Abstract
Circadian rhythms pervade all aspects of mammalian physiology and behaviour, providing a near 24 h temporal architecture for all major brain and body systems. Mammalian chronobiology research has focused on nocturnal laboratory rodent models, and this has yielded valuable insight into the neural basis of circadian timekeeping. This has identified the suprachiasmatic nuclei (SCN) as the dominant circadian pacemaker and characterised its neurophysiology, neurochemistry, and potential mechanisms of behavioural control. However, nocturnality is only one of four possible temporal niches, and understanding of the neural circadian system in diurnal, crepuscular, and cathemeral mammalian species is very much in its infancy. In this chapter, we review the fundamental properties of the neural circadian system in nocturnal rodents and then compare this with what is known about neural timekeeping in diurnal species. Through this, we identify gaps in our knowledge and key problems to investigate in order to gain a more complete understanding of circadian control of behaviour and physiology, particularly with regard to temporal niche preference.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction to the Suprachiasmatic Nucleus as the Central Brain Clock
Since the early twentieth century, behavioural research has shown that many wild-caught as well as laboratory-bred rodent species have a propensity for voluntarily exercising in running wheels with the onset of this activity tightly linked with the time of lights off. Further, when assessed in the absence of a light-dark cycle, rhythms in locomotor activity persisted, with a period that deviated from the 24 h pattern seen under light-dark conditions. Based on this and a multitude of other findings, researchers in the 1970s surmised that the daily brain clock controlling these rhythms should receive visual information. Through mapping visual pathways and making discrete lesions in brain nuclei receiving retinal innervation, it was discovered in 1972 that the suprachiasmatic nuclei (SCN) of the hypothalamus contained the circadian pacemaker controlling daily rhythms in the physiology and behaviour of nocturnal rats [1, 2]. Subsequent demonstration that behavioural rhythms are rescued in SCN-lesioned adult rodents by implants of fetal SCN tissue cemented the SCN as the master pacemaker in mammals and catalysed modern neuroscience investigation of the biological timekeeping mechanisms.
Neuroanatomists in the late nineteenth and early twentieth century had identified the SCN as distinct structures in the mammalian hypothalamus [3], but their function was unknown until the 1970s. The SCN are readily identifiable in histological preparations, in part, because the density of cell packing in this structure is relatively high compared to adjacent anterior hypothalamic structures. Consequently, the SCN are now well-characterised, particularly in nocturnal rodent species such as rat (Rattus norvegicus), mouse (Mus musculus), Syrian hamster (Mesocricetus auratus) and Siberian hamster (Phodopus sungorus). The SCN are bilobed, situated on either side of the ventral floor of the third ventricle in the periventricular zone of the anterior hypothalamus. In the adult laboratory rat, they are ~0.7 to 1 mm in length along its rostrocaudal axis, ~0.5–0.6 mm on the dorsoventral plane and ~0.3–0.5 mm wide on the mediolateral plane. The SCN vary in morphology along their rostrocaudal axis (described as ‘rugby ball shaped’ in the rat) as well as between species, and consequently, these parameters are general estimates.
Cytologically, the SCN contain both neurons and astroglia with an estimated ratio of 7–8:1 in the rat SCN (reviewed in [4]). In rodents, SCN neurons are among the smallest of the brain, with cell body diameter of ~8–10 μm. The size of these cells in the medial SCN is smaller (~8 μm in diameter) than neurons in more lateral areas (~10 μm). Similarly, the density of cell packing is heterogeneous in the rat SCN with more cells per volume in the dorsomedial compared to lateral areas. It has been speculated that this dense cell packing and reduced extracellular space between neurons in the dorsomedial SCN may enable ephaptic communication between these cells, an idea that has yet to be experimentally scrutinised.
Physiologically, four key features define circadian timekeeping in the nocturnal rodent SCN: (1) The SCN exhibits daily changes in the uptake of 2-deoxyglucose, a marker of metabolic activity [5]. Since this is higher during the day than at night, this indicates that the SCN is metabolically most active during the animal’s behavioural quiescent phase of the circadian cycle. (2) Consistent with the metabolic profile, in vivo and in vitro electrophysiological recordings show that SCN neurons of nocturnal rodents are spontaneously active and intrinsically generate ~24 h rhythms in the frequency of action potential (AP) discharge [6, 7]. This AP firing rate rhythm peaks during the behaviourally quiescent day, with the nadir during the active phase at night. (3) The 24 h variation in electrical activity does not depend on ‘network’ properties as dissociated SCN neurons isolated in culture also vary daily discharge of AP firing [8]. (4) SCN neuronal clocks are predisposed to synchronise their activity with another, and intercellular communication is necessary for this process. Thus, the SCN are composed of several thousand cell autonomous oscillators, and the ability of SCN neurons to maintain synchronous activity in brain slices has enabled the development of ‘clock-in-a-dish’ preparations. With this in vitro model, the resetting actions of drugs on the firing rate rhythm can be readily assessed (see [9] for review).
Behaviourally, examination of nocturnal rodent wheel-running rhythms has allowed the description of several key properties of circadian rhythms. First, wheel running is synchronised or entrained to lights off such that the initiation of vigorous wheel running is coincident with lights off. Second, these rhythms are sustained in constant darkness, and their timing can be adjusted by exposure to short pulses of light. In nocturnal rodents, the onset of the active phase is defined as circadian time (CT12), and light pulses given in the early night (CT13-17) causes phase delays in the onset of the wheel-running rhythm, while the same stimulus given during the latter part of the active phase (CT18-22) causes phase advances. When given during the middle of the circadian day (CT4-8), light pulses are without phase-resetting effects. This phase response curve (PRC) to light is characteristic of many nocturnal rodent species (for review, see [10]).
Different types of stimuli that elicit increases in internal arousal, such as changing the cage or providing a novel running wheel, have very different phase-resetting actions [11]. During the subjective night, such stimuli have little phase-shifting effect, but, when given during the middle of the subjective day, they evoke phase advances in behavioural rhythms. As this PRC tends to be in anti-phase to that of light, such stimuli are called non-photic cues [12].
2 Connections of the SCN
2.1 Inputs
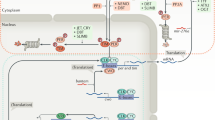
Neuroanatomically, antero- and retrograde tracer compounds as well as viral tools have revealed SCN efferents and afferents. Three main input pathways have been extensively characterised (Fig. 12.1):
Mammalian circadian system: main inputs and outputs to the master circadian clock, the suprachiasmatic nucleus (SCN). Photic cues are conveyed to the SCN through the retinohypothalamic tract (RHT) with glutamate (Glu) and pituitary adenylate cyclase-activating polypeptide (PACAP) as main neurotransmitters. The SCN also receives photic information indirectly from the intergeniculate leaflet (IGL) via the geniculohypothalamic tract (GHT). The neurotransmitters used in this pathway are γ-aminobutyric acid (GABA) and neuropeptide Y (NPY). GABAergic and peptidergic projections from the IGL and serotonergic (5-HT) projections from the median raphe (MR) convey non-photic cues to the SCN. Master circadian clock is organised in two main subdivisions: the dorsomedial area or ‘shell’ characterised by the presence of neurons expressing mainly arginine vasopressin (AVP) and the ventrolateral part or ‘core’ where most neurons contain vasoactive intestinal polypeptide (VIP) and gastrin-releasing peptide (GRP). Virtually all neurons across the SCN express the neurotransmitter GABA. Timekeeping signals from the SCN are transmitted to the rest of the brain and body by neural outputs as well as by diffusible humoral signals
-
1.
The retinohypothalamic tract (RHT) is a monosynaptic pathway from melanopsin-containing retinal ganglion cells to the SCN. In some rodent species, such as the rat, the ventral aspect of the SCN is more densely innervated than the dorsal [3], whereas in the others, such as the mouse, retinal innervation is dense in both dorsal and ventral aspects [13]. The main neurotransmitter of the RHT is glutamate, and it primarily acts on ionotropic glutamate receptors expressed by SCN neurons. Glutamate depolarises and increases action potential firing of SCN neurons [14]. In addition, the neuropeptide, pituitary adenylatecyclase-activating polypeptide (PACAP), is co-localised with glutamate in RHT terminals in the SCN where it acts to modulate the actions of glutamate [15]. This is the direct route via which environmental light information is conveyed to the SCN.
-
2.
The geniculohypothalamic tract (GHT) mostly innervates the ventral and central aspects of the rodent SCN and originates from neurons in the intergeniculate leaflet (IGL) of the visual thalamus [16]. The IGL is also retinally innervated and thus represents an indirect route through which environmental light cues are communicated to the SCN. The characteristic neurochemical of the GHT is neuropeptide Y (NPY). This neuropeptide predominantly inhibits SCN neurons (see [9]), while functionally, activation of the GHT, or microinjection of NPY into the SCN [17], resets rodent behavioural rhythms at different times of the circadian cycle to light (non-photic PRC, phase advancing the SCN and circadian rhythms in wheel running during the subjective day, with much smaller effects during the subjective night). Stimuli that elevate internal arousal (such as cleaning a rodent’s cage) activate the IGL, and the GHT signals this information to the SCN. In addition to NPY, GABA is also present in the GHT, reinforcing the inhibitory nature of this pathway.
-
3.
The median raphe (MR) innervates the ventral and central SCN aspects, and the neurotransmitter serotonin (5-hydroxytryptophan or 5-HT) is the characteristic neurochemical of this pathway [18]. This neurochemical mostly suppresses SCN neuronal activity, although there is a range of pre- and postsynaptic 5-HT receptors expressed in the SCN region, adding complexity to its actions. Similar to the GHT, input from the median raphe signals internal arousal state, although the magnitude of the resetting actions of 5-HT is less pronounced than those of the NPY. Nonetheless, current evidence indicates that both the GHT- and MR-SCN pathways are necessary for the resetting actions of non-photic stimuli to be fully realised [19].
In addition to these three pathways, there are also inputs arising from other hypothalamic structures. For example, the orexin neurons of the lateral hypothalamus provide a diffuse innervation to the SCN, with orexin-containing fibres in close proximity to clock gene-expressing neurons. Orexin alters SCN neuronal activity and acts to accentuate the resetting actions of NPY on the SCN, implicating this projection in non-photic entrainment of circadian rhythms [20].
In the SCN, the terminations of the RHT, GHT, and MR pathways overlap, particularly in the ventral aspects. Perhaps unsurprisingly, activation of non-photic pathways can limit the resetting effects of light pulses, while acute light exposure can reduce or eliminate shifts to non-photic stimuli [12]. Thus, SCN neurons actively integrate photic and non-photic cues to shape the phase of the molecular clock and the entrainment of the circadian system to the external world.
2.2 Major Projections
From mapping of rat SCN efferents, the idea emerged that the dorsal and ventral aspects of the SCN may differ in their outputs. A crescent-shaped dorsal area, labelled the ‘shell’ by Moore and colleagues [3], was found to project mainly to the hypothalamus including the dorsomedial hypothalamus (DMH), the preoptic area (POA), the medial subparaventricular zone (mSPVz) and the paraventricular hypothalamic nucleus (PVH). In addition, the ‘shell’ efferents also targeted the paraventricular nucleus in the thalamus (PVT) and the bed nucleus of the stria terminalis (BST). By contrast, the more ventral ‘core’ SCN projected mainly to peri-SCN as well as the lateral SPVz and basal forebrain including the lateral septal nuclei. The implication of these anatomical studies is that different compartments of the SCN share and communicate different types of information.
3 Molecular Basis of Timekeeping
Single SCN neurons function as cell autonomous clocks as they contain the molecular clockworks. Building on key findings on the molecular basis for circadian timekeeping in Drosophila melanogaster, from the mid to late 1990s, several labs identified and characterised the interactions of fundamental molecular components of the mammalian circadian clock. These include the period (Per1-2), cryptochrome (Cry1-2), Clock and Bmal1 genes and their associated proteins [21, 22]. There is now a generally accepted model of this molecular clockwork with PER1-2/CRY1-2 constituting the negative limb and CLOCK and BMAL1 the positive limb of the clock. This molecular clock is named the transcriptional-translational feedback loop (TTFL) and conceptually is similar to the molecular circadian clock of flies, yeast and plants. Additional components, such as Rev-Erbα [23], Chrono [24], dec1/dec2, and others, function to fine-tune the molecular clock. Further, post-translational modulation of the clock function is provided by casein kinase 1δ/ε enzymes which influence PER-CRY complex stability and translocation to the nucleus to influence circadian clock speed [25]. Similarly, F-box proteins that alter protein ubiquitination and degradation influence CRY protein stability and clock speed in the SCN and other circadian oscillators [26–28]. In addition to this TTFL clock, a second non-TTFL oscillator is present in red blood cells and may be present in neurons [29]. Intriguingly, current evidence indicates that the circadian variation in SCN neuronal activity acts to facilitate the internal molecular clockworks such that chronic interference with electrical activity damps molecular oscillations and desynchronises SCN neurons [9].
3.1 Compartments of the SCN
Visualisation of gene expression by in situ hybridisation indicates that not all regions of the SCN rhythmically express clock genes at the same phase or perhaps at all (for review, see [30]). In particular, a predominantly dorsal area that forms a crescent-like shape expresses detectable and robust rhythms in Per1/Per2 which peak in the late day, while the ventromedial area that is largely enveloped by the dorsal area expresses rhythms that are weak or difficult to discern. These areas map well onto the ‘shell’ and ‘core’ regions previously described in relation to the differential regional pattern of SCN efferents. Similarly, imaging of luciferase reporters of Per1 promoter activity or PER2 expression in living rodent SCN brain slices reveals ‘shell’ and ‘core’ regions that are strongly and weakly rhythmic, respectively. Through attempting to model these temporal patterns in bioluminescent signals, terms such as ‘waves’ and ‘tides’ have been used to capture the complex dynamic nature of spatio-temporal organisation of molecular activity in the SCN [30]. Subsequent studies have conceptualised that the dorsal SCN (roughly corresponding to the area designated the ‘shell’ on the basis of SCN extrinsic connections and intrinsic neurochemistry) as initiating the daily change in Per1/PER2 expression such that the peak in these signals in some occurs several hours after the peak in the ‘shell’. Thus, while the SCN as a whole functions as the mammalian brain’s master circadian clock, intra-SCN timekeeping is heterogeneous with some areas appearing to lead daily changes in molecular clock activity, while others follow.
4 Intrinsic Neurochemistry
Neurochemically, all SCN neurons contain GABA, but they can, to an extent, be distinguished by the neuropeptides that they synthesise. The prominent neuropeptides contained in SCN neurons include vasoactive intestinal polypeptide (VIP), gastrin-releasing peptide (GRP), and arginine vasopressin (AVP) [31]. VIP-expressing neurons appear anatomically partitioned from AVP-expressing neurons lending further credence to the idea of ‘core’ and ‘shell’ regions of the SCN (Fig. 12.1). As there are many studies of the organisation of the intrinsic neurochemistry of the SCN as well as the local neurophysiological actions of these substances, we direct the reader to recent comprehensive reviews on these topics.
4.1 VIP
VIP-containing neurons are present throughout the ventral aspect of the nocturnal rodent SCN. In rat, it is estimated that VIP neurons constitute around 24 % of SCN neurons and these cells delineate the ‘core’ SCN. VIP neurons have extensive axonal arborisations throughout the SCN, and their axons form part of the SCN output to innervate other hypothalamic sites such as the dorsomedial hypothalamus. The cognate receptor for VIP, VPAC2, is expressed throughout the dorsal and ventral aspects of mouse and rat SCN. Electron microscopy studies have indicated that some VIP neurons are directly retinorecipient and suggest that they form part of an interface with light input to the SCN. Consistent with this, the immediate early gene c-fos is induced by light in VIP neurons, while rat SCN firing rate rhythms are reset by VIP and VIP agonists in a pattern resembling the actions of light on behavioural rhythms (for review, see [32]). Further, microinjection of VIP into the SCN region evokes moderate phase shifts in wheel-running rhythms with a PRC resembling light [33].
The acute actions of VIP on SCN neurons are not well understood. Extracellular recordings from rat SCN neurons in vitro indicate that VIP evokes both activations and suppressions in the spontaneous firing rate of SCN neurons, with a larger portion of cells responding when tested during the night. The suppressive actions of VIP are mimicked by a VPAC2 receptor agonist. Voltage clamp recordings indicate that VIP elicits the release of GABA, and, therefore, the mixed acute effects of VIP likely represent a combination of pre-and postsynaptic actions [33].
As an intrinsic signal, VIP-VPAC2 communication plays a key role in synchronising SCN neurons; in transgenic mice lacking VIP or VPAC2 expression (Vip −/− and Vipr2 −/−, respectively), coordination among SCN cellular timekeepers is dysfunctional [34], and these mice express severely disrupted behavioural activity patterns, ranging from arrhythmic to rhythmic with an abnormally short tau (~22 h) [33, 35, 36]. Some of the alterations in cellular rhythmicity and synchrony arising from VIP-VPAC2 signalling deficits can be circumvented by treatment with other non-VIP agonists [37, 38], revealing functional redundancy in intercellular communication within the SCN.
4.2 GRP
Gastrin-releasing peptide (GRP) is synthesised by ~14 % of neurons in the central region of the SCN [3], and its cognate receptor, BB2, is also expressed in the SCN. Similar to VIP neurons, some GRP neurons are retinorecipient, and GRP microinjected into the SCN causes light-like phase shifts in wheel-running behaviour [39]. Further, GRP resets rodent SCN firing rate rhythms in vitro with a temporal pattern of sensitivity resembling that of glutamate. GRP and associated peptides activate SCN neurons to elevate firing rate. A greater proportion of SCN cells respond to GRP when tested during the subjective night, and these actions are blocked by a BB2 receptor antagonist. In mice, transgenic deletion of BB2 expression does not affect ongoing rhythmicity but does reduce the phase-resetting actions of light [9].
4.3 AVP
Arginine vasopressin (AVP) is arguably the most abundant of SCN neuropeptides (~37 % of rat SCN neurons [3]). AVP is expressed in neurons along the medial border of the SCN as well as in a small cluster of neurons near the ventrolateral pole of the SCN. This neuropeptide is often used as the neurochemical marker of the ‘shell’ compartment. AVP expression overtly varies over the 24 h light-dark (as well as constant dark) cycle, with high levels of mRNA/protein during the middle of the day and lower levels at night. AVP is a clock-controlled gene and its expression is predictably altered by clock gene mutations. Electrophysiological investigation reveals that AVP activates rodent SCN neurons [40]. These effects are mediated by AVP V1a and V1b receptors that are expressed in the SCN [41].
The role of AVP in the circadian rhythms is suggested to be noncritical since Brattleboro rats that do not express AVP continue to show rhythmic behaviour. However, recently, a study has indicated that AVP-V1a/1b receptor signals are necessary for effectively controlling the magnitude of resetting to changes in the external light-dark cycle [41]. In mice lacking both V1a and V1b expression, the SCN resets to large changes in the phase of the LD cycle much more quickly than in wild-type animals.
5 Outputs
The peptide prokineticin-2 (PK2) is synthesised in the mouse SCN and is implicated in conveying circadian information to the rest of the brain. Levels of PK2 mRNA in the SCN vary across the light-dark and circadian cycles. The main receptor for PK2 in the brain is PK2r. PK2r is expressed by distinct populations of the SCN including AVP- and GRP-containing neurons, and PK2 acting via the PK2r alters SCN electrical activity [42]. PK2 is predominantly excitatory in the SCN and appears to act by reducing GABA release. Further, injections of PK2 into the lateral ventricle of rats at night, when the level of PK2 in the SCN is very low, reduce motor activity. Since PK2 mRNA peaks during the day when rats are behaviourally quiescent, then this suggests that PK2 functions as an inhibitory output signal. It is unclear whether this represents a direct inhibition of motor centres or arises through indirect pathways. PK2 expressing cells in the SCN potentially innervate a number of extra-SCN sites including the dorsomedial hypothalamus and the habenula (Hb), and PK2r mRNA is also expressed in these structures. There has been very little examination of the influence of PK2 at the target sites of SCN efferents, but, in the lateral Hb, PK2 acts presynaptically to enhance GABA release and reduce neuronal activity [43]. The model that has emerged is that PK2 released from SCN efferents during the day acts to suppress motor centres of the brain and that as PK2 release declines during the late day, this disinhibits these motor centres and is permissive of the expression of locomotor activity. Unfortunately, there has been limited follow-up on PK2’s role, and while this model remains an intriguing possibility, more insightful experiments are required to be fully confident that PK2 does indeed function as an SCN output signal.
There is limited evidence that other factors such as transforming growth factor-alpha (TGFα) [44] and cardiotrophin-like cytokine [45] are involved, but, aside from these early initial investigations, there is little additional evidence to support such a role for these chemicals.
6 Circadian Organisation in Diurnal Species
Most of our current knowledge of the biological timekeeping mechanisms in mammals arises from laboratory investigations focused on nocturnal rodent models, but studies in diurnal species are much more limited. One layer of complexity in understanding the neural mechanism underpinning diurnal temporal niche preference in mammals is that diurnalism has re-emerged in several independent evolutionary lineages [46]. Thus, comparative analysis of diurnal species from different taxonomic groups is necessary to identify convergent adaptations that are common to a diurnal niche and therefore more likely to be shared by most diurnal species, including humans [47].
From a theoretical point of view, the different patterns of behaviour and physiology in diurnal and nocturnal species could be the result of (1) changes in the central master oscillator and in the mechanisms involved in its entrainment to the LD cycle, but similar coupling of the clock to the functions it controls, and (2), alternatively, a circadian master oscillator with similar fundamental features and entrainment to the LD cycle but with different coupling between the clock and the functions that it regulates (Fig. 12.2) [48].
Schematic diagram of the possible neural mechanisms underlying temporal niche preference. Different behavioural outputs in diurnal and nocturnal animals could arise from (a) a reversed phase relationship between the SCN and the external light-dark cycle (LD) and, (b) alternatively, changes in the coupling between the SCN and the functions that it controls, including reversed expression and release of SCN timekeeping signals (b1) or altered responsiveness in target areas downstream from the SCN (b2) (Adapted from [81])
6.1 Inputs to the Central Pacemaker
In the natural environment, nocturnal and diurnal species differ considerably in the lighting conditions which they routinely experience. Indeed, diurnal species are exposed to light for longer durations and at higher intensities than nocturnal ones. It is therefore not surprising that evolutionary adaptation to a diurnal temporal niche has resulted in a visual system that is distinctly different from nocturnal animals. This includes, for example, lens pigmentation which confers strong filtering properties and high cone density in the retina for colour vision processing [49]. In addition, anatomical studies have revealed that some diurnal species possess thicker optic nerves and more prominent optic chiasm and tracts than nocturnal animals [50, 51]. However, the significance of such differences, from a circadian point of view, is though unknown.
Adaptation to light-saturated environments by diurnal species could also account for the finding that the circadian systems of such species appear to be less sensitive to light than those of nocturnal animals. This is reflected in higher thresholds for inducing sustained photic responses in SCN neurons in diurnal rodents [52]. Intriguingly, differences between diurnal and nocturnal species are reported in the polarity of electrophysiological responses of their SCN neurons to photic stimulation. For example, the majority of photically responsive SCN neurons are suppressed by light in diurnal species, including the degu (Octodon degus [52]) and 13-lined ground squirrel (Spermophilus tridecemlineatus [53]), in contrast to predominantly excitatory responses observed in SCN neurons of nocturnal animals. This finding was also replicated in vitro by recording SCN responses to optic nerve stimulation [54]. Additional studies using other diurnal species are needed to confirm if these differences in the physiological effects of light inputs are a common feature of diurnalism.
A fundamental property of the circadian system is the PRC which describes the resetting effects of light on the SCN clock. As stated earlier, the shifting effects of light on the SCN clock depend on the time of day when light is applied. With pulses of light given during the night, the pattern of PRC appears to be quite similar across a wide range of diurnal and nocturnal species. Thus, a light pulse early and late in the subjective night causes phase delays and phase advances, respectively, regardless of the timing of activity. However, with light pulses given during the subjective day, differences between diurnal and nocturnal species emerge. For several hours during the subjective day, nocturnal animals are generally unresponsive to the phase-shifting actions of light pulses (the so-called dead zone), whereas in diurnal species, including degus and humans, the duration of photic insensitivity during daytime is much shorter or even absent [47, 55]. Indeed, this daytime sensitivity of the diurnal SCN to light is also mirrored when the activity of the immediate early gene, c-fos, is examined. Light pulses during the subjective day cause changes in c-fos expression in the diurnal SCN, but have no effect in nocturnal rodents [56]. However, these subtle variations in the shape of the PRC do not seem sufficient to explain the 180° phase reversal in the entrained activity pattern between nocturnal and diurnal species [47].
Anatomical pathways involved in conveying non-photic internal arousal to the SCN in nocturnal animals are conserved in diurnal species. However, it is unclear if their neurophysiological characteristics and the processing of information conveyed to the SCN are also conserved. As stated previously, arousal-promoting cues are conveyed to the SCN by the GHT, which co-releases NPY and GABA in the SCN, as well as via a brainstem pathway originating in the median raphe, which uses serotonin as its neurochemical signal. To date, studies comparing the role of GABA or serotonin in the SCN of diurnal and nocturnal animals are few, and the limited evidence available suggests that these two neurochemicals elicit distinct behavioural effects in diurnal and nocturnal animals [57].
Microinjections of GABAA agonists, such as muscimol, directly into the SCN during the mid-subjective day cause phase shifts of behavioural activity in both diurnal and nocturnal animals. Interestingly, these shifts are in opposite directions, phase advance in nocturnal hamsters and large phase delay in diurnal Nile grass rat, Arvicanthis niloticus. These effects of muscimol are not prevented by blocking voltage-gated sodium channels with tetrodotoxin, indicating that they most likely represent direct actions of GABAA activation in the SCN region (for review, see [51]). These phase alterations have been associated with downregulation of both Per1 and Per2 expressions in nocturnal animals and only decrease of Per2 in the SCN of A. niloticus. GABAergic stimulation reduces the shifting effects of light at night in both diurnal and nocturnal animals [58].
The circadian window of sensitivity to serotonin-resetting actions also appears to differ between the nocturnal and diurnal species studied to date [57]. In nocturnal species, such as hamster, in vivo administration of the 5-HT agonist (8-OH-DPAT) causes phases advances only during the mid-subjective day. By contrast, in the diurnal Arvicanthis ansorgei, serotonergic activation induces phase advances during the subjective night. Intriguingly, serotonergic stimulation potentiates photic-resetting effect at night in diurnal A. ansorgei [59], instead of reducing it as it occurs in nocturnal animals.
Similarly to nocturnal animals, orexin fibres are found in the SCN of diurnal species [60]. Unfortunately, how orexin influences SCN activity and its potential role in conveying/modulating non-photic cues to the SCN in diurnal species is unknown. Importantly, the temporal pattern of activation of orexin cells is phase reversed in nocturnal and diurnal animals, that is, higher activation at night in nocturnal animals and during the day in diurnal rodents [48]. Thus, it will be interesting to study if orexinergic cues would induce phase shifts of the SCN only at night and if such cues can potentiate light-induced phase shifts as is seen for serotonergic stimulation.
6.2 The Central Pacemaker
Interspecific differences in the morphological organisation of the SCN at both the structural and neurochemical levels have been found; however, none seem clearly associated with diurnality. For example, the SCN in some diurnal species, including O. degus [61] and Funambulus palmarum [50], have an elliptical shape in the coronal plane, in contrast with the typical oval shape described for nocturnal rats and hamsters. However, this feature cannot be generalised to all diurnal species as diurnal Nile grass rats, for example, also have oval-shaped SCN [62]. The typical organisation of the SCN into ‘core’ and ‘shell’ described in nocturnal species seems to be present in some but not all diurnal species. For example, cell density in degus SCN does not appear greater in any part of the nucleus. Interestingly, AVP cells in this species are found not only in the dorsomedial part but also in the central region of the SCN [61]. Recently, a study in capuchin monkeys has also reported the presence of AVP in both ventral and dorsal SCN regions [63]. Such pattern contrasts with the typical distribution of the AVP cell bodies limited to the dorsomedial SCN found in nocturnal species. Future studies are needed to explore the functional impact of these differences.
Despite the varied activity patterns across animal species, some features of the SCN are fundamentally conserved. For example, global SCN metabolic activity (assessed by 2-deoxyglucose) peaks during the day in both nocturnal and diurnal rodent species [5]. However, whether more subtle regional differences in SCN metabolism are related to temporal niche remains to be examined. Further, the possibility that the SCN output signal generated differs between nocturnal and diurnal species needs to be interrogated. For example, such output signals may vary in terms of their ratio of synthesis and release of inhibitory vs excitatory neurotransmitters [46].
Studies of electrical activity rhythms in diurnal species are scarce and the available data variable. In vivo multiunit activity SCN recordings made from the diurnal chipmunk (Eutamias sibericus) mirror the population-level metabolic rhythm. Thus, chipmunk SCN neurons generate daily rhythms in electrical outputs, being significantly more active during the day than at night [64]. However, a lack of significant daily variation in spontaneous firing was found in the degu SCN (in both light-responsive and light-unresponsive cells) [52]. At present, there is insufficient data to conclude on the nature of SCN neuronal rhythms in diurnal species, and many more such assessments from other diurnal species are necessary. Further, the possibility that temporal niche preference may be captured by one cell population but not another needs examining. Therefore, more detailed investigations are required to determine the SCN neuronal properties that contribute to the emergence of nocturnal and diurnal behavioural patterns.
At the molecular level, there do not appear to be any major alterations in the daily pattern of the SCN TTFL that can account for differences in temporal niche. Indeed, the phasing of core-clock gene expression, and their corresponding protein rhythms in the SCN, is remarkably similar in diurnal and nocturnal mammals despite major differences in their activity patterns (e.g. [65, 66]). Patterns of Per1 and Per2 expression, with high levels during the light phase and low levels at night, have been found in all diurnal rodent species studied so far including degus, barbary-striped grass mouse, ground squirrel, A. ansorgei and A. niloticus [58, 65, 67–70] and in typical nocturnal species, such as rats, mice and hamsters. In addition, recent studies comparing individuals with different activity patterns (e.g. day active vs night active) within the same species have found no differences in the phases of Per1 and Per2 clock gene expression or in their protein rhythms [66, 67].
Interestingly, however, the expression patterns of these core-clock genes outside the SCN appear to peak in phase with the animal’s activity pattern. Indeed, in extra-SCN brain regions (e.g. the hippocampus and cortex), Per1/Per2 rhythms in nocturnal animals peak at night, which is nearly 180° out of phase with the peaks seen in diurnal species [65, 67, 69]. Together, this suggests that the different temporal niche preferences in most diurnal or nocturnal species should result from different coupling between the clock and functions that it regulates.
Two cases are noteworthy to mention where changes in the pattern of coupling between the SCN and the LD cycle potentially influence an animal’s activity phase preference: (i) Mice lacking the inner-retinal photopigment melanopsin (OPN4) and RPE65, a key protein used in retinal chromophore recycling, exhibit a switch from nocturnal to diurnal activity. The phase of clock gene expression rhythms is also reversed in the SCN of these animals suggesting that neural responses to light upstream of the SCN can produce acute temporal-niche switching. In the same way, a switch from nocturnal to diurnal phenotype also occurs when wild-type mice are transferred from an LD cycle with standard light intensity to an LD cycle with scotopic levels of light during the 12 h light phase [71]. (ii) In the case of the subterranean mole rat Spalax ehrenbergi, most individuals are active during the light phase, but a subset of them can switch from being active during daytime to becoming active at night. Interestingly, the molecular clock gene, Per2, also shifts to exhibit a peak during the dark phase in the SCN, while Per1 oscillates in a biphasic manner [72].
6.3 Circadian Outputs
The SCN convey temporal information to other brain regions and to peripheral organs through rhythmic release of output signals (potentially including AVP, VIP, TGFα, PK2, GABA). Oppositely phased activity rhythms in nocturnal and diurnal species may result from reversed expression and release of these output signals (Fig. 12.2(b1)). However, to date, comparative analyses of PK2 and TGFα expression in diurnal and nocturnal species have revealed similar rhythmic profiles regardless of their pattern of activity [73, 74]. In the same way, the daily pattern of expression of VIP mRNA under LD conditions in the SCN of diurnal and nocturnal animals seems overtly similar [75]. AVP expression is under clock control. Measurement of its concentration in cerebrospinal fluid, as well as AVP mRNA expression in the SCN, reveals circadian rhythms in AVP peaking during daytime. Hence, both transcription and release of AVP appear to peak during daytime regardless on the animal’s behavioural pattern. Collectively, these studies suggest that the fundamental mechanisms for diurnality should be located downstream the SCN. Thus, target areas would show reversed responses to SCN output signals (Fig. 12.2(b2)). An interesting example supporting this idea is the control of corticosterone rhythms by AVP-containing projections from the SCN to the paraventricular nucleus of the hypothalamus (PVN). Plasma corticosteroids (corticosterone in rats and mice; cortisol in degus, humans, and non-human primates) show daily variations with maximal values at dusk in nocturnal species and at dawn in diurnal mammals. This prepares the organisms for the forthcoming period of increased activity (night or day, respectively). Intriguingly, PVN neurons of diurnal and nocturnal rodents show different responsiveness to AVP administration. Indeed, AVP infused in the PVN of the diurnal grass rat (A. ansorgei) stimulates release of plasma corticosterone. By contrast, the same treatment has a strong inhibitory effect on corticosterone levels in nocturnal rats [76]. The authors propose that changes in the phenotype of the neurons targeted by the SCN efferents (i.e. glutamatergic instead of GABAergic) could underlie these reversed responses to AVP in animals with different activity patterns.
7 Summary and Questions of Interests
-
SCN neurons exhibit intrinsic circadian variation in molecular, metabolic, and electrophysiological characteristics.
-
Regional differences in neurochemical and timekeeping characteristics in the SCN are pronounced in some species.
-
SCN molecular clock does not appear to differ between nocturnal and diurnal species.
-
1.
What processes and mechanisms make an animal diurnal?
-
2.
How do SCN output signals influence activity in specific target areas?
-
3.
Why are ‘core’ and ‘shell’ compartments more discernable in some species and not others?
-
4.
What are the mechanisms underlying temporal niche switching within the same species?
Suggested Further Reading
Very recent studies indicate new roles for known neuropeptides in the SCN [77, 78] and identify a new G-protein-coupled receptor and its function in the SCN [79]. Further, interactions between the SCN and other hypothalamic regions shape diurnal temperature preference [80].
References
Moore RY, Eichler VB (1972) Loss of a circadian adrenal corticosterone rhythm following suprachiasmatic lesions in the rat. Brain Res 42:201–206
Stephan FK, Zucker I (1972) Circadian rhythms in drinking behavior and locomotor activity of rats are eliminated by hypothalamic lesions. Proc Natl Acad Sci U S A 69:1583–1586
Moore RY (2013) The suprachiasmatic nucleus and the circadian timing system. Prog Mol Biol Transl Sci 119:1–28
van den Pol AN (1991) The suprachiasmatic nucleus: morphological and cytochemical substrates for cellular interaction. In: Klein DC, Moore RY, Reppert SM (eds) Suprachiasmatic nucleus: the mind’s clock. Oxford University Press, New York, pp 17–50
Schwartz WJ (1991) SCN metabolic activity in vivo. In: Klein DC, Moore RY, Reppert SM (eds) Suprachiasmatic nucleus: the mind’s clock. Oxford University Press, New York, pp 144–156
Groos G, Hendriks J (1982) Circadian rhythms in electrical discharge of rat suprachiasmatic neurones recorded in vitro. Neurosci Lett 34:283–288
Inouye ST, Kawamura H (1979) Persistence of circadian rhythmicity in a mammalian hypothalamic “island” containing the suprachiasmatic nucleus. Proc Natl Acad Sci U S A 76:5962–5966
Welsh DK, Logothetis DE, Meister M, Reppert SM (1995) Individual neurons dissociated from rat suprachiasmatic nucleus express independently phased circadian firing rhythms. Neuron 14:697–706
Brown TM, Piggins HD (2007) Electrophysiology of the suprachiasmatic circadian clock. Prog Neurobiol 82:229–255
Golombek DA, Rosenstein RE (2010) Physiology of circadian entrainment. Physiol Rev 90:1063–1102
Hughes AT, Piggins HD (2012) Feedback actions of locomotor activity to the circadian clock. Prog Brain Res 199:305–336
Mrosovsky N (1996) Locomotor activity and non-photic influences on circadian clocks. Biol Rev Camb Philos Soc 71:343–372
Hattar S, Kumar M, Park A, Tong P, Tung J, Yau KW, Berson DM (2006) Central projections of melanopsin-expressing retinal ganglion cells in the mouse. J Comp Neurol 497:326–349
Ebling FJ (1996) The role of glutamate in the photic regulation of the suprachiasmatic nucleus. Prog Neurobiol 50:109–132
Hannibal J (2002) Neurotransmitters of the retino-hypothalamic tract. Cell Tissue Res 309:73–88
Harrington ME (1997) The ventral lateral geniculate nucleus and the intergeniculate leaflet: interrelated structures in the visual and circadian systems. Neurosci Biobehav Rev 21:705–727
Albers HE, Ferris CF (1984) Neuropeptide Y: role in light-dark cycle entrainment of hamster circadian rhythms. Neurosci Lett 50:163–168
Morin LP (2013) Neuroanatomy of the extended circadian rhythm system. Exp Neurol 243:4–20
Marchant EG, Watson NV, Mistlberger RE (1997) Both neuropeptide Y and serotonin are necessary for entrainment of circadian rhythms in mice by daily treadmill running schedules. J Neurosci 17:7974–7987
Belle MD, Hughes AT, Bechtold DA, Cunningham P, Pierucci M, Burdakov D, Piggins HD (2014) Acute suppressive and long-term phase modulation actions of orexin on the mammalian circadian clock. J Neurosci 34:3607–3621
Mohawk JA, Takahashi JS (2011) Cell autonomy and synchrony of suprachiasmatic nucleus circadian oscillators. Trends Neurosci 34:349–358
Reppert SM, Weaver DR (2002) Coordination of circadian timing in mammals. Nature 418:935–941
Preitner N, Damiola F, Molina LL, Zakany J, Duboule D, Albrecht U, Schibler U (2002) The orphan nuclear receptor REV-ERB alpha controls circadian transcription within the positive limb of the mammalian circadian oscillator. Cell 110:251–260
Goriki A, Hatanaka F, Myung J, Kim JK, Yoritaka T, Tanoue S, Abe T, Kiyonari H, Fujimoto K, Kato Y, Todo T, Matsubara A, Forger D, Takumi T (2014) A novel protein, CHRONO, functions as a core component of the mammalian circadian clock. PLoS Biol 12, e1001839
Meng QJ, Logunova L, Maywood ES, Gallego M, Lebiecki J, Brown TM, Sladek M, Semikhodskii AS, Glossop NR, Piggins HD, Chesham JE, Bechtold DA, Yoo SH, Takahashi JS, Virshup DM, Boot-Handford RP, Hastings MH, Loudon AS (2008) Setting clock speed in mammals: the CK1 epsilon tau mutation in mice accelerates circadian pacemakers by selectively destabilizing PERIOD proteins. Neuron 58:78–88
Siepka SM, Yoo SH, Park J, Song W, Kumar V, Hu Y, Lee C, Takahashi JS (2007) Circadian mutant overtime reveals F-box protein FBXL3 regulation of cryptochrome and period gene expression. Cell 129:1011–1023
Godinho SI, Maywood ES, Shaw L, Tucci V, Barnard AR, Busino L, Pagano M, Kendall R, Quwailid MM, Romero MR, O’Neill J, Chesham JE, Brooker D, Lalanne Z, Hastings MH, Nolan PM (2007) The after-hours mutant reveals a role for Fbxl3 in determining mammalian circadian period. Science 316:897–900
Guilding C, Scott F, Bechtold DA, Brown TM, Wegner S, Piggins HD (2013) Suppressed cellular oscillations in after-hours mutant mice are associated with enhanced circadian phase-resetting. J Physiol 591:1063–1080
Rey G, Reddy AB (2015) Interplay between cellular redox oscillations and circadian clocks. Diabetes Obes Metab 17(Suppl 1):55–64
Yan L, Karatsoreos I, Lesauter J, Welsh DK, Kay S, Foley D, Silver R (2007) Exploring spatiotemporal organization of SCN circuits. Cold Spring Harb Symp Quant Biol 72:527–541
Moore RY, Silver R (1998) Suprachiasmatic nucleus organization. Chronobiol Int 15:475–487
Piggins HD, Cutler DJ (2003) The roles of vasoactive intestinal polypeptide in the mammalian circadian clock. J Endocrinol 177:7–15
Vosko AM, Schroeder A, Loh DH, Colwell CS (2007) Vasoactive intestinal peptide and the mammalian circadian system. Gen Comp Endocrinol 152:165–175
Aton SJ, Colwell CS, Harmar AJ, Waschek J, Herzog ED (2005) Vasoactive intestinal polypeptide mediates circadian rhythmicity and synchrony in mammalian clock neurons. Nat Neurosci 8:476–483
Harmar AJ, Marston HM, Shen S, Spratt C, West KM, Sheward WJ, Morrison CF, Dorin JR, Piggins HD, Reubi JC, Kelly JS, Maywood ES, Hastings MH (2002) The VPAC(2) receptor is essential for circadian function in the mouse suprachiasmatic nuclei. Cell 109:497–508
Hughes AT, Piggins HD (2008) Behavioral responses of Vipr2−/− mice to light. J Biol Rhythms 23:211–219
Brown TM, Hughes AT, Piggins HD (2005) Gastrin-releasing peptide promotes suprachiasmatic nuclei cellular rhythmicity in the absence of vasoactive intestinal polypeptide-VPAC2 receptor signaling. J Neurosci 25:11155–11164
Maywood ES, Chesham JE, O’Brien JA, Hastings MH (2011) A diversity of paracrine signals sustains molecular circadian cycling in suprachiasmatic nucleus circuits. Proc Natl Acad Sci U S A 108:14306–14311
Piggins HD, Antle MC, Rusak B (1995) Neuropeptides phase-shift the mammalian circadian pacemaker. J Neurosci 15:5612–5622
Ingram CD, Ciobanu R, Coculescu IL, Tanasescu R, Coculescu M, Mihai R (1998) Vasopressin neurotransmission and the control of circadian rhythms in the suprachiasmatic nucleus. Prog Brain Res 119:351–364
Yamaguchi Y, Suzuki T, Mizoro Y, Kori H, Okada K, Chen Y, Fustin JM, Yamazaki F, Mizuguchi N, Zhang J, Dong X, Tsujimoto G, Okuno Y, Doi M, Okamura H (2013) Mice genetically deficient in vasopressin V1a and V1b receptors are resistant to jet lag. Science 342:85–90
Li JD, Hu WP, Zhou QY (2012) The circadian output signals from the suprachiasmatic nuclei. Neurobiol Circadian Timing 199:119–127
Sakhi K, Wegner S, Belle MD, Howarth M, Delagrange P, Brown TM, Piggins HD (2014) Intrinsic and extrinsic cues regulate the daily profile of mouse lateral habenula neuronal activity. J Physiol 592:5025–5045
Kramer A, Yang FC, Snodgrass P, Li X, Scammell TE, Davis FC, Weitz CJ (2001) Regulation of daily locomotor activity and sleep by hypothalamic EGF receptor signaling. Science 294:2511–2515
Kraves S, Weitz CJ (2006) A role for cardiotrophin-like cytokine in the circadian control of mammalian locomotor activity. Nat Neurosci 9:212–219
Smale L, Lee T, Nunez AA (2003) Mammalian diurnality: some facts and gaps. J Biol Rhythm 18:356–366
Hagenauer MH, Lee TM (2008) Circadian organization of the diurnal Caviomorph rodent, Octodon degus. Biol Rhythm Res 39:269–289
Smale L, Nunez AA, Schwartz MD (2008) Rhythms in a diurnal brain. Biol Rhythm Res 39:305–318
Hut RA, Kronfeld-Schor N, van der Vinne V, De la Iglesia H (2012) In search of a temporal niche: environmental factors. Neurobiology of Circadian Timing 199:281–304
Mammen AP, Jagota A (2011) Immunocytochemical evidence for different patterns in daily rhythms of VIP and AVP peptides in the suprachiasmatic nucleus of diurnal Funambulus palmarum. Brain Res 1373:39–47
Novak CM, Ehlen JC, Albers HE (2008) Photic and nonphotic inputs to the diurnal circadian clock. Biol Rhythm Res 39:291–304
Jiao YY, Lee TM, Rusak B (1999) Photic responses of suprachiasmatic area neurons in diurnal degus (Octodon degus) and nocturnal rats (Rattus norvegicus). Brain Res 817:93–103
Meijer JH, Rusak B, Harrington ME (1989) Photically responsive neurons in the hypothalamus of a diurnal ground squirrel. Brain Res 501:315–323
Jiao YY, Rusak B (2003) Electrophysiology of optic nerve input to suprachiasmatic nucleus neurons in rats and degus. Brain Res 960:142–151
Jewett ME, Rimmer DW, Duffy JF, Klerman EB, Kronauer RE, Czeisler CA (1997) Human circadian pacemaker is sensitive to light throughout subjective day without evidence of transients. Am J Physiol 273:R1800–R1809
Krajnak K, Dickenson L, Lee TM (1997) The induction of Fos-like proteins in the suprachiasmatic nuclei and intergeniculate leaflet by light pulses in degus (Octodon degus) and rats. J Biol Rhythms 12:401–412
Challet E (2007) Minireview: entrainment of the suprachiasmatic clockwork in diurnal and nocturnal mammals. Endocrinology 148:5648–5655
Novak CM, Ehlen JC, Paul KN, Fukuhara C, Albers HE (2006) Light and GABA)(A) receptor activation alter period mRNA levels in the SCN of diurnal Nile grass rats. Eur J Neurosci 24:2843–2852
Cuesta M, Mendoza J, Clesse D, Pevet P, Challet E (2008) Serotonergic activation potentiates light resetting of the main circadian clock and alters clock gene expression in a diurnal rodent. Exp Neurol 210:501–513
Nixon JP, Smale L (2007) A comparative analysis of the distribution of immunoreactive orexin A and B in the brains of nocturnal and diurnal rodents. Behav Brain Funct 3:28
Goel N, Lee TM, Smale L (1999) Suprachiasmatic nucleus and intergeniculate leaflet in the diurnal rodent Octodon degus: retinal projections and immunocytochemical characterization. Neuroscience 92:1491–1509
Smale L, Boverhof J (1999) The suprachiasmatic nucleus and intergeniculate leaflet of Arvicanthis niloticus, a diurnal murid rodent from East Africa. J Comp Neurol 403:190–208
Rocha VA, Frazao R, Campos LM, Mello P, Donato J Jr, Cruz-Rizzolo RJ, Nogueira MI, Pinato L (2014) Intrinsic organization of the suprachiasmatic nucleus in the capuchin monkey. Brain Res 1543:65–72
Sato T, Kawamura H (1984) Circadian rhythms in multiple unit activity inside and outside the suprachiasmatic nucleus in the diurnal chipmunk (Eutamias sibiricus). Neurosci Res 1:45–52
Vosko AM, Hagenauer MH, Hummer DL, Lee TM (2009) Period gene expression in the diurnal degu (Octodon degus) differs from the nocturnal laboratory rat (Rattus norvegicus). Am J Physiol Regul Integr Comp Physiol 296:R353–R361
Ramanathan C, Stowie A, Smale L, Nunez AA (2010) Phase preference for the display of activity is associated with the phase of extra-suprachiasmatic nucleus oscillators within and between species. Neuroscience 170:758–772
Otalora BB, Hagenauer MH, Rol MA, Madrid JA, Lee TM (2013) Period gene expression in the brain of a dual-phasing rodent, the Octodon degus. J Biol Rhythms 28:249–261
Chakir I, Dumont S, Pevet P, Ouarour A, Challet E, Vuillez P (2015) The circadian gene clock oscillates in the suprachiasmatic nuclei of the diurnal rodent Barbary striped grass mouse, Lemniscomys barbarus: a general feature of diurnality? Brain Res 1594:165–172
Mrosovsky N, Edelstein K, Hastings MH, Maywood ES (2001) Cycle of period gene expression in a diurnal mammal (Spermophilus tridecemlineatus): implications for nonphotic phase shifting. J Biol Rhythms 16:471–478
Caldelas I, Poirel VJ, Sicard B, Pevet P, Challet E (2003) Circadian profile and photic regulation of clock genes in the suprachiasmatic nucleus of a diurnal mammal Arvicanthis ansorgei. Neuroscience 116:583–591
Doyle SE, Castrucci AM, McCall M, Provencio I, Menaker M (2006) Nonvisual light responses in the Rpe65 knockout mouse: rod loss restores sensitivity to the melanopsin system. Proc Natl Acad Sci U S A 103:10432–10437
Oster H, Avivi A, Joel A, Albrecht U, Nevo E (2002) A switch from diurnal to nocturnal activity in S.ehrenbergi is accompanied by an uncoupling of light input and the circadian clock. Curr Biol 12:1919–1922
Lambert CM, Machida KK, Smale L, Nunez AA, Weaver DR (2005) Analysis of the prokineticin 2 system in a diurnal rodent, the unstriped Nile grass rat (Arvicanthis niloticus). J Biol Rhythms 20:206–218
Tournier BB, Dardente H, Vuillez P, Pevet P, Challet E (2007) Expression of Tgfalpha in the suprachiasmatic nuclei of nocturnal and diurnal rodents. Neuroscience 145:1138–1143
Dardente H, Menet JS, Challet E, Tournier BB, Pevet P, Masson-Pevet M (2004) Daily and circadian expression of neuropeptides in the suprachiasmatic nuclei of nocturnal and diurnal rodents. Brain Res Mol Brain Res 124:143–151
Kalsbeek A, Verhagen LA, Schalij I, Foppen E, Saboureau M, Bothorel B, Buijs RM, Pevet P (2008) Opposite actions of hypothalamic vasopressin on circadian corticosterone rhythm in nocturnal versus diurnal species. Eur J Neurosci 27:818–827
Lee IT, Chang AS, Manandhar M, Shan Y, Fan J, Izumo M, Ikeda Y, Motoike T, Dixon S, Seinfeld JE, Takahashi JS, Yanagisawa M (2015) Neuromedin s-producing neurons act as essential pacemakers in the suprachiasmatic nucleus to couple clock neurons and dictate circadian rhythms. Neuron 85:1086–1102
Mieda M, Ono D, Hasegawa E, Okamoto H, Honma K, Honma S, Sakurai T (2015) Cellular clocks in AVP neurons of the SCN are critical for interneuronal coupling regulating circadian behavior rhythm. Neuron 85:1103–1116
Doi M, Murai I, Kunisue S, Setsu G, Uchio N, Tanaka R, Kobayashi S, Shimatani H, Hayashi H, Chao HW, Nakagawa Y, Takahashi Y, Hotta Y, Yasunaga J, Matsuoka M, Hastings MH, Kiyonari H, Okamura H (2016) Gpr176 is a Gz-linked orphan G-protein-coupled receptor that sets the pace of circadian behaviour. Nat Commun 7:10583
Guzman-Ruiz MA, Ramirez-Corona A, Guerrero-Vargas NN, Sabath E, Ramirez-Plascencia OD, Fuentes-Romero R, Leon-Mercado LA, Basualdo SM, Escobar C, Buijs RM (2015) Role of the suprachiasmatic and arcuate nuclei in diurnal temperature regulation in the rat. J Neurosci 35:15419–15429
Chiesa JJ, Aguzzi J, Garcia JA, Sarda F, de la Iglesia HO (2010) Light intensity determines temporal niche switching of behavioral activity in deep-water Nephrops norvegicus (Crustacea: Decapoda). J Biol Rhythms 25:277–287
Funding
BBO is funded by Fundacion Seneca 19701/PD/14 and HDP by project grants from the Biotechnology and Biological Sciences Research Council (BBSRC) (BB/M02329X and BB/L007665).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer (India) Pvt. Ltd.
About this chapter
Cite this chapter
Bano-Otalora, B., Piggins, H.D. (2017). The Mammalian Neural Circadian System: From Molecules to Behaviour. In: Kumar, V. (eds) Biological Timekeeping: Clocks, Rhythms and Behaviour. Springer, New Delhi. https://doi.org/10.1007/978-81-322-3688-7_12
Download citation
DOI: https://doi.org/10.1007/978-81-322-3688-7_12
Published:
Publisher Name: Springer, New Delhi
Print ISBN: 978-81-322-3686-3
Online ISBN: 978-81-322-3688-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)