Abstract
Bile acids are planar amphipathic molecules that have a polar and a nonpolar face. They are the end products of cholesterol metabolism and are called biological detergents. In the duodenum and upper jejunum, they participate in the digestion and absorption of lipids, including triacylglycerols, phospholipids, cholesterol, and fat-soluble vitamins. The formation of mixed micelles is the best-known property of bile acids, and the activation of pancreatic lipases is another important role. Triacylglycerols account for 90–95% of dietary lipids and are hydrolyzed to 2-monoacylglycerol and free fatty acids. Conjugated bile acids and phospholipids form mixed micelles with these hydrolysates of triacylglycerols, cholesterol, and fat-soluble vitamins. The mixed micelles effectively pass through the unstirred water layer overlying the microvillus border of the enterocytes, and all of the nutrients except for the conjugated bile acids are finally taken up by the cells. Conjugation of bile acids with glycine or taurine maintains water solubility at an acidic pH and prevents nonionic passive absorption from the proximal small intestine, which allows bile acids to be absorbed efficiently by an active transport system at the terminal ileum after the completion of their roles.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Bile acids are the end products of cholesterol metabolism and possess a number of chemical, physiological, and pathophysiological functions. The recent discovery of nuclear and transmembrane G protein-coupled bile acid receptors has interested many scientists in the regulation of lipid and carbohydrate metabolism, inflammation, fibrosis, and carcinogenesis through transcriptional networks and/or signaling cascades [1]. In contrast to the recent rapid development within these topics, the roles of bile acids in digestion and absorption may be the most basic and historic subject that has been studied for more than half a century [2,3,4,5]. In this review, we will summarize the progress in our understanding of the mechanisms of digestion and absorption in the gastrointestinal tract with a particular emphasis on bile acids.
2 Bile Acids as Biological Detergents
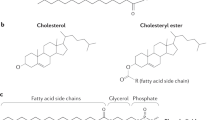
Bile acids are planar amphipathic molecules having a polar and a nonpolar face [6]. The polar face contains hydroxyl and carboxyl groups and is water soluble, while the other face contains two methyl groups and is fat soluble. Thus, bile acids are called biological detergents. In water or a low salt concentration, bile acids form smaller micelles relative to classical detergent micelles because of the planar polarity. However, bile acids also form mixed micelles with a variety of other soluble and insoluble lipidic substances [7].
In human bile, bile acids are almost completely conjugated with either glycine or taurine [8] at the carboxyl group of the side chain through an amide bond. The pKa values of free (unconjugated) bile acids are between 5 and 6.5, while those of glycine- and taurine-conjugated bile acids are approximately 4 and 2, respectively [7]. It indicates that amino acid conjugation promotes ionization and increases the aqueous solubility of bile acids at an acidic pH [9]. Because the postprandial pH of the duodenum is between 3 and 5 [10], the conjugation allows bile acids to maintain water solubility to facilitate digestion in the duodenum and upper jejunum. In addition, the conjugation prevents nonionic passive absorption of bile acids [11]. Patients with genetic defects in bile acid conjugation experience fat-soluble vitamin deficiency because of an inability to form mixed micelles due to rapid nonionic passive absorption of unconjugated bile acids from the proximal small intestine [12]. Thus, conjugation with glycine or taurine allows bile acids to be actively absorbed in the terminal ileum after the completion of their roles.
3 Digestion and Absorption of Lipids
Typical adult Japanese and American diets contain approximately 54 and 85 g of fat per day, providing approximately 26% and 35% of the calorie intake of each individual, respectively [13, 14]. Approximately 90–95% of the dietary lipids are triacylglycerols (triglycerides) that consist of three fatty acids esterified to a glycerol. Dietary lipids also include phospholipids (predominantly phosphatidylcholine), sterols (cholesterol and plant sterols), and other lipids (e.g., fat-soluble vitamins). Phospholipids (essentially phosphatidylcholine) and cholesterol are also provided endogenously via the bile. Daily 7–22 g of phospholipids and approximately 1 g of cholesterol are loaded on the duodenum by biliary secretion, while 4–8 g of phospholipids and 200–400 mg of cholesterol per day are of dietary origin [5, 13, 15].
Digestion and absorption of lipids are complex processes with sequential and interdependent steps [3]. Emulsification, hydrolysis (lipolysis), and solubilization (micellization) are intraluminal key steps before the translocation of lipids across the apical membranes of enterocytes (absorption) [16, 17].
Emulsification
mainly takes place in the stomach. A coarse emulsion (chyme) is produced by antral peristalsis against a closed pylorus, and the squirting of the antral contents through a partially opened pyloric canal into the duodenum produces a fine emulsion [5]. In the duodenum, the fine emulsion particles are generally less than 500 nm in diameter and are extremely stable.
Hydrolysis
is carried out enzymatically by preduodenal and pancreatic lipases (Fig. 3.1). Preduodenal lipases are secreted from the tongue, pharynx, and stomach depending on the species; humans possess predominantly gastric lipase [18]. Human gastric lipase hydrolyzes triacylglycerols, but does not hydrolyze phospholipids or cholesterol ester. It has a pH optimum of 3–6, and conjugated bile acids inhibit the reaction [19, 20]. Pancreatic triacylglycerol lipase (EC 3.1.1.3) is secreted into the duodenum with colipase (as a procolipase) and conjugated bile acids. Most of the dietary triacylglycerols are hydrolyzed by pancreatic triacylglycerol lipase rather than gastric lipase [18]. This enzyme works at the interface between oil and water and an optimum pH optimum of 8–9. The presence of conjugated bile acids inhibits the lipase activity, but colipase overcomes the inhibition with a shift in the pH optimum to 6–7 [21] (see Sect. 3.3.1.1). In general, pancreatic lipases include phospholipase A2 (EC 3.1.1.4) and carboxyl ester lipase (EC 3.1.1.13) [16]. The latter is also called pancreatic nonspecific lipase or cholesterol esterase [5]. Phospholipase A2 preferentially hydrolyzes phospholipids, whereas carboxyl ester lipase has a wide substrate specificity hydrolyzing cholesterol esters; tri-, di-, and monoacylglycerols; phospholipids; lysophospholipids; ceramide; and fat-soluble vitamins [22].
Enzymatic hydrolysis and micellar solubilization of dietary lipids. BA conjugated bile acid, C cholesterol, CE cholesterol ester, DG diacylglycerol, FFA free fatty acid, LPC lysophosphatidylcholine, MG monoacylglycerol, PC phosphatidylcholine, TG triacylglycerol, V fat-soluble vitamin, VE fat-soluble vitamin ester
Solubilization
of lipids and lipolytic products due to the formation of mixed micelles is a critical step for the absorption. Conjugated bile acids play a key role in this process, but polar lipids, including fatty acids and monoglycerides, are also important to increase the solubility of nonpolar lipids such as cholesterol. In contrast to emulsion, mixed micelle solution is optically clear. The diameter of the mixed micelles is 4–5 nm, which means that they have an approximately 100-fold reduced size and 10,000-fold increased surface area relative to the fine emulsion particle. It is also estimated that one fine emulsion particle can form approximately 1 × 106 micelles [23]. The unstirred water layer overlying the microvillus border of the epithelial cells is an intestinal diffusion barrier (Fig. 3.2). The mixed micelles effectively pass into the intermicrovillous spaces and are able to reach to the epithelial cells [24]. However, it seems to be an oversimplification to divide the intestinal phase of lipids into an emulsion and mixed micelles. Under an adequate concentration of bile acids, the lipids are incorporated into mixed micelles. When the bile acid concentration is relatively low but still exceeds the critical micellar concentration, large mixed disclike micelles are formed at approximately 40 nm in diameter. Furthermore, when the relative bile acid concentration is much lower, this results in the formation of vesicles (liposomes) with a diameter of approximately 80–120 nm [5]. Because patients with low upper intestinal bile acid concentration show reasonably good absorption of lipids [25, 26], vesicles may play an important role in the uptake of free fatty acids and monoglycerides by enterocytes [5]. However, the relative roles of the mixed micelles and the vesicles have not been clarified [16].
Micellar solubilization and absorption of lipids in the upper small intestine. ABCG5/ABCG8 ATP-binding cassette G5 and G8, BA conjugated bile acid, C cholesterol, CD36 cluster determinant 36, FFA free fatty acid, LPC lysophosphatidylcholine, MG monoacylglycerol, NPC1L1 Niemann-Pick C1-like 1, PC phosphatidylcholine, SR-BI scavenger receptor class B type I, V fat-soluble vitamin
3.1 Triacylglycerols
Most of the triacylglycerols found in food have long-chain fatty acids with 16–18 carbon atoms. However, a small but variable proportion of triacylglycerols contain fatty acids with only 6–10 carbon atoms and are called medium-chain triacylglycerols [27]. Because medium-chain triacylglycerols are less hydrophobic than long-chain triacylglycerols, the processes of digestion and absorption of these two types of triacylglycerols are somewhat different.
3.1.1 Long-Chain Triacylglycerols
The digestion of dietary triacylglycerols begins in the stomach. In addition to emulsification, gastric lipase hydrolyzes a significant portion of dietary triacylglycerols. This enzyme hydrolyzes medium-chain triacylglycerols better than long-chain triacylglycerols [28] and preferentially acts on the sn-3 position of the triacylglycerols [29] to release diacylglycerols and free fatty acids [19, 20, 30]. The relative contributions of gastric lipase and pancreatic triacylglycerol lipase to the hydrolysis of dietary triacylglycerols were reported to be approximately 1:3, and approximately 40% of the hydrolysis by gastric lipase occurred in the duodenum [31].
In the duodenum and proximal jejunum, the rest of triacylglycerols and diacylglycerols are hydrolyzed by pancreatic triacylglycerol lipase. This enzyme works at the oil-water interface of the emulsion. Conjugated bile acids adsorb onto fat droplets and remove proteins, emulsifiers, and lipolysis products from the lipid surface [32]. However, only triacylglycerol lipase is not removed from the oil-water interface and instigates lipolysis because colipase binds a bile-acid-covered oil-water interface and provides a high-affinity anchor site for triacylglycerol lipase [5, 21, 32]. Pancreatic triacylglycerol lipase preferentially cleaves the ester bond in the sn-1 and sn-3 positions of the triacylglycerols at equal rates [29, 33] so that 2-monoacylglycerol and free fatty acids are formed. A part of 2-monoacylglycerol is further hydrolyzed into glycerol and a free fatty acid by pancreatic triacylglycerol lipase either directly or after isomerization to 1-monoacylglycerol [33, 34]. Carboxyl ester lipase also hydrolyzes the acyl group at the sn-2 position to release glycerol and free fatty acid [35]. However, 80–90% of dietary glycerides retain their fatty acid in the sn-2 position during the entire digestion and absorption process [33].
Long-chain triacylglycerols and diacylglycerols are insoluble in aqueous solution regardless of whether bile acids are present [3]. Therefore, most of these acylglycerols reside in emulsified oil phase. In contrast, monoacylglycerols and free fatty acids possess polar groups that make them highly soluble in the presence of conjugated bile acids to form mixed micelles. The mixed micelles effectively pass through the unstirred water layer overlying the microvillus border of the enterocytes, and monoacylglycerols and free fatty acids are finally taken up by the cells [24].
The mechanisms by which free fatty acids and monoacylglycerols are translocated into the enterocytes have not been elucidated completely. Cluster determinant 36 (CD36) or fatty acid translocase (FAT) is known to be a membrane protein that facilitates cellular uptake of long-chain fatty acids. This protein is also highly expressed on the luminal surface of enterocytes in the proximal small intestine [36, 37]. However, CD36-null mice exhibited normal overall absorption of long-chain fatty acids and impaired chylomicron secretion. These findings suggest that CD36 plays critical roles for the absorption of long-chain fatty acids and the formation of chylomicron in the proximal small intestine, but CD36-independent absorption mechanisms predominate in the distal segments [37]. In comparison to free fatty acids, studies on intestinal uptake of monoacylglycerols are limited. An in vitro study using human intestinal Caco-2 cells showed that long-chain fatty acid and 2-monoacylglycerol were taken up in a saturable and competitive manner. The results suggest that long-chain fatty acids and 2-monoacylglycerol are transported into the enterocyte, at least in part, via a protein-mediated pathway that is shared by both lipids [38].
3.1.2 Medium-Chain Triacylglycerols
Higher concentrations of medium-chain length fatty acids are found in coconut oil (14%) and palm kernel oil (7%), butter (3%), and fresh cream (2%); cow and breast milk fat (1–3%) also contain significant amounts of the fatty acids [39]. However, the ingestion of medium-chain fatty acids is reported to be less than 2% of the total fatty acid intake in the United States [40].
Gastric lipase and pancreatic triacylglycerol lipase work more efficiently with medium-chain triacylglycerols than long-chain triacylglycerols. As a consequence, medium-chain triacylglycerols are absorbed mainly as free fatty acids and glycerol and only rarely as mono- or diacylglycerols [41]. Because of their smaller molecular size, medium-chain fatty acids and glycerol have greater solubility in water, and micellization with bile acids is unnecessary. In contrast to long-chain fatty acids that are resynthesized to triacylglycerol in the enterocytes and follow the lymphatic system as chylomicrons, medium-chain fatty acids are bound with albumin and follow the portal venous system [41].
3.2 Phospholipids
Dietary phospholipids are not hydrolyzed by gastric lipase but aid the emulsification of dietary fat. Therefore, they are forwarded to the duodenum as a component of emulsified oil droplets. In contrast, biliary phospholipids (essentially phosphatidylcholine) are supplied in mixed micelles along with cholesterol and conjugated bile acids. In the upper small intestine, dietary phospholipids are redistributed much in favor of the micellar phase [42].
The digestion of phospholipids is carried out mainly by pancreatic phospholipase A2, but carboxyl ester lipase may contribute to the hydrolysis of phosphatidylcholine and lysophosphatidylcholine to some extent [22]. In fact, mice deficient in phospholipase A2 show no abnormality in dietary phospholipid absorption [43]. Phospholipase A2 undergoes a substantial increase in the catalytic activity on binding to the surface of phospholipid membranes or micelles [44] and the presence of bile acids [45]. However, the enzyme shows a low activity on biliary phosphatidylcholine because the high bile acid/phosphatidylcholine molar ratio in native bile presents unfavorable conditions for hydrolysis [46]. Phospholipase A2 preferentially cleaves the ester bond in the sn-2 position of the phospholipids to yield lysophosphatidylcholine and free fatty acid [47].
Deacylation of lysophosphatidylcholine in the gut lumen is believed to be quite limited, and lysophosphatidylcholine and free fatty acids are taken up by enterocytes and resynthesized to phospholipids or triacylglycerols, which follow the lymphatic system as chylomicrons. The remaining absorbed lysophosphatidylcholine is hydrolyzed to form glycero-3-phosphorylcholine by phospholipase A2/lysophospholipase (phospholipase B) [48,49,50], which is readily transported via the portal blood for use in the liver [51]. Although specific intestinal transporters for phosphatidylcholine and lysophosphatidylcholine have not been identified, lysophosphatidylcholine uptake by enterocytes is much greater than phosphatidylcholine absorption [52, 53].
3.3 Cholesterol and Plant Sterols
Most dietary cholesterol is present in the free form, but 10–15% exists as cholesterol ester [16]. Gastric lipase does not hydrolyze cholesterol ester; rather, the hydrolysis is performed by pancreatic carboxyl ester lipase (cholesterol esterase). In this process, bile acids strongly stimulate the lipase activity [54, 55]. Chemical modification studies suggest that positive-charged arginine residues in carboxyl ester lipase are important for its interaction with bile acids [56,57,58,59, 22]. In contrast, biliary cholesterol is exclusively free form and is secreted as mixed micelles with phosphatidylcholine and conjugated bile acids. In the proximal small intestine, dietary cholesterol is initially emulsified with triglycerides in oil droplets, but free cholesterol originated from the diet is finally incorporated into mixed micelles or vesicles with biliary cholesterol [60].
In humans, cholesterol absorption is not complete, and the percent of absorption varies from 15% to 75% [61]. Relative to monoacylglycerols, free fatty acids, and lysophosphatidylcholine, the aqueous solubility of cholesterol is extremely low. Therefore, the formation of mixed micelles and vesicles is critically important for the transport of cholesterol through the unstirred water layer overlying the microvillus border of enterocytes. Therefore, intestinal cholesterol absorption is markedly affected by coexisting bile acids, phospholipids, free fatty acids, and plant sterols.
It has been reported that trihydroxy bile acids more effectively promote cholesterol absorption than dihydroxy bile acids [62,63,64], and the size of the cholic acid pool significantly correlates with cholesterol absorption in patients with liver cirrhosis [65]. On the other hand, the intestinal uptake of cholesterol was linearly dependent on micellar cholesterol concentration and was not dependent on the bile acid concentration [64]. However, taurochenodeoxycholic acid is a better micellar solubilizer of cholesterol than taurocholic acid, although the latter is a better promoter of cholesterol absorption [66,67,68]. In addition, when cholesterol was completely solubilized in micelles with a nontoxic nonionic detergent, Pluronic F68, cholesterol was not taken up by enterocytes [64]. These results suggest that not only the solubilization capacity but also the interaction between micelle and acceptor (transporter) serves as determinants of the absorption efficiency of cholesterol.
There are at least four transporters that are key players in the control of cholesterol absorption from the intestine. Niemann-Pick C1-like 1 (NPC1L1) is a major cholesterol uptake transporter [69], while scavenger receptor class B type I (SR-BI) also plays a role in cholesterol uptake to a lesser extent [70, 71]. On the other hand, ATP-binding cassette (ABC) proteins ABCG5 and ABCG8 are cholesterol efflux transporters [72]. Although little is known about the direct effects of bile acids on intestinal NPC1L1 and SR-BI activities, ABCG5-/ABCG8-specific cholesterol efflux is stimulated by bile acids in cell models [73, 74]. ABCG5/ABCG8 transfers cholesterol in an ATP-dependent manner, and the hydrolysis of ATP is stimulated by bile acids [75]. It has been suggested that bile acids may promote an active conformation of ABCG5/ABCG8 either by global stabilization of the transporter or by binding to a specific site on ABCG5/ABCG8. Furthermore, CD36 may also play a role in cholesterol uptake. Overexpression of CD36 enhanced cholesterol uptake from micellar substrates in COS-7 cells [76]. Conversely, CD36-null mice showed significant reduced cholesterol transport from the intestinal lumen to the lymphatic system [77]. However, its absence was not sufficient to cause an overall reduction in intestinal cholesterol uptake.
Although phospholipids are essential molecules for the effective solubilization of cholesterol in the bile and intestine, excess phospholipids cause the suppression of cholesterol absorption. There are at least three possible mechanisms [15]. First, excess phospholipids may interfere with efficient hydrolysis of micellar phospholipids, which is a prerequisite for efficient mucosal uptake of cholesterol. Second, surplus phospholipids may alter the physicochemical properties of mixed micelles resulting in reduced absorption of cholesterol. Third, phospholipids may act on the membrane characteristics of enterocytes or have a direct effect on cellular cholesterol transporters. Free fatty acids may also affect intestinal cholesterol absorption. Mixed micelles containing medium-chain fatty acids have a reduced solubilizing capacity for cholesterol relative to those containing long-chain fatty acids [78].
Food of a plant origin includes plant sterols that are structurally related to cholesterol but differ from cholesterol only in their unsaturation level and/or side-chain configuration [76]. Typical Western diets contain approximately 300 mg of plant sterols per day [79, 80], but the absorption percentage of plant sterols is less than 2% in humans [81], which is considerably lower than that of cholesterol (15–75%). Plant sterols transported into enterocytes with cholesterol via NPC1L1 are pumped back to the lumen via ABCG5/ABCG8, whereas a significant proportion of the internalized cholesterol is esterified and incorporated into chylomicrons [82, 83]. Plant sterols are known to inhibit cholesterol absorption, but the mechanisms are not fully understood. Because the digestion process of plant sterols and cholesterol is virtually the same, it has been suggested that plant sterols compete with intestinal cholesterol for incorporation into mixed micelles [84, 85], but other possible mechanisms have also been proposed [86].
3.4 Fat-Soluble Vitamins
Because vitamins A, D, E, and K are fat soluble, micelle formation is required for intestinal absorption. Vitamins A, D, and E have hydroxyl groups that can be esterified with fatty acid, and pancreatic carboxyl ester lipase catalyzes the hydrolysis under the presence of bile acids [35]. While most dietary vitamin E (tocopherol and tocotrienol) is in the free form, vitamin A (retinol) is often esterified and must be hydrolyzed to retinol and fatty acid before absorption [22, 87]. Except for carboxyl ester lipase, pancreatic triacylglycerol lipase and intestinal phospholipase B also contribute to the hydrolysis of retinyl esters [22]. Free retinol is then incorporated with other lipids into the mixed micelles, passes through the unstirred water layer, and is taken up by enterocytes [87]. Although the intestinal retinol-specific transporter has not been clarified, stimulated by retinoic acid 6 (STRA6) [88] and retinol-binding protein 4-receptor 2 (RBPR2) [89] are candidate proteins.
Micelle formation is also required for the absorption of vitamins E, D, and K [12, 90]. It has been reported that intestinal cholesterol transporters, NPC1L1 and SR-BI, play a role in the uptake of micellar vitamin E [91, 92]. However, recent report suggests that additional intestinal transporters are also involved in the uptake of vitamin E [93]. In addition, there are reports that both SR-BI and CD36 contribute to the intestinal absorption of vitamins D [94] and K [95].
4 Protein Digestion
In addition to promoting lipid digestion and absorption, conjugated bile acids also bind to dietary proteins in the small intestine. The binding of bile acids denatures the protein and dramatically enhances the proteolysis by pancreatic proteases [96]. The effect was most pronounced in the presence of dihydroxy bile acids and was observed at concentrations below the critical micellar concentration.
5 Absorption of Polyvalent Metals
Polyvalent metals such as calcium (Ca2+) and iron (Fe2+) are poorly soluble at the intestinal pH. However, premicellar concentrations of taurocholic acid solubilize calcium [97] and iron [98, 99] in the proximal small intestine and promote their absorption. The mechanism of solubilization is explained by high-affinity binding of these polyvalent cations to the interposition between terminal carboxyl and 7- or 12-hydroxyl groups of the steroid ring of taurocholic acid. Taurodehydrocholic acid, lacking ring hydroxyl groups, did not bind either cation with a high affinity and did not promote their absorption [100].
6 Conclusions
In the duodenum and upper jejunum, the conjugated bile acids facilitate lipolysis by pancreatic lipases and formation of mixed micelles with phospholipids, lipolytic products of triacylglycerols (2-monoacylglycerols and free fatty acids), cholesterol, and fat-soluble vitamins. The mixed micelles can effectively approach the enterocytes, and each nutrient is finally taken up by the cells primarily by protein-mediated processes. Conjugated bile acids also promote digestion of proteins and absorption of polyvalent metals, such as calcium and iron. In addition to these direct effects on digestion and absorption, duodenal conjugated bile acids are known to inhibit the release of cholecystokinin [101] and motilin [102], which modulate contractions of the gallbladder and indirectly control digestion and absorption. Conjugated bile acids maintain water solubility at an acidic pH in the upper small intestine and are not absorbed together with the solubilized lipids, which allows for efficient active absorption of bile acids from the terminal ileum after the completion of their roles.
References
Schaap FG, Trauner M, Jansen PL. Bile acid receptors as targets for drug development. Nat Rev Gastroenterol Hepatol. 2014;11:55–67. doi:10.1038/nrgastro.2013.151.
Hofmann AF. The function of bile salts in fat absorption. The solvent properties of dilute micellar solutions of conjugated bile salts. Biochem J. 1963;89:57–68.
Hofmann AF. A physicochemical approach to the intraluminal phase of fat absorption. Gastroenterology. 1966;50:56–64.
Friedman HI, Nylund B. Intestinal fat digestion, absorption, and transport. A review. Am J Clin Nutr. 1980;33:1108–39.
Carey MC, Small DM, Bliss CM. Lipid digestion and absorption. Annu Rev Physiol. 1983;45:651–77. doi:10.1146/annurev.ph.45.030183.003251.
Hofmann AF, Hagey LR. Bile acids: chemistry, pathochemistry, biology, pathobiology, and therapeutics. Cell Mol Life Sci. 2008;65:2461–83. doi:10.1007/s00018-008-7568-6.
Carey MC, Small DM. Micelle formation by bile salts. Physical-chemical and thermodynamic considerations. Arch Intern Med. 1972;130:506–27.
Matoba N, Une M, Hoshita T. Identification of unconjugated bile acids in human bile. J Lipid Res. 1986;27:1154–62.
Hofmann AF, Mysels KJ. Bile acid solubility and precipitation in vitro and in vivo: the role of conjugation, pH, and Ca2+ ions. J Lipid Res. 1992;33:617–26.
Ovesen L, Bendtsen F, Tage-Jensen U, Pedersen NT, Gram BR, Rune SJ. Intraluminal pH in the stomach, duodenum, and proximal jejunum in normal subjects and patients with exocrine pancreatic insufficiency. Gastroenterology. 1986;90:958–62.
Hofmann AF, Roda A. Physicochemical properties of bile acids and their relationship to biological properties: an overview of the problem. J Lipid Res. 1984;25:1477–89.
Setchell KD, Heubi JE, Shah S, Lavine JE, Suskind D, Al-Edreesi M, et al. Genetic defects in bile acid conjugation cause fat-soluble vitamin deficiency. Gastroenterology. 2013;144:945–55 e6. quiz e14-5 doi:10.1053/j.gastro.2013.02.004.
National Health and Nutrition Survey Japan 2013 Ministry of Health, Labour and Welfare, Japan. http://www.mhlw.go.jp/stf/houdou/0000067890.html/. Accessed 15 Dec 2015.
Freedman MR, King J, Kennedy E. Popular diets: a scientific review. Obes Res. 2001;9(Suppl 1):1S–40S. doi:10.1038/oby.2001.113.
Cohn JS, Kamili A, Wat E, Chung RW, Tandy S. Dietary phospholipids and intestinal cholesterol absorption. Nutrients. 2010;2:116–27. doi:10.3390/nu2020116.
Phan CT, Tso P. Intestinal lipid absorption and transport. Front Biosci. 2001;6:D299–319.
Iqbal J, Hussain MM. Intestinal lipid absorption. Am J Physiol Endocrinol Metab. 2009;296:E1183–94. doi:10.1152/ajpendo.90899.2008.
Verger R, Aoubala M, Carriere F, Ransac S, Dupuis L, De Caro J, et al. Regulation of lumen fat digestion: enzymic aspects. Proc Nutr Soc. 1996;55:5–18.
Hamosh M, Scanlon JW, Ganot D, Likel M, Scanlon KB, Hamosh P. Fat digestion in the newborn. Characterization of lipase in gastric aspirates of premature and term infants. J Clin Invest. 1981;67:838–46.
Abrams CK, Hamosh M, Lee TC, Ansher AF, Collen MJ, Lewis JH, et al. Gastric lipase: localization in the human stomach. Gastroenterology. 1988;95:1460–4.
Borgström B, Erlanson C. Pancreatic lipase and co-lipase. Interactions and effects of bile salts and other detergents. Eur J Biochem. 1973;37:60–8.
Hui DY, Howles PN. Carboxyl ester lipase: structure-function relationship and physiological role in lipoprotein metabolism and atherosclerosis. J Lipid Res. 2002;43:2017–30.
Ganguly J. Biochemistry of vitamin A. Boca Raton: CRC Press, Inc.; 1989.
Thomson AB, Schoeller C, Keelan M, Smith L, Clandinin MT. Lipid absorption: passing through the unstirred layers, brush-border membrane, and beyond. Can J Physiol Pharmacol. 1993;71:531–55.
Porter HP, Saunders DR, Tytgat G, Brunser O, Rubin CE. Fat absorption in bile fistula man. A morphological and biochemical study. Gastroenterology. 1971;60:1008–19.
Mansbach 2nd CM, Newton D, Stevens RD. Fat digestion in patients with bile acid malabsorption but minimal steatorrhea. Dig Dis Sci. 1980;25:353–62.
Scheig R. What is dietary fat? Am J Clin Nutr. 1969;22:651–3.
Liao TH, Hamosh P, Hamosh M. Fat digestion by lingual lipase: mechanism of lipolysis in the stomach and upper small intestine. Pediatr Res. 1984;18:402–9.
Paltauf F, Esfandi F, Holasek A. Stereospecificity of lipases. Enzymic hydrolysis of enantiomeric alkyl diacylglycerols by lipoprotein lipase, lingual lipase and pancreatic lipase. FEBS Lett. 1974;40:119–23.
Cohen M, Morgan RG, Hofmann AF. Lipolytic activity of human gastric and duodenal juice against medium and long chain triglycerides. Gastroenterology. 1971;60:1–15.
Carriere F, Barrowman JA, Verger R, Laugier R. Secretion and contribution to lipolysis of gastric and pancreatic lipases during a test meal in humans. Gastroenterology. 1993;105:876–88.
Gargouri Y, Julien R, Bois AG, Verger R, Sarda L. Studies on the detergent inhibition of pancreatic lipase activity. J Lipid Res. 1983;24:1336–42.
Borgström B. Influence of bile salt, pH, and time on the action of pancreatic lipase; physiological implications. J Lipid Res. 1964;5:522–31.
Hofmann AF, Borgstrom B. Hydrolysis of long-chain monoglycerides in micellar solution by pancreatic lipase. Biochim Biophys Acta. 1963;70:317–31.
Lombardo D, Guy O. Studies on the substrate specificity of a carboxyl ester hydrolase from human pancreatic juice. II. Action on cholesterol esters and lipid-soluble vitamin esters. Biochim Biophys Acta. 1980;611:147–55.
Chen M, Yang Y, Braunstein E, Georgeson KE, Harmon CM. Gut expression and regulation of FAT/CD36: possible role in fatty acid transport in rat enterocytes. Am J Physiol Endocrinol Metab. 2001;281:E916–23.
Nassir F, Wilson B, Han X, Gross RW, Abumrad NA. CD36 is important for fatty acid and cholesterol uptake by the proximal but not distal intestine. J Biol Chem. 2007;282:19493–501. doi:10.1074/jbc.M703330200.
Murota K, Storch J. Uptake of micellar long-chain fatty acid and sn-2-monoacylglycerol into human intestinal Caco-2 cells exhibits characteristics of protein-mediated transport. J Nutr. 2005;135:1626–30.
Takeuchi H, Sekine S, Kojima K, Aoyama T. The application of medium-chain fatty acids: edible oil with a suppressing effect on body fat accumulation. Asia Pac J Clin Nutr. 2008;17(Suppl 1):320–3.
U.S. Department of Agriculture ARS. Nutrient intakes from foods: mean amounts consumed per individual, one day, 2011–2012 http://www.ars.usda.gov/Services/docs.htm?docid=18349 Accessed 15 Dec 2015.
Bach AC, Babayan VK. Medium-chain triglycerides: an update. Am J Clin Nutr. 1982;36:950–62.
Arnesjo B, Nilsson A, Barrowman J, Borgstrom B. Intestinal digestion and absorption of cholesterol and lecithin in the human. Intubation studies with a fat-soluble reference substance. Scand J Gastroenterol. 1969;4:653–65.
Richmond BL, Boileau AC, Zheng S, Huggins KW, Granholm NA, Tso P, et al. Compensatory phospholipid digestion is required for cholesterol absorption in pancreatic phospholipase A2-deficient mice. Gastroenterology. 2001;120:1193–202. doi:10.1053/gast.2001.23254.
Tatulian SA. Toward understanding interfacial activation of secretory phospholipase A2 (PLA2): membrane surface properties and membrane-induced structural changes in the enzyme contribute synergistically to PLA2 activation. Biophys J. 2001;80:789–800. doi:10.1016/S0006-3495(01)76058-4.
Arnesjo B, Grubb A. The activation, purification and properties of rat pancreatic juice phospholipase A 2. Acta Chem Scand. 1971;25:577–89.
Nalbone G, Lairon D, Charbonnier-Augeire M, Vigne JL, Leonardi J, Chabert C, et al. Pancreatic phospholipase A2 hydrolysis of phosphatidylcholines in various physicochemical states. Biochim Biophys Acta. 1980;620:612–25.
van den Bosch H, Postema NM, de Haas GH, van Deenen LL. On the positional specificity of phospholipase A from pancreas. Biochim Biophys Acta. 1965;98:657–9.
Ottolenghi A. Estimation and subcellular distribution of lecithinase activity in rat intestinal mucosa. J Lipid Res. 1964;5:532–7.
Subbaiah PV, Ganguly J. Studies on the phospholipases of rat intestinal mucosa. Biochem J. 1970;118:233–9.
Gassama-Diagne A, Fauvel J, Chap H. Purification of a new, calcium-independent, high molecular weight phospholipase A2/lysophospholipase (phospholipase B) from guinea pig intestinal brush-border membrane. J Biol Chem. 1989;264:9470–5.
Le Kim D, Betzing H. Intestinal absorption of polyunsaturated phosphatidylcholine in the rat. Hoppe Seylers Z Physiol Chem. 1976;357:1321–31.
Reynier MO, Lafont H, Crotte C, Sauve P, Gerolami A. Intestinal cholesterol uptake: comparison between mixed micelles containing lecithin or lysolecithin. Lipids. 1985;20:145–50.
Homan R, Hamelehle KL. Phospholipase A2 relieves phosphatidylcholine inhibition of micellar cholesterol absorption and transport by human intestinal cell line Caco-2. J Lipid Res. 1998;39:1197–209.
Erlanson C. Purification, properties, and substrate specificity of a carboxylesterase in pancreatic juice. Scand J Gastroenterol. 1975;10:401–8.
Blackberg L, Lombardo D, Hernell O, Guy O, Olivecrona T. Bile salt-stimulated lipase in human milk and carboxyl ester hydrolase in pancreatic juice: are they identical enzymes? FEBS Lett. 1981;136:284–8.
Lombardo D, Campese D, Multigner L, Lafont H, De Caro A. On the probable involvement of arginine residues in the bile-salt-binding site of human pancreatic carboxylic ester hydrolase. Eur J Biochem. 1983;133:327–33.
Blackberg L, Hernell O. Bile salt-stimulated lipase in human milk. Evidence that bile salt induces lipid binding and activation via binding to different sites. FEBS Lett. 1993;323:207–10.
Gjellesvik DR, Lorens JB, Male R. Pancreatic carboxylester lipase from Atlantic salmon (Salmo salar). cDNA sequence and computer-assisted modelling of tertiary structure. Eur J Biochem. 1994;226:603–12.
Liang Y, Medhekar R, Brockman HL, Quinn DM, Hui DY. Importance of arginines 63 and 423 in modulating the bile salt-dependent and bile salt-independent hydrolytic activities of rat carboxyl ester lipase. J Biol Chem. 2000;275:24040–6. doi:10.1074/jbc.M003187200.
Yao L, Heubi JE, Buckley DD, Fierra H, Setchell KD, Granholm NA, et al. Separation of micelles and vesicles within lumenal aspirates from healthy humans: solubilization of cholesterol after a meal. J Lipid Res. 2002;43:654–60.
Grundy SM. Absorption and metabolism of dietary cholesterol. Annu Rev Nutr. 1983;3:71–96. doi:10.1146/annurev.nu.03.070183.000443.
Swell L, Flick DF, Field Jr H, Treadwell CR. Influence of dietary bile salts on blood cholesterol levels. Proc Soc Exp Biol Med. 1953;84:428–31.
Gallo-Torres HE, Miller ON, Hamilton JG. Further studies on the role of bile salts in cholesterol esterification and absorption from the gut. Arch Biochem Biophys. 1971;143:22–36.
Watt SM, Simmonds WJ. The specificity of bile salts in the intestinal absorption of micellar cholesterol in the rat. Clin Exp Pharmacol Physiol. 1976;3:305–22.
Ponz de Leon M, Loria P, Iori R, Carulli N. Cholesterol absorption in cirrhosis: the role of total and individual bile acid pool size. Gastroenterology. 1981;80:1428–37.
Cohen BI, Raicht RF, Mosbach EH. Sterol metabolism studies in the rat. Effects of primary bile acids (sodium taurochenodeoxycholate and sodium taurocholate) on sterol metabolism. J Lipid Res. 1977;18:223–31.
Reynier MO, Montet JC, Gerolami A, Marteau C, Crotte C, Montet AM, et al. Comparative effects of cholic, chenodeoxycholic, and ursodeoxycholic acids on micellar solubilization and intestinal absorption of cholesterol. J Lipid Res. 1981;22:467–73.
Watt SM, Simmonds WJ. Effects of four taurine-conjugated bile acids on mucosal uptake and lymphatic absorption of cholesterol in the rat. J Lipid Res. 1984;25:448–55.
Altmann SW, Davis Jr HR, Zhu LJ, Yao X, Hoos LM, Tetzloff G, et al. Niemann-Pick C1 like 1 protein is critical for intestinal cholesterol absorption. Science. 2004;303:1201–4. doi:10.1126/science.1093131.
Altmann SW, Davis Jr HR, Yao X, Laverty M, Compton DS, Zhu LJ, et al. The identification of intestinal scavenger receptor class B, type I (SR-BI) by expression cloning and its role in cholesterol absorption. Biochim Biophys Acta. 2002;1580:77–93.
Bietrix F, Yan D, Nauze M, Rolland C, Bertrand-Michel J, Comera C, et al. Accelerated lipid absorption in mice overexpressing intestinal SR-BI. J Biol Chem. 2006;281:7214–9. doi:10.1074/jbc.M508868200.
Lee MH, Lu K, Hazard S, Yu H, Shulenin S, Hidaka H, et al. Identification of a gene, ABCG5, important in the regulation of dietary cholesterol absorption. Nat Genet. 2001;27:79–83. doi:10.1038/83799.
Vrins C, Vink E, Vandenberghe KE, Frijters R, Seppen J, Groen AK. The sterol transporting heterodimer ABCG5/ABCG8 requires bile salts to mediate cholesterol efflux. FEBS Lett. 2007;581:4616–20. doi:10.1016/j.febslet.2007.08.052.
Tachibana S, Hirano M, Hirata T, Matsuo M, Ikeda I, Ueda K, et al. Cholesterol and plant sterol efflux from cultured intestinal epithelial cells is mediated by ATP-binding cassette transporters. Biosci Biotechnol Biochem. 2007;71:1886–95.
Johnson BJ, Lee JY, Pickert A, Urbatsch IL. Bile acids stimulate ATP hydrolysis in the purified cholesterol transporter ABCG5/G8. Biochemistry. 2010;49:3403–11. doi:10.1021/bi902064g.
van Bennekum A, Werder M, Thuahnai ST, Han CH, Duong P, Williams DL, et al. Class B scavenger receptor-mediated intestinal absorption of dietary beta-carotene and cholesterol. Biochemistry. 2005;44:4517–25. doi:10.1021/bi0484320.
Nauli AM, Nassir F, Zheng S, Yang Q, Lo CM, Vonlehmden SB, et al. CD36 is important for chylomicron formation and secretion and may mediate cholesterol uptake in the proximal intestine. Gastroenterology. 2006;131:1197–207. doi:10.1053/j.gastro.2006.08.012.
von Bonsdorff-Nikander A, Christiansen L, Huikko L, Lampi AM, Piironen V, Yliruusi J, et al. A comparison of the effect of medium- vs. long-chain triglycerides on the in vitro solubilization of cholesterol and/or phytosterol into mixed micelles. Lipids. 2005;40:181–90.
Klingberg S, Andersson H, Mulligan A, Bhaniani A, Welch A, Bingham S, et al. Food sources of plant sterols in the EPIC Norfolk population. Eur J Clin Nutr. 2008;62:695–703. doi:10.1038/sj.ejcn.1602765.
Valsta LM, Lemstrom A, Ovaskainen ML, Lampi AM, Toivo J, Korhonen T, et al. Estimation of plant sterol and cholesterol intake in Finland: quality of new values and their effect on intake. Br J Nutr. 2004;92:671–8.
Ostlund Jr RE, McGill JB, Zeng CM, Covey DF, Stearns J, Stenson WF, et al. Gastrointestinal absorption and plasma kinetics of soy Delta(5)-phytosterols and phytostanols in humans. Am J Physiol Endocrinol Metab. 2002;282:E911–6. doi:10.1152/ajpendo.00328.2001.
Calandra S, Tarugi P, Speedy HE, Dean AF, Bertolini S, Shoulders CC. Mechanisms and genetic determinants regulating sterol absorption, circulating LDL levels, and sterol elimination: implications for classification and disease risk. J Lipid Res. 2011;52:1885–926. doi:10.1194/jlr.R017855.
Gylling H, Plat J, Turley S, Ginsberg HN, Ellegard L, Jessup W, et al. Plant sterols and plant stanols in the management of dyslipidaemia and prevention of cardiovascular disease. Atherosclerosis. 2014;232:346–60. doi:10.1016/j.atherosclerosis.2013.11.043.
Nissinen M, Gylling H, Vuoristo M, Miettinen TA. Micellar distribution of cholesterol and phytosterols after duodenal plant stanol ester infusion. Am J Physiol Gastrointest Liver Physiol. 2002;282:G1009–15. doi:10.1152/ajpgi.00446.2001.
Ikeda I. Factors affecting intestinal absorption of cholesterol and plant sterols and stanols. J Oleo Sci. 2015;64:9–18. doi:10.5650/jos.ess14221.
De Smet E, Mensink RP, Plat J. Effects of plant sterols and stanols on intestinal cholesterol metabolism: suggested mechanisms from past to present. Mol Nutr Food Res. 2012;56:1058–72. doi:10.1002/mnfr.201100722.
Reboul E. Absorption of vitamin A and carotenoids by the enterocyte: focus on transport proteins. Nutrients. 2013;5:3563–81. doi:10.3390/nu5093563.
Isken A, Golczak M, Oberhauser V, Hunzelmann S, Driever W, Imanishi Y, et al. RBP4 disrupts vitamin A uptake homeostasis in a STRA6-deficient animal model for Matthew-Wood syndrome. Cell Metab. 2008;7:258–68. doi:10.1016/j.cmet.2008.01.009.
Alapatt P, Guo F, Komanetsky SM, Wang S, Cai J, Sargsyan A, et al. Liver retinol transporter and receptor for serum retinol-binding protein (RBP4). J Biol Chem. 2013;288:1250–65. doi:10.1074/jbc.M112.369132.
Goncalves A, Gontero B, Nowicki M, Margier M, Masset G, Amiot MJ, et al. Micellar lipid composition affects micelle interaction with class B scavenger receptor extracellular loops. J Lipid Res. 2015;56:1123–33. doi:10.1194/jlr.M057612.
Reboul E, Klein A, Bietrix F, Gleize B, Malezet-Desmoulins C, Schneider M, et al. Scavenger receptor class B type I (SR-BI) is involved in vitamin E transport across the enterocyte. J Biol Chem. 2006;281:4739–45. doi:10.1074/jbc.M509042200.
Narushima K, Takada T, Yamanashi Y, Suzuki H. Niemann-pick C1-like 1 mediates alpha-tocopherol transport. Mol Pharmacol. 2008;74:42–9. doi:10.1124/mol.107.043034.
Reboul E, Soayfane Z, Goncalves A, Cantiello M, Bott R, Nauze M, et al. Respective contributions of intestinal Niemann-Pick C1-like 1 and scavenger receptor class B type I to cholesterol and tocopherol uptake: in vivo v. in vitro studies. Br J Nutr. 2012;107:1296–304. doi:10.1017/S0007114511004405.
Reboul E, Borel P. Proteins involved in uptake, intracellular transport and basolateral secretion of fat-soluble vitamins and carotenoids by mammalian enterocytes. Prog Lipid Res. 2011;50:388–402. doi:10.1016/j.plipres.2011.07.001.
Goncalves A, Margier M, Roi S, Collet X, Niot I, Goupy P, et al. Intestinal scavenger receptors are involved in vitamin K1 absorption. J Biol Chem. 2014;289:30743–52. doi:10.1074/jbc.M114.587659.
Gass J, Vora H, Hofmann AF, Gray GM, Khosla C. Enhancement of dietary protein digestion by conjugated bile acids. Gastroenterology. 2007;133:16–23. doi:10.1053/j.gastro.2007.04.008.
Sanyal AJ, Hirsch JI, Moore EW. Premicellar taurocholate enhances calcium uptake from all regions of rat small intestine. Gastroenterology. 1994;106:866–74.
Sanyal AJ, Hirsch JI, Moore EW. Premicellar taurocholate avidly binds ferrous (Fe++) iron: a potential physiologic role for bile salts in iron absorption. J Lab Clin Med. 1990;116:76–86.
Sanyal AJ, Shiffmann ML, Hirsch JI, Moore EW. Premicellar taurocholate enhances ferrous iron uptake from all regions of rat small intestine. Gastroenterology. 1991;101:382–9.
Sanyal AJ, Hirsch JI, Moore EW. High-affinity binding is essential for enhancement of intestinal Fe2+ and Ca2+ uptake by bile salts. Gastroenterology. 1992;102:1997–2005.
Koop I, Schindler M, Bosshammer A, Scheibner J, Stange E, Koop H. Physiological control of cholecystokinin release and pancreatic enzyme secretion by intraduodenal bile acids. Gut. 1996;39:661–7.
Portincasa P, Peeters TL, van Berge-Henegouwen GP, van Solinge WW, Palasciano G, van Erpecum KJ. Acute intraduodenal bile salt depletion leads to strong gallbladder contraction, altered antroduodenal motility and high plasma motilin levels in humans. Neurogastroenterol Motil. 2000;12:421–30.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer Japan KK
About this chapter
Cite this chapter
Honda, A., Ikegami, T., Matsuzaki, Y. (2017). Intestinal Digestion and Absorption. In: Tazuma, S., Takikawa, H. (eds) Bile Acids in Gastroenterology. Springer, Tokyo. https://doi.org/10.1007/978-4-431-56062-3_3
Download citation
DOI: https://doi.org/10.1007/978-4-431-56062-3_3
Published:
Publisher Name: Springer, Tokyo
Print ISBN: 978-4-431-56060-9
Online ISBN: 978-4-431-56062-3
eBook Packages: MedicineMedicine (R0)