hiltbrunner, E. (2002). Effects of air pollutants on biotic stress. In J. N. B. Bell & M. Treshow (Eds.), Air pollution and plant life (2nd edn., pp. 379–406). Chichester: Wiley.
Fowler, D. (2002). Pollutant deposition and uptake by vegetation. In J. N. B. Bell & Treshow, M. (Eds.), Air pollution and plant life (2nd edn., pp. 43–67). Chichester: Wiley.
Gigon, A., & Rorison, I. H. (1972). The response of some ecologically distinct plant species to nitrate- and to ammonium-nitrogen. Journal of Ecology, 60, 93–102.
Kinzel, S. (1982). Pflanzenökologie und Mineralstoffwechsel. Stuttgart: Ulmer.
Kleijn, D., Bekker, R. M., Bobbink, R., De Graaf, M. C. C., & Roelofs, J. G. M. (2008). In search for key biogeochemical factors affecting plant species persistence in heathland and acidic grasslands: A comparison of common and rare species. Journal of Applied Ecology, 45, 680–687.
Krupa, S. V. (2003). Effects of atmospheric ammonia (NH3) on terrestrial vegetation: A review. Environmental Pollution, 124, 179–221.
Nihlgård, B. (1985). The ammonium hyphothesis—an explanation to the forest dieback in Europe. Ambio, 14, 2–8.
Nijssen, M., Alders, K., Van der Smissen, N., & Esselink, H. (2001). Effects of grass encroachment and grazing management on carabid assemblages of dry dune grasslands. Proceedings of the Section Experimental and Applied Entomology of the Netherlands Entomological Society. Amsterdam, 12, 113–120.
Nordin, A., Sheppard, L. J., Strengbom, J., Bobbink, R., Gunnarsson, U., Hicks, W. K., & Sutton, M. A. (2011). New science on the effects of nitrogen deposition and concentrations on Natura 2000 sites (THEME 3), Background document. In W. K. Hicks, C. P. Whitfield, W. J. Bealey, & M. A. Sutton (Eds.), Nitrogen deposition and natura 2000: Science & practice in determining environmental impacts. COST729/Nine/ESF/CCW/JNCC/SEI Workshop Proceedings, published by COST.http://cost729.ceh.ac.uk/n2kworkshop. Accessed 1 June 2011
Ockinger, E., Hammarstedt, O., Nilsson, S. G., & Smith, H. G. (2006). The relationship of local extinctions of grassland butterflies and increased soil nitrogen levels. Biological Conservation, 128, 564–573.
Olsson, M. O., & Falkengren-Grerup, U. (2000). Potential nitrification as an indicator of preferential uptake of ammonium or nitrate by plants in an oak understory. Annals of Botany, 85, 299–305.
Paulissen, M. P. C. P., Van der Ven, P. J. M., Dees, A. J., & Bobbink, R. (2004). Differential effects of nitrate and ammonium on three fen bryophyte species in relation to pollutant nitrogen input. New Phytologist, 164, 551–458.
Pearson, J., & Stewart, G. R. (1993). The deposition of atmospheric ammonia and its effects on plants. New Phytologist, 125, 283–305.
Roelofs, J. G. M. (1983). Impact of acidification and eutrophication on macrophyte communities in softwaters in The Netherlands. Part I. Field observations. Aquatic Botany, 17, 139–155.
Roelofs, J. G. M., Kempers, A. J., Houdijk, A. L. F. M., & Jansen, J. (1985). The effects of air-borne ammonium sulphate on Pinus nigra in the Netherlands. Plant and Soil, 42, 372–377.
Smith, V. H., & Schindler, D. W. (2009). Eutrophication science: Where do we go from here? Trends in Ecology and Evolution, 24, 201–207.
Suding, K. N., Collins, S. L., Gough, L., Clark, C., Cleland, E. E., Gross, K. L., Milchunas, D. G., & Pennings, S. (2005). Functional- and abundance-based mechanisms explain diversity loss due to N fertilization. Proceedings of the National Academy of Sciences of the United States of America (PNAS), 102, 4387–4392.
Sutton, M. A., Erisman, J. W., Dentener, F., & Möller, D. (2008). Ammonia in the environment: From ancient times to present. Environmental Pollution, 156, 583–604.
Throop, H. L., & Lerdau, M. T. (2004). Effects of nitrogen deposition on insect herbivory: Implications for community and ecosystem processes. Ecosystems, 7, 109–133.
Ulrich, B. (1983). Soil acidity and its relation to acid deposition. In B. Ulrich & J. Pankrath (Eds.), Effects of accumulation of air pollutants in ecosystems (pp. 127–146). Boston: Reidel Publishing.
Ulrich, B. (1991). An ecosystem approach to soil acidification. In B. Ulrich & M. E. Summer (Eds.), Soil acidity (pp. 28–79). Berlin: Springer.
Van Breemen, N., Burrough, P. A., Velthorst, E. J., Van Dobben, H. F., De Wit, T., & Ridder, T. B. (1982). Soil acidification from atmospheric ammonium sulphate in forest canopy throughfall. Nature, 299, 548–550.
Van den Berg, L. J. L., Peters, C. J. H., Ashmore, M. R., & Roelofs, J. G. M. (2008). Reduced nitrogen has a greater effect than oxidised nitrogen on dry heathland vegetation. Environmental Pollution, 154, 359–369.
Van Dijk, H. F. G., De Louw, M. H. J., Roelofs, J. G. M., & Verburgh, J. J. (1990). Impact of artificial ammonium-enriched rainwater on soils and young coniferous trees in a greenhouse. Part 2—effects on the trees. Environmental Pollution, 63, 41–60.
Van Herk, C. M., Mathijssen-Spiekman, E. A. M., & de Zwart, D. (2003). Long distance nitrogen air pollution effects on lichens in Europe. Lichenologist, 35, 347–359.
Weiss, S. B. (1999). Cars, cows, and checkerspot butterflies: Nitrogen deposition and management of nutrient-poor grasslands for a threatened species. Conservation Biology, 13, 1476–1486.
Acknowledgments
The authors gratefully acknowledge support from the European Science Foundation to allow presentation at the workshop and from the NinE and COST 729 programmes.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media Dordrecht
About this chapter
Cite this chapter
Bobbink, R., Hicks, W. (2014). Factors Affecting Nitrogen Deposition Impacts on Biodiversity: An Overview. In: Sutton, M., Mason, K., Sheppard, L., Sverdrup, H., Haeuber, R., Hicks, W. (eds) Nitrogen Deposition, Critical Loads and Biodiversity. Springer, Dordrecht. https://doi.org/10.1007/978-94-007-7939-6_14
Download citation
DOI: https://doi.org/10.1007/978-94-007-7939-6_14
Published:
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-007-7938-9
Online ISBN: 978-94-007-7939-6
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)