Abstract
The structural maintenance of dendrite arbors and dendritic spines is crucial for proper brain function. Dendrite arbors and dendritic spines transition from dynamic plasticity to greater stability during maturation. Dendrite branches stabilize before dendritic spines to retain overall dendrite arbor integrity while preserving the ability to fine-tune synaptic connections. Nonetheless, shared signaling mechanisms exist to coordinate shaping and stabilization of both dendritic spines and dendrite arbors. Long-term dendrite arbor and dendritic spine stability is maintained by dynamic cytoskeletal elements that turn over quickly. This process is exquisitely controlled by small GTPases, kinases, and proteins that regulate actin and microtubule polymerization, stability, and turnover. These proteins collectively ensure the precise formation and balanced turnover of the actin and microtubule cytoskeletons to maintain dendritic spine and dendrite structure. In addition, cell surface adhesion receptors, including Eph receptors, immunoglobulin superfamily receptors, cadherins, and integrins, play important roles in dendritic spine formation and stability by signaling to cytoplasmic nonreceptor tyrosine kinases that mediate changes in cytoskeletal structure. The loss of dendritic spine and/or dendrite arbor stability in humans is a major contributing factor to normal aging and the pathology of psychiatric illnesses, neurodegenerative diseases, and stroke. In this review, we discuss the cellular and molecular mechanisms that differentially regulate dendritic spine and dendrite arbor stability, examine how these mechanisms interact functionally, and highlight how they become disrupted in different pathological states.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Maintenance
- Dendritic spines
- Small GTPases
- Cytoskeleton
- Actin stability
- Alzheimer’s disease
- Rac1
- BDNF
- CaMKII
1 Introduction
1.1 Dendrite Arbors and Dendritic Spines Transition from Dynamic Plasticity to Greater Stability During Development
The proper formation and long-term maintenance of neuronal connectivity is crucial for proper brain function. The size and shape of a neuron’s dendrite arbor establish the blueprint for its connections by determining the number and distribution of receptive synaptic contacts it can make with afferent axons. Dendrites in developing neurons undergo continual dynamic changes in shape to facilitate proper wiring, synapse formation, and establishment of neural circuits. Growing dendrite arbors continually extend and retract branches as they mature, and this behavior is greatly influenced by synaptic activity. Importantly, only a subset of nascent dendrite branches become stabilized (Dailey and Smith 1996; Wu et al. 1999; Wong and Wong 2000; Wong et al. 2000; Niell et al. 2004). Many observations in diverse model systems have affirmed that afferent innervation or synapse formation on a nascent dendrite branch promotes its stabilization, whereas the loss or reduction of synaptic inputs destabilizes target dendrites (Clark 1957; Matthews and Powell 1962; Jones and Thomas 1962; Wiesel and Hubel 1963; Coleman and Riesen 1968; Vaughn 1989; Rajan et al. 1999; Wu et al. 1999; Niell et al. 2004; Cline and Haas 2008). To explain the observation that dendrite branches that receive synaptic inputs are preferentially stabilized, Vaughn proposed the “synaptotrophic hypothesis,” offering a model by which afferents can influence dendrite arbor development (Vaughn 1989).
The structural plasticity of dendrites decreases greatly as circuits mature. Dendrite branches stabilize first, while dendritic spines continue to form, change shape, and turn over as circuits refine (Trachtenberg et al. 2002; Oray et al. 2004; Holtmaat et al. 2005, 2006; Majewska et al. 2006). Spine formation and pruning is especially dependent on experience and activity patterns during this early stage of development (Trachtenberg et al. 2002; Zuo et al. 2005a; Holtmaat et al. 2006; Yang et al. 2009). This period of spine dynamics is followed by a phase of extensive synapse and dendritic spine pruning, which extends through adolescence and early adulthood in some human brain regions (Huttenlocher 1979, 1990; Rakic et al. 1986; Markus and Petit 1987; Rakic et al. 1994; Holtmaat et al. 2005; Yang et al. 2009; Gourley et al. 2012). Dendritic spine and dendrite branch stability become mechanistically uncoupled during late refinement. Such uncoupling is crucial for long-term circuit stability, as it enables mature neurons to retain overall long-term dendrite arbor integrity and integration within networks while preserving the ability to fine-tune synaptic connections. The dynamic behavior of dendritic spines in the rodent cortex becomes greatly reduced by early adulthood. Transcranial two-photon imaging indicates that a significant fraction (50–85 %) of dendritic spines in adult rodent cortex is stable for extended time periods of several months and possibly years (Trachtenberg et al. 2002; Holtmaat et al. 2005, 2006; Zuo et al. 2005a; Majewska et al. 2006). Together, these findings suggest a scenario in which the majority of dendritic spines and dendrite arbors become stabilized for long periods within an organism’s lifetime, perhaps decades in humans.
The loss of dendritic spine and/or dendrite arbor stability in humans is a major contributing factor to the pathology of psychiatric, neurological, and neurodegenerative disorders, as well as damage associated with stroke and normal aging. Importantly, different patterns of dendritic spine and dendrite branch loss are observed in different psychiatric and neurodegenerative disorders (reviewed in Kulkarni and Firestein 2012), suggesting that spine and branch stabilization mechanisms are differentially disrupted in different disease pathologies. The altered synaptic connectivity resulting from dendrite arbor and dendritic spine destabilization or loss is thought to contribute to the impaired perception, cognition, memory, mood, and decision-making that characterize these pathological conditions. Additionally, despite the evidence discussed for long-term dendritic spine stability during an organism’s lifetime, a recent study has shown that cortical dendritic spines return to a more dynamic state in aged mice (>20 months) (Mostany et al. 2013). It is presently unclear how this increased dynamic spine behavior contributes to age-dependent decrements in cognition, learning, or memory.
Biochemical, imaging, and electrophysiological approaches have been used to dissect the mechanisms that mediate long-term dendritic spine and dendrite branch stability. We provide a perspective on the cellular and molecular mechanisms that differentially regulate dendritic spine versus dendrite branch stability and highlight how these mechanisms are disrupted in different pathological states.
2 Cytoskeletal Dynamics Play a Key Role in the Regulation of Dendrite Arbor and Dendritic Spine Stability
2.1 A Dynamic Actin Framework Supports Dendritic Spine Structure and Function
Dendritic spines are small protrusions from the dendrite branch that serve as postsynaptic contacts at the majority of excitatory synapses (Harris 1999; Tashiro and Yuste 2003; Bourne and Harris 2007; Izeddin et al. 2011). Spines are morphologically heterogeneous (e.g., thin, mushroom, and stubby), yet commonly have a narrow neck and bulbous head (Harris and Kater 1994; Harris 1999; Star et al. 2002; Tashiro and Yuste 2003; Bourne and Harris 2007; Arellano et al. 2007; Tønnesen et al. 2014). Spine shape is supported by a filamentous actin network , which reinforces overall spine structure and stability, organizes the postsynaptic signaling machinery, and powers changes in spine shape (Fifková and Delay 1982; Matus et al. 1982; Fischer et al. 1998; Racz and Weinberg 2006; Hotulainen and Hoogenraad 2010; Izeddin et al. 2011). The actin network consists of both linear and branched filaments, which interact with microtubules at the base of the spine neck and extend to the postsynaptic density (PSD) (Korobova and Svitkina 2010; Izeddin et al. 2011). Additionally, the actin network closely associates with the plasma membrane, and activity-dependent remodeling of the actin network drives changes in spine structure (Izeddin et al. 2011).
Even in stable spines, the actin cytoskeleton undergoes dynamic remodeling (Okamoto et al. 2004). Actin filaments are polarized with their barbed ends located near the membrane; polymerization at the barbed ends contributes to a retrograde flow from the plasma membrane to the spine center with an approximate turnover time in tens of seconds, as measured by both fluorescence recovery after photobleaching and two-photon photoactivation of fluorescent protein-tagged actin (Star et al. 2002; Pollard and Borisy 2003; Okamoto et al. 2004; Honkura et al. 2008). Changes in the polymerization and depolymerization rates or the degree of actin filament branching are believed to mediate the long-lasting changes in spine shape that accompany changes in synaptic efficacy (i.e., the degree to which a presynaptic afferent affects a postsynaptic spine) (Okamoto et al. 2004; Bourne and Harris 2007). For example, long-term potentiation (LTP) induced by tetanic stimulation causes increased actin polymerization and a rapid and selective enlargement of stimulated spine heads that coincides with increased synaptic efficacy (Matsuzaki et al. 2004; Okamoto et al. 2004; Bourne and Harris 2007). In contrast, long-term depression (LTD) induced by prolonged low-frequency stimulation results in actin depolymerization, spine shrinkage, and reduced synaptic efficacy (Fig. 14.1) (Okamoto et al. 2004; Zhou et al. 2004; Bourne and Harris 2007).
Key regulators of the actin and microtubule cytoskeletons. The actin cytoskeleton in dendritic spines is composed of two distinct pools of actin: a dynamic “shell” at the tip and periphery of the spine and a more stable “core” extending from the center of the spine head to the base of the spine neck. Actin-binding proteins (ABPs) that increase actin dynamics, such as cofilin, which severs actin filaments, localize to the spine shell and postsynaptic density (PSD) to support actin remodeling. ABPs that stabilize actin, such as cortactin, localize to the spine core. Cortactin also activates the Arp2/3 complex, which localizes to a toroidal zone between the core and the shell where it nucleates new actin branches. The microtubule cytoskeleton is composed of mixed polarity microtubules that support dendrite arbor structure. Microtubule-associated proteins, including MAP1A and MAP2, form crossbridges between microtubules and mediate their assembly and stabilization. End-binding protein 3 (EB3) localizes to microtubule plus ends, which transiently enter spines. Finally, changes in the actin and microtubule cytoskeleton correlate with alterations in spine structure and synaptic efficacy. In particular, the spine shrinkage and reduced synaptic efficacy observed with long-term depression (LTD) correlate with actin depolymerization, increased actin severing by cofilin, and decreased actin stabilization by cortactin and the Arp2/3 complex. Accordingly, the spine enlargement and increased synaptic efficacy observed with long-term potentiation (LTP) correlate with increased actin polymerization, increased actin nucleation by the Arp2/3 complex, and increased cortactin-mediated actin stability
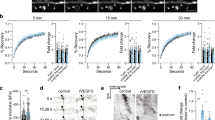
Interestingly, the dynamic behavior of actin is heterogeneous within different regions of the spine (Izeddin et al. 2011). Elegant measurements of the dispersal of photoactivated actin within discrete subspine regions strongly suggest that spines contain at least two distinct pools of actin filaments: a dynamic “shell” and a stable “core” (Fig. 14.1) (Izeddin et al. 2011). The more dynamic shell, consisting of the tip and periphery of the spine, determines spine shape and provides the protrusive force to change shape (Honkura et al. 2008). Not surprisingly, actin in the shell region displays a very fast turnover time (~tens of seconds) (Honkura et al. 2008). In contrast, the spine core, consisting of the central zone of the spine head extending to the base of the spine neck, turns over >20-fold more slowly than the shell (Honkura et al. 2008). Moreover, single actin molecule tracking in dendritic spines using photoactivation localization microscopy has supported this model by revealing that half of the actin molecules are stationary and one-third of the actin molecules undergo a retrograde motion, consistent with spines having a stable actin core and a more dynamic actin shell, respectively (Tatavarty et al. 2009).
The concept of distinct functional pools of actin in spines is reinforced by the observation that specific actin-binding proteins preferentially localize to discrete subdomains within the spine (Rácz and Weinberg 2013). For example, cortactin , which is implicated in actin filament nucleation, branching, and stabilization, localizes to the stable spine core where it helps to maintain actin filament stability (Rácz and Weinberg 2004; Hering and Sheng 2003; Lin et al. 2013). In contrast, cofilin , which severs actin filaments to initiate actin remodeling, localizes to the dynamic spine shell and PSD (Racz and Weinberg 2006). Additionally, the actin-related protein 2/3 (Arp2/3) complex is localized in a toroidal zone between the stable spine core and dynamic spine shell where it specializes in nucleating new actin branches that can extend into the shell from the stable core (Fig. 14.1) (Rácz and Weinberg 2008). Overall, these studies suggest that the distinct pools of actin may have discrete functions in dendritic spines. The stable actin pool allows spines to maintain a constant overall position with respect to the spine core and serves as a scaffold from which the more dynamic pool can elaborate. The dynamic actin pool allows spines to both probe their environment and make activity-dependent size adjustments at the spine periphery. In addition, the dynamic actin pool plays key roles in endocytosis and receptor internalization in the spine, as endocytic sites are enriched for actin regulatory proteins that are necessary for endocytosis (for a recent review, see Mooren et al. 2012).
2.2 Microtubule Arrays Support Dendrite Arbor Stability and Membrane Trafficking
Dendrite arbors in the mature nervous system are supported by dense, parallel arrays of regularly spaced microtubule bundles (Sasaki et al. 1983). These microtubules are required for dendrite arbor growth, dendrite arbor maintenance, and trafficking of cargoes, including organelles (Conde and Caceres 2009), PSD-associated proteins (El-Husseini et al. 2001), neurotransmitter receptors (Setou et al. 2000, 2002), specific mRNAs (Setou et al. 2002; Kanai et al. 2004), and proteins for local synthesis within the dendrite (Hirokawa and Takemura 2005). While axonal microtubules are uniformly polarized with their plus ends oriented away from the soma, dendritic microtubules are generally of mixed polarity to facilitate, in part, bidirectional trafficking of cargoes (Baas et al. 1988; Burton 1988). Nonetheless, microtubule polarization within dendrites is observed both close to the growth cone and in apical dendrites with large diameters, which may reflect the importance of transporting cargoes to the growth cone and away from the soma, respectively (Baas et al. 1988; Kwan et al. 2008).
2.3 Long-Term Dendrite Arbor and Dendritic Spine Stability Is Maintained by Dynamic Cytoskeletal Elements
While individual dendritic spines and dendrite branches can be stable for months to years, the individual cytoskeletal elements underlying these stable structures turn over in minutes to hours in both the developing and mature nervous system. In dendritic spines, more than 80 % of the actin turns over every minute, with the remaining portion undergoing turnover in tens of minutes (Star et al. 2002; Honkura et al. 2008; Tatavarty et al. 2009). Approximately 75 % of dendritic microtubules turn over within tens of minutes, while the rest turns over at a slower rate, likely to be on the order of a few hours, based on measurements for axonal microtubules (Okabe and Hirokawa 1990; Edson et al. 1993).
The structural and functional stability of dendrite branches and dendritic spines would appear to be in direct conflict with the high turnover rates observed for their underlying cytoskeletal elements. The resolution of this apparent paradox arises from the fact that formation and turnover of the actin and microtubule structures is under exquisite control. In particular, a huge collection of proteins is dedicated to the formation, organization, and stabilization of actin in dendritic spines and microtubules in dendrite branches. These regulatory proteins ensure carefully balanced turnover and maintenance of the actin and microtubule cytoskeletons in dendritic spines and dendrite branches, respectively, so that they can serve as scaffolds for their own replenishment and reorganization. A myriad of such proteins, some discussed below, ensure that only a fraction of the cytoskeleton is remodeled at any particular time.
Regulatory proteins that control the actin cytoskeleton in dendritic spines include the actin-stabilizing protein cortactin, the actin-severing protein cofilin, and the Arp2/3 complex. Of particular interest, the Arp2/3 complex localizes to dendritic spines (Rácz and Weinberg 2008) and mediates nucleation and initiation of new actin branches on preexisting filaments in vitro (Pollard and Borisy 2003). Conditional knockout of the Arp2/3 complex in excitatory neurons disrupts the ability of spines to refurbish actin, resulting in gradual spine and synapse loss in cortical and hippocampal neurons (Kim et al. 2013a), highlighting the critical role that the Arp2/3 complex plays in actin stability. In dendrite branches, the microtubule cytoskeleton is stabilized by microtubule-associated proteins (MAPs) , such as MAP1A and MAP2, which localize in high concentrations to dendrite branches where they stabilize existing microtubules, form crossbridges between microtubules, and promote tubulin assembly into microtubules (Fig. 14.1) (Bloom et al. 1984; Huber and Matus 1984; De Camilli et al. 1984; Harada et al. 2002). Activity-dependent dendrite growth, branching, and stabilization depend critically on both MAP1A and MAP2 expression (Vaillant et al. 2002; Szebenyi et al. 2005). Correspondingly, upregulation of both MAPs in cultured neurons strongly correlates with dendrite branch stabilization, whereas knockout of both MAPs causes reduced microtubule density, disrupted microtubule spacing and bundling, and decreased overall dendrite length (Teng et al. 2001; Vaillant et al. 2002; Harada et al. 2002; Szebenyi et al. 2005). Additionally, MAP overexpression protects microtubules from microtubule-severing proteins, such as katanins, which can destabilize dendrite arbors by severing microtubules (Sudo and Baas 2010). In addition to the stabilizing effects of MAPs, microtubules can undergo covalent modifications that affect their stability. For example, detyrosination of the α-tubulin subunit stabilizes microtubules and inhibits their disassembly by decreasing their affinity for the kinesin-13 family of microtubule depolymerases (Peris et al. 2009). Overall, despite the dynamic nature of the individual cytoskeletal elements, the careful balance between stabilization and destabilization of microtubules and actin is essential for the long-term structural stability of dendrite branches and dendritic spines.
3 Actin Cytoskeletal Regulators are Essential for Dendritic Spine Maintenance
As the actin cytoskeleton determines spine structure, actin control machinery is critical for spine stability. Small GTPases, kinases, and actin-binding proteins regulate actin in spines to confer long-term structural stability.
3.1 Small GTPases Exert Differential Effects on Dendritic Spine Stability
Small GTPases , including Ras and the Rho family GTPases RhoA, Rac1, and Cdc42, are central regulators of cytoskeletal dynamics in neurons (Govek et al. 2005) and mediate both initial spine formation and activity-based changes in spine size. Even after initial spine formation, these GTPases continue to regulate spine shape and stability. For example, activation of single synapses by glutamate uncaging activates Ras, RhoA, Rac1, and Cdc42. Selective knockdown experiments reveal that each of these GTPases is critical for persistent spine head enlargement induced by repeated glutamate uncaging at single synapses (Murakoshi et al. 2011). Moreover, expression of dominant negative Rac1 in hippocampal neurons leads to progressive spine loss, indicating that Rac1 activity is required for long-term spine stability (Nakayama and Luo 2000). Rac1 activates the WAVE regulatory complex (WRC) to stimulate the Arp2/3 complex to nucleate new actin filament branches from the side of existing actin filaments (Machesky and Insall 1998; Machesky et al. 1999; Chen et al. 2014). WAVE1 and the Arp2/3 complex both localize to dendritic spines; knockout of the Arp2/3 complex and knockdown or knockout of wave1 cause reductions in dendritic spine density (Kim et al. 2006; Soderling et al. 2007; Kim et al. 2013a). At the ultrastructural level, loss of WAVE1 function leads to abnormalities in the structure of both axon terminals and dendritic spines and greatly alters the normal contacts between them (Hazai et al. 2013). These observations strongly suggest that Rac1 acts through WAVE1 and the Arp2/3 complex to refresh the spinoskeleton core and support long-term spine stability. Rac1 and Cdc42 also act through the PAK3 kinase to activate LIM kinase (LIMK) to phosphorylate and inhibit the actin-severing protein cofilin (Fig. 14.2). Knockdown or genetic inhibition of PAK3 activity converts hippocampal spines to a more immature thin morphology and reduces their stability (Boda et al. 2004; Kreis et al. 2007; Dubos et al. 2012).
Small GTPases are central regulators of actin dynamics. The Rho family GTPases, including Rac1, Cdc42, and RhoA, regulate changes in the actin cytoskeleton. In particular, GTP-bound Rac1 activates the WAVE regulatory complex (WRC) such that WAVE1 can stimulate the Arp2/3 complex and mediate actin nucleation. Additionally, GTP-bound Rac1 and Cdc42 stimulate PAK3 kinase to activate LIM kinase (LIMK), which phosphorylates cofilin and inhibits its actin-severing ability. GTP-bound RhoA activates ROCK, which also stimulates LIMK-based inhibition of cofilin. Finally, GTP-bound Ras GTPase influences actin dynamics through stimulation of T lymphoma invasion and metastasis-inducing protein 1 (Tiam1), which promotes activation of Rac1 by catalyzing the dissociation of GDP and allowing for the binding of GTP to Rac1
Activated RhoA mutants or elevated RhoA activity causes reductions in dendritic spine density (Tashiro et al. 2000; Xing et al. 2012), while either RhoA inhibition or knockdown of the RhoA activator GEF1/Lfc increases spine density (Tashiro et al. 2000; Kang et al. 2009). Inhibition of the major RhoA target Rho-associated kinase (ROCK) can block spine loss resulting from elevated RhoA protein levels or activity (Xing et al. 2012; Lin et al. 2013). These studies suggest that RhoA-ROCK signaling antagonizes the stability of previously formed dendritic spines, although the target of RhoA signaling in regulating spine stability is not clear. Nonetheless, RhoA signaling through ROCK stimulates LIMK to phosphorylate cofilin and inhibit its actin-severing activity (Fig. 14.2) (Arber et al. 1998; Yang et al. 1998), but this might be expected to increase actin stability and promote overall spine stability. However, the effects of RhoA on spine stability may depend on the neuron type, developmental stage, or context. For example, in a number of cases, RhoA activation or loss of a RhoA inhibitory cascade in mature neurons leads to dramatic dendrite arbor shrinkage, but does not impact dendritic spine stability (Nakayama and Luo 2000; Sfakianos et al. 2007; Lin et al. 2013). In fact, repeated activation of individual spines by glutamate uncaging actually leads to increased RhoA activity, which is required for activity-based spine enlargement (Murakoshi et al. 2011). Another study found that localized ROCK inhibition decreased dendritic spine stability (Kang et al. 2009). Ongoing work in the field should resolve what factors determine these differential effects of RhoA-ROCK activation on overall spine stability.
3.2 Actin-Binding Proteins Play Key Roles in Long-Term Dendritic Spine Stabilization
Dendritic spines are enriched with several actin-binding proteins, including Abp1, cortactin, drebrin, and β-adducin, that play important roles in the long-term stability of dendritic spines (Shirao et al. 1988; Matsuoka et al. 1998; Yamazaki et al. 2001; Tanokashira et al. 2012). Abp1 plays a critical role in dendritic spine morphogenesis, particularly in the transition of thin immature dendritic spines to larger more stable mushroom-shaped spines in the hippocampus (Haeckel et al. 2008). Abp1 interacts with the Arp2/3 complex activator N-WASp and the synaptic scaffold protein ProSAP1/Shank2 during this process, presumably to promote formation of branched actin networks that support spine head enlargement and anchor them to the postsynaptic signaling machinery.
Cortactin is an actin-binding protein that stabilizes actin filaments, stimulates actin branch nucleation by the Arp2/3 complex, and stabilizes the resulting branches (MacGrath and Koleske 2012a; Courtemanche et al. 2015). Cortactin is enriched in the spinoskeleton core, where about 50 % of cortactin is largely immobile, as measured by fluorescence recovery after photobleaching (Rácz and Weinberg 2004; Iki et al. 2005). Cortactin knockdown leads to significant reductions in dendritic spine density (Hering and Sheng 2003). Interestingly, excessive NMDA receptor (NMDAR) stimulation or blockade of the tropomyosin receptor kinase B (TrkB) leads to cortactin redistribution from dendritic spines to dendrite shafts (Hering and Sheng 2003; Iki et al. 2005; Seese et al. 2012; Lin et al. 2013). This cortactin redistribution correlates with loss of spine actin content and spine destabilization, strongly suggesting that cortactin is a critical mediator of spine stability. Cortactin also interacts with several proteins with known or suspected roles in long-term spine stabilization, including cortactin-binding protein 2, the PSD scaffolding protein Shank3, p140Cap, and the Abl2/Arg nonreceptor tyrosine kinase (Du et al. 1998; Naisbitt et al. 1999; Jaworski et al. 2009; Chen and Hsueh 2012; Lin et al. 2013; Courtemanche et al. 2015).
DrebrinA , the main drebrin splice isoform expressed in neurons, has complex roles in dendritic spine development, function, and stability. DrebrinA promotes clustering of actin and the postsynaptic scaffolding protein 95 (PSD95) in dendritic filopodia and promotes pairing of these filopodia with presynaptic partners (Takahashi et al. 2003). Knocking down DrebrinA in immature cultured hippocampal neurons has been reported by one group to decrease the proportion of filopodia with PSD95 clusters, which may represent nascent synapses (Takahashi et al. 2003). However, another study demonstrated that DrebrinA knockdown in immature cultured hippocampal neurons at a similar age increased the proportion of mature spines (Biou et al. 2008). The basis for these different findings is at present unclear. Nonetheless, drebrin binding to actin filaments stabilizes them from depolymerization and protects them from cofilin-mediated severing (Mikati et al. 2013; Grintsevich and Reisler 2014), but drebrin also competes with several proteins, including tropomyosin, cofilin, and α-actinin, for binding to actin filaments (Ishikawa et al. 1994; Sasaki et al. 1996; Grintsevich and Reisler 2014). Interestingly, DrebrinA overexpression in established hippocampal neuron cultures selectively dislocates α-actinin from spines. This correlates with an elongation of spine shape and reduced spine stability (Hayashi and Shirao 1999; Biou et al. 2008), likely due to the loss of α-actinin as a key stabilizing link between the actin cytoskeleton and the N-cadherin adhesion receptor (Knudsen et al. 1995). Together, these data indicate that DrebrinA levels must be carefully balanced for it to contribute its critical roles to dendritic spine formation and stability. Interestingly, drebrin levels are significantly decreased not only in patients with Alzheimer’s disease, but also in patients with mild cognitive impairment (Harigaya et al. 1996; Counts et al. 2012). This suggests that even slight reductions in drebrin levels can disrupt normal synaptic function.
β-adducin is an actin filament capping protein that serves as a bridge between actin and the membrane-associated cytoskeletal protein spectrin (Matsuoka et al. 2000). Spectrin/β-adducin complexes are key nodes in the very stable linkages between the red blood cell membrane and the underlying actin cytoskeleton. β-adducin likely plays a similar role in stabilizing membrane-actin cytoskeletal linkages in dendritic spines. Phosphorylation of β-adducin by protein kinase C, a key trigger for synaptic plasticity, can disrupt β-adducin’s linkages to actin and spectrin, which may be critical to reshape spines during plasticity. Indeed, phosphorylated β-adducin is enriched in the dendritic spines of hippocampal neurons (Matsuoka et al. 1998). Although relatively normal in appearance, dendritic spines exhibit greatly increased rates of formation and turnover in b-adducin knockout mice (Bednarek and Caroni 2011). Stimuli can induce new spine formation in b- adducin knockout mice, but a significant fraction of these new spines fail to develop PSD95-positive postsynaptic densities (Bednarek and Caroni 2011; Jung et al. 2013). This increased spine lability and defective synapse formation likely contribute to the observed reductions in spine density in b-adducin knockout mice (Jung et al. 2013).
4 Activity-Dependent Mechanisms Regulate the Maintenance of Dendrite Arbors and Dendritic Spines
Neuronal activity controls the remodeling and stabilization of both dendritic spines and dendrite arbors. Specifically, the coordinated remodeling and stabilization of neuronal circuits requires crosstalk between dendritic spines and dendrite arbors through shared, activity-dependent signaling molecules that influence cytoskeletal shaping and stabilization. This crosstalk is, in part, mediated by brain-derived neurotrophic factor (BDNF) signaling through TrkB, which can influence both actin and microtubule dynamics. In support of this theory, BDNF release correlates with synaptic activity in cultured hippocampal neurons (Kohara et al. 2001; Kojima et al. 2001; Hartmann et al. 2001). In addition to BDNF-TrkB signaling, activation of calcium/calmodulin-dependent protein kinase II (CaMKII) in response to synaptic activity likely mediates dendritic spine and dendrite arbor stabilization, as premature dendrite stabilization is achieved by expression of constitutively active CaMKII (Wu and Cline 1998).
4.1 BDNF-TrkB Signaling Regulates Dendritic Spine Size and Stability
In response to synaptic activity, BDNF is released from both the pre- and postsynaptic terminals (Kohara et al. 2001; Kojima et al. 2001; Hartmann et al. 2001). BDNF acts on presynaptic TrkB to promote axon arbor maturation and to stabilize presynaptic release sites during development (Hu et al. 2005; Marshak et al. 2007). Intriguingly, blocking NMDAR activity disrupts the clustering of presynaptic release sites, but this effect can be overcome by treatment with BDNF, suggesting that NMDAR-mediated BDNF release stabilizes presynaptic boutons (Hu et al. 2005). While the mechanisms underlying these presynaptic changes mediated by BDNF-TrkB signaling remain unclear, TrkB may signal through cortactin or other actin regulatory proteins to mediate stabilization of the presynaptic actin network, which organizes synaptic vesicles and their release machinery.
The delivery of BDNF to postsynaptic TrkB modulates several pathways that mediate dendritic spine enlargement and stabilization. In particular, BDNF stimulation of TrkB activates the Rac1 GTPase, which stimulates the PAK3-LIMK cascade and inhibitory phosphorylation of cofilin , resulting in increased spine size and stability (Rex et al. 2007; Lai et al. 2012). Additionally, BDNF-induced Rac1 activation promotes Arp2/3 complex activation by WAVE1 and the simultaneous release of mRNAs encoding cytoskeletal elements from translational inhibition by the fragile X mental retardation protein complex (De Rubeis et al. 2013). BDNF signaling through TrkB also mediates activity-dependent activation of the Ras GTPase to promote spine enlargement (Fig. 14.3) (Atwal et al. 2000; Yasuda et al. 2006). Overall, BDNF-TrkB signaling employs these and likely additional mechanisms to mediate the activity-induced maintenance of dendritic spines. In addition, other activity-dependent mechanisms may potentiate the effects of BDNF-TrkB signaling, as research has shown that actin content increases following high-frequency stimulation of presynaptic afferents in hippocampal slices, yet this increase is only attenuated by TrkB blockade (Rex et al. 2007). Also, when combined with theta burst stimulation, BDNF treatment of hippocampal slices increases the actin content in spines more than either manipulation alone, suggesting that activity and BDNF act synergistically (Rex et al. 2007). Overall, BDNF-TrkB signaling regulates, at least in part, the activity-induced remodeling and stabilization of dendritic spines by controlling regulators of the actin cytoskeleton.
Activity-dependent dendritic spine stabilization. During glutamatergic neurotransmission, glutamate is released from presynaptic terminals and binds to AMPA receptors and NMDA receptors (NMDARs) on the postsynaptic membrane. At the resting membrane potential, the NMDAR is blocked by Mg2+; however, once sufficiently depolarized, the Mg2+ blockade is released and Ca2+ enters the dendritic spine. Ca2+ influx through NMDARs activates a variety of signaling mechanisms, including those controlled by calcium/calmodulin-dependent protein kinase II (CaMKII). CaMKII phosphorylates p135 SYNGAP to catalyze the activation of Ras GTPase, which in turn activates Tiam1, a Rac1 guanine nucleotide exchange factor (GEF). CaMKII can also directly and rapidly phosphorylate and activate Kalirin-7, another Rac1 GEF. Kalirin-7 and Tiam1 catalyze Rac1 activation, which signals through the PAK3-LIMK pathway to inhibit the actin-severing ability of cofilin and through the WAVE regulatory complex (WRC) and WAVE1 to stimulate the actin-nucleating ability of the Arp2/3 complex. In addition, CaMKII can activate RhoA, which inhibits the actin-severing ability of cofilin through the ROCK-LIMK pathway. Activity-dependent release of brain-derived neurotrophic factor (BDNF) and stimulation of the tropomyosin receptor kinase B (TrkB) also activate Ras and Rac1 to mediate their downstream effects on cofilin and the Arp2/3 complex
4.2 BDNF-TrkB Signaling Mediates Dendrite Arbor Stability Through the Regulation of Microtubule-Binding Proteins
Neuronal activity, which promotes BDNF-TrkB signaling (Kohara et al. 2001; Kojima et al. 2001; Hartmann et al. 2001), is essential for dendrite arbor growth, branching, and stabilization in developing and mature neurons (Wu and Cline 1998; Xu et al. 2000; Vaillant et al. 2002; Szebenyi et al. 2005). Specifically, knockout of TrkB from cortical pyramidal neurons leads to dendrite shrinkage (Xu et al. 2000). In addition, dendrites develop normally on cortical layer 2/3 neurons in forebrain-specific BDNF knockout mice, but they shrink by 3 weeks of age, indicating that cortical BDNF supports dendrite maintenance rather than initial development (Gorski et al. 2003). The mechanism(s) by which synaptic activity-dependent neurotrophin signaling influence microtubule stability are still being elucidated. Nonetheless, direct application of BDNF or nerve growth factor to developing neurons has been observed to promote increased expression of MAP2 and MAP1A as well as their association with microtubules to stabilize dendrite arbors (Bloom et al. 1984; Huber and Matus 1984; Vaillant et al. 2002; Szebenyi et al. 2005). Additionally, BDNF exposure increases mRNA and protein levels of cypin, a guanine deaminase that promotes microtubule polymerization to regulate dendrite branching and stabilization (Akum et al. 2004; Kwon et al. 2011). Overall, BDNF-TrkB signaling appears to influence dendrite arbor growth and stabilization by regulating proteins that mediate microtubule polymerization and stability.
4.3 Neuronal Activity Induces the Transient Invasion of Microtubules into Dendritic Spines and Mediates Crosstalk Between Dendrite Arbors and Dendritic Spines
In addition to the discrete effects of neuronal activity on the actin and microtubule cytoskeletons, activity-dependent mechanisms also mediate crosstalk between dendrite arbors and dendritic spines, influencing their maintenance. Even though spines are actin-rich and contain few microtubules, the dynamic plus ends of microtubules can make activity-dependent invasions into a subset of dendritic spines. In particular, dynamic or growing microtubule plus ends are associated with tip-binding proteins, such as end-binding protein 3 (EB3 ; also known as MAPRE3) (Fig. 14.1) (Jaworski et al. 2009). Live imaging studies have revealed that neuronal activity and BDNF application modulate the number and duration of transient invasions of EB3-labeled microtubule tips into dendritic spines (Gu et al. 2008; Hu et al. 2008, 2011; Jaworski et al. 2009). Invasion of microtubules into individual spines causes increased spine accumulation of PSD95 and the EB3-binding protein p140Cap, while no significant change in PSD95 or p140Cap is observed in spines that do not exhibit microtubule invasions (Jaworski et al. 2009; Hu et al. 2011). Both PSD95 and p140Cap are required for spine maintenance and stability, and their accumulation is associated with increased actin content and spine enlargement (Jaworski et al. 2009; Hu et al. 2011). In particular, p140Cap interacts with cortactin, which binds and stabilizes actin and recruits the Arp2/3 complex to facilitate actin branch formation, to promote spine stability (Weed et al. 2000; Uruno et al. 2001; Weaver et al. 2001; Jaworski et al. 2009; Courtemanche et al. 2015). Pharmacological inhibition of microtubule dynamics and EB3 knockdown impair PSD95 and p140Cap accumulation, respectively, leading to spine shrinkage or destabilization (Gu et al. 2008; Hu et al. 2008, 2011; Jaworski et al. 2009). In addition, under conditions that induce NMDAR-dependent LTD, EB3 dissociates from growing microtubules and associates with MAP2-positive microtubules in the dendritic shaft, resulting in the inhibition of dynamic microtubule elongation, loss of microtubule targeting to spines, and shrinkage and loss of dendritic spines (Kapitein et al. 2011). Finally, because microtubule invasions are associated with increased spine stability, a decrease in BDNF-induced microtubule invasions could, in part, explain the diminished spine density in animals with a reduced gene dosage of TrkB (von Bohlen und Halbach et al. 2003). Overall, the transient invasion of microtubules into dendritic spines occurs in response to synaptic activity and mediates crosstalk between the microtubule-rich dendrite arbor and the actin-rich dendritic spine.
4.4 CaMKII Mediates Synaptic Activity-Based Alterations in Dendrite Arbor and Dendritic Spine Structure
Reductions in synaptic input, such as experimental deafferentation and sensory input removal, result in fewer dendrites, shorter dendrite length, and decreased dendritic branching in many model systems (Clark 1957; Matthews and Powell 1962; Jones and Thomas 1962; Coleman and Riesen 1968; Trachtenberg et al. 2002). The maintenance of this input from presynaptic boutons to dendritic spines is crucial for the long-term stability of dendritic spines. Specifically, in vivo time-lapse two-photon imaging of dendritic spines has revealed that changes in sensory experience, such as whisker trimming, alter spine formation and elimination (Trachtenberg et al. 2002; Zuo et al. 2005b; Holtmaat et al. 2006). At the molecular level, activity-dependent mechanisms activate CaMKII and the MEK-ERK pathway , both of which are required for activity-dependent stabilization of dendrites in cultured sympathetic neurons (Vaillant et al. 2002). CaMKII and ERK stabilize microtubules via the increased phosphorylation of MAP2, which promotes its binding to microtubules (Schulman 1984; Ray and Sturgill 1987; Sánchez et al. 2000; Vaillant et al. 2002). Precise temporal and activity-dependent control of CaMKII is important for dendrite maturation and stabilization. Expression of constitutively active CaMKII causes premature dendrite stabilization, while knocking down CaMKII expression correlates with reduced branch dynamics of developing dendrite arbors (Wu and Cline 1998). Specifically, CaMKIIα predominantly localizes to spines and dendritic subdomains near activated spines. Lemieux et al. have proposed that subspine CaMKIIα clusters may decode compartmental calcium transients to both support synapse remodeling through local changes in the actin cytoskeleton and interact with microtubules during activity-induced plasticity in cultured neurons (Takemoto-Kimura et al. 2010; Lemieux et al. 2012). The effect of CaMKIIα on dendrite arbor and dendritic spine stabilization may be mediated through its cytoskeleton-associated substrates, such as the Rac1 guanine nucleotide exchange factors Kalirin-7 (Xie et al. 2007) and T lymphoma invasion and metastasis-inducing protein 1 (Tiam1) (Fleming et al. 1999), as well as the Ras GTPase-activating protein p135 SYNGAP (Chen et al. 1998) (Fig. 14.3). Overall, CaMKIIα mediates activity-induced alterations in both the actin cytoskeleton of dendritic spines and the microtubule cytoskeleton of dendrite arbors. Further studies are needed to elucidate which specific targets of CaMKIIα mediate dendrite arbor and dendritic spine stabilization.
5 Adhesion Receptor and Neurotrophin Receptor Signaling to the Cytoskeleton Confer Long-Term Dendritic Spine Stability
Cell surface adhesion receptors, including ephrins and their Eph receptors, immunoglobulin superfamily receptors, cadherins, and integrins, play important roles in dendritic spine formation and stability (Biederer and Stagi 2008; Shi et al. 2009; Benson and Huntley 2012). Upon engagement with their extracellular binding partners, these receptors employ cytoplasmic nonreceptor tyrosine kinases of the focal adhesion kinase (FAK), Src, and Abl families to coordinate changes in cytoskeletal structure that impact spine stability.
5.1 EphB Signaling Through FAK Stabilizes Mature Dendritic Spines
The ephrin ligands for Eph receptors are composed of ephrin As, which activate EphA receptors and are anchored to the cell surface via covalent linkage to glycophosphoinositide lipids, and ephrin Bs, which activate EphB receptors and contain a transmembrane domain that can engage SH2 domain and PDZ domain-containing proteins to signal into the cell (Klein 2009). Importantly, just as ephrin binding activates the Eph receptor in so-called “forward” signaling, binding to an Eph receptor can also generate “reverse” signaling through the ephrin. Although originally identified as a kinase associated with integrin signaling, FAK can be activated by a variety of cell surface receptors, including EphB receptors, that regulate spine stability. Disruption of EphB2 signaling or knockdown of FAK in established hippocampal neuron cultures leads to significant loss of mature mushroom-shaped dendritic spines and an increase in immature thin “filopodia-like” spines (Shi et al. 2009). FAK may mediate spine stabilization by stimulating p190RhoGEF, which activates the RhoA-ROCK pathway and causes downstream phosphorylation and inactivation of cofilin by LIMK. Consistent with this model, an inactive phospho-mimetic cofilin mutant can block the spine destabilization induced by loss of EphB2-FAK signaling (Shi et al. 2009). In addition, activated FAK recruits and activates Src family kinases, such as Fyn, to phosphorylate p130CAS, a scaffolding molecule that promotes Rac1 activation (Tomar and Schlaepfer 2009). Interestingly, knockout of the Src family kinase Fyn leads to a progressive age-dependent loss of dendritic spines in mice (Babus et al. 2011), and knockdown of p130CAS leads to spine loss in established hippocampal neuron cultures (Bourgin et al. 2007). Both manipulations are expected to disrupt Rac1 activation in the spine, which suggests that FAK and Rac1 signaling are important mediators of dendritic spine stability. FAK can also phosphorylate the key spine protein cortactin and promote its binding to the Abl2/Arg kinase and the Nck1 adaptor, which activates the Arp2/3 complex to promote actin assembly (Fig. 14.4) (MacGrath and Koleske 2012b; Tomar et al. 2012). Overall, FAK signaling in response to EphB2 stimulation mediates stabilization of the actin cytoskeleton and dendritic spines through the Rac1 GTPase.
Adhesion-dependent dendritic spine stabilization. Cell surface adhesion receptors, including ephrin B, EphB2 receptor, and integrin α3β1, coordinate changes in cytoskeletal structure and impact spine stability by employing cytoplasmic nonreceptor tyrosine kinases, such as the focal adhesion kinase (FAK) and Abl2/Arg kinase. Ephrin B on the presynaptic membrane stimulates the EphB2 receptor on the postsynaptic membrane to activate FAK. FAK activates Src family kinases, such as Fyn, to phosphorylate p130CAS, which, in complex with DOCK1 and the adaptor molecule CRK, mediates the activation of the Rac1 GTPase. Rac1 signals through the WAVE regulatory complex (WRC) and WAVE1 to stimulate actin nucleation by the Arp2/3 complex and through the PAK3-LIMK pathway to inhibit actin severing by cofilin. FAK also stimulates p190RhoGEF to activate RhoA, which signals through the ROCK-LIMK pathway to inhibit actin severing by cofilin. FAK also phosphorylates cortactin, which promotes its binding to Abl2/Arg kinase and activates the Arp2/3 complex to promote actin stabilization, branching, and nucleation. In addition, upon binding of its extracellular matrix (ECM) ligand, integrin α3β1 signals through Abl2/Arg kinase, which both cooperatively binds actin filaments to prevent their depolymerization and promotes cortactin binding to actin filaments to further stabilize them
5.2 Astrocyte-Derived Ephrin A2 Regulates Dendritic Spine Stability by Controlling Glutamate Transmission in the Synaptic Cleft
Knockout of ephrin A2 in mice leads to dendritic spine loss in the mouse sensory cortex beginning at 1 month of age and leading to a significant reduction by 4 months of age. Interestingly, this spine loss requires activity, as trimming of whiskers to reduce sensory input or treatment with NMDAR antagonists greatly reduces the enhanced spine loss observed in ephrina2 knockouts. Ephrin A2 localizes to perisynaptic astrocytic processes where it regulates dendritic spine stability via a novel mechanism. Ephrin A2 stabilizes two distinct astrocytic glutamate transporters that help to clear glutamate from the synaptic cleft. Expression of these glutamate transporters is reduced in ephrina2 knockout mice, leading to elevated levels of synaptic glutamate, which triggers spine elimination. It appears that one or more EphA receptors in the dendritic spine engage this astrocytic ephrin A2-glutamate transporter system to prevent excessive glutamate buildup and maintain optimal synaptic function and spine stability (Yu et al. 2013).
5.3 Integrin α3β1 Signaling Through Abl2/Arg and Cortactin Mediates Dendritic Spine Stability
Integrins are heterodimeric receptors for the extracellular matrix that are composed of α and β subunits and are widely expressed in the nervous system (Pinkstaff et al. 1999). Interestingly, inhibition or loss of the integrin α3 or β1 subunits destabilizes spines and reduces synapse density (Bourgin et al. 2007; Warren et al. 2012; Kerrisk et al. 2013). Integrins signal through the Abl family kinases, Abl1/Abl and Abl2/Arg in vertebrates, to coordinate changes in cytoskeletal structure (Bradley and Koleske 2009). Arg is particularly abundant in the nervous system (Koleske et al. 1998), where it localizes to dendritic spines (Moresco et al. 2003; Warren et al. 2012; Lin et al. 2013). arg knockout mice exhibit normal spine and synapse development through postnatal day 21, followed by significant spine and synapse loss as the mice mature to adulthood (Sfakianos et al. 2007; Gourley et al. 2009b), phenotypes that closely resemble those resulting from neuron-specific ablation of integrin α3 or β1 (Warren et al. 2012; Kerrisk et al. 2013). Importantly, biochemical and genetic experiments indicate that the integrin β1 cytoplasmic tail binds directly to Arg and these interactions with Arg confer dendritic spine and dendrite stability (Warren et al. 2012; Simpson et al. 2015).
Dendritic spine stabilization by Arg in response to integrin α3β1 signaling occurs through two known mechanisms. First, Arg binds cooperatively to actin filaments and prevents their depolymerization (Fig. 14.4) (Wang et al. 2001; MacGrath and Koleske 2012a; Courtemanche et al. 2015), and second, Arg promotes increased cortactin binding to actin filaments to further stabilize them (MacGrath and Koleske 2012a). Knockdown of Arg in cultured neurons leads to cortactin loss from spines, a parallel decrease in spine actin content, and a 50 % loss of spines. NMDAR antagonists can prevent cortactin depletion from spines and rescue spine loss resulting from Arg knockdown. These data strongly suggest that activity-driven cortactin loss from spines is the cause of synapse loss in Arg knockdown neurons. Indeed, fusion of the Arg actin-binding domains to cortactin drives it into spines and rescues spine instability in Arg knockdown neurons (Lin et al. 2013). Whether and how Arg also interfaces functionally with NMDARs or other components of the glutamatergic signaling apparatus is a major unresolved question.
6 Dendritic Spines Are Pathologically Destabilized with Normal Aging and in Disease
6.1 Region-Specific Dendrite Arbor, Dendritic Spine, and Synaptic Alterations Occur with Normal Aging
Aging is the primary risk factor for most neurodegenerative diseases and is accompanied by cognitive decline even in individuals unaffected by neurodegeneration. Nonetheless, very little research has been done to understand whether and how aging is associated with alterations in dendrite and dendritic spine morphology and stability, and if so, what molecular mechanisms are involved. Despite early evidence that age-related cell loss occurred in all cortical layers (Brody et al. 1955) and the hippocampus (Ball 1977) of humans, stereological methods of neuronal quantification have since shown that cell death is not a characteristic of normal aging, but rather of neurodegeneration (West et al. 1994). More recent studies suggest that age-related impairments in cognitive performance may instead result from subtle dendritic and synaptic alterations in the hippocampus and prefrontal cortex (Morrison and Hof 1997; Burke and Barnes 2006; Morrison and Baxter 2012). In particular, region-specific alterations with advanced aging have been shown to occur in dendrite morphology (Flood et al. 1987b; Hanks and Flood 1991; De Brabander et al. 1998; Uylings and De Brabander 2002), dendritic spine density (Dumitriu et al. 2010; Bloss et al. 2011; Young et al. 2014), and dendritic spine stabilization (Mostany et al. 2013).
The functions of the human dorsolateral prefrontal cortex, such as working memory, decline with age (Gazzaley and D’Esposito 2007; Arnsten et al. 2012), and this is associated with changes in dendrite arbor and dendritic spine structure. In humans, decline of prefrontal function is accompanied by age-related decreases in cortical dendrite arbor branching (De Brabander et al. 1998; Uylings and De Brabander 2002). Studies of the prefrontal cortex in nonhuman primates have revealed a 30–33 % reduction of dendritic spines on pyramidal neurons with advancing age (Peters et al. 1998; Peters et al. 2008; Dumitriu et al. 2010; Young et al. 2014). These significant reductions occur primarily with long, thin spines in this region, while no decrease is observed in mushroom or stubby spine density (Dumitriu et al. 2010; Bloss et al. 2011; Young et al. 2014). Additionally, studies in the rodent cortex have shown that aged rats have decreased overall spine density but increased dendritic spine stability in a chronic stress paradigm in comparison to young rats, who exhibit dendritic spine loss during stress (Bloss et al. 2011). A recent study investigated these age-related changes in spine dynamics using transcranial in vivo two-photon imaging of mouse somatosensory cortex. Spine density was shown to increase through adulthood, stabilizing in mature and old mice. Interestingly, the authors found a decrease in the long-term stability and size of dendritic spines in aged mice and an increase in turnover spine dynamics in very old animals, suggesting a return of spines to a more plastic state later in life (Mostany et al. 2013). These studies suggest that cortical network flexibility, spine remodeling, and spine stabilization are altered during normal aging.
In contrast to the prefrontal cortex, no changes in dendritic branching are observed with aging in hippocampal area CA1 (Hanks and Flood 1991) or CA3 (Flood et al. 1987b). Additionally, even though age-related CA3 spine loss is observed in the rat hippocampus (Smith et al. 2000; Adams et al. 2010), no CA1 spine loss is observed with normal aging (Geinisman et al. 2004). Instead, recent studies suggest that either the maintenance of complex synapses or the conversion from simple axospinous to complex synapses may be impaired with advancing age in the hippocampus, specifically in CA1. Complex synapses, such as perforated synapses or multisynaptic boutons, are enriched following LTP induction, and their density correlates with increased efficacy of synaptic transmission (Toni et al. 2001; Geinisman et al. 1993; Ganeshina et al. 2004a, b). Specifically, LTP induction increases the number of both multi-synapse boutons and perforated spines, which are characterized by a fenestrated PSD, with high AMPA receptor expression in CA1 (Toni et al. 2001). Additionally, cognitive decline in aged spatial learning-impaired rats correlates with decreased PSD size of CA1 perforated synapses (Nicholson et al. 2004), and aged rats display a reduction in perforated synapses in the sublayer of the dentate gyrus receiving input from entorhinal cortex (Geinisman et al. 1986). Aged nonhuman primates also exhibit age-related reductions in multiple-synapse boutons (Hara et al. 2011). Overall, these data suggest that alterations in the number and composition of complex synapses may underlie age-related changes in the hippocampus. Nonetheless, the molecular mechanisms underlying age-related alterations in both the cortex and hippocampus merit further investigation.
6.2 Dendritic Spines and Dendrite Arbors Are Pathologically Destabilized in Alzheimer’s Disease
Alzheimer’s disease (AD) is characterized by region-specific alterations in the hippocampus, including synapse loss, dendrite arbor atrophy, and neuronal death, that are not observed during normal aging (Flood et al. 1987a, b; Flood 1991; West et al. 1994; Anderton et al. 1998). The decreases in synaptic density, dendrite arborization, and neuronal density correlate with the degree of dementia and AD pathology, with reductions in synapse number being the strongest pathological correlate of cognitive and memory impairment (Terry et al. 1991; Falke et al. 2003). Accordingly, advances in the past decade have highlighted how disrupting molecular mechanisms mediating dendritic spine and dendrite arbor stability could lead to AD pathology. In particular, the observed synapse loss appears to be pathologically induced by the presence of soluble Aβ-derived diffusible ligands (ADDLs), which are small, soluble oligomers derived from Aβ that correlate with memory loss in AD patients, adversely impact synaptic plasticity, cause dendritic spine loss, and contribute to dendritic atrophy in AD (Gong et al. 2003; Haass and Selkoe 2007) (for a recent review, see Pozueta et al. 2013). The application of ADDLs to cultured hippocampal neurons results in rapid alterations in dendritic spine morphology accompanied by a significant decrease in spine density (Lacor et al. 2007; Calabrese et al. 2007), which is also observed in organotypic slice (Shankar et al. 2007). In particular, the effects of ADDLs on dendritic spines may be mediated, at least in part, by the selective binding of ADDLs to certain receptors, such as the α7 nicotinic acetylcholine receptor (Wang et al. 2000), leukocyte immunoglobulin-like receptor B2 (Kim et al. 2013b), and cellular prion protein (PrPC) (Freir et al. 2011; Um et al. 2012). For example, ADDLs bind to PrPC, which causes Fyn kinase activation and subsequent phosphorylation of the NMDAR NR2B subunit (Freir et al. 2011; Um et al. 2012). This ADDL-induced NMDAR phosphorylation causes an initial increase in NMDAR expression at the postsynaptic membrane of excitatory neurons, followed by a subsequent decrease in NMDAR expression mediated by receptor endocytosis (Lacor et al. 2007; Shankar et al. 2007; Um et al. 2012). The initial increase in NMDAR surface activity destabilizes dendritic spines by triggering loss of drebrin and cortactin from spines (Lacor et al. 2007; Um et al. 2012; Ishizuka et al. 2014) and causing the calcineurin-mediated dephosphorylation and activation of cofilin leading to actin disassembly (Fig. 14.4) (Wang et al. 2005; Wu et al. 2010). In support of this model, both calcineurin and cofilin are required for ADDL-mediated spine loss (Shankar et al. 2007). Additionally, the surface localization of other receptors implicated in dendritic spine stability and memory formation, such as EphB2, is disrupted by application of ADDLs (Lacor et al. 2007). Specifically, the application of ADDLs to neurons results in a 60 % decrease in EphB2 puncta within 6 h (Lacor et al. 2007), and disruption of EphB2 causes a decrease in mushroom-shaped dendritic spine density and an increase in “filopodia-like” spine formation in cultured hippocampal neurons (Shi et al. 2009).
In addition to their effects on dendritic spine structure, ADDLs have been shown to cause reductions in dendrite arborization through interactions with the microtubule-associated protein tau (Falke et al. 2003; Wu et al. 2010). Hyperphosphorylation of tau and its subsequent accumulation into neurofibrillary tangles is a hallmark of AD pathology that correlates with cognitive decline and the pathological loss of neurons and synapses (Giannakopoulos et al. 2003; Ingelsson et al. 2004). Hyperphosphorylated tau relocalizes from axons to somatodendritic compartments where it aggregates into neurofibrillary tangles, blocks trafficking on microtubules, and causes dystrophic patterning and simplification of dendrites (Wu et al. 2010; Kopeikina et al. 2012; Carlyle et al. 2014). These effects may be mediated by the ADDL-based activation of certain kinases, including MARK1, p70S6K, BRSK, and Fyn, that can phosphorylate tau, causing tau relocalization and microtubule depletion in response to elevated calcium levels (Fig. 14.5) (Zempel et al. 2010; Larson et al. 2012). In addition, these tau missorting events occur in proximity to areas of decreased spine density, suggesting that local elevations in calcium levels may mediate both spine loss and microtubule depletion (Zempel et al. 2010). Recently, tau has been suggested to play a physiologic role in the targeting of Fyn kinase to the PSD to regulate the NMDAR (Ittner et al. 2010). Thus, the ADDL-induced accumulation of hyperphosphorylated tau in spines could disrupt normal Fyn localization and thereby compromise NMDAR anchoring and activity at the synapse, leading to dendritic spine destabilization (Ittner et al. 2010; Hoover et al. 2010). Overall, pathologic ADDLs specifically target mechanisms that regulate dendritic spine and dendrite arbor stabilization and maintenance, resulting in loss of dendritic spines and dendrite arbors in AD.
Dendrite arbor and dendritic spine destabilization in Alzheimer’s disease. Aβ-derived diffusible ligands (ADDLs) selectively bind to certain receptors, such as the cellular prion protein (PrPC), which activate a variety of kinases, including Fyn kinase, that mediate the alterations in dendrite arbor and dendritic spine morphology observed in Alzheimer’s disease. In particular, Fyn phosphorylates the NMDA receptor (NMDAR) GluN2B subunit, which causes an initial increase in NMDAR expression at the postsynaptic membrane (1). The increased surface activity of NMDARs destabilizes spines by triggering actin disassembly through the calcineurin-dependent activation of cofilin and the loss of actin-binding proteins (ABPs), such as drebrin and cortactin. Following this period of increased NMDAR expression, NMDAR expression decreases as the receptors undergo endocytosis (2). Fyn and other kinases, such as MARK1, BRSK, and p70S6K, also hyperphosphorylate tau in response to ADDL-based activation of PrPC. Hyperphosphorylated tau accumulates into neurofibrillary tangles (NFTs) in the dendrite arbor, which blocks microtubule-based trafficking, triggers microtubule destabilization and depletion, and causes dystrophic patterning of dendrites. Additionally, ADDL application disrupts surface localization of receptors other than PrPC, including EphB2
6.3 Decreased Expression of Synaptic Proteins and Reduced BDNF Signaling Lead to Dendritic Spine Destabilization and Dendrite Atrophy in Depression
Major depressive disorder (MDD) is associated with reduced synapse density (Kang et al. 2012) and reduced dendrite arbor size in the prefrontal cortex and hippocampus (Drevets et al. 1997; Drevets 2000; Cotter et al. 2001, 2002). Interestingly, expression of the GATA1 transcriptional repressor is significantly increased in the brains of individuals with MDD (Kang et al. 2012). GATA1 is a master regulator of a cluster of genes that play critical roles in dendrite arbor maintenance and dendritic spine stability. Several GATA1 target genes are significantly downregulated in the MDD brain, including tubulin β4, the major β tubulin in neurons, and the trafficking regulators Rab3A and Rab4B (Kang et al. 2012). These reductions may decrease the size and stability of the microtubule network and perturb the flow of traffic within dendrites. This model is supported by the observation that GATA1 overexpression alone is sufficient to significantly reduce dendrite arbor size in cortical neurons (Kang et al. 2012). In addition, overexpression of GATA1 or the related GATA2 in the hippocampus is sufficient to reduce dendrite arbor size, decrease dendritic spine density, and lead to depression-like behaviors in mice (Choi et al. 2014). These observations support a model in which transcriptional repression by GATA1/GATA2 mediates the loss of connectivity and compromised neuronal function in MDD.
Chronic stress and elevated circulating corticosteroid levels are major risk factors for the development of MDD. Both conditions reduce BDNF and TrkB levels in the brain (Smith et al. 1995; Nibuya et al. 1999; Gourley et al. 2009a), which likely explains the reduction in BDNF in the brains of MDD patients (Schmidt and Duman 2007). Reduced BDNF signaling would be expected to compromise the BDNF-mediated support of dendritic spine and dendrite arbor stability, via mechanisms detailed in previous sections, thereby contributing to the neuronal destabilization found in this disease. Recent work shows that chronic corticosterone treatment also decreases expression of caldesmon, an actin-stabilizing molecule, increasing actin turnover in dendritic spines and resulting in smaller, more unstable spines (Tanokashira et al. 2012). Thus, reduced BDNF-TrkB signaling also appears to be a key factor in the altered connectivity and circuit dysfunction in MDD.
6.4 Dendritic Spine and Dendrite Arbor Stability Are Disrupted in Stroke
In addition to causing death of some neurons, stroke causes destabilization of spines and dendrites on neurons within and adjacent to the infarct area. For an individual neuron, this destabilization may clear dendrites away from areas where damage or death of afferents has disrupted connectivity and free up synaptic proteins and metabolic resources to remodel the dendrite arbor. In a photothrombotic murine ischemia model, light is used to occlude small vessels leading to lesions that are 1 mm in diameter. The reductions in dendrite arbor size in the peri-infarct region that follow this photothrombotic stroke are closely matched by growth of distal dendrites during the recovery period (Brown et al. 2010), suggesting that the neuron may attempt to reach some homeostatic “set point” in dendrite size and synaptic inputs as it reintegrates into networks. In contrast, occlusion of the middle cerebral artery leads to larger lesions averaging 7 mm3; with this ischemia model, apical dendrites in the peri-infarct region become permanently reduced in size and lose higher order branches. Interestingly, intravital imaging reveals that dendrite shrinkage results both from tissue shrinkage in the peri-infarct region and also from retraction of the distal tips (Mostany and Portera-Cailliau 2011).
Ischemia also leads to alterations in dendritic spine stability and dynamics. Introduction of photothrombotic lesions leads to significantly increased dendritic spine turnover rates in the peri-infarct region for up to 4 weeks following the lesion. This increased dynamic behavior correlates with functional recovery and remapping of the sensory input from the affected area to adjacent cortical regions (Brown et al. 2009). In the middle cerebral artery occlusion model, significant spine loss is observed already at 4 days after stroke with 20 % of spines lost in the peri-infarct region by 12 days after stroke. These spine losses recover by about 4 weeks after stroke (Kalia et al. 2008). The observed reductions in spine number result both from an increase in spine loss and a significant decrease in new spine formation. Importantly, spines do not recover at similar rates on all neurons, and the best predictor of recovery is the amount of local blood flow at 1 h after stroke induction. In addition, neurons in areas distal to the stroke gain supranumerary spine densities, suggesting that these areas may be contributing to the functional remodeling of cortical circuits.
Elevated NMDAR activity is associated with neuronal damage and death following stroke (for a recent review, see Kalia et al. 2008). Application of NMDA to cultured neurons or focal application of NMDA to tissues in vivo has been widely used to model this glutamate-induced excitotoxicity. While prolonged, excessive stimulation leads to neuron death, sublethal NMDA stimulation of neurons, either in culture or focally in tissues, causes spine loss, dendrite branch swelling, and dendrite shrinkage (Hasbani et al. 1998, 2001; Halpain et al. 1998; Hering and Sheng 2003; Kitaoka et al. 2004; Iki et al. 2005; Tseng and Firestein 2011). These changes are closely associated with disruptions in actin and microtubule structure in spines and dendrites, respectively (Hering and Sheng 2003; Graber et al. 2004).
Excessive NMDA stimulation destabilizes spines by disrupting the function of actin regulatory proteins in the spine. For example, bath application of NMDA to cultured hippocampal neurons causes cortactin to relocalize from dendritic spines to the dendrite shaft, and this correlates with loss of spine stability (Hering and Sheng 2003; Iki et al. 2005). In addition, inhibitors of the cathepsin B family of proteases attenuate NMDA-induced spine loss, indicating that proteolysis of one or more key actin regulatory proteins may contribute to spine disruption (Graber et al. 2004). Indeed, NMDA stimulation leads to reduced levels of the spine stabilizing protein MARCKS, but this decrease can be prevented by cathepsin B inhibition. Elevated NMDA stimulation also disrupts the targeting of dynamic microtubules to spines, which prevents delivery of key proteins, including PSD95 and p140Cap (Kapitein et al. 2011), required for spine stability.
Exactly how elevated NMDA stimulation triggers dendrite destabilization is less clear. One potential candidate for mediating these effects is the RhoA GTPase. Focal synaptic stimulation can activate RhoA in the spine, and activated RhoA can then diffuse into the dendritic shaft (Murakoshi et al. 2011). The summed signaling from a group of spines in aggregate, such as that following excessive NMDA stimulation, could trigger RhoA-ROCK signaling at levels sufficient to disrupt MAP function (Yamamoto et al. 1983; Murthy and Flavin 1983) and cypin expression (Chen and Firestein 2007), thereby destabilizing dendrites. In support of this theory, the ROCK inhibitor fasudil can reduce the severity of damage to the dendrite-rich inner plexiform layer of the retina resulting from NMDA injection (Kitaoka et al. 2004). Moreover, RhoA activation and NMDA stimulation have both been shown to decrease cypin levels in cultured neurons (Tseng and Firestein 2011). Finally, it is possible that NMDA-mediated disruption of microtubule structure impacts dendrites by disrupting the microtubule motor-based delivery of key building blocks to the dendrite. Overall, the stabilization of dendrite arbors and dendritic spines is severely disrupted following stroke through a mechanism downstream of excessive NMDAR activation.
7 Summary
While dendrite arbors and dendritic spines are highly dynamic in early development, structural plasticity decreases and stability increases as circuits mature. Afferent innervation or synapse formation onto dendrite arbors or dendritic spines stabilizes them, while loss or decrease of synaptic inputs destabilizes dendrites and dendritic spines. During maturation, dendrite arbors stabilize before dendritic spines and help retain the overall dendrite arbor field integrity while preserving the ability to refine synaptic connections. A diverse collection of cellular and molecular mechanisms regulates this differential stabilization of dendritic spines and dendrite branches. Nonetheless, the coordinated stabilization of neuronal circuits requires the existence of shared signaling mechanisms, such as BDNF-TrkB signaling and CaMKII activation, that act on cytoskeletal elements and contribute to the stabilization of both dendritic spines and dendrite arbors. Accordingly, the long-term structural and functional stability of dendrite arbors and dendritic spines is maintained by these dynamic cytoskeletal elements that turn over in seconds to hours. To confer stability, the formation and turnover of actin and microtubule structures are exquisitely controlled by regulatory proteins, including Rho family GTPases, kinases, actin-binding proteins, and microtubule-binding proteins, that ensure the balanced turnover and maintenance of the actin and microtubule cytoskeletons so that they can serve as scaffolds for their own replenishment and reorganization. Cell surface adhesion receptors, including Eph receptors, immunoglobulin superfamily receptors, cadherins, and integrins, also play important roles in dendritic spine formation and stability through the employment of cytoplasmic nonreceptor tyrosine kinases, such as those of the FAK, Src, and Abl families, to mediate changes in cytoskeletal structure. Finally, the loss of dendritic spine and/or dendrite arbor stability in humans is a major contributing factor to normal aging and the pathology of psychiatric illnesses, such as major depressive disorder; neurodegenerative diseases, such as Alzheimer’s disease; and stroke. Overall, the cellular and molecular mechanisms that mediate dendrite arbor and dendritic spine maintenance are crucial for proper brain function.
References
Adams MM, Donohue HS, Linville MC et al (2010) Age-related synapse loss in hippocampal CA3 is not reversed by caloric restriction. Neuroscience 171:373–382. doi:10.1016/j.neuroscience.2010.09.022
Akum BF, Chen M, Gunderson SI et al (2004) Cypin regulates dendrite patterning in hippocampal neurons by promoting microtubule assembly. Nat Neurosci 7:145–152. doi:10.1038/nn1179
Anderton BH, Callahan L, Coleman P et al (1998) Dendritic changes in Alzheimer’s disease and factors that may underlie these changes. Prog Neurobiol 55:595–609
Arber S, Barbayannis FA, Hanser H et al (1998) Regulation of actin dynamics through phosphorylation of cofilin by LIM-kinase. Nature 393:805–809
Arellano JI, Benavides-Piccione R, Defelipe J, Yuste R (2007) Ultrastructure of dendritic spines: correlation between synaptic and spine morphologies. Front Neurosci 1:131–143. doi:10.3389/neuro.01.1.1.010.2007
Arnsten AFT, Wang MJ, Paspalas CD (2012) Neuromodulation of thought: flexibilities and vulnerabilities in prefrontal cortical network synapses. Neuron 76:223–239. doi:10.1016/j.neuron.2012.08.038
Atwal JK, Massie B, Miller FD, Kaplan DR (2000) The TrkB-Shc site signals neuronal survival and local axon growth via MEK and P13-kinase. Neuron 27:265–277
Baas PW, Deitch JS, Black MM, Banker GA (1988) Polarity orientation of microtubules in hippocampal neurons: uniformity in the axon and nonuniformity in the dendrite. Proc Natl Acad Sci U S A 85:8335–8339
Babus LW, Little EM, Keenoy KE et al (2011) Decreased dendritic spine density and abnormal spine morphology in Fyn knockout mice. Brain Res 1415:96–102. doi:10.1016/j.brainres.2011.07.059
Ball MJ (1977) Neuronal loss, neurofibrillary tangles and granulovacuolar degeneration in the hippocampus with ageing and dementia. A quantitative study. Acta Neuropathol 37:111–118
Bednarek E, Caroni P (2011) β-Adducin is required for stable assembly of new synapses and improved memory upon environmental enrichment. Neuron 69:1132–1146. doi:10.1016/j.neuron.2011.02.034
Benson DL, Huntley GW (2012) Building and remodeling synapses. Hippocampus 22:954–968. doi:10.1002/hipo.20872
Biederer T, Stagi M (2008) Signaling by synaptogenic molecules. Curr Opin Neurobiol 18:261–269. doi:10.1016/j.conb.2008.07.014
Biou V, Brinkhaus H, Malenka RC, Matus A (2008) Interactions between drebrin and Ras regulate dendritic spine plasticity. Eur J Neurosci 27:2847–2859. doi:10.1111/j.1460-9568.2008.06269.x
Bloom GS, Schoenfeld TA, Vallee RB (1984) Widespread distribution of the major polypeptide component of MAP 1 (microtubule-associated protein 1) in the nervous system. J Cell Biol 98:320–330
Bloss EB, Janssen WG, Ohm DT et al (2011) Evidence for reduced experience-dependent dendritic spine plasticity in the aging prefrontal cortex. J Neurosci 31:7831–7839. doi:10.1523/JNEUROSCI.0839-11.2011
Boda B, Alberi S, Nikonenko I et al (2004) The mental retardation protein PAK3 contributes to synapse formation and plasticity in hippocampus. J Neurosci 24:10816–10825. doi:10.1523/JNEUROSCI.2931-04.2004
Bourgin C, Murai KK, Richter M, Pasquale EB (2007) The EphA4 receptor regulates dendritic spine remodeling by affecting beta1-integrin signaling pathways. J Cell Biol 178:1295–1307. doi:10.1083/jcb.200610139
Bourne J, Harris KM (2007) Do thin spines learn to be mushroom spines that remember? Curr Opin Neurobiol 17:381–386. doi:10.1016/j.conb.2007.04.009
Bradley WD, Koleske AJ (2009) Regulation of cell migration and morphogenesis by Abl-family kinases: emerging mechanisms and physiological contexts. J Cell Sci 122:3441–3454. doi:10.1242/jcs.039859
Brody DA, Harris TR, Romans WE (1955) A simple method for obtaining esophageal electrocardiograms of good diagnostic quality. Am Heart J 50:923–926
Brown CE, Aminoltejari K, Erb H et al (2009) In vivo voltage-sensitive dye imaging in adult mice reveals that somatosensory maps lost to stroke are replaced over weeks by new structural and functional circuits with prolonged modes of activation within both the peri-infarct zone and distant sites. J Neurosci 29:1719–1734. doi:10.1523/JNEUROSCI.4249-08.2009
Brown CE, Boyd JD, Murphy TH (2010) Longitudinal in vivo imaging reveals balanced and branch-specific remodeling of mature cortical pyramidal dendritic arbors after stroke. J Cereb Blood Flow Metab 30:783–791. doi:10.1038/jcbfm.2009.241
Burke SN, Barnes CA (2006) Neural plasticity in the ageing brain. Nat Rev Neurosci 7:30–40. doi:10.1038/nrn1809
Burton PR (1988) Dendrites of mitral cell neurons contain microtubules of opposite polarity. Brain Res 473:107–115
Calabrese B, Shaked GM, Tabarean IV et al (2007) Rapid, concurrent alterations in pre- and postsynaptic structure induced by naturally-secreted amyloid-beta protein. Mol Cell Neurosci 35:183–193. doi:10.1016/j.mcn.2007.02.006
Carlyle BC, Nairn AC, Wang M et al (2014) cAMP-PKA phosphorylation of tau confers risk for degeneration in aging association cortex. Proc Natl Acad Sci U S A 111:5036–5041. doi:10.1073/pnas.1322360111
Chen H, Firestein BL (2007) RhoA regulates dendrite branching in hippocampal neurons by decreasing cypin protein levels. J Neurosci 27:8378–8386. doi:10.1523/JNEUROSCI.0872-07.2007
Chen Y-K, Hsueh Y-P (2012) Cortactin-binding protein 2 modulates the mobility of cortactin and regulates dendritic spine formation and maintenance. J Neurosci 32:1043–1055
Chen HJ, Rojas-Soto M, Oguni A, Kennedy MB (1998) A synaptic Ras-GTPase activating protein (p135 SynGAP) inhibited by CaM kinase II. Neuron 20:895–904
Chen B, Brinkmann K, Chen Z et al (2014) The WAVE regulatory complex links diverse receptors to the actin cytoskeleton. Cell 156:195–207. doi:10.1016/j.cell.2013.11.048
Choi M, Wang SE, Ko SY et al (2014) Overexpression of human GATA-1 and GATA-2 interferes with spine formation and produces depressive behavior in rats. PLoS One 9:e109253. doi:10.1371/journal.pone.0109253
Clark WL (1957) Inquiries into the anatomical basis of olfactory discrimination. Proc R Soc Lond B Biol Sci 146:299–319
Cline H, Haas K (2008) The regulation of dendritic arbor development and plasticity by glutamatergic synaptic input: a review of the synaptotrophic hypothesis. J Physiol 586:1509–1517. doi:10.1113/jphysiol.2007.150029
Coleman PD, Riesen AH (1968) Environmental effects on cortical dendritic fields. I. Rearing in the dark. J Anat 102:363–374
Conde C, Cáceres A (2009) Microtubule assembly, organization and dynamics in axons and dendrites. Nat Rev Neurosci 10:319–332
Cotter D, Mackay D, Landau S et al (2001) Reduced glial cell density and neuronal size in the anterior cingulate cortex in major depressive disorder. Arch Gen Psychiatry 58:545–553
Cotter D, Mackay D, Chana G et al (2002) Reduced neuronal size and glial cell density in area 9 of the dorsolateral prefrontal cortex in subjects with major depressive disorder. Cereb Cortex 12:386–394
Counts SE, He B, Nadeem M et al (2012) Hippocampal drebrin loss in mild cognitive impairment. Neurodegener Dis 10:216–219. doi:10.1159/000333122
Courtemanche N, Gifford SM, Simpson MA et al (2015) Abl2/Abl-related gene stabilizes actin filaments, stimulates actin branching by actin-related protein 2/3 complex and promotes actin filament severing by cofilin. J Biol Chem 290:4038–4046. doi:10.1074/jbc.M114.608117
Dailey ME, Smith SJ (1996) The dynamics of dendritic structure in developing hippocampal slices. J Neurosci 16:2983–2994. doi:10.1002/cne.901950106
De Brabander JM, Kramers RJ, Uylings HB (1998) Layer-specific dendritic regression of pyramidal cells with ageing in the human prefrontal cortex. Eur J Neurosci 10:1261–1269
De Camilli P, Miller PE, Navone F et al (1984) Distribution of microtubule-associated protein 2 in the nervous system of the rat studied by immunofluorescence. Neuroscience 11:817–846
De Rubeis S, Pasciuto E, Li KW et al (2013) CYFIP1 coordinates mRNA translation and cytoskeleton remodeling to ensure proper dendritic spine formation. Neuron 79:1169–1182. doi:10.1016/j.neuron.2013.06.039
Drevets WC (2000) Functional anatomical abnormalities in limbic and prefrontal cortical structures in major depression. Prog Brain Res 126:413–431. doi:10.1016/S0079-6123(00)26027-5
Drevets WC, Price JL, Simpson JRJ et al (1997) Subgenual prefrontal cortex abnormalities in mood disorders. Nature 386:824–827. doi:10.1038/386824a0
Du Y, Weed SA, Xiong WC et al (1998) Identification of a novel cortactin SH3 domain-binding protein and its localization to growth cones of cultured neurons. Mol Cell Biol 18:5838–5851
Dubos A, Combeau G, Bernardinelli Y et al (2012) Alteration of synaptic network dynamics by the intellectual disability protein PAK3. J Neurosci 32:519–527. doi:10.1523/JNEUROSCI.3252-11.2012
Dumitriu D, Hao J, Hara Y et al (2010) Selective changes in thin spine density and morphology in monkey prefrontal cortex correlate with aging-related cognitive impairment. J Neurosci 30:7507–7515. doi:10.1523/JNEUROSCI.6410-09.2010
Edson KJ, Lim SS, Borisy GG, Letourneau PC (1993) FRAP analysis of the stability of the microtubule population along the neurites of chick sensory neurons. Cell Motil Cytoskeleton 25:59–72. doi:10.1002/cm.970250108
El-Husseini AE-D, Craven SE, Brock SC, Bredt DS (2001) Polarized targeting of peripheral membrane proteins in neurons. J Biol Chem 276:44984–44992
Falke E, Nissanov J, Mitchell TW et al (2003) Subicular dendritic arborization in Alzheimer’s disease correlates with neurofibrillary tangle density. Am J Pathol 163:1615–1621. doi:10.1016/S0002-9440(10)63518-3
Fifková E, Delay RJ (1982) Cytoplasmic actin in neuronal processes as a possible mediator of synaptic plasticity. J Cell Biol 95:345–350
Fischer M, Kaech S, Knutti D, Matus A (1998) Rapid actin-based plasticity in dendritic spines. Neuron 20:847–854
Fleming IN, Elliott CM, Buchanan FG et al (1999) Ca2+/calmodulin-dependent protein kinase II regulates Tiam1 by reversible protein phosphorylation. J Biol Chem 274:12753–12758
Flood DG (1991) Region-specific stability of dendritic extent in normal human aging and regression in Alzheimer’s disease. II. Subiculum. Brain Res 540:83–95
Flood DG, Buell SJ, Horwitz GJ, Coleman PD (1987a) Dendritic extent in human dentate gyrus granule cells in normal aging and senile dementia. Brain Res 402:205–216
Flood DG, Guarnaccia M, Coleman PD (1987b) Dendritic extent in human CA2-3 hippocampal pyramidal neurons in normal aging and senile dementia. Brain Res 409:88–96
Freir DB, Nicoll AJ, Klyubin I et al (2011) Interaction between prion protein and toxic amyloid β assemblies can be therapeutically targeted at multiple sites. Nat Commun 2:336. doi:10.1038/ncomms1341
Ganeshina O, Berry RW, Petralia RS et al (2004a) Synapses with a segmented, completely partitioned postsynaptic density express more AMPA receptors than other axospinous synaptic junctions. Neuroscience 125:615–623. doi:10.1016/j.neuroscience.2004.02.025
Ganeshina O, Berry RW, Petralia RS et al (2004b) Differences in the expression of AMPA and NMDA receptors between axospinous perforated and nonperforated synapses are related to the configuration and size of postsynaptic densities. J Comp Neurol 468:86–95. doi:10.1002/cne.10950
Gazzaley A, D’Esposito M (2007) Top-down modulation and normal aging. Ann N Y Acad Sci 1097:67–83. doi:10.1196/annals.1379.010
Geinisman Y, de Toledo-Morrell L, Morrell F (1986) Loss of perforated synapses in the dentate gyrus: morphological substrate of memory deficit in aged rats. Proc Natl Acad Sci U S A 83:3027–3031
Geinisman Y, deToledo-Morrell L, Morrell F et al (1993) Structural synaptic correlate of long-term potentiation: formation of axospinous synapses with multiple, completely partitioned transmission zones. Hippocampus 3:435–445. doi:10.1002/hipo.450030405
Geinisman Y, Ganeshina O, Yoshida R et al (2004) Aging, spatial learning, and total synapse number in the rat CA1 stratum radiatum. Neurobiol Aging 25:407–416. doi:10.1016/j.neurobiolaging.2003.12.001
Giannakopoulos P, Herrmann FR, Bussière T et al (2003) Tangle and neuron numbers, but not amyloid load, predict cognitive status in Alzheimer’s disease. Neurology 60:1495–1500
Gong Y, Chang L, Viola KL et al (2003) Alzheimer’s disease-affected brain: presence of oligomeric A beta ligands (ADDLs) suggests a molecular basis for reversible memory loss. Proc Natl Acad Sci U S A 100:10417–10422. doi:10.1073/pnas.1834302100
Gorski JA, Zeiler SR, Tamowski S, Jones KR (2003) Brain-derived neurotrophic factor is required for the maintenance of cortical dendrites. J Neurosci 23:6856–6865
Gourley SL, Kedves AT, Olausson P, Taylor JR (2009a) A history of corticosterone exposure regulates fear extinction and cortical NR2B, GluR2/3, and BDNF. Neuropsychopharm: Off Publ Am Coll Neuropsychopharm 34:707–716. doi:10.1038/npp.2008.123
Gourley SL, Koleske AJ, Taylor JR (2009b) Loss of dendrite stabilization by the Abl-related gene (Arg) kinase regulates behavioral flexibility and sensitivity to cocaine. Proc Natl Acad Sci U S A 106:16859–16864. doi:10.1073/pnas.0902286106
Gourley SL, Olevska A, Warren MS et al (2012) Arg kinase regulates prefrontal dendritic spine refinement and cocaine-induced plasticity. J Neurosci 32:2314–2323. doi:10.1523/JNEUROSCI.2730-11.2012
Govek EE, Newey SE, Van Aelst L (2005) The role of the Rho GTPases in neuronal development. Genes Dev 19:1–49. doi:10.1101/gad.1256405
Graber S, Maiti S, Halpain S (2004) Cathepsin B-like proteolysis and MARCKS degradation in sub-lethal NMDA-induced collapse of dendritic spines. Neuropharmacology 47:706–713. doi:10.1016/j.neuropharm.2004.08.004
Grintsevich EE, Reisler E (2014) Drebrin inhibits cofilin-induced severing of F-actin. Cytoskeleton (Hoboken) 71:472–483. doi:10.1002/cm.21184
Gu J, Firestein BL, Zheng JQ (2008) Microtubules in dendritic spine development. J Neurosci 28:12120–12124. doi:10.1523/JNEUROSCI.2509-08.2008
Haass C, Selkoe DJ (2007) Soluble protein oligomers in neurodegeneration: lessons from the Alzheimer’s amyloid beta-peptide. Nat Rev Mol Cell Biol 8:101–112. doi:10.1038/nrm2101
Haeckel A, Ahuja R, Gundelfinger ED et al (2008) The actin-binding protein Abp1 controls dendritic spine morphology and is important for spine head and synapse formation. J Neurosci 28:10031–10044. doi:10.1523/JNEUROSCI.0336-08.2008
Halpain S, Hipolito A, Saffer L (1998) Regulation of F-actin stability in dendritic spines by glutamate receptors and calcineurin. J Neurosci 18:9835–9844
Hanks SD, Flood DG (1991) Region-specific stability of dendritic extent in normal human aging and regression in Alzheimer’s disease. I. CA1 of hippocampus. Brain Res 540:63–82
Hara Y, Park CS, Janssen WGM et al (2011) Synaptic characteristics of dentate gyrus axonal boutons and their relationships with aging, menopause, and memory in female rhesus monkeys. J Neurosci 31:7737–7744. doi:10.1523/JNEUROSCI.0822-11.2011
Harada A, Teng J, Takei Y et al (2002) MAP2 is required for dendrite elongation, PKA anchoring in dendrites, and proper PKA signal transduction. J Cell Biol 158:541–549. doi:10.1083/jcb.200110134
Harigaya Y, Shoji M, Shirao T, Hirai S (1996) Disappearance of actin-binding protein, drebrin, from hippocampal synapses in Alzheimer’s disease. J Neurosci Res 43:87–92. doi:10.1002/jnr.490430111
Harris KM (1999) Structure, development, and plasticity of dendritic spines. Curr Opin Neurobiol 9:343–348
Harris KM, Kater SB (1994) Dendritic spines: cellular specializations imparting both stability and flexibility to synaptic function. Annu Rev Neurosci 17:341–371. doi:10.1146/annurev.ne.17.030194.002013
Hartmann M, Heumann R, Lessmann V (2001) Synaptic secretion of BDNF after high-frequency stimulation of glutamatergic synapses. EMBO J 20:5887–5897. doi:10.1093/emboj/20.21.5887
Hasbani MJ, Hyrc KL, Faddis BT et al (1998) Distinct roles for sodium, chloride, and calcium in excitotoxic dendritic injury and recovery. Exp Neurol 154:241–258. doi:10.1006/exnr.1998.6929
Hasbani MJ, Schlief ML, Fisher DA, Goldberg MP (2001) Dendritic spines lost during glutamate receptor activation reemerge at original sites of synaptic contact. J Neurosci 21:2393–2403
Hayashi K, Shirao T (1999) Change in the shape of dendritic spines caused by overexpression of drebrin in cultured cortical neurons. J Neurosci 19:3918–3925
Hazai D, Szudoczki R, Ding J et al (2013) Ultrastructural abnormalities in CA1 hippocampus caused by deletion of the actin regulator WAVE-1. PLoS One 8:e75248. doi:10.1371/journal.pone.0075248
Hering H, Sheng M (2003) Activity-dependent redistribution and essential role of cortactin in dendritic spine morphogenesis. J Neurosci 23:11759–11769
Hirokawa N, Takemura R (2005) Molecular motors and mechanisms of directional transport in neurons. Nat Rev Neurosci 6:201–214
Holtmaat AJGD, Trachtenberg JT, Wilbrecht L et al (2005) Transient and persistent dendritic spines in the neocortex in vivo. Neuron 45:279–291. doi:10.1016/j.neuron.2005.01.003
Holtmaat A, Wilbrecht L, Knott GW et al (2006) Experience-dependent and cell-type-specific spine growth in the neocortex. Nature 441:979–983. doi:10.1038/nature04783
Honkura N, Matsuzaki M, Noguchi J et al (2008) The subspine organization of actin fibers regulates the structure and plasticity of dendritic spines. Neuron 57:719–729. doi:10.1016/j.neuron.2008.01.013
Hoover BR, Reed MN, Su J et al (2010) Tau mislocalization to dendritic spines mediates synaptic dysfunction independently of neurodegeneration. Neuron 68:1067–1081. doi:10.1016/j.neuron.2010.11.030
Hotulainen P, Hoogenraad CC (2010) Actin in dendritic spines: connecting dynamics to function. J Cell Biol 189:619–629. doi:10.1083/jcb.201003008
Hu B, Nikolakopoulou AM, Cohen-Cory S (2005) BDNF stabilizes synapses and maintains the structural complexity of optic axons in vivo. Development 132:4285–4298. doi:10.1242/dev.02017
Hu X, Viesselmann C, Nam S et al (2008) Activity-dependent dynamic microtubule invasion of dendritic spines. J Neurosci 28:13094–13105. doi:10.1523/JNEUROSCI.3074-08.2008
Hu X, Ballo L, Pietila L et al (2011) BDNF-induced increase of PSD-95 in dendritic spines requires dynamic microtubule invasions. J Neurosci 31:15597–15603. doi:10.1523/JNEUROSCI.2445-11.2011
Huber G, Matus A (1984) Differences in the cellular distributions of two microtubule-associated proteins, MAP1 and MAP2, in rat brain. J Neurosci 4:151–160
Huttenlocher PR (1979) Synaptic density in human frontal cortex – developmental changes and effects of aging. Brain Res 163:195–205
Huttenlocher PR (1990) Morphometric study of human cerebral cortex development. Neuropsychologia 28:517–527
Iki J, Inoue A, Bito H, Okabe S (2005) Bi-directional regulation of postsynaptic cortactin distribution by BDNF and NMDA receptor activity. Eur J Neurosci 22:2985–2994. doi:10.1111/j.1460-9568.2005.04510.x
Ingelsson M, Fukumoto H, Newell KL et al (2004) Early Abeta accumulation and progressive synaptic loss, gliosis, and tangle formation in AD brain. Neurology 62:925–931
Ishikawa R, Hayashi K, Shirao T et al (1994) Drebrin, a development-associated brain protein from rat embryo, causes the dissociation of tropomyosin from actin filaments. J Biol Chem 269:29928–29933
Ishizuka Y, Shimizu H, Takagi E et al (2014) Histone deacetylase mediates the decrease in drebrin cluster density induced by amyloid beta oligomers. Neurochem Int 76:114–121. doi:10.1016/j.neuint.2014.07.005
Ittner LM, Ke YD, Delerue F et al (2010) Dendritic function of tau mediates amyloid-beta toxicity in Alzheimer’s disease mouse models. Cell 142:387–397. doi:10.1016/j.cell.2010.06.036
Izeddin I, Specht CG, Lelek M et al (2011) Super-resolution dynamic imaging of dendritic spines using a low-affinity photoconvertible actin probe. PLoS One 6:e15611. doi:10.1371/journal.pone.0015611
Jaworski J, Kapitein LC, Gouveia SM et al (2009) Dynamic microtubules regulate dendritic spine morphology and synaptic plasticity. Neuron 61:85–100. doi:10.1016/j.neuron.2008.11.013
Jones WH, Thomas DB (1962) Changes in the dendritic organization of neurons in the cerebral cortex following deafferentation. J Anat 96:375–381
Jung Y, Mulholland PJ, Wiseman SL et al (2013) Constitutive knockout of the membrane cytoskeleton protein beta adducin decreases mushroom spine density in the nucleus accumbens but does not prevent spine remodeling in response to cocaine. Eur J Neurosci 37:1–9. doi:10.1111/ejn.12037
Kalia LV, Kalia SK, Salter MW (2008) NMDA receptors in clinical neurology: excitatory times ahead. Lancet Neurol 7:742–755. doi:10.1016/S1474-4422(08)70165-0
Kanai Y, Dohmae N, Hirokawa N (2004) Kinesin transports RNA: isolation and characterization of an RNA-transporting granule. Neuron 43:513–525
Kang MG, Guo Y, Huganir RL (2009) AMPA receptor and GEF-H1/Lfc complex regulates dendritic spine development through RhoA signaling cascade. Proc Natl Acad Sci U S A 106:3549–3554. doi:10.1073/pnas.0812861106
Kang HJ, Voleti B, Hajszan T et al (2012) Decreased expression of synapse-related genes and loss of synapses in major depressive disorder. Nat Med 18:1413–1417. doi:10.1038/nm.2886
Kapitein LC, Yau KW, Gouveia SM et al (2011) NMDA receptor activation suppresses microtubule growth and spine entry. J Neurosci 31:8194–8209. doi:10.1523/JNEUROSCI.6215-10.2011
Kerrisk ME, Greer CA, Koleske AJ (2013) Integrin alpha3 is required for late postnatal stability of dendrite arbors, dendritic spines and synapses, and mouse behavior. J Neurosci 33:6742–6752. doi:10.1523/JNEUROSCI.0528-13.2013
Kim Y, Sung JY, Ceglia I et al (2006) Phosphorylation of WAVE1 regulates actin polymerization and dendritic spine morphology. Nature 442:814–817. doi:10.1038/nature04976
Kim IH, Rácz B, Wang H et al (2013a) Disruption of Arp2/3 results in asymmetric structural plasticity of dendritic spines and progressive synaptic and behavioral abnormalities. J Neurosci 33:6081–6092. doi:10.1523/JNEUROSCI.0035-13.2013
Kim T, Vidal GS, Djurisic M et al (2013b) Human LilrB2 is a β-amyloid receptor and its murine homolog PirB regulates synaptic plasticity in an Alzheimer’s model. Science 341:1399–1404. doi:10.1126/science.1242077
Kitaoka Y, Kitaoka Y, Kumai T et al (2004) Involvement of RhoA and possible neuroprotective effect of fasudil, a Rho kinase inhibitor, in NMDA-induced neurotoxicity in the rat retina. Brain Res 1018:111–118. doi:10.1016/j.brainres.2004.05.070
Klein R (2009) Bidirectional modulation of synaptic functions by Eph/ephrin signaling. Nat Neurosci 12:15–20
Knudsen KA, Soler AP, Johnson KR, Wheelock MJ (1995) Interaction of alpha-actinin with the cadherin/catenin cell-cell adhesion complex via alpha-catenin. J Cell Biol 130:67–77
Kohara K, Kitamura A, Morishima M, Tsumoto T (2001) Activity-dependent transfer of brain-derived neurotrophic factor to postsynaptic neurons. Science 291:2419–2423. doi:10.1126/science.1057415
Kojima M, Takei N, Numakawa T et al (2001) Biological characterization and optical imaging of brain-derived neurotrophic factor-green fluorescent protein suggest an activity-dependent local release of brain-derived neurotrophic factor in neurites of cultured hippocampal neurons. J Neurosci Res 64:1–10
Koleske AJ, Gifford AM, Scott ML et al (1998) Essential roles for the Abl and Arg tyrosine kinases in neurulation. Neuron 21:1259–1272
Kopeikina KJ, Hyman BT, Spires-Jones TL (2012) Soluble forms of tau are toxic in Alzheimer’s disease. Transl Neurosci 3:223–233. doi:10.2478/s13380-012-0032-y
Korobova F, Svitkina T (2010) Molecular architecture of synaptic actin cytoskeleton in hippocampal neurons reveals a mechanism of dendritic spine morphogenesis. Mol Biol Cell 21:165–176. doi:10.1091/mbc.E09-07-0596
Kreis P, Thévenot E, Rousseau V et al (2007) The p21-activated kinase 3 implicated in mental retardation regulates spine morphogenesis through a Cdc42-dependent pathway. J Biol Chem 282:21497–21506. doi:10.1074/jbc.M703298200
Kulkarni VA, Firestein BL (2012) The dendritic tree and brain disorders. Mol Cell Neurosci 50:10–20. doi:10.1016/j.mcn.2012.03.005
Kwan AC, Dombeck DA, Webb WW (2008) Polarized microtubule arrays in apical dendrites and axons. Proc Natl Acad Sci U S A 105:11370–11375
Kwon M, Fernández JR, Zegarek GF et al (2011) BDNF-promoted increases in proximal dendrites occur via CREB-dependent transcriptional regulation of cypin. J Neurosci 31:9735–9745. doi:10.1523/JNEUROSCI.6785-10.2011
Lacor PN, Buniel MC, Furlow PW et al (2007) Abeta oligomer-induced aberrations in synapse composition, shape, and density provide a molecular basis for loss of connectivity in Alzheimer’s disease. J Neurosci 27:796–807. doi:10.1523/JNEUROSCI.3501-06.2007
Lai K-O, Wong ASL, Cheung M-C et al (2012) TrkB phosphorylation by Cdk5 is required for activity-dependent structural plasticity and spatial memory. Nat Neurosci 15:1506–1515. doi:10.1038/nn.3237
Larson M, Sherman MA, Amar F et al (2012) The complex PrP(c)-Fyn couples human oligomeric Aβ with pathological tau changes in Alzheimer’s disease. J Neurosci 32:16857–71a. doi:10.1523/JNEUROSCI.1858-12.2012
Lemieux M, Labrecque S, Tardif C et al (2012) Translocation of CaMKII to dendritic microtubules supports the plasticity of local synapses. J Cell Biol 198:1055–1073. doi:10.1083/jcb.201202058
Lin Y-C, Yeckel MF, Koleske AJ (2013) Abl2/Arg controls dendritic spine and dendrite arbor stability via distinct cytoskeletal control pathways. J Neurosci 33:1846–1857. doi:10.1523/JNEUROSCI.4284-12.2013
MacGrath SM, Koleske AJ (2012a) Arg/Abl2 modulates the affinity and stoichiometry of binding of cortactin to f-actin. Biochemistry 51:6644–6653. doi:10.1021/bi300722t
MacGrath SM, Koleske AJ (2012b) Cortactin in cell migration and cancer at a glance. J Cell Sci 125:1621–1626. doi:10.1242/jcs.093781
Machesky LM, Insall RH (1998) Scar1 and the related Wiskott-Aldrich syndrome protein, WASP, regulate the actin cytoskeleton through the Arp2/3 complex. Curr Biol 8:1347–1356
Machesky LM, Mullins RD, Higgs HN et al (1999) Scar, a WASp-related protein, activates nucleation of actin filaments by the Arp2/3 complex. Proc Natl Acad Sci U S A 96:3739–3744
Majewska AK, Newton JR, Sur M (2006) Remodeling of synaptic structure in sensory cortical areas in vivo. J Neurosci 26:3021–3029. doi:10.1523/JNEUROSCI.4454-05.2006
Markus EJ, Petit TL (1987) Neocortical synaptogenesis, aging, and behavior: lifespan development in the motor-sensory system of the rat. Exp Neurol 96:262–278
Marshak S, Nikolakopoulou AM, Dirks R et al (2007) Cell-autonomous TrkB signaling in presynaptic retinal ganglion cells mediates axon arbor growth and synapse maturation during the establishment of retinotectal synaptic connectivity. J Neurosci 27:2444–2456. doi:10.1523/JNEUROSCI.4434-06.2007
Matsuoka Y, Li X, Bennett V (1998) Adducin is an in vivo substrate for protein kinase C: phosphorylation in the MARCKS-related domain inhibits activity in promoting spectrin-actin complexes and occurs in many cells, including dendritic spines of neurons. J Cell Biol 142:485–497
Matsuoka Y, Li X, Bennett V (2000) Adducin: structure, function and regulation. Cell Mol Life Sci 57:884–895
Matsuzaki M, Honkura N, Ellis-Davies GCR, Kasai H (2004) Structural basis of long-term potentiation in single dendritic spines. Nature 429:761–766. doi:10.1038/nature02617
Matthews MR, Powell TP (1962) Some observations on transneuronal cell degeneration in the olfactory bulb of the rabbit. J Anat 96:89–102
Matus A, Ackermann M, Pehling G et al (1982) High actin concentrations in brain dendritic spines and postsynaptic densities. Proc Natl Acad Sci U S A 79:7590–7594
Mikati MA, Grintsevich EE, Reisler E (2013) Drebrin-induced stabilization of actin filaments. J Biol Chem 288:19926–19938. doi:10.1074/jbc.M113.472647
Mooren OL, Galletta BJ, Cooper JA (2012) Roles for actin assembly in endocytosis. Annu Rev Biochem 81:661–686. doi:10.1146/annurev-biochem-060910-094416
Moresco EM, Scheetz AJ, Bornmann WG et al (2003) Abl family nonreceptor tyrosine kinases modulate short-term synaptic plasticity. J Neurophysiol 89:1678–1687. doi:10.1152/jn.0892.2002
Morrison JH, Baxter MG (2012) The ageing cortical synapse: hallmarks and implications for cognitive decline. Nat Rev Neurosci 13:240–250. doi:10.1038/nrn3200
Morrison JH, Hof PR (1997) Life and death of neurons in the aging brain. Science 278:412–419
Mostany R, Portera-Cailliau C (2011) Absence of large-scale dendritic plasticity of layer 5 pyramidal neurons in peri-infarct cortex. J Neurosci 31:1734–1738. doi:10.1523/JNEUROSCI.4386-10.2011
Mostany R, Anstey JE, Crump KL et al (2013) Altered synaptic dynamics during normal brain aging. J Neurosci 33:4094–4104. doi:10.1523/JNEUROSCI.4825-12.2013
Murakoshi H, Wang H, Yasuda R (2011) Local, persistent activation of Rho GTPases during plasticity of single dendritic spines. Nature 472:100–104. doi:10.1038/nature09823
Murthy AS, Flavin M (1983) Microtubule assembly using the microtubule-associated protein MAP-2 prepared in defined states of phosphorylation with protein kinase and phosphatase. Eur J Biochem 137:37–46
Naisbitt S, Kim E, Tu JC et al (1999) Shank, a novel family of postsynaptic density proteins that binds to the NMDA receptor/PSD-95/GKAP complex and cortactin. Neuron 23:569–582
Nakayama AY, Luo L (2000) Intracellular signaling pathways that regulate dendritic spine morphogenesis. Hippocampus 10:582–586
Nibuya M, Takahashi M, Russell DS, Duman RS (1999) Repeated stress increases catalytic TrkB mRNA in rat hippocampus. Neurosci Lett 267:81–84
Nicholson DA, Yoshida R, Berry RW et al (2004) Reduction in size of perforated postsynaptic densities in hippocampal axospinous synapses and age-related spatial learning impairments. J Neurosci 24:7648–7653. doi:10.1523/JNEUROSCI.1725-04.2004
Niell CM, Meyer MP, Smith SJ (2004) In vivo imaging of synapse formation on a growing dendritic arbor. Nat Neurosci 7:254–260. doi:10.1038/nn1191
Okabe S, Hirokawa N (1990) Turnover of fluorescently labelled tubulin and actin in the axon. Nature 343:479–482. doi:10.1038/343479a0
Okamoto K-I, Nagai T, Miyawaki A, Hayashi Y (2004) Rapid and persistent modulation of actin dynamics regulates postsynaptic reorganization underlying bidirectional plasticity. Nat Neurosci 7:1104–1112. doi:10.1038/nn1311
Oray S, Majewska A, Sur M (2004) Dendritic spine dynamics are regulated by monocular deprivation and extracellular matrix degradation. Neuron 44:1021–1030. doi:10.1016/j.neuron.2004.12.001
Peris L, Wagenbach M, Lafanechère L et al (2009) Motor-dependent microtubule disassembly driven by tubulin tyrosination. J Cell Biol 185:1159–1166. doi:10.1083/jcb.200902142
Peters A, Sethares C, Moss MB (1998) The effects of aging on layer 1 in area 46 of prefrontal cortex in the rhesus monkey. Cereb Cortex 8:671–684
Peters A, Sethares C, Luebke JI (2008) Synapses are lost during aging in the primate prefrontal cortex. Neuroscience 152:970–981. doi:10.1016/j.neuroscience.2007.07.014
Pinkstaff JK, Detterich J, Lynch G, Gall C (1999) Integrin subunit gene expression is regionally differentiated in adult brain. J Neurosci 19:1541–1556
Pollard TD, Borisy GG (2003) Cellular motility driven by assembly and disassembly of actin filaments. Cell 112:453–465
Pozueta J, Lefort R, Shelanski ML (2013) Synaptic changes in Alzheimer’s disease and its models. Neuroscience 251:51–65. doi:10.1016/j.neuroscience.2012.05.050
Rácz B, Weinberg RJ (2004) The subcellular organization of cortactin in hippocampus. J Neurosci 24:10310–10317. doi:10.1523/JNEUROSCI.2080-04.2004
Rácz B, Weinberg RJ (2006) Spatial organization of cofilin in dendritic spines. Neuroscience 138:447–456. doi:10.1016/j.neuroscience.2005.11.025
Rácz B, Weinberg RJ (2008) Organization of the Arp2/3 complex in hippocampal spines. J Neurosci 28:5654–5659. doi:10.1523/JNEUROSCI.0756-08.2008
Rácz B, Weinberg RJ (2013) Microdomains in forebrain spines: an ultrastructural perspective. Mol Neurobiol 47:77–89
Rajan I, Witte S, Cline HT (1999) NMDA receptor activity stabilizes presynaptic retinotectal axons and postsynaptic optic tectal cell dendrites in vivo. J Neurobiol 38:357–368. doi:10.1002/(SICI)1097-4695(19990215)38:3<357::AID-NEU5>3.0.CO;2-#
Rakic P, Bourgeois JP, Eckenhoff MF et al (1986) Concurrent overproduction of synapses in diverse regions of the primate cerebral cortex. Science 232:232–235
Rakic P, Bourgeois JP, Goldman-Rakic PS (1994) Synaptic development of the cerebral cortex: implications for learning, memory, and mental illness. Prog Brain Res 102:227–243. doi:10.1016/S0079-6123(08)60543-9
Ray LB, Sturgill TW (1987) Rapid stimulation by insulin of a serine/threonine kinase in 3T3-L1 adipocytes that phosphorylates microtubule-associated protein 2 in vitro. Proc Natl Acad Sci U S A 84:1502–1506
Rex CS, Lin C-Y, Kramár EA et al (2007) Brain-derived neurotrophic factor promotes long-term potentiation-related cytoskeletal changes in adult hippocampus. J Neurosci 27:3017–3029. doi:10.1523/JNEUROSCI.4037-06.2007
Sánchez C, Díaz-Nido J, Avila J (2000) Phosphorylation of microtubule-associated protein 2 (MAP2) and its relevance for the regulation of the neuronal cytoskeleton function. Prog Neurobiol 61:133–168
Sasaki S, Stevens JK, Bodick N (1983) Serial reconstruction of microtubular arrays within dendrites of the cat retinal ganglion cell: the cytoskeleton of a vertebrate dendrite. Brain Res 259:193–206
Sasaki Y, Hayashi K, Shirao T et al (1996) Inhibition by drebrin of the actin-bundling activity of brain fascin, a protein localized in filopodia of growth cones. J Neurochem 66:980–988
Schmidt HD, Duman RS (2007) The role of neurotrophic factors in adult hippocampal neurogenesis, antidepressant treatments and animal models of depressive-like behavior. Behav Pharmacol 18:391–418. doi:10.1097/FBP.0b013e3282ee2aa8
Schulman H (1984) Phosphorylation of microtubule-associated proteins by a Ca2+/calmodulin-dependent protein kinase. J Cell Biol 99:11–19
Seese RR, Babayan AH, Katz AM et al (2012) LTP induction translocates cortactin at distant synapses in wild-type but not Fmr1 knock-out mice. J Neurosci 32:7403–7413. doi:10.1523/JNEUROSCI.0968-12.2012
Setou M, Nakagawa T, Seog DH, Hirokawa N (2000) Kinesin superfamily motor protein KIF17 and mLin-10 in NMDA receptor-containing vesicle transport. Science 288:1796–1802
Setou M, Seog DH, Tanaka Y et al (2002) Glutamate-receptor-interacting protein GRIP1 directly steers kinesin to dendrites. Nature 417:83–87
Sfakianos MK, Eisman A, Gourley SL et al (2007) Inhibition of Rho via Arg and p190RhoGAP in the postnatal mouse hippocampus regulates dendritic spine maturation, synapse and dendrite stability, and behavior. J Neurosci 27:10982–10992. doi:10.1523/JNEUROSCI.0793-07.2007
Shankar GM, Bloodgood BL, Townsend M et al (2007) Natural oligomers of the Alzheimer amyloid-beta protein induce reversible synapse loss by modulating an NMDA-type glutamate receptor-dependent signaling pathway. J Neurosci 27:2866–2875. doi:10.1523/JNEUROSCI.4970-06.2007
Shi Y, Pontrello CG, DeFea KA et al (2009) Focal adhesion kinase acts downstream of EphB receptors to maintain mature dendritic spines by regulating cofilin activity. J Neurosci 29:8129–8142
Shirao T, Kojima N, Kato Y, Obata K (1988) Molecular cloning of a cDNA for the developmentally regulated brain protein, drebrin. Brain Res 464:71–74
Simpson MA, Bradley WD, Harburger D et al (2015) Direct interactions with the integrin β 1 cytoplasmic tail activate the Abl2/Arg kinase. J Biol Chem 290:8360–8372. doi:10.1074/jbc.M115.638874
Smith MA, Makino S, Kvetnansky R, Post RM (1995) Stress and glucocorticoids affect the expression of brain-derived neurotrophic factor and neurotrophin-3 mRNAs in the hippocampus. J Neurosci 15:1768–1777
Smith TD, Adams MM, Gallagher M et al (2000) Circuit-specific alterations in hippocampal synaptophysin immunoreactivity predict spatial learning impairment in aged rats. J Neurosci 20:6587–6593
Soderling SH, Guire ES, Kaech S et al (2007) A WAVE-1 and WRP signaling complex regulates spine density, synaptic plasticity, and memory. J Neurosci 27:355–365. doi:10.1523/JNEUROSCI.3209-06.2006
Star EN, Kwiatkowski DJ, Murthy VN (2002) Rapid turnover of actin in dendritic spines and its regulation by activity. Nat Neurosci 5:239–246. doi:10.1038/nn811
Sudo H, Baas PW (2010) Acetylation of microtubules influences their sensitivity to severing by katanin in neurons and fibroblasts. J Neurosci 30:7215–7226. doi:10.1523/JNEUROSCI.0048-10.2010
Szebenyi G, Bollati F, Bisbal M et al (2005) Activity-driven dendritic remodeling requires microtubule-associated protein 1A. Curr Biol 15:1820–1826. doi:10.1016/j.cub.2005.08.069
Takahashi H, Sekino Y, Tanaka S et al (2003) Drebrin-dependent actin clustering in dendritic filopodia governs synaptic targeting of postsynaptic density-95 and dendritic spine morphogenesis. J Neurosci 23:6586–6595
Takemoto-Kimura S, Suzuki K, Kamijo S et al (2010) Differential roles for CaM kinases in mediating excitation-morphogenesis coupling during formation and maturation of neuronal circuits. Eur J Neurosci 32:224–230. doi:10.1111/j.1460-9568.2010.07353.x
Tanokashira D, Morita T, Hayashi K et al (2012) Glucocorticoid suppresses dendritic spine development mediated by down-regulation of caldesmon expression. J Neurosci 32:14583–14591. doi:10.1523/JNEUROSCI.2380-12.2012
Tashiro A, Yuste R (2003) Structure and molecular organization of dendritic spines. Histol Histopathol 18:617–634
Tashiro A, Minden A, Yuste R (2000) Regulation of dendritic spine morphology by the rho family of small GTPases: antagonistic roles of Rac and Rho. Cereb Cortex 10:927–938
Tatavarty V, Kim E-J, Rodionov V, Yu J (2009) Investigating sub-spine actin dynamics in rat hippocampal neurons with super-resolution optical imaging. PLoS One 4:e7724. doi:10.1371/journal.pone.0007724
Teng J, Takei Y, Harada A et al (2001) Synergistic effects of MAP2 and MAP1B knockout in neuronal migration, dendritic outgrowth, and microtubule organization. J Cell Biol 155:65–76. doi:10.1083/jcb.200106025
Terry RD, Masliah E, Salmon DP et al (1991) Physical basis of cognitive alterations in Alzheimer’s disease: synapse loss is the major correlate of cognitive impairment. Ann Neurol 30:572–580. doi:10.1002/ana.410300410
Tomar A, Schlaepfer DD (2009) Focal adhesion kinase: switching between GAPs and GEFs in the regulation of cell motility. Curr Opin Cell Biol 21:676–683. doi:10.1016/j.ceb.2009.05.006. PMID: 19525103
Tomar A, Lawson C, Ghassemian M, Schlaepfer DD (2012) Cortactin as a target for FAK in the regulation of focal adhesion dynamics. PLoS One 7:e44041. doi:10.1371/journal.pone.0044041. PMID: 22952866
Toni N, Buchs PA, Nikonenko I et al (2001) Remodeling of synaptic membranes after induction of long-term potentiation. J Neurosci 21:6245–6251
Tønnesen J, Katona G, Rózsa B, Nägerl UV (2014) Spine neck plasticity regulates compartmentalization of synapses. Nat Neurosci 17:678–685. doi:10.1038/nn.3682
Trachtenberg JT, Chen BE, Knott GW et al (2002) Long-term in vivo imaging of experience-dependent synaptic plasticity in adult cortex. Nature 420:788–794. doi:10.1038/nature01273
Tseng CY, Firestein BL (2011) The role of PSD-95 and cypin in morphological changes in dendrites following sublethal NMDA exposure. J Neurosci 31:15468–15480. doi:10.1523/JNEUROSCI.2442-11.2011
Um JW, Nygaard HB, Heiss JK et al (2012) Alzheimer amyloid-β oligomer bound to postsynaptic prion protein activates Fyn to impair neurons. Nat Neurosci 15:1227–1235. doi:10.1038/nn.3178
Uruno T, Liu J, Zhang P et al (2001) Activation of Arp2/3 complex-mediated actin polymerization by cortactin. Nat Cell Biol 3:259–266. doi:10.1038/35060051
Uylings HBM, De Brabander JM (2002) Neuronal changes in normal human aging and Alzheimer’s disease. Brain Cogn 49:268–276
Vaillant AR, Zanassi P, Walsh GS et al (2002) Signaling mechanisms underlying reversible, activity-dependent dendrite formation. Neuron 34:985–998
Vaughn JE (1989) Fine structure of synaptogenesis in the vertebrate central nervous system. Synapse 3:255–285. doi:10.1002/syn.890030312
von Bohlen und Halbach O, Minichiello L, Unsicker K (2003) Haploinsufficiency in trkB and/or trkC neurotrophin receptors causes structural alterations in the aged hippocampus and amygdala. Eur J Neurosci 18:2319–2325
Wang HY, Lee DH, Davis CB, Shank RP (2000) Amyloid peptide Abeta(1–42) binds selectively and with picomolar affinity to alpha7 nicotinic acetylcholine receptors. J Neurochem 75:1155–1161
Wang Y, Miller AL, Mooseker MS, Koleske AJ (2001) The Abl-related gene (Arg) nonreceptor tyrosine kinase uses two F-actin-binding domains to bundle F-actin. Proc Natl Acad Sci U S A 98:14865–14870
Wang Y, Shibasaki F, Mizuno K (2005) Calcium signal-induced cofilin dephosphorylation is mediated by Slingshot via calcineurin. J Biol Chem 280:12683–12689. doi:10.1074/jbc.M411494200
Warren MS, Bradley WD, Gourley SL et al (2012) Integrin beta1 signals through Arg to regulate postnatal dendritic arborization, synapse density, and behavior. J Neurosci 32:2824–2834. doi:10.1523/JNEUROSCI.3942-11.2012
Weaver AM, Karginov AV, Kinley AW et al (2001) Cortactin promotes and stabilizes Arp2/3-induced actin filament network formation. Curr Biol 11:370–374
Weed SA, Karginov AV, Schafer DA et al (2000) Cortactin localization to sites of actin assembly in lamellipodia requires interactions with F-actin and the Arp2/3 complex. J Cell Biol 151:29–40
West MJ, Coleman PD, Flood DG, Troncoso JC (1994) Differences in the pattern of hippocampal neuronal loss in normal ageing and Alzheimer’s disease. Lancet 344:769–772
Wiesel TN, Hubel DH (1963) Effects of visual deprivation on morphology and physiology of cells in the cats lateral geniculate body. J Neurophysiol 26:978–993
Wong WT, Wong RO (2000) Rapid dendritic movements during synapse formation and rearrangement. Curr Opin Neurobiol 10:118–124. doi:10.1016/S0959-4388(99)00059-8
Wong WT, Faulkner-Jones BE, Sanes JR, Wong RO (2000) Rapid dendritic remodeling in the developing retina: dependence on neurotransmission and reciprocal regulation by Rac and Rho. J Neurosci 20:5024–5036. doi:10.1016/0006-8993(76)90383-8
Wu GY, Cline HT (1998) Stabilization of dendritic arbor structure in vivo by CaMKII. Science 279:222–226
Wu GY, Zou DJ, Rajan I, Cline H (1999) Dendritic dynamics in vivo change during neuronal maturation. J Neurosci 19:4472–4483. doi:10.1016/S0896-6273(00)80471-7
Wu H-Y, Hudry E, Hashimoto T et al (2010) Amyloid beta induces the morphological neurodegenerative triad of spine loss, dendritic simplification, and neuritic dystrophies through calcineurin activation. J Neurosci 30:2636–2649. doi:10.1523/JNEUROSCI.4456-09.2010
Xie Z, Srivastava DP, Photowala H et al (2007) Kalirin-7 controls activity-dependent structural and functional plasticity of dendritic spines. Neuron 56:640–656. doi:10.1016/j.neuron.2007.10.005
Xing L, Yao X, Williams KR, Bassell GJ (2012) Negative regulation of RhoA translation and signaling by hnRNP-Q1 affects cellular morphogenesis. Mol Biol Cell 23:1500–1509. doi:10.1091/mbc.E11-10-0867
Xu B, Zang K, Ruff NL et al (2000) Cortical degeneration in the absence of neurotrophin signaling: dendritic retraction and neuronal loss after removal of the receptor TrkB. Neuron 26:233–245
Yamamoto H, Fukunaga K, Tanaka E, Miyamoto E (1983) Ca2+- and calmodulin-dependent phosphorylation of microtubule-associated protein 2 and tau factor, and inhibition of microtubule assembly. J Neurochem 41:1119–1125
Yamazaki H, Takahashi H, Aoki T, Shirao T (2001) Molecular cloning and dendritic localization of rat SH3P7. Eur J Neurosci 14:998–1008
Yang N, Higuchi O, Ohashi K et al (1998) Cofilin phosphorylation by LIM-kinase 1 and its role in Rac-mediated actin reorganization. Nature 393:809–812
Yang G, Pan F, Gan W-B (2009) Stably maintained dendritic spines are associated with lifelong memories. Nature 462:920–924. doi:10.1038/nature08577
Yasuda R, Harvey CD, Zhong H et al (2006) Supersensitive Ras activation in dendrites and spines revealed by two-photon fluorescence lifetime imaging. Nat Neurosci 9:283–291. doi:10.1038/nn1635
Young ME, Ohm DT, Dumitriu D et al (2014) Differential effects of aging on dendritic spines in visual cortex and prefrontal cortex of the rhesus monkey. Neuroscience 274:33–43. doi:10.1016/j.neuroscience.2014.05.008
Yu X, Wang G, Gilmore A et al (2013) Accelerated experience-dependent pruning of cortical synapses in ephrin-A2 knockout mice. Neuron 80:64–71. doi:10.1016/j.neuron.2013.07.014
Zempel H, Thies E, Mandelkow E, Mandelkow E-M (2010) Abeta oligomers cause localized Ca(2+) elevation, missorting of endogenous Tau into dendrites, Tau phosphorylation, and destruction of microtubules and spines. J Neurosci 30:11938–11950. doi:10.1523/JNEUROSCI.2357-10.2010
Zhou Q, Homma KJ, Poo M-M (2004) Shrinkage of dendritic spines associated with long-term depression of hippocampal synapses. Neuron 44:749–757. doi:10.1016/j.neuron.2004.11.011
Zuo Y, Lin A, Chang P, Gan W-B (2005a) Development of long-term dendritic spine stability in diverse regions of cerebral cortex. Neuron 46:181–189. doi:10.1016/j.neuron.2005.04.001
Zuo Y, Yang G, Kwon E, Gan WB (2005b) Long-term sensory deprivation prevents dendritic spine loss in primary somatosensory cortex. Nature 436:261–265. doi:10.1038/nature03715
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer Japan
About this chapter
Cite this chapter
Katrancha, S.M., Koleske, A.J. (2016). Dendrite Maintenance. In: Emoto, K., Wong, R., Huang, E., Hoogenraad, C. (eds) Dendrites. Springer, Tokyo. https://doi.org/10.1007/978-4-431-56050-0_14
Download citation
DOI: https://doi.org/10.1007/978-4-431-56050-0_14
Published:
Publisher Name: Springer, Tokyo
Print ISBN: 978-4-431-56048-7
Online ISBN: 978-4-431-56050-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)