Abstract
White coat hypertension (WCHT) and masked hypertension (MH) are phenomena that were identified and detected once blood pressure (BP) measurements became possible outside an office setting. The threshold values generally used to diagnose WCHT are >140/90 mmHg in the office setting and <135/85 mmHg in an ambulatory or home setting. Subjects with MH have a normal office BP of <140/90 mmHg and an abnormal out-of-office BP ≥135/85 mmHg. The prevalence of WCHT and MH varies according to the definition of WCHT and MH and the population studied, but it is generally around 30 % for mild, sustained hypertensive (SH) subjects. Several measures of target organ damage have been compared for normotensive, WCHT, MH, and sustained hypertension subjects; these include left ventricular mass, microalbuminuria, and carotid intima-media atherosclerosis. In general, target organ damage in WCHT is less than that observed in MH or SH. Authors of prospective studies have concluded that WCHT subjects have a lower risk of morbidity than MH subjects. Recent meta-analyses confirm that the incidence cardiovascular (CV) events is more important in patients with MH, and that subjects with MH have the same CV risks that subjects with SH. When WCHT subjects are prescribed antihypertensive medications, there is usually a decrease in clinical BP, but little or no change in ABP. Thus, drug treatment is not necessarily indicated. For MH subjects, in the absence of a randomized trial, treatment recommendations might be premature but the existence of MH in a patient with CV risk or signs of CV damage must be an incentive to promote lifestyle changes and even to start pharmacological treatment. In any case, these patients should be followed closely over time.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
The phenomena of white coat hypertension (WCHT) and masked hypertension (MH) were first identified once blood pressure (BP) measurements became possible outside of a clinic/office setting. Ambulatory blood pressure (ABP) or home blood pressure (HBP) measurements have really changed the way we interpret the significance of BP data. Thus, four BP categories can be measured: MH, true normotension (NT), sustained hypertension (SH) and WCHT ([1]; see Table 2.1).
Generally, clinic/office BP is defined as normal when <140/90 mmHg and out-of-office BP, ABP, or HBP are defined as normal when <135/85 mmHg (daytime or home BP) [1]. The question then is whether these entities are clinically unimportant or whether they are associated with an increased cardiovascular (CV) risk.
2 White Coat Hypertension
2.1 Terminology
WCHT is the most commonly used term for describing patients with elevated BP in a clinic or office setting but not in other settings [2, 3].The term isolated office hypertension is also used.
2.2 Definition
The criterion originally used by Pickering and colleagues [2] to define WCHT was a clinic or office BP that remained above 140/90 mmHg, together with a daytime ABP or HBP below 134/90 mmHg. The threshold values generally used are >140/90 mmHg within an office setting and <135/85 mmHg outside the office [1]. Most investigators have used both systolic and diastolic BP to make a diagnosis of WCHT. WCHT should be distinguished from the white-coat effect, which is the difference between clinic BP and daytime ABP.
2.3 Prevalence
The prevalence of WCHT depends on the definition and on the demographic features of the population being surveyed.
Authors of early studies suggested that WCHT occurs in 20 % or more of the hypertensive population [2, 4–6]. Not surprisingly, it is more common (33.3 %) among patients with the mildest hypertension (from 140/90 to 159/99 mmHg) and virtually nonexistent among those with the most severe hypertension (above 210/120 mmHg) [7–10]. The prevalence of WCHT does not appear to increase with age [11], nor does it differ for sex or weight from SH.
2.4 Metabolic and Biochemical Features
Weber and colleagues [12] reported that patients with WCHT had higher levels of total and low-density lipoprotein (LDL) cholesterol than normotensive patients. Levels of high-density lipoprotein (HDL) cholesterol and triglycerides did not differ between the WCHT and normotensive groups. Even though several studies have been performed in this area, the authors did not confirm their initial findings.
Cavallini and colleagues [13] and Pierdomenico and colleagues [14] reported that all three groups (NT, SH, and WCHT) had the same levels of total cholesterol, HDL cholesterol, and triglycerides. Pierdomenico and colleagues [14] also found no difference in the level of LDL cholesterol among groups. In a third study, Marchesi and colleagues [15] reported that, of 84 newly diagnosed hyper-tensive patients, 24 % who were classified as having WCHT had glucose and lipid blood levels that were similar to those in SH patients, though the WCHT patients had lower insulin levels. Pierdomenico and colleagues [16] observed that the LDL of WCHT patients does not exhibit greater than normal propensity toward oxidation.
In the Pressioni Arteriose Monitorate E Loro Associazioni (PAMELA) study, Mancia and colleagues [17] reported that a prevalence of diabetes (≥126 mg/dL or use of antidiabetic drugs), impaired fasting blood glucose (≥110 to <126 mg/dL), and hypercholesterolemia (total serum cholesterol ≥240 mg/dL or 200 mg/dL) increased progressively from optimal to normal, high-normal, and then elevated office systolic or diastolic BP. Fasting blood glucose and total serum cholesterol also increased progressively from the first to the fourth group, with HDL cholesterol values showing a concomitant progressive decrease. Even if the majority of the results show that there is no specific metabolic abnormality associated with WCHT, a unanimous consensus has not been reached.
2.5 Target Organ Damage
Verdecchia and colleagues [4] reported that there was no correlation between the magnitude of the white-coat effect and left ventricular mass (LVM). A similar finding was reported by Guida and colleagues [18].
Kuwajima and colleagues [19] performed an echocardiographic study of 67 patients aged 60 years and older: 17 patients with WCHT, 34 patients with ST, and 16 NT control subjects. The patients with WCHT had a moderately increased left atrial mass and LVM in association with a tendency for disturbed diastolic function. These findings suggest that WCHT in older patients may not be unharmful. Cavallini and colleagues [13] found that intima-medial thicknesses (IMT) of the carotid artery in the NT group (0.76 mm) and WCHT group (0.84 mm) were similar, whereas the IMT in the SH group was greater (0.98 mm). Pierdomenico and colleagues [14] compared 50 SHs with 25 sex-matched and age-matched WCHTs and 25 NTs, using a comprehensive battery of tests for target organ damage, including both echocardiography and carotid artery ultrasonography. WCHT subjects did not differ from NT subjects in any of these measures. Cuspidi and colleagues [20], whose study involved 82 patients, reported that the prevalence of left ventricular hypertrophy and cardiac remodeling was significantly more frequent in SH patients (51 %) than in WCHT patients.
Soma and colleagues [21] investigated left ventricular function in 26 subjects with WCHT. They discovered that the arterial pressure response in subjects with WCHT was associated with increased left ventricular external work, increased end-systolic wall stress and alterations of left ventricular filling, though they recorded normal ejection fraction and mean velocity of circumferential fiber shortening. Ormezzano and colleagues [22] did not find cardiovascular alterations in WCHT subjects with reference to the left ventricular mass index (LVMI), carotid IMT, and aortic stiffness (pulse wave velocity). The results obtained by Sega and colleagues [6] from the PAMELA study did not match with the previous data reported by Ormezzano. In these subjects, LVMI was greater than in subjects with normotension both inside and outside the office setting.
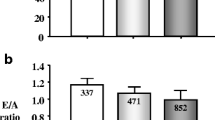
Another sign of target organ damage is microalbuminuria. Hoegholm and colleagues [23] evaluated microalbuminuria in 411 subjects who were NT subjects, SH subjects, or WCHT subjects. The ratios for the NT and WCHT subjects were the same, while those for the SH subjects were greater [9]. Pierdomenico and colleagues [14] and Martinez and colleagues [8] also found lower levels of microalbuminuria in WCHT subjects than they did for SH subjects.
2.6 Morbidity and Mortality
Verdecchia and colleagues [24] were the first to propose the hypothesis that WCHT is associated with a relatively low risk of morbidity, with an intermediate level of morbidity for NT and SH subjects. Following up a group of 1,187 NT and HT individuals over 3 years, they reported an event rate of 0.49 per 100 patient years for WCHT subjects (similar to the rate of 0.47 for NT subjects), and rates of 1.79 among hypertensive dippers, who constituted the majority, and 4.99 among non-dippers.
In the Self-Measurement of Blood Pressure at Home in the Elderly: Assessment and Follow-up (SHEAF) study, Bobrie and colleagues [25] reported that even when the characteristics of patients with isolated office hypertension were similar to those of patients with controlled hypertension, patients with isolated office hypertension had fewer previous cardiovascular complications.
In a subsequent report, Bobrie and colleagues [26] performed a multivariable analysis and found that the hazard ratio of cardiovascular events was almost the same in patients with elevated office BP and normal HBP than in patients having normal home and office BP.
After a 10-year follow-up study of 420 patients with grade 1–2 hypertension newly diagnosed by their general practitioner and 146 NT controls for which ABP monitoring was performed at baseline, Gustavsen and colleagues [27] concluded that there was an increased CV risk in WCHT patients compared to NT controls. Ohkubo and colleagues [28] followed 1,332 subjects by means of ABP for a mean duration of 10 years and showed that the CV risk in WCHT subjects does not differ significantly from that of NT subjects.
In the PAMELA study, Mancia and colleagues [29] did not confirm their previously obtained results and, like Gustavsen and colleagues [27], found that WCHT and MH, when identified by office and ABP or by office and HBP, are not prognostically innocent in term of CV risks. Fagard and Cornelissen [30], by means of a meta-analysis, concluded that the outcome of CV events in WCHT does not significantly differ from NT.
A more recent meta-analysis study carried out by Pierdomenico and Cuccurullo [31] confirmed that CV risk is not significantly different between WCHT and NT, regardless of normotensive population type and follow-up length. However, it is noteworthy that during follow-up, drug therapy was more frequent in WCHT than in NT subjects and thus its possible impact on the outcome should be evaluated in future studies. Consequently, when WCHT is identified at an office setting and presents a normal ambulatory blood pressure monitoring (ABPM) and a normal HBP is not perhaps completely innocent.
2.7 Management and Treatment
The most controversial issue about the management of WCHT is whether treatment with antihypertensive drugs should be prescribed. Authors of several studies have analyzed the effects of anti-hypertensive medications on patients with SH and WCHT. Pickering and colleagues [32] reported that administration of doxazosin mesylate, a long-acting α-blocker, lowered clinic BP for subjects in both groups and to the same extent, but lowered ABP only in those with SH.
Two other groups, both using calcium antagonists, found that the effects of medication on clinic BP in patients with high and normal ABP were similar. However, when ABP was normal to begin with (i.e., in the case of WCHT), the drug did not lower it further [33, 34]. Herpin and colleagues [35] compared patients treated with a variety of calcium antagonists with treatment with angiotensin-converting enzyme inhibitors, and confirmed that these two drug classes had similar effects on clinic BP. However, calcium antagonists had little effect on ABP if this was low to begin with (i.e., WCHT), whereas angiotensin-converting enzyme inhibitors lowered it irrespective of whether ABP had started out low or high. Kristensen and colleagues [36] found that treatment with benazepril lowered ABP in WCHT patients more than did treatment with felodipine. This finding raises the interesting possibility that there might be differences among the various classes of antihypertensives in terms of the degree to which they affect WCHT.
The other major issue regarding WCHT is patient follow-up. Authors of at least three studies have investigated this by repeating ABPM. Bidlingmeyer and colleagues [37] re-examined 81 patients with WCHT after an average period of 5 years and found that ABP in 60 of them had increased to more than 140/90 mmHg, although no similar change in clinic BP had been recorded. Verdecchia and colleagues [38] followed 83 patients for 2.5 years and found that for two patients, the classification had changed from WCHT to SH. Polonia and colleagues [39] followed 36 patients with WCHT and 52 NT patients for 3.5 years and found that 11 and 6 %, respectively, developed ambulatory hypertension.
In a third study performed by White and colleagues [40], only 12.5 % of WCHT patients became truly hypertensive. The apparent transition of a patient from WCHT to SH could have several explanations. The most widely advocated hypothesis is that WCHT is a prehypertensive state, though an equally plausible hypothesis is that the transition is nothing more than a regression to the mean. So, it is reasonable to suggest a treatment plan based on lifestyle recommendations in patients with WCHT in the absence of comorbid conditions and target organ damage and monitor them on a regular basis. It is clear that all patients diagnosed with WCHT should be followed up indefinitely, either with both clinic and HBP or with ABPM.
3 Masked Hypertension
3.1 Definition
Subjects with MH have a normal clinic BP of <140/90 mmHg and an abnormal, out-of-office (ambulatory or at home) daytime systolic BP >135 mmHg or daytime diastolic blood pressure >85 mmHg [1]. MH has also been known as white coat normotension, inverse white coat hypertension, isolated clinic normotension, isolated home hypertension, isolated ambulatory hypertension and reverse white-coat effect.
3.2 Prevalence
The use of different BP limits affects the prevalence of MH and MH is certainly often underestimated. Prevalence can be different according to different studies, the range being between 10 and 40 % [5, 6, 22, 26, 28, 29, 41, 42]. According to Eguchi and colleagues [43], prevalence is higher in diabetic patients (47 %), whereas Baguet and colleagues [44] have reported a 30 % prevalence in sleep apnea syndrome.
3.3 Target Organ Damage
The first study that looked at the issue of target organ damage in MH was carried out by Liu and colleagues [45], who examined 295 clinically normotensive individuals and 64 patients with SH. They found that individuals with MH had greater LVM and higher prevalence of carotid IMT atherosclerosis than those with normal BP.
Sega and colleagues [6], with reference to the PAMELA study, observed that MH presented higher LVMI and structural cardiac alterations. Bjorklund and colleagues [46] investigated 578 untreated 70-year-old hypertensive men and reported a higher LVM and relative wall thickness for MH patients.
In a population of 688 subjects, Silva and colleagues [47] observed an increased LVM with MH. Ormezzanno and colleagues [22] observed that cardiovascular alterations, such as LVMI, carotid IMT, aortic stiffness, and pulse wave velocity are found in association with MH. These data were confirmed by Lurbe and colleagues [48] in younger patients. Consequently, we can suggest that subjects with MH have more extensive target organ damage than true normotensive individuals.
3.4 Morbidity and Mortality
The clinical significance of MH is based on five longitudinal studies and two meta-analyses. First, individuals with MH are at increased risk of developing SH. In the Hypertension and Ambulatory Recording Venetia Study (HARVEST) [49], 35 % of participants with MH had persistent SH when followed up over 6 years whereas only 19 % of those with true NT at baseline developed SH. This has been confirmed by Mancia and colleagues [29] in the PAMELA study where subjects with WCHT and MH were at increased risk of developing SH. However, Messerli and Makani [50] concluded that for undetermined reasons, some patients are able to remain in the same BP category or are even able to go back to a category that confers reduced morbidity and mortality.
The prognostic value of MH was assessed by Bjorklund and colleagues [46] in a cohort of 70-year-old men followed for 8.4 years. They found that MH was as great a predictor of CV events (relative risk: 2.77) as SH (relative risk: 2.94).
In a cohort of treated hypertensive patients followed up for a mean of 3.2 years, Bobrie and colleagues [26] found that individuals with controlled office BP but uncontrolled HBP were doubly at risk of developing CV events in comparison with patients having their BP controlled both at home and in the office.
The composite risk of cardiovascular mortality and stroke morbidity was calculated for 1,332 subjects from the town of Ohasama, Japan, who were followed up for a period of 10 years [28]. The risk was significantly greater for subjects with MH or SH than for those with NT or WCHT.
Lurbe and colleagues [48] investigated the prevalence, persistence and clinical significance of MH in 592 children and adolescents. They concluded that in children and adolescents, MH is a precursor of SH and LVM.
Finally, in the PAMELA study, Mancia and colleagues [29] discovered that WCHT and MH, when identified by office and ambulatory measurement or by office and HBP, are not prognostically innocent.
Hara and colleagues [51] demonstrated in the Ohasama study that CV risk is higher with MH and similar to that for SH. Taking into account 11 studies and 11,502 participants, Fagard and colleagues [30] established, by means of meta-analysis, that the incidence of CV events is more important in patients with MH. More recently, in a meta-analysis of six cohort studies reporting quantitative data for MH prognosis, Bobrie and colleagues [52] showed that subjects with MH have a higher risk of CV incidents [hazard ratios: 1.92 (1.51–2.44)] than normotensive subjects or subjects with controlled hypertension. Consequently, we suggest that MH and SH subjects have the same CV risk.
3.5 Identification of Patients with Masked Hypertension
For practical and financial reasons, ABPM and HBP monitoring cannot be performed in all individuals with normal office BP. The decision to carry out such measurements should be taken according to specific criteria. In the French SHEAF study involving almost 5,000 treated hypertensive patients (mean age 70 years, 49 % men), Mallion and collegues [53] found that three variables at inclusion identified a MH profile: age >60 years, office systolic BP >130 mmHg, male sex. Rasmussen and colleagues [54] showed that a higher daytime than clinic BP was a much more frequent event among men aged 42 years (82 %) than among men aged 72 years (51 %). Thus, MH is expected to be more frequent in men. Recent results from the PAMELA study, however, do not support this hypothesis [55]. In this study, 40.1 % of women and 39 % of men showed a reversed white-coat condition.
Lifestyle factors, and smoking in particular, have been shown to largely influence the relationship between clinic and ABP [56]. Drinking alcohol [57], contraceptive use in women [58], and being sedentary [59] are other lifestyle factors that have been shown to selectively raise ABP. According to some investigators [60], obesity is another determinant of higher daytime BP, but this association has not been confirmed by others [49].
Moreover, MH should be diagnosed in patients who are at increased risk of CV complications, including patients with coronary, cerebrovascular, or kidney disease, patients with diabetes and other individuals with a high CV risk profile.
3.6 Management and Treatment
In the absence of randomized trials, treatment recommendations might be premature. However, the existence of MH in a patient with CV risk or sign of CV damage must be an incentive to promote lifestyle recommendations and changes and even to start treatment. In any case, these patients should be closely monitored..
4 Conclusions
From a practical standpoint, it is much easier to suspect the diagnosis of WCHT, because patients will usually state that their BP is normal at home. In contrast, MH needs to be looked for, and there are very few clinical hints as to its presence.
A normal BP in the clinical setting does not mean that a patient is not at risk from an elevated BP, which can occur at other times of the day. This is particularly true in patients who are treated with antihypertensive drugs that do not include a full 24-h period. Because most patients take their medication in the morning, BP values in the physician’s office are often normal, though they may be substantially elevated at the end of the dosing interval (i.e., during the night and early morning hours). Thus, in many hypertensive patients, clinic BP may be seemingly well controlled, but early morning BP, before the patients takes the medication, may be elevated thereby increasing the risk of CV events. For both patient and physician, MH may become a blind spot in the antihypertensive regimen. Pickering and colleagues [60] have suggested that, for the detection of MH, home BP monitoring is likely to prove more cost-effective than ABPM.
As to the therapeutic approach, we should remember that WCHT has a benign prognosis and may often be overtreated; therefore, a conservative approach is probably justified. Conversely, MH has a more serious prognosis and is often undertreated; it deserves, therefore, a thorough evaluation and a more aggressive therapeutic approach.
References
Mancia G, De Backer G, Dominiczak A et al (2007) 2007 guidelines for the management of arterial hypertension: the task force for the management of arterial hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). J Hypertens 25:1105–1187
Pickering TG, James GD, Boddie C et al (1988) How common is white coat hypertension? JAMA 259:225–228
Mancia G, Zanchetti A (1996) White-coat hypertension: misnomers, misconceptions and misunderstandings. What should we do next? (editorial). J Hypertens 14:1049–1052
Verdecchia P, Schillaci G, Borgioni C et al (1995) White coat hypertension and white coat effect similarities and differences. Am J Hypertens 8:790–798
Pierdomenico SD, Mezzetti A, Lapenna D et al (1995) ‘White-coat’ hypertension in patients with newly diagnosed hypertension: evaluation of prevalence by ambulatory monitoring and impact on cost of health care. Eur Heart J 16:692–697
Sega R, Trocino G, Lanzarotti A et al (2001) Alterations of cardiac structure in patients with isolated office, ambulatory, or home hypertension: data from the general population (Pressione Arteriose Monitorate E Loro Associazioni (PAMELA) Study). Circulation 104:1385–1392
Hoegholm A, Kristensen KS, Madsen NH, Svendsen TL (1992) White coat hypertension diagnosed by 24-h ambulatory monitoring. Examination of 159 newly diagnosed hypertensive patients. Am J Hypertens 5:64–70
Martinez MA, Garcia-Puig J, Martin JC et al (1999) Frequency and déterminants of white coat hypertension in mild to moderate hypertension: a primary care-based study. Am J Hypertens 12:251–259
Hoegholm A, Kristensen KS, Bang LE, Nielsen JW (1998) White-coat hypertension and target organ involvement: the impact of différent cut-off levels on albuminuria and left ventricular mass and geometry. J Hum Hypertens 12:433–439
Staessen JA, O’Brien ET, Amery AK, Atkins N et al (1994) Ambulatory blood pressure in normotensive and hypertensive subjects: results from an international database. J Hypertens suppl:S1–S12
Trenkwalder P, Plaschke M, Steffes-Tremer I, Lydtin H (1993) ‘White coat’ hypertension and alerting reaction in elderly and very elderly hypertensive patients. Blood Press 2:262–271
Weber MA, Neutel JM, Smith DH, Graettinger WF (1994) Diagnosis of mild hypertension by ambulatory blood pressure monitoring. Circulation 90:2291–2298
Cavallini MC, Roman MJ, Pickering TG et al (1995) Is white coat hypertension associated with arterial disease or left ventricular hypertrophy? Hypertension 26:413–419
Pierdomenico SD, Lapenna D, Guglielmi MD et al (1995) Target organ status and serum lipids in patients with white coat hypertension. Hypertension 26:801–807
Marchesi E, Perani G, Falaschi F et al (1994) Metabolic risk factors in white coat hypertensives. J Hum Hypertens 8:475–479
Pierdomenico SD, Costantini F, Bucci A et al (1998) Low-density lipoprotein oxidation and vitamins E and C in sustained and white-coat hypertension. Hypertension 31:621–626
Mancia G, Facchetti R, Bombelli M et al (2005) Relationship of office, home, and ambulatory blood pressure to blood glucose and lipid variables in the PAMELA population. Hypertension 45:1072–1077
Guida L, Lannuzzi R, Crivaro M et al (1999) Clinic-daytime blood pressure différences and cardiovascular damage. J Hypertens 17:331–337
Kuwajima I, Suzuki Y, Fujisawa A, Kuramoto K (1993) Is white coat hypertension innocent? structure and function of the heart in the elderly. Hypertension 22:826–831
Cuspidi C, Marabini M, Lonati L et al (1995) Cardiac and carotid structure in patients with established hypertension and white-coat hypertension. J Hypertens 13:1707–1711
Soma J, Wideroe TE, Dahl K et al (1996) Left ventricular systolic and diastolic function assessed with two-dimensional and doppler echocardiography in “white coat” hypertension. J Am Coll Cardiol 28:190–196
Ormezzano O, Baguet JP, François P et al (2004) Is there any real target organ damage associated with white-coat normotension? Clin Auton Res 14:160–166
Hoegholm A, Bang LE, Kristensen KS et al (1994) Microalbuminuria in 411 untreated individuals with established hypertension, white coat hypertension, and normotension. Hypertension 24:101–105
Verdecchia P, Porcellati C, Schillaci G et al (1994) Ambulatory blood pressure. an independent predictor of prognosis in essential hypertension. Hypertension 4:793–801 (published erratum Hypertension 1995; 25:462)
Bobrie G, Genès N, Vaur L et al (2001) Is “isolated home” hypertension as opposed to “isolated office” hypertension a sign of greater cardiovascular risk? Arch Intern Med 161:2205–2211
Bobrie G, Chatellier G, Genes N et al (2004) Cardiovascular prognosis of “masked hypertension” detected by blood pressure self-measurement in elderly treated hypertensive patients. JAMA 291:1342–1349
Gustavsen PH, Høegholm A, Bang LE, Kristensen KS (2003) White coat hypertension is a cardiovascular risk factor: a 10-year follow-up study. J Hum Hypertens 17:811–817
Ohkubo T, Kikuya M, Metoki H et al (2005) Prognosis of “masked” hypertension and “white-coat” hypertension detected by 24-h ambulatory blood pressure monitoring 10-year follow-up from the Ohasama study. J Am Coll Cardiol 46:508–515
Mancia G, Facchetti R, Bombelli M et al (2006) Long-term risk of mortality associated with selective and combined elevation in office, home, and ambulatory blood pressure. Hypertension 47:846–853
Fagard RH, Cornelissen VA (2007) Incidence of cardiovascular events in white-coat, masked and sustained hypertension versus true normotension: a meta-analysis. J Hypertens 25:2193–2198
Pierdomenico SD, Cuccurullo F (2001) Prognostic value of white-coat and masked hypertension diagnosed by ambulatory monitoring in initially untreated subjects: an updated meta analysis. Am J Hypertens 24:52–58
Pickering TG, Levenstein M, Walmsley R (1994) Differential effects of doxazosin on clinic and ambulatory pressure according to age, gender, and présence of white coat hypertension. Results of the HALT study. Am J Hypertens 7:848–852
Weber MA, Cheung DG, Graettinger WF, Lipson JL (1988) Characterization of antihypertensive therapy by whole-day blood pressure monitoring. JAMA 259:3281–3285
Hoegholm A, Wiinberg N, Kristensen KS (1996) The effect of antihypertensive treatment with dihydropyridine calcium antagonists on white-coat hypertension. Blood Press Monit 1:375–380
Herpin D, Vaisse B, Pitiot M et al (1992) Comparison of angiotensin-converting enzyme inhibitors and calcium antagonists in the treatment of mild to moderate systemic hypertension, according to baseline ambulatory blood pressure level. Am J Cardiol 69:923–926
Kristensen KS, Wiinberg N, Hoegholm A et al (1998) Benazepril versus felodipine as supplément to bendroflumethiazide: évaluation by office and ambulatory blood pressure. Blood Press Monit 3:115–120
Bidlingmeyer I, Burnier M, Bidlingmeyer M et al (1996) Isolated office hypertension: a prehypertensive state? J Hypertens 14:327–332
Verdecchia P, Schillaci G, Borgioni C et al (1996) Identification of subjects with white-coat hypertension and persistently normal ambulatory blood pressure. Blood Press Monit 1:217–222
Polonia JJ, Santos AR, Gama GM et al (1997) Follow-up clinic and ambulatory blood pressure in untreated white-coat hypertensive patients (évaluation after 2–5 years). Blood Press Monit 2:289–295
White WB, Daragjati C, Mansoor GA, McCabe EJ (1996) The management and follow-up of patients with white-coat hypertension. Blood Press Monit 1(suppl 2):S33–S36
Selenta C, Hogan BE, Linden W (2000) How often do office blood pressure measurements fail to identify true hypertension? An exploration of white-coat normotension. Arch Fam Med 9:533–540
Wing LMH, Brown MA, Beilin U, Ryan P, Reid CM (2002) On behalf of the ANBP2 management committee and investigators ‘Reverse white coat hypertension’ in older hypertensives. J Hypertens 20:639–644
Eguchi K, Ishikawa J, Hoshide S et al (2007) Masked hypertension in diabetes mellitus: a potential risk. J Clin Hypertens 9:601–607
Baguet JP, Lévy P, Barone-Rochette G et al (2008) Masked hypertension in obstructive sleep apnea syndrome. J Hypertens 26:885–892
Liu JE, Roman MJ, Pini R et al (1999) Cardiac and arterial target organ damage in adults with elevated ambulatory and normal office blood pressure. Ann Intern Med 131:564–572
Bjorklund K, Lind L, Zethelius B et al (2003) Isolated ambulatory hypertension predicts cardiovascular morbidity in elderly men. Circulation 107:1297–1302
Silva JA, Barbosa L, Bertoquini S et al (2004) Relationship between aortic stiffness and cardiovascular risk factors in a population of normotensives, white-coat normotensives, white-coat hypertensives, sustained hypertensives and diabetic patients. Rev Port Cardiol 23:1533–1547
Lurbe E, Torro I, Alvarez V et al (2005) Prevalence, persistence, and clinical significance of masked hypertension in youth. Hypertension 45:493–498
Palatini P, Winnicki M, Santonastaso M et al (2004) Prevalence and clinical significance of isolated ambulatory hypertension in young subjects screened for stage 1 hypertension. Hypertension 44:170–174
Messerli FH, Makani H (2009) Relentless progression toward sustained hypertension. Hypertension 54:217–218
Hara A, Ohkubo T, Kondo T et al (2009) Detection of silent cerebrovascular lesions in individuals with ‘masked’ and ‘white-coat’ hypertension by home blood pressure measurement: the Ohasama study. J Hypertens 27:1049–1055
Bobrie G, Clerson P, Ménard J et al (2008) Masked hypertension: a systematic review. J Hypertens 26:1715–1725
Mallion JM, Clerson P, Bobrie G et al (2006) Predictive factors for masked hypertension within a population of controlled hypertensives. J Hypertens 24:2365–2370
Rasmussen SL, Torp-Pedersen C, Borch-Johnsen K, Ibsen H (1998) Normal values for ambulatory blood pressure and differences between casual blood pressure and ambulatory blood pressure: results from a Danish population survey. J Hypertens 16:1415–1424
Bombelli M, Sega R, Facchetti R et al (2005) Prevalence and clinical significance of a greater ambulatory versus office blood pressure (reverse white coat condition) in a general population. J Hypertens 23:513–520
Narkiewicz K, Maraglino G, Biasion T et al (1995) Interactive effect of cigarettes and coffee on daytime systolic blood pressure in patients with mild essential hypertension. HARVEST Study Group (Italy). Hypertension ambulatory recording VEnetia STudy. J Hypertens 13:965–970
Vriz O, Piccolo D, Cozzutti E et al (1998) The effects of alcohol consumption on ambulatory blood pressure and target organs in subjects with borderline to mild hypertension HARVEST study group. Am J Hypertens 11:230–234
Narkiewicz K, Graniero G, D’esté D et al (1994) Ambulatory blood pressure in mild hypertensive women taking oral contraceptives. A case control study. Am J Hypertens 8:249–253
Palatini P, Graniero G, Mormino P et al (1994) Relation between physical training and ambulatory blood pressure in stage I hypertensive subjects results of the HARVEST trial. Circulation 90:2870–2876
Pickering T, Davidson K, Gerin W, Schwartz GE (2002) Masked hypertension. Hypertension 40:795–796
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2012 Springer-Verlag Italia
About this chapter
Cite this chapter
Mallion, JM. (2012). Clinical Significance and Treatment Requirements in White Coat and Masked Hypertension. In: Berbari, A., Mancia, G. (eds) Special Issues in Hypertension. Springer, Milano. https://doi.org/10.1007/978-88-470-2601-8_2
Download citation
DOI: https://doi.org/10.1007/978-88-470-2601-8_2
Published:
Publisher Name: Springer, Milano
Print ISBN: 978-88-470-2600-1
Online ISBN: 978-88-470-2601-8
eBook Packages: MedicineMedicine (R0)