Abstract
Glomerular basement membrane (GBM) plays a critical role in preventing serum protein leakage into Bowman’s space. By using “in vivo cryotechnique (IVCT),” the periodic acid-Schiff (PAS) fluorescence emission was well-represented GBM instead of immunostaining of collagen type IV, which was difficult to observe without the microwave treatment in specimens. Serum protein distribution in living mouse glomeruli was better visualized with IVCT compared with other conventional methods. Under normal condition, immunoreaction products of albumin and immunoglobulin G heavy and light chains (IgG (H+L)) were localized within glomerular capillary loops (GCL) but not colocalized with the PAS fluorescence emission of the GBM. Under heart-arrest condition and with quick-freezing of resected tissues, albumin, IgG (H+L), immunoglobulin kappa light chain, and IgG1 heavy chain (IgG1) were immunolocalized within GCL and mesangial areas, but only albumin and kappa light chain were additionally immunolocalized in Bowman’s space, indicating their passage through GBM. Under acute hypertensive condition, both albumin and kappa light chain, but not IgG1, were clearly immunolocalized along GBM and in Bowman’s space, indicating their increased passage through GBM. The overlapped areas of PAS fluorescence emission and albumin or kappa light chain were appeared to be larger with quick-freezing and under the heart-arrest or acute hypertensive condition than under normal circulation, whereas those of PAS emission and IgG1 were not different among those conditions. These results suggest that PAS fluorescence emission combined with IVCT allowed us to investigate the serum proteins passing through GBM induced by hemodynamic changes.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Glomerular basement membrane
- Serum proteins
- Hemodynamic changes
- In vivo cryotechnique
- PAS fluorescence emission
1 Introduction
The hemodynamic changes in animal kidneys are well known to cause abnormal leakage of serum protein s through glomerular filtration barrier s , leading to proteinuria , which is closely related to progression of renal diseases in experimental animal studies and clinical human cases [1, 2]. The charge- and size-selective glomerular filtration barriers against serum protein s were composed of highly fenestrated endothelium , glomerular basement membrane s (GBM ), and podocytes [3, 4]. Although there are some evidences [1, 5] suggesting key roles of GBM for the filtration barriers of serum proteins, one of the technical problems to reveal the immunolocalization of serum proteins would be due to their preparation procedures of the in situ drip-fixation with aqueous aldehyde fixatives [6].
The “in vivo cryotechnique ” (IVCT) has been already developed for the past decade [7–9], which is designed to capture transiently dynamic morphology of living animal organ s . Using IVCT followed by freeze substitution , the diffusion of soluble component s would be mostly restricted [10], and many antigenic sites would be exposed in the frozen cells and tissues [11]. However, in living mouse kidney specimens prepared by IVCT, collagen type IV , laminin 5, fibronectin, and perlecan, the widely used markers for GBM [12–15], were not clearly immunostained. To elucidate the distribution alternation of serum protein s in living mouse kidney under various hemodynamic condition s , the fluorescence emission of periodic acid-Schiff (PAS ) staining was utilized for representing GBM, and serum protein immunolocalizations were examined in living mouse kidneys by IVCT.
2 Application of PAS Fluorescence Emission for Glomerular Basement Membrane
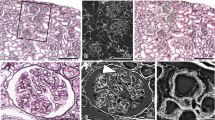
The PAS staining of nephrons is specific for glomerular or tubular basement membrane s , mesangial areas (MA), and proximal tubular brush borders in living mouse kidney s [16]. By using the common PAS staining (Fig. 22.1a) followed with immunostaining for zonula occludens-1 (ZO-1) (Fig. 22.1c), a specific marker of foot process es in glomeruli [17–20], the PAS fluorescence emission can be shown to be localized at the GBM and MA in the glomerulus (Fig. 22.1b). The fluorescence-merged image clearly showed that the immunofluorescence for ZO-1 mostly outlines the PAS fluorescence emission of the GBM (Fig. 22.1d), indicating fluorescence emission of PAS was effectively represented GBM. The immunostaining for GBM with specific antibodies against collage type IV was hardly successful in the specimens prepared with the IVCT (unpublished data), although the immunoreactivity of collagen type IV was increased at GBM after the microwave treatment on paraffin sections prepared with IVCT. The differences in “immunoreactivity” would be due to that small reagents in the PAS staining processes could be easily bound to the target molecular sites of the compact GBM with serum protein s , whereas larger molecules of antibodies were difficult to reach them using IVCT followed by freeze substitution.
Representative light micrograph (a) and confocal laser scanning micrographs (b–d) showing periodic acid-Schiff staining ( PAS ; a), its fluorescence emission ( PAS fluorescence emission; b), and immunofluorescence staining for zonula occludens-1 (ZO-1; c) on the same section prepared by “in vivo cryotechnique ” under the normotensive condition. The layer at glomerular basement membrane positive for PAS fluorescence emission mostly surrounds the glomerular capillary loop s and closely localizes near the foot process layers visualized by the ZO-1 immunostaining (d, arrows and arrowheads). Gl glomeruli, PT proximal tubule s , asterisks blood capillaries . Scale bars 10 μm
3 Alternation of Serum Protein Distribution in Glomeruli Under Various Hemodynamic Conditions
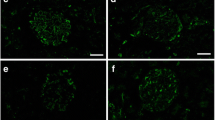
As PAS fluorescence emission has been well identified to represent GBM , the distribution of serum protein s in living mouse glomeruli could be estimated by comparing the immunolocalization of serum proteins and GBM using PAS fluorescence emission combined with IVCT. Serial sections of kidneys under different hemodynamic condition s were prepared with IVCT and immunostained to detect the distribution of albumin , immunoglobulin G heavy and light chains (IgG (H+L)), as well as the kappa light chain and IgG1 heavy chain s (IgG1) (Fig. 22.2).
Representative confocal laser scanning micrographs showing the alternation of serum protein (green color) distribution in living mouse kidney under various hemodynamic condition s using PAS fluorescence emission (red color). Nuclei are labeled with TO-PRO-3 (blue color). Under the normotensive condition (normotension), the immunolocalization of albumin (a) is primarily restricted within glomerular capillary loop s (GCL; arrowhead) and slightly overlapped with the PAS fluorescence emission at glomerular basement membrane ( GBM ; arrows); immunolocalizations of immunoglobulin G high and light chains (IgG (H+L), e), the kappa light chain (kappa, i), and IgG1 heavy chain (IgG1, m) are detected within GCL and at some mesangial areas (MA; arrowheads). Under the heart-arrest condition (heart arrest) and in the kidney tissues quickly frozen after tissue resection (resection), the GCL are collapsed, and the leaked albumin is additionally immunolocalized in Bowman’s space (b and c, arrowheads); immunoreaction products of IgG (H+L) are also localized in Bowman’s space (f and g, arrows); kappa are clearly localized in Bowman’s space and also proximal convoluted tubule s (j and k, arrows); IgG1 are localized at MA (n and o, arrowheads), but neither in Bowman’s space nor in proximal convoluted tubules. Under the acute hypertensive condition (hypertension), albumin is more clearly immunolocalized in Bowman’s space (d, arrowheads); IgG (H+L) immunostaining is clearly detected in Bowman’s space and also in proximal convoluted tubules (h, arrows); kappa is immunolocalized in Bowman’s space (l, right upper inset, arrows), the MA (arrowhead), and the apical cytoplasm of some proximal tubule s (arrows); IgG1 is detected exclusively within the GCL and at some MA (p, arrowheads), but not in Bowman’s space. In addition, under the heart-arrest or acute hypertensive conditions and in the kidney tissues quickly frozen after tissue resection, the glomerular areas, where the immunoreactivity of albumin (b–d), IgG (H+L) (f–h), kappa (j–l), and PAS overlapped each other (arrows), respectively, appear to become wider than those under the normotensive condition (a, e, i, arrows), whereas the overlapping of IgG1 and PAS appears to be unchanged (m–p, double arrowheads). Gl glomeruli, PT proximal tubules, asterisks blood capillaries . Scale bars 10 μm
Under normal hemodynamic condition , both serum albumin and IgG were almost kept within GCL (Fig. 22.2a, e), and their immunoreactivity was more widely overlapped with the PAS fluorescence emission under the acute hypertensive condition (Fig. 22.2d, h). Although the main filtration barrier was reported to be GBM itself by electron microscopy [5, 21], other reports also demonstrated that slit diaphragms of foot process es formed the ultimate filtration barrier for macromolecular permeability [22, 23]. Considering these previous reports, our findings suggest that more serum protein s reached the outer layer of GBM under the acute hypertensive condition and the slit diaphragms would play a significant role especially in such pathological or physiological states. The translocation of serum proteins may be due to the acutely increased pressures in the GCL, and if the blood pressures were temporarily so high, lots of serum proteins would leak out into Bowman’s space.
Under abnormal conditions, the leakage of albumin (Fig. 22.2b–d) and kappa light chain (Fig. 22.2j–l), but not IgG1 (Fig. 22.2n–p), was clearly detected in Bowman’s space. Serum proteins with low molecular weights, such as albumin and kappa light chains, could easily translocate to the urinary space and reabsorbed in the convoluted proximal tubule s [24–27]. On the other hand, larger or cationic molecules, such as IgGs, were trapped in the GBM and hardly leaked out into Bowman’s spaces under some physiological or pathological conditions, such as membranoproliferative glomerulonephritis, membranous nephropathy, and lupus nephritis [28–30]. The differences of protein immunolocalization are undoubtedly due to the size and charge selectivity of the GBM [31].
In the present experiment, the overlapping of PAS fluorescence emission with the IgG (H+L) immunoreactivity close to Bowman’s space appeared to be prominent at some parts on one side of the GCL under the acute hypertensive condition (Fig. 22.2h). As such heterogeneity of leaking areas was also observed using extrinsic tracers [21], the leakage degree of serum protein s would exhibit their heterogeneous immunolocalizations not only among glomeruli but also among GCL under the acute hypertensive condition.
The leakage of both albumin and IgG (H+L) through GBM into Bowman’s spaces was detected under the heart-arrest condition (Fig. 22.2b, f) or with the quick-freezing of the resected kidney tissues (Fig. 22.2c, g), which might be caused by low blood pressures and ischemia , resulting in serious alteration of GCL structures and molecular organization [5, 32]. Moreover, the ischemia or hypoxia , including the hypotensive condition, was reported to induce changes of the glomerular structures and also damages of intercellular junctions in renal vasculatures, resulting in the increased vascular permeability and local interstitial edema of kidneys [7, 33–35].
4 Concluding Remarks
In conclusion, the fluorescence emission of PAS staining allowed us to examine the precise immunolocalization of serum protein s at the GBM under various hemodynamic condition s of living mouse kidney s in combination with the IVCT followed by freeze substitution. In addition, the IVCT would be a reliable tool to observe soluble serum protein s in situ and capture transient images of functioning glomeruli in the living mice. Further detailed analyses, e.g., by using immunoelectron microscopy in conjunction with the “in vivo cryotechnique ,” would enable us to characterize the changes of soluble serum proteins around GBM, which may be new findings of clinical importance. The present figures were already published in our paper, Arch Histol Cytol (2006) 69:147–161, and cited with their permissions.
References
Olivetti G, Kithier K, Giacomelli F, Wiener J (1981) Glomerular permeability to endogenous proteins in the rat: effects of acute hypertension. Lab Invest 44:127–137
Remuzzi G, Bertani T (1998) Pathophysiology of progressive nephropathies. N Engl J Med 339:1448–1456
Reiser J, Kriz W, Kretzler M, Mundel P (2000) The glomerular slit diaphragm is a modified adherens junction. J Am Soc Nephrol 11:1–8
Rico M, Mukherjee A, Konieczkowski M, Bruggeman LA, Miller RT, Khan S, Schelling JR, Sedor JR (2005) WT1-interacting protein and ZO-1 translocate into podocyte nuclei after puromycin aminonucleoside treatment. Am J Physiol Renal Physiol 289:F431–F441
Fujigaki Y, Nagase M, Kobayasi S, Hidaka S, Shimomura M, Hishida A (1993) Intra-GBM site of the functional filtration barrier for endogenous proteins in rats. Kidney Int 43:567–574
Ryan GB, Karnovsky MJ (1976) Distribution of endogenous albumin in the rat glomerulus: role of hemodynamic factors in glomerular barrier function. Kidney Int 9:36–45
Ohno S, Terada N, Fujii Y, Ueda H, Takayama I (1996) Dynamic structure of glomerular capillary loop as revealed by an in vivo cryotechnique. Virchows Arch 427:519–527
Ohno N, Terada N, Fujii Y, Baba T, Ohno S (2004) “In vivo cryotechnique” for paradigm shift to “living morphology” of animal organs. Biomed Rev 15:1–19
Terada N, Ohno N, Li Z, Fujii Y, Baba T, Ohno S (2006) Application of in vivo cryotechnique to the examination of cells and tissues in living animal organs. Histol Histopathol 21:265–272
Bridgman PC, Dailey ME (1989) The organization of myosin and actin in rapid frozen nerve growth cones. J Cell Biol 108:95–109
Ohno N, Terada N, Murata S, Katoh R, Ohno S (2005) Application of cryotechniques with freeze-substitution for the immunohistochemical demonstration of intranuclear pCREB and chromosome territory. J Histochem Cytochem 53:55–62
Sorokin LM, Pausch F, Durbeej M, Ekblom P (1997) Differential expression of five laminin alpha (1-5) chains in developing and adult mouse kidney. Dev Dyn 210:446–462
Conde-Knape K (2001) Heparan sulfate proteoglycans in experimental models of diabetes: a role for perlecan in diabetes complications. Diabetes Metab Res Rev 17:412–421
van Vliet AI, van Alderwegen IE, Baelde HJ, de Heer E, Bruijn JA (2002) Fibronectin accumulation in glomerulosclerotic lesions: self-assembly sites and the heparin II binding domain. Kidney Int 61:481–489
Chow FY, Nikolic-Paterson DJ, Atkins RC, Tesch GH (2004) Macrophages in streptozotocin-induced diabetic nephropathy: potential role in renal fibrosis. Nephrol Dial Transplant 19:2987–2996
Nagato Y, Mitsui T, Kushida T, Kushida H (1985) Localization of periodate-Schiff reactive glycosaminoglycans in semi-thin sections embedded in GMA-Quetol 523-MMA–application of a method for correlative light and electron microscopy of identical sites. Tokai J Exp Clin Med 10:37–46
Schnabel E, Anderson JM, Farquhar MG (1990) The tight junction protein ZO-1 is concentrated along slit diaphragms of the glomerular epithelium. J Cell Biol 111:1255–1263
Angeles G, Owens SA, Ewers FW (2004) Fluorescence shell: a novel view of sclereid morphology with the Confocal Laser Scanning Microscope. Microsc Res Tech 63:282–288
Evans CM, Williams OW, Tuvim MJ, Nigam R, Mixides GP, Blackburn MR, DeMayo FJ, Burns AR, Smith C, Reynolds SD, Stripp BR, Dickey BF (2004) Mucin is produced by clara cells in the proximal airways of antigen-challenged mice. Am J Respir Cell Mol Biol 31:382–394
Schaart G, Hesselink RP, Keizer HA, van Kranenburg G, Drost MR, Hesselink MK (2004) A modified PAS stain combined with immunofluorescence for quantitative analyses of glycogen in muscle sections. Histochem Cell Biol 122:161–169
Farquhar MG, Wissig SL, Palade GE (1961) Glomerular permeability. I. Ferritin transfer across the normal glomerular capillary wall. J Exp Med 113:47–66
Graham RC Jr, Karnovsky MJ (1966) Glomerular permeability. Ultrastructural cytochemical studies using peroxidases as protein tracers. J Exp Med 124:1123–1134
Mundel P, Shankland SJ (2002) Podocyte biology and response to injury. J Am Soc Nephrol 13:3005–3015
Batuman V, Dreisbach AW, Cyran J (1990) Light-chain binding sites on renal brush-border membranes. Am J Physiol 258:F1259–F1265
Batuman V, Guan S (1997) Receptor-mediated endocytosis of immunoglobulin light chains by renal proximal tubule cells. Am J Physiol 272:F521–F530
Birn H, Fyfe JC, Jacobsen C, Mounier F, Verroust PJ, Orskov H, Willnow TE, Moestrup SK, Christensen EI (2000) Cubilin is an albumin binding protein important for renal tubular albumin reabsorption. J Clin Invest 105:1353–1361
Brunskill NJ (2000) Albumin and proximal tubular cells–beyond endocytosis. Nephrol Dial Transplant 15:1732–1734
Andrews PM, Bates SB (1985) Dose-dependent movement of cationic molecules across the glomerular wall. Anat Rec 212:223–231
Imai H, Hamai K, Komatsuda A, Ohtani H, Miura AB (1997) IgG subclasses in patients with membranoproliferative glomerulonephritis, membranous nephropathy, and lupus nephritis. Kidney Int 51:270–276
Bijl M, Dijstelbloem HM, Oost WW, Bootsma H, Derksen RH, Aten J, Limburg PC, Kallenberg CG (2002) IgG subclass distribution of autoantibodies differs between renal and extra-renal relapses in patients with systemic lupus erythematosus. Rheumatology (Oxford) 41:62–67
Kanwar YS (1984) Biophysiology of glomerular filtration and proteinuria. Lab Invest 51:7–21
Zea-Aragon Z, Terada N, Ohno N, Fujii Y, Baba T, Ohno S (2004) Effects of anoxia on serum immunoglobulin and albumin leakage through blood-brain barrier in mouse cerebellum as revealed by cryotechniques. J Neurosci Methods 138:89–95
Griffith LD, Bulger RE, Trump BF (1967) The ultrastructure of the functioning kidney. Lab Invest 16:220–246
Pagtalunan ME, Olson JL, Tilney NL, Meyer TW (1999) Late consequences of acute ischemic injury to a solitary kidney. J Am Soc Nephrol 10:366–373
Sutton TA, Mang HE, Campos SB, Sandoval RM, Yoder MC, Molitoris BA (2003) Injury of the renal microvascular endothelium alters barrier function after ischemia. Am J Physiol Renal Physiol 285:F191–F198
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer Japan
About this chapter
Cite this chapter
Li, Z., Terada, N., Ohno, S. (2016). Application of Periodic Acid-Schiff Fluorescence Emission for Immunohistochemistry of Living Mouse Renal Glomeruli. In: Ohno, S., Ohno, N., Terada, N. (eds) In Vivo Cryotechnique in Biomedical Research and Application for Bioimaging of Living Animal Organs. Springer, Tokyo. https://doi.org/10.1007/978-4-431-55723-4_22
Download citation
DOI: https://doi.org/10.1007/978-4-431-55723-4_22
Publisher Name: Springer, Tokyo
Print ISBN: 978-4-431-55722-7
Online ISBN: 978-4-431-55723-4
eBook Packages: MedicineMedicine (R0)