Abstract
Dialysis-related amyloidosis (DRA) is a clinical syndrome of pain, loss of function and other symptoms due to the deposition of amyloid consisting of β2-microglobulin (β2m) in the musculoskeletal system. The condition is seen in patients who suffer from chronic kidney disease and are treated with hemodialysis for a long time. Even though β2m easily can be manipulated to form amyloid in laboratory experiments under non-physiological conditions the precise mechanisms involved in the formation of β2m-amyloid in patients with DRA have been difficult to unravel. The current knowledge which is reviewed here indicates that conformational fluctuations centered around the D-strand, the DE-loop, and around the cis-configured Pro32 peptide bond are involved in β2m amyloidosis. Also required are highly increased concentrations of circulating β2m and possibly various post-translational modifications mediated by the pro-inflammatory environment in uremic blood, together with the influence of divalent metal ions (specifically Cu2 +), uremic toxins, and dialysis-enhanced redox-processes. It seems plausible that domain-swapped β2m dimers act as building blocks of β-spine cross-β -sheet fibrils consisting of otherwise globular, roughly natively folded protein. An activated complement system and cellular activation perpetuate these reactions which due to the affinity of β2m-amyloid for the collagen of synovial surfaces result in the DRA syndrome.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Despite normally being a small soluble protein in the circulation or in complex with membrane proteins on cell surfaces, β2-microglobulin (β2m) is prone to form amyloid under specific conditions in vitro as well as in vivo. Thus, β2m is the fibrillating culprit in Aβ2m, a systemic type of amyloidosis that is first and foremost associated with chronic hemodialysis in a clinical syndrome named dialysis-related amyloidosis (DRA). Due to its small size (Mr = 11,729) β2m is present in the glomerular filtrate of the normal kidney and β2m was originally discovered as a component present in the urine of patients with tubular proteinuria, i.e. patients with defects in the reabsorption of β2m in the proximal tubules of the kidneys (Berggård 1965; Berggard and Bearn 1968). While the DRA syndrome was defined earlier (Assenat et al. 1980; Charra et al. 1984; Kuntz et al. 1984; Warren and Otieno 1975) it was not until 1985 that it was discovered that β2m is the major constituent of the amyloid deposits in osteoarticular tissues encountered in long-term hemodialysis patients with DRA symptoms (Gejyo et al. 1985; Gorevic et al. 1985, 1986).
In addition to its importance for patients with DRA β2m is also an easily accessible model protein for in vitro studies of protein amyloidogenesis. Many of these aspects have been covered in several recent reviews (Drueke and Massy 2009; Eichner and Radford 2011; Heegaard 2009; Yamamoto et al. 2009). While Chap. 8 of this book focuses on the experimental aspects of β2m misfolding and fibrillogenesis in vitro the present chapter is focused on the molecular pathology of β2m under in vivo conditions where β2m amyloidosis is encountered.
2 β2-Microglobulin in Physiology
β2m is present on the surface of all nucleated cells where it is the invariant chain in complex with and stabilizing membrane-anchored peptide- and lipid-presenting polypeptide chains (MHC class I and CD1 complexes) and Fc-receptors. It is also found as a conformationally less restricted (Hodkinson et al. 2009; Verdone et al. 2002) free monomer in blood and other biofluids, including synovial fluid (Gobezie et al. 2007), at low concentration (1.5–3 mg/ml (128–256 nM) in serum) (Karlsson et al. 1980). The normal physiological function, if any, of the freely circulating β2m is unknown.
2.1 β2-Microglobulin in the Healthy Organism
β2m associates non-covalently with MHC class I and CD1 complex heavy chains during synthesis to form MHC molecules that present peptides of 8–10 residues processed from intracellular proteins for cytotoxic T-lymphocytes (Doherty and Zinkernagel 1975) or antigenic lipids for natural killer T-cells in the case of the CD1 complex (Gumperz 2006; Kang and Cresswell 2002). β2m is required for efficient transport of nascent MHC class I heavy chains to the cell surface (Williams et al. 1989; Zijlstra et al. 1990). β2m is also present in association with other types of MHC Class I-like cell receptors, such as the IgG Fc receptor (Praetor and Hunziker 2002) and the HFE hemochromatosis membrane protein (Feder et al. 1997). β2m is critical for the stability and function of these receptors but is not directly involved in their binding of ligands (Otten et al. 1992).
A variable number of MHC class I molecules are expressed on cell membranes from different cells. In the case of human lymphocytes it has been estimated that there are 105–106 β2m-molecules/cell (Bjorck et al. 1979). When MHC is degraded, the MHC-associated β2m is released to the circulation (Cresswell et al. 1974). This results in a quite constant production of free (monomeric) β2m at a level of 0.13 mg/h ´ kg body weight under normal conditions. More than 99.9 % of this circulating β2m is filtered through the kidney glomeruli into the proximal tubules where it is reabsorbed by pinocytosis or by binding to the endocytic multiligand receptor megalin. Subsequently, it is degraded by the tubular cells (Karlsson et al. 1980; Saito et al. 1994). As a result, less than 0.1 % of the glomeruli-filtered β2m is excreted in the urine under normal conditions. A half-time for free β2m in the circulation of only about 60 min can be estimated on the basis of its serum concentration and production rate and the normal glomerular filtration rate and discounting exchange with cell-surface associated β2m. These parameters fluctuate only minimally under normal physiological conditions (Karlsson et al. 1980). In renal disorders (e.g., cadmium poisoning) where proximal tubular function is destroyed and tubular proteinemia ensues, the GFR and β2m serum concentrations are normal, but β2m is lost in the urine. Urine from patients suffering from defects in the function of proximal tubules is therefore a convenient source of β2m that can be purified to homogeneity by a combination of size-exclusion chromatography and chromatofocusing (Nissen et al. 1987, 1997).
2.2 β2-Microglobulin Structure
A detailed review of β2m conformational structure and dynamics is provided by Chap. 7 by Esposito et al. Here, the features that are most relevant and important for the generation of β2m amyloid in vivo are summarized.
β2m is a 99 residue protein and has a molecular weight of 11,729 Da. It folds natively into a seven-stranded antiparallel β-sandwich consisting of two β-sheets linked covalently by one intramolecular disulfide bond between Cys25 and Cys80. One sheet contains four strands (A, B, E and D) and the other sheet three (C, F and G) (Fig. 19.1). Native β2m as well as amyloid β2m are highly structured, tightly packed proteins. Strands B and F, connected by the disulfide bond, are the most protected part of the protein. Strands A, C, E, and G are moderately protected, leaving the D-strand connecting the two β-sheets of the β2m sandwich as the most exposed part of the monomeric native protein (Villanueva et al. 2004). The D-strand interacts with the α-heavy chain in the MHC class II cell membrane complex (Khan et al. 2000; Saper et al. 1991). The loop between the D and E strands is a strained but dynamic part of the molecule that may switch between several conformations (Okon et al. 1992; Verdone et al. 2002).
Schematic β2m structure (PDB: 1DUZ) (Saper et al. 1991). The A-G β-strands are labelled and the DE-loop with the Lysine-58 cleavage point is marked by the arrow
3 β2-Microglobulin in Pathology
Levels of plasma β2m increase in lymphoproliferative disorders, in chronic renal failure (with decreased filtration), in inflammation and infection and in other conditions with high cell turnover (Wu 2006). A relationship between tumor burden in certain lymphoproliferative disorders, particularly multiple myeloma, and serum levels of β2m thus has been noted. β2m is lost in the urine in conditions where proximal tubular function is impaired.
While elevated plasma β2m levels are absolutely necessary for the development of DRA it is not sufficient since increased circulating levels of β2m where the kidney function is normal e.g. in chronic immunoinflammatory conditions, in infections and hematopoietic malignancies (Bartl et al. 1989; Bhalla et al. 1985; Bourantas et al. 1999) do not lead to β2m amyloidosis. In agreement with this there are no signs of β2m amyloidosis in engineered mice models with highly increased human β2m in the circulation (Zhang et al. 2010). A sustained increase in β2m concentration in the circulation combined with specific abnormalities (e.g. inflammation and oxidative stress (Annuk et al. 2005; Terawaki et al. 2004)) encountered in renal insufficiency and/or the process of hemodialysis thus are necessary for the development of DRA.
4 β2-Microglobulin in Dialysis-Related Amyloidosis
The incidence of DRA is seemingly declining with the increased use of high-flux dialysis, with changes in dialysate composition, and as the components of dialysis machines that come into contact with patient blood (dialysis membranes and tubings) have become more biocompatible (Fujimori 2011; Schwalbe et al. 1997; Winchester et al. 2003).
4.1 The Clinical Syndrome
DRA results from the deposition and accumulation of β2m amyloid (Floege and Ehlerding 1996; Gejyo et al. 1985; Gorevic et al. 1985). Deposition primarily takes place in osteoarticular tissue. The clinical manifestations of DRA include erosive and destructive osteoarthropathy, destructive spondyloarthropathy, cystic bone lesions, carpal tunnel syndrome, and other neuropathies (Kiss et al. 2005) (Fig. 19.2) leading to joint pain, decreased function, and fractures.
Magnetic resonance imaging of humeral erosions in a 63-year-old man on hemodialysis for 31 years for chronic glomerulonephritis with biopsy-proven dialysis-related amyloidosis. Coronal T1-weighted MR image (470/12) shows osteolysis in superior-posterior humeral head, which communicates with joint (arrow). Low-signal-intensity tissue representing amyloid appears within lesion. Amyloid deposits are also visible within subdeltoid bursa between deltoid muscle and humerus (arrowheads). (Reproduced with permission from Fig. 4 in Kiss et al. 2005)
In addition, β2m amyloid deposits may be found in extra-osteoarticular tissues, including the heart, gastrointestinal tract, ovaries, liver, ureter and subcutaneous tissue (Buchholz et al. 1995; Kawano et al. 1998; Mogyorosi and Schubert 1999; Mount et al. 2002; Shimizu et al. 2003). Amyloid deposits outside the musculoskeletal system are often asymptomatic but may cause serious complications such as heart failure or gastrointestinal bleeding. Extensive deposition of β2m amyloid in the small blood vessels in the myocardium may cause calcification of the heart and cardiac dysfunction, thereby contributing to the occurrence of heart disease in long-term dialysis patients (Takayama et al. 2001).
The prevalence of DRA may also be influenced by patient age and duration of dialysis treatment (Jadoul 1998; Jadoul et al. 1997). Recent studies, however, fail to demonstrate a clear correlation between clinical manifestations (carpal tunnel syndrome) and duration of end-stage kidney disease and hemodialysis (Kwon et al. 2011). Furthermore, the type of hemodialysis, e.g. high-flux/low-flux dialyzers, membrane type, dialysate composition and purity, and biocompatibility of the material in contact with the biofluids are all factors that may influence the development of amyloid deposits (Baz et al. 1991; Campistol et al. 1999; Locatelli et al. 1999; van Ypersele et al. 1991) even though there are no clear effects on circulating oxidative stress parameters (Schneider et al. 2011). There is no direct correlation between the efficiency of β2m removal and the rate of progress of DRA (Danesh and Ho 2001; Kwon et al. 2011).
4.2 β2-Microglobulin Aggregation, Fibrillation, and Amyloidogenesis
A great number of in vitro conditions have been shown to induce amyloid fibrillation of β2m (cf. Chap. 8). These conditions include extremes of pH, amino acid substitutions, anionic detergent, addition of trifluoroethanol, ultrasonication, agitation, heat treatment, incubation with Cu2 + (Morgan et al. 2001; Ohhashi et al. 2005; Sasahara et al. 2007; Yamamoto et al. 2004a) and other factors (Heegaard 2009). Most of these conditions are unlikely to be encountered in vivo except for slightly decreased pH at inflammatory sites (Treuhaft and McCarty 1971), increased divalent cations (especially Cu2 +), Proline-32 cis-to-trans isomerisation, and Lysine-58 cleaved forms of β2m (cf. Table 19.1) (Eichner and Radford 2011; Heegaard 2009).
Some of the fibrillation pathways do not necessarily involve major conformational changes (Calabrese et al. 2008; Eakin et al. 2006; Liu et al. 2011). This is in accord with the lack of correlation between thermodynamic stability and tendency to amyloid formation of β2m variants (Smith et al. 2003). Thus, highly conformationally unstable β2m variants may readily unfold and aggregate but do not in every case form amyloid. Amyloid fibrillation appears to be linked to the conformational dynamics of retained globular folds e.g. leading to domain swapped dimers as the building blocks in fibril formation (Hafner-Bratkovic et al. 2011; Liu et al. 2011; Sambashivan et al. 2005; Wahlbom et al. 2007; Domanska et al. 2011; Mendoza et al. 2011). Such mechanisms are compatible with limited structural rearrangements e.g. cis-to-trans isomerisation of the Pro-32 bond or rearrangement or pertubations of the DE-loop in the globular fold (Barbet-Massin et al. 2010; Colombo et al. 2011; Eichner and Radford 2009; Eichner and Radford 2011; Mimmi et al. 2006; White et al. 2009) and has recently also been shown to be a plausible mechanism involved in ΔN6β2m amyloid formation (Domanska et al. 2011). While increasingly supported by experimental data these mechanisms have not yet been conclusively tested with in vivo-generated material.
The kinetics of in vitro generation of amyloid-like fibrils displays characteristic traits in which a lag phase precedes a period of exponential growth. During this lag phase the early aggregates or nuclei are generated, and the subsequent rapid fibril elongation corresponds to the exponential growth period. The majority of ex vivo β2m amyloid (i.e. material isolated from clinical specimens and studied in the laboratory) appears to consist of aggregated full-length, wild-type β2m (Gorevic et al. 1986). However, natively folded β2m is not prone to self-aggregate at neutral pH even at vastly supraphysiological concentrations (Eakin and Miranker 2005; Kad et al. 2001; Myers et al. 2006) and, as mentioned above, in vivo fibrillogenesis will not be initiated simply by the presence of high β2m plasma concentrations. It is likely that the assembly of β2m monomers into bona fide amyloid fibril requires encounters between partially unfolded molecules. Under native conditions this is extremely rare, and possibly explains why the generation of amyloid in vivo is a process that occurs over prolonged periods of time, usually several years.
4.2.1 β2m Post-Translational Modifications
The conformational dynamics of β2m has been studied using H/D-exchange experiments monitored by e.g. mass spectrometry (Heegaard et al. 2005; Hodkinson et al. 2009; Jørgensen et al. 2007). These experiments show the capability of β2m to undergo transient regional unfolding-refolding under physiological conditions and also document the increased rate of unfolding—even though the molecule has the same overall conformation as the wild-type (Mimmi et al. 2006)—of a cleaved variant of β2m termed ΔK58-β2m in which Lys58 has been proteolytically removed (Heegaard et al. 2005). This post-translationally modified β2m has been found in the circulation of many (20–40 %) hemodialysis patients (Corlin et al. 2005), and serum levels are markedly higher in patients dialyzed with the less biocompatible, copper containing low-flux membranes (Cuprophane), than in those treated with synthetic high-flux membranes. ΔK58-β2m is thought to be generated in vivo as a consequence of the activation of the complement system by cleavage of β2m by the serine protease C1s between Lys58 and Asp59 in the DE-loop followed by the rapid removal of Lys58 by carboxypeptidase B activities (Nissen et al. 1990). The removal of Lys58 changes the protein from a single chain molecule with an intrachain disulphide bond to two chains held together by an interchain disulphide bond. The two chains consist of residues 1–57 and 59–99 of native β2m. The removal of Lys58 only results in minor global conformational changes (Mimmi et al. 2006) but a positively charged residue that otherwise could contribute to electrostatic repulsion between β2m molecules is lost. This makes the AA59–70 region of the molecule more hydrophobic and aggregation-prone. Also, the molecule is considerably more conformationally unstable as shown by capillary electrophoresis experiments (Heegaard et al. 2002). Thus the cleavage of β2m induces significantly accelerated (about 10 times) rates of cooperative unfolding at physiological temperature, increasing molecular aggregation and the ability to generate fibrils with amyloid features (Heegaard et al. 2005).
Other posttranslationally modified species e.g. β2m with deletions of N-terminal residues (Esposito et al. 2000) (Table 19.1) are also of interest for the in vivo situation because catalytic amounts (1 %) are sufficient to induce conversion of native β2m into Aβ2m in vitro (Eichner and Radford 2011). A catalytic effect was also shown for fibrils of Lys-58 cleaved β2m formed in the presence of heparin sulfate (Corlin et al. 2010). N-terminally truncated β2m has been found to be a significant constituent of Aβ2m fibrils ex vivo (Stoppini et al. 2000, 2005) (cf. below). The ΔN6 truncation of β2m (Bellotti et al. 1998; Linke et al. 1987, 1989; Stoppini et al. 2000, 2005) results in structural rearrangements of the protein rendering the N-terminal region more disordered at the same time as the β-bulge of strand D, the successive loop, and strand E all adopt a less flexible conformation. The ΔN6β2m-variant has been shown to have a reduced free energy of stabilization as compared to native β2m and to possess an enhanced tendency to precipitate and self-aggregate (Esposito et al. 2000). At pH lower than 7, ΔN6β2m generates amorphous aggregates and short fibrillar structures, and at neutral pH this variant possesses the ability to further extend ex vivo β2m fibrils. Moreover, examination of a putative role of collagen on β2m fibril formation showed that the presence of Δ N6β2m together with collagen induced the generation of β2m amyloid-like fibrils (Canale et al. 2011; Relini et al. 2006).
Posttranslational modifications in the form of truncations and cleavages thus significantly decrease β2m solubility at physiological conditions and are likely to occur in vivo. Measurements suggest that up to 30 % of the β2m in amyloid fibers extracted from ex vivo deposits are present as N-terminally modified or truncated forms while Lys-58 cleaved β2m could not be detected in the same extracts (Bellotti et al. 1998; Floege and Ehlerding 1996; Giorgetti et al. 2007; Linke et al. 1986). Conversely, Lys-58 cleaved β2m has been demonstrated in the circulation of many dialysis patients while N-terminally truncated β2m variants have only been demonstrated in amyloid deposits.
4.2.2 β2m Pro32-Isomers and Conformers
In native, wild-type β2m the peptide bond preceding Pro32 attains the less common, thermodynamically unfavorable cis configuration (Jahn et al. 2006). Engineered mutations changing Pro32 to glycine or alanine, causing the peptide bond to stay in the trans configuration, result in markedly increased rates of amyloid fiber growth and in a repacking of the hydrophobic core of the protein wherein the backbone changes in the BC loop affect the conformation of strand D and the DE loop (Jahn et al. 2006; Eakin et al. 2006). Thus, a β2m folding intermediate in which Pro32 is kept in trans configuration does not bind the MHC heavy chain well (Esposito et al. 2008) and is much more prone to aggregation than the wild-type β2m molecule (Jahn et al. 2006). Since the isomerization of this bond is the slowest step in the folding of native β2m (Eakin et al. 2006; Eichner and Radford 2009; Kameda et al. 2005), a population of protein intermediates in the non-native trans configuration may exist and be of significance in fibril formation. Interestingly, chelation of Cu2 + by His31 promotes Pro32 isomerization (Eakin et al. 2002).
A β-bulge in this D strand is straightened out in infrequently encountered β2m structures and this may favour oligomerization by edge strand docking (Richardson and Richardson 2002) possibly facilitating amyloid formation (Trinh et al. 2002). However, recent data do not support a role for the β-bulge in protecting against amyloid formation (Azinas et al. 2011).
4.2.3 β2m-Cu2 + Interactions
Many proteins are known to have specific binding sites for metal ions, and binding of ions can significantly alter the properties of the protein. β2m binds metal cations, in particular Cu2 +, and this binding will, under the proper experimental conditions, result in the generation of fibrous β2m aggregates (Eakin et al. 2002). The binding of Cu2 + to β2m is specific, while other divalent cations such as Ca2 + and Zn2 + bind nonspecifically (Morgan et al. 2001). Studies have indicated that binding of Cu2 + to β2m lower the energy barrier of transition (Deng et al. 2006) to a partially unfolded state. The binding of Cu2 + (chiefly by His31) promoting these local molecular rearrangements has also been proposed to enhance the cis-trans isomerization at Pro32 leading to a partial unfolding of the hydrophobic core (Eakin et al. 2004) and thus increasing conformational instability (De Lorenzi et al. 2008). From a pathophysiological point of view, the relevance of Cu2 + to β2m amyloidogenesis in DRA is evident, not only because of the former use of the Cuprophane membranes containing copper, but also since a dialysis patient is exposed to more than 100 L of dialysate during each dialysis session (Cheung et al. 2006). The dialysate contains copper to a maximum level of 1.6 mM (Vorbeck-Meister et al. 1999), being within a factor of two of the measured affinity of Cu2 + and β2m (Eakin et al. 2002).
4.2.4 β2m-amyloid Seeding
Addition of nucleating seeds (preformed β2m fibrils) to a solution of native β2m will result in extensive fibrillation by elongation of the existing fibrils. In a study by Jones et al. (2003), a range of peptides of β2m were examined for their ability to assemble into amyloid-like fibrils in vitro and for their ability to act as seeds for full-length β2m. The study showed that only peptides having a sequence corresponding to strand E in wild-type β2m (E peptides) were capable of fibrillation, even when studied over a wide pH range. These fibrils also had the ability to seed the formation of fibrils in a solution of full-length β2m. The E peptides contain a very large number of aromatic amino acids, i.e., the aromatic residues Phe56, Trp60, Phe62 and Tyr63 which are all important for hydrophobic interactions of β2m with the heavy chain in the MHC class I complex (Saper et al. 1991). The importance of the DE-loop region is further underscored by a substantial number of experiments showing the effect of mutations and posttranslational modifications in the D-strand and DE-loop region (AA50–63) (Azinas et al. 2011; Santambrogio et al. 2010) including other nucleation/elongation experiments strongly implicating this aromatic-rich region in amyloidogenetic interactions (Platt et al. 2008). The region was shown directly by 2D NMR measurements to be the longest contiguous region involved in intermolecular contacts in low pH-generated fibrils (Debelouchina et al. 2010) and is also central in the putative amyloidogenic structure involved in domain-swapped propagating dimers (see below).
4.2.5 Structure of β2m Amyloid Fibrils
All amyloid fibrils, despite having rather different morphologies, share a common cross-β spine structure (Jahn et al. 2010). Albeit not fully understood, the propensity of a protein or peptide to form amyloid fibrils is dependent on an interplay between secondary structure, charge, sequence and hydrophobicity.
Experiments using low pH and high salt have shown that in the early aggregation processes β2m monomers first assemble into dimers and tetramers, then as β2m aggregates over time, changes in secondary structure can be observed indicative of the emergence of antiparallel intermolecular β-sheet structures (Fabian et al. 2008). In a study using H/D exchange of amide protons combined with NMR analysis the core of the β2m amyloid fibril was mapped. The result was that in addition to the regions protected from exchange in the native monomeric β2m, the residues in the native loops also become highly protected in the fibrillar state indicating an increase in the hydrogen bond network in the fibrils, leaving only the N- and C-terminal ends unprotected from exchange. Also, the CD spectrum of β2m amyloid fibrils showed increased β-sheet content, supporting the suggestion that both the native loops and the native β-strands are transformed into β-sheets in the β2m amyloid fibrils (Hoshino et al. 2002).
The structure of β2m amyloid fibrils has been suggested to involve the assembly of six protofilaments, arranged in pairs of three protofilaments, wherein each protofilament is build from globular subunits in a dimer-of-dimers packing (Mendoza et al. 2011; White et al. 2009). Domain-swapped dimers are also at the heart of a model for the β2m fibril core formation stabilized by disulfide exchange leading to intermolecular disulfide bridges. This leads to a “steric zipper” arrangement with the AA54–59 segment most likely involved and thereby forming the β-spine with retention of the globular features of the other parts of the protein that participates in the amyloid fibril structure (Liu et al. 2011). In another model tetramers are formed by dimer-dimer interactions involving D- and G-strands from the two different dimer-units (Mendoza et al. 2011) while D-D interactions were found unlikely to form dimer interfaces (Mendoza et al. 2010).
Three-dimensional domain swapping is emerging as a common mechanism for amyloid fibril formation (Ecroyd et al. 2010; Hafner-Bratkovic et al. 2011) and entails disulfide exchange to get covalently linked dimer β2m building blocks forming the steric-zipper spine of amyloid fibrils (Fig. 19.3). This is consistent with the accelerating effect of reducing agents on Aβ2m formation in vitro at physiological pH in some studies (Liu et al. 2011) and with the fiber polymorphism characteristic of various amyloids. Disulfide rearrangement is well known to occur in vivo e.g. in immunoglobulin arm rearrangements (Liu et al. 2010) and in some amyloidogenic proteins (Knaus et al. 2001; Nilsson et al. 2004). The precise mechanisms associated with this model in vivo and its relevance for DRA remain to be settled. Domain swapping and stabilization by disulfide exchange have not been shown in ex vivo Aβ2m, and some studies actually show that thiol compounds (reductants) inhibit β2m amyloid fibril formation at neutral pH (Yamamoto et al. 2008). Also, while there is no doubt that chronic kidney disease and hemodialysis lead to a proinflammatory and oxidatively stressed environment in the circulation (possibly further enhanced by oxidizing Cu2 +-ions) (Lee et al. 2011) the factors associated with chronic kidney disease and hemodialysis that would be especially conducive to breaking intramolecular disulfide bonds in the core of a tightly packed globular protein remain to be characterized.
Schematical model of β2m amyloidogenesis in vivo in chronic hemodialysis patients. Upper panel (based on Liu et al. 2011) shows the disulfide exchange involved in domain-swapped β2m dimer formation and the proposed structure of a runaway domain-swapped oligomer with inter-dimer disulfide bonds indicated. Lower panel shows a schematic of amyloid formation by β2m. Three stages are envisioned: (A), normal conditions, monomeric β2m displays conformational fluctuations (depicted as closed and open domain forms) but is present at normal concentrations; (B) Increased concentrations in renal disease lead to formation of β2m dimers possibly stabilized by intermolecular disulfide bonding between swapped domains (cf. upper panel); (C) Association, organization and consolidation on collagen of modified and/or dimerized β2m and then further oligomerzation with runaway dimerization as proposed by Eisenberg et al. (Liu et al. 2011) on the seeding surface of collagen-attached β2m
4.3 Accessory Molecules
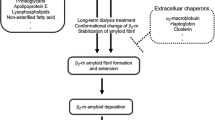
In the attempt to elucidate the early fibrillating events in DRA a number of co-factors have been studied for their ability to elicit and facilitate β2m fibrillogenesis (Table 19.2). Glycosaminoglycans (GAGs) are long unbranched polysaccharide chains consisting of repeating disaccharide units. Heparan sulfate is a glycosaminoglycan which is expressed on cell surfaces and binds non-covalently to a variety of proteins (Jackson et al. 1991). In many of the amyloidoses heparan sulfate has been shown to be a universal component of amyloid (Jackson et al. 1991; Magnus et al. 1991; Snow et al. 1987, 1988, 1991; Snow and Kisilevsky 1985; Young et al. 1989, 1992) and thus this GAG has been proposed to play an active role in amyloid generation by promoting fibrillogenesis (Castillo et al. 1998; Cohlberg et al. 2002; Goedert et al. 1996) rather than being passively accumulated. Heparan sulfate and heparin (which is commonly administered as an anticoagulant during hemodialysis treatment) have been shown to promote β2m amyloid formation in vitro (Borysik et al. 2007) and to exert a stabilizing effect on such β2m fibrils (Myers et al. 2006; Yamaguchi et al. 2003; Yamamoto et al. 2004b). Both uremic serum and synovial fluid also have amyloid-enhancing effects in seeding experiments (Myers et al. 2006). Interestingly, a mouse human β2m model with vastly supraphysiological circulating β2m levels did not show β2m-amyloid formation even in the cases where preformed amyloid fibril seeds were injected (Zhang et al. 2010). This again shows that the specific uremic environment of kidney failure patients is an indispensable prerequisite for the triggering of Aβ2m.
Collagen fibers, which are found in the joint environment, have also been shown to promote β2m fibrillogenesis (Relini et al. 2006) and could be relevant for DRA pathology. The positively charged collagen molecules are proposed to act as an immobilized surface on which the β2m molecules bind and become oriented in a fashion facilitating fibril formation. Also apolipoprotein E (ApoE) which is a cholesterol transport protein has been suggested to be of relevance for amyloidogenesis, since it has been found to be ubiquitously co-localized with amyloid deposits in both systemic and localized amyloidoses. However, reports are contradictory showing both promoting and inhibitory effects of ApoE on fibril formation (Naiki et al. 1997; Wisniewski et al. 1994).
Serum amyloid P component (SAP) is a common component of extracellular matrix in the microfibrillar mantle of elastic fibers and in the glomerular basement membrane. SAP binds to amyloid in a calcium-dependent manner and is a universal constituent of all amyloid deposits, comprising up to ~ 15 % of the amyloid tissue mass (Skinner et al. 1980). SAP itself is highly resistant to proteolysis (Kinoshita et al. 1992) and possesses the ability to also prevent proteolysis of the amyloid fibrils to which it binds (Tennent et al. 1995), thereby possibly contributing to their persistence in vivo. Due to its specificity and affinity for amyloid deposits SAP may be used for radioimaging in vivo of some systemic amyloidoses (Pepys 2006).
4.4 Treatment of β2-microglobulin Amyloidosis
Treatment of amyloidosis is difficult. The general strategy is to treat symptoms secondary to organ damage and to reduce the production of amyloid by restricting the production of precursor molecules (Pepys 2006). In the case of Aβ2m there is no curative treatment except for reconstitution of renal function by renal transplantation (Pepys 2006). This will halt DRA progression but does not revert established β2m amyloid deposits (Labriola et al. 2007; Mourad and Argiles 1996) that must be treated by surgery. It is worth noting that amyloid deposits are not static entities but rather go from a more reversible state when newly formed to much more stable irreversibly consolidated fibril formations over time (Kardos et al. 2011). Dye molecules and GAG analogues have been used experimentally based on the notion of inhibiting interactions with accessory molecules. Screening of sulfonated small molecule, tetracycline analogies, and other antibiotics-based potential fibrillogenesis inhibitors (Giorgetti et al. 2011; Regazzoni et al. 2011) has identified promising compounds that may divert aggregation into non-amyloidogenic pathways (Woods et al. 2011). Also tetracycline (Giorgetti et al. 2011) and proteins such as α2-macroglobulin (Ozawa et al. 2011a) and compounds stabilizing native protein structures have been launched, especially for treating the transthyretin amyloidoses (Adamski-Werner et al. 2004; Sacchettini and Kelly 2002) but have not yet been explored in DRA. In DRA most work is directed at reducing disease progression by depleting the precursors of β2m amyloid by optimized dialysis including Cu2 +-free dialysis membranes (Miura et al. 1992; van Ypersele et al. 1991).
Experimental therapies may be derived from preliminary reports of amyloid fibril destruction by laser irradiation of thioflavin T-fibril complexes which has been shown in vitro both for keratoepithelin and β2m-derived peptide fibrils (Ozawa et al. 2011b; Ozawa et al. 2009). The clinical implementation of fibril destruction by amyloid dye-excitation, however, is far from straightforward.
5 Conclusions and Future Directions
The list of factors that have been shown to influence the conformation of intact β2m is very long. In DRA-patients, the increased serum concentration is important, but there is no simple correlation between DRA-severity and circulating β2m concentration (Gejyo et al. 1986), and β2m-amyloid has not been observed in other diseases where there are sustained, elevated β2m concentrations. Furthermore, many of the in vitro conditions that are highly favorable for amyloid formation from normal β2m such as very low pH and very high ionic strength are not encountered in vivo. Some of the factors that may be relevant enhancers and inhibitors of β2m amyloidogenicity in vivo are listed in Table 19.2. In this regard the catalysis of amyloidogenic conformations by divalent cations, especially Cu2 + and its relationship to Pro-32 trans-to-cis conversion, at neutral pH (Eakin et al. 2002, 2004, 2006) is very interesting. Most, if not all, amyloid proteins have affinities for divalent cations, and metal ions have been proposed as a triggering factor in Alzheimer’s disease (Atwood et al. 2000) even though the evidence linking Cu2 + to enhancement or inhibition of amyloid formation by amyloid β-peptides is ambiguous (Pedersen et al. 2011). Metals are also involved in Parkinson’s disease (Uversky et al. 2001), immunoglobulin light chain (Davis et al. 2001), and prion protein amyloidogenesis (Jackson et al. 2001). Furthermore, the presence of a subfraction of cleaved β2m-species in the amyloid deposits (Linke et al. 1986, 1987, 1989; Stoppini et al. 2000) and of another cleaved variant in the circulation of hemodialysis patients—especially those treated with Cu2 +-containing, complement-activating dialysis membranes and the demonstration of the decreased conformational stability in vitro of such truncated variants (Corazza et al. 2004; Esposito et al. 2000; Heegaard et al. 2005) together with their catalytic effects on amyloidogenesis (Eichner and Radford 2011) provide strong indications of the pathogenetic importance of post-translationally modified β2m for the development of β2m amyloidosis in dialysis patients.
The possibility that several factors may interplay in different ways in vivo and yet lead to the β2m amyloidosis syndrome DRA is unlikely since the syndrome is limited to such a well-defined group of patients. In that regard it is an interesting model disease for all types of amyloid.
With a normal half-life in solution of only one hour it is likely that monomeric β2m may be regarded as a conformationally unstable waste product with unfavorable exposure of hydrophobic side chains in the solvent exposed DE-loop which is otherwise designed to be stabilized by fitting into the MHC class I receptor complex in which β2m fulfils its proper physiological function.
Abbreviations
- AA:
-
Amino Acid
- Aβ2m:
-
β2-microglobulin amyloidosis
- β2m:
-
β2-microglobulin
- DRA:
-
Dialysis-Related Amyloidosis
- GAG:
-
Glycosaminoglycan
- GFR:
-
Glomerular Filtration Rate
- MHC:
-
Major Histocompatibility Complex
- SAP:
-
Serum Amyloid P component
References
Adamski-Werner SL, Palaninathan SK, Sacchettini JC, Kelly JW (2004) Diflunisal analogues stabilize the native state of transthyretin. Potent inhibition of amyloidogenesis. J Med Chem 47:355–374
Ahn M, Kang S, Koo HJ, Lee JH, Lee YS, Paik SR (2010) Nanoporous protein matrix made of amyloid fibrils of beta2-microglobulin. Biotechnol Prog 26:1759–1764
Annuk M, Soveri I, Zilmer M, Lind L, Hulthe J, Fellstrom B (2005) Endothelial function, CRP and oxidative stress in chronic kidney disease. J Nephrol 18:721–726.
Argiles A, Mourad G, Axelrud-Cavadore C, Watrin A, Mion C, Cavadore JC (1989) High-molecular-mass proteins in haemodialysis-associated amyloidosis. Clin Sci (Lond) 76:547–552
Argiles A, Derancourt J, Jauregui-Adell J, Mion C, Demaille JG (1992) Biochemical characterization of serum and urinary beta 2 microglobulin in end-stage renal disease patients. Nephrol Dial Transplant 7:1106–1110
Argiles A, Garcia-Garcia M, Derancourt J, Mourad G, Demaille JG (1995) Beta 2 microglobulin isoforms in healthy individuals and in amyloid deposits. Kidney Int 48:1397–1405
Assenat H, Calemard E, Charra B, Laurent G, Terrat JC, Vanel T (1980) Hémodialyse, syndrome du canal carpien et substance amyloïd. Nouv Presse Med 24:1715
Atwood CS, Scarpa RC, Huang X, Moir RD, Jones WD, Fairlie DP, Tanzi RE, Bush AI (2000) Characterization of copper interactions with alzheimer amyloid beta peptides: identification of an attomolar-affinity copper binding site on amyloid beta1-42. J Neurochem 75:1219–1233
Azinas S, Colombo M, Barbiroli A et al (2011) D-strand perturbation and amyloid propensity in beta-2 microglobulin. FEBS J 278:2349–2358
Barbet-Massin E, Ricagno S, Lewandowski JR, Giorgetti S, Bellotti V, Bolognesi M, Emsley L, Pintacuda G (2010) Fibrillar vs crystalline full-length beta-2-microglobulin studied by high-resolution solid-state NMR spectroscopy. J Am Chem Soc 132:5556–5557
Bartl R, Frisch B, Diem H, Mundel M, Fateh-Moghadam A (1989) Bone marrow histology and serum beta 2 microglobulin in multiple myeloma—a new prognostic strategy. Eur J Haematol Suppl 51:88–98
Baz M, Durand C, Ragon A, Jaber K, Andrieu D, Merzouk T, Purgus R, Olmer M, Reynier JP, Berland Y (1991) Using ultrapure water in hemodialysis delays carpal tunnel syndrome. Int J Artif Organs 14:681–685
Bellotti V, Stoppini M, Mangione P, Sunde M, Robinson C, Asti L, Brancaccio D, Ferri G (1998) Beta2-microglobulin can be refolded into a native state from ex vivo amyloid fibrils. Eur J Biochem 258:61–67
Berggård I (1965) Identification and isolation of urinary proteins. In: Peeters H (ed) Protides of the biological fluids (Proceedings of the 12th Colloquium, Bruges, 1964), Elsevier, Amsterdam, pp 285–291
Berggard I, Bearn AG (1968) Isolation and properties of a low molecular weight beta-2-globulin occurring in human biological fluids. J Biol Chem 243:4095–4103
Bhalla RB, Safai B, Pahwa S, Schwartz MK (1985) Beta 2-microglobulin as a prognostic marker for development of AIDS. Clin Chem 31:1411–1412
Bjorck L, Akerstrom B, Berggard I (1979) Occurrence of beta 2-microglobulin in mammalian lymphocytes and erythrocytes. Eur J Immunol 9:486–490
Borysik AJ, Morten IJ, Radford SE, Hewitt EW (2007) Specific glycosaminoglycans promote unseeded amyloid formation from beta2-microglobulin under physiological conditions. Kidney Int 72:174–181
Bourantas KL, Hatzimichael EC, Makis AC, Chaidos A, Kapsali ED, Tsiara S, Mavridis A (1999) Serum beta-2-microglobulin, TNF-alpha and interleukins in myeloproliferative disorders. Eur J Haematol 63:19–25
Brancaccio D, Gallieni M, Niwa T, Braidotti P, Coggi G (2000) Ultrastructural localization of advanced glycation end products and beta2-microglobulin in dialysis amyloidosis. J Nephrol 13:129–136
Buchholz NP, Moch H, Gasser TC, Linke RP, Thiel GT, Mihatsch MJ (1995) Ureteral amyloid deposits of beta 2-microglobulin origin in both kidney recipients of 1 donor. J Urol 153:399–401
Calabrese MF, Eakin CM, Wang JM, Miranker AD (2008) A regulatable switch mediates self-association in an immunoglobulin fold. Nat Struct Mol Biol 15:965–971
Campistol JM, Torregrosa JV, Ponz E, Fenollosa B (1999) Beta-2-microglobulin removal by hemodialysis with polymethylmethacrylate membranes. Contrib Nephrol 125:76–85
Canale C, Relini A, Gliozzi A (2011) Atomic force microscopy of ex vivo amyloid fibrils. Methods Mol Biol 736:81–95
Castillo GM, Cummings JA, Yang W, Judge ME, Sheardown MJ, Rimvall K, Hansen JB, Snow AD (1998) Sulfate content and specific glycosaminoglycan backbone of perlecan are critical for perlecan’s enhancement of islet amyloid polypeptide (amylin) fibril formation. Diabetes 47:612–620
Charra B, Calemard F, Uzan M, Terrat JC, Vanel T, Laurent G (1984) Carpal tunnel syndrome, shoulder pain and amyloid deposits in long-term hemodialysis patients. Proc Eur Dial Transplant Assoc 21:291–295
Cheung AK, Rocco MV, Yan G et al (2006) Serum beta-2 microglobulin levels predict mortality in dialysis patients: results of the HEMO study. J Am Soc Nephrol 17:546–555
Cohlberg JA, Li J, Uversky VN, Fink AL (2002) Heparin and other glycosaminoglycans stimulate the formation of amyloid fibrils from alpha-synuclein in vitro. Biochemistry 41:1502–1511
Colombo M, Ricagno S, Barbiroli A, Santambrogio C, Giorgetti S, Raimondi S, Bonomi F, Grandori R, Bellotti V, Bolognesi M (2011) The effects of an ideal beta-turn on beta-2 microglobulin fold stability. J Biochem 150:39–47
Corazza A, Pettirossi F, Viglino P et al (2004) Properties of some variants of human beta2-microglobulin and amyloidogenesis. J Biol Chem 279:9176–9189
Corlin DB, Sen JW, Ladefoged S, Lund GB, Nissen MH, Heegaard NH (2005) Quantification of cleaved b2-microglobulin in serum from patients undergoing chronic hemodialysis. Clin Chem 51:1177–1184
Corlin DB, Johnsen CK, Nissen MH, Heegaard NH (2009) A beta2-microglobulin cleavage variant fibrillates at near-physiological pH. Biochem Biophys Res Commun 381:187–191
Corlin DB, Johnsen CK, Nissen MH, Heegaard NH (2010) Glycosaminoglycans enhance the fibrillation propensity of the beta2-microglobulin cleavage variant--DeltaK58-beta2m. Biochem Biophys Res Commun 402:247–251
Cresswell P, Springer T, Strominger JL, Turner MJ, Grey HM, Kubo RT (1974) Immunological identity of the small subunit of HL-A antigens and beta2-microglobulin and its turnover on the cell membrane. Proc Natl Acad Sci U S A 71:2123–2127
Danesh F, Ho LT (2001) Dialysis-related amyloidosis: history and clinical manifestations. Semin Dial 14:80–85
Davis DP, Gallo G, Vogen SM, Dul JL, Sciarretta KL, Kumar A, Raffen R, Stevens FJ, Argon Y (2001) Both the environment and somatic mutations govern the aggregation pathway of pathogenic immunoglobulin light chain. J Mol Biol 313:1021–1034
De Lorenzi E, Colombo R, Sabella S, Corlin DB, Heegaard NH (2008) The influence of Cu(2 +) on the unfolding and refolding of intact and proteolytically processed beta(2)-microglobulin. Electrophoresis 29:1734–1740
Debelouchina GT, Platt GW, Bayro MJ, Radford SE, Griffin RG (2010) Intermolecular alignment in beta(2)-microglobulin amyloid fibrils. J Am Chem Soc. [Epub ahead of print]
Deng NJ, Yan L, Singh D, Cieplak P (2006) Molecular basis for the Cu2 + binding-induced destabilization of beta2-microglobulin revealed by molecular dynamics simulation. Biophys J 90:3865–3879
Doherty PC, Zinkernagel RM (1975) H-2 compatibility is required for T-cell-mediated lysis of target cells infected with lymphocytic choriomeningitis virus. J Exp Med 141:502–507
Domanska K, Vanderhaegen S, Srinivasan V et al (2011) Atomic structure of a nanobody-trapped domain-swapped dimer of an amyloidogenic beta2-microglobulin variant. Proc Natl Acad Sci U S A 108:1314–1319
Drueke TB, Massy ZA (2009) Beta2-microglobulin. Semin Dial 22:378–380
Eakin CM, Miranker AD (2005) From chance to frequent encounters: origins of beta2-microglobulin fibrillogenesis. Biochim Biophys Acta 1753:92–99
Eakin CM, Knight JD, Morgan CJ, Gelfand MA, Miranker AD (2002) Formation of a copper specific binding site in non-native states of beta-2-microglobulin. Biochemistry 41:10646–10656
Eakin CM, Attenello FJ, Morgan CJ, Miranker AD (2004) Oligomeric assembly of native-like precursors precedes amyloid formation by beta-2 microglobulin. Biochemistry 43:7808–7815
Eakin CM, Berman AJ, Miranker AD (2006) A native to amyloidogenic transition regulated by a backbone trigger. Nat Struct Mol Biol 13:202–208
Ecroyd H, Thorn DC, Liu Y, Carver JA (2010) The dissociated form of kappa-casein is the precursor to its amyloid fibril formation. Biochem J 429:251–260
Eichner T, Radford SE (2009) A generic mechanism of beta2-microglobulin amyloid assembly at neutral pH involving a specific proline switch. J Mol Biol 386:1312–1326
Eichner T, Radford SE (2011) Understanding the complex mechanisms of beta(2) -microglobulin amyloid assembly. FEBS J 278:3868–3883
Esposito G, Michelutti R, Verdone G et al (2000) Removal of the N-terminal hexapeptide from human beta2-microglobulin facilitates protein aggregation and fibril formation. Protein Sci 9:831–845
Esposito G, Ricagno S, Corazza A et al (2008) The controlling roles of Trp60 and Trp95 in beta2-microglobulin function, folding and amyloid aggregation properties. J Mol Biol 378:887–897
Fabian H, Gast K, Laue M, Misselwitz R, Uchanska-Ziegler B, Ziegler A, Naumann D (2008) Early stages of misfolding and association of beta2-microglobulin: insights from infrared spectroscopy and dynamic light scattering. Biochemistry 47:6895–6906
Feder JN, Tsuchihashi Z, Irrinki A et al (1997) The hemochromatosis founder mutation in HLA-H disrupts beta2-microglobulin interaction and cell surface expression. J Biol Chem 272:14025–14028
Floege J, Ehlerding G (1996) Beta-2-microglobulin-associated amyloidosis. Nephron 72:9–26
Fujimori A (2011) Beta-2-microglobulin as a uremic toxin: the Japanese experience. Contrib Nephrol 168:129–33. [Epub 2010, Oct 7]
Garcia-Garcia M, Gouin-Charnet A, Mourad G, Argilés A (1997) Monomeric and dimeric b2-microglobulin may be extracted from amyloid deposits in vitro. Nephrol Dial Transplant 12:1192–1198
Gejyo F, Yamada T, Odani S et al (1985) A new form of amyloid protein associated with chronic hemodialysis was identified as beta 2-microglobulin. Biochem Biophys Res Commun 129:701–706
Gejyo F, Homma N, Suzuki Y, Arakawa M (1986) Serum levels of beta 2-microglobulin as a new form of amyloid protein in patients undergoing long-term hemodialysis. N Engl J Med 314:585–586
Giorgetti S, Rossi A, Mangione P et al (2005) Beta2-microglobulin isoforms display an heterogeneous affinity for type I collagen. Protein Sci 14:696–702
Giorgetti S, Stoppini M, Tennent GA et al (2007) Lysine 58-cleaved beta2-microglobulin is not detectable by 2D electrophoresis in ex vivo amyloid fibrils of two patients affected by dialysis-related amyloidosis. Protein Sci 16:343–349
Giorgetti S, Raimondi S, Pagano K et al (2011) Effect of tetracyclines on the dynamics of formation and destructuration of beta2-microglobulin amyloid fibrils. J Biol Chem 286:2121–2131
Gobezie R, Kho A, Krastins B, Sarracino DA, Thornhill TS, Chase M, Millett PJ, Lee DM (2007) High abundance synovial fluid proteome: distinct profiles in health and osteoarthritis. Arthritis Res Ther 9:R36
Goedert M, Jakes R, Spillantini MG, Hasegawa M, Smith MJ, Crowther RA (1996) Assembly of microtubule-associated protein tau into Alzheimer-like filaments induced by sulphated glycosaminoglycans. Nature 383:550–553
Gorevic PD, Casey TT, Stone WJ, DiRaimondo CR, Prelli FC, Frangione B (1985) Beta-2 microglobulin is an amyloidogenic protein in man. J Clin Invest 76:2425–2429
Gorevic PD, Munoz PC, Casey TT, DiRaimondo CR, Stone WJ, Prelli FC, Rodrigues MM, Poulik MD, Frangione B (1986) Polymerization of intact b2-microglobulin in tissue causes amyloidosis in patients on chronic hemodialysis. Proc Natl Acad Sci U S A 83:7908–7912
Gouin-Charnet A, Laune D, Granier C, Mani JC, Pau B, Mourad G, Argiles A (2000) Alpha2-macroglobulin, the main serum antiprotease, binds beta2-microglobulin, the light chain of the class I major histocompatibility complex, which is involved in human disease. Clin Sci (Lond) 98:427–433
Gumperz JE (2006) The ins and outs of CD1 molecules: bringing lipids under immunological surveillance. Traffic 7:2–13
Hafner-Bratkovic I, Bester R, Pristovsek P et al (2011) Globular domain of the prion protein needs to be unlocked by domain swapping to support prion protein conversion. J Biol Chem 286:12149–12156
Hasegawa K, Tsutsumi-Yasuhara S, Ookoshi T, Ohhashi Y, Kimura H, Takahashi N, Yoshida H, Miyazaki R, Goto Y, Naiki H (2008) Growth of beta(2)-microglobulin-related amyloid fibrils by non-esterified fatty acids at a neutral pH. Biochem J 416:307–315
Hashimoto N, Naiki H, Gejyo F (1999) Modification of beta 2-microglobulin with D-glucose or 3-deoxyglucosone inhibits A beta 2M amyloid fibril extension in vitro. Amyloid 6:256–264
Heegaard NH (2009) Beta(2)-microglobulin: from physiology to amyloidosis. Amyloid 16:151–173
Heegaard NH, Roepstorff P, Melberg SG, Nissen MH (2002) Cleaved beta 2-microglobulin partially attains a conformation that has amyloidogenic features. J Biol Chem 277:11184–11189
Heegaard NHH, Jørgensen TJD, Rozlosnik N, Corlin DB, Pedersen JS, Tempesta AG, Roepstorff P, Bauer R, Nissen MH (2005) Unfolding, aggregation, and seeded amyloid formation of Lysine-58-cleaved b2-microglobulin. Biochemistry 44:4397–4407
Hodkinson JP, Jahn TR, Radford SE, Ashcroft AE (2009) HDX-ESI-MS reveals enhanced conformational dynamics of the amyloidogenic protein beta(2)-microglobulin upon release from the MHC-1. J Am Soc Mass Spectrum 20:278–286
Hoshino M, Katou H, Hagihara Y, Hasegawa K, Naiki H, Goto Y (2002) Mapping the core of the beta(2)-microglobulin amyloid fibril by H/D exchange. Nat Struct Biol 9:332–336
Jackson RL, Busch SJ, Cardin AD (1991) Glycosaminoglycans: molecular properties, protein interactions, and role in physiological processes. Physiol Rev 71:481–539
Jackson GS, Murray I, Hosszu LL, Gibbs N, Waltho JP, Clarke AR, Collinge J (2001) Location and properties of metal-binding sites on the human prion protein. Proc Natl Acad Sci U S A 98:8531–8535
Jadoul M (1998) Dialysis-related amyloidosis: importance of biocompatibility and age. Nephrol Dial Transplant 13(Suppl 7):61–64
Jadoul M, Garbar C, Noel H, Sennesael J, Vanholder R, Bernaert P, Rorive G, Hanique G, van Ypersele de SC (1997) Histological prevalence of beta 2-microglobulin amyloidosis in hemodialysis: a prospective post-mortem study. Kidney Int 51:1928–1932
Jahn TR, Parker MJ, Homans SW, Radford SE (2006) Amyloid formation under physiological conditions proceeds via a native-like folding intermediate. Nat Struct Mol Biol 13:195–201
Jahn TR, Makin OS, Morris KL, Marshall KE, Tian P, Sikorski P, Serpell LC (2010) The common architecture of cross-beta amyloid. J Mol Biol 395:717–727
Jones S, Manning J, Kad NM, Radford SE (2003) Amyloid-forming peptides from beta2-microglobulin-Insights into the mechanism of fibril formation in vitro. J Mol Biol 325:249–257
Jørgensen TJD, Cheng L, Heegaard NHH (2007) Mass spectrometric characterization of conformational preludes to b2-microglobulin aggregation. 268:207–216
Kad NM, Thomson NH, Smith DP, Smith DA, Radford SE (2001) Beta(2)-microglobulin and its deamidated variant, N17D form amyloid fibrils with a range of morphologies in vitro. J Mol Biol 313:559–571
Kameda A, Hoshino M, Higurashi T, Takahashi S, Naiki H, Goto Y (2005) Nuclear magnetic resonance characterization of the refolding intermediate of beta2-microglobulin trapped by non-native prolyl peptide bond. J Mol Biol 348:383–397
Kang SJ, Cresswell P (2002) Calnexin, calreticulin, and ERp57 cooperate in disulfide bond formation in human CD1d heavy chain. J Biol Chem 277:44838–44844
Kardos J, Micsonai A, Pal-Gabor H, Petrik E, Graf L, Kovacs J, Lee YH, Naiki H, Goto Y (2011) Reversible heat-induced dissociation of beta2-microglobulin amyloid fibrils. Biochemistry 19(50):3211–3220
Karlsson FA, Wibell L, Evrin PE (1980) b2-microglobulin in clinical medicine. Scand J Clin Lab Invest 40(Suppl 154):27–37
Kawano M, Muramoto H, Yamada M, Minamoto M, Araki H, Koni I, Mabuchi H, Nonomura A (1998) Fatal cardiac beta2-microglobulin amyloidosis in patients on long-term hemodialysis. Am J Kidney Dis 31:E4
Khan AR, Baker BM, Ghosh P, Biddison WE, Wiley DC (2000) The structure and stability of an HLA-A*0201/octameric tax peptide complex with an empty conserved peptide-N-terminal binding site. J Immunol 164:6398–6405
Kinoshita CM, Gewurz AT, Siegel JN, Ying SC, Hugli TE, Coe JE, Gupta RK, Huckman R, Gewurz H (1992) A protease-sensitive site in the proposed Ca(2 +)-binding region of human serum amyloid P component and other pentraxins. Protein Sci 1:700–709
Kiss E, Keusch G, Zanetti M, Jung T, Schwarz A, Schocke M, Jaschke W, Czermak BV (2005) Dialysis-related amyloidosis revisited. AJR Am J Roentgenol 185:1460–1467
Knaus KJ, Morillas M, Swietnicki W, Malone M, Surewicz WK, Yee VC (2001) Crystal structure of the human prion protein reveals a mechanism for oligomerization. Nat Struct Biol 8:770–774
Kuntz D, Naveau B, Bardin T, Drueke T, Treves R, Dryll A (1984) Destructive spondylarthropathy in hemodialyzed patients. A new syndrome. Arthritis Rheum 27:369–375
Kwon HK, Pyun SB, Cho WY, Boo CS (2011) Carpal tunnel syndrome and peripheral polyneuropathy in patients with end stage kidney disease. J Korean Med Sci 26:1227–1230
Labriola L, Garbar C, Jadoul M (2007) Persistence of beta2-microglobulin amyloidosis 20 years after successful kidney transplantation. Am J Kidney Dis 50:167–168
Lee S, Eisenberg D (2003) Seeded conversion of recombinant prion protein to a disulfide-bonded oligomer by a reduction-oxidation process. Nat Struct Biol 10:725–730
Lee DM, Jackson KW, Knowlton N, Wages J, Alaupovic P, Samuelsson O, Saeed A, Centola M, Attman PO (2011) Oxidative stress and inflammation in renal patients and healthy subjects. PLoS ONE 6:e22360.
Linke RP, Bommer J, Ritz E, Waldherr R, Eulitz M (1986) Amyloid kidney stones of uremic patients consist of beta 2-microglobulin fragments. Biochem Biophys Res Commun 136:665–671
Linke RP, Hampl H, Bartel-Schwarze S, Eulitz M (1987) Beta 2-microglobulin, different fragments and polymers thereof in synovial amyloid in long-term hemodialysis. Biol Chem Hoppe Seyler 368:137–144
Linke RP, Hampl H, Lobeck H, Ritz E, Bommer J, Waldherr R, Eulitz M (1989) Lysine-specific cleavage of beta 2-microglobulin in amyloid deposits associated with hemodialysis. Kidney Int 36:675–681
Liu C, Sawaya MR, Eisenberg D (2011) beta-Microglobulin forms three-dimensional domain-swapped amyloid fibrils with disulfide linkages. Nat Struct Mol Biol 18:49–55
Liu H, Chumsae C, Gaza-Bulseco G, Hurkmans K, Radziejewski CH (2010) Ranking the susceptibility of disulfide bonds in human IgG1 antibodies by reduction, differential alkylation, and LC-MS analysis. Anal Chem 82:5219–5226
Locatelli F, Marcelli D, Conte F, Limido A, Malberti F, Spotti D (1999) Comparison of mortality in ESRD patients on convective and diffusive extracorporeal treatments. The Registro Lombardo Dialisi E Trapianto. Kidney Int 55:286–293
Magnus JH, Stenstad T, Kolset SO, Husby G (1991) Glycosaminoglycans in extracts of cardiac amyloid fibrils from familial amyloid cardiomyopathy of Danish origin related to variant transthyretin Met 111. Scand J Immunol 34:63–69
McParland VJ, Kad NM, Kalverda AP, Brown A, Kirwin-Jones P, Hunter MG, Sunde M, Radford SE (2000) Partially unfolded states of beta(2)-microglobulin and amyloid formation in vitro. Biochemistry 39:8735–8746
Mendoza VL, Antwi K, Baron-Rodriguez MA, Blanco C, Vachet RW (2010) Structure of the preamyloid dimer of beta-2-microglobulin from covalent labeling and mass spectrometry. Biochemistry 49:1522–1532
Mendoza VL, Baron-Rodriguez MA, Blanco C, Vachet RW (2011) Structural insights into the pre-amyloid tetramer of beta-2-microglobulin from covalent labeling and mass spectrometry. Biochemistry 50:6711–6722
Mimmi MC, Jorgensen TJ, Pettirossi F et al (2006) Variants of beta-microglobulin cleaved at lysine-58 retain the main conformational features of the native protein but are more conformationally heterogeneous and unstable at physiological temperature. FEBS J 273:2461–2474
Miura Y, Ishiyama T, Inomata A, Takeda T, Senma S, Okuyama K, Suzuki Y (1992) Radiolucent bone cysts and the type of dialysis membrane used in patients undergoing long-term hemodialysis. Nephron 60:268–273
Miyata T, Oda O, Inagi R, Iida Y, Araki N, Yamada N, Horiuchi S, Taniguchi N, Maeda K, Kinoshita T (1993) beta 2-Microglobulin modified with advanced glycation end products is a major component of hemodialysis-associated amyloidosis. J Clin Invest 92:1243–1252
Miyata T, Taneda S, Kawai R, Ueda Y, Horiuchi S, Hara M, Maeda K, Monnier VM (1996) Identification of pentosidine as a native structure for advanced glycation end products in b2-microglobulin-containing amyloid fibrils in patients with dialysis-related amyloidosis. Proc Natl Acad Sci U S A 93:2353–2358
Mogyorosi A, Schubert ML (1999) Dialysis-related amyloidosis: an important cause of gastrointestinal symptoms in patients with end-stage renal disease. Gastroenterology 116:217–220
Morgan CJ, Gelfand M, Atreya C, Miranker AD (2001) Kidney dialysis-associated amyloidosis: a molecular role for copper in fiber formation. J Mol Biol 309:339–345
Motomiya Y, Ando Y, Haraoka K, Sun X, Iwamoto H, Uchimura T, Maruyama I (2003) Circulating level of alpha2-macroglobulin-beta2-microglobulin complex in hemodialysis patients. Kidney Int 64:2244–2252
Mount SL, Eltabbakh GH, Hardin NJ (2002) Beta-2 microglobulin amyloidosis presenting as bilateral ovarian masses: a case report and review of the literature. Am J Surg Pathol 26:130–133
Mourad G, Argiles A (1996) Renal transplantation relieves the symptoms but does not reverse beta 2-microglobulin amyloidosis. J Am Soc Nephrol 7:798–804
Myers SL, Jones S, Jahn TR, Morten IJ, Tennent GA, Hewitt EW, Radford SE (2006) A systematic study of the effect of physiological factors on b2-microglobulin amyloid formation at neutral pH. Biochemistry 45:2311–2321
Naiki H, Gejyo F, Nakakuki K (1997) Concentration-dependent inhibitory effects of apolipoprotein E on Alzheimer’s beta-amyloid fibril formation in vitro. Biochemistry 36:6243–6250
Nilsson M, Wang X, Rodziewicz-Motowidlo S, Janowski R, Lindstrom V, Onnerfjord P, Westermark G, Grzonka Z, Jaskolski M, Grubb A (2004) Prevention of domain swapping inhibits dimerization and amyloid fibril formation of cystatin C: use of engineered disulfide bridges, antibodies, and carboxymethylpapain to stabilize the monomeric form of cystatin C. J Biol Chem 279:24236–24245
Nissen MH, Thim L, Christensen M (1987) Purification and biochemical characterization of the complete structure of a proteolytically modified beta-2-microglobulin with biological activity. Eur J Biochem 163:21–28
Nissen MH, Roepstorff P, Thim L, Dunbar B, Fothergill JE (1990) Limited proteolysis of beta 2-microglobulin at Lys-58 by complement component C1s. Eur J Biochem 189:423–429
Nissen MH, Johansen B, Bjerrum OJ (1997) A simple method for the preparation and purification of C1 complement cleaved beta 2-microglobulin from human serum. J Immunol Methods 205:29–33
Niwa T, Katsuzaki T, Momoi T, Miyazaki T, Ogawa H, Saito A, Miyazaki S, Maeda K, Tatemichi N, Takei Y (1996) Modification of beta 2m with advanced glycation end products as observed in dialysis-related amyloidosis by 3-DG accumulating in uremic serum. Kidney Int 49:861–867
Niwa T, Katsuzaki T, Miyazaki S, Momoi T, Akiba T, Miyazaki T, Nokura K, Hayase F, Tatemichi N, Takei Y (1997) Amyloid b2-microglobulin is modified with imidazolone, a novel advanced glycation end product, in dialysis-related amyloidosis. Kidney Int 51:187–194
Niwa T, Tsukushi S (2001) 3-Deoxyglucosone and AGEs in uremic complications: inactivation of glutathione peroxidase by 3-deoxyglucosone. Kidney Int Suppl 78:S37-S41
Odani H, Oyama R, Titani K, Ogawa H, Saito A (1990) Purification and complete amino acid sequence of novel b2-microglobulin. Biochem Biophys Res Commun 168:1223–1229
Ogawa H, Saito A, Oda O, Nakajima M, Chung TG (1988) Detection of novel beta 2-microglobulin in the serum of hemodialysis patients and its amyloidogenic predisposition. Clin Nephrol 30:158–163
Ohhashi Y, Kihara M, Naiki H, Goto Y (2005) Ultrasonication-induced amyloid fibril formation of beta2-microglobulin. J Biol Chem 280:32843–32848
Okon M, Bray P, Vucelic D (1992) 1H NMR assignments and secondary structure of human beta 2-microglobulin in solution. Biochemistry 31:8906–8915
Otten GR, Bikoff E, Ribaudo RK, Kozlowski S, Margulies DH, Germain RN (1992) Peptide and beta 2-microglobulin regulation of cell surface MHC class I conformation and expression. J Immunol 148:3723–3732
Ozawa D, Yagi H, Ban T, Kameda A, Kawakami T, Naiki H, Goto Y (2009) Destruction of amyloid fibrils of a beta2-microglobulin fragment by laser beam irradiation. J Biol Chem 284:1009–1017
Ozawa D, Hasegawa K, Lee YH, Sakurai K, Yanagi K, Ookoshi T, Goto Y, Naiki H (2011a) Inhibition of beta2-microglobulin amyloid fibril formation by alpha2-macroglobulin. J Biol Chem 286:9668–9676
Ozawa D, Kaji Y, Yagi H, Sakurai K, Kawakami T, Naiki H, Goto Y (2011b) Destruction of amyloid fibrils of keratoepithelin peptides by laser irradiation coupled with amyloid-specific thioflavin T. J Biol Chem 286:10856–10863
Pedersen JT, Østergaard J, Rozlosnik N, Gammelgaard B, Heegaard NH (2011) Cu(II) mediates kinetically distinct, non-amyloidogenic aggregation of amyloid-beta peptides. J Biol Chem 286:26952–26963
Pepys MB (2006) Amyloidosis. Annu Rev Med 57:223–241
Platt GW, Routledge KE, Homans SW, Radford SE (2008) Fibril growth kinetics reveal a region of beta2-microglobulin important for nucleation and elongation of aggregation. J Mol Biol 378:251–263
Praetor A, Hunziker W (2002) beta(2)-Microglobulin is important for cell surface expression and pH-dependent IgG binding of human FcRn. J Cell Sci 115:2389–2397
Regazzoni L, Colombo R, Bertoletti L et al (2011) Screening of fibrillogenesis inhibitors of beta2-microglobulin: integrated strategies by mass spectrometry capillary electrophoresis and in silico simulations. Anal Chim Acta 685:153–161
Relini A, Canale C, De SS et al (2006) Collagen plays an active role in the aggregation of beta2-microglobulin under physiopathological conditions of dialysis-related amyloidosis. J Biol Chem 281:16521–16529
Richardson JS, Richardson DC (2002) Natural beta-sheet proteins use negative design to avoid edge-to-edge aggregation. Proc Natl Acad Sci U S A 99:2754–2759
Sacchettini JC, Kelly JW (2002) Therapeutic strategies for human amyloid diseases. Nat Rev Drug Discov 1:267–275
Saito A, Pietromonaco S, Loo AK, Farquhar MG (1994) Complete cloning and sequencing of rat gp330/“megalin,” a distinctive member of the low density lipoprotein receptor gene family. Proc Natl Acad Sci U S A 91:9725–9729
Sambashivan S, Liu Y, Sawaya MR, Gingery M, Eisenberg D (2005) Amyloid-like fibrils of ribonuclease A with three-dimensional domain-swapped and native-like structure. Nature 437:266–269
Santambrogio C, Ricagno S, Colombo M, Barbiroli A, Bonomi F, Bellotti V, Bolognesi M, Grandori R (2010) DE-loop mutations affect beta2 microglobulin stability, oligomerization, and the low-pH unfolded form. Protein Sci 19:1386–1394
Saper MA, Bjorkman PJ, Wiley DC (1991) Refined structure of the human histocompatibility antigen HLA-A2 at 2.6A resolution. J Mol Biol 219:277–319
Sasahara K, Yagi H, Naiki H, Goto Y (2007) Heat-induced conversion of beta(2)-microglobulin and hen egg-white lysozyme into amyloid fibrils. J Mol Biol 372:981–991
Schneider A, Drechsler C, Krane V, Krieter DH, Scharnagl H, Schneider MP, Wanner C (2011) The effect of high-flux hemodialysis on hemoglobin concentrations in patients with CKD: results of the MINOXIS study. Clin J Am Soc Nephrol 7(1):52–59
Schwalbe S, Holzhauer M, Schaeffer J, Galanski M, Koch KM, Floege J (1997) Beta 2-microglobulin associated amyloidosis: a vanishing complication of long-term hemodialysis? Kidney Int 52:1077–1083
Shimizu S, Yasui C, Yasukawa K, Nakamura H, Shimizu H, Tsuchiya K (2003) Subcutaneous nodules on the buttocks as a manifestation of dialysis-related amyloidosis: a clinicopathological entity? Br J Dermatol 149:400–404
Skinner M, Pepys MB, Cohen AS, Heller LM, Lian JB (1980) In: Freitas AF de, Glenner GG, Costa PP (eds) Amyloid and amyloidosis: proceedings of the Third International Symposium on Amyloidosis, Póvoa de Varzim, Portugal, 23–28 September 1979. Amsterdam: Excerpta Medica, pp 384–391
Smith DP, Jones S, Serpell LC, Sunde M, Radford SE (2003) A systematic investigation into the effect of protein destabilisation on beta 2-microglobulin amyloid formation. J Mol Biol 330:943–954
Snow AD, Kisilevsky R (1985) Temporal relationship between glycosaminoglycan accumulation and amyloid deposition during experimental amyloidosis. A histochemical study. Lab Invest 53:37–44
Snow AD, Willmer J, Kisilevsky R (1987) A close ultrastructural relationship between sulfated proteoglycans and AA amyloid fibrils. Lab Invest 57:687–698
Snow AD, Mar H, Nochlin D, Kimata K, Kato M, Suzuki S, Hassell J, Wight TN (1988) The presence of heparan sulfate proteoglycans in the neuritic plaques and congophilic angiopathy in Alzheimer’s disease. Am J Pathol 133:456–463
Snow AD, Bramson R, Mar H, Wight TN, Kisilevsky R (1991) A temporal and ultrastructural relationship between heparan sulfate proteoglycans and AA amyloid in experimental amyloidosis. J Histochem Cytochem 39:1321–1330
Stoppini MS, Arcidiaco P, Mangione P, Giorgetti S, Brancaccio D, Bellotti V (2000) Detection of fragments of beta2-microglobulin in amyloid fibrils. Kidney Int 57:349–350
Stoppini M, Mangione P, Monti M et al (2005) Proteomics of beta2-microglobulin amyloid fibrils. Biochim Biophys Acta 1753:23–33
Takayama F, Miyazaki S, Morita T, Hirasawa Y, Niwa T (2001) Dialysis-related amyloidosis of the heart in long-term hemodialysis patients. Kidney Int Suppl 78:S172-S176
Tang X, Sadeghi M, Olumee Z, Vertes A, Braatz JA, McIlwain LK, Dreifuss PA (1996) Detection and quantitation of beta-2-microglobulin glycosylated end products in human serum by matrix-assisted laser desorption/ionization mass spectrometry. Anal Chem 68:3740–3745
Tennent GA, Lovat LB, Pepys MB (1995) Serum amyloid P component prevents proteolysis of the amyloid fibrils of Alzheimer disease and systemic amyloidosis. Proc Natl Acad Sci U S A 92:4299–4303
Terawaki H, Yoshimura K, Hasegawa T, Matsuyama Y, Negawa T, Yamada K, Matsushima M, Nakayama M, Hosoya T, Era S (2004) Oxidative stress is enhanced in correlation with renal dysfunction: examination with the redox state of albumin. Kidney Int 66:1988–1993
Treuhaft PS, MCCarty DJ (1971) Synovial fluid pH, lactate, oxygen and carbon dioxide partial pressure in various joint diseases. Arthritis Rheum 14:475–484
Trinh CH, Smith DP, Kalverda AP, Phillips SE, Radford SE (2002) Crystal structure of monomeric human beta-2-microglobulin reveals clues to its amyloidogenic properties. Proc Natl Acad Sci U S A 99:9771–9776
Uversky VN, Li J, Fink AL (2001) Metal-triggered structural transformations, aggregation, and fibrillation of human alpha-synuclein. A possible molecular NK between Parkinson’s disease and heavy metal exposure. J Biol Chem 276:44284–44296
van Ypersele dS, Jadoul M, Malghem J, Maldague B, Jamart J (1991) Effect of dialysis membrane and patient’s age on signs of dialysis-related amyloidosis. The Working Party on Dialysis Amyloidosis. Kidney Int 39:1012–1019
Verdone G, Corazza A, Viglino P, Pettirossi F, Giorgetti S, Mangione P, Andreola A, Stoppini M, Bellotti V, Esposito G (2002) The solution structure of human beta2-microglobulin reveals the prodromes of its amyloid transition. Protein Sci 11:487–499
Villanueva J, Hoshino M, Katou H, Kardos J, Hasegawa K, Naiki H, Goto Y (2004) Increase in the conformational flexibility of beta 2-microglobulin upon copper binding: a possible role for copper in dialysis-related amyloidosis. Protein Sci 13:797–809
Vincent C, Dennoroy L, Revillard J-P (1994) Molecular variants of b2-microglobulin in renal insufficiency. Biochem J 298:181–187
Vorbeck-Meister I, Sommer R, Vorbeck F, Horl WH (1999) Quality of water used for haemodialysis: bacteriological and chemical parameters. Nephrol Dial Transplant 14:666–675
Wahlbom M, Wang X, Lindstrom V, Carlemalm E, Jaskolski M, Grubb A (2007) Fibrillogenic oligomers of human cystatin C are formed by propagated domain swapping. J Biol Chem 282:18318–18326
Warren DJ, Otieno LS (1975) Carpal tunnel syndrome in patients on intermittent haemodialysis. Postgrad Med J 51:450–452
White HE, Hodgkinson JL, Jahn TR, Cohen-Krausz S, Gosal WS, Muller S, Orlova EV, Radford SE, Saibil HR (2009) Globular tetramers of beta(2)-microglobulin assemble into elaborate amyloid fibrils. J Mol Biol 389:48–57
Williams DB, Barber BH, Flavell RA, Allen H (1989) Role of beta 2-microglobulin in the intracellular transport and surface expression of murine class I histocompatibility molecules. J Immunol 142:2796–2806
Winchester JF, Salsberg JA, Levin NW (2003) Beta-2 microglobulin in ESRD: an in-depth review. Adv Ren Replace Ther 10:279–309
Wisniewski T, Castano EM, Golabek A, Vogel T, Frangione B (1994) Acceleration of Alzheimer’s fibril formation by apolipoprotein E in vitro. Am J Pathol 145:1030–1035
Woods LA, Platt GW, Hellewell AL, Hewitt EW, Homans SW, Ashcroft AE, Radford SE (2011) Ligand binding to distinct states diverts aggregation of an amyloid-forming protein. Nat Chem Biol 7:730–739
Wu AHB (2006) Tietz Clinical Guide to Laboratory Tests, 4th edn. Saunders, St. Louis
Yamaguchi I, Suda H, Tsuzuike N et al (2003) Glycosaminoglycan and proteoglycan inhibit the depolymerization of beta2-microglobulin amyloid fibrils in vitro. Kidney Int 64:1080–1088
Yamamoto S, Hasegawa K, Yamaguchi I, Tsutsumi S, Kardos J, Goto Y, Gejyo F, Naiki H (2004a) Low concentrations of sodium dodecyl sulfate induce the extension of beta 2-microglobulin-related amyloid fibrils at a neutral pH. Biochemistry 43:11075–11082
Yamamoto S, Yamaguchi I, Hasegawa K, Tsutsumi S, Goto Y, Gejyo F, Naiki H (2004b) Glycosaminoglycans enhance the trifluoroethanol-induced extension of beta 2-microglobulin-related amyloid fibrils at a neutral pH. J Am Soc Nephrol 15:126–133
Yamamoto K, Yagi H, Ozawa D, Sasahara K, Naiki H, Goto Y (2008) Thiol compounds inhibit the formation of amyloid fibrils by beta 2-microglobulin at neutral pH. J Mol Biol 376:258–268
Yamamoto S, Kazama JJ, Narita I, Naiki H, Gejyo F (2009) Recent progress in understanding dialysis-related amyloidosis. Bone. 45(Suppl 1):S39–S42. [Epub 2009, Mar 19]
Young ID, Willmer JP, Kisilevsky R (1989) The ultrastructural localization of sulfated proteoglycans is identical in the amyloids of Alzheimer’s disease and AA, AL, senile cardiac and medullary carcinoma-associated amyloidosis. Acta Neuropathol 78:202–209
Young ID, Ailles L, Narindrasorasak S, Tan R, Kisilevsky R (1992) Localization of the basement membrane heparan sulfate proteoglycan in islet amyloid deposits in type II diabetes mellitus. Arch Pathol Lab Med 116:951–954
Zhang P, Fu X, Sawashita J et al (2010) Mouse model to study human A beta 2M amyloidosis: generation of a transgenic mouse with excessive expression of human beta2-microglobulin. Amyloid 17:50–62
Zijlstra M, Bix M, Simister NE, Loring JM, Raulet DH, Jaenisch R (1990) Beta 2-microglobulin deficient mice lack CD4-8 + cytolytic T cells. Nature 443:742–746
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2012 Springer Science+Business Media Dordrecht
About this chapter
Cite this chapter
Corlin, D., Heegaard, N. (2012). β2-Microglobulin Amyloidosis. In: Harris, J. (eds) Protein Aggregation and Fibrillogenesis in Cerebral and Systemic Amyloid Disease. Subcellular Biochemistry, vol 65. Springer, Dordrecht. https://doi.org/10.1007/978-94-007-5416-4_19
Download citation
DOI: https://doi.org/10.1007/978-94-007-5416-4_19
Published:
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-007-5415-7
Online ISBN: 978-94-007-5416-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)