Abstract
The sensory epithelium of the cochlear duct, called the organ of Corti, is the hearing organ of mammals including human. The organ of Corti is a masterpiece of cellular micro-architecture. The organ of Corti consists of two subtypes of hair cells, a single row of inner and three rows of outer hair cells, and several subtypes of supporting cells strategically positioned on the basilar membrane. The organization of these cells maximizes the extraction of sound energy by amplifying sound-induced basilar membrane motion and transmitting those movements, via the help of the tectorial membrane, to inner hair cells. In this section, an overview of cochlear development is presented and four important events during cochlear development are described: extension of cochlear duct, prosensory specification, cell-cycle exit, and cellular differentiation. Some of the most recent and enlightening results regarding the molecular mechanism underlying the formation of the organ of Corti are also discussed. Most data are from the experiments using mice, the most comprehensive model system for the developing mammalian cochlea.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Extension of Cochlear Duct
The mammalian cochlear duct is a coiled structure resembling the shape of a snail, while the avian cochlear duct (basilar papilla) is relatively straight. In mice, the cochlear duct first arises as a ventral outpocketing from a posterolateral region of the developing otocyst beginning around E11 [1] and descends ventromedially forming an L-shaped organ (a half turn) by embryonic day 12. Then, the cochlear duct continues to extend and coil with the mature mouse structure consisting of one and three quarter turns (Fig. 12.1). Similar to other mammals, the mouse cochlear duct is tonotopically organized such that the base of the cochlear duct is most sensitive to high frequency sounds and the apical region to low frequency sounds [3].
1.1 Factors Regulating Cochlear Duct Extension
Multiple extrinsic factors regulate proper outgrowth and extension of the cochlear duct. One clear example is Shh secreted from the notochord and floor plate. Similar ventral phenotypes occur in Shh−/− ears and ears in chicken embryos in which the ventral midline has been ablated, including agenesis of the cochlear duct [4, 5].
Additionally, it is well established that a mesenchymal contribution to cochlear duct formation is also important [6]. Two transcription factors, Tbx1 and Pou3f4 (Brn4), of which the latter is expressed only in the otic mesenchyme, have been implicated so far. Lack of Tbx1 or Pou3f4 in the otic mesenchyme can lead to abnormal coiling or shortening of the cochlear duct, and these two pathways have been shown to interact genetically [7–9]. One possible mediator of these effects is RA, as both of these transcription factors are believed to induce expression of the RA degradation enzyme Cyp26 in the periotic mesenchyme [9].
1.2 Convergent Extension
The cochlear duct is composed of pseudostratified epithelial cells derived from the otic placode. Even at the earliest time points following the initiation of cochlear outgrowth, the dorsal half of the duct, typically referred to as the floor, is already comprised of a notably thickened epithelium that contains five or six layers of cells [10, 11]. The organ of Corti arises from the floor of cochlear duct and the final pattern contains two layers of cells: a hair cell layer and a supporting cell layer.
Accumulating evidence suggests that the growth of the cochlear duct is regulated by not only proliferation of cells but also convergent extension movements [6, 12–14]. Genes within the planar cell polarity pathway such as Dishevelled1–3, Vangl2, and Celsr1 are thought to be involved in this process [15–17] (Fig. 12.2).
2 Prosensory Specification
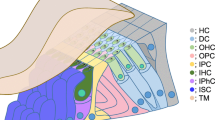
The first step in the development of the organ of Corti is believed to be the specification of the prosensory domain [18] (Fig. 12.3).
Development of the organ or Corti. (Modified from [19])
Notch signaling seemed to the main effector of sensory specification, and other signaling pathways such as FGF [20–22] and Bmp signaling [23] also contribute to the sensory specification.
Before E12.5 all of the epithelial cells that compose the floor of the cochlear duct have a similar morphology; however, even at these early time points, a subset of cells express the prosensory markers Sox2, Jagged1 (Jag1, also referred to as Serrate1 in the chick), Lunatic Fringe (Lfng), Fgf10, and bone morphogenetic protein 4 (Bmp4), and they have been implicated in the specification of prosensory domain [1, 23] (Fig. 12.4).
Markers in prosensory formation. (Modified from [23])
2.1 Notch Signaling and Prosensory Formation
Jag1 and Lfng are expressed in patterns that are largely consistent with a role in specification of prosensory patches. Jag1 and Lfng are both components of the Notch signaling pathway, with Jag1 acting as a ligand for Notch, while Lfng modulates the activity of some Notch ligands [24, 25].
Lateral inhibition is the well-established role of Notch signaling in the regulation of cellular differentiation within prosensory domains and will be discussed in a subsequent section. But more recent experiments have illuminated an additional role for Notch signaling in the specification of prosensory domains, called Notch induction.
Although less well understood, the mechanism of inductive signaling differs from lateral inhibition and typically involves positive feedback rather than negative feedback [26].
Analyses of inner ears from Jag1-deficient mice reveal a decrease in the overall size of the sensory epithelia [27–31]. Similarly, deletion of Rbp-Jk, a transcriptional repressor that is required for all Notch function, leads to a complete absence of all vestibular epithelia and to a nearly complete loss of all cochlear hair cells as well [32].
To the contrary, overexpression of an activated form of Notch1, NICD (Notch-intracellular domain), in non-sensory regions of chick and mouse inner ear leads to the formation of ectopic sensory patches [31, 33–36]. Interestingly, ectopic activation of Notch can only induce ectopic sensory patches within a certain time window of development, suggesting other effectors may contribute to sensory specification [35, 36].
The HMG-box transcription factor Sox2 is thought to specify the auditory prosensory domain. Sensory precursors fail to develop in Sox2-deficient inner ears, leading to loss of hair and supporting cells [37] and forced expression of Sox2 in vivo induces ectopic sensory patches [36].
Sox2 has been suggested to be a downstream target of Jag1-Notch signaling. Sox2 expression is reduced in Jag1 conditional mutants [29] and activation of Notch leads to ectopic Jag1 and Sox2 expression [34, 36], suggesting that Jag1-Notch signaling acts upstream of Sox2.
2.2 FGF Signaling and Prosensory Formation
Fibroblast growth factors (FGFs) have been shown to be of critical importance for the development of the organ of Corti. In mice, FGF signaling has been implicated in the early inductive events of the otic vesicle (Chap. 11). In the next phase of cochlear development, at the sensory specification phase, FGF signaling is again thought to be required. Tissue-specific deletion of Fgfr1 results in severe defects in the development of both hair cells and support cells [20]. Subsequent studies have identified the likely ligand for this receptor as Fgf20 [21]. Fgf20 is specifically required for the differentiation of the lateral compartment of the cochlea (which includes OHCs and Deiters’cells) [22]. In vitro rescue experiments indicate that FGF signaling is downstream of Notch, as disruption of prosensory development by the Notch inhibitor g-secretase can be partially rescued by exogenous application of Fgf20 [38]. Interestingly, exogenous application of Fgf20 also restores Sox2 expression indicating that Fgf can independently control Sox2 expression [38]. However, the phenotype of Fgf20 knockout cochlea is relatively mild and the lateral compartment is partially formed [22]. Other ligands may be act for prosensory specification and Fgf10 is a good candidate because of the expression pattern in E11.5–E13.5 prosensory domain overlapping with Jag1 and Sox2 [23] (Fig. 12.4). Fgf10 knockout cochlea is shortened but otherwise normal indicating the possibility of compensation by other FGFs [39].
2.3 Bmp4 Signaling and Prosensory Formation
Bmp4 is one of the regulators for development of sensory domain in the cochlea. Bmp4 is expressed adjacent to the developing prosensory domain of the cochlea in cells destined to become Hensen’s and Claudius’ cells of the outer sulcus [1]. Ohyama et al. showed that BMP signaling is necessary for the development of the outer sulcus and the prosensory domain and that BMP4 suppresses medial markers but promotes lateral markers, suggesting that a gradient of BMP signaling is an important step in patterning the cochlea across its modiolar-to-strial axis.
3 Cell-Cycle Exit
Following specification of the prosensory domain, a subsequent important step is cell-cycle exit within this domain. Cells in the presumptive organ of Corti can first be distinguished by cell-cycle exit during mid-embyrogenesis. This termination of mitosis occurs sequentially across the length of the cochlea, with cells at the apex exiting first from the cell cycle at E12 and cells at the base terminating mitosis last at E15 [40] (Fig. 12.3).
3.1 p27Kip1
The cyclin-dependent kinase inhibitor, p27Kip1, is involved in the maintenance of this nonproliferative state [41] (Fig. 12.3). p27Kip1 is one of the earliest known markers of the presumptive organ of Corti, and its expression correlates with the onset of mitotic exit [41, 42]. In p27Kip1−/− null mutants, hair and supporting cells continue proliferation after E15 resulting in extra hair and supporting cells [41].
p27Kip1 expression overlaps Sox2 expression and, as differentiation advances, Sox2 and p27Kip1 are gradually limited to supporting cells and become undetectable in HCs by birth [29, 37, 41, 43]. p27Kip1 has been shown to keep postnatal supporting cells quiescent [44, 45]. Induced ablation of Sox2 in postnatal inner pillar cells causes p27Kip1 downregulation and proliferation suggesting Sox2 is one of the upstream regulators of p27Kip1 to maintain the quiescent state of postmitotic inner pillar cells [46].
3.2 pRb
The retinoblastoma protein pRb is the protein product of Rb tumor suppressor gene and a key cell-cycle inhibitor. pRb is present in all cells in the E12.5 otocyst and its expression is prominent in hair cells during embryonic and adult ages [47]. Deletion of Rb in cochlear prosensory cells produces supernumerary hair cells and supporting cells and later apoptotic hair cell death [48]. Inducible inactivation of Rb in postnatal cochlear hair cells causes cell-cycle reentry and cell death [49]. Inducible inactivation of Rb in postnatal pillar cells and Deiters’ cells results in cell-cycle reentry and maintain their supporting cell fate [50].
3.3 p21cip1 and p19ink4d
Two additional cyclin-dependent kinase inhibitor, p21cip1 and p19ink4d, are also expressed in the prosensory domain beginning between E14.5 and E16.5 [51, 52] and suggested to maintain the nonproliferative state. While deletion of p21cip1 has no apparent effect of maintenance of mitotic quiescence [52], loss of p19ink4d results in an increasing rate of spontaneous hair cell mitoses beginning in the postnatal period [51]. Deletion of both p19ink4d and p21cip1 results in initiation of mitosis in hair cells beginning on P3 [52].
3.4 Wnt Signaling and Lgr5
Wnt/b-catenin regulates proliferation within the mitotic prosensory domain of E12 mouse cochlea, and Wnt activity becomes reduced as development progresses [53]. Later in development, low-level Wnt reporter activity is still maintained at E17in pillar cells and in the third row of Deiters’ cells [53].
Leucine-rich repeat-containing G-protein-coupled receptor 5 (Lgr5), a Wnt target gene, has been shown to mark endogenous stem cells in rapidly proliferating organs [54, 55]. In the postnatal cochlea, Lgr5 expression is Wnt dependent and limited to supporting cell subtypes [56]. Lgr5-positive cells isolated by flow cytometry from neonatal Lgr5EGFP-CreERT2/+ mice proliferated, formed clonal colonies, and differentiated into hair cells [57, 58]. Moreover, both in vitro and in vivo, Wnt signaling enhanced proliferation of Lgr5-positive cells [58]. These data indicate that Lgr5 marks Wnt-regulated sensory precursor cells in the postnatal cochlea and Wnt signaling can promote their proliferation.
4 Cellular Differentiation
Following specification of the prosensory domain and cell-cycle exit, individual prosensory cells become determined to develop as all of the unique cell types within the organ of Corti. The first indication of cellular differentiation within the prosensory domain is inner hair cells observed in the mid-basal region of the cochlea between E14 and E15. Inner hair cell differentiation then proceeds in a gradient that extends toward both the apex and the base of the cochlear spiral [59] (Fig. 12.4). The differentiation of outer hair cells and supporting cells is about 1 day behind inner hair cells and can be observed by E15 to E16.
4.1 Hair Cell Differentiation
The earliest known gene expressed in the prosensory domain associated with hair cells is the bHLH transcription factor Atoh1, which is first detected as a gradient that is strongest near the cochlear base and over time extends toward the apex [60, 61]. As development proceeds, Atoh1 expression is restricted to cells that will develop as hair cells.
Several factors that positively or negatively regulate Atoh1 have been identified. Sox2, which is expressed in all prosensory regions, is required for Atoh1 expression. However, Sox2 also seems to negatively regulate Atoh1, as prolonged expression of Sox2 inhibits the ability of Atoh1 to induce hair cell formation, whereas decreased expression of Sox2 leads to an increase in hair cell formation [43]. Other regulators of Atoh1 are the Ids (inhibitors of differentiation), a family of bHLH-related genes that act as antagonists of other bHLH genes [62]. Three of the four mammalian Id genes, Id1, Id2, and Id3, are broadly expressed in the developing cochlear duct but become downregulated in cells that will develop as hair cells [63]. Moreover, forced persistent expression of Id3 leads to an inhibition of hair cell formation, indicating that Ids act to negatively regulate Atoh1 [64].
Lateral inhibition of the Notch signaling pathway is involved in the regulation of hair cell development and Atoh1 expression. Within the inner ear, localization studies showed expression of Notch1 throughout the epithelium and expression of two Notch ligands, Jagged2 and Delta-like1, in developing hair cells [65, 66]. Moreover, several inhibitory bHLHs, including Hes1, Hes5, and Hey1, are expressed in developing supporting cells [2, 61, 67–69]. Deletion of different members of Notch signaling pathway results in varying increases in the number of hair cells, an effect that is very consistent with classic Notch-mediated lateral inhibition [65, 67, 68, 70–72].
4.2 Supporting Cell Differentiation
In the cochlea supporting cells take on several unique morphologies. Though the nature and developmental regulation of supporting cells is less well understood in comparison with hair cells, the FGF signaling pathway has been shown to regulate the formation of the pillar cells that give rise to the tunnel of Corti. Before pillar cell formation, Fgfr3 is expressed in the population of cochlear progenitor cells that will develop as pillar cells, outer hair cells, and Deiters’ cells [73, 74]. At the same time, developing inner hair cells express FGF8, a ligand with a strong binding affinity for FGFR3. Deletion of either Fgf8 or Fgfr3 leads to a defect in pillar cell development, whereas deletion of Sprouty2, a molecule that has been shown to act as an FGF antagonist and is expressed in the cochlea, leads to an overproduction of pillar cells [75]. These results are consistent with the hypothesis that FGF8 secreted by inner hair cells binds to and activates FGFR3 in adjacent cells, leading to the formation of pillar cells (Fig. 12.4).
In addition to inducing prosensory cells to develop as pillar cells, FGF8/FGFR3 signaling also acts to prevent these same cells from developing as hair cells [76, 77]. This effect is mediated through activation of the inhibitory bHLH Hey2, a gene that is normally regulated through the Notch pathway. Interestingly, deletion of Hey2 alone does not lead to any changes in cell fate, but when Hey2 is deleted along with inhibition of Notch signaling, pillar cells will convert into outer hair cells [69].
4.3 Temporal Regulation of Cellular Differentiation
Cell-cycle exit in the cochlea occurs from the apex in an apical-to-basal gradient, while cellular differentiation begins from the base around E14.5, just after the cell-cycle exit has completed. It means that prosensory cells located in the apex of the cochlea that become postmitotic have to be undifferentiated until the cellular differentiation wave comes from the base. Recent works showed that this unique temporal pattern of sensory cell differentiation requires Hh signaling and the source of the signaling is Sonic hedgehog (Shh) from adjacent spiral ganglion [78–80]. In vivo activation and inactivation of the Hh effector smoothened (Smo) in the developing cochlear epithelium after prosensory domain formation revealed that Hh signaling inhibits prosensory cell differentiation into hair cells or supporting cells and maintains their properties as prosensory cells [81]. Smo conditional knockout (Smo CKO) mice showed that hair cell differentiation was preferentially accelerated in the apical region of the cochlea and exhibited hair cell disarrangement in the apical region, a decrease in hair cell number, and hearing impairment [81]. These results indicate that Hh signaling determines the timing of cellular differentiation and the basal-to-apical wave of hair cell development is required for the proper differentiation, arrangement, and survival of hair cells and for hearing ability [81].
References
Morsli H, Choo D, Ryan A, Johnson R, Wu DK. Development of the mouse inner ear and origin of its sensory organs. J Neurosci. 1998;18:3327–35.
Zine A, Aubert A, Qiu J, Therianos S, Guillemot F, Kageyama R, de Ribaupierre F. Hes1 and Hes5 activities are required for the normal development of the hair cells in the mammalian inner ear. J Neurosci. 2001;21:4712–20.
Davis RL. Gradients of neurotrophins, ion channels, and tuning in the cochlea. Neuroscientist. 2003;9:311–6.
Riccomagno MM, Martinu L, Mulheisen M, Wu DK, Epstein DJ. Specification of the mammalian cochlea is dependent on Sonic hedgehog. Genes Dev. 2002;16:2365–78.
Bok J, Bronner-Fraser M, Wu DK. Role of the hindbrain in dorsoventral but not anteroposterior axial specification of the inner ear. Development. 2005;132:2115–24.
Montcouquiol M, Kelley MW. Planar and vertical signals control cellular differentiation and patterning in the mammalian cochlea. J Neurosci. 2003;23:9469–78.
Phippard D, Lu L, Lee D, Saunders JC, Crenshaw EB. Targeted mutagenesis of the POU-domain gene Brn4/Pou3f4 causes developmental defects in the inner ear. J Neurosci. 1999;19:5980–9.
Braunstein EM, Crenshaw EB, Morrow BE, Adams JC. Cooperative function of Tbx1 and Brn4 in the periotic mesenchyme is necessary for cochlea formation. J Assoc Res Otolaryngol. 2008;9:33–43.
Braunstein EM, Monks DC, Aggarwal VS, Arnold JS, Morrow BE. Tbx1 and Brn4 regulate retinoic acid metabolic genes during cochlear morphogenesis. BMC Dev Biol. 2009;9:31.
Kikuchi K, Hilding D. The development of the organ of Corti in the mouse. Acta Otolaryngol. 1965;60:207–22.
Lim DJ, Anniko M. Developmental morphology of the mouse inner ear. A scanning electron microscopic observation. Acta Otolaryngol Suppl. 1985;422:1–69.
Chen P, Johnson JE, Zoghbi HY, Segil N. The role of Math1 in inner ear development: Uncoupling the establishment of the sensory primordium from hair cell fate determination. Development. 2002;129:2495–505.
McKenzie E, Krupin A, Kelley MW. Cellular growth and rearrangement during the development of the mammalian organ of Corti. Dev Dyn. 2004;229:802–12.
Yamamoto N, Okano T, Ma X, Adelstein RS, Kelley MW. Myosin II regulates extension, growth and patterning in the mammalian cochlear duct. Development. 2009;136:1977–86.
Wang J, Mark S, Zhang X, Qian D, Yoo SJ, Radde-Gallwitz K, Zhang Y, Lin X, Collazo A, Wynshaw-Boris A, et al. Regulation of polarized extension and planar cell polarity in the cochlea by the vertebrate PCP pathway. Nat Genet. 2005;37:980–5.
Jones C, Chen P. Planar cell polarity signaling in vertebrates. Bioessays. 2007;29:120–32.
Etheridge SL, Ray S, Li S, Hamblet NS, Lijam N, Tsang M, Greer J, Kardos N, Wang J, Sussman DJ et al. Murine dishevelled 3 functions in redundant pathways with dishevelled 1 and 2 in normal cardiac outflow tract, cochlea, and neural tube development. PLoS Genet 2008;4:e1000259.
Kelley MW, Xu XM, Wagner MA, Warchol ME, Corwin JT. The developing organ of Corti contains retinoic acid and forms supernumerary hair cells in response to exogenous retinoic acid in culture. Development. 1993;119:1041–53.
Kelly MC, Chen P. Development of form and function in the mammalian cochlea. Curr Opin Neurobiol. 2009;19:395–401.
Pirvola U, Ylikoski J, Trokovic R, Hébert JM, McConnell SK, Partanen J. FGFR1 is required for the development of the auditory sensory epithelium. Neuron. 2002;35:671–80.
Hayashi T, Ray CA, Bermingham-McDonogh O. Fgf20 is required for sensory epithelial specification in the developing cochlea. J Neurosci. 2008;28:5991–9.
Huh SH, Jones J, Warchol ME, Ornitz DM. Differentiation of the lateral compartment of the cochlea requires a temporally restricted FGF20 signal. PLoS Biol. 2012;10:e1001231.
Ohyama T, Basch ML, Mishina Y, Lyons KM, Segil N, Groves AK. BMP signaling is necessary for patterning the sensory and nonsensory regions of the developing mammalian cochlea. J Neurosci. 2010;30:15044–51.
Kageyama R, Ohtsuka T, Shimojo H, Imayoshi I. Dynamic regulation of Notch signaling in neural progenitor cells. Curr Opin Cell Biol. 2009;21:733–40.
Imayoshi I, Shimojo H, Sakamoto M, Ohtsuka T, Kageyama R. Genetic visualization of notch signaling in mammalian neurogenesis. Cell Mol Life Sci. 2013;70:2045–57.
Kiernan AE. Notch signaling during cell fate determination in the inner ear. Semin Cell Dev Biol. 2013;24:470–9.
Kiernan AE, Ahituv N, Fuchs H, Balling R, Avraham KB, Steel KP, Hrabé de Angelis M. The Notch ligand Jagged1 is required for inner ear sensory development. Proc Natl Acad Sci U S A. 2001;98:3873–8.
Tsai H, Hardisty RE, Rhodes C, Kiernan AE, Roby P, Tymowska-Lalanne Z, Mburu P, Rastan S, Hunter AJ, Brown SD, et al. The mouse slalom mutant demonstrates a role for Jagged1 in neuroepithelial patterning in the organ of Corti. Hum Mol Genet. 2001;10:507–12.
Kiernan AE, Xu J, Gridley T. The Notch ligand JAG1 is required for sensory progenitor development in the mammalian inner ear. PLoS Genet. 2006;2:e4.
Brooker R, Hozumi K, Lewis J. Notch ligands with contrasting functions: Jagged1 and Delta1 in the mouse inner ear. Development. 2006;133:1277–86.
Pan W, Jin Y, Stanger B, Kiernan AE. Notch signaling is required for the generation of hair cells and supporting cells in the mammalian inner ear. Proc Natl Acad Sci U S A. 2010;107:15798–803.
Yamamoto N, Chang W, Kelley MW. Rbpj regulates development of prosensory cells in the mammalian inner ear. Dev Biol. 2011;353:367–79.
Daudet N, Lewis J. Two contrasting roles for Notch activity in chick inner ear development: specification of prosensory patches and lateral inhibition of hair-cell differentiation. Development. 2005;132:541–51.
Hartman BH, Reh TA, Bermingham-McDonogh O. Notch signaling specifies prosensory domains via lateral induction in the developing mammalian inner ear. Proc Natl Acad Sci U S A. 2010;107:15792–7.
Liu Z, Owen T, Fang J, Zuo J. Overactivation of Notch1 signaling induces ectopic hair cells in the mouse inner ear in an age-dependent manner. PLoS One. 2012;7:e34123.
Pan W, Jin Y, Chen J, Rottier RJ, Steel KP, Kiernan AE. Ectopic expression of activated notch or SOX2 reveals similar and unique roles in the development of the sensory cell progenitors in the mammalian inner ear. J Neurosci. 2013;33:16146–57.
Kiernan AE, Pelling AL, Leung KK, Tang AS, Bell DM, Tease C, Lovell-Badge R, Steel KP, Cheah KS. Sox2 is required for sensory organ development in the mammalian inner ear. Nature. 2005;434:1031–5.
Munnamalai V, Hayashi T, Bermingham-McDonogh O. Notch prosensory effects in the Mammalian cochlea are partially mediated by Fgf20. J Neurosci. 2012;32:12876–84.
Pauley S, Wright TJ, Pirvola U, Ornitz D, Beisel K, Fritzsch B. Expression and function of FGF10 in mammalian inner ear development. Dev Dyn. 2003;227:203–15.
Ruben RJ. Development of the inner ear of the mouse: a radioautographic study of terminal mitoses. Acta Otolaryngol. 1967;Suppl 220:1–44.
Chen P, Segil N. p27(Kip1) links cell proliferation to morphogenesis in the developing organ of Corti. Development. 1999;126:1581–90.
Lee YS, Liu F, Segil N. A morphogenetic wave of p27Kip1 transcription directs cell cycle exit during organ of Corti development. Development. 2006;133:2817–26.
Dabdoub A, Puligilla C, Jones JM, Fritzsch B, Cheah KS, Pevny LH, Kelley MW. Sox2 signaling in prosensory domain specification and subsequent hair cell differentiation in the developing cochlea. Proc Natl Acad Sci U S A. 2008;105:18396–401.
Ono K, Nakagawa T, Kojima K, Matsumoto M, Kawauchi T, Hoshino M, Ito J. Silencing p27 reverses post-mitotic state of supporting cells in neonatal mouse cochleae. Mol Cell Neurosci. 2009;42:391–8.
Oesterle EC, Chien WM, Campbell S, Nellimarla P, Fero ML. p27(Kip1) is required to maintain proliferative quiescence in the adult cochlea and pituitary. Cell Cycle. 2011;10:1237–48.
Liu Z, Walters BJ, Owen T, Brimble MA, Steigelman KA, Zhang L, Mellado Lagarde MM, Valentine MB, Yu Y, Cox BC, et al. Regulation of p27Kip1 by Sox2 maintains quiescence of inner pillar cells in the murine auditory sensory epithelium. J Neurosci. 2012;32:10530–40.
Sage C, Huang M, Karimi K, Gutierrez G, Vollrath MA, Zhang DS, García-Añoveros J, Hinds PW, Corwin JT, Corey DP. Proliferation of functional hair cells in vivo in the absence of the retinoblastoma protein. Science. 2005;307:1114–8.
Mantela J, Jiang Z, Ylikoski J, Fritzsch B, Zacksenhaus E, Pirvola U. The retinoblastoma gene pathway regulates the postmitotic state of hair cells of the mouse inner ear. Development. 2005;132:2377–88.
Sage C, Huang M, Vollrath MA, Brown MC, Hinds PW, Corey DP, Vetter DE, Chen ZY. Essential role of retinoblastoma protein in mammalian hair cell development and hearing. Proc Natl Acad Sci U S A. 2006;103:7345–50.
Weber T, Corbett MK, Chow LM, Valentine MB, Baker SJ, Zuo J. Rapid cell-cycle reentry and cell death after acute inactivation of the retinoblastoma gene product in postnatal cochlear hair cells. Proc Natl Acad Sci U S A. 2008;105:781–5.
Yu Y, Weber T, Yamashita T, Liu Z, Valentine MB, Cox BC, Zuo J. In vivo proliferation of postmitotic cochlear supporting cells by acute ablation of the retinoblastoma protein in neonatal mice. J Neurosci. 2010;30:5927–36.
Chen P, Zindy F, Abdala C, Liu F, Li X, Roussel MF, Segil N. Progressive hearing loss in mice lacking the cyclin-dependent kinase inhibitor Ink4d. Nat Cell Biol. 2003;5:422–6.
Laine H, Doetzlhofer A, Mantela J, Ylikoski J, Laiho M, Roussel MF, Segil N, Pirvola U. p19(Ink4d) and p21(Cip1) collaborate to maintain the postmitotic state of auditory hair cells, their codeletion leading to DNA damage and p53-mediated apoptosis. J Neurosci. 2007;27:1434–44.
Jacques BE, Puligilla C, Weichert RM, Ferrer-Vaquer A, Hadjantonakis AK, Kelley MW, Dabdoub A. A dual function for canonical Wnt/β-catenin signaling in the developing mammalian cochlea. Development. 2012;139:4395–404.
Barker N, van Es JH, Kuipers J, Kujala P, van den Born M, Cozijnsen M, Haegebarth A, Korving J, Begthel H, Peters PJ, et al. Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature. 2007;449:1003–7.
Jaks V, Barker N, Kasper M, van Es JH, Snippert HJ, Clevers H, Toftgård R. Lgr5 marks cycling, yet long-lived, hair follicle stem cells. Nat Genet. 2008;40:1291–9.
Chai R, Xia A, Wang T, Jan TA, Hayashi T, Bermingham-McDonogh O, Cheng AG. Dynamic expression of Lgr5, a Wnt target gene, in the developing and mature mouse cochlea. J Assoc Res Otolaryngol. 2011;12:455–69.
Shi F, Kempfle JS, Edge AS. Wnt-responsive Lgr5-expressing stem cells are hair cell progenitors in the cochlea. J Neurosci. 2012;32:9639–48.
Chai R, Kuo B, Wang T, Liaw EJ, Xia A, Jan TA, Liu Z, Taketo MM, Oghalai JS, Nusse R, et al. Wnt signaling induces proliferation of sensory precursors in the postnatal mouse cochlea. Proc Natl Acad Sci U S A. 2012;109:8167–72.
Kelley MW. Cellular commitment and differentiation in the organ of Corti. Int J Dev Biol. 2007;51:571–83.
Anniko M. Cytodifferentiation of cochlear hair cells. Am J Otolaryngol. 1983;4:375–88.
Lanford PJ, Shailam R, Norton CR, Gridley T, Kelley MW. Expression of Math1 and HES5 in the cochleae of wildtype and Jag2 mutant mice. J Assoc Res Otolaryngol. 2000;1:161–71.
Norton JD. ID helix-loop-helix proteins in cell growth, differentiation and tumorigenesis. J Cell Sci. 2000;113(Pt 22):3897–905.
Jones JM, Montcouquiol M, Dabdoub A, Woods C, Kelley MW. Inhibitors of differentiation and DNA binding (Ids) regulate Math1 and hair cell formation during the development of the organ of Corti. J Neurosci. 2006;26:550–8.
Kamaid A, Neves J, Giráldez F. Id gene regulation and function in the prosensory domains of the chicken inner ear: a link between Bmp signaling and Atoh1. J Neurosci. 2010;30:11426–34.
Lanford PJ, Lan Y, Jiang R, Lindsell C, Weinmaster G, Gridley T, Kelley MW. Notch signalling pathway mediates hair cell development in mammalian cochlea. Nat Genet. 1999;21:289–92.
Morrison A, Hodgetts C, Gossler A, Hrabé de Angelis M, Lewis J. Expression of Delta1 and Serrate1 (Jagged1) in the mouse inner ear. Mech Dev. 1999;84:169–72.
Hayashi T, Kokubo H, Hartman BH, Ray CA, Reh TA, Bermingham-McDonogh O. Hesr1 and Hesr2 may act as early effectors of Notch signaling in the developing cochlea. Dev Biol. 2008;316:87–99.
Li S, Mark S, Radde-Gallwitz K, Schlisner R, Chin MT, Chen P. Hey2 functions in parallel with Hes1 and Hes5 for mammalian auditory sensory organ development. BMC Dev Biol. 2008;8:20.
Doetzlhofer A, Basch ML, Ohyama T, Gessler M, Groves AK, Segil N. Hey2 regulation by FGF provides a Notch-independent mechanism for maintaining pillar cell fate in the organ of Corti. Dev Cell. 2009;16:58–69.
Kiernan AE, Cordes R, Kopan R, Gossler A, Gridley T. The Notch ligands DLL1 and JAG2 act synergistically to regulate hair cell development in the mammalian inner ear. Development. 2005;132:4353–62.
Takebayashi S, Yamamoto N, Yabe D, Fukuda H, Kojima K, Ito J, Honjo T. Multiple roles of Notch signaling in cochlear development. Dev Biol. 2007;307:165–78.
Tateya T, Imayoshi I, Tateya I, Ito J, Kageyama R. Cooperative functions of Hes/Hey genes in auditory hair cell and supporting cell development. Dev Biol. 2011;352:329–40.
Mueller KL, Jacques BE, Kelley MW. Fibroblast growth factor signaling regulates pillar cell development in the organ of corti. J Neurosci. 2002;22:9368–77.
Jacques BE, Montcouquiol ME, Layman EM, Lewandoski M, Kelley MW. Fgf8 induces pillar cell fate and regulates cellular patterning in the mammalian cochlea. Development. 2007;134:3021–9.
Shim K, Minowada G, Coling DE, Martin GR. Sprouty2, a mouse deafness gene, regulates cell fate decisions in the auditory sensory epithelium by antagonizing FGF signaling. Dev Cell. 2005;8:553–64.
Hayashi T, Cunningham D, Bermingham-McDonogh O. Loss of Fgfr3 leads to excess hair cell development in the mouse organ of Corti. Dev Dyn. 2007;236:525–33.
Puligilla C, Feng F, Ishikawa K, Bertuzzi S, Dabdoub A, Griffith AJ, Fritzsch B, Kelley MW. Disruption of fibroblast growth factor receptor 3 signaling results in defects in cellular differentiation, neuronal patterning, and hearing impairment. Dev Dyn. 2007;236:1905–17.
Driver EC, Pryor SP, Hill P, Turner J, Rüther U, Biesecker LG, Griffith AJ, Kelley MW. Hedgehog signaling regulates sensory cell formation and auditory function in mice and humans. J Neurosci. 2008;28:7350–8.
Bok J, Zenczak C, Hwang CH, Wu DK. Auditory ganglion source of Sonic hedgehog regulates timing of cell cycle exit and differentiation of mammalian cochlear hair cells. Proc Natl Acad Sci U S A. 2013;110:13869–74.
Tateya T, Imayoshi I, Tateya I, Hamaguchi K, Torii H, Ito J, Kageyama R. Hedgehog signaling regulates prosensory cell properties during the basal-to-apical wave of hair cell differentiation in the mammalian cochlea. Development. 2013;140:3848–57.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Japan
About this chapter
Cite this chapter
Tateya, T. (2014). Cochlear Development. In: Ito, J. (eds) Regenerative Medicine for the Inner Ear. Springer, Tokyo. https://doi.org/10.1007/978-4-431-54862-1_12
Download citation
DOI: https://doi.org/10.1007/978-4-431-54862-1_12
Published:
Publisher Name: Springer, Tokyo
Print ISBN: 978-4-431-54861-4
Online ISBN: 978-4-431-54862-1
eBook Packages: MedicineMedicine (R0)