Abstract
A small number of patients with chronic kidney disease (CKD), particularly those on maintenance hemodialysis, are in the habit of exercise, and this sedentary lifestyle and poor physical fitness is associated with a high prevalence of fall-associated fracture and an increased mortality risk. However, exercise training has various therapeutic benefits in these patients. It delays the progression of microalbuminuria or CKD, increases physical fitness and strength, improves mental health, enhances dialysis efficacy, results in a better glycemic control, and reduces both cardiovascular and mortality risks.
If possible, undergoing a cardiopulmonary exercise test (CPX) before participating in an exercise program may lead to a safer and more effective training of these patients. Aerobic exercise training should be started with a low level of exercise load, and its intensity should be gradually increased along with the assessment of the patient’s perceived exertion during the sessions. Not only ensuring the safety and enough consideration toward the diabetic complications but also the involvement of exercise professionals and attractive programs are included as the key elements of the sustainability of exercise in these patients.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Physical Frailty Among Patients with Chronic Kidney Disease (CKD)
1.1 Physical Inactivity Among Patients with CKD
1.1.1 A Small Number of the Patients with CKD Are Habitual of Exercise
Regular exercise is an established non-pharmacological therapeutic strategy for patients with diabetes with or without chronic kidney disease (CKD) and is expected to bring about favorable effects on their physical functions and reduce the cardiovascular and mortality risk factors. In 2005, the National Kidney Foundation recommended in the Kidney Disease Outcomes Quality Initiative (K/DOQI) cardiovascular guidelines that patients with CKD should perform at least 30 min of physical activity of moderate intensity every day, if possible [1]. However, this population, particularly, those on dialysis, has a significantly lower rate of being habitual to exercise as compared with their healthy sedentary counterparts. According to the cross-sectional data of 20,920 participants in the Dialysis Outcomes and Practice Patterns Study (DOPPS), 52.4 % of the patients on maintenance hemodialysis never exercised or exercised less than once a week, and this sedentary lifestyle was remarkable when the facility the patients belonged did not provide specific exercise programs [2]. In addition to the aforementioned findings, obtained using self-reported physical activity levels, investigations by activity monitors have quantitatively revealed the physical inactivity of patients on hemodialysis. Johansen et al. [3] and recently Kim et al. [4] reported in their studies using a three-dimensional accelerometer that the daily physical activity and the estimated daily energy expenditure were reduced in dialysis patients with values corresponding to 60–70 % of those in normal controls.
Several prospective observational studies, including a large cohort of dialysis patients, revealed that sedentary behavior was associated with an increased risk of mortality among not only the general population but also these patients [5–7].
1.1.2 Why Are They Sedentary?
Most patients on hemodialysis are forced to spend 3–4 h on bed rest during one dialysis session, which amounts to 9–12 h a week. Actually, the parameters pertaining to the physical activity level, such as the number of daily steps and activity-related energy expenditure, are lower on dialysis days as compared with non-dialysis days [8]. In addition, particularly, patients with diabetes may face difficulties in getting out of bed due to various complications such as blindness, stroke sequelae, and leg amputation. However, physical inactivity among these patients on dialysis is not only a result of this lengthy bed rest period. Delgado et al. conducted a survey on 100 dialysis patients to identify the barriers, which prevent them from participating in exercise [9]. In their report, almost all patients believed that a sedentary lifestyle was a health risk and that exercising was beneficial. Nevertheless, a majority of them had at least one barrier toward physical activity. Shortness of breath, lack of motivation, and having multiple medical problems were the most common barriers, in addition to fatigue or insufficient time to exercise on dialysis days.
Although the patients also answered they would exercise if counseled to do so, the counseling behavior by nephrologists does not seem enough. Few nephrologists make an assessment of their patients’ physical activity levels and advise them to increase exercise in spite of the published K/DOQI guidelines [10]. This lack of guidance by the nephrologists is associated with their concerns about risks associated with exercise participation and a lack of confidence in the ability to discuss the topics pertaining to physical activity. Consequently, patients with CKD have little opportunity to increase their physical activity.
1.2 Low Exercise Capacity and Motor Ability in the Population
1.2.1 Low Peak Oxygen Consumption (Peak VO2) and Impaired Motor Ability
There are several studies regarding limited aerobic fitness in patients with CKD. It has been reported that patients with stage 3–5 CKD have a lower peak VO2 as compared with their healthy counterparts, with an average of 50–80 % of reference values [11]. Furthermore, Leehey et al. in an exercise intervention study showed that obese diabetic patients with stage 2–4 CKD had an extremely low peak VO2, averaging 45 % of healthy levels [12]. Their low exercise capacity corresponded to the Class III determined in New York Heart Function Assessment (NYHA) classification and probably limited their daily activities to slow walking.
In addition to a low aerobic capacity, muscle atrophy (sarcopenia) and reduced muscle strength are common complications among patients with CKD, particularly in the dialysis patients [13]. The cross-sectional areas of thigh muscle are smaller in dialysis patients than in dialysis-independent patients with CKD and are associated with aging and low serum albumin concentration [14]. Quadriceps muscle strength in dialysis patients is also less than 70 % of that in their anthropometrically matched healthy counterparts, and serum albumin concentration is the only significant predictor of the loss of muscle strength [15]. Thus, muscle weakness, a well-recognized feature in dialysis patients, is most likely derived from protein-energy malnutrition.
1.2.2 Clinical Consequences of Low Exercise Capacity
In elderly people, physical performance and functional independence in daily life are mainly determined by muscle strength. The same is true for dialysis patients, and muscle weakness contributes to impairment in mobility such as walking at a slow speed. Dialysis patients also have higher chances of falling. Cook et al. have shown by cross-sectional interviews with seniors (65 years and older) on dialysis that 27 % of them had fallen in the past 12 months and that 4 % of them had reported a fall-associated fracture or head injury [16]. They also revealed that the rate of falling in elderly dialysis patients was higher than in the elderly population without CKD [17]. Trauma or fracture by falling inevitably causes hospitalization and functional decline and often results in being bedridden or other life-threatening conditions.
On the other hand, exercise capacity evaluated by peak VO2 is an important predictor of survival in dialysis patients as well as in the population without CKD [18]. This is because peak VO2 reflects other factors (e.g., cardiovascular function or complicated medical conditions), which have been previously established as the predictors of survival for the patients. Sietsema et al. analyzed 175 dialysis cohorts over a median follow-up of 39 months and found that a low peak VO2 was a significant contributor to mortality along with age and chronic heart failure [19]. Recently, a much simpler test measuring the distance walked in 6 min has become widely used as the index of functional capacity. This test is closely associated with peak VO2, and it has been reported as a useful predictor of survival in dialysis patients, in whom a 100-m increment increased approximately 5 % of the survival rate [20].
In addition, it is a major problem that these impaired physical performances adversely affect the mental health and health-related quality of life (QOL) in patients with CKD, particularly in those on dialysis [21–23], which finally results in dependency on family members and caregivers.
1.2.3 Why Are They Unfit?
Besides physical inactivity, various specific factors in CKD seem to contribute to low physical fitness, although the exact mechanism has not been fully resolved. To date, anemia, a very common complication in patients with CKD, has been considered to be very important in the reduction of exercise capacity in these patients. A number of studies have reported an improvement in peak VO2 by intense treatment of anemia in patients with dialysis-dependent and dialysis-independent CKD. However, in many of these studies, exercise capacity failed to improve as much as was expected post the extent of the achieved amount of correction in hemoglobin concentration. Furthermore, some reports found that exercise performance fell with the progression of chronic renal failure despite the maintenance of hemoglobin concentration [24]. Therefore, the impact of anemia in the impairment of exercise capacity is still controversial. Other potential causes include uremic toxins [25], which have been suggested as a mechanism for impaired physical fitness in CKD, independent of physical inactivity; however, the exact mechanisms remain unknown. Cardiovascular factors such as endothelial dysfunction and high cardiac sympathetic nerve activity, which are common features in CKD, may also contribute to low exercise capacity in this population.
There was an intriguing report which evaluated whether dialysis itself affected exercise capacity. Moore et al. performed peak exercise testing in dialysis patients immediately before and during dialysis and demonstrated that peak VO2 and cardiovascular responses such as cardiac output or stroke volume on testing were not different between the two conditions [26]. These results suggest that dialysis itself has no effect on the acute response to exercise, i.e., the hemodynamic changes on dialysis or water retention could not explain the low physical fitness of dialysis patients.
2 Various Aspects of the Efficacy of Exercise in Patients with CKD
2.1 Delaying the Initiation of Dialysis
Can exercise slow the progression of renal dysfunction in predialytic patients with CKD? There are only a few interventional studies with small cohorts on this issue. A non-randomized aerobic exercise trial [27] and a randomized-controlled resistance training trial [28] have shown an increase in glomerular filtration rate (GFR) in patients with CKD after 12-week interventions. In the predialytic state, a low-protein diet is prescribed in almost all cases to delay the progression of CKD, which leads to muscle atrophy and frailty. As a specific effect on this population, exercise can reduce catabolism by low-protein diet and uremia. No previous study has supported the adverse effect of exercise on the progression of CKD.
2.2 Increasing Physical Fitness and Strength
The effect of exercise on maximum aerobic capacity has been a major concern in many clinical trials with aerobic exercise protocols in dialysis patients. In a systematic review by Cheema et al., 17 of 19 uncontrolled, non-randomized controlled, and randomized controlled trials (RCTs) succeeded in increasing peak VO2 by 17–23 % mainly due to aerobic training for a period of 10 weeks to 4 years in patients maintained on hemodialysis [29]. In another review article, Johansen demonstrated that trials with aerobic exercise training for 8 weeks to 6 months enhanced peak VO2 by 17 %, regardless of the use of erythropoietin to control renal anemia [30]. Smart et al. reported in a more recent systematic review on 365 patients from 8 studies that aerobic training increased peak VO2 from 70 to 88 % of age-predicted value, corresponding to +5.22 ml/kg/min of peak VO2 [31]. The improvements in peak VO2 reported in these studies, although significant, were relatively minor, and the patients could not reach the predicted age-adjusted peak VO2 even after the intervention.
Muscle strength is the most important contributor to physical performance and independence in daily life. Particularly in the last 5 years, several RCTs on dialysis patients concerning the effect of exercise on muscle strength could be found. These studies reported the positive effect of intradialytic resistance training, with or without cycle ergometer, on the muscle strength of lower extremities [32–35]. Furthermore, the ACTINUT study, an RCT against protein-energy wasting and muscle atrophy, is currently ongoing [36]. In addition, a biopsy study also showed the regeneration of degenerated muscle fibers, increase in capillary density, and favorable changes in the structure and number of mitochondria following exercise training in hemodialysis patients [37]. The improvement in functional abilities evaluated by gait speed, stair-climbing, 10-m walking, or timed up and go test was also found to be associated with increased muscle strength.
2.3 Positive Psychiatric Effects
Like many other chronic diseases, CKD probably affects the psychological state of the patients. Based on the high incidence of depression and anxiety among this population, a concept called “psychonephrology” was proposed to refer to this problem, particularly, in patients on maintenance hemodialysis and those who have had a kidney transplant [38]. There are only a few reports that have demonstrated the favorable effects of aerobic exercise on mental health and health-related QOL. Most of these used the Short Form-36 questionnaire, which is frequently used to evaluate QOL in patients on dialysis as well as in the general population [39–41]. For many elderly patients on dialysis, clinical outcomes such as mental status and QOL may become more important than mortality. From this perspective, it is desirable that they increase opportunities to participate in exercise more actively in the clinical settings of dialysis, although further studies on the effects of exercise on mental health in this population are necessary.
2.4 Enhancing Dialysis Efficacy
Reduced hemodialysis efficiency with insufficient removal of toxins leads to the accumulation of uremic toxins and systemic damages in a concentration-dependent manner. To address this issue, the methodologies involved in hemodialysis (HD) such as high-flux HD and online hemodiafiltration (HDF) have been developed, in addition to increasing the dialysis time and frequency. However, the benefits of these therapies are restricted by inter-compartmental resistance, i.e., toxins distributed in intracellular and interstitial compartments are restricted from being removed by cellular membranes or capillary endothelium. Intradialytic exercise increases cardiac output and blood flow to low-perfusion skeletal muscle regions and could possibly increase the exchange between these compartments and the systemic circulation, therefore, enhancing the removal of uremic toxins. Several previous studies have reported the improvement of dialysis efficiency, assessed by Kt/V, clearance of urea and creatinine, and removal of potassium and phosphate, by intradialytic exercise [42, 43]. A prospective-randomized study, comparing toxin removal in online HDF and intradialytic exercise in high-flux HD, is also ongoing [44].
2.5 Improving Glycemic Control
Exercise is an established therapeutic strategy for type 2 diabetes, and several meta-analyses have reported that aerobic exercise, resistance exercise, and a combination of the two improved glycemic control in this population with HbA1c levels reduced by 0.6–0.8 % [45–47]. An elevated HbA1c level is an independent contributor to mortality in diabetic patients with and without CKD and in those on maintenance hemodialysis [48–50]. Therefore, improved glycemic control is a very important aspect of health management in patients with diabetes and CKD.
Insulin resistance is a common finding in diabetes with CKD due to impaired insulin signaling in skeletal muscle cells along with muscle atrophy and metabolic acidosis [51]. These features are often masked by the prolonged half-life of insulin secondary to decrease in both the degeneration of insulin and its renal clearance. DeFronzo et al. demonstrated that peripheral insulin-mediated glucose uptake was decreased in patients on dialysis [10]. For these reasons, exercise may be more suitable than therapies with insulin or insulin secretagogues, for glycemic management in patients with CKD. A recent interventional study revealed that the effects of strength training on glucose tolerance by changing in muscle fiber-type composition in type 2 diabetes on dialysis [52].
2.6 Reducing Cardiovascular Risks
Patients with CKD not on dialysis are at the highest risk of cardiovascular diseases (CVD), and they are more likely to die of CVD than to progress to dialysis [53]. These patients have a high prevalence of traditional risk factors for CVD, such as diabetes, hypertension, and dyslipidemia. Various conditions closely related to CKD itself, including endothelial dysfunction, high sympathetic activity, oxidative stress, and inflammation, may also increase the risk for cardiovascular diseases in this population. Of course physical inactivity itself contributes to the development of CVD; therefore, exercise or physical activity could ameliorate CKD-associated cardiovascular dysfunction [54, 55]. However, there is no available evidence that exercise intervention can reduce the incidence of CVD in patients with CKD.
Many studies have suggested a counterintuitive relationship between obesity, hypertension, or hypercholesterolemia and CVD risk in dialysis patients. The lack of these CVD risk factors, which are generally expected to be ameliorated by exercise, rather involve malnutrition associated with a higher risk of mortality in dialysis patients, and this phenomenon is known as “reverse epidemiology” [56]. Nevertheless, a sedentary lifestyle is still a risk factor for impaired cardiovascular health and high mortality rate in this population [30].
2.7 Reducing Mortality Risks
As mentioned in Sect. 19.1.1.1, several observational studies, including the latest one with a large cohort of dialysis patients, have shown that physical inactivity, assessed by self-report or interview, was associated with an increased risk of mortality among these patients [5–7]. On the other hand, there are only a few studies regarding the effect of exercise intervention on life expectancy in patients on maintenance dialysis. We could only find one study reporting mortality risk reduction by cardiac rehabilitation in dialysis patients who have undergone coronary artery bypass grafting (CABG). Huang et al. have shown that over a 42-month follow-up period, cardiac rehabilitation was associated with a longer cumulative survival time with an increase of up to 76 days in post-CABG dialysis patients [57]. Further longitudinal studies are needed to elucidate whether exercise increases life span in these patients.
3 Exercise Practice for Patients with CKD
3.1 Cardiopulmonary Exercise Test (CPX)
Exercise testing has mainly been used for screening and identification of myocardial ischemia for a long time. However, there are no available guidelines specifically for conducting CPX in patients with CKD or end-stage renal disease. If “asymptomatic” patients intend to perform low-intensity physical activity such as walking, routine CPX before the participation is not necessarily recommended, because it does not seem to have any significant benefits on serious clinical outcomes even among patients with diabetes who are at a high risk for silent ischemia [58]. In addition, requiring routine CPX before exercise may rather be obstructive to participating in exercises for the patients. Consequently, it is unlikely that pre-exercise CPX is essential for all patients with CKD or end-stage renal disease.
Even so, in a joint statement in 2010, the American College of Sports Medicine (ACSM) and the American Diabetes Association (ADA) proposed recommendations for exercise testing in patients with diabetes. They stated that exercise testing was advisable in patients with type 2 diabetes who had previously been sedentary and now planned to undertake physical activity of an intensity more than brisk walking [59]. The American Heart Association (AHA) guidelines also recommend that individuals at a greater risk for CVD, such as the patients with diabetes or known CVD, should be considered for exercise testing before participating in vigorous exercise programs [60]. According to these statements, it is preferable that patients with CKD, who are also at a high risk for CVD, undergo CPX not only for screening but also for the evaluation of prognosis and effect of therapy, including exercise interventions, if all necessary equipment for the testing is available. According to the AHA guidelines for exercise testing in older people, the testing must include an adequate warm-up period at low resistance and progress with a modest and almost imperceptible increment in exercise load [60]. Such modulation is also ideal for exercise testing in patients with CKD.
A previous survey reported that one to two myocardial infarctions and sustained ventricular tachycardia per 10,000 tests are expected [61]. Therefore, exercise testing is considered an extremely safe procedure. However, a defibrillator and other appropriate medications should be immediately available, and physicians supervising the test must be trained in cardiopulmonary resuscitation.
3.2 Exercise Prescription for Dialysis Patients
The aforementioned K/DOQI guidelines recommend that dialysis patients should participate in cardiovascular (aerobic) exercise at a moderate intensity for 30 min most, if not all, days per week [1]. Moderate intensity generally means exercising to reach a target heart rate of 60–70 % of the maximal heart rate (220-age). However, exercise prescriptions based on heart rate may not be suitable for dialysis patients, because their fluid status and medications such as β-adrenergic blocking agents can affect their heart rate response to exercise. If the dialysis patients have undergone CPX, the physicians can prescribe the exercise load at an aerobic threshold as an appropriate intensity. Another way to determine exercise intensity for dialysis patients is to use the highly reproducible Borg’s rating of perceived exertion (RPE) scale, which ranges from 6 (no exertion at all) to 20 (maximal exertion) and is widely used to assess perceived patients’ exertion during an exercise test in clinical settings. Because a rating of “13 (somewhat hard)” corresponds approximately to an aerobic threshold, around “13” on the scale can be adopted as the target exercise intensity. It is also important to provide several minutes of warm-up at very low levels before the true exercise period followed by an adequate cooling-down period.
In addition to aerobic exercise, resistance training is also recommended with the aim of strengthening the muscles of the lower extremities. This program has to be individualized and gradually stepped up from bodyweight exercise, using the patient’s own weight against gravity as resistance to weight training of the hand and ankle weights or Thera-Bands. Furthermore, combining this with flexibility exercises may enhance the efficacy of resistance training and activity level during walking by improving the range of motion and stride.
3.3 Special Concerns About Exercise for Diabetes with CKD
In the ACSM/ADA joint position statement of 2010, it was suggested that exercise for diabetes with nephropathy should be started with a low intensity and volume because of reduced aerobic capacity and muscle function in this population [59]. These are essential to avoid the patients from feeling that the exercise program is difficult and losing motivation and to prevent them from unexpected injuries. The statement also mentioned exercise with Valsalva maneuver, which may not only cause retinal hemorrhage in patients with active retinopathy but also cause an excessive elevation of blood pressure (BP) [59]. Although, in many cases, an increase in urinary albumin excretion by BP elevation during exercise is transient, the patients should keep away from further otiose hypertension caused by vigorous exercise. Tight BP control along with the treatment and low-intensity exercise are more beneficial in delaying the progression of microalbuminuria [62].
If a patient is relatively fit and is able to exercise for longer than 1 h even at low intensity, frequent monitoring of blood glucose may minimize the risk of exercise-induced hypoglycemia. The patient may need to decrease insulin dosage by one or two units and take additional carbohydrate snacks before each exercise session. For better glucose control, both the patient and physician must regularly discuss the timing of drug and insulin administration and food intake prior to exercise.
3.4 When Should Dialysis Patients Exercise?
3.4.1 Exercise During Dialysis
Patients on hemodialysis can participate in facility-based exercise programs during their dialysis sessions. Investigators have shown favorable efficacy of intradialytic exercise training on exercise capacity, muscle strength, functional ability, and mental health [63]. In many cases, exercise professionals supervise intradialytic exercise; therefore, the patients can be advised on how to perform appropriate exercises based on individual exercise prescriptions. In addition, intradialytic exercise is expected to enable the patients to save time, improve adherence to exercise, and avoid boredom during the dialysis treatment. Indeed, exercise programs during dialysis sessions often have lower dropout rates than outpatient exercise programs on non-dialysis days [64].
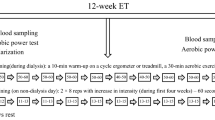
We also have incorporated an intradialytic exercise program into our partnered artificial dialysis facilities. Many patients begin the training program early in the dialysis treatment, which is composed of aerobic exercise using a recumbent cycle ergometer, strength training using silicone rubber bands or ankle weights, and flexibility exercises (Fig. 19.1a, b). The athletic trainers supervise each session and encourage the participants to perform exercise with adequate intensity and procedure. The program has been very successful, particularly, in improving peak VO2 and physical function.
Exercise professionals often need to be involved in intradialytic exercise programs because dialysis staff may not have the skills or the inclination to attend the exercising patients. Furthermore, exercise performed during dialysis is usually limited to leg exercise because of vascular access complications; therefore, the patient needs to have another opportunity to exercise the upper extremities.
3.4.2 Home-Based Exercise on Non-dialysis Days
Home-based exercise training is an alternative for hemodialysis patients. This type of training enables them to exercise at their own convenience without being concerned about fatigue during dialysis, therefore resulting in a low dropout rate. Koh et al. in a randomized 6-month interventional study reported that home-based exercise training equally improved exercise capacity as intradialytic exercise in hemodialysis patients, if they could conduct home-based exercise with appropriate volume or intensity as advised [65]. However, because such programs cannot be supervised at all, it is difficult to ensure patient compliance with exercise. Whether the patients conduct exercise or not may be influenced by their motivation and mood. Therefore, in home-based exercise, practicing time and intensity are often insufficient, and there are a few studies, which report the effects of home-based exercise on cardiorespiratory function to be less than intradialysis exercise [64].
3.4.3 In-facility Exercise Programs on Non-dialysis Days
Although some dialysis facilities provide outpatient exercise programs, availability is still too low for the majority of dialysis patients to benefit from them. The survey in DOPPS (2005–2006) reported that only 10–20 % of facilities offered exercise programs to patients not on dialysis in most DOPPS countries [2]. Furthermore, patients’ adherence to such programs is not satisfactory. They often stop participating because of lack of spare time on non-dialysis days, transportation difficulties, high morbidity, and lack of motivation [63, 64]. Nevertheless, such supervised exercise training on non-dialysis days enables them to perform more aggressive, high-intensity exercise and further sports activities involving the movement of upper extremities, which is difficult during the dialysis treatment because of vascular access. Therefore, this method of exercise program has the greatest effect on improving cardiorespiratory capacity in maintenance hemodialysis patients as compared with intradialytic or home-based exercise [64].
3.5 The Aspects of Sustainability of Exercise Programs
Because it is widely known that the benefits of exercise are lost within a few weeks of non-training (detraining), it is important to incorporate exercise programs into routine care in the population on dialysis. Bennett et al. tried to identify contributing factors for sustaining hemodialysis exercise programs by examining 28 original studies and 14 review articles [66]. Other than the aforementioned elements necessary for improving patients’ adherence to exercise, such as exercise professionals’ involvement and conducting exercise programs during dialysis sessions, they identified other factors as follows: commitment to exercise by dialysis and medical staff, adequate space and equipment, interesting and stimulating programs or interventions, and addressing cost implications. Although it is not easy to meet all these requirements, the staffs in dialysis facilities need to make some effort toward creating an atmosphere in which the patients feel like participating continuously in physical activity or exercise.
In regard to the patients on continuous ambulatory peritoneal dialysis (CAPD), exercise capacity estimated by peak VO2 and aerobic threshold remain unchanged regardless of whether the dialysis solution was retained in the peritoneal cavity or drained. However, exercise time was significantly shortened when the solution was retained directly due to the carrying of extra weight [67]. Therefore, exercising under conditions in which the solution is drained may be sustainable for this population.
In addition, difficulty in sustaining physical activity in patients with CKD has been attributed to anemia. A meta-analysis by Johansen et al. demonstrated that the treatment of anemia using erythropoiesis-stimulating agents significantly increased peak VO2 by 24 % in hemodialysis patients, although many studies included were not controlled trials [68]. The improvement in anemia probably contributes to the sustainability of exercise, because the patients are freed from shortness of breath and fatigue, which are both major barriers to continuously participating in exercise programs, and could overcome discomfort or pain during exercise.
3.6 Safety of Exercise for Patients with CKD
There is a very small amount of data from controlled studies to discuss or quantify the risks of exercise-induced adverse events in patients with CKD. However, to date, no published study on exercise training in hemodialysis patients has reported deaths or cardiovascular events. Therefore, it does not seem that exercise in dialysis patients necessarily carries additional risks.
According to a scientific statement from AHA, although the incidence of fatal and nonfatal cardiovascular events during exercise varies with the prevalence of cardiac disease in a study population, the rate is extremely low even among patients with chronic heart failure undertaking cardiac rehabilitation programs [69]. In contrast, the relative risk of cardiac arrest during vigorous exercise as compared with risk at other times is notably greater in patients who are unaccustomed to exercise [70]. Regular exercise participation, performed at low to moderate intensity, reduces the long-term risk of cardiac events both during exercise and at rest.
The exercise guide for nephrologists developed by the Life Options Rehabilitation Advisory Council suggested that to minimize the risks of exercise, nephrologists must be sure to provide adequate dialysis, manage ongoing medical concerns, control hypertension, and respond to symptoms suggestive of cardiac disease [71].
4 Summary
Patients with CKD, particularly, those on dialysis, have been exposed to the environment where they had a few opportunities to participate in physical activity due to conventional therapeutic policy. However, now it is evident that a sedentary lifestyle and low physical fitness result in a poor prognosis for this population. To engage all the patients in regular exercise, it is important that both patients and nephrologists discuss exercise positively and that the medical staff encourage the patients to exercise with a customized training program according to individual physical strength, medical concerns, and convenience.
References
K/DOQI Workgroup (2005) K/DOQI clinical practice guidelines for cardiovascular disease in dialysis patients. Am J Kidney Dis 45:S1–S153
Tentori F, Elder SJ, Thumma J, Pisoni RL, Bommer J, Fissell RB et al (2010) Physical exercise among participants in the Dialysis Outcomes and Practice Patterns Study (DOPPS): correlates and associated outcomes. Nephrol Dial Transplant 25:3050–3062
Johansen KL, Chertow GM, Ng AV, Mulligan K, Carey S, Schoenfeld PY et al (2000) Physical activity levels in patients on hemodialysis and healthy sedentary controls. Kidney Int 57:2564–2570
Kim JC, Shapiro BB, Zhang M, Li Y, Porszasz J, Bross R et al (2014) Daily physical activity and physical function in adult maintenance hemodialysis patients. J Cachex Sarcopenia Muscle 5:209–220
O’Hare AM, Tawney K, Bacchetti P, Johansen KL (2003) Decreased survival among sedentary patients undergoing dialysis: results from the dialysis morbidity and mortality study wave 2. Am J Kidney Dis 41:447–454
Stack AG, Molony DA, Rives T, Tyson J, Murthy BV (2005) Association of physical activity with mortality in the US dialysis population. Am J Kidney Dis 45:690–701
Lopes AA, Lantz B, Morgenstern H, Wang M, Bieber BA, Gillespie BW et al (2014) Associations of self-reported physical activity types and levels with quality of life, depression symptoms, and mortality in hemodialysis patients: the DOPPS. Clin J Am Soc Nephrol 9:1702–1712
Avesani CM, Trolonge S, Deleaval P, Baria F, Mafra D, Faxen-Irving G et al (2012) Physical activity and energy expenditure in hemodialysis patients: an international survey. Nephrol Dial Transplant 27:2430–2434
Delgado C, Johansen KL (2012) Barriers to exercise participation among dialysis patients. Nephrol Dial Transplant 27:1152–1157
Delgado C, Johansen KL (2010) Deficient counseling on physical activity among nephrologists. Nephron Clin Pract 116:c330–c336
Johansen KL, Painter P (2012) Exercise in individuals with CKD. Am J Kidney Dis 59:126–134
Leehey DJ, Moinuddin I, Bast JP, Qureshi S, Jelinek CS, Cooper C et al (2009) Aerobic exercise in obese diabetic patients with chronic kidney disease: a randomized and controlled pilot study. Cardiovasc Diabetol 8:62
Johansen KL, Shubert T, Doyle J, Soher B, Sakkas GK, Kent-Braun JA (2003) Muscle atrophy in patients receiving hemodialysis: effects on muscle strength, muscle quality, and physical function. Kidney Int 63:291–297
McIntyre CW, Selby NM, Sigrist M, Pearce LE, Mercer TH, Naish PF (2006) Patients receiving maintenance dialysis have more severe functionally significant skeletal muscle wasting than patients with dialysis-independent chronic kidney disease. Nephrol Dial Transplant 21:2210–2216
Spindler A, Paz S, Berman A, Lucero E, Contino N, Penalba A et al (1997) Muscular strength and bone mineral density in hemodialysis patients. Nephrol Dial Transplant 12:128–132
Cook WL, Jassal SV (2005) Prevalence of falls among seniors maintained on hemodialysis. Int Urol Nephrol 37:649–652
Cook WL, Tomlinson G, Donaldson M, Markowitz SN, Naglie G, Sobolev B et al (2006) Falls and fall-related injuries in older dialysis patients. Clin J Am Soc Nephrol 1:1197–1204
Myers J, Prakash M, Froelicher V, Do D, Partington S, Atwood JE (2002) Exercise capacity and mortality among men referred for exercise testing. N Engl J Med 346:793–801
Sietsema KE, Amato A, Adler SG, Brass EP (2004) Exercise capacity as a predictor of survival among ambulatory patients with end-stage renal disease. Kidney Int 65:719–724
Kohl Lde M, Signori LU, Ribeiro RA, Silva AM, Moreira PR, Dipp T et al (2012) Prognostic value of the six-minute walk test in end-stage renal disease life expectancy: a prospective cohort study. Clinics 67:581–586
Laegreid IK, Aasarod K, Bye A, Leivestad T, Jordhoy M (2014) The impact of nutritional status, physical function, comorbidity and early versus late start in dialysis on quality of life in older dialysis patients. Ren Fail 36:9–16
Oliveira CM, Costa SP, Costa LC, Pinheiro SM, Lacerda GA, Kubrusly M (2012) Depression in dialysis patients and its association with nutritional markers and quality of life. J Nephrol 25:954–961
Da Silva-Gane M, Wellsted D, Greenshields H, Norton S, Chandna SM, Farrington K (2012) Quality of life and survival in patients with advanced kidney failure managed conservatively or by dialysis. Clin J Am Soc Nephrol 7:2002–2009
Leikis MJ, McKenna MJ, Petersen AC, Kent AB, Murphy KT, Leppik JA et al (2006) Exercise performance falls over time in patients with chronic kidney disease despite maintenance of hemoglobin concentration. Clin J Am Soc Nephrol 1:488–495
Moore GE, Parsons DB, Stray-Gundersen J, Painter PL, Brinker KR, Mitchell JH (1993) Uremic myopathy limits aerobic capacity in hemodialysis patients. Am J Kidney Dis 22:277–287
Moore GE, Brinker KR, Stray-Gundersen J, Mitchell JH (1993) Determinants of VO2 peak in patients with end-stage renal disease: on and off dialysis. Med Sci Sports Exerc 25:18–23
Toyama K, Sugiyama S, Oka H, Sumida H, Ogawa H (2010) Exercise therapy correlates with improving renal function through modifying lipid metabolism in patients with cardiovascular disease and chronic kidney disease. J Cardiol 56:142–146
Castaneda C, Gordon PL, Uhlin KL, Levey AS, Kehayias JJ, Dwyer JT et al (2001) Resistance training to counteract the catabolism of a low-protein diet in patients with chronic renal insufficiency. A randomized, controlled trial. Ann Intern Med 135:965–976
Cheema BS, Singh MA (2005) Exercise training in patients receiving maintenance hemodialysis: a systematic review of clinical trials. Am J Nephrol 25:352–364
Johansen KL (2007) Exercise in the end-stage renal disease population. J Am Soc Nephrol 18:1845–1854
Smart N, Steele M (2011) Exercise training in hemodialysis patients: a systematic review and meta-analysis. Nephrology 16:626–632
Cheema B, Abas H, Smith B, O’Sullivan A, Chan M, Patwardhan A et al (2007) Randomized controlled trial of intradialytic resistance training to target muscle wasting in ESRD: the Progressive Exercise for Anabolism in Kidney Disease (PEAK) study. Am J Kidney Dis 50:574–584
Dong J, Sundell MB, Pupim LB, Wu P, Shintani A, Ikizler TA (2011) The effect of resistance exercise to augment long-term benefits of intradialytic oral nutritional supplementation in chronic hemodialysis patients. J Ren Nutr 21:149–159
Song WJ, Sohng KY (2012) Effects of progressive resistance training on body composition, physical fitness and quality of life of patients on hemodialysis. J Korean Acad Nurs 42:947–956
Paglialonga F, Lopopolo A, Scarfia RV, Consolo S, Galli MA, Salera S et al (2014) Intradialytic cycling in children and young adults on chronic hemodialysis. Pediatr Nephrol 29:431–438
Magnard J, Deschamps T, Cornu C, Paris A, Hristea D (2013) Effects of a six-month intradialytic physical ACTIvity program and adequate NUTritional support on protein-energy wasting, physical functioning and quality of life in chronic hemodialysis patients: ACTINUT study protocol for a randomized controlled trial. BMC Nephrol 14:259
Kouidi E, Albani M, Natsis K, Megalopoulos A, Gigis P, Guiba-Tziampiri O et al (1998) The effects of exercise training on muscle atrophy in hemodialysis patients. Nephrol Dial Transplant 13:685–699
Levy NB (2008) What is psychonephrology? J Nephrol 21(Suppl 13):S51–S53
Levendoglu F, Altintepe L, Okudan N, Ugurlu H, Gokbel H, Tonbul Z et al (2004) A twelve week exercise program improves the psychological status, quality of life and work capacity in hemodialysis patients. J Nephrol 17:826–832
Matsumoto Y, Furuta A, Furuta S, Miyajima M, Sugino T, Nagata K et al (2007) The impact of pre-dialytic endurance training on nutritional status and quality of life in stable hemodialysis patients (Sawada study). Ren Fail 29:587–593
Carney RM, Templeton B, Hong BA, Harter HR, Hagberg JM, Schechtman KB et al (1987) Exercise training reduces depression and increases the performance of pleasant activities in hemodialysis patients. Nephron 47:194–198
Farese S, Budmiger R, Aregger F, Bergmann I, Frey FJ, Uehlinger DE (2008) Effect of transcutaneous electrical muscle stimulation and passive cycling movements on blood pressure and removal of urea and phosphate during hemodialysis. Am J Kidney Dis 52:745–752
Giannaki CD, Stefanidis I, Karatzaferi C, Liakos N, Roka V, Ntente I et al (2011) The effect of prolonged intradialytic exercise in hemodialysis efficiency indices. ASAIO J 57:213–218
Maheshwari V, Samavedham L, Rangaiah GP, Loy Y, Ling LH, Sethi S et al (2012) Comparison of toxin removal outcomes in online hemodiafiltration and intradialytic exercise in high-flux hemodialysis: a prospective randomized open-label clinical study protocol. BMC Nephrol 13:156
Boule NG, Kenny GP, Haddad E, Wells GA, Sigal RJ (2003) Meta-analysis of the effect of structured exercise training on cardiorespiratory fitness in Type 2 diabetes mellitus. Diabetologia 46:1071–1081
Snowling NJ, Hopkins WG (2006) Effects of different modes of exercise training on glucose control and risk factors for complications in type 2 diabetic patients: a meta-analysis. Diabetes Care 29:2518–2527
Umpierre D, Ribeiro PA, Kramer CK, Leitao CB, Zucatti AT, Azevedo MJ et al (2011) Physical activity advice only or structured exercise training and association with HbA1c levels in type 2 diabetes: a systematic review and meta-analysis. J Am Med Assoc 305:1790–1799
Morioka T, Emoto M, Tabata T, Shoji T, Tahara H, Kishimoto H et al (2001) Glycemic control is a predictor of survival for diabetic patients on hemodialysis. Diabetes Care 24:909–913
Oomichi T, Emoto M, Tabata T, Morioka T, Tsujimoto Y, Tahara H et al (2006) Impact of glycemic control on survival of diabetic patients on chronic regular hemodialysis: a 7-year observational study. Diabetes Care 29:1496–1500
Kalantar-Zadeh K, Kopple JD, Regidor DL, Jing J, Shinaberger CS, Aronovitz J et al (2007) A1C and survival in maintenance hemodialysis patients. Diabetes Care 30:1049–1055
Bailey JL (2013) Insulin resistance and muscle metabolism in chronic kidney disease. ISRN Endocrinol 2013:329606
Molsted S, Harrison AP, Eidemak I, Dela F, Andersen JL (2013) Improved glucose tolerance after high-load strength training in patients undergoing dialysis. Nephron Clin Pract 123:134–141
Schiffrin EL, Lipman ML, Mann JF (2007) Chronic kidney disease: effects on the cardiovascular system. Circulation 116:85–97
Mustata S, Chan C, Lai V, Miller JA (2004) Impact of an exercise program on arterial stiffness and insulin resistance in hemodialysis patients. J Am Soc Nephrol 15:2713–2718
Kouidi E, Karagiannis V, Grekas D, Iakovides A, Kaprinis G, Tourkantonis A et al (2010) Depression, heart rate variability, and exercise training in dialysis patients. Eur J Cardiovasc Prev Rehabil 17:160–167
Levin NW, Handelman GJ, Coresh J, Port FK, Kaysen GA (2007) Reverse epidemiology: a confusing, confounding, and inaccurate term. Semin Dial 20:586–592
Huang Y, Zhang R, Culler SD, Kutner NG (2008) Costs and effectiveness of cardiac rehabilitation for dialysis patients following coronary bypass. Kidney Int 74:1079–1084
Lievre MM, Moulin P, Thivolet C, Rodier M, Rigalleau V, Penfornis A et al (2011) Detection of silent myocardial ischemia in asymptomatic patients with diabetes: results of a randomized trial and meta-analysis assessing the effectiveness of systematic screening. Trials 12:23
Colberg SR, Albright AL, Blissmer BJ, Braun B, Chasan-Taber L, Fernhall B et al (2010) Exercise and type 2 diabetes: American College of Sports Medicine and the American Diabetes Association: joint position statement. Exercise and type 2 diabetes. Med Sci Sports Exerc 42:2282–2303
Fletcher GF, Ades PA, Kligfield P, Arena R, Balady GJ, Bittner VA et al (2013) Exercise standards for testing and training: a scientific statement from the American Heart Association. Circulation 128:873–934
Myers J, Voodi L, Umann T, Froelicher VF (2000) A survey of exercise testing: methods, utilization, interpretation, and safety in the VAHCS. J Cardiopulm Rehabil 20:251–258
Lazarevic G, Antic S, Vlahovic P, Djordjevic V, Zvezdanovic L, Stefanovic V (2007) Effects of aerobic exercise on microalbuminuria and enzymuria in type 2 diabetic patients. Ren Fail 29:199–205
Cheema BS, Smith BC, Singh MA (2005) A rationale for intradialytic exercise training as standard clinical practice in ESRD. Am J Kidney Dis 45:912–916
Konstantinidou E, Koukouvou G, Kouidi E, Deligiannis A, Tourkantonis A (2002) Exercise training in patients with end-stage renal disease on hemodialysis: comparison of three rehabilitation programs. J Rehabil Med 34:40–45
Koh KP, Fassett RG, Sharman JE, Coombes JS, Williams AD (2010) Effect of intradialytic versus home-based aerobic exercise training on physical function and vascular parameters in hemodialysis patients: a randomized pilot study. Am J Kidney Dis 55:88–99
Bennett PN, Breugelmans L, Barnard R, Agius M, Chan D, Fraser D et al (2010) Sustaining a hemodialysis exercise program: a review. Semin Dial 23:62–73
Ohmura N, Tamura H, Kawaguchi Y, Ohta M, Miyahara T (1989) The influence of dialysis solution on the exercise capacity in patients on CAPD. Adv Perit Dial Conf Perit Dial 5:46–48
Johansen KL, Finkelstein FO, Revicki DA, Gitlin M, Evans C, Mayne TJ (2010) Systematic review and meta-analysis of exercise tolerance and physical functioning in dialysis patients treated with erythropoiesis-stimulating agents. Am J Kidney Dis 55:535–548
Thompson PD, Franklin BA, Balady GJ, Blair SN, Corrado D, Estes NA 3rd et al (2007) Exercise and acute cardiovascular events placing the risks into perspective: a scientific statement from the American Heart Association Council on Nutrition, Physical Activity, and Metabolism and the Council on Clinical Cardiology. Circulation 115:2358–2368
Siscovick DS, Weiss NS, Fletcher RH, Lasky T (1984) The incidence of primary cardiac arrest during vigorous exercise. N Engl J Med 311:874–877
Painter P, Blagg CR, Moore GE (1995) The risks. In: Exercise for the dialysis patients: a guide for the nephrologist. The life options administered by the Medical Education Institute, Inc. http://lifeoptions.org/catalog/. Accessed 16 Jan 2015
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer Japan
About this chapter
Cite this chapter
Yokoyama, H. (2016). Clinical Application of Exercise Therapy in Diabetes with Chronic Kidney Disease. In: Inaba, M. (eds) Musculoskeletal Disease Associated with Diabetes Mellitus. Springer, Tokyo. https://doi.org/10.1007/978-4-431-55720-3_19
Download citation
DOI: https://doi.org/10.1007/978-4-431-55720-3_19
Published:
Publisher Name: Springer, Tokyo
Print ISBN: 978-4-431-55719-7
Online ISBN: 978-4-431-55720-3
eBook Packages: MedicineMedicine (R0)