Abstract
Acorus calamus Linn. is a traditional medicinal plant included in “lekhaneyagana” (which means “reduce excess fat”), a pharmacological classification mentioned by Charaka in Charakasamhita. Traditionally, this plant has been prescribed as a first-line treatment for many ailments such as digestive problems, diabetes, obesity, and related problems. The purpose of the present study was to check the effect of methanolic fraction of A. calamus on serum lipids, Lecithin-cholesterol acyltransferase (LCAT) enzyme, and apolipoproteins. We found that treatment of A. calamus at concentrations of 200 and 400 mg/kg body weight was able to reduce total cholesterol (T-c), triglycerides (TG), and low-density lipoprotein cholesterol (LDL-c) levels and increase high-density lipoprotein cholesterol (HDL-c) levels compared to model control group. A. calamus treatment results in activation of LCAT enzyme, which helps in the maturation of HDL-c. ApoA1 level was found to be increased considerably by the administration of A. calamus extract, and the Apo B level was decreased. GCMS analysis of the A. calamus extract showed the presence of compound, Friedelan-3-one. The result suggests the use of methanol fraction of A .calamus, as a potential drug for controlling the cholesterol level.
Access provided by Autonomous University of Puebla. Download conference paper PDF
Similar content being viewed by others
Keywords
Introduction
Acorus calamus Linn. (Family: Araceae), also known as sweet flag, is a semiaquatic perennial herb with creeping and branched aromatic rhizome. The roots and rhizomes of this plant have been used in the Indian and Chinese systems of medicine for at least 2,000 years. The essential oils isolated from A. calamus showed anti-hypercholesterolemia, anti-inflammatory, and antioxidative activity, respectively [1–3]. Furthermore, A. calamus extract showed antifungal activity and insecticidal properties [4].
Obesity is an excess proportion of total body fat and is reaching epidemic proportions. Direct health costs attributable to obesity have been estimated at $52 billion in 1995 and $75 billion in 2003 and increasing in alarming rate [5]. A. calamus is a plant included in lekhaneyagana, a pharmacological classification mentioned in “Charakasamhita” which means “reduce excess fat” [6]. In the present study, we screened the methanolic fraction of A. calamus for hypolipidemic effects in diet-induced rat models. The fraction which shows hypolipidemic effect was used to perform GCMS analysis.
Materials and Methods
Preparation of the Root Extract of A. calamus
Root samples of A. calamus were purchased from local market and authenticated by the Centre for Medicinal Plants Research (CMPR), Kottakkal, India. Voucher specimen was processed and deposited (No: CMPR 1486). 2.3 kg of the fine powdered sample was used for Soxhlet extraction using methanol. The homogenate was then filtered using Millipore filtration system 2 (Millipore, USA) and dried using a rotary evaporator at 40°C (273 g). The methanol fraction was loaded to a silica gel column eluted with MeOH: water (18 × 500 mm column), increasing water gradually to supply five fractions, since the 30% MeOH-soluble fraction exhibited considerable hypolipidemic activity [1, 4]. The fourth fraction was used to feed the diet-induced obese rats, and the same was used for GCMS analysis.
Establishment of Experimental Model and Drug Treatment
Sprague Dawley (SD) strain rats of body weight 160 ± 10 g were used for experiments. The animals were housed in polypropylene cages at controlled temperature (22 ± 3°C) and humidity (50 ± 10%) and were kept in 12-h light cycle. The experiment was approved by the Institutional Animal Ethics Committee (IAEC No-KULS/IAEC 2011-04) and performed based on CPCSEA accepted guidelines for care and use of laboratory animals.
Rats were randomly divided into two groups: normal control group (n = 10) and high-fat group (n = 40). The normal control rats were fed with standard diet, and hyperlipidemic rats were fed with high-fat diet [7, 8]. The rats in high-fat model group were randomly divided into four groups with ten rats per group: high-fat model control group, A. calamus I group (200 mg/kg body weight (BW)), A. calamus II group (400 mg/kg BW), and mevinolin (3.0 mg/kg body weight)-treated positive control group [7]. The acute toxicity of the extract was performed, and the doses were chosen according to the acute toxicological study [9, 10]. At the end of the experiment, the overnight fasted rats were sacrificed and blood samples were taken for the estimation of serum T-c, TG, HDL-c, LDL-c [11–14], Lecithin-cholesterol acyltransferase [15], and apolipoproteins [16].
GCMS Analysis of Methanolic Extract of A. calamus
GCMS and MSMS analysis of the methanolic extract of A. calamus was performed using Perkin Elmer Clarus 600 GCMS and Turbomatrix 40 trap GC-HS controlled by Turbo mass software version 5.4.2. One microliter of the sample was injected by direct liquid injection mode into Perkin Elmer Clarus 600 Gas Chromatogram equipped with Elite-5MS, 30 m × 0.25-mm ID × 0.25-um film thickness. The GC conditions were, injection temperature 275°C, helium as carrier gas with flow of 15 psi, and a split ratio of 10. GC effluent was introduced to the mass spectrometer for EI mass spectrometry analysis where data were recorded in full scan or linear mode in the mass range of 10–600 m/z.
Statistical Analysis
All values are presented as mean ± s.d. Statistical comparisons of the groups were made by ANOVA, and each group was compared with the others by Posthoc Fisher’s PLSD test (SPSS Inc-IBM, USA). Statistical significance was defined as P < 0.05.
Results and Discussion
Effect of A. calamus on Serum Lipid Levels in Diet-Induced Obese Rats
The estimated lipid levels in normal control, high-fat model control, extract-treated groups, and positive control are given in Table 47.1. The T-c and TG levels of A. calamus extract-treated groups were lower than the high-fat group; the dose response was also obvious. Moreover, A. calamus facilitates enhancement of cardio-protective lipid HDL-c, and this effect may be due to the increase in activity of lecithin: cholesterol acyl transferase (LCAT) enzyme, which may contribute to the regulation of blood lipids. The effect of A. calamus on HDL-c may be largely attributed to its central function in the reverse cholesterol transport, a process whereby excess cell cholesterol is taken up and processed by HDL particles for further delivery to the liver for metabolism [17, 18].
Treatment of hyperlipidemic rats with A. calamus extract at a dose of 200 and 400 mg/kg body weight lowered the serum LDL-c levels. The mevinolin-treated group had also showed a significant hypocholesterolemic effect in serum as compared to model control group [19, 20].
Effect of A. calamus on LCAT Activity in Diet-Induced Obese Rats
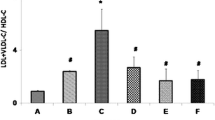
Oral administration of A. calamus extracts that result in significant increase in LCAT activity supports the above result (Fig. 47.1). LCAT is an enzyme that catalyzes the formation of cholesteryl esters on HDL and by that promotes maturation of HDL particles in plasma and facilitates reverse cholesterol transport by maintaining a concentration gradient for the diffusion of cellular unesterified cholesterol to HDL. It helps to improve cholesterol exportation in biliary acids [18, 19]. Similar results were observed for lipid profiles treating Curcuma kwangsiensis polysaccharides [7] and LCAT activity in case of Gymnema sylvestre and Monascus-fermented soybean extracts [21, 22] (Fig. 47.1).
Effect of A. calamus on Serum Apolipoproteins in Diet-Induced Obese Rats
The administration of A. calamus extract significantly increases the apoA1 and drops down Apo B levels. Both lipid-poor apoAI and HDL act as acceptors for cellular lipids, and lipid efflux from cells can be mediated via the cell surface proteins. Apo A-I and Apo A-II are the main protein components of HDL cholesterol, which helps in removing excess cholesterol from the tissues for removal by the liver [23]. On the other hand, Apo B, which is a component of LDL-c, enables tissue cells to take up cholesterol [24].
GCMS Analysis of Methanolic Extract of A. calamus
The GCMS and MSMS analysis done using A.calamus extract shows the presence of Friedelan-3-one (Fig. 47.2). The spectrum and library search of 34.41 min RT peak represents the compound, Friedelan-3-one.
Conclusions
The administration of A.calamus extract reduced serum T-c, TG, and LDL-c levels. Furthermore, A.calamus helps to improve cardio-protective lipid HDL-c. The mechanism of hypolipidemic activity of A. calamus was not studied till date. Our study addresses the shortfall; it is presumed that a higher hypolipidemic effect of A. calamus might be due to the activation of LCAT enzyme. GCMS analysis data of same fraction of A. calamus extract shows that major peak present is Friedelan-3-one. The decrease of serum TG level is also a promising result, since triglycerides are independently related to cardiovascular disease and most of hypercholesterolemic drugs were not able to reduce TG levels. Based on this observation, it can be comprehended that the A.calamus fraction containing compound Friedelan-3-one is able to activate LCAT and exhibit potential hypolipidemic activity. The results constitute a valid scientific groundwork for its medicinal application and valid support for “lekhaneya” action of A.calamus.
Abbreviations
- Apo A1:
-
Apolipoprotein A1
- Apo B:
-
Apolipoprotein B
- GCMS:
-
Gas chromatography mass spectrometry
- HDL-c:
-
High-density lipoprotein cholesterol
- LCAT:
-
Lecithin-cholesterol acyltransferase
- LDL-c:
-
Low-density lipoprotein cholesterol
- MSMS:
-
Tandem mass spectrometry
- RT:
-
Retention time
- T-c:
-
Total cholesterol
- TG:
-
Triglycerides
References
Parab RS, Mengi SA. Hypolipidemic activity of Acorus calamus L. in rats. Fitoterapia. 2002;73:451–5.
Kim H, Han TH, Lee SG. Anti-inflammatory activity of a water extract of Acorus calamus L. leaves on keratinocyte HaCaT cells. J Ethnopharmacol. 2009;122:149–56.
Manikandan S, Srikumar R, Jeya Parthasarathy N, Sheela Devi R. Protective effect of Acorus calamus Linn on free radical scavengers and lipid peroxidation in discrete regions of brain against noise stress exposed rat. Biol Pharm Bull. 2005;28:2327–30.
Sharma PR, Sharma OP, Saxena BP. Effect of sweet flag rhizome oil (Acorus calamus) on hemogram and ultra structure of hemocytes of the tobacco armyworm, Spodoptera litura (Lepidoptera: Noctuidae). Micron. 2008;39:544–51.
Wolf AM, Colditz GA. Current estimates of the economic cost of obesity in the United States. Obes Res. 1998;6:97–106.
Sharma PV. Caraka samhita. Varanasi: Chaukhambha Orientalia; 2000.
Xu C, Haiyan Z, Hua Z, Jianhong Z, Pin D. Effect of Curcuma kwangsiensis polysaccharides on blood lipid profiles and oxidative stress in high-fat rats. Int J Biol Macromol. 2009;44:138–42.
Lien EL, Boyle FG, Wrenn JM, Perry RW, Thompson CA, Borzelleca JF. Comparison of AIN-76A and AIN-93G diets: a 13-week study in rats. Food Chem Toxicol. 2001;39:385–92.
Tajuddin AS, Latif A, Qasmi IA, Amin KMY. An experimental study of sexual function improving effect of Myristica fragrans Houtt. (nutmeg). BMC Complement Altern Med. 2005;5:16.
Pahua-Ramos ME, Ortiz-Moreno A, Chamorro-Cevallos G, Hernández-Navarro MD, Garduño-Siciliano L, Necoechea-Mondragón H, Hernández-Ortega M. Hypolipidemic effect of Avocado (Persea americana Mill) seed in a Hypercholesterolemic mouse model. Plant Food Hum Nutr. 2012;67:10–6.
Rubinstein A. National cholesterol education program, second report of the expert panel on detection, evaluation, and treatment of high blood cholesterol in adults. Circulation. 1995;91:908–9.
Stein EA, Myers GL. National cholesterol education program recommendations for triglyceride measurement: executive summary. The national cholesterol education program working group on lipoprotein measurement. Clin Chem. 1995;41:1421–6.
Rifai N, Warnick GR, Dominiczak MH. Handbook of lipoprotein testing. 2nd ed. Washington, DC: AACC Press; 2001.
Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem. 1972;18:499–502.
Nagasaki T, Akanuma Y. A new colorimetric method for the determination of plasma lecithin-cholesterol acyltransferase activity. Clin Chim Acta. 1977;75:1–5.
Marcovina SM, Albers JJ, Dati F, Ledue TB, Ritchie RF. International federation of clinical chemistry standardization project for measurements of apolipoproteins A-1 and B. Clin Chem. 1991;37:1676–82.
Rader DJ, Alexander ET, Weibel GL, Billheimer J, Rothblat GH. The role of reverse cholesterol transport in animals and humans and relationship to atherosclerosis. J Lipid Res. 2009;50:189–94.
Bainton D, Miller NE, Bolton CH, Yarnell JW, Sweetnam PM, Baker IA, Lewis B, Elwood PC. Plasma triglyceride and high density lipoprotein cholesterol as predictors of ischaemic heart disease in British men. The caerphilly and speedwell collaborative heart disease studies. Br Heart J. 1992;68:60–6.
Wilson PW. High-density lipoprotein, low-density lipoprotein and coronary artery disease. Am J Cardiol. 1990;66:7–10.
Oh PS, Lee SJ, Lim KT. Hypolipidemic and antioxidative effects of the plant glycoprotein (36 kDa) from Rhus verniciflua stokes fruit in Triton WR-1339-induced hyperlipidemic mice. Biosci Biotechnol Biochem. 2006;70:447–56.
Pyo YH, Seong KS. Hypolipidemic effects of Monascus-fermented soybean extracts in rats fed a high-fat and -cholesterol diet. J Agric Food Chem. 2009;57:8617–22.
Shigematsu N, Asano R, Shimosaka M, Okazaki M. Effect of administration with the extract of Gymnema sylvestre R. Br leaves on lipid metabolism in rats. Biol Pharm Bull. 2001;24:713–7.
Grundy SM, Cleeman JI, Merz CN, Brewer Jr HB, Clark LT, Hunninghake DB, Pasternak RC, Smith Jr SC, Stone NJ. Implications of recent clinical trials for the national cholesterol education program adult treatment panel III guidelines. J Am Coll Cardiol. 2004;44:720–32.
Smith JD. Apolipoprotein A-I and its mimetics for the treatment of atherosclerosis. Curr Opin Investig Drugs. 2010;11:989–96.
Acknowledgements
DBT-BIF, Govt. of India is gratefully acknowledged for the support for computational and other allied facilities. Arun KS is thankful to UGC-BSR, New Delhi, for providing the fellowship to carry out the research work.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2012 Springer India
About this paper
Cite this paper
Arun, K.S., Augustine, A. (2012). Hypolipidemic Effect of Methanol Fraction of Acorus calamus Linn. in Diet-Induced Obese Rats. In: Sabu, A., Augustine, A. (eds) Prospects in Bioscience: Addressing the Issues. Springer, India. https://doi.org/10.1007/978-81-322-0810-5_47
Download citation
DOI: https://doi.org/10.1007/978-81-322-0810-5_47
Published:
Publisher Name: Springer, India
Print ISBN: 978-81-322-0809-9
Online ISBN: 978-81-322-0810-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)