Abstract
To compare the influence of the different fixation procedures on the distribution of endogenous proteins of albumin and IgG in mouse glomeruli and identify the advantage of tissue fixation method in living mouse renal glomeruli by “in vivo cryotechnique” (IVCT), four different fixation methods were performed in mouse kidney tissues, such as (i) conventional immersion or (ii) quick-freezing following resected kidney tissues, (iii) quick-freezing following perfusion-fixation, and (iv) “in vivo cryotechnique” for living mouse kidneys. Kidney glomeruli were noticeably contracted after conventional immersion-fixation or quick-freezing following resection compared to glomeruli from tissues preserved by the IVCT. With the IVCT, both albumin and IgG were colocalized exclusively along or within the glomerular capillary loops; however, immunoreactivity of these proteins in the other three methods was clearly detected in Bowman’s space and apical cytoplasm of the proximal tubules. With the IVCT, immunoreactivity of collagen type IV was very weak at the glomerular basement membrane (GBM) until microwave treatment, which increased its immunoreactivity. Using conventional fixation method, aquaporin-1 (AQP-1) is abundant in the apical and basolateral membranes of renal proximal tubules and descending thin limbs, but the proteins were prominent immunolocalization on the apical areas by “in vivo cryotechnique.” The “in vivo cryotechnique” should be a reliable tool to maintain soluble serum proteins and AQP-1 in situ and capture transient images of functioning kidney in living mice.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
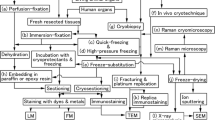
Immersion-fixation and perfusion -fixation are well known to be tissue fixation techniques that have been applied widely in clinical and experimental research. However, these fixation procedures usually cause transient ischemia and hypoxia and translocation of components in cells and tissues, which can lead to changes in the tissues’ construction and function [1, 2]. Furthermore, renal glomerular selectivity for permeable serum solutes and translocation of filtered serum protein s have never been directly visualized on paraffin sections by light microscopy (LM) due to the technical limitations in preparing specimens for immunohistochemistry using routine fixation methods. The common fixatives in buffer solution, such as glutaraldehyde and paraformaldehyde , need considerable time to completely cross-link and preserve the soluble substances during routine perfusion-fixation or immersion-fixation . Many of these soluble substances are washed from the specimens, and the translocation of soluble serum protein s occurs during the fixation period. In addition, the specimens from freeze-substitution technique are routinely embedded in paraffin wax, presumably leaving many antigenic sites exposed due to formation of tiny ice crystal s in the frozen cells and tissues [3, 4]. To adequately define the functional structures of tissues and cells in vivo, the in situ preservation of cells and tissues in living animal organ s is necessary. We previously developed the “in vivo cryotechnique ”(IVCT) [5–7], which is designed to transiently arrest dynamic structures in living animal organs by the combination of a cryoknife precooled in liquid nitrogen (−196 °C) and liquid isopentane -propane cryogen (−193 °C). With this technique, all biological processes in living animal organs instantly stop and are embedded in the ice microenvironment while maintaining all their functional components in situ [2, 6].
In the present, the periodic acid-Schiff (PAS ) staining was clearly detected in the specimens prepared by the “in vivo cryotechnique .” Therefore, we have used the conventional PAS staining with its fluorescence emission as a marker for GBM . We compared the fluorescence emission of PAS staining with immunofluorescence of serum protein s , on the same section to analyze their distribution in the living mouse kidney s . On the other hand, the localization of AQP-1 in proximal tubule s has never been directly visualized on paraffin sections by “in vivo cryotechnique .”
2 PAS Staining and Immunolocalization of Albumin and IgG in Glomeruli
To visualize topographical migrations of serum protein s such as albumin or IgG passing through GBM filtration barriers under various fixation conditions, we used the PAS fluorescence emission as a marker for the GBM in combination with immunostaining for several serum proteins on the same sections. Under normotensive condition, immunoreaction products of both albumin and IgG were exclusively colocalized along or within the GCL, and only IgG immunolocalization was also seen in some mesangial areas. Neither albumin nor IgG was immunolocalized in Bowman’s space. On the contrary, under quick-freezing after tissue resection and conventional immersion-fixation conditions, immunoreaction products of both albumin and IgG were found to be localized in Bowman’s space. Under quick-freezing following perfusion -fixation condition, however, both of albumin and IgG were partially washed out and immunostaining was weakly detected in glomerular blood capillary loops than that under other conditions due to the perfusion-fixation. In addition, the immunostaining of both albumin and IgG was more clearly seen on apical cell membranes of proximal convoluted tubule s and some Bowman’s spaces at the artificial state washing out all of the circulating proteins (Fig. 21.1).
Confocal laser scanning micrographs showing the distribution of endogenous albumin (red color) and IgG (green color) in living mouse glomeruli under four different fixation methods. (a) Both albumin and IgG were exclusively colocalized along or within the glomerular capillary loop s (GCLs) with the “in vivo cryotechnique ” (IVCT), (b) under quick-freezing following resection (QF), and (c) conventional immersion-fixation (IF); (d) we found both albumin and IgG in Bowman’s space. Under quick-freezing following perfusion -fixation (PF), we detected a weak indication of both albumin and IgG in the GCLs, but more clearly detected these proteins in Bowman’s space and on apical cell membranes of proximal convoluted tubule s . Scale bars 10 mm
Under the normotensive condition by “in vivo cryotechnique ,” the immunofluorescence for albumin and IgG was exclusively detected in GCL with little colocalization with the PAS fluorescence emission, and IgG was also at MA. To the contrary, under the quick-freezing of resected kidney tissues or with immersion-fixation condition, both of the albumin and IgG immunoreactivity were also detected in Bowman’s space. These findings suggest that some amounts of albumin or IgG, which are almost kept within GCL under the normotensive condition, easily pass through the GBM and translocate into Bowman’s space under the quick-freezing of resected kidney tissues or immersion-fixation condition (Fig. 21.2).
Confocal laser scanning micrographs showing albumin or IgG (green color) passing through GBM filtration barriers under various fixation conditions using the PAS fluorescence emission (red color) as a marker for the GBM. With the IVCT (a, b), the immunofluorescence for albumin and IgG was exclusively detected in GCLs with little colocalization with the PAS fluorescence emission, and IgG was also in mesangial areas (b). After QF (c, d) or IF (e, f), both albumin and IgG immunoreactivities were detected in Bowman’s space on the opposite side of the GCLs and bordered by the PAS-positive GBM. The overlapping images of albumin or IgG immunoreactivity with PAS fluorescence emission, represented by a yellow color, was more widely apparent with IF (e, f) than with QF (c, d). Scale bars 10 mm
3 Metal-Enhanced 3,3′-Diaminobenzidine (DAB) Staining of Collagen Type IV and Aquaporin-1(AQP-1 ) in Living Mouse Glomeruli and Proximal Tubules
The common HE staining was performed to examine native morphology in the living mouse kidney s , as obtained by the “in vivo cryotechnique .” Well-preserved areas of renal cortices under normotensive conditions could be obtained within 300–400 μm from the frozen surface tissues without visible ice crystal s at a light microscopic level, and the GCLs are kept open. Although collagen type IV is reported to be clearly immunolocalized in the GBM and MAs after a conventional fixation method, when we employed the “in vivo cryotechnique ,” we found marked immunolocalization of collagen type IV in the MAs, but only weakly immunolocalized on the GBM. To visualize the expression of water channel AQP-1 in living mouse proximal tubule s , the immunostaining for AQP-1 was performed, respectively, on paraffin section with “in vivo cryotechnique” and perfusion-fixation methods. Using perfusion-fixation method, the immunoreaction product of AQP-1 was observed clearly along the basolateral membrane of proximal tubule, and its distribution was disorganized and irregular on the brush border and apical cell membrane along the proximal tubule due to the artificial state (Fig. 21.3).
Light micrographs showing immunolocalization of aquaporin-1 (AQP-1 ) in living mouse proximal tubule s on paraffin section. Under PF, AQP-1 had a disorganized and irregular distribution on the brush border and apical cell membrane along the proximal tubules and was also clearly found along the basolateral membrane of the proximal tubules, which is an artificial state (a). With the IVCT, AQP-1 was labeled intensely and regularly on the brush border and apical cell membrane along the proximal tubules, but only weakly labeled along the basolateral membrane. There was also little immunostaining detected in the initial part of the proximal tubules (b)
4 Concluding Remarks
The fluorescence emission of PAS staining allowed us to examine the precise immunolocalization of serum protein s at the GBM under various hemodynamic condition s of living mouse kidney s in combination with the “in vivo cryotechnique” followed by freeze substitution. In addition, the “in vivo cryotechnique ” would be a reliable tool to observe soluble serum protein s and water channel AQP-1 in situ and capture transient images of functioning glomeruli in the living mice [1, 2, 5, 7–9]. The present figures were already published in our paper, Microsc Res Tech (2013) 76:113–120, and cited with their permissions.
References
Li Z, Ohno N, Terada N, Ohno S (2006) Immunolocalization of serum proteins in living mouse glomeruli under various hemodynamic conditions by “in vivo cryotechnique”. Histochem Cell Biol 126(3):399–406
Ohno S, Terada N, Ohno N, Saitoh S, Saitoh Y, Fujii Y (2010) Significance of “in vivo cryotechnique” for morphofunctional analyses of living animal organs. J Electron Microsc (Tokyo) 59(5):395–408
Bridgman PC, Dailey ME (1989) The organization of myosin and actin in rapid frozen nerve growth cones. J Cell Biol 108(1):95–109
Ohno N, Terada N, Murata S, Katoh R, Ohno S (2005) Application of cryotechniques with freeze-substitution for the immunohistochemical demonstration of intranuclear pCREB and chromosome territory. J Histochem Cytochem 53(1):55–62
Ohno S, Terada N, Fujii Y, Ueda H, Takayama I (1996) Dynamic structure of glomerular capillary loop as revealed by an in vivo cryotechnique. Virchows Arch 427(5):519–527
Terada N, Ohno N, Li Z, Fujii Y, Baba T, Ohno S (2005) Detection of injected fluorescence-conjugated IgG in living mouse organs using “in vivo cryotechnique” with freeze-substitution. Microsc Res Tech 66(4):173–178
Terada N, Ohno N, Li Z, Fujii Y, Baba T, Ohno S (2006) Application of in vivo cryotechnique to the examination of cells and tissues in living animal organs. Histol Histopathol 21(3):265–272
Zhai XY, Fenton RA, Andreasen A, Thomsen JS, Christensen EI (2007) Aquaporin-1 is not expressed in descending thin limbs of short-loop nephrons. J Am Soc Nephrol 18(11):2937–2944
Mobasheri A, Marples D (2004) Expression of the AQP-1 water channel in normal human tissues: a semiquantitative study using tissue microarray technology. Am J Physiol Cell Physiol 286(3):C529–C537
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer Japan
About this chapter
Cite this chapter
Li, Z., Zhai, X., Ohno, N., Terada, N., Ohno, S. (2016). Application of Novel “In Vivo Cryotechnique” in Living Animal Kidneys. In: Ohno, S., Ohno, N., Terada, N. (eds) In Vivo Cryotechnique in Biomedical Research and Application for Bioimaging of Living Animal Organs. Springer, Tokyo. https://doi.org/10.1007/978-4-431-55723-4_21
Download citation
DOI: https://doi.org/10.1007/978-4-431-55723-4_21
Publisher Name: Springer, Tokyo
Print ISBN: 978-4-431-55722-7
Online ISBN: 978-4-431-55723-4
eBook Packages: MedicineMedicine (R0)