Abstract
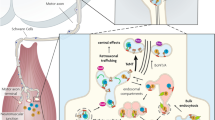
Tetanus and botulinum neurotoxins are clostridial toxins that cause tetanus and botulism, respectively. Tetanus neurotoxin binds specifically to peripheral motoneuron nerve terminals at the neuromuscular junction and is endocytosed within vesicles, which transport the toxin retroaxonally to the spinal cord. Here, it enters the inhibitory interneurons that ensure the balanced contraction of opposing skeletal muscle, and the metalloprotease domain of the toxin inactivates VAMP/synaptobrevin, a protein essential for neurotransmitter release. The synapse of the inhibitory circuit at the spinal cord is blocked resulting in a characteristic spastic paralysis. The botulinum neurotoxins are produced in dozens of different isoforms that can be grouped into seven distinct serotypes. They bind to neurospecific receptors enriched in the presynaptic membrane of cholinergic nerve terminals and are then mainly internalized inside the synaptic vesicles wherefrom their metalloprotease domain translocates in the cytosol and cleaves one of the three SNARE proteins that form the core of the nanomachine which mediates the neuroexocytosis with ensuing flaccid paralysis. In spite of the opposing clinical symptoms, botulinum and tetanus neurotoxins intoxicate neuronal cells in the same way and have similar functional and structural organizations. Common features and molecular diversities between tetanus and botulinum neurotoxin mechanism of action will be discussed in this present chapter.
Similar content being viewed by others
References
Araye, A., Goudet, A., Barbier, J., Pichard, S., Baron, B., England, P., Perez, J., Zinn-Justin, S., Chenal, A., & Gillet, D. (2016). The translocation domain of botulinum neurotoxin a moderates the propensity of the catalytic domain to interact with membranes at acidic pH. PLoS One, 11(4), e0153401.
Benefield, D. A., Dessain, S. K., Shine, N., Ohi, M. D., & Lacy, D. B. (2013). Molecular assembly of botulinum neurotoxin progenitor complexes. Proc Natl Acad Sci U S A, 110, 5630–5.
Bercsenyi, K., Schmieg, N., Bryson, J. B., Wallace, M., Caccin, P., Golding, M., et al. (2014). Tetanus toxin entry. Nidogens are therapeutic targets for the prevention of tetanus. Science, 346, 1118–23.
Binz, T., & Rummel, A. (2009). Cell entry strategy of clostridial neurotoxins. J Neurochem, 109, 1584–95.
Bilsland, L. G., Sahai, E., Kelly, G., Golding, M., Greensmith, L., & Schiavo, G. (2010). Deficits in axonal transport precede ALS symptoms in vivo. Proc Natl Acad Sci U S A, 107(47), 20523–8.
Bizzini, B., Grob, P., & Akert, K. (1981). Papain-derived fragment IIc of tetanus toxin: its binding to isolated synaptic membranes and retrograde axonal transport. Brain Res, 210, 291–9.
Bohnert, S., & Schiavo, G. (2005). Tetanus toxin is transported in a novel neuronal compartment characterized by a specialized pH regulation. J Biol Chem, 280, 42336–44.
Breidenbach, M. A., & Brunger, A. T. (2005). 2.3 A crystal structure of tetanus neurotoxin light chain. Biochemistry, 44(20), 7450–7.
Bruggemann, H., Baumer, S., Fricke, W. F., Wiezer, A., Liesegang, H., Decker, I., Herzberg, C., Martinez-Arias, R., Merkl, R., Henne, A., & Gottschalk, G. (2003). The genome sequence of Clostridium tetani, the causative agent of tetanus disease. Proc Natl Acad Sci USA, 100, 1316–21.
Chaddock, J. (2013). Transforming the domain structure of botulinum neurotoxins into novel therapeutics. Curr Top Microbiol Immunol, 364, 287–306.
Chen, S. (2014). Clostridial neurotoxins: mode of substrate recognition and novel therapy development. Curr Protein Pept Sci, 15(5), 490–503.
Colasante, C., Rossetto, O., Morbiato, L., Pirazzini, M., Molgo, J., & Montecucco, C. (2013). Botulinum neurotoxin type A is internalized and translocated from small synaptic vesicles at the neuromuscular junction. Mol Neurobiol, 48, 120–7.
Comella, J. X., Molgó, J., & Faille, L. (1993). Sprouting of mammalian motor nerve terminals induced by in vivo injection of botulinum type D toxin and the functional recovery of paralysed neuromuscular junctions. Neurosci Lett, 153, 61–4.
Eisele, K. H., Fink, K., Vey, M., & Taylor, H. V. (2011). Studies on the dissociation of botulinum neurotoxin type A complexes. Toxicon, 57, 555–65.
Eleopra, R., Tugnoli, V., Quatrale, R., Rossetto, O., & Montecucco, C. (2004). Different types of botulinum toxin in humans. Mov Disord, 19, S53–9.
Fischer, A., & Montal, M. (2013). Molecular dissection of botulinum neurotoxin reveals interdomain chaperone function. Toxicon, 75, 101–7.
Foran, P. G., Mohammed, N., Lisk, G. O., Nagwaney, S., Lawrence, G. W., Johnson, E., et al. (2003). Evaluation of the therapeutic usefulness of botulinum neurotoxin B, C1, E, and F compared with the long-lasting type A. J Biol Chem, 278, 1363–71.
Foster, K. A. (2014). Clinical applications of botulinum neurotoxin (Current topics in neurotoxicity). New York: Springer.
Fujinaga, Y., Sugawara, Y., & Matsumura, T. (2013). Uptake of botulinum neurotoxin in the intestine. Curr Top Microbiol Immunol, 364, 45–59.
Georgiou, J., Robitaille, R., Trimble, W. S., & Charlton, M. P. (1994). Synaptic regulation of glial protein expression in vivo. Neuron, 12, 443–55.
Hallett, M., Albanese, A., Dressler, D., Segal, K. R., Simpson, D. M., Truong, D., & Jankovic, J. (2013). Evidence-based review and assessment of botulinum neurotoxin for the treatment of movement disorders. Toxicon, 67, 94–114.
Harper, C. B., Martin, S., Nguyen, T. H., Daniels, S. J., Lavidis, N. A., Popoff, M. R., Hadzic, G., Mariana, A., Chau, N., McCluskey, A., Robinson, P. J., & Meunier, F. A. (2011). Dynamin inhibition blocks botulinum neurotoxin type A endocytosis in neurons and delays botulism. J Biol Chem, 286, 35966–76.
Hughes, R., & Whaler, B. C. (1962). Influence of nerve-ending activity and of drugs on the rate of paralysis of rat diaphragm preparations by Cl. botulinum type A toxin. J Physiol, 160, 221–33.
Johnson, E. A., & Montecucco, C. (2008). Botulism. Handb Clin Neurol, 91, 333–68.
Juzans, P., Comella, J. X., Molgó, J., Faille, L., & Angaut-Petit, D. (1996). Nerve terminal sprouting in botulinum type-A treated mouse levator auris longus muscle. Neuromuscul Disord, 6, 177–85.
Kalb, S. R., Baudys, J., Raphael, B. H., Dykes, J. K., Luquez, C., Maslanka, S. E., & Barr, J. R. (2015). Functional characterization of botulinum neurotoxin serotype H as a hybrid of known serotypes F and A (BoNT F/A). Anal Chem, 87(7), 3911–7.
Kalb, S. R., Baudys, J., Webb, R. P., Wright, P., Smith, T. J., Smith, L. A., Fernandez, R., Raphael, B. H., Maslanka, S. E., Pirkle, J. L., & Barr, J. R. (2012). Discovery of a novel enzymatic cleavage site for botulinum neurotoxin F5. FEBS Lett, 586(2), 109–15.
Keller, J. E., Cai, F., & Neale, E. A. (2004). Uptake of botulinum neurotoxin into cultured neurons. Biochemistry, 43(2), 526–32.
Keller, J. E., Neale, E. A., Oyler, G., & Adler, M. (1999). Persistence of botulinum neurotoxin action in cultured spinal cord cells. FEBS Lett, 456, 137–42.
Kitamura, M., Igimi, S., Furukawa, K., & Furukawa, K. (2005). Different response of the knockout mice lacking b-series gangliosides against botulinum and tetanus toxins. Biochim Biophys Acta, 1741(1-2), 1–3.
Kumaran, D., Eswaramoorthy, S., Furey, W., Navaza, J., Sax, M., & Swaminathan, S. (2009). Domain organization in clostridium botulinum neurotoxin type E is unique: its implication in faster translocation. J Mol Biol, 386(1), 233–45.
Lacy, D., Tepp, W., Cohen, A., DasGupta, B., & Stevens, R. (1998). Crystal structure of botulinum neurotoxin type A and implications for toxicity. Nat Struct Mol Biol, 5(10), 898–902.
Lee, K., Gu, S., Jin, L., Le, T., Cheng, L., Strotmeier, J., et al. (2013). Structure of a bimodular botulinum neurotoxin complex provides insights into its oral toxicity. PLoS Pathog, 9(10), e1003690.
Masuyer, G., Chaddock, J. A., Foster, K. A., & Acharya, K. R. (2014). Engineered botulinum neurotoxins as new therapeutics. Annu Rev Pharmacol Toxicol, 54, 27–51.
Matteoli, M., Verderio, C., Rossetto, O., Iezzi, N., Coco, S., Schiavo, G., & Montecucco, C. (1996). Synaptic vesicle endocytosis mediates the entry of tetanus neurotoxin into hippocampal neurons. Proc Natl Acad Sci U S A, 93(23), 13310–5.
Mazzocchio, R., & Caleo, M. (2015). More than at the neuromuscular synapse: actions of botulinum neurotoxin A in the central nervous system. Neuroscientist, 21, 44–61.
Megighian, A., Zordan, M., Pantano, S., Scorzeto, M., Rigoni, M., Zanini, D., Rossetto, O., & Montecucco, C. (2013). Evidence for a radial SNARE super-complex mediating neurotransmitter release at the Drosophila neuromuscular junction. J Cell Sci, 126, 3134–40.
Meunier, F. A., Lisk, G., Sesardic, D., & Dolly, J. O. (2003). Dynamics of motor nerve terminal remodeling unveiled using SNARE-cleaving botulinum toxins: the extent and duration are dictated by the sites of SNAP-25 truncation. Mol Cell Neurosci, 22, 454–66.
Montal, M. (2010). Botulinum neurotoxin: a marvel of protein design. Annu Rev Biochem, 79, 591–617.
Montecucco, C. (1986). How do tetanus and botulinum toxins bind to neuronal membranes? Trends Biochem Sci, 11, 314–7.
Montecucco, C., & Rasotto, M. (2015). On botulinum neurotoxin variability. mBio, 6(1), e02131–.
Morbiato, L., Carli, L., Johnson, E. A., Montecucco, C., Molgo, J., & Rossetto, O. (2007). Neuromuscular paralysis and recovery in mice injected with botulinum neurotoxins A and C. Eur J Neurosci, 25(9), 2697–704.
Muraro, L., Tosatto, S., Motterlini, L., Rossetto, O., & Montecucco, C. (2009). The N-terminal half of the receptor domain of botulinum neurotoxin A binds to microdomains of the plasma membrane. Biochem Biophys Res Commun, 380, 76–80.
Pantano, S., & Montecucco, C. (2014). The blockade of the neurotransmitter release apparatus by botulinum neurotoxins. Cell Mol Life Sci, 71(5), 793–811.
Pirazzini, M., Bordin, F., Rossetto, O., Shone, C. C., Binz, T., & Montecucco, C. (2013a). The thioredoxin reductase-thioredoxin system is involved in the entry of tetanus and botulinum neurotoxins in the cytosol of nerve terminals. FEBS Lett, 587, 150–5.
Pirazzini, M., Rossetto, O., Bertasio, C., Bordin, F., Shone, C., Binz, T., et al. (2013b). Time course and temperature dependence of the membrane translocation of tetanus and botulinum neurotoxins C and D in neurons. Biochem Biophys Res Commun, 430(1), 38–42.
Pirazzini, M., Rossetto, O., Bolognese, P., Shone, C. C., & Montecucco, C. (2011). Double anchorage to the membrane and intact inter-chain disulfide bond are required for the low pH induced entry of tetanus and botulinum neurotoxins into neurons. Cell Microbiol, 13, 1731–43.
Pirazzini, M., Tehran, D., Leka, O., Zanetti, G., Rossetto, O., & Montecucco, C. (2016). On the translocation of botulinum and tetanus neurotoxins across the membrane of acidic intracellular compartments. Biochim Biophys Acta, 1858(3), 467–74.
Popoff, M. R., & Marvaud, J. C. (1999). Structural and genomic features of clostridial neurotoxins. In J. E. Alouf & J. H. Freer (Eds.), The comprehensive sourcebook of bacterial protein toxins (2nd ed., pp. 174–201). London: Academic.
Restani, L., Giribaldi, F., Manich, M., Bercsenyi, K., Menendez, G., Rossetto, O., Caleo, M., & Schiavo, G. (2012). Botulinum neurotoxins A and E undergo retrograde axonal transport in primary motor neurons. PLoS Pathog, 8, e1003087.
Rossetto, O., Pirazzini, M., & Montecucco, C. (2014). Botulinum neurotoxins: genetic, structural and mechanistic insights. Nat Rev Microbiol, 12(8), 535–49.
Rossetto, O., Schiavo, G., Montecucco, C., Poulain, B., Deloye, F., Lozzi, L., & Shone, C. C. (1994). SNARE motif and neurotoxins. Nature, 372(6505), 415–6.
Rummel, A. (2013). Double receptor anchorage of botulinum neurotoxins accounts for their exquisite neurospecificity. Curr Top Microbiol Immunol, 364, 61–90.
Schmitt, A., Dreyer, F., & John, C. (1981). At least three sequential steps are involved in the tetanus toxin induced block of neuromuscular transmission. Naunyn Schmiedebergs Arch Pharmacol, 317, 326–30.
Schmieg, N., Bercsenyi, K., & Schiavo, G. (2015). Uptake and transport of clostridial neurotoxins. In J. E. Alouf, D. Ladant, & M. R. Popoff (Eds.), The comprehensive sourcebook of bacterial protein toxins (4th ed.). New York: Academic.
Scott, A. B., Rosenbaum, A., & Collins, C. C. (1973). Pharmacologic weakening of extraocular muscles. Invest Ophthalmol, 12, 924–7.
Shoemaker, C. B., & Oyler, G. A. (2013). Persistence of Botulinum neurotoxin inactivation of nerve function. Curr Top Microbiol Immunol, 364, 179–96.
Simpson, L. L. (2013). The life history of a botulinum toxin molecule. Toxicon, 68, 40–59.
Smith, T. J., Hill, K. K., & Raphael, B. H. (2015). Historical and current perspectives on Clostridium botulinum diversity. Res Microbiol, 166, 290–302.
Son, Y. J., & Thompson, W. J. (1995). Nerve sprouting in muscle is induced and guided by processes extended by Schwann cells. Neuron, 14, 133–41.
Sun, S., Tepp, W. H., Johnson, E. A., & Chapman, E. R. (2012). Botulinum neurotoxins B and E translocate at different rates and exhibit divergent responses to GT1b and low pH. Biochemistry, 51, 5655–62.
Sutton, R. B., Fasshauer, D., Jahn, R., & Brunger, A. T. (1998). Crystal structure of a SNARE complex involved in synaptic exocytosis at 2.4 A resolution. Nature., 395, 347–53.
Swaminathan, S., & Eswaramoorthy, S. (2000). Structural analysis of the catalytic and binding sites of Clostridium botulinum neurotoxin B. Nat Struct Biol, 7, 693–9.
Toivonen, J. M., Olivan, S., & Osta, R. (2010). Tetanus toxin C-fragment: the courier and the cure? Toxins (Basel), 2(11), 2622–44.
Umland, T. C., Wingert, L. M., Swaminathan, S., Furey, W. F., Schmidt, J. J., & Sax, M. (1997). Structure of the receptor binding fragment HC of tetanus neurotoxin. Nat Struct Biol, 4(10), 788–92.
Vagin, O., Tokhtaeva, E., Garay, P. E., Souda, P., Bassilian, S., Whitelegge, J. P., Lewis, R., Sachs, G., Wheeler, L., Aoki, R., & Fernandez-Salas, E. (2014). Recruitment of septin cytoskeletal proteins by botulinum toxin A protease determines its remarkable stability. J Cell Sci, 127(Pt 15), 3294–308.
Wheeler, A., & Smith, H. S. (2013). Botulinum toxins: mechanisms of action, antinociception and clinical applications. Toxicology, 306, 124–46.
Whitemarsh, R. C., Tepp, W. H., Johnson, E. A., & Pellett, S. (2014). Persistence of botulinum neurotoxin A subtypes 1-5 in primary rat spinal cord cells. PLoS One, 9(2), e90252.
Yeh, F. L., Dong, M., Yao, J., Tepp, W. H., Lin, G. Y., Johnson, E. A., et al. (2010). SV2 mediates entry of tetanus neurotoxin into central neurons. PLoS Pathog, 6, e1001207.
Zanetti, G., Azarnia Tehran, D., Pirazzini, M., Binz, T., Shone, C. C., Fillo, S., Lista, F., Rossetto, O., & Montecucco, C. (2015). Inhibition of botulinum neurotoxins interchain disulfide bond reduction prevents the peripheral neuroparalysis of botulism. Biochem Pharmacol, 98, 522–30.
Acknowledgments
Work in the authors’ laboratory is supported by the University of Padova, Fondazione Cariparo, and the Italian Ministry of Defence.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer Science+Business Media Dordrecht
About this entry
Cite this entry
Rossetto, O., Montecucco, C. (2016). Tetanus and Botulinum Neurotoxins. In: Gopalakrishnakone, P., Stiles, B., Alape-Girón, A., Dubreuil, J., Mandal, M. (eds) Microbial Toxins. Toxinology. Springer, Dordrecht. https://doi.org/10.1007/978-94-007-6725-6_19-1
Download citation
DOI: https://doi.org/10.1007/978-94-007-6725-6_19-1
Received:
Accepted:
Published:
Publisher Name: Springer, Dordrecht
Online ISBN: 978-94-007-6725-6
eBook Packages: Springer Reference Biomedicine and Life SciencesReference Module Biomedical and Life Sciences