Abstract
The present Mediterranean molluscan biodiversity, comprising more than 2,000 species, was shaped by the multiple events connected with the geodynamic and climatic evolution of this basin in the last million years coupled with the process of biological evolutionary processes on the global scale. Climate, hydrology, basin physiography, and connections via seaways were and are the main forcing factors tuning the type of molluscs making the Mediterranean diversity at any geological instant, resulting in (often cyclical) taxonomic additions and subtractions. However, this basic motif operating in the Mediterranean since its formation as an independent basin over millions of years, has been seriously perturbed by the deliberate or accidental introduction of non-indigenous (alien or non native) species in the last decades. The taxonomic addition of mostly warm-water species is taking place at an unprecedented pace with respect to even the dramatic changes that have marked glacial-interglacial transitions, thus making the present day Mediterranean Sea a planetary biogeographic unicum.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
The Mediterranean Sea is considered as a biodiversity hotspot of exceptional planetary value (Coll et al. 2010). With a present estimate of ca. 17,000 marine species it accounts for as much as 7.5 % of total marine diversity (Coll et al. 2010; Danovaro et al. 2010). Such comparably high richness when contrasted with the relatively small surface area of the basin (ca. 2.5 million km2, 0,7 % of the global ocean surface) is the net result of the basin’s complex geological history, mid-latitude geographic position, hydrological regimes, varied coastal (rocky shores, sandy beaches, lagoons, islands etc.) and submarine topography (shelves, bathyal plains, canyons, seamounts, active submarine volcanoes, anoxic bottoms, mud volcanoes etc.), wide bathymetric range (the mean depth is about 1,525 m, and the deepest point is the Matapan Deep System or Matapan–Vavilov Deep in the Hellenic Trench at ca. 5,121 m), and the past and present regional variability of climate within the basin (Taviani 2002; Sardà et al. 2004; Danovaro et al. 2010) (Fig. 16.1).
Snapshots of Mediterranean mollusc biodiversity. The established present situation (a) the quintessential Mediterranean endemic bivalve Pinna nobilis (Linnaeus 1758) embedded in coarse sandy sediment at 10 m within a seagrass meadow, Montecristo island, Tyrrhenian Sea (© E. Lipparini 2009). The recent past (b) a shell assemblage dominated by the pectinid Pseudamussium peslutrae, one of the most common bivalves in the Quaternary Mediterranean basin, CNR Cruise SETE of R/V Urania, southern Adriatic Sea, ca.–350 m. The on-going change (c) screen from an industrial plant in Augusta, Sicily heavily biofouled by the non-indigenous invasive mussel Brachidontes pharaonis(© M. Faimali 2008)
The Mollusca is one of the most species-rich phyla inhabiting the Mediterranean waters from brackish lagoons to bathyal depths. For instance, among macro-invertebrates, molluscs outnumber all other taxonomic groups (Relini 2008, 2010). The Mediterranean molluscan fauna is considered as the best known in the world (Oliverio 2003), numbering more than 2,000 species (Coll et al. 2010), although this figure is being steadily increased by the addition of new taxa, including introduced species. Interestingly but not surprisingly, even among the non-indigenous species, Mollusca play a leading role accounting for almost one fifth of exotic taxa presently known from the Mediterranean (Zenetos et al. 2010; CIESM 2012).
In this chapter we discuss the present day Mediterranean molluscan biodiversity, tracing its history and evaluating its potential developments in the mid term. One major advantage of using molluscs for such an analysis is the phylum’s high biodiversity and ecological adaptability promoting colonization of virtually all marine environments. Another is the occurrence in most member species of a durable mineral skeleton, the shell, which notoriously permits an easier evaluation of taxonomy (therefore diversity and biogeography) through time at various temporal scales. By large, the focus of our discussion is centred upon shell-bearing benthic molluscs that also permits a comparison with the fossil record.
From Past to Present
The Mediterranean marine biodiversity as presently known is the result of many interacting factors of both regional and global scale. The present situation is but the very last of the many “diversity snapshots” configuring at any temporal instant the biological load of this basin. Today’s biodiversity is intimately linked but not equal to past and future changes. Various studies have tried to describe and explain the history of the marine biota of the Mediterranean in light of its very complex and dynamic geologic and climatic evolution and the reader should refer to these papers for details (Pérès 1989; Por and Dimentman 1989; Bianchi and Morri 2000; Corselli 2001; Taviani 2002, with references).
Most Authors agree upon the pivotal event represented by the late Miocene Messinian Salinity Crisis (CIESM 2008; Roveri et al. 2008). In short, a widely shared view is that the marine stenoecious Mediterranean marine fauna was annihilated within a hydrologicaly-compromised basin, and then completely renewed by the following Pliocene inundation from the Atlantic Ocean at 5.33 million years (Hsü et al. 1977; Taviani 2004; Monegatti and Raffi 2010, with references therein). Some Authors contend, however, that this Mediterranean reflooding might have taken place already in the terminal Miocene during the so-called Lago-mare event (Carnevale et al. 2008; Ligios et al. 2012). At any event, the post-Messinian crisis Mediterranean marine molluscan biodiversity basically evolved from this wave of invaders. On the other hand, this view is not unanimously accepted and is contrasted on some aspects as it does not explain all observations. For instance, Por (2009), on the basis of records of fossil fish and of certain benthic species (e.g., echinoids: Néraudeau et al. 2001) in the westernmost part of the basin, admits the permanence of trans-Messinian marine stocks in coastal areas, beside the presence of supposedly Tethyan relics such as the present endemic seagrass Posidonia oceanica (L) Delile 1813, to the Mediterranean. Tethyan relics, basically marine organisms echoing the times when the proto-Mediterranean was still in direct connection with the Indo-Pacific region, may include molluscs both in the Plio-Pleistocene and recent Mediterranean fauna (e.g., Ponder 1989; La Perna 2005, 2006; La Perna and D’Abramo 2011). However, the significance of Tethyan relics has been discussed by Taviani (2002) that opposes the view that these faunal elements did survive permanently inside the Mediterranean.
We can probably accept that at least a substantial part of the post-crisis Mediterranean benthic fauna is of Atlantic origin and that the core of the Mediterranean mollusc diversity in the Pliocene is represented by Miocene species (Raffi 1993), most of which hypothetically to have necessarily survived the crisis outside the basin in the Atlantic (Sabelli and Taviani 1984; Jones 1984; Taviani 2004). Monegatti and Raffi (2010) argue that most of the paleoendemic molluscan fauna inhabiting the pre-Messinian proto-Mediterranean basin vanished before the salinity crisis and that the molluscan fauna replenishing the Mediterranean in the early Pliocene was shaped in the Miocene of the adjacent Atlantic Ocean. This Miocene core was later modified by the addition of neo-endemics (putatively) evolved within the basin (Oliverio 1995b; Le Renard et al. 1996; Pedriali and Robba 2001; Forli et al. 2002, 2004; Landau et al. 2004, and many others).
In any case, within 3 million years this Pliocene fauna was exposed to the dramatic oceanographic and climatic modifications associated with the change from the tropical-subtropical situation of the Pliocene (Zanclean-Piacenzian, from ca. 5.33 to 2.59 Ma) to the cooler times of the Pleistocene starting with the Gelasian (2.59–1.80 Ma), which had important repercussions on the marine biota (Taviani 2002; Monegatti and Raffi 2010). These events have been documented to have strongly impacted the molluscan diversity of the Mediterranean causing for instance the selective extinction within this basin of a number of taxa of warm-water affinity inhabiting the continental margin (Le Renard et al. 1996; Monegatti and Raffi 2001, 2010; La Perna et al. 2002, 2004; Ceregato et al. 2007). The deep water mollusc fauna does not necessarily conform completely to the trend observed in shallower water environments although, at least some bivalves, such as Acharax doderleini Mayer 1861, seems to be disappeared in the Piacenzian (Taviani et al. 2011a). As suggested by diversity in family Nuculidae, the Plio-Pleistocene Mediterranean mollusc fauna contained putative endemic shallow water taxa up to the Early Pleistocene, some of which did not survive further into the basin (La Perna 2007)
It has been repeatedly observed that many Neogene Mediterranean gastropods only differ from their Pleistocene to Recent counterparts in the typology of their embryonic shell (Solsona and Martinell 1999). This is the case for instance of Neverita olla (de Serres, 1829) vs N. josephinia Risso 1826 (Pedriali and Robba 2009), Pagodula vaginata (De Cristofori and Jan 1832) vs P. echinata (Kiener 1842) (La Perna 1996), Cyclope migliorinii (Bevilacqua 1928) vs C. neritea Linnaeus 1758 (Gili and Martinell 2000), some Terebridae (Bouchet 1981) and many others. This rather mysterious trend has not yet been satisfactory explained apart from a generic attribution to climatic driven causes. The real taxonomic significance of such chronological pairs still needs to be properly addressed and fully understood.
Ultimately, as the Pleistocene progressed, the Mediterranean achieved a great part of its present status of a temperate marine basin at least as regards seawater temperature. The onset of cyclical glacial ages on Earth caused the periodic entry inside the basin of benthic elements, mostly molluscs, from the adjacent Eastern Atlantic Ocean (e.g., Gignoux 1913; Pelosio and Raffi 1973; Raffi 1986; Malatesta and Zarlenga 1988; Taviani et al. 1991; Bellomo 1993; Marabini et al. 1995). With respect to the Mediterranean mollusc fauna, these periodical cold times of intense colonization of its coastal habitats by such Atlantic Boreo-Celtic species (referred to in the literature as “cold” or “northern guests”), i.a. Arctica islandica (Linnaeus 1767), Modiolus modiolus Linnaeus 1758, Pseudamussium peslutrae (Linnaeus 1771), Mya truncata Linnaeus 1758, Panomya norvegica (Spengler 1793), Buccinum undatum Linnaeus 1758, Buccinum humphreysianum Bennet 1824, Neptunea contraria (Linnaeus 1771), profoundly impacted the structure of the basin’s biota for the longest duration of the post-Gelasian Quaternary. The last expansion of these north-eastern Atlantic molluscs took place during the last glacial epoch (Mars 1958; Colantoni 1973; Colantoni et al. 1975; Taviani 1978; Domènech and Martinell 1980, 1982; Curzi et al. 1984; Taviani et al. 1991), after which they (temporarily) withdrew from the Mediterranean except B. humphreysianum and P. peslutrae (see below). Shorter-lasting temperature reversals (interglacials) allowed the colonisation of the Mediterranean by sub-tropical elements the best known examples of which is Strombus and the associate ‘Senegalese’ fauna of the last interglacial (also known as “warm guests” in the literature) comprising a cohort of subtropical species (Gignoux 1913; Issel 1914; Ruggieri and Buccheri 1968; Bellomo 1996; Taviani 2002 with references). More in detail, the Strombus fauna includes a number of extant West African taxa, such as Persististrombus latus (Gmelin 1791) (better known in the literature as Strombus bubonius Lamarck 1822), Polinices lacteus (Guilding 1834), Monoplex trigonus (Gmelin 1791), Gemophos viverratus (Kiener, 1834), Conus ermineus Born 1778, Brachidontes puniceus (Gmelin 1791), and many others that apparently only once reached and established themselves successfully in the Mediterranean, during the short time of the penultimate interglacial (ca. 125 ka BP: Marine Isotope Stage 5e, commonly recorded in the Mediterranean stratigraphy as the Tyrrhenian Stage: Antonioli et al. 2008). This could have also been the time when Patella ferruginea Gmelin 1791, spread in the Mediterranean where it apparently acclimatized successfully up to present, although only in the western basin.
As for the deep sea, it is noteworthy that during the Pliocene (at least from the Piacenzian) and part of the Pleistocene, bottom water temperature kept the Mediterranean basin within psycrospheric conditions (Di Geronimo and La Perna 1996, 1997; Rosso and Di Geronimo 1998; Corselli 2001; La Perna 2003, 2004, 2007; La Perna et al. 2004). Such cooler than present temperature favoured the establishment of a diverse molluscan fauna in the Pliocene and up to the Sicilian stage in the Pleistocene decreasing in diversity from then onwards with the establishment of warm homothermal conditions (Raffi and Taviani 1984; Di Geronimo and La Perna 1997; La Perna 2004; Tabanelli 2008). Glacial times seem to be more propitious in the last 700 ka for temporal increases in the deep sea benthic diversity as documented by Late Glacial submerged subfossil taphocoenoses containing various Atlantic species no longer living at present in the Mediterranean (Taviani 1974; Raffi and Taviani 1984; Taviani et al. 2011b).
In summary, what we see today in the Mediterranean Sea is the (temporary) result of various filters. Above all, the geodynamic evolution of the basin controls through time the opening and closure of seaways at various times and the establishment of a variety of physiographic conditions, the global climate with its regional responses influenced land and sea (Taviani 2002), and both factors acted together in driving the oceanographic properties of Mediterranean water masses that sustain its benthic fauna.
The Marine Molluscs of the Mediterranean: How Many Species at Present?
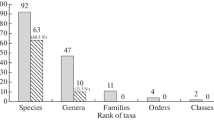
The term biodiversity usually refers to the main three levels of genes, species and ecosystems (Wilson 1988), and is here limited to the species level (species diversity or species richness). Molluscs represent the 18 % of the entire marine fauna of the Mediterranean sea, they are only surpassed by crustaceans (19 %) but exceed the annelids (10 %) representing the third most diverse marine invertebrate taxon (Coll et al. 2010). The most recent catalogues of the marine Mollusca species inhabiting the Mediterranean Sea list more than 2,100 species (see Coll et al. 2010). Mediterranean molluscs include members of all known existing classes, i.e. Caudofoveata 9 species (0.4 %), Solenogastres 29 species (1.4 %), Monoplacophora 1 species (0.04 %: no living individuals collected so far), Polyplacophora 30 species (1.4 %), Gastropoda 1,558 species (73 %), Bivalvia 430 species (20 %), Scaphopoda 14 species (0.7 %) and Cephalopoda 65 species (3 %). Molluscs are mainly benthic, but some gastropods are holoplanktic: essentially Heteropoda (14 species) and Thecosomata (49 species), while cephalopods are mostly nektonic (45 species). Holoplanktic molluscs are not treated here and the reader may refer to the monograph on the eastern Mediterranean by Janssen (2012) and to Janssen and Peijnenburg (Chap. 20, this volume) for the recentmost update.
Besides depending upon the authorial concept of species, any number given is far from being definitive and the tally is in need of constant updating mostly because of the steady addition of novel taxa through deliberate or unintentional human action. Furthermore, some species are only known from their mineralized exoskeleton (the shell), rather than from living individuals (Table 16.1). This fact introduces a potential bias (that is, an over-estimation) in assessing the present biodiversity especially as regards deep water taxa and holoplanktonic species which could well represent, in some cases, pre-modern remains. This holds particularly true for deep sea molluscs whose empty shells may at time even pertain to former late Pleistocene communities (e.g., Taviani 1974, 1978; Taviani and Colantoni 1979; Di Geronimo and Bellagamba 1985; Taviani and Taviani 1986; Bouchet and Taviani 1989; Bonfitto et al. 1994; Pons-Moya and Pons 2000; Panetta et al. 2003).
With respect to the shelled molluscs, three contributions deserve special attention. Monterosato (1878) published the first complete compendium by listing 989 species. About a century later Piani (1980) published a list of 1,502 species, a figure which has now risen to 1667 due to the work of Coll et al. (2010) representing an average increase of about five species/year.
The discrepancies apparent from a simple comparison of recently produced lists underlies the current uncertainness in defining species in Mollusca. For example, the most recent compilations by Coll et al. (2010) and Cossignani and Ardovini (2011) report 1,221 vs. 1,341 shelled gastropods and 430 vs. 441 bivalves, respectively. Such substantial differences derive from the various criteria accepted by different authors as to as to which entities are valid species. Alpha taxonomy of shell bearing molluscs was, and still is, primarily based on shell morphology and colour pattern, and occasionally on morphometric analysis (Carvajal-Rodriguez et al. 2005, 2006; Criscione et al. 2009; Criscione and Patti 2010). It is always important to keep in mind that shelled molluscs have for millennia captured human attention for their aesthetic value (Taviani and Fiocchi 1997). This attitude, similarly for Lepidoptera (butterflies) or Coleoptera (beetles), has direct implications for the taxonomy of the Phylum that may be both positive and negative. The positive aspect is that fascination with this group has resulted in a vast number of workers, a large proportion of whom are amateurs, actively searching for shells, thus making enormous quantities of data and material available for study. The other side of the coin might be a sort of distressed superficiality or incompleteness in defining species based upon hasty subjective evaluation of shell features only (the frenzy for “discovering” new species as evoked by Ghisotti 1974a). In the last decades taxonomy benefitted by the routinely application of advanced technologies such as scanning electron microscopy that permit study of morphology at a great level of detail representing a substantial step forwards in assessing species, especially in morphologically complex groups. An excellent case of this is the family Triphoriidae where SEM images of larval stages (Thiriot-Quivrieux and Rodriguez-Babio 1975; Bouchet 1984), coupled with the radular morphology and of the colour patterns of the living mollusc, resulted in the identification of a number of Atlanto-Mediterranean species and genera out of what was previously considered a single very variable species Triphora perversa with its variants (Bouchet and Guillemot 1978; Bouchet 1984, 1995). In fact, besides the shell, the radula is often used to delimit taxa at species level in Gastropoda but its value is somewhat controversial primarily because of its variability in relation to ontogeny (Warén 1990), sex (Fretter 1984; Mutlu 2004), and phenotypic plasticity (Padilla 1998). The integration of shell and soft part morphologic features has been used for unravelling the systematics of complex groups such as for example Marginellidae and Cerithiidae (Gofas 1992; Garilli and Galletti 2006).
A further issue with strong repercussions on gastropod taxonomy is the occurrence of species pairs characterized by possessing an identical teleconch but distinct protoconchs (size, sculpture and number of whorls). The protoconch is assumed to reflect contrasting developmental pathways, i.e. direct and/or lecithotrophic vs. planktotrophic and this bears on taxonomic evaluation (see Oliverio 1996 and bibliography therein). The current view is that such mollusc pairs that only differ in shell protoconch type could be considered as distinct specific entities (Bouchet 1989; Solsona and Martinell 1999; Mariottini et al. 2009). The alternative hypothesis postulated by some authors that the same mollusc species could present two different kind of development (poecilogony) is still to be demonstrated (Hoagland and Robertson 1988; Bouchet 1989). However, some authors admit to this possibility attesting the uncertainness still reigning on this crucial issue (Cumming 1993; Gibson and Chia 1995; Chester 1996; Krug 2007). The adoption of the protoconch morphology (= developmental strategy) as diagnostic in separating species has in the last 30 years resulted in establishing many new taxa of Mediterranean gastropods solely or almost entirely based upon this criterion. The most emblematic case in point is offered by the species-rich family Rissoidae. Before the 1960s, the family included 98 nominal species, but this figure has now reached 154; many of these additions are based upon species pairs as described before. Similar considerations apply to other families as well (i.e. Nassariidae, Conidae, Pyramidellidae). Some species have also been split primarily by their very different egg capsules, such as the gastropods Nassarius cuvierii (Payraudeau, 1826) and N. unifasciatus (Kiener, 1834) (Moreno and Templado 1994).
The isozyme electrophoresis approach, although scantily adopted by European malacologists, has contributed to clarifying the status of some Mediterranean taxa, especially when integrated with the more traditional descriptors just outlined. For examples, it has been possible to resolve such controversial taxa as the bivalves Chamelea gallina (Linnaeus 1758) and C. striatula (Da Costa 1778) (Backeljau et al. 1994) and, among the gastropods, Nassarius reticulatus (Linnaeus 1758) and N. nitidus (Jeffreys, 1867) (Sanjuan et al. 1997), species within the Rissoidae (Oliverio 1984; Colognola et al. 1986; Munksgaard 1990) and Cerithiidae (Boisselier-Dubayle and Gofas 1999).
To date, the growing application of molecular techniques is bringing fresh breath to the alpha taxonomy of Mediterranean molluscs. Sequencing of DNA or RNA represents the most recent approach widely and successfully used for supraspecific and phylogenetic recontructions (e.g., Schiaparelli et al. 2005; Plazzi et al. 2011; Donald et al. 2012; Sharma et al. 2012). The application of bio-molecular methods for species-level definition is gaining momentum but still presents its own problems; nevertheless, such methods have revealed sibling species in bivalves (the Mactra corallina group: Guarniero et al. (2010), and resolved debated morphospecies in Polyplacophora (Acanthochitona crinita (Pennant 1777)-A. oblonga (Leloup 1981)): Bonfitto et al. 2011) and in the gastropod genera Ocinebrina (Crocetta et al. 2012) and Cerithiopsis (Modica et al. 2013). The intertidal habitat-engineering vermetid gastropod Dendropoma petraeum (Monterosato 1884) has been shown to likely contain four criptic species at least (Calvo et al. 2009), and the existence of one possible cryptic species in the eastern Mediterranean has also been proposed for the limpet Patella rustica Linnaeus 1758 (Sá-Pinto et al. 2010). Interestingly two main lineages within the common brackish water bivalve Cerastoderma glaucum (Poiret 1789), one Aegean-Ponto-Caspian, the other central-western Mediterranean-Boreo Atlantic, diverged considerably, possibly suggesting an incipient speciation (Nikula and Väinölä 2003). Finally, the genetic screening of a non-native bivalve of unknown original provenance known for long as Anadara demiri Piani 1981 has finally identified this widespread invasive taxon as being the common east American arcid Anadara transversa Say, 1822 (Albano et al. 2009).
The combination of multiple descriptors (genetics, shell morphology, anatomy, egg masses and ecological function) is probably the best approach thus far to try categorizing a species. As regards Mediterranean molluscan diversity, some points strictly linked to the foregoing discussion need to be explored further: the role and meaning of endemic, deep-sea molluscs and non-indigenous species.
Endemics
The Mediterranean marine fauna is considered to host a large number of endemic taxa which also include molluscs (Bianchi and Morri 2000; Coll et al. 2010, with references therein). To the best of our knowledge, no synoptic review dealing specifically with the Mediterranean endemic malacofauna as a whole has been published to date. This topic has been partly discussed by Gofas (1998) while treating the malacofauna around the Gibraltar Strait. Most of such endemics are distributed in shallow waters, while only a few have been recorded from bathyal depths (see below). Endemic molluscs total several tens of putative species, and belong largely to the Gastropoda (iconography in Cossignani and Ardovini 2011). A full discussion of the taxonomic status of all species to date only reported from the Mediterranean basin is beyond the scope of this paper. Here we prefer to limit ourselves to some emblematic cases. Perhaps the best known is Pinna nobilis Linnaeus 1758, the largest Mediterranean bivalve, considered an endangered species under strict protection (Council Directive 92/43/EEC 1992) since its populations appear in decline (Vicente 1990).
Up to the present, many ‘endemic’ species have been recognized as such based solely upon distinct morphological features of their shells. This is, for example, the case of the ‘historic’ endemic trochids Gibbula spratti (Forbes 1844) from the Aegean Sea and G. albida (Gmelin 1791) from the Adriatic (and in the Black Sea where it was originally described as G. euxinica Andrjewski 1937). The three ascertained extant species within Clathromangelia are non planktotrophic endemics of which C. granum is distributed in the central and eastern Mediterranean, while the other two (C. strigillata Pallary 1904 and C. loiselieri Oberling 1970) are localized in the Aegean-Levant and the Gabés basin (Oliverio 1995b). It equally applies to the many Rissoidae (especially taxa in the genus Alvania); Muricidae (e.g., Muricopsis cevikeri Houart 2000), Fasciolariidae (Fusinus spp. and others, most of which have been described in the last decades (i.e., Houart 2000; Buzzurro and Russo 2007)). Supplementary developmental and ecological information additional to shell morphology is provided only in some cases (e.g., Rissoa auriscalpium-R. italiensis: Russo and Patti 2005). The application of electrophoresis lends support to specific status of the bivalve Chamelea gallina (Linnaeus 1758) relative to its prevalently Atlantic couterpart C. striatula (da Costa 1778) making this abundant species Mediterranean endemics (Backeljau et al. 1994).
An interesting condition showing the operational relevance in systematics of integrating more descriptors is offered by one of the most classic of Mediterranean endemics, the sublittoral trochid Gibbula nivosa (A. Adams, 1851) only known from Malta (a rare case of punctiform geographic range: Evans et al. 2011), and considered as probably extinct (Schembri et al. 2007) until the recent finding of live populations (Evans et al. 2010). Although established on its peculiar shell morphology and coloration on-going molecular research provides full support for its specific status (Barco et al. 2013). Such an integrated approach would surely solve the many taxonomic problems of the presumed endemic fauna of semi-confined marine coastal settings in the Mediterranean Sea, like the Venice lagoon (home to for example the gastropods Calliostoma virescens Coen 1833 and Ocinebrina ingloria (Crosse 1865)), the Ganzirri and Pantano del Faro marine lakes near Messina (home to the gastropod Nassarius tinei (Maravigna 1840)), and above all the Gulf of Gabés, on the north African shore of the Mediterranean. The latter situation has been described as a potential species factory (Sabelli and Taviani 1980) because of the high proportion of apparently exclusive species inhabiting this Tunisian site (Ghisotti 1972a, b, c). The recent work by Cecalupo et al. (2008) overall lists 584 species, of which 33 are considered as endemics, that is to say about the 7 % a value similar to the 6 % suggested by Sabelli and Taviani (1980). These putative neoendemics all have a sibling species in the Mediterranean from which they likely derived.
The Mediterranean deep sea also includes some ascertained mollusc endemic molluscs, especially those associated with hydrocarbon/sulphide seepage sites (treated below).
An intriguing case of a very localized and putative endemic-rich site that merits to be studied by integrating available taxonomic techniques, are the volcanic “caves” along the Sicilian coast (Palazzi and Villari 2001) said to host 5 exclusive species, some not formally described (Puncturella picciridda, Monophorus sp., Muricopsis sp., Neolepton discriminatum, Lucinoma spelaeum). Another endemic gastropod (Skeneoides digeronimoi La Perna 1998) is reported from a volcanic ‘cave’ at Ustica island in the Tyrrhenian Sea (La Perna 1998b), and an arcid bivalve (Asperarca magdalenae) has been described from the Gymnasium calcareous cave off southeast Sicily and as a Pleistocene fossil also from Sicily (La Perna 1998a).
Most such endemic taxa established solely on their shell morphology are currently accepted as valid (WoRMS 2012). Clearly, the final acceptance of the specific status of morphologically different entities regarded as endemics to the Mediterranean, urgently requires a comparative and in-depth screening of their genetics.
The Deep Sea
In general, the deep-sea benthic fauna of the Mediterranean Sea (200–4,000 m: Danovaro et al. 2010) is considered to be an impoverished annex of the deep Atlantic Ocean, further characterized by a longitudinal gradient of decreasing biodiversity from the Strait of Gibraltar eastwards (Fredj and Laubier 1985; Bouchet and Taviani 1992a; Sardà et al. 2004; Tecchio et al. 2011). At present, the supposedly marked taxonomic depauperation of the Eastern Mediterranean vs the Western Mediterranean deep sea fauna has been re-dimensioned when considering prokaryota and eukarya all together (Danovaro et al. 2010) but still seems to hold true for megabenthos (Tecchio et al. 2011), and it also applies to molluscs which show a comparably higher diversity in the west, especially in the Alboran and Gibraltar areas (Bouchet and Taviani 1992a,b; Salas 1996), a trait shared with other taxonomic groups as well (Abelló et al. 2002; Arvanitidis et al. 2002; Danovaro et al. 2007).
Based upon deep-sea gastropod distribution Bouchet and Taviani (1992a) have proposed that at least part of the Mediterranean deep sea benthos derives from planktotrophic larval influxes from the Atlantic Ocean entering the Mediterranean via the surface Atlantic water (MAW = Modified Atlantic Water, Font et al. 1998), thus breaching the otherwise insurmountable Gibraltar sill. Relying upon empirical data showing a prominence of planktotrophic gastropods and concomitant eastward decrease in diversity, these authors hypothesize therefore the existence of pseudopopulations (i.e. not self-maintaining in the basin) in the Mediterranean deep-sea benthos. While this is a possible dispersal and settling mechanism for some species, as admitted by Bouchet and Taviani (1992a) this concept does not apply to the deep sea benthos as a whole. In fact, Sardà et al. (2004, with references therein) make the point that various benthic organisms in the deep Levantine basin consist of self-maintaining viable autochthonous populations (see also Koutsoubas et al. 2000 regarding more specifically molluscs). La Perna (2004) observes that the Mediterranean deep water fauna of protobranchiate bivalve is partly inherited from the past and therefore is a permanent and viable resident component, without excluding the potential presence of neo-endemics as a result on intrabasinal evolution. In this respect, only a few species of deep sea molluscs are at present considered endemic to the Mediterranean (i.e. Yoldiella ovulum, Yoldiella wareni, Ledella marisnostri: La Perna 2004). In fact, the most consistent number of endemics at bathyal depths pertain to specialized molluscs inhabiting chemosynthetic cold seep habitats. Many data on deep sea molluscs traditionally derive from sampling of soft-sediment habitats and accordingly mostly reflect life in the relatively monotonous muddy bathyal bottoms. In the recent years however, substantial improvements in submarine technology had led to the exploration of many habitats previously either poorly known or totally unsampled. A case in point are the deep water coral banks, canyon heads, seamounts and steep cliffs (e.g., Zibrowius and Taviani 2005; Freiwald et al. 2009; Mastrototaro et al. 2010). Research on such hard bottoms is steadily providing important information on remarkable and elusive molluscan life as well, including giant oysters (Gofas et al. 2007), large sessile bivalves (López-Correa et al. 2005) and rare gastropods (Taviani et al. 2009). Even more remarkable and productive in terms of upgrading our knowledge on deep-sea biodiversity is the on-going research on cold seep chemosynthetic habitats, such as mud volcanoes and pockmarks releasing hydrogen sulphide and hydrocarbons. These habitats are widespread in the Mediterranean deep sea and host an exclusive mollusc fauna largely made up of endemic species (Taviani 2004; Taviani et al. 2011a). These taxa include mainly bivalves, most of which associated with bacterial symbionts, e.g., “Solemya” sp., Idas modiolaeformis (Sturany 1896), Lucinoma kazani Salas and Woodside 2002, Myrtea amorpha (Sturany 1896), Thyasira striata (Sturany 1896), and subordinately gastropods, e,g. Lurifax vitreus Warén and Bouchet 2001, Clelandella myriamae Gofas 2004 (Corselli and Basso 1996; Salas and Woodside 2002; Gofas 2004; Olu-le Roy et al. 2004; Rodrigues et al. 2011; Taviani et al. 2011a; Chap. 5 by Taviani, this volume). It should be noticed that many of these Mediterranean endemics were described long ago (Sturany 1896) and have thus been included in the classic Mediterranean catalogues of molluscs. However, most of these species have yet to be studied genetically to ascertain their species status as distinct species compared to closely related Atlantic taxa.
Non-indigenous (Alien or Non-native) Species
The recent conquest of the Mediterranean Sea by an increasing armada of non-native species is an unprecedented event in the biogeography of the Mediterranean, because of the number of species involved (Zibrowius 1991; CIESM 2002; Leppäkoski et al. 2002; Zenetos et al. 2003; Dumont et al. 2004; Galil 2008; Zenetos et al. 2010; Occhipinti-Ambrogi et al. 2011; Chap. 22 by Goren, this volume; Chap. 27 by Galil, this volume). Probably no other large marine basin on Earth could claim to house in excess of 209 species of exotic origin, of which 109 established, as does the Mediterranean Sea (Zenetos et al. 2010). Some of these species are ecologically (and some economically: Garaventa et al. 2012) impacting the Mediterranean, locally assuming a key role in the marine costal ecosystems (Streftaris and Zenetos 2006; Galil 2007). The highest number of ascertained alien molluscs in the Mediterranean have come from the Red Sea via the Suez Canal and are so-called Lessepsian migrants (Gofas and Zenetos 2003). The latest estimate records 117 such species (Zenetos et al. 2010), all of them exclusively occurring in shallow water habitats (Chemello and Oliverio 1996). In addition to Lessepsians, other Indo-West Pacific immigrants have reached the Mediterranean possibly through ship-mediated transport as it has been postulated for the gastropod Cantharus tranquebaricus (Gmelin 1791) (Mienis 2003), and the strombid Conomurex persicus (Swainson 1821) (see Zibrowius 2002) sometimes uncorrectly cited as C. decorus (Röding 1798) (Moolenbeck and Dekker 1993). As for a few other tropical elements, C. persicus is spreading quite successfully in the eastern Mediterranean (Oliverio 1995a). Remarkably, it represents a renewed situation with Strombus s.l. forming part of the Mediterranean marine fauna as seen up to the Pliocene and once again in the last interglacial, providing an argument in favour of the concept of Godot basins (Taviani 2002; Oliverio and Taviani 2004). The arrival of Atlantic tropical aliens is on the contrary a rare phenomenon. An example is the gastropod Mitrella psilla (Duclos 1846) found in the Bay of Tunis and thought to have been accidentally introduced by shipping (Antit et al. 2010). Besides such tropical elements, many other molluscan taxa now form part of the Mediterranean fauna, often reaching the status of stable reproducing populations in the basin (Gofas and Zenetos 2003; Occhipinti-Ambrogi et al. 2011). Major dispersal pathways for such molluscs are deliberate, such as aquaculture (UNEP/MAP-RAC/SPA 2008), or unintentional anthropic introduction (CIESM 2002). This phenomenon is far from being concluded as new taxa are continuously introduced (i.a. Kabasakal et al. 2005; Louizidou et al. 2007; Ovalis and Zenetos 2007; Tzomos et al. 2011; Öztürk 2012), and some acclimatize themselves in the Mediterranean (Fabbri and Landi 1999; Keppel et al. 2012), while already-established non-native species steadily expand their range within the basin (e.g., Russo 1999; Russo and Mel 2002; Öztürk and Poutiers 2005; Zenetos et al. 2005; Çeviker and Albayrak 2006; Öztürk et al. 2007; Sarà et al. 2008; Tzomos et al. 2011; Crocetta 2011; Tsiakkiros and Zenetos 2011; Turk and Furlan 2011). Beside the case of Strombus discussed before, some non-tropical aliens appear to be particularly successful even while confronting the long-established autochthonous mollusc fauna. An emblematic example is presented by the arcid bivalve Anadara kagoshimensis (Tokunaga 1906), an invasive species mainly recorded in the Mediterranean literature as Scapharca cornea (Reeve 1844) or Anadara inaequivalvis (Bruguière 1789). First spotted in clean and well-sorted sands of fully marine coastal waters in the Adriatic Sea (e.g., Ghisotti 1973; Ghisotti and Rinaldi 1976) and in the Black Sea (Gomoiu 1981) surprisingly it soon successfully invaded very confined brackish and muddy waters in northern Adriatic lagoons (Poluzzi et al. 1981; Taviani et al. 1985; Poluzzi and Taviani 1986). Nowadays, it is a common and well-established faunal element of many coastal and lagoonal systems in the Mediterranean (e.g., Occhipinti-Ambrogi et al. 2011). A rather similar case is that of the Japanese clam Ruditapes philippinarum, originally introduced for aquaculture purposes in the 1980s (Bodoy et al. 1981; Lazzari and Rinaldi 1994; Breber 1985, 2002; Cesari and Pellizzato 1985), and expanding quite successfully in the Mediterranean from coastal marine settings to confined lagoons (Zamouri-Langar et al. 2006). Similar considerations could be applied to other non-tropical species (some of which are very invasive) such as the gastropods Crepidula fornicata (Linnaeus 1758) and Rapana venosa (Valenciennes 1846), and the bivalves Arcuatula senhousia (Benson in Cantor 1842), Limnoperna securis (Lamarck 1819), Crassostrea gigas (Thunberg 1793) and Mya arenaria (Linnaeus 1758) (Ghisotti 1974b; Stora 1976; Clanzig 1989; Cachia 1981; Madhioub and Zaouali 1988; Hoenselaar and Hoenselaar 1989; Zibrowius 1991; Blanchard 1997; Porcheddu et al. 1999; Zenetos et al. 2005; Crocetta 2011).
Biogeographic Considerations
Multiple factors influence the biogeography of marine benthos, including historical legacy, physical barriers and seaways, oceanographic vectors (currents), and dispersal mechanisms. The Mediterranean Sea is a distinct province included in the Temperate Northern Atlantic realm (Spalding et al. 2007). The overall biogeographic complexity of this region is evident when considering that the Mediterranean province itself is subdivided into as many as seven ecoregions that are west to east: Alboran Sea, Western Mediterranean, Ionian Sea, Tunisian Plateau/Gulf of Sidra, Adriatic Sea, Levantine Sea, Aegean Sea (Spalding et al. 2007).
As a consequence, the total benthic molluscan stock inhabiting the Mediterranean is unevenly distributed in the basin (see Table 16.2). It should be said that the geographic range of individual molluscan taxa only seldom, if ever, strictly coincides with the boundary of any ecoregion. In fact, there is a substantial number of species whose range is almost basinwide, although each region seems characterized by its own peculiarities.
To provide examples, the Alboran Sea, a region much influenced by the Atlantic fauna, contains many species that are localized or endemic in that general area, some expanding on both sides (Atlantic and Mediterranean) of the Gibraltar Strait (Gofas 1998; Gofas et al. 2011a, b). This area is to date one of the best known in the entire Mediterranean Sea. It has been receiving attention from malacologists beginning from the pioneering works of Pallary (1902, 1920). Van Aartsen et al. (1984) list around 380 species, an estimate substantially upgraded by Peñas et al. (2006) who listed 603 species found around the island of Alboran. Finally, the recent monographs by Gofas et al. (2011a, b) provide a synoptic and critical overview of all molluscs known from Andalusia which is largely located in the Alboran Sea ecoregion. Some 30 molluscs inhabit a very restricted geographic range (1 to 2° in latitude) centred on the Strait of Gibraltar and could be considered endemics to this area (Gofas 1998; Peñas et al. 2006). Besides, about 40 tropical Atlantic species have spread into the Alboran Sea but decrease in number along a west to east gradient with about 25 reaching up to Algeria and five into the westernmost part of the Mediterranean (Gofas and Zenetos 2003). As much as 11 % of the Alboran mollusc fauna appears to be exclusive of this Mediterranean ecozone and 4 % are endemic.
The far distant northern Adriatic hosts some remarkable endemics as well, as described above (e.g. Calliostoma virescens Coen 1933), or species that are preferentially distributed in that sector of the basin influenced by cooler waters and reduced salinity, like Gibbula albida (Gmelin 1791), Gibbula adriatica (Philippi 1844), and Littorina saxatilis (Olivi 1792). Furthermore, Alvania schwartziana Brusina 1866, Trivia multilirata (Sowerby G. B. II 1870) and Ocinebrina ingloria (Crosse 1865) also appear to be limited to this sub-basin. It is noteworthy that all these species, apart from T. multilirata, are non planktotrophic or are direct developers like L. saxatilis. This latter presents a puzzling disjunct distribution (Atlantic Ocean and Venice lagoon: Cesari 1994). However, biomolecular evidence seems to support a recent, possibly anthropic northern Adriatic introduction in historical times (Panova et al. 2011).
Noticeably, some warm temperate species are modally distributed along the south-western Mediterranean coasts, although reaching up to eastern Sicilian waters (e.g. Bivetiella cancellata (Linnaeus 1767)) or Sardinia and the Tuscan Archipelago (e.g. Patella ferruginea). On the other hand, cold temperate species, possibly leftovers from the last glacial times, display a localized Mediterranean distribution, such as, Pseudamussium peslutrae Linnaeus 1771 (=P. septemradiatum (Müller 1776)), in the deep Alboran Sea (Salas 1996), and Buccinum humphreysianum Bennet 1824, extending as north as the Gulf of Lion (Casamor and Ghisotti 1968).
One of the leading physical factors governing the distribution of molluscs in the Mediterranean, and therefore their biogeography, is obviously hydrography. Firstly, on the basinal scale the current regime (summarized in Font et al. 1998) is offering the avenues for dispersal. Secondly, seawater attributes such as salinity and temperature set additional constraints on the successful establishment of any given taxon. Salinity for instance selects the marine benthic molluscs capable of living within euryaline seawater bodies such as lagoons. Brackish lagoons do not seem to host endemic species but molluscan assemblages nevertheless differ biogeographically inside the Mediterranean basin (Bedulli and Sabelli 1990). Temperature may act to impede or favour the dispersal of taxa within the basin even in the face of propitious current directions, as seen in the sub-tropical non-native species of the Levantine basin.
Biologically, the type of development and the length of larval life are of paramount importance in determining the biodiversity achievable within the Mediterranean Sea. A few papers address specifically the issue of larval life length related to the distribution of molluscs (Bouchet 1981, 1989; Oliverio 1996). The latter compares the larval strategies of gastropods found in Posidonia beds along a west–east transect from Spain to Levantine Turkey observing an increase in the ratio of non planktotrophic to planktotrophic species. This trend is interpreted as being directly dependent on the evolution of different larval developmental pathways.
The developmental strategy in Mollusca is so relevant to the biogeography and biodiversity of the Mediterranean as to deserve to be treated in some detail.
Developmental Strategies and Their Biogeographic Consequences
All marine molluscs develop inside an egg envelope in a first larval form (known as pericalymma in protobranch bivalves), and in a trochophore-like larva in gastropods and non-protobranch bivalves (Page 2009). The following larval stage, the veliger, is in most cases a swimming one, a strategy enabling in principle a widespread dispersion in benthic species of reduced vagility. Veligers in various species have different lifespans ranging from a few hours to a week in lecithotrophic larvae, and from one to several weeks or even years in planktotrophic larvae (Strathmann and Strathmann 2007). The shell keeps memory of the type of development. Thus, the occurrence of only one or two kinds of protoconch (prodissoconch in bivalves) in shells is generally informative about direct/lecithotrophic, or planktotrophic development. The planktotrophic larval stage is securing a dispersal over long distances as well documented for instance by amphiatlantic species among deep water gastropods (e.g. Bouchet and Warén 1980, 1985; Taviani and Taviani 1986).
Obviously, great caution should be applied when trying inferring the actual dispersal capability of any given species, based solely upon indirect information as larval shell morphology. In fact, the effectiveness of dispersal is constrained primarily by the requirements of the metamorphosing larva. It seems that the more a veliger swims away from the parental habitat, the more difficult is for them finding a suitable place for metamorphosis. Remarkably, a wide geographic area characterized by little if any habitat discontinuity in its extension has been documented to be settled successfully by “direct” developers or short swimming larvae in just a few generations. Examples of such are provided by Foighil (1989) for the bivalve genus Lasea, and by Scheltema and Williams (2009) for the family Nuculidae. These observations support the “paradox of Rockall”, an island in the northeastern Atlantic whose littoral zone is uniquely inhabited by benthic invertebrates lacking a planktonic larva (Johanesson 1988). In this island, only the brooding Littorina saxatilis (Olivi 1792) is present while the planktonic L. littorea Linnaeus 1758, is absent, in spite that they normally co-exist along both the western and eastern continental coasts.
Regarding constraints to dispersion, another case of some interest is presented by the gastropod Cerithium scabridum Philippi 1848, an alien species firstly reported from the Levantine basin about 140 years ago (Keller 1883). This locally invasive species possesses a planktotrophic larval phase lasting 45–60 days (Ayal and Safriel 1982) in principle providing a potential wide dispersal capability. Adults exhibit unspecialized feeding habits, occurring on all kind of substrata in intertidal or shallow water of fully marine and brackish habitats and displaying a high level of genetic variability (Lavie and Nevo 1986). All together such characteristics should enable an almost unlimited distribution within the Mediterranean but this does not seem to be the case. In fact, Cerithium scabridum exhibits a continuous distribution within the Levantine basin but a rather patchy distribution in the southern Mediterranean with local populations often rich in individuals. To account for this spotty distribution, Gofas and Zenetos (2003) hypothesized that this species was likely introduced by shipping, first in Sicily and later in Tunisia. Recent findings of dead shells within harbours at Vibo Valentia, Tyrrhenian Sea (Crocetta et al. 2009) and of a living population in Otranto, Apulia (Albano and Trono 2008) lend support to such a hypothesis.
The swimming or buoyancy capability of larvae and their resulting passive drift with currents is not the only mode of dispersal. Molluscs lacking a planktonic larval stage can be dispersed by mucous threads (Martel and Chia 1991), as propagules: young crawling stages or even sexually mature adults in the case of small sized species (e.g. Sullivan 1948; Bradley and Cooke 1959; Sellmer 1967 for bivalves and Newel1 1964; Anderson 1971; Little and Nix 1976; Levinton 1979 for gastropods). A peculiar dispersal mechanism has been described in Mytilus edulis Linnaeus 1758, whose post-larvae are able to remain pelagic by secreting a gas bubble into the mantle cavity (Nelson 1925). Tidal currents spread young specimens sometimes over tens of kilometers (Beukema and Vlas 1989). Noticeably, a basic circadian periodicity in migratory rhythms of drifting juvenile molluscs has been observed (Armonies 1992). Frequent drifting excursions enhance rafting opportunities thus favouring long distance dispersal (Martel and Chia 1991). Floating material (plastic debris, algae, logs and other) is usually concentrated by wind and currents into drift lines that can then travel hundred kilometers (Birkeland 1971), becoming available to floating animals within a few hours or days (Highsmith 1985). This mechanism can be more effcient than normally envisaged. A high proportion of widely distributed Antarctic benthic invertebrate taxa brood or produce egg capsules from which benthic juveniles hatch, preventing in principle a large diffusion: On the contrary, many such taxa are widely distributed so that it has been envisaged that algal rafting likely plays a role as dispersal mechanism (Dell 1972; Simpson 1977; Pearse 1979; Picken 1980). Furthermore, Dell (1972) posits that a brooding female is a better colonist than a larva.
The classic passive transport by wading birds (Rees 1965; Green and Figuerola 2005) or by other migrant marine animals like turtles (Oliverio et al. 1992), is by now evolved to include humans as a significant dispersal mechanism of overwhelming importance.
About 15 % of the exotic molluscs currently found in the Mediterranean basin have been introduced by shipping/ballast waters or aquaculture, and for three species, i.e. Gibbula albida, Siphonaria pectinata (Linnaeus 1758) and Perna perna (Linnaeus 1758), an intra-Mediterranean transfer has been reported (Gofas and Zenetos 2003). Moreover, this kind of transfer is also hypothesized for other alien species: Cerithium scabridum, Rapana venosa (Valenciennes 1846) and Fulvia fragilis (Forskål in Niehbur 1775) (Crocetta et al. 2008).
If dispersal mechanism represents the main source of construction of the past and present Mediterranean biodiversity, ecology constitutes a constraint to the diversity and distribution of molluscs mostly affecting larval settlement and metamorphosis or juvenile life. As shown by Zenetos (1997) there is a substantial uniformity in the diversity of marine Bivalvia in a geographic area as large as Greece, but ecological patterns, such as depth and degree of confinement, nevertheless play here a major role in the distribution of individual species. Under this respect, Table 16.3 presents a comparison among shelled gastropods, bivalves and their ratio in several Mediterranean localities with different attributes. The role of ecological factors in shaping distribution of marine organisms is well exemplified by the noteworthy transformation of bottom communities in the Adriatic Sea. In about 50 years the variation in sedimentation rate, organic load and flocculation resulted in the substitution of the Turritella community described by Vatova (1949) with a Corbula-Amphiura filiformis community (Scaccini 1967; Schiaparelli et al. 2000; Chiantore et al. 2001), with rarefaction and possible local extinction of some of the species associated with the former. Another striking case is oyster reefs (Ostrea edulis Linnaeus 1758) much more abundant in the Mediterranean coastal areas in the recent past and at present in strong recession with few exceptions (Airoldi and Beck 2007).
Threats to the Mediterranean Mollusc Diversity
There is a widespread perception that in the current Anthropocene (sensu Crutzen and Stoermer 2000) the marine biodiversity is under attack on a global scale often because of direct or indirect human actions (Gray 1997; Hoegh-Guldberg and Bruno 2010). Among the main causes often considered by scientists as serious threats to marine biodiversity at all levels from species to ecosystems we can list: (1) global warming, (2) ocean acidification, (3) habitat loss, (4) pollution, and (5) overfishing (Gray 1997; Sala and Knowlton 2006). In this scenario, the Mediterranean is not exception (Airoldi and Beck 2007; Claudet and Fraschetti 2010; UNEP 2010; Coll et al. 2011).
Global Warming
The present trend of rising temperature as a result of increasing levels of greenhouse gases in the atmosphere is generating a growing concern about its pernicious impact on marine ecosystems. According to projections, global warming may potentially depressing habitat complexity, rearranging trophic webs, fostering new ecological assemblages, favouring exotic newcomers, and introducing diseases (Hoegh-Guldberg and Bruno 2010). In the Mediterranean, an increase of seawater temperature is already causing visible alterations in the marine biota (Danovaro et al. 2004; Peirano et al. 2005; Bianchi 2007; CIESM 2008; Parravicini et al. 2009; Por 2009; Coll et al. 2010; Faimali and Taviani 2011; Chap. 17 by Albano, this volume). The macroscopic evidence regarding molluscs is surely the amazing increase in the number of exotic species, both Lessepsians or accidentally-introduced, steadily added to the autochtonous molluscs fauna of the basin and by large represented by warm-affinity organisms (Zibrowius 1991; Galil 2008, 2009; Occhipinti-Ambrogi et al. 2011). Although such species-increment appears as the most evident outcome of tropicalization, it is the influence of rising seawater temperature on Mediterranean habitats as a whole that deserves attention. In fact, many important habitats, as for example seagrass beds house various exclusive or preferentially associating autochtonous molluscs (e.g., Pérès and Picard 1964; Terlizzi and Russo 1995; Beqiraj et al. 2008; Gofas et al. 2011a, b; Albano and Sabelli 2012) that may respond to thermal variations by reduction, displacement or eradication.
Ocean Acidification
Ocean acidification (OA), induced by uptake by seawater of anthropogenic CO2, might adversely impact the calcification potential of carbonate shelled organisms (Orr et al. 2005), and have important repercussions on other physiological functions and ultimately on the trophic web (Fabry et al. 2008; Rossoll et al. 2012; Seibel et al. 2012). Therefore, OA is clearly a potential factor controlling the Mediterranean mollusc fauna. Aragonitic holoplanktonic species for instance, such as thecosomatous pteropods, are supposedly at risk from increasingly acidified water masses (Comeau et al. 2009, 2012). Tank experiments on larvae of the Mediterranean Cavolinia inflexa (Leseur 1813) exposed to decreasing seawater pH documented a strong reduction in shell secretion (Comeau et al. 2010). Moreover, ocean acidification has been shown to alter other metabolic processes beside calcification. However, in face of such negative scenarios, a recent metabolic study on Pacific pteropods which include also taxa represented in the present-day Mediterranean fauna (Hyalocylis striata (Rang 1828), Clio pyramidata Linnaeus 1767, Diacavolinia longirostris (Blainville 1821), and Creseis virgula Rang 1828) seem to demonstrate an individual resilience to augmented acidification (Maas et al. 2012), as also other calcifiers and non-calcifying organisms (Nielsen et al. 2010; McCulloch et al. 2012a, b). Regarding benthic molluscs, data from naturally acidified waters do not straightforwardly lend support that they are detrimental to the calcification capacity of Mediterranean shelled molluscs (Rodolfo-Metalpa et al. 2011). Much still needs to be learned about the mid and long term effects of OA on organisms with carbonate skeletons, but it is unlikely that it could be considered, on the short term at least, as a serious threat to Mediterranean molluscs
Habitat Loss
Habitat degradation, and in some cases complete loss, induced by human activity is for sure one of the most serious, if not the most important, menaces to marine biodiversity (Airoldi and Beck 2007; Coll et al. 2010). Human-induced habitat fragmentation is documented for the important endemic habitat-forming Posidonia oceanica that already suffers from competition by invasive species (Peirano et al. 2005; Airoldi and Beck 2007; Montefalcone et al. 2010a, b). It is likely that the decline and the possible loss of local stands of this seagrass might result in the rarefaction or even extinction of those molluscan taxa associated with it, which are often restricted to specific geographic sectors of the Mediterranean Sea. Fishing malpractices (including overfishing) can, in some cases, be considered as causing habitat loss through inducing sensitive changes both directly in the ecological structure of biological communities (species removal, trophic web disruption, etc.) and indirectly in the physical environment (Coll et al. 2010). For instance, the over-trawling of sectors of the Adriatic continental shelf is stressing the exploitable (as well as the co-occurring non-commercial) living stocks (e.g., Chamelea gallina) with local decline in populations inhabiting soft sediments, as well as impacting the recruitment through unnatural stirring and ploughing of the sediment (Romanelli et al. 2009). Another case in point centred on molluscs is the now-illegal collection through rock-hammering or rock blasting of edible boring molluscs (such as the date-mussel Lithophaga lithophaga Linnaeus 1758) that has caused considerable and long-lasting ecological damage on limestone coasts (Russo and Cicogna 1991; Fanelli et al. 1994; Bianchi and Morri 2000; Fraschetti et al. 2001; Guidetti and Boero 2004; Devescovi et al. 2005; Rovere et al. 2009).
Pollution
Habitat degradation due to pollution is another factor adversely impacting marine ecosystems from coastal lagoons down to bathyal depths (WWF/IUCN 2004; Coll et al. 2010). It encompasses a variety of phenomena, from the release at sea of dangerous and toxic substances to oil spills and solid waste dumping (Valavanidis and Vlachogianni 2011 with references therein). At the species level, chemicals may interfere with body functions generally in a negative way (including lethality) and cause a decrease in the viability of organisms due to histo-pathological alterations in their organs, chromosomal aberrations and breaks, DNA damage (Bresler et al. 1999, 2003), or the biochemistry of the gonads during the spawning phase (Hamdani and Soltani-Mazouni 2011). Biotoxins and heavy metals may cause physiological stress in bivalves, as seen, for example, in Mytilus galloprovincialis Lamarck 1819, from the Albanian coast (Sadikaj et al. 2009). At least one organotin pollutant, tributyltin (TBT), has been documented to act directly on molluscs eventually driving a decline in populations (e.g. the muricid Stramonita haemastoma Linnaeus 1767, Terlizzi et al. 2001). TBT is a xenobiotic used (but now banned in many countries) as a biocide in marine antifouling paints and wood preservatives; it is recognized as inducing an alteration in the reproductive system of prosobranch gastropods known as imposex (Braber 1970). Imposex has been demonstrated in several Atlanto-Mediterranean species including several muricids and nassariids (Gibbs et al. 1990; Oehlmann et al. 1996, 1998; Garaventa et al. 2007; Lemghich and Benajiba 2007; Wirzinger et al. 2007; Rodríguez et al. 2009). As clearly demonstrated by Oehlmann et al. (1996) in Ocinebrina aciculata (Lamarck 1822), TBT not only induces the development of an additional male sexual organs (mainly the penis and/or vas deferens) in females, sterilization and sex change, but also damages of capsule gland (inhibition of breeding) and the occlusion of vulva by vas deferens tissue (a barrier to copulation) (Bryan et al. 1986) even if recent findings about TBT effect, pointed out that, at least in Hexaplex trunculus Linnaeus 1758, it could be not the only imposex cue (Garaventa et al. 2006). Sewage related pollution has been documented to negatively affect rocky shore assemblages (Terlizzi et al. 2002), including the structuring of molluscan assemblages (Terlizzi et al. 2005). Changes in soft bottom molluscan assemblages has been also quantified as a results of offshore gas platforms (Terlizzi et al. 2008). There is however no strong evidence that pollution is really threatening extinction of any Mediterranean mollusc, although some concern exists for the endangered Patella ferruginea as, for example, in the populations in the Ceuta harbour (Espinosa et al. 2007).
Final Remarks
The present Mediterranean diversity is transient. Biodiversity per se is in a sense ephemeral, and the durability and resilience of its components are accordingly transitory. As the natural historical evolution of our planet teaches, every diversity of every single place at any particular time is permanently liable to be partly or totally jeopardized by a variety of factors thus vanishing, evolving or adapting. The present (mollusc) marine diversity is thus only the result of a collection of evolving diversities without solution of continuity accompanying the highly complex history of this basin.
The Mediterranean is by now in an ultra-dynamic and accelerated situation of aggrading diversity especially in response to an alien expansion that has no equal in its geological past. But is this present trend going to set the pace for this basin on a longer time scale? The answer is probably negative as soon as we consider the global paleoclimatic behaviour as understood to date. In fact, according to the theory that cyclity of Quaternary ice ages is astronomically tuned by precessional forcing at ca. 11 kyrs periodicity, the current interglacial (Holocene or Marine Isotope Stage 1) should by now be approaching its end (Kukla et al. 1972). Because of the uncertainties linked to increasing atmospheric carbon dioxide concentrations, however, this termination could be deferred by many centuries (Tzedakis et al. 2012). At some time in the future, however, a new ice age will impose a drastic climatic deterioration over the Earth which in turn will in all likeliness dictate once again the typology of marine ecosystems of the Mediterranean basin. The expected depression of the temperature will seal the fate of the hundreds of Lessepsian and other ‘warm water’ alien species presently inhabiting the Mediterranean and causing so much concern at present (Oliverio and Taviani 2004). Thus, the evocative prophecy by Por (2009) of a Mediterranean Sea returning to the warm days of Tethys times might in fact collide with a major change in climatic direction governed by astronomical causes. In such a scenario temperate invasive species could in principle be spared by climatic deteriorations as those routinely experienced by the Mediterranean in the Pleistocene.
Bibliography
Abelló P, Carbonell A, Torres P (2002) Biogeography of epibenthic crustaceans on the shelf and upper slope of the Iberian Peninsula Mediterranean coasts: implications for the establishment of natural management areas. Sci Mar 66(Suppl 2):183–198
Airoldi L, Beck MW (2007) Loss, status and trends for coastal marine habitats of Europe. Oceanogr Mar Biol Annu Rev 45:345–405
Albano PG, Sabelli B (2012) The molluscan assemblages inhabiting the leaves and rhizomes of a deep water Posidonia oceanica settlement in the central Tyrrhenian Sea. Sci Mar 76(4):721–732
Albano PG, Trono D (2008) Record of the alien species Cerithium scabridum Philippi, 1848 (Gastropoda: Cerithiidae) from Otranto, southern Adriatic Sea. Boll Malcol 44:1–4
Albano PG, Rinaldi E, Evangelisti F, Kuan M, Sabelli B (2009) On the identity and origin of Anadara demiri (Bivalvia: Arcidae). J Mar Biol Assoc UK 89:1289–1298
Anderson A (1971) Intertidal activity, breeding and the floating habit of Hydrobia ulvae in the Ythan estuary. J Mar Biol Assoc UK 51:423–437
Antit M, Gofas S, Azzouna A (2010) A gastropod from the tropical Atlantic becomes an established alien in the Mediterranean. Biol Invasions 12:991–994
Antonioli F, Deino A, Ferranti L, Keller J, Marabini S, Mastronuzzi G, Negri A, Piva A, Vai GB, Vigliotti L (2008) Lo studio della sezione “Il Fronte” per la definizione del Piano Tarantiano (Puglia, Italy). Il Quat 20:31–34
Armonies W (1992) Migratory rhythms of drifting juvenile molluscs in tidal waters of the Wadden Sea. Mar Ecol Prog Ser 83:197–206
Arvanitidis C, Bellan G, Drakopoulos P, Valavanis V, Dounas C, Koukouras A, Eleftheriou A (2002) Seascape biodiversity patterns along the Mediterranean and the Black sea: lessons from the biogeography of benthic polychaetes. Mar Ecol Prog Ser 244:139–152
Ayal Y, Safriel UN (1982) r-curves and the cost of the planktonic stage. Am Nat 119:391–401
Backeljau T, Bouchet P, Gofas S, de Bruyn L (1994) Genetic variation, systematics and distribution of the venerid clam Chamelea gallina. J Mar Biol Assoc UK 74:211–223
Barco A, Evans J, Schembri PJ, Taviani M, Oliverio M (2013) Testing the applicability of DNA barcoding for mediterranean species of top-shells (Gastropoda, Trochidae, Gibbula s.l.). Mar Biol Res 9:785–793
Barash A, Danin Z (1992) Fauna Palestina. Mollusca I. annotated list of Mediterranean molluscs of Israel and Sinai. The Israel Academy of Sciences and Humanities, Jerusalem
Bedulli D, Sabelli B (1990) E’ possibile una zoogeografia delle lagune mediterranee attraverso la distribuzione dei Molluschi? Oebalia 16(Suppl):133–141
Bellomo E (1993) Presenza di “Ospiti boreali” (Mollusca, Gastropoda) nel Pleistocene di Contrada Case Alte (Villa S. Giovanni). Boll Soc Paleontol Ital 32:393–399
Bellomo E (1996) Predation traces on specimens of Polinices lacteus (Guilding, 1831), (Gastropoda, Prosobranchia, Naticidae) from last interglacial assemblage of Bovetto (Reggio Calabria, Southern Italy). Boll Soc Paleontol Ital 3(Spec):9–17
Beqiraj S, Kashta L, Kuci M, Rasemi D, Mato X, Gace A (2008) Benthic macrofauna of Posidonia oceanica meadows in the Albanian coast. Nat Montenegr 7:55–69
Beukema JI, de Vlas J (1989) Tidal-current transport of thread-drifting postlarval juveniles of the bivalve Macoma balthica from the Wadden Sea to the North Sea. Mar Ecol Prog Ser 52:193–200
Bianchi CN (2007) Biodiversity issues for the forthcoming tropical Mediterranean Sea. Hydrobiologia 580:7–12
Bianchi CN, Morri C (2000) Marine biodiversity of the Mediterranean Sea: situation, problems and prospects for future research. Mar Pollut Bull 40:367–376
Birkeland C (1971) Biological observations on Cobb Seamount. Northwest Sci 45:193–199
Blanchard M (1997) Spread of the slipper limpet Crepidula fornicata (L. 1758) in Europe. Current state and consequences. Sci Mar 61(Suppl 2):109–118
Bodoy A, Maître-Allain T, Riva A (1981) Croissance comparée de la palourde européenne Ruditapes decussatus et de la palourde japonaise Ruditapes philippinarum dans un écosystème artificiel méditerranéen. Vie Mar 2:39–51
Boisselier-Dubayle MC, Gofas S (1999) Genetic relationships between marine and marginal marine populations of Cerithium species from the Mediterranean Sea. Mar Biol 135:671–682
Bonfitto A, Oliverio M, Sabelli B, Taviani M (1994) A quaternary deep-sea marine molluscan assemblage from East Sardinia (Western Tyrrhenian Sea). Boll Malacol 11:305–309
Bonfitto A, Dell’Angelo B, Evangelisti F, Sabelli B (2011) The genus Acanthochitona (Mollusca: Polyplacophora) in the Mediterranean Sea: morphological and molecular data. Sci Mar 75:171–180
Bouchet P (1981) Evolution of larval development in Eastern Atlantic Terebridae (Gastropoda), Neogene to Recent. Malacologia 21:363–369
Bouchet P (1984) Les Triphoridae de Mediterranée et du proche Atlantique (Mollusca, Gastropoda). Lav Soc Ital Malacol 21:5–58
Bouchet P (1989) A review of poecilogony in Gastropods. J Molluscan Stud 55:67–78
Bouchet P (1995) Nouvelles observations sur la systematique des Triphoridae de Mediterranée et du proche Atlantique. Boll Malacol 31:205–220
Bouchet P, Guillemot H (1978) The Triphora perversa-complex in Western Europe. J Molluscan Stud 44:344–356
Bouchet P, Taviani M (1989) Atlantic deep-sea gastropods in the Mediterranean: new findings. Boll Malacol 2:137–148
Bouchet P, Taviani M (1992a) The Mediterranean deep-sea fauna: pseudopopulations of Atlantic species? Deep Sea Res Part I Oceanogr Res Pap 39:169–184
Bouchet P, Taviani M (1992b) La colonizzazione dei bacini a soglia. Il caso del Mar Mediterraneo. Lav Soc Malacol Ital 24:120–130
Bouchet P, Warén A (1980) Revision of the North-East Atlantic bathyal and abyssal Turridae (Mollusca: Gastropoda). J Molluscan Stud 8(Suppl):1–119
Bouchet P, Warén A (1985) Revision of the northeast Atlantic bathyal and abyssal neogastropoda (excluding Turridae). Boll Malacol 2(Suppl):123–296
Braber SJM (1970) The occurrence of a penis-like outgrowth behind the right tentacle in spent females of Nucella lapillus (L). Proc Malacol Soc Lond 39:231–233
Bradley WH, Cooke P (1959) Living and ancient populations of the clam Gemma gemma in a Maine coast tidal flat. Fish Bull 137:305–334
Breber P (1985) L’introduzione e l’allevamento in Italia dell’arsella del Pacifico Tapes decussatus, Reeve (Bivalvia, Veneridae). Oebalia 9:675–680
Breber P (2002) Introduction and acclimatisation of the Pacific carpet clam, Tapes philippinarum, to Italian waters. In: Leppäkoski E, Gollasch S, Olenin S (eds) Invasive aquatic species of Europe – distribution, impact and management. Kluwer, Dordrecht
Bresler V, Bissinger V, Abelson A, Dizer H, Sturm A, Kratke R, Fishelson L, Hansen P-D (1999) Marine molluscs and fish as biomarkers of pollution stress in littoral regions of the Red Sea, Mediterranean Sea and North Sea. Helgoland Mar Res 53:218–243
Bresler V, Abelson A, Fishelson L, Feldstein T, Rosenfeld M, Mokady O (2003) Marine molluscs in environmental monitoring. I. Cellular and molecular responses. Helgoland Mar Res 57:157–165
Bryan GW, Gibbs PE, Hummerstone LG, Burt GR (1986) The decline of the gastropod Nucella lapillus around South-West England: evidence for the effect of trybutiltin from antifouling paints. J Mar Biol Assoc UK 66:611–640
Buzzurro G, Russo P (2007) Fusinus del mediterraneo – Mediterranean Fusinus. Grafiche ATA, Milano
Cachia C (1981) Notes on some uncommon species of molluscs from the Maltese islands. Boll Malacol 17:291–294
Cachia C, Mifsud C, Sammut PM (2004) The marine mollusca of the Maltese Islands. Part four. The classes: Caudofoveata, Solenogastres, Bivalvia, Scaphopoda and Cephalopoda. Backhuys, Leiden
Calvo M, Templado J, Oliverio M, Machordom A (2009) Hidden Mediterranean biodiversity: molecular evidence for a cryptic species complex within the reef building vermetid gastropod Dendropoma petraeum (Mollusca: Caenogastropoda). Biol J Linn Soc 96:898–912
Carnevale G, Longinelli A, Caputo D, Barbieri M, Landini W (2008) Did the Mediterranean marine reflooding preceded the Mio-Pliocene boundary? Paleontological and geochemical evidence from upper Messinian sequences of Tuscany, Italy. Paleogeogr Paleoclimatol Paleoecol 257:81–105
Carvajal-Rodriguez A, Conde-Padin P, Rolan-Alvarez E (2005) Decomposing shell form into size and shape by geometric morphometric methods in two sympatric ecotypes of Littorina saxatilis. J Molluscan Stud 71:313–318
Carvajal-Rodriguez A, Guerra-Varela J, Fernandez B, Rolan E, Rolan-Alvarez E (2006) An example of application of geometric morphometric tools to the morphological diagnosis of two sibling species in Nassarius (Mollusca, Prosobranchia). Iberus 24:81–88
Casamor L, Ghisotti F (1968) Buccinum humphreysianum Bennet, 1825. Schede malacologiche del Mediterraneo. Toffoletto, Milano
Casellato S, Stefanon A (2008) Coralligenous habitat in the northern Adriatic Sea: an overview. Mar Ecol 29:321–341
Cattaneo Vietti R, Giovine F (2008) Opisthobranchia. Biol Mar Med 15(Suppl):279–295
Cecalupo A, Buzzurro G, Mariani M (2008) Contributo alla conoscenza della malacofauna del Golfo di Gabès (Tunisia). Quad Civ Staz Idrobiol Milano 31:1–175
Ceregato A, Raffi S, Scarponi D (2007) The circalittoral/bathyal paleocommunities in the Middle Pliocene of Northern Italy: the case of the Korobkovia oblonga–Jupiteria concava paleocommunity type. Geobios 40:555–572
Cesari P (1994) I Molluschi della Laguna di Venezia. Arsenale, Venezia
Cesari P, Mizzan L (1994) Dati sulla malacofauna marina costiera del Veneziano. Boll Mus Civ St Nat Venezia 43:179–190
Cesari P, Pellizzato M (1985) Molluschi pervenuti in laguna di Venezia per apporti volontari o casuali. Acclimazione di Saccostrea commercialis (Iredale & Roughely, 1933) e di Tapes philippinarum (Adams & Revve, 1850). Boll Malacol 21:237–274
Çeviker D, Albayrak S (2006) Three alien molluscs from Iskenderun Bay (SE Turkey). Aquat Invasions 1:76–79
Chemello R, Oliverio M (1996) Lessepsian migrations: a theoretical “island-jumping” model. Biol Mar Med 3:444–446
Chester CM (1996) The effect of adult nutrition on the reproduction and development of the estuarine nudibranch, Tenellia adspersa (Nordmann, 1845). J Exp Mar Biol Ecol 198:113–130
Chiantore M, Bedulli D, Cattaneo-Vietti R, Schiaparelli S, Albertelli G (2001) Long-term changes in the Mollusc-Echinoderm assemblages in the north and coastal middle Adriatic Sea. Atti Ass Ital Oceanogr Limn 14:63–75
CIESM (2002) Alien marine organisms introduced by ships in the Mediterranean and Black seas. CIESM, Monaco
CIESM (2008) The Messinian salinity crisis from mega-deposits to microbiology. In: Briand F (ed) A consensus report. CIESM, Monaco
CIESM (2012) Atlas of exotic species in the Mediterranean Sea. http://www.ciesm.org/online/atlas/index.htm. Accessed 27 Sept 2012
Clanzig S (1989) Introduction récente d’invertébré dans les lagunes Mediterranéennes du Languedoc-Roussilon (France). Bull Soc Zool Fr 114:151–152
Claudet J, Fraschetti S (2010) Human-driven impacts on marine habitats: a regional m eta-analysis in the Mediterranean Sea. Biol Conserv 143:2195–2206
Coen G (1937) Nuovo saggio di una Sylloge Molluscorum Adriaticorum. Mem Regio Com Talass Ital 240:1–173
Coen G, Vatova A (1932) Malacofauna Arupinensis. Thalassia 1:1–53
Colantoni P (1973) A glacial mollusc fauna from Baronie seamount (off Eastern Sardinia). Rapp Comm Int Mer Médit 21:897–900
Colantoni P, Noto P, Taviani M (1975) Prime datazioni assolute di una fauna fossile a Pseudamussium septemradiatum dragata nel basso Adriatico. G Geol 40(Supplt 3):133–140
Coll M, Piroddi C, Steenbeek J, Kashner K, Lasram FBR, Aguzzi J, Ballesteros E, Bianchi CN, Corbera J, Dailinis T, Danovaro R, Estrada M, Froglia C, Galil BS, Gasol JM, Gerwagen R, Gil J, Guilhaumon F, Kesner-Rayes K, Kitsos MS, Koukouras A, Lampradariou N, Laxamana E, Lopez Fe de la Cuadra CM, Lotze HK, Martin D, Mouillot D, Oro D, Raichevich S, Rius-Barile J, Saiz-Salinas JI, San Vincente C, Somot S, Templado J, Turon X, Vafidis D, Villanueva R, Voultsiadou E (2010) The biodiversity of the Mediterranean Sea: estimates, patterns, and threats. PLoS One 5:1–36
Coll M, Piroddi C, Albouy C, Lasram FBR, Cheung WWL, Christensen V, Karpouzi VS, Guilhaumon F, Mouillot D, Paleczny M, Palomares ML, Steenbeek J, Trujillo P, Watson R, Pauly D (2011) The Mediterranean Sea under siege: spatial overlap between marine biodiversity, cumulative threats and marine reserves. Glob Ecol Biogeogr 21:465–480
Colognola R, Masturzo P, Russo G, Scardi M, Vinci D, Fresi E (1986) Biometric and genetic analysis of the marine rissoid Rissoa auriscalpium and its ecological implications. Mar Ecol 7:265–285
Comeau S, Gorsky G, Jeffree R, Teyssié JL, Gattuso JP (2009) Impact of ocean acidification on a key Arctic pelagic mollusc (Limacina helicina). Biogeosciences 6:1877–1882
Comeau S, Gorsky G, Alliouane S, Gattuso JP (2010) Larvae of the pteropod Cavolinia inflexa exposed to aragonite undersaturation are viable but shell-less. Mar Biol 157:2341–2345
Comeau S, Gattuso JP, Nisumaa AM, Orr J (2012) Impact of aragonite saturation state changes on migratory pteropods. Proc R Soc B Biol Sci 279:732–738
Corselli C (2001) Change and diversity: the Mediterranean deep corals from the Miocene to the Present. In: Faranda FM, Guglielmo L, Spezie G (eds) Mediterranean ecosystems: structures and processes. Springer Italia, Genova
Corselli C, Basso D (1996) First evidence of benthic communities based on chemosynthesis in the Napoli mud volcano (Eastern Mediterranean). Mar Geol 132:227–239
Cossignani T, Ardovini R (2011) Malacologia Mediterranea. L’Informatore Piceno, Ancona
Council Directive 92/43/EEC (1992) http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=CELEX:31992L0043:EN:html. Accessed 10 Oct 2012
Criscione F, Patti FP (2010) Similar shells are not necessarily a reliable guide to phylogeny: Rissoa guerinii Récluz, 1843, and Rissoa lia (Monterosato, 1884) (Caenogastropoda: Rissoidae): a case study. Nautilus 124:117–128
Criscione F, Scuderi D, Patti FP (2009) Revising alpha-taxonomy in shelled gastropods: the case of Rissoa panhormensis Verduin, 1985 (Caenogastropoda: Rissoidae). Nautilus 123:303–312
Crocetta F (2011) Marine alien Mollusca in the Gulf of Trieste and neighbouring areas: a critical review and state of knowledge (updated in 2011). Acta Adriat 52:247–260
Crocetta F, Bonomolo G, Albano PG, Barco A, Houart R, Oliverio M (2012) The status of the northeastern Atlantic and Mediterranean small mussel drills of the Ocinebrina aciculata complex (Mollusca: Gastropoda: Muricidae), with the description of a new species. Sci Mar 76:177–189
Crutzen PJ, Stoermer EF (2000) The “Anthropocene”. IGBP Newsl 41:17–18
Cumming RL (1993) Reproduction and variable larval development of an ectoparasitic snail, Turbonilla sp. (Pyramidellidae, Opisthobranchia), on cultured giant clams. Bull Mar Sci 52:760–771
Curzi P, D’Onofrio S, Roveri M, Taviani M (1984) Core ADS 74–24: a window on the latest Quaternary history of the Adriatic Sea. G Geol 46(Suppl 3):61–77
Danovaro R, Dell’Anno A, Pusceddu A (2004) Biodiversity response to climate change in a warm deep sea. Ecol Lett 7:821–828
Danovaro R, Gambi C, Lampadariou N, Tselepides A (2007) Deep-sea nematode biodiversity in the Mediterranean basin: testing for longitudinal, bathymetric and energetic gradients. Ecography 31:231–244
Danovaro R, Company JB, Corinaldesi C, D’Onghia G, Galil B, Gambi C, Gooday AJ, Lampadariou N, Luna GM, Morigi C, Olu K, Polymenakou P, Ramirez-Llodra E, Sabbatini A, Sardà F, Sibuet M, Tselepides A (2010) Deep-sea biodiversity in the Mediterranean Sea: the known, the unknown, and the unknowable. PLoS One 5:e11832
Dell RK (1972) Antarctic benthos. In: Russell FS, Yonge M (eds) Advances in marine biology, vol 10. Academic, London
Demir M (2003) Shells of Mollusca collected from the seas of Turkey. Turk J Zool 27:101–140
Devescovi M, Ozretic B, Iveša L (2005) Impact of date mussel harvesting on the rocky bottom structural complexity along the Istrian coast (Northern Adriatic, Croatia). J Exp Mar Biol Ecol 325:134–145
Di Geronimo I, Bellagamba M (1985) Malacofaune dei dragaggi BS 77–1 e BS 77–2 (Sardegna nord orientale). Boll Soc Paleontol Ital 24:111–129
Di Geronimo I, La Perna R (1996) Bathyspinula excisa (Philippi 1844) (Bivalvia, Protobranchia): a witness of the Plio-Quaternary history of the deep Mediterranean benthos. Riv Ital Paleontol Stratigr 102:105–118
Di Geronimo I, La Perna R (1997) Pleistocene bathyal molluscan assemblages from Southern Italy. Riv Ital Paleontol Stratigr 103:389–426
di Maria Monterosato TA (1878) Enumerazione e sinonimia delle conchiglie mediterranee. Giorn Sci Nat Econ Palermo 13:61–115
Domènech R, Martinell J (1980) Contribución al conocimiento de la fauna malacológica sumergida del Würm del litoral gerundense. Com Prim Congr Nac Malac, Madrid
Domènech R, Martinell J (1982) Fauna malacòlogica submergida del Würm del litoral Gironí. Descriptiva i sistematica. Bull Inst Cat Hist Nat 48:31–60
Donald KM, Preston J, Williams ST, Reid DG, Winter D, Alvarez R, Buge B, Hawkins SJ, Templado J, Spencer HG (2012) Phylogenetic relationships elucidate colonization patterns in the intertidal grazers Osilinus Philippi, 1847 and Phorcus Risso, 1826 (Gastropoda: Trochidae) in the northeastern Atlantic Ocean and Mediterranean Sea. Mol Phylogenet Evol 62:35–45
Dumont HJ, Shiganova TA, Niermann U (eds) (2004) Aquatic invasions in the Black, Caspian and Mediterranean Seas. Kluwer, Dordrecht
Espinosa F, Guerra-García JM, García-Gómez JC (2007) Sewage pollution and extinction risk: an endangered limpet as a bioindicator? Biodivers Conserv 16:377–397
Evans J, Borg JA, Schembri PJ (2010) Rediscovery of live Gibbula nivosa (Gastropoda: Trochidae). Rapp Proc verb Réun CIESM 39:507
Evans J, Borg JA, Schembri PJ (2011) Distribution, habitat preferences and behaviour of the critically endangered Maltese top-shell Gibbula nivosa (Gastropoda: Trochidae). Mar Biol 158:603–611
Fabbri R, Landi L (1999) Nuove segnalazioni di molluschi, crostacei e pesci esotici in Emilia-Romagna e prima segnalazione di Corbicula fluminea (O.F. Müller, 1774) in Italia. Quad Studi Nat Romagna 12:9–20
Fabry VJ, Seibel BA, Feely RA, Orr JC (2008) Impacts of ocean acidification on marine fauna and ecosystem processes. ICES J Mar Sci 65:414–432
Faimali M, Taviani M (2011) Una rivoluzione su scala planetaria. Darwin 45:84–89
Fanelli G, Piraino S, Belmonte G, Geraci S, Boero F (1994) Human predation along Apulian rocky coasts (SE Italy): desertification caused by Lithophaga lithophaga (Mollusca) fisheries. Mar Ecol Prog Ser 110:1–8
Foighil DO (1989) Planktotrophic larval development is associated with a restricted geographic range in Lasaea, a genus of brooding, hermaphroditic bivalves. Mar Biol 103:349–358
Font J, Millot C, Salas J, Julià A, Chic O (1998) The drift of modified Atlantic water from the Alboran Sea to the eastern Mediterranean. Sci Mar 62:211–216
Forli M, Dell’Angelo B, Ciappelli F, Taviani M (2002) A new species of Haliotidae (Mollusca, Vetigastropoda) in the Italian Pliocene. Boll Malacol 38:149–154
Forli M, Dell’Angelo B, Montagna P, Taviani M (2004) A new large Patella (Mollusca: Archaeogastropoda) in the Pliocene of the Mediterranean Basin. Boll Malacol 40:1–9
Fraschetti S, Bianchi CN, Terlizzi A, Fanelli G, Morri C, Boero F (2001) Spatial variability and human disturbance in shallow subtidal hard substrate assemblages: a regional approach. Mar Ecol Prog Ser 212:1–12
Fredj G, Laubier L (1985) The deep Mediterranean benthos. In: Moraitou-Apostopulou M, Kiortsis V (eds) Mediterranean marine ecosystems. NATO Conf Ser Plenum Press, New York
Freiwald A, Beuck L, Rüggeberg A, Taviani M, R/V Meteor M70-1 Participants (2009) The white coral community in the central Mediterranean revealed by ROV surveys. Oceanography 22:58–74
Fretter V (1984) Prosobranchs. In: Wilbur KM (ed) The Mollusca, vol 1, Reproduction. Academic, New York
Galil B (2007) Seeing red: alien species along the Mediterranean coast of Israel. Acquat Invasions 2:281–312
Galil BS (2008) Alien species in the Mediterranean Sea – which, when, where, why? Hydrobiologia 597:105–116
Galil BS (2009) Taking stock: inventory of alien species in the Mediterranean sea. Biol Invasions 11:359–372
Garaventa F, Faimali M, Terlizzi A (2006) Imposex in pre-pollution times. Is TBT to blame? Mar Pollut Bull 52:696–718
Garaventa F, Centanni E, Pellizzato F, Faimali M, Terlizzi A, Pavoni A (2007) Imposex and accumulation of organotin compounds in populations of Hexaplex trunculus (Gastropoda, Muricidae) from the Lagoon of Venice (Italy) and Istrian Coast (Croatia). Mar Pollut Bull 54:602–625
Garaventa F, Corrà C, Piazza V, Giacco E, Greco G, Pane L, Faimali M (2012) Settlement of the alien mollusc Brachidontes pharaonis in a Mediterranean industrial plant: bioassays for antifouling treatment optimization and management. Mar Environ Res 76:90–96
Garilli V, Galletti L (2006) Taxonomical characters for distinguishing Cerithium lividulum Risso, 1826, and C. renovatum Monterosato, 1884 (Gastropoda, Caenogastropoda, Cerithiidae). Basteria 70:109–122
Ghisotti F (1972a) Le conchiglie del golfo di Gabès (prima parte). Conchiglie 8:63–89
Ghisotti F (1972b) Le conchiglie del golfo di Gabès (seconda parte). Conchiglie 8:101–113
Ghisotti F (1972c) Le conchiglie del golfo di Gabès (terza parte). Conchiglie 8:133–144
Ghisotti F (1973) Scapharca cfr. cornea (Reeve), ospite nuova del Mediterraneo. Conchiglie 9:68
Ghisotti F (1974a) La frenesia della nuova specie. Conchiglie 10:65–70
Ghisotti F (1974b) Rapana venosa (Valenciennes) nuova ospite adriatica? Conchiglie 10:125–126
Ghisotti F, Rinaldi E (1976) Osservazioni sulla popolazione di Scapharca, insediatasi in questi ultimi anni su un tratto del litorale romagnolo. Conchiglie 12:183–195
Gibbs PE, Bryan GW, Pascoe PL, Burt GR (1990) Reproductive abnormalities in female Ocenebra erinacea (Gastropoda) resulting from tributyltin-induced imposex. J Mar Biol Assoc UK 70:639–656
Gibson GD, Chia F-S (1995) Developmental variability in the poecilogonous opisthobranch Haminaea callidegenita: life-history traits and effects of environmental parameters. Mar Ecol Prog Ser 121:139–155
Gignoux M (1913) Les formations marines pliocénes et quaternaires de l’Italie du Sud e de la Sicile. Ann Univ Lyon 36(ns 1):1–693
Gili C, Martinell J (2000) Phylogeny, speciation and turnover. The case of Mediterranean gastropods of the genus Cyclope Risso, 1826. Lethaia 33:236–250
Gofas S (1992) Le genre Granulina (Marginellidae) en Méditerranée et dans l’Atlantique Oriental. Boll Malacol 28:1–26
Gofas S (1998) Marine molluscs with a very restricted range in the Strait of Gibraltar. Divers Distrib 4:255–266
Gofas S (2004) Geographical differentiation in Clelandella (Gastropoda: Trochidae) in the northeastern Atlantic. J Molluscan Stud 71:133–144
Gofas S, Zenetos A (2003) Exotic molluscs in the Mediterranean Basin: current status and perspectives. Oceanogr Mar Biol Annu Rev 41:237–277
Gofas S, Freiwald A, López Correa M, Remia A, Salas C, Taviani M, Wisshak M, Zibrowius H (2007) Oyster beds in the deep sea. World Congress of Malacology, Antwerp
Gofas S, Moreno D, Salas C (2011a) Moluscos marinos de Andalucia, vol 1. Servicio de Publicaciones/Interscambio Cientifico Universidad de, Malaga
Gofas S, Moreno D, Salas C (eds) (2011b) Moluscos marinos de Andalucia, vol 2. Servicio de Publicaciones e Interscambio Cientifico/Universidad de Malaga, Malaga
Gomoiu M-T (1981) Some problems concerning actual ecological changes in the Black sea. Cercet Mar IRCM 14:109–127
Gray JS (1997) Marine biodiversity: patterns, threats and conservation needs. Biodivers Conserv 6:153–175
Green AJ, Figuerola J (2005) Recent advances in the study of long-distance dispersal of aquatic invertebrates via birds. Divers Distrib 11:149–156
Guarniero I, Plazzi F, Bonfitto A, Rinaldi A, Trentini M, Passamonti M (2010) The bivalve mollusc Mactra corallina: genetic evidence of existing sibling species. J Mar Biol Assoc UK 90:633–644
Guidetti P, Boero F (2004) Desertification of Mediterranean rocky reefs caused by date-mussel, Lithophaga lithophaga (Mollusca: Bivalvia), fishery: effects on adult and juvenile abundance of a temperate fish. Mar Pollut Bull 48:978–982
Hamdani A, Soltani-Mazouni N (2011) Changes in biochemical composition of the gonads of Donax trunculus L. (Mollusca, Bivalvia) from the Gulf of Annaba (Algeria) in relation to reproductive events and pollutiom. Jordan J Biol Sci 4:149–156
Highsmith RF (1985) Floating and algal rafting as potential dispersal mechanisms in brooding invertebrates. Mar Ecol Prog Ser 25:169–179
Hoagland KE, Robertson R (1988) An assessment of poecilogony in marine invertebrates: phenomenon or fantasy? Biol Bull 174:109–125
Hoegh-Guldberg O, Bruno JF (2010) The impact of climate change on the world’s marine ecosystems. Science 328:1523–1528
Hoenselaar HJ, Hoenselaar J (1989) Musculista senhousia (Benson in Cantor, 1842) in the western Mediterranean (Bivalvia, Mytilidae). Basteria 53:73–76
Houart R (2000) New species of Muricidae (Gastropoda) from the northeastern Atlantic and the Mediterranean Sea. Zoosystema 22:459–469
Hsü KJ, Montadert L, Bernoulli D, Cita MB, Erickson A, Garrison RE, Kidd RB, Mèlierés F, Müller C, Wright R (1977) History of the Messinian salinity crisis. Nature 267:399–403
Issel A (1914) Lembi fossiliferi quaternari e recenti nella Sardegna meridionale. Acc Naz Lincei 23(Suppl 5):759–770
Janssen AW (2012) La Quaternary to Recent holoplanktonic Mollusca (Gastropoda) from bottom samples of the eastern Mediterranean Sea: systematics, morphology. Boll Malacol 48(Suppl 9):1–105
Johanesson K (1988) The paradox of Rockall: why is a brooding gastropod (Littorina saxatilis) more widespread than one having a planktonic larval dispersal stage (L. littorea)? Mar Biol 99:507–513
Jones CC (1984) Messinian refugia: evidence of some Venerinae bivalves. Ann Geol Pays Hell 22:69–77
Kabasakal H, Karhan SU, Kabasakal E, Kalakan E, Buzzurro G (2005) First record of Strombus (Doxander) vittatus vittatus Linnaeus, 1758 from the Mediterranean Sea, with a brief review of strombids in the Levant Basin. Nach Ersten Vorarlb Malakol Gesellsch 13:63–66
Keller C (1883) Die Fauna im Suez-Kanal und die Diffusion der mediterranen und erythräischen Thierwelt. Neue Denkschr algem schweiz Ges gesammt Naturwiss 29:1–39
Keppel E, Sigovini M, Tagliapietra D (2012) A new geographical record of Polycera hedgpethi Er. Marcus, 1964 (Nudibranchia: Polyceridae) and evidence of its established presence in the Mediterranean Sea, with a review of its geographical distribution. Mar Biol Res 8(10):969–981
Koutsoubas D, Tselepides A, Eleftheriou A (2000) Deep sea molluscan fauna of the Cretan Sea (Eastern Mediterranean): faunal, ecological and zoogeographical remarks. Senkenb Mar 30:85–98
Krug PJ (2007) Poecilogony and larval ecology in the gastropod genus Alderia. Am Malacol Bull 23:99–111
Kukla G, Mattheus RK, Mitchell JM (1972) The end of the present interglacial. Quat Res 2:261–269
La Perna R (1996) Phyletic relationships and ecological implications between Pagodula vaginata (De Cristofori & Jan, 1832) and Pagodula echinata (Kiener, 1840) (Gastropoda, Muricidae). Boll Soc Paleontol Ital 35:81–92
La Perna R (1998a) On Asperarca Sacco, 1898 (Bivalvia, Arcidae) and two new Mediterranean species. Boll Malacol 33:11–18
La Perna R (1998b) A new Mediterranean Skeneoides (Gastropoda: Skeneidae) from a shallow-water cave. J Conchol 36:21–27
La Perna R (2003) The Quaternary deep-sea protobranch fauna from the Mediterranean: composition, depth-related distribution and changes. Boll Malacol 39:17–34
La Perna R (2004) The identity of Yoldia micrometrica Sequenza, 1877 and three new deep-sea protobranchs from the Mediterranean (Bivalvia). J Nat Hist 38:1045–1057
La Perna R (2005) A gigantic deep-sea Nucinellidae from the tropical West Pacific (Bivalvia: Protobranchia). Zootaxa 881:1–10
La Perna R (2006) Taxonomy of the family Neilonellidae (Bivalvia, Protobranchia): Miocene and Plio-Pleistocene species of Pseudoneilonella Laghi, 1896 from Italy. Veliger 49:196–208
La Perna R (2007) Revision of the Nuculidae (Bivalvia: Protobranchia) from the Cerulli Irelli collection (Pleistocene, Mediterranean). Palaeontogr Ital 91:109–140
La Perna R, D’Abramo M (2011) The genus Nemocardium Meek 1876 in the Plio-Pleistocene of Italy (Bivalvia, Cardiidae). J Conchol 40:559–568
La Perna R, Landau B, Marquet R (2002) Species of Granulina (Gastropoda, Marginellidae) from the Pliocene of Málaga (southern Spain) with description of four new species. Cainozoic Res 1:111–120
La Perna R, Ceregato A, Tabanelli C (2004) Mediterranen Pliocene protobranchs: the genera Jupiteria Bellardi, 1875, Ledella Verril & Bush, 1897 and Zealeda Marwick, 1924 (Mollusca, Bivalvia). Boll Malacol 40:25–36
Landau B, Beu A, Marquet R (2004) The early Pliocene Gastropoda (Mollusca) of Estepona, Southern Spain. Part 5: Tonnoidea, Ficoidea. Palaeontos 5:35–102
Lavie B, Nevo E (1986) Genetic diversity of marine gastropods: contrasting strategies in Cerithium rupestre and Cerithium scabridum in the Mediterranean Sea. Mar Ecol Prog Ser 28:99–103
Lazzari G, Rinaldi E (1994) Alcune considerazioni sulla presenza di specie extramediterranee nelle lagune salmastre di Ravenna. Boll Malacol 30:195–202
Le Renard J, Sabelli B, Taviani M (1996) On Candinia (Sacoglossa: Juliidae), a new fossil genus of bivalved gastropods. J Paleontol 70:230–235
Lemghich I, Benajiba MH (2007) Survey of imposex in prosobranch molluscs along the northern Mediterranean coast of Morocco. Ecol Indic 7:209–214
Leppäkoski E, Gollasch S, Olenin S (eds) (2002) Invasive aquatic species of Europe – distribution, impact and management. Kluwer, Dordrecht
Levinton JS (1979) The effect of density upon deposit-feeding populations: movement, feeding and floating of Hydrobia ventrosa Montagu (Gastropoda: Prosobranchia). Oecologia 43:27–39
Ligios S, Anadón P, Castorina F, D’Amico C, Esu D, Gliozzi E, Gramigna P, Mola M, Monegato G (2012) Ostracoda and Mollusca biodiversity and hydrochemical features in Lat Miocene brackish basins of Italy. Geobiology 45:351–367
Little C, Nix W (1976) The burrowing and floating behaviour of the gastropod Hydrobia ulvae. Estuar Coast Mar Sci 4:537–544
López Correa M, Freiwald A, Hall-Spencer J, Taviani M (2005) Distribution and habitats of Acesta excavata (Bivalvia: Limidae) with new data on its shell ultrastructure. In: Freiwald A, Roberts M (eds) Cold-water corals & ecosystems. Springer, Berlin
Louizidou P, Thessalou-Legaki M, Zenetos A (2007) First record of the Red Sea immigrant Chrysallida fischeri (Hornung and Memod, 1925) (Gastropoda: Pyramidellidae) from Greek waters. Aquat Invasions 2:95–98
Maas AE, Wishner KF, Seibel BA (2012) The metabolic response of pteropods to acidification reflects natural CO2-exposure in oxygen minimun zone. Biogeosciences 9:747–757
Madhioub MN, Zaouali J (1988) Captage de l’huître creuse Crassostrea gigas dans le lac d’Ichkeul, Tunisie. Bull Inst Natl Sci Tech Oceanogr Pêche Salambó 15:47–60
Malatesta A, Zarlenga F (1988) Northern guests in the Pleistocene Mediterranean sea. Geol Rom (1986) 25:91–154
Marabini S, Taviani M, Vai GB, Vigliotti L (1995) Yellow sand facies with Arctica islandica: low stand signature in an early Pleistocene Front-Apennine Basin. G Geol 57(Suppl 3):259–275
Mariottini P, Smriglio C, Di Giulio A, Oliverio M (2009) A new fossil conoidean from the Pliocene of Italy, with comments on the Bela menkhorsti complex (Gastropoda: Conidae). J Conchol 40:5–14
Mars P (1958) Les faunes malacologiques quaternaires “froides” de Méditerranée. Le gisement du cap de Creus. Vie Milieu 9:67–89
Martel A, Chia F-S (1991) Drifting and dispersal of small bivalves and gastropods with direct development. J Exp Mar Biol Ecol 150:131–147
Mastrototaro F, D’Onghia G, Corriero G, Matarrese A, Maiorano P, Panetta P, Gherardi M, Longo C, Rosso A, Sciuto F, Sanfilippo R, Gravili C, Boero F, Taviani M, Tursi A (2010) Biodiversity of the white coral bank off Cape Santa Maria di Leuca (Mediterranean Sea): an update. Deep Sea Res Part II Trop Stud Oceanogr 57:412–430
Mazziotti C, Agamennone F, Tisselli M (2008) Checklist della malacofauna delle Isole Tremiti (Medio Adriatico). Boll Malacol 44(5–8):71–86
McCulloch M, Falter J, Trotter J, Montagna P (2012a) Coral resilience to ocean acidification and global warming through pH up-regulation. Nat Clim Change 2(8):623–627
McCulloch M, Trotter J, Montagna P, Falter J, Dunbar R, Freiwald A, Forsterra G, López Correa M, Maier C, Rüggeberg A, Taviani M (2012b) Resilience of cold-water scleractinian corals to ocean acidification: Boron isotopic systematics of pH and saturation state up-regulation. Geochim Cosmochim Ac 87:21–34
Mienis HK (2003) Cantharus tranquebaricus: first records of another Indian ocean species in the eastern Mediterranean. Triton 8:11
Modica MV, Mariottini P, Prkíc J, Oliverio M (2013) DNA-barcoding of sympatric species of ectoparasitic gastropods of the genus Cerithiopsis (Mollusca: Gastropoda: Cerithiopsidae) from Croatia. J Mar Biol Assoc UK 93:1059–1065
Monegatti P, Raffi S (2001) Taxonomic diversity and stratigraphic distribution of mediterranean Pliocene bivalves. Paleogeogr Paleoclimatol Paleoecol 297:1–11
Monegatti P, Raffi S (2010) The Messinian marine molluscs record and the dawn of the eastern Atlantic biogeography. Paleogeogr Paleoclimatol Paleoecol 165:171–193
Montefalcone M, Albertelli G, Morri C, Bianchi CN (2010a) Patterns of wide-scale substitution within meadows of the seagrass Posidonia oceanica in NW Mediterranean Sea: invaders are stronger than natives. Aquat Conserv 20:507–515
Montefalcone M, Parravicini V, Vacchi M, Albertelli G, Ferrari M, Morri C, Bianchi CN (2010b) Human influence on seagrass habitat fragmentation in NW Mediterranean Sea. Estuar Coast Shelf Sci 86:292–298
Moolenbeck RG, Dekker H (1993) On the identity of Strombus decorus and Strombus persicus, with description of Strombus decorus masirensis n. ssp. and a note on Strombus fasciatus. Vita Marina 42:3–10
Moreno D, Templado J (1994) El completo de especies “Nassarius cuvierii–N. unifasciatus” (Gastropoda, Nassariidae) en el SE de España. Iberus 12:33–47
Munksgaard C (1990) Electrophoretic separation of morphologically similar species of the genus Rissoa (Gastropoda; Prosobranchia). Ophelia 31:97–104
Mutlu E (2004) Sexual dimorphisms in radula of Conomurex persicus (Gastropoda: Strombidae) in the Mediterranean Sea. Mar Biol 145:693–698
Nelson TC (1925) On the occurrence and food habits of ctenophores in New Jersey inland coastal waters. Biol Bull 48:92–111
Néraudeau D, Goubert E, Lacour D, Rouchy J-M (2001) Changing biodiversity of Mediterranean irregular echinoids from the Messinian to the Present-Day. Paleogeogr Paleoclimatol Paleoecol 175:43–60
Newell R (1964) Some factors controlling the upstream distribution of Hydrobia ulvae (Pennant). (Gastropoda. Prosobranchia). Proc Zool Soc Lond 142:85–106
Nielsen LT, Jakobsen HH, Hansen PJ (2010) High resilience of two coastal plankton communities to twenty-first century seawater acidification: Evidence from microcosm studies. Mar Biol Res 6:542–555
Nikula R, Väinölä R (2003) Phylogeography of Cerastoderma glaucum (Bivalvia: Cardiidae) across Europe: a major break in the Eastern Mediterranean. Mar Biol 143:339–350
Occhipinti-Ambrogi A, Marchini A, Cantone G, Castelli A, Chimenz C, Cormaci M, Froglia C, Furnari G, Gambi MC, Giaccone G, Giangrande A, Gravili C, Mastrototaro F, Mazziotti C, Orsi-Relini L, Piraino S (2011) Alien species along the italian coasts: an overview. Biol Invasions 13:215–237
Oehlmann J, Fioroni P, Stroben E, Markert B (1996) Tributyltin (TBT) effects on Ocinebrina aciculata (Gastropoda: Muricidae): imposex develpment, sterilization, sex change and population decline. Sci Total Environ 188:205–223
Oehlman J, Stroben E, Schulte-Oehlman U, Bauer B (1998) Imposex development in response to TBT pollution in Hinia incrassate (Strōm, 1768) (Prosobranchia, Stenoglossa). Aquat Toxicol 43:239–260
Oliver Baldovì JD (2007) Catàlogo de los Gasteròpodos testàceos marinos de la parte Sur del Golfo de Valencia (Espana). Iberus 25:29–61
Oliverio M (1984) Developmental vs. genetic variation in two Mediterranean rissoid complexes. J Molluscan Stud 60:461–465
Oliverio M (1995a) The status of the living mediterranean Strombus, or: what is a lessepsian migrant? Notiz CISMA 16:35–40
Oliverio M (1995b) The systematics of the radula-less gastropod Clathromangelia (Caenogastropoda, Conoidea). Zool Scr 24:193–201
Oliverio M (1996) Contrasting developmental strategies and speciation in N. E. Atlantic Prosobranchs: a preliminary analysis. In: Taylor J (ed) Origin and evolutionary radiation of the Mollusca. Oxford University Press, Oxford
Oliverio M (2003) The Mediterranean molluscs: the best known malacofauna of the world … so far. Biogeographia 24:195–208
Oliverio M (2008) Gastropoda prosobranchia. Biol Mar Med 15(Suppl):235–278
Oliverio M, Taviani M (2004) The Eastern Mediterranean Sea: tropical invasions and niche opportunities in a “Godot Basin”. Biogeographia 24:313–318
Oliverio M, Gerosa G, Cocco M (1992) First record of Pinctada radiata (Bivalvia, Pteriidae) epibiont on the loggerhead sea turtle Caretta caretta (Chelonia, Cheloniidae). Boll Malacol 28:149–152
Olu-Le Roy K, Sibuet M, Fiala-Médioni A, Gofas S, Salas C, Mariotti A, Foucher JP, Woodside J (2004) Cold seep communities in the deep eastern Mediterranean Sea: composition, symbiosis and spatial distribution on mud volcanoes. Deep Sea Res Part I Oceanogr Res Pap 51:1915–1936
Orr JC, Fabry VJ, Aumont O, Bopp L, Doney SC et al (2005) Anthropogenic ocean acidification over the twenty-first century and its impact on calcifying organisms. Nature 437:681–686
Ovalis P, Zenetos A (2007) On the establishment of two more alien molluscs (Chama aspera Reeve, 1846 and Chama asperella Lamarck, 1819) in the eastern Mediterranean. Mediterr Mar Sci 8:97–100
Öztürk B (2012) A new alien gastropod Pseudorhaphitoma iodolabiata (Hornung & Mermod, 1928) (Mangeliidae, Mollusco) in the Mediterranean Sea. Mediterr Mar Sci 13:30–32
Öztürk B, Poutiers J-M (2005) Fulvia fragilis (Bivalvia: Cardiidae): a lessepsian mollusc species from Izmir Bay (Aegean Sea). J Mar Biol Assoc UK 85:351–356
Öztürk B, Buzzurro G, Benli A (2004) Marine molluscs from Cyprus: new data and checklist. Boll Malacol 39:49–78
Öztürk B, Önen M, Dogan A (2007) Addition to the knowledge of Turkish mollusc fauna. Rapp Comm Int Mer Médit CIESM 38:566
Padilla D (1998) Inducible phenotypic plasticity of the radula in Lacuna (Gastropoda: Littorinidae). Veliger 41:201–204
Page LR (2009) Molluscan larvae: pelagic juveniles or slowly metamorphosing larvae? Biol Bull 216:216–225
Palazzi S, Villari A (2001) Molluschi e brachiopodi delle grotte sottomarine del Taorminese. La Conchiglia 32(Suppl a 297):1–56
Pallary P (1902) Liste des mollusques testacés de la baie de Tanger. J Conchyl 50:1–39
Pallary P (1920) Exploration scientifique du Maroc organisée par la Societé de Géographie de Paris et continuée par la Scocieté des Sciences Naturelles du Maroc. Deuxiéme fascicule. Malacologie. Larose Paris and Institut Scientifique Chérifen Rabat
Panetta P, Mastrototaro F, Matarrese A, Tursi A (2003) Tanatocenosi a molluschi presenti nelel acque adriatiche albanesi. Biol Mar Med 10:597–601
Panetta P, Mastrototaro F, Matarrese A (2007) Malacofauna dei fondi incoerenti del Golfo di Manfredonia. Boll Malacol 43:57–61
Panova M, Blakeslee AMH, Miller AW, Mäkinen T, Ruiz GM, Johannesson K, André C (2011) Glacial history of the North Atlantic marine snail Littorina saxatilis, inferred from distribution of mitochondrial DNA lineages. PLoS One 6:e17511
Parravicini V, Guidetti P, Morri C, Montefalcone M, Donato M, Bianchi CN (2009) Consequnces of sea water temperature anomalies on a Mediterranean submarine cave ecosystem. Estuar Coast Shelf Sci 86:276–282
Pearse JS (1979) Polyplacophora. In: Giese AC, Pearse JS (eds) Reproduction of marine invertebrates, vol V. Academic, New York
Pedriali L, Robba E (2001) Euspira magenesi, a new species of the Naticidae (Gastropoda) from the Pliocene of Italy. Riv Ital Paleontol Stratigr 107:483–487
Pedriali L, Robba E (2009) A revision of the Pliocene naticids of northern and central Italy. III. The subfamilies Poliniceinae and Sininae. Riv Ital Paleontol Stratigr 15:371–429
Peirano A, Damasso V, Montefalcone M, Morri C, Bianchi CN (2005) Effects of climate, invasive species and antropogenic impacts on the growth of the seagrass Posidonia oceanica (L.) Delile in Liguria (NW Mediterranean Sea). Mar Pollut Bull 50:817–822
Pelosio G, Raffi S (1973) Considerazioni sul limite Plio-Pleistocene nella serie del T. Crostolo (Preappennino reggiano). Aten Parm Acta Nat 9:39–67
Peñas A, Rolan E, Luque AA, Templado J, Moreno D, Rubio F, Salas C, Sierra A, Gofas S (2006) Moluscos marinos de la isla de Alboran. Iberus 24:23–151
Peñas A, Rolan E, Almera J (2009) Fauna malacologica de un fondo detrìtico fangoso en El Maresma, Barcelona (nordeste de la Penìnsula Ibérica). Iberus 27:19–56
Pérès JM (1989) History of the Mediterranean biota and the colonization of the depths. In: Por FD, Dimentman D (eds) The legacy of Tethys. Kluwer, Dordrecht
Pérès JM, Picard J (1964) Nouveau manuel de bionomie benthique de la mer Méditerranée. Rec Trav Sta Mar Endoume 47:3–137
Piani P (1980) Catalogo dei molluschi conchiferi viventi nel Mediterraneo. Boll Malacol 16:113–224
Picken GB (1980) Reproductive adaptations of Antarctic benthic invertebrates. Biol J Linn Soc 14:67–75
Plazzi F, Ceregato A, Taviani M, Passamonti M (2011) A molecular phylogeny of bivalve mollusks: ancient radiations and divergences as revealed by mitochondrial genes. PLoS One 6:e27147
Poluzzi A, Taviani M (1986) Analisi quantitativa delle associazioni a molluschi di due sacche (Scardovari e Canarin) del delta meridionale del fiume Po. Mem Soc Geol It 27:403–410
Poluzzi A, Sabelli B, Taviani M (1981) Auto-sinecologia dei molluschi dei fondi mobili del delta settentrionale del Po (estate 1980). Boll Soc Paleont It 20:169–178
Ponder WF (1989) Mediterranean Cingulopsidae, a relict eastern Tethyan fauna (Gastropoda, Cingulopsidae). Boll Malacol 25:85–90
Pons-Moya J, Pons GX (2000) Molluscs de fons fangosos batials del Coll de Mallorca (SW de Mallorca, Illes Baleares, Mediterrània Occidental). Boll Soc Hist Nat Baleares 43:105–110
Por FD (2009) Tethys returns to the Mediterranean: success and limits of tropical re-colonization. BioRisk 3:5–19
Por FD, Dimentman C (1989) The legacy of Tethys. Kluwer, Dordrecht
Porcheddu AS, Francour P, Soltan D (1999) Considerazioni sul ritrovamento di una populazione di Mya arenaria L., 1758 negli stagni di Berri e di Vaïne (France meridionale). Boll Malacol 34:167–171
Raffi S (1986) The significance of marine boreal molluscs in the early Pleistocene faunas of the Mediterranean area. Paleogeogr Paleoclimatol Paleoecol 52:267–289
Raffi S (1993) L’origine della fauna attuale del mar Mediterraneo: il caso dei Bivalvi. XIX Seminario sulla evoluzione biologica e i grandi problemi della biologia. Faune attuali e faune fossili. Accademia nazionale dei Lincei. Contributi del centro Linceo interdisciplinare “Beniamino Segre” Rome 86:189–207
Raffi S, Taviani M (1984) An outline of the late Tertiary to Recent history of the Mediterranean deep-sea mollusk faunas. Ann Géol Pays Hellén 32:257–261
Rees WJ (1965) The aerial dispersal of Mollusca. Proc Malacol Soc Lond 36:269–282
Relini G (ed) (2008) Checklist della flora e della fauna dei mari italiani. I Parte. Biol Mar Med 15(Suppl 1):1–385
Relini G (ed) (2010) Checklist della flora e della fauna dei mari italiani/checklist of the flora and fauna in italian seas. Parte II. Biol Mar Med 17(Suppl 1):387–828
Rinaldi E (1991) Le conchiglie della costa romagnola. Essegi, Ravenna
Rodolfo-Metalpa R, Houlbrèque F, Tambutté E, Boisson F, Baggini C, Patti FP, Jeffree R, Fine M, Foggo A, Gattuso J-P, Hall-Spencer JM (2011) Coral and mollusc resistance to ocean acidification adversely affected by warming. Nat Clim Change 1:308–312
Rodrigues CF, Duperron S, Gaudron SM (2011) First documented record of a living solemyid bivalve in a pockmark of the Nile Deep-sea Fan (eastern Mediterranean Sea). Mar Biodivers Rec. doi:10.1017/S175526721100008X
Rodríguez JG, Tueros I, Borja Á, Franco J, Alonso JIG, Garmendia JM, Muxica I, Sariego C, Valencia V (2009) Butyltin compounds, sterility and imposex assessment in Nassarius reticulatus (Linnaeus, 1758), prior to the 2008 European ban on TBT antifouling paints, within Basque ports and along coastal areas. Cont Shelf Res 29:1165–1173
Romanelli M, Cordisco CA, Giovanardi O (2009) The long-term decline of the Chamelea gallina L. (Bivalvia: Veneridae) clam fishery in the Adriatic Sea: is a synthesis possible? Acta Adriat 50:171–205
Rosso A, Di Geronimo I (1998) Deep-sea Pleistocene Bryozoa of southern Italy. Geobios 30:303–317
Rossoll D, Bermúdez R, Hauss H, Schulz KG, Riebesell U, Sommer U, Winder M (2012) Ocean acidification-induced food quality deterioration constrains trophic transfer. PLoS One 7:e34737
Rovere A, Bellati S, Parravicini V, Firpo M, Morri C, Bianchi CN (2009) Abiotic and biotic links work two ways: effects on the deposit at the cliff foot induced by mechanical action of date mussel harvesting (Lithophaga lithophaga). Estuaries Coasts 32:333–339
Roveri M, Manzi V, Gennari R, Iaccarino S, Lugli S (2008) Recent advancements in the Messinian stratigraphy of Italy and their Mediterranean-scale implications. Boll Soc Paleontol Ital 47:71–85
Ruggieri G, Buccheri G (1968) Una malacofauna tirreniana dell’isola di Ustica (Sicilia). Geol Rom 7:27–58
Russo P (1999) Nuovi dati sulla distribuzione geografica di Strombus (Conomurex) persicus Swainson, 1821. Boll Malacol 34:103–104
Russo GF, Cicogna F (1991) The date mussel (Lithophaga lithophaga) a “case” in the Gulf of Naples. In: Boudouresque CF, Avon M, Gravez V (eds) Les Espèces Marines à Protéger en Méditerranée. GIF Posidonie publ, Marseille
Russo P, Mel P (2002) Ritrovamento di Musculista senhousia (Benson in Cantor, 1842) nella laguna di Venezia. La Conchiglia 34:53–55
Russo GF, Patti FP (2005) Early life history of two closely related gastropods, Rissoa auriscalpium and Rissoa italiensis (Caenogastropoda: Rissoidae). Mar Biol 147:429–437
Sabelli B, Taviani M (1980) Reflexions sur les endemiques (mollusques marins) du Golfe de Gabes (Tunisie). CIESM, Cagliari
Sabelli B, Taviani M (1984) The paleobiogeographic distribution of the Mediterranean benthic molluscs and the Messinian salinity crisis or where did the molluscs go? Ann Géol Pays Hellén 32:263–269
Sadikaj R, Panariti E, Arapi D (2009) Biotoxin levels in bivalve molluscs in Albania and the related health impact. Maced J Anim Sci 1:205–211
Sala E, Knowlton N (2006) Global marine biodiversity trends. Ann Rev Environ Resour 31:93–122
Salas C (1996) The Bivalves from off the Southern Iberian Peninsula collected by the FAUNA I and BALGIM expeditions. Haliotis 25:33–100
Salas C, Woodside J (2002) Lucinoma kazani n. sp. (Mollusca: Bivalvia): evidence of a living benthic community associated with a cold seep in the eastern Mediterranean Sea. Deep Sea Res Part I Oceanogr Res Pap 49:991–1005
Sanjuan A, Pérez-Losada M, Rolàn E (1997) Allozyme evidence for cryptic speciation in sympatric populations of Nassarius spp. (Mollusca: Gastropoda). J Mar Biol Assoc UK 77:773–784
Sá-Pinto A, Baird SJE, Pinho C, Alexandrino P, Branco M (2010) A three-way contact zone between forms of Patella rustica (Mollusca: Patellidae) in the central Mediterranean Sea. Biol J Linn Soc 100:154–169
Sarà G, Romano C, Mazzola A (2008) A new Lessepsian species in the western Mediterranean (Brachidontes pharaonis Bivalvia: Mytilidae): density, resource allocation and biomass. Mar Biodivers Rec. doi:10.1017/S175526720600087X
Sardà F, Calafat A, Mar Flexas M, Tselepides A, Canals M, Espino M, Tursi A (2004) An introduction to Mediterranean deep-sea biology. Sci Mar 68(Suppl 1):7–38
Scaccini A (1967) Dati preliminari sulel zoocenosi bentoniche e sulla biomassa in una zona dell’alto e medio Adriatico. Note Lab Biol Mar Pesca Fano 2:25–56
Scheltema RS, Williams IP (2009) Reproduction among protobranch bivalves of the family Nuculidae from sublittoral, bathyal, and abyssal depths off the New England coast of North America. Deep Sea Res Part II Trop Stud Oceanogr 56:1835–1846
Schembri PJ, Borg JA, Deidun A, Knittweis L, Mellado Lopez T (2007) Is the endemic Maltese Topshell Gibbula nivosa extinct? Rapp Proc Verb Réun CIESM 38:592
Schiaparelli S (2008) Bivalvia. Biol Mar Med 15(Suppl):296–314
Schiaparelli S, Chiantore M, Cattaneo-Vietti R, Bedulli D, Albertelli G (2000) The autoecology of some mud-dwelling species helps understanding changes in benthic communities of the northern Adriatic Sea. Atti Ass Ital Oceanogr Limn 13:195–205
Schiaparelli S, Barucca M, Olmo E, Boyer M, Canapa A (2005) Phylogenetic relationships within Ovulidae (Gastropoda: Cypraeoidea) based on molecular data from the 16S rRNA gene. Mar Biol 147:411–420
Seibel BA, Maas AE, Dierssen HM (2012) Energetic plasticity underlies a variable response to ocean acidification in the pteropod, Limacina helicina antarctica. PLoS One 7:e30464
Sellmer GP (1967) Functional morphology and ecological life history of the gem clam, Gemma gemma (Eulamellibranchia: Veneridae). Malacologia 5:137–223
Sharma PP, González VL, Kawauchi GY, Andrade SC, Guzmán A, Collins TM, Glover EA, Harper EM, Healy JM, Mikkelsen PM, Taylor JD, Bieler R, Giribet G (2012) Phylogenetic analysis of four nuclear protein-encoding genes largely corroborates the traditional classification of Bivalvia (Mollusca). Mol Phylogenet Evol 65:64–74
Simpson RD (1977) The reproduction of some littoral molluscs from Macquarie Island (sub-Antarctic). Mar Biol 44:125–142
Solsona M, Martinell J (1999) Protoconch as a taxonomic tool in Gastropoda systematics. Application in the Pliocene Mediterranean Naticidae. Geobios 32:409–419
Soppelsa O, Crocetta F, Fasulo G (2007) I molluschi marini di Punta Pioppeto (Isola di Procida – Campania). Boll Malacol 43:21–32
Spalding MD, Fox HE, Allen GR, Davidson N, Ferdaña AA, Finlayson M, Halpern BS, Jorge MA, Lombana A, Luorie SA, Martin KD, McManus E, Molnar J, Recchia CA, Robertson J (2007) Marine ecoregions of the world: a bioregionalization of coastal and shelf areas. Bioscience 57:573–583
Stjepeević J, Parenzan P (1980) Il golfo delle Bocche di Cattaro – condizioni generali e biocenosi bentoniche con carta ecologica delle sue due baie interne: di Kotor (Cattaro) e di Risan (Risano). Studia Marina 9–10:3–146
Stora G (1976) Evolution des peuplements benthiques d’un étang marin soumis à un effluent d'eaux douces. Bull Ecol 7:275–285
Strathmann MG, Strathmann RR (2007) An extraordinarily long larval duration of 4.5 years from hatching to metamorphosis for teleplanic veligers of Fusitriton oregonensis. Biol Bull 213:152–159
Streftaris N, Zenetos A (2006) Alien marine species in the Mediterranean – the 100 ‘worst invasives’ and their impact. Med Mar Sci 7:87–118
Sturany R (1896) Zoologische Ergebnisse VII. Mollusken I (Proso-branchier und Opisthobranchier; Scaphopoden; Lamellibranchier) gesammelt von S.M. Schiff “Pola” 1890–1894. Denkschr Kaiserl Akad Wissensch Math-Naturwiss Cl 63:1–36
Sullivan CM (1948) Bivalve larvae of Malpeque Bay, P. E. I. Bull Fish Res Bd Can 77:1–58
Tabanelli C (2008) Associazioni di paleocomunità batiali a molluschi bentonici nel Pliocene della Romagna. Metodologie ed indicazioni per nuove tematiche. Quad Studi Nat Romagna 26:1–80
Tamayo Goya JC (2008) Catàlogo de los bivalvos marinos del sector central del Golfo de Valencia (España). Iberus 26:69–80
Taviani M (1974) Nota sul ritrovamento di cinque specie di Molluschi Gastropoda, Prosobranchia poco conosciuti o nuovi per le acque del Mediterraneo. Quad Civ Staz Idrobiol Milano 5:39–50
Taviani M (1978) Associazioni a Molluschi pleistoceniche-attuali dragate nell’Adriatico meridionale. Boll Zool 45:297–306
Taviani M (2002) The Mediterranean benthos from late Miocene up to present: ten million years of dramatic climatic and geologic vicissitudes. Biol Mar Med 9:445–463
Taviani M (2004) Shaping the biogeography of the Mediterranean basin: one geologist’s perspective. In: Marine biogeography of the Mediterranean Sea: patterns and dynamics of biodiversity, vol 24, Biogeographia. Nuova Immagine Ed, Siena, pp 15–22
Taviani M, Colantoni P (1979) Thanatocoenoses würmiennes associées aux coraux blancs. Rapp Proc Verb Réun CIESM 25:141–142
Taviani M, Fiocchi C (1997) L’Uomo e la conchiglia. In: Endrizzi L, Marzatico F (eds) Ori delle Alpi. Quad Sez Arch Castello Buonconsiglio Monum Collez Provinc Trento 6:143–149
Taviani M, Taviani N (1986) Remarks on Coralliophila richardi (P. Fischer, 1882) (Gastropoda, Prosobranchia). Boll Malacol 22:65–72
Taviani M, Poluzzi A, Taviani N, Calamaras G (1985) Scapharca inaequivalvis; a recent invader of the southern lagoons in the Po river delta plain. Rapp Comm intern Mer Med CIESM 29:179–181
Taviani M, Bouchet P, Metivier B, Fontugne M, Delibrias G (1991) Intermediate steps of southwards faunal shifts testified by last glacial submerged thanatocoenoses in the Atlantic Ocean. Paleogeogr Paleoclimatol Paleoecol 86:331–338
Taviani M, Angeletti L, Freiwald A, Dimech M, Harasewych MG, Mifsud C, Oliverio M (2009) Coralliophilinae (Gastropoda: Muricidae) associated with deep-water coral banks in the Mediterranean. Nautilus 123:106–112
Taviani M, Angeletti L, Ceregato A (2011a) Chemosynthetic bivalves of the family Solemyidae (Bivalvia, Protobranchia) in the Neogene of the Mediterranean Basin. J Paleontol 85:1067–1076
Taviani M, Vertino A, López Correa M, Savini De Mol B, Remia A, Montagna P, Angeletti L, Zibrowius H, Alves T, Salomidi M, Ritt B, Henry P (2011b) Pleistocene to Recent scleractinian deep-water corals and coral facies in the Eastern Mediterranean. Facies 57:579–603
Tecchio S, Ramírez-Llodra E, Sardà F, Company JB, Palomera I, Mechó A, Pedrosa-Pàmies R, Sanchez-Vidal A (2011) Drivers of deep Mediterranean megabenthos communities along longitudinal and bathymetric gradients. Mar Ecol Prog Ser 439:181–192
Tenekidis NE (1989) On a collection of shells from the Greek seas. Protopata, Athens
Terlizzi A, Russo GF (1995) Variabilità strutturale del popolamento a molluschi di alcune praterie di Posidonia oceanica del Mediterraneo. Biol Mar Med 2:423–426
Terlizzi A, Fraschetti S, Gianguzza P, Faimali M, Boero F (2001) Environmental impact of antifouling technologies: state of the art and perspectives. Aquat Conserv Mar Freshw Ecosyst 11:311–317
Terlizzi A, Fraschetti S, Guidetti P, Boero F (2002) The effects of sewage discharge on shallow hard substrate sessile assemblages. Mar Pollut Bull 44:544–550
Terlizzi A, Scuderi D, Fraschetti S, Anderson MJ (2005) Quantifying effects of pollution on biodiversity: a case study of highly diverse molluscan assemblages in the Mediterranean. Mar Biol 148:293–305
Terlizzi A, Bevilacqua S, Scuderi D, Fiorentino D, Guarnieri G, Giangrande A, Licciano M, Felline S, Fraschetti S (2008) Effects of offshore platforms on soft bottom macro-benthic assemblages: a case study in a Mediterranean gas field. Mar Pollut Bull 56:1303–1309
Terreni G (1981) Molluschi conchiferi del mare antistante la costa toscana (Gastropoda, Scaphopoda, Amphineura, Bivalvia, Cephalopoda). Benvenuti e Cavaciocchi, Livorno
Thiriot-Quievreux C, Rodriguez-Babio C (1975) Etude des protoconques de quelques prosobranches de la région de roscoff. Cah Biol Mar 16:135–148
Tsiakkiros L, Zenetos A (2011) Further additions to the alien mollusc fauna along the Cypriot coast: new opisthobranchia species. Acta Adriat 52:115–124
Turk T, Furlan B (2011) New records of Indo-Pacific and Atlantic mollusc species (Opisthobranchia) in the Eastern Mediterranean and Adriatic Sea. Ann Se Hist Nat 21:5–10
Tzedakis PC, Channell JET, Hodell DA, Kleiven HF, Skinner LC (2012) Determining the natural length of the current interglacial. Nat Geosci 5:138–141
Tzomos T, Chartosia N, Christodoulou M, Kitsos M-S (2011) New records and range expansion of lessepsian migrants in the Levantine and Aegean Seas. Mar Biodivers Rec 3:e10
UNEP (2010) The Mediterranean Sea biodiversity: state of the ecosystems, pressures, impacts and future priorities. UNEP-MAP RAC/SPA, Tunis
UNEP/MAP-RAC/SPA (2008) Guidelines for controlling the vectors of introduction into the Mediterranean of non-indigenous and invasive marine species. RAC/SPAC, Tunis
Valavanidis A, Vlachogianni T (2011) Marine litter: man-made solid waste pollution in the Mediterranean Sea and coastline. Abundance, composition and sources identification. http://chem-tox-ecotox.org/wp/?p=749. Accessed 27 Sept 2012
van Aartsen JJ, Menkhorst HPM, Gittenberger E (1984) The marine Mollusca of the Bay of Algeciras, Spain, with general notes on Mitrella, Marginellidae and Turridae. Basteria 1(Suppl 2):135
Vatova A (1949) La fauna bentonica dell’Alto e Medio Adriatico. Nova Thal 1:1–110
Vazzana A (2010) La malacofauna del circalitorale di Scilla (Stretto di Messina). Boll Malacol 46:65–74
Vicente N (1990) Estudio ecológico y protección del molusco lamelibranquio Pinna nobilis L. 1758 en la costa mediterránea. Iberus 9:269–279
Vio E, De Min R (1996) Contributo alla conoscenza dei Molluschi marini del golfo di Trieste. Atti Mus Civ St Nat Trieste 47:173–233
Warén A (1990) Ontogenetic changes in the trochoidean (Archaeogastropoda) radula, with some phylogenetic interpretations. Zool Scr 19:179–187
Wilson EO (ed) (1988) Biodiversity. National Academic Press, New York
Wirzinger G, Vogt C, Bachmann J, Hasenbank M, Liers C, Stark C, Ziebart S, Oehlmann J (2007) Imposex of the netted whelk Nassarius reticulatus (Prosobranchia) in Brittany along a transect from a point source. Cah Biol Mar 48:85–94
WoRMS (2012) World register of marine species. http://www.marinespecies.org/aphia.php?p=taxdetails&id=565481. Accessed 29 Sept 2012
WWF/IUCN (2004) The Mediterranean deep-sea ecosystems: an overview of their diversity, structure, functioning and anthropogenic impacts, with a proposal for conservation. IUCN, Malaga
Zamouri-Langar N, Chouba L, Jarboui O, Mirabet R (2006) Presence du bivalve Veneridae Ruditapes philippinarum (Adams & Reeve, 1850) sur le littoral sud de la Tunisie. Bull Inst Natn Scien Tech Mer Salambô 33:23–27
Zenetos A (1997) Diversity of marine bivalvia in Greek waters: effects of geography and environment. J Mar Biol Assoc UK 77:463–472
Zenetos A, Gofas S, Russo G, Templado J (2003) CIESM atlas of exotic species in the Mediterranean, vol 3, Mollusca. CIESM, Monaco
Zenetos A, Koutsoubas D, Vardala-Theodoru E (2005) Origin and vectors of introduction of exotic molluscs in greek waters. Belg J Zool 135:279–286
Zenetos A, Gofas S, Verlaque M, Cinar ME, Raso EG, Bianchi CN, Morri C, Azzurro E, Bilecenoglu M, Froglia C, Siokou I, Violanti D, Sfriso A, San Martin G, Giangrande A, Katagan T, Ballesteros E, Ramos-Esplá A, Mastrototaro F, Ocana O, Zingone A, Gambi MC, Streftaris N (2010) Alien species in the Mediterranean Sea by 2010 A contribution to the application of European Union’s Marine Strategy Framework Directive (MSFD). Part I. Spatial distribution. Mediterr Mar Sci 11:381–493
Zibrowius H (1991) Ongoing Modification of the Mediterranean marine fauna and flora by the establishment of exotic species. Mésogée 51:83–107
Zibrowius H (2002) Assessing scale and impact of ship-transported alien fauna in the Mediterranean. CIESM Workshop Monogr 20:63–68
Zibrowius H, Taviani M (2005) Remarkable sessile fauna associated with deep coral and other calcareous substrates in the Strait of Sicily, Mediterranean Sea. In: Freiwald A, Roberts M (eds) Deep-water corals & ecosystems. Springer, Berlin
Acknowledgments
This paper benefitted over the years of enlightening discussion with a number of professional and amatorial malacologists. Alessandro Ceregato (ISMAR-CNR Bologna), Francesca Garaventa (ISMAR-CNR Genova), Serge Gofas (University of Malaga), Marco Faimali (ISMAR-CNR Genova), Rafael La Perna (University of Bari), Marco Oliverio (University La Sapienza, Rome), Patrick Schembri (University of Malta), José ‘Pepe’ Templado (Museo Nacional de Ciencias Naturales-CSIC, Madrid), Antonio Terlizzi (University of Lecce) kindly revised the ms providing many constructive suggestions. This article contributes to the EU Hermione (contract n° 226354) and Coconet (contract n° 287844) programmes. ISMAR-CNR Bologna scientific contribution n. 1769.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media Dordrecht
About this chapter
Cite this chapter
Sabelli, B., Taviani, M. (2014). The Making of the Mediterranean Molluscan Biodiversity. In: Goffredo, S., Dubinsky, Z. (eds) The Mediterranean Sea. Springer, Dordrecht. https://doi.org/10.1007/978-94-007-6704-1_16
Download citation
DOI: https://doi.org/10.1007/978-94-007-6704-1_16
Published:
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-007-6703-4
Online ISBN: 978-94-007-6704-1
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)