Abstract
Beat-to-beat intervals of cardiac sinus rhythm are not constant but show complex and continuous fluctuations called heart rate variability (HRV). Because HRV disappears with cardiac denervation by complete autonomic blockades or cardiac transplantation, HRV is thought to originate from the brain and to transfer to the heart through the autonomic nervous system. HRV includes a plenty of information not only about autonomic neural cardiac regulations but also about health state and hazard that are captured by the brain. To extract information that meets with particular purposes, various methods have been developed for the analysis of HRV. This chapter explains the basic mechanisms generating HRV and introduces the purposes and corresponding methods for HRV analyses.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
1 What Is Heart Rate Variability?
1.1 Definitions
The rhythm of heart beat at rest is essentially regular, and irregularity of the rhythm is called arrhythmia. In fact, however, cardiac cycle length even under sinus rhythm shows continuous fluctuations, as indicated by the presence of term “physiological arrhythmia.” Respiratory sinus arrhythmia (RSA) is the representative among such physiological arrhythmias, and it has first been described by Ludwig [1] in 1847. Cardiac cycle length shows various physiological fluctuations ranging from circadian rhythm of heart rate to such changes in beat-to-beat intervals as RSA.
Cardiac cycle length and its physiological fluctuations are mediated by the neurohumoral factors, particularly autonomic nervous system, that regulates the rate of discharge of the sinus node. Under complete pharmacological autonomic blocking with sufficient doses of sympathetic and vagal blockades, the beat-to-beat fluctuations of cardiac cycle length disappear, and the heart ticks a regular rhythm like a metronome [2, 3]. This is also the case for transplanted heart that has no neural connections with the recipient [4]. These indicate that physiological fluctuations in beat-to-beat cardiac cycle length are mediated by the fluctuations in autonomic neural outflow to the sinus node.
In a narrow sense, the term heart rate variability (HRV) is defined as the amount of variability in beat-to-beat fluctuations of cardiac cycle length under normal sinus rhythm. The term is also used as almost the same meaning as physiological arrhythmia. In contrast, fluctuations in cardiac cycle length that are caused by ectopic rhythms and beats, conduction blocks, and sinus dysfunction are not included in HRV and should be excluded from the analysis of HRV. In a broad sense, however, cardiac cycle length fluctuations caused by a part of pathological arrhythmias have also been categorized as studies of HRV. For example, heart rate turbulence (HRT) that is known as a powerful predictor of mortality risk after acute myocardial infarction analyzes the cardiac cycle length fluctuations flowing ectopic beats observed during Holter ECG monitoring [5, 6]. The methods developed for HRV analysis have been used for ventricular response cycle length fluctuations during atrial fibrillation and have produced useful indices for both pathophysiologic understandings and clinical risk stratifications of this arrhythmia [7–11].
1.2 HRV and Autonomic Nervous Functions
Heart rate is regulated antagonistically by the sympathetic and vagal divisions of autonomic nervous system. Heart rate reflects the static balance between their activities to the sinus node, and HRV reflects the fluctuations in the balance. HRV, however, includes information that cannot be obtained from heart rate itself, i.e., pure vagal modulation of heart rate separated from sympathetic influence.
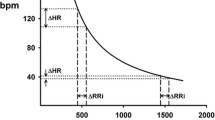
The principle of autonomic functional assessment by HRV may be compared to the estimation of driver’s manipulations of accelerator and brake pedals from the time series recording of changes in vehicle’s speed, where heart rate corresponds to vehicle’s speed, sympathetic activity to accelerator, and vagal activity to brakes. Different from automatic transmission vehicles that creep when the driver takes off both accelerator and brake pedals, heart rate increases to a value called “intrinsic heart rate” when both sympathetic and vagal activities are blocked. The intrinsic heart rate is higher in young people and declines with aging; the means are 107 bpm for 20-year, 101 bpm for 30-year, 90 bpm for 50-year, and 78 bpm for 70-year olds. This indicates that young people maintain their cardiac vagal activity at a certain level continuously during rest to keep their heart rate below the intrinsic heart rate.
During vehicles are moving, if you tap the accelerator rapidly, vehicle’s speed shows almost no changes. In contrast, if you tap the brake pedals, the speed changes faithfully with the manipulations. This is explained as the difference in frequency characteristic of transfer functions between two systems. Similar difference in frequency characteristic exists between sympathetic and vagal modulations of heart rate. Changes in sympathetic activity cause the effects through several phosphorylating enzymatic processes in the intracellular signal transduction mechanisms existing downstream of beta-adrenergic receptors. Consequently, the sympathetic modulation of heart rate cannot transfer fluctuations at frequencies >0.15 Hz (cycle length <6.7 s) [12]. While on the other hand, changes in vagal activity cause the effects simply by the conformation change of the membrane potassium channels with Ach. Consequently, the vagal modulation of heart rate can transfer fluctuations at frequency up to 1 Hz [12].
When a decrease was observed in the speed of a vehicle, there may be three possibilities that the driver (1) took his/her foot off accelerator, (2) pressed the brake pedal, or (3) both. If the deceleration was faster than a certain level, however, it is attributable only to the effect of (2). Similarly, when heart rate fluctuations at frequencies >0.15 Hz were observed, it is not explained by sympathetic modulation and it is attributable only to the effect of vagal modulation. These indicate that we can separate frequency components of heart rate fluctuations purely mediated by the vagal activities from those affected by sympathetic activities.
Methods for analyzing the fluctuations of time series data by resolving them into components by the difference in frequency are called power spectral analyses. Although there are several methods for this analysis, including fast Fourier transformation (FFT) [13], autoregressive (AR) model [14], and maximum entropy methods (MEM), the principle is the same as spectroscopic analysis by prisms that separate light into components with their colors (wave frequencies). Power spectral analyses resolve fluctuation into frequency components and quantify the power or amplitude of each component.
1.3 Purposes of HRV Analysis
The analysis of HRV is currently used for two major purposes: (1) assessment of cardiac autonomic functions and (2) risk stratification of patients with cardiac and other diseases.
The former includes the assessment of stress or arousal level, relaxation depth, and sleep quality as well as clinical evaluation for autonomic dysfunctions. For these purposes, short-term (5–10 min) ECG data that are recorded under standardized conditions (room temperature, body posture, time after food, alcohol and caffeine intake, exercise and smoking, and breathing frequency according to the circumstances) are used. HRV analyzed from short-term ECG data thus obtained is referred to as short-term HRV.
The risk stratification includes the prediction of mortality and adverse prognosis. For these purposes, long-term (24 h) ambulatory ECG data recorded under daily activities are used. HRV analyzed from long-term ECG data thus obtained is referred to as long-term HRV.
2 Mechanisms Generating HRV
2.1 Short-Term HRV
Power spectrum of short-term HRV at rest includes two major frequency components (Fig. 7.1): low-frequency (LF) component (0.04–0.15 Hz) and high-frequency (HF) component (>0.15 Hz) [2, 15–17]. This is a characteristic of HRV that is unlike vehicle’s speed indicating that HRV does not comprise only random fluctuations but includes frequency components with specific frequencies. The cutoff frequency (0.15 Hz) separating between two components has been defined from the frequency characteristic of transfer function for sympathetic heart rate modulation discussed above [12]. Although the frequency of LF component has been defined as 0.04–0.15 Hz, it usually appears between 0.06 and 0.11 Hz.
Power spectrum of short-term HRV and frequency ranges modulated by sympathetic and vagal heart rate controls. While cardiac vagal control system can modulate heart rate in the entire frequency band, cardiac sympathetic control system can modulate heart rate at frequencies <0.15 Hz. Consequently, while the LF component of HRV is mediated by both sympathetic and vagal nerves, the HF component is mediated purely by the vagus
2.1.1 HF Component of HRV
The HF component of HRV usually corresponds to RSA, and thus, its frequency is identical to breathing frequency (e.g., when breathing frequency is 15 cycle/min, the frequency of HF component is 15 cycle/60 s = 0.25 Hz). This means that when breathing frequency decreases below 9 cycle/min (0.15 Hz), HRV caused by RSA is detected as a part of LF component and physiologic HF component disappears; HF component under such conditions, if observed, should be interpreted as a different physiologic entity from RSA or an artifact.
The mechanisms generating RSA is discussed in Chap. 8 in detail. Briefly, RSA is mediated by the vagus originated from the nucleus ambiguus in the medulla oblongata that is modulated by the input from the respiratory center and generates vagal outflow fluctuating with respiration [18]. By this mechanism, vagal flow increases during expiration and decreases during inspiration, generating RSA. Although respiratory fluctuation exists also in sympathetic outflow, it does not transfer to HRV when the breathing frequency is above 0.15 Hz.
2.1.2 LF Component of HRV
The LF component of HRV is thought to be heart rate variation caused by Mayer wave [19] in blood pressure fluctuation through the arterial baroreceptor reflex mechanism (Fig. 7.2) [20, 21]. Mayer wave is a component of physiological arterial blood pressure fluctuation with a period around 10 s. It is also called the third-order variation of blood pressure and has been found by Cion in 1874 and described by Mayer in 1876 [19].
Arterial blood pressure and R-R interval of ECG in a healthy subject. Mayer wave is observed in the compressed strip of arterial blood pressure (upper panel after 5 s) at a period around 13 s. In the trend of R-R intervals (lower panel), fluctuations corresponding HF (period around 3 s) and LF (period around 13 s) components are observed. The LF fluctuation of R-R interval shows several-second delay behind Mayer wave of arterial blood pressure
The mechanisms generating Mayer wave are still controversial, and there are theories proposing peripheral, central, and resonance origins. Blood vessel contraction by sympathetic vasomotor function is known to occur with 5-s delay after sympathetic neural activation. Simulation studies have reported that such delay systems cause spontaneous fluctuation in the baroreceptor reflex feedback loop at a period of 10 s [20]. The LF component of HRV is decreased in patients with low baroreceptor reflex sensitivity independently of the presence of sympathetic innervation to the heart [21].
2.2 Long-Term HRV
Although the LF and HF components are the major constituents of short-term HRV, long-term HRV comprises nonperiodic components with a broad range of spectrum as the major constituents [22]. For descriptive purposes, fluctuations at frequencies lower than LF component are divided into two components: very-low-frequency (VLF) component (0.003–0.04 Hz) and ultralow frequency (ULF) component (≤0.003 Hz). Unlike LF and HF components, VLF and ULF are nonperiodic components that form no distinct peaks in power spectrum. The nonperiodic component of long-term HRV is also called 1/f fluctuation or fractal component, because it has power negatively correlated with frequency in the power spectrum and it furnishes the properties of fractal dynamics including long-term negative correlation and scale-independent self-affine structure [23].
Long-term HRV from ambulatory ECG recordings under daily activities includes the influences of circadian and ultradian variations in physiological functions, physical and mental activities, and environmental factors. Accordingly, analysis of long-term HRV does not suit for the evaluations of specific autonomic functions, but it may be useful for the evaluations of overall performance of autonomic regulations. In fact, long-term HRV provides prognostic information, particularly all-cause mortality risk in patients with cardiac and other diseases, which have stronger predictive power than short-term HRV indices [24–26].
3 Data Collection for HRV
3.1 Data Sources of HRV
HRV can be analyzed when at least one lead of continuous ECG recording is available. The ECG needs to be stored as digital data at a sampling frequency of no less than 125 Hz (desirably, 500–1000 Hz) to avoid artificial cycle length fluctuations caused by under sampling. The ECG data are converted into time series of beat-to-beat cycle length by measuring all R-R intervals (Fig. 7.3). For this purpose, accurate automated classifications (normal sinus rhythm, atrial or ventricular ectopic beat, and artifact) are requited for all beats, the results of classification should be reviewed, and all errors in the annotation need to be edited completely.
Measurement of HRV from ECG. For the analysis of HRV, time series of beat-to-beat cycle lengths are measured as R-R intervals under sinus rhythm. The temporal position of each R-R interval is usually defined as the position of subsequent R wave. For the purpose of frequency domain analyses, R-R interval time series are interpolated and resampled at equidistantly devided time points
Although pulse wave signal may be used as a surrogate of ECG, several limitations should be recognized, which include difficulty in beat classifications, limited accuracy in measurement of beat-to-beat cycle length, and modifications by the frequency characteristic of pulse wave conduction [27].
3.1.1 Standardization for Short-Term HRV Measurement
HRV is affected by various intrinsic and extrinsic factors such as environmental temperature, physical activities [28, 29], mental activities [30, 31], food intake [32], smoking [33, 34], and sleep/awake rhythm. For the assessment of autonomic function, subjects need to avoid strenuous exercise, smoking, alcohol and caffeine intake from the previous night, and food intake from 3 h before the study, and the measurement should be performed at constant time of day in an air-conditioned and calm experimental room after >15 min supine rest for equilibrium. Although the length of recording is determined by the purposes, continuous 5-min recording after the stabilization of heart rate is the standard.
For short-term HRV, controlled breathing with metronome signal may be used so that breathing frequency of subjects is kept at >0.15 Hz and heart rate to respiration ratio at >2. There are three reasons for this:
-
1.
To evaluate cardiac vagal function separately from sympathetic influences, breathing frequency needs to be kept at >0.15 Hz (the upper frequency limit of sympathetic heart rate control).
-
2.
The magnitude of HF component decreases with increasing breathing frequency independently of cardiac vagal activity.
-
3.
If heart rate to respiration ratio decreases to <2, respiratory fluctuation in cardiac vagal activity, if exists, is not reflected by HRV.
Thus, when breathing frequency is not controlled, the frequency of HF component and heart rate should be checked if these conditions are satisfied.
3.1.2 ECG Recordings for Long-Term HRV
For the assessment of long-term HRV, 24-h Holter ECG recordings are useful. ECG signals are digitized at 125–500 Hz in the recorders, and all R waves are classified automatically. Recent Holter ECG scanners have software for HRV analysis as the standard or optional function. For 24-h long-term HRV, there may be defect of data due to insufficient recording length or to temporary electrode troubles. In such case, care should be needed for the effects of day/night difference in HRV. Because the long-term HRV indices have been standardized as 24-h data, when the substantial data defects occurred disproportionally during daytime or nighttime, it causes bias of sampling.
4 Methods for HRV Analysis
A variety of measures have been used for quantifying the characteristics of HRV. They are classified by the method of analysis as shown in Table 7.1. From time domain analysis, statistical and geometric measures are calculated. These measures mainly applied to 24-h long-term recordings and used for risk stratification for mortality and adverse prognosis among patients with cardiac and other diseases. Frequency domain analysis is used for both short-term and long-term recordings. Short-term measures of HRV are used for the assessment of autonomic function, and long-term measures are used for risk stratification among patients with cardiac and other diseases. Nonlinear and fractal dynamics analyses are used mainly for long-term recordings and provide prognostic indices such as α1 of detrended fluctuation analysis (DFA) [36, 37], which is a powerful predictor of mortality risk in patients after myocardial infarction [39] and those with end-stage renal disease on chronic hemodialysis therapy [40].
Study of HRV has started in 1970s, but new methods of analysis and novel measures have been still proposed almost every year. Non-Gaussianity index of λ reflects an increase in large abrupt changes in heart rate, and an increase in this measure predicts increased mortality risk among patients with chronic heart failure [38, 41]. This measure is discussed in Chap. 9 in detail. Among most of other measures of HRV whose decrease is associated with increased health risk, λ is the only measure whose increase predicts increased risk. Because λ decreases with β-blockers, this measure is thought to be a unique index reflecting sympathetic over activities. Deceleration capacity (DC) [26] and heart rate turbulence (HRT) [5, 6] are currently the most powerful predictors of mortality risk after acute myocardial infarction. Figure 7.4 shows examples of analysis of long-term HRV in male patients after acute myocardial infarction; one survived >5 years, and the other died suddenly 25 months after the Holter monitoring.
Long-term 24-h HRV in patients after acute myocardial infarction. The patient on the left side survived for >5 years, while the patients on the right side died suddenly 25 months after Holter ECG monitoring. (a) 24-h trend graphs of N-N intervals, (b) histograms of N-N intervals and time domain measures, (c) power spectra of N-N intervals and frequency domain measures, (d) power spectra in log-log scale and spectral exponent β, (e) detrended fluctuation analysis (DFA) and short-term and long-term fractal scaling exponents, α1 and α2, (f) heart rate turbulence and turbulence onset (TO) and turbulence slope (TS), and (g) deceleration capacity (DC). See Table 7.1 for the explanation of measures
4.1 Time Domain Analysis
For time domain analysis, N-N interval data were used without interpolation. Because time domain analysis depends only on the order of data but not on the temporal positions, the statistical and geometric measures are less affected by the defect of data caused by the removal of abnormal data caused by ectopic beats, etc. Conversely, such abnormal data need to be removed because the contaminations of such data could affect substantially to statistical measures. Geometric measures are robust to the contaminations of abnormal data.
To select and interpret the time domain measures appropriately, the knowledge of crude correspondences between time domain and frequency domain measures is useful, although the relationships are affected by heart rate. SDNN, HRV triangular index, and TINN correspond to total power and SDNN index to the mean of the total powers for all 5 min segments during 24 h. RMSSD, SDSD, NN50 count, and pNN50 reflect N-N interval variations in the frequency band analyzed as HF component.
4.2 Frequency Domain Analysis
Frequency domain analysis in general indicates spectral analysis. In spectral analysis, fluctuations observed in time series data are recognized as a set of elementary waves (Fig. 7.5). Spectral analysis provides the mean frequency and amplitude (≈ sqrt [2*power]) of each elementary wave averaged over the entire length of data segment that was analyzed. In case of HRV, VLF, LF, and HF components are elementary waves, and the waveform of original time series data is the ensemble of these elementary waves.
Power spectral analysis of HRV. In spectral analysis, fluctuation observed in time series data is recognized as a set of elementary waves. As the results of analysis, the mean frequency and amplitude (≈ sqrt [2*power]) of each elementary wave averaged over the entire length of segment analyzed. In case of HRV, VLF, LF, and HF components are elementary waves, and the waveform of original time series data is the ensemble of these elementary waves
Periodic wave elements form peaks in spectrum; the position of peak represents frequency, and the height (in FFT spectrum) or area (in AR model and MEM spectra) represents amplitude/power of the elementary wave. The term “power” means variance caused by the wave, and when the wave can be assumed as a sinusoid, power and amplitude can be transformed to each other by the equation
Waves and their spectra are summable. Thus, even if two waves with different frequencies are added, the characteristics (such as frequency and amplitude) of individual wave are preserved. Also, the variance or total power of combined wave is the sum of the power of two original waves.
4.3 Interpolation and Resampling
Frequency domain analyses of HRV require interpolation and resampling of time series data before analysis. Including N-N interval and arterial blood pressure, time series data of the circulatory system arise one datum per heartbeat. Because the intervals of heartbeat are fluctuating as HRV, the sampling intervals of data points become unequal, which causes problem in spectral analysis that requires equidistantly sampled time series data.
Data interpolation is the method for estimate value at time points between two consecutive observations. Many methods have been proposed as the methods for interpolating N-N intervals, which include step, linear, and spline interpolations. Although there is no ideal method for interpolation, the difference in the interpolation method is known to cause no substantial difference in the final results of spectral analysis. Interpolation is also used for the portions of data defect caused by removal of ectopic beats and noise. The interpolated N-N interval time series data are resampled at appropriate frequency (such as 2 Hz), yielding equidistantly sampled N-N interval time series for which spectral analysis is performed.
4.4 Analysis of Dynamic Autonomic Functions
4.4.1 Limitation of Spectral Analysis
Autonomic nervous system shows dynamic responses to various stimuli. Although such responses are reflected in HRV, they are not detected by the conventional methods of spectral analysis. This is because spectral analysis assumes that data are in a steady state throughout the analyzed period, and it provides the characteristics of waves representing their averages over the period. To overcome this limitation, methods such as spectral analysis for overlapping shifting windows have been devised, but there are limitations for detecting rapid autonomic responses with high temporal resolution.
4.4.2 Analysis of Time-Dependent Changes in Frequency Component of HRV
The methods for analyzing continuous changes in frequency and amplitude of HRV are divided into two categories:
-
1.
Analysis of time/frequency distributions
-
2.
Complex demodulation (CDM)
The former includes short-term FFT, wavelet transformation, and instantaneous AR spectral analysis. These methods estimate information in three dimensions (time, frequency, and power) from N-N interval time series, that is, information in two dimensions (time and N-N interval). Thus, they need to assume the conditions of analysis. Also, even if time/frequency distribution is determined by these methods, time-dependent changes in frequency and amplitude of LF and HF components need to be extracted from that after all. In this view point, CDM that is a simpler and directly extracts necessary results may be often advantageous.
4.4.3 Complex Demodulation of HRV
The principle of CDM analysis of HRV is compared with the demodulation of electric wave by radios in amplitude modulation (AM) broadcasting system [42, 43]. In AM broadcasting, audio signal is converted into the changes in amplitude of carrier electric wave whose frequency is unique to each broadcast station. This relationship is similar to vagal modulation of the amplitude of HF component of HRV. AM radio obtains audio signals of selected broadcast station by extracting time-dependent changes in amplitude of the electric wave of the station. If similar procedure is done for HRV in HF frequency band, time-dependent changes in vagal modulation can be obtained as continuous data.
Figure 7.6 shows simulation of CDM analysis. On both sides, simulated time series data of N-N intervals were generated by combining two time series data simulating LF and HF components, on which spectral analysis and CDM were performed. On the left side, the amplitude of LF and HF components shows time-dependent changes, while on the right side, the frequency of both components shows time-dependent changes. CDM detects the changes in amplitude and frequency in each component faithfully.
Complex demodulation (CDM) of simulated data with time-dependent changes in amplitude (left-side panels a–f) and in frequency (right-side panels g–l). Lt, simulated low-frequency (LF) components with a fluctuating amplitude and a fixed frequency of 0.09 Hz and (a) with a constant amplitude and a fluctuating frequency between 0.06 and 0.12 Hz; (g) Ht, simulated high-frequency (HF) components with a fluctuating amplitude and a fixed frequency of 0.25 Hz and (b) with a constant amplitude and a fluctuating frequency between 0.16 and 0.44 Hz; (h) Ht + Lt: time series data generated by adding two component signals above (C = A + B, I = G + H); PSD, autoregressive power spectral density of the generated time series (panels d and j); and CDM LF and CDM HF, instantaneous amplitude (AMP, solid line) and frequency (FREQ, dashed line) of the LF (panels e and k); and the HF (panels f and l) components obtained by CDM (Modified from reference [43])
Figure 7.7 shows CDM analysis of R-R interval and respiration signals during symptom-limited rump exercise in a healthy young male subject. Progressive decreases in the amplitude of HF and LF components with increasing workload are observed.
CDM of R-R interval variability and spirogram (RESP) signal obtained during a supine ergometer exercise test with increasing workload (20 W/min) in a healthy young male subject. Panels a and b: R-R interval (RR) and spirogram (RESP). Panels c–g: autoregressive power spectral density (PSD) of R-R interval (solid line, RR) and spirogram (dashed line, RESP). Panels h, i, and j: instantaneous amplitude (solid line) and frequency (dashed line) of spirogram (RESP), and the LF and HF components of R-R interval variability (RR LF and RR HF) (Modified from reference [43])
CDM detects time-dependent changes in amplitude and frequency of LF component of HRV at a time resolution of 15 s and those of HF component at a higher resolution [42]. This method is useful for the analysis of autonomic responses to stress and those accompanying various pathologic episodes [44].
5 Conclusion
Although analysis of HRV provide powerful, useful, and unique tools for the assessment of autonomic functions, understandings of its physiology and methodology are important for the selection of suitable methods and measures and for the appropriate interpretation of results.
References
Ludwig C. Beiträge zur Kenntniss des Einflusses der Respirationsbewegungen auf den Blutlauf im Aortensysteme. Arch Anat Physiol Leipzig. 1847;13:242–302.
Pomeranz B, Macaulay RJ, Caudill MA, Kutz I, Adam D, Gordon D, et al. Assessment of autonomic function in humans by heart rate spectral analysis. Am J Physiol. 1985;248(1 Pt 2):H151–3.
Hayano J, Sakakibara Y, Yamada A, Yamada M, Mukai S, Fujinami T, et al. Accuracy of assessment of cardiac vagal tone by heart rate variability in normal subjects. Am J Cardiol. 1991;67(2):199–204.
Sands KEF, Appel ML, Lilly LS, Schoen FJ, Mudge Jr GH, Cohen RJ. Power spectrum analysis of heart rate variability in human cardiac transplant recipients. Circulation. 1989;79:76–82.
Schmidt G, Malik M, Barthel P, Schneider R, Ulm K, Rolnitzky L, et al. Heart-rate turbulence after ventricular premature beats as a predictor of mortality after acute myocardial infarction. Lancet. 1999;353:1390–6.
Bauer A, Malik M, Schmidt G, Barthel P, Bonnemeier H, Cygankiewicz I, et al. Heart rate turbulence: standards of measurement, physiological interpretation, and clinical use: international society for holter and noninvasive electrophysiology consensus. J Am Coll Cardiol. 2008;52(17):1353–65.
Hayano J, Yamasaki F, Sakata S, Okada A, Mukai S, Fujinami T. Spectral characteristics of ventricular response to atrial fibrillation. Am J Physiol. 1997;273:H2811–6.
Yamada A, Hayano J, Sakata S, Okada A, Mukai S, Ohte N, et al. Reduced ventricular response irregularity is associated with increased mortality in patients with chronic atrial fibrillation. Circulation. 2000;102:300–6.
Hayano J, Ishihara S, Fukuta H, Sakata S, Mukai S, Ohte N, et al. Circadian rhythm of atrioventricular conduction predicts long-term survival in patients with chronic atrial fibrillation. Chronobiol Int. 2002;19(3):633–48.
Sato K, Yamasaki F, Furuno T, Hamada T, Mukai S, Hayano J, et al. Rhythm-independent feature of heart rate dynamics common to atrial fibrillation and sinus rhythm in patients with paroxysmal atrial fibrillation. J Cardiol. 2003;42(6):269–76.
Watanabe E, Kiyono K, Hayano J, Yamamoto Y, Inamasu J, Yamamoto M, et al. Multiscale entropy of the heart rate variability for the prediction of an ischemic stroke in patients with permanent atrial fibrillation. PLoS One. 2015;10(9):e0137144.
Berger RD, Saul JP, Cohen RJ. Transfer function analysis of autonomic regulation. I: canine atrial rate response. Am J Physiol. 1989;256:H142–52.
Cooley JW, Turkey JW. An algorithm for the machine calculation of complex Fourier series. Math Comput. 1965;19:297–301.
Akaike H. Power spectrum estimation through autoregressive model fitting. Ann Inst Stat Math. 1969;21:407–19.
Sayers BM. Analysis of heart rate variability. Ergonomics. 1973;16:17–32.
Akselrod S, Gordon D, Ubel FA, Shannon DC, Barger AC, Cohen RJ. Power spectrum analysis of heart rate fluctuation: a quantitative probe of beat-to-beat cardiovascular control. Science. 1981;213:220–2.
Pagani M, Lombardi F, Guzzetti S, Rimoldi O, Furlan R, Pizzinelli P, et al. Power spectral analysis of heart rate and arterial pressure variabilities as a marker of sympatho-vagal interaction in man and conscious dog. Circ Res. 1986;59:178–93.
Hayano J, Yasuma F. Hypothesis: respiratory sinus arrhythmia is an intrinsic resting function of cardiopulmonary system. Cardiovasc Res. 2003;58(1):1–9.
Penaz J. Mayer waves: history and methodology. Automedica. 1978;2:135–41.
Madwed JB, Albrecht P, Mark RG, Cohen RJ. Low-frequency oscillation in arterial pressure and heart rate: a simple computer model. Am J Physiol. 1991;256:H1573–9.
Moak JP, Goldstein DS, Eldadah BA, Saleem A, Holmes C, Pechnik S, et al. Supine low-frequency power of heart rate variability reflects baroreflex function, not cardiac sympathetic innervation. Heart Rhythm: Off J Heart Rhythm Soc. 2007;4(12):1523–9.
Yamamoto Y, Nakamura Y, Sato H, Yamamoto M, Kato K, Hughson RL. On the fractal nature of heart rate variability in humans: effects of vagal blockade. Am J Physiol. 1995;269:R830–7.
Saul JP, Albrecht P, Berger RJ. Analysis of long term heart rate variability: methods, 1/f scaling and implications. Comput Cardiol. 1987;14:419–22.
Camm AJ, Malik M, Bigger Jr JT, Breithardt G, Cerutti S, Cohen RJ, et al. Task force of the European society of cardiology and the north American society of pacing and electrophysiology. Heart rate variability: standards of measurement, physiological interpretation and clinical use. Circulation. 1996;93(5):1043–65.
Huikuri HV, Mäkikallio TH, Peng CK, Goldberger AL, Hintze U, Moller M, et al. Fractal correlation properties of R-R interval dynamics and mortality in patients with depressed left ventricular function after an acute myocardial infarction. Circulation. 2000;101:47–53.
Bauer A, Kantelhardt JW, Barthel P, Schneider R, Makikallio T, Ulm K, et al. Deceleration capacity of heart rate as a predictor of mortality after myocardial infarction: cohort study. Lancet. 2006;367(9523):1674–81.
Hayano J, Barros AK, Kamiya A, Ohte N, Yasuma F. Assessment of pulse rate variability by the method of pulse frequency demodulation. Biomed Eng OnLine. 2005;4:62.
Arai Y, Saul JP, Albrecht P, Hartley LH, Lilly LS, Cohen RJ, et al. Modulation of cardiac autonomic activity during and immediately after exercise. Am J Physiol. 1989;256:H132–41.
Yamamoto Y, Hughson RL, Peterson JC. Autonomic control of heart rate during exercise studied by heart rate variability spectral analysis. J Appl Physiol. 1991;71:1136–42.
Sakakibara M, Takeuchi S, Hayano J. Effect of relaxation training on cardiac parasympathetic tone. Psychophysiology. 1994;31:223–8.
Sakakibara M, Kanematsu T, Yasuma F, Hayano J. Impact of real-world stress on cardiorespiratory resting function during sleep in daily life. Psychophysiology. 2008;45(4):667–70.
Hayano J, Sakakibara Y, Yamada M, Kamiya T, Fujinami T, Yokoyama K, et al. Diurnal variations in vagal and sympathetic cardiac control. Am J Physiol. 1990;258:H642–6.
Hayano J, Yamada M, Sakakibara Y, Fujinami T, Yokoyama K, Watanabe Y, et al. Short- and long-term effects of cigarette smoking on heart rate variability. Am J Cardiol. 1990;65:84–8.
Niedermaier ON, Smith ML, Beightol LA, Zukowska-Grojec Z, Goldstein DS, Eckberg DL. Influence of cigarette smoking on human autonomic function. Circulation. 1993;88:562–71.
Pincus SM, Goldberger AL. Physiological time-series analysis: what does regularity quantify. Am J Physiol. 1994;266:H1643–56.
Peng CK, Havlin S, Stanley HE, Goldberger AL. Quantification of scaling exponents and crossover phenomena in nonstationary heartbeat time series. Chaos. 1995;5(1):82–7. doi:10.1063/1.166141.
Iyengar N, Peng CK, Morin R, Goldberger AL, Lipsitz LA. Age-related alterations in the fractal scaling of cardiac interbeat interval dynamics. Am J Physiol. 1996;271:R1078–84.
Kiyono K, Hayano J, Watanabe E, Struzik ZR, Yamamoto Y. Non-Gaussian heart rate as an independent predictor of mortality in patients with chronic heart failure. Heart Rhythm. 2008;5(2):261–8.
Huikuri HV, Mäkikallio TH, Airaksinen KEJ, Seppänen T, Puukka P, Räihä IJ, et al. Power-law relationship of heart rate variability as a predictor of mortality in the elderly. Circulation. 1998;97:2031–6.
Suzuki M, Hiroshi T, Aoyama T, Tanaka M, Ishii H, Kisohara M, et al. Nonlinear measures of heart rate variability and mortality risk in hemodialysis patients. Clin J Am Soc Nephrol. 2012;7(9):1454–60. doi:10.2215/CJN.09430911.
Hayano J, Kiyono K, Struzik ZR, Yamamoto Y, Watanabe E, Stein PK, et al. Increased non-gaussianity of heart rate variability predicts cardiac mortality after an acute myocardial infarction. Front Physiol. 2011;2:65.
Hayano J, Taylor JA, Yamada A, Mukai S, Hori R, Asakawa T, et al. Continuous assessment of hemodynamic control by complex demodulation of cardiovascular variability. Am J Physiol. 1993;264:H1229–38.
Hayano J, Taylor JA, Mukai S, Okada A, Watanabe Y, Takata K, et al. Assessment of frequency shifts in R-R interval variability and respiration with complex demodulation. J Appl Physiol. 1994;77:2879–88.
Lipsitz LA, Hayano J, Sakata S, Okada A, Morin RJ. Complex demodulation of cardiorespiratory dynamics preceding vasovagal syncope. Circulation. 1998;98:977–83.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer Japan
About this chapter
Cite this chapter
Hayano, J. (2017). Introduction to Heart Rate Variability. In: Iwase, S., Hayano, J., Orimo, S. (eds) Clinical Assessment of the Autonomic Nervous System. Springer, Tokyo. https://doi.org/10.1007/978-4-431-56012-8_7
Download citation
DOI: https://doi.org/10.1007/978-4-431-56012-8_7
Published:
Publisher Name: Springer, Tokyo
Print ISBN: 978-4-431-56010-4
Online ISBN: 978-4-431-56012-8
eBook Packages: MedicineMedicine (R0)