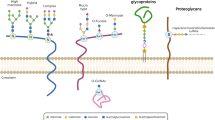
Abstract
Aberrant glycosylation is the basic phenomenon found in almost all types of experimental and clinical cancer tissues and/or cell lines, and many glycosyl epitopes are known to constitute so-called tumor-associated carbohydrate antigens (TACAs). Many lines of evidence suggest that these cancer-related glycosyl epitopes play functional roles in a variety of pathophysiologic events in cancer, such as cell metastasis and invasion, apart from their potential values as diagnostic and/or prognostic glyco-biomarkers. This chapter describes a brief summary on the aberrant glycosylation in cancer, the related glycosyltransferases, and their roles in cancer development and progression. The chapter focuses on the glycosyltransferases responsible for N-glycan branching; those include N-acetylglucosaminyltransferase (GnT)-III, GnT-IV, and GnT-V and fucosyltransferase (FUT) 8.
Similar content being viewed by others
References
Abbott KL, Matthews RT, Pierce M (2008) Receptor tyrosine phosphatase β (RPTPβ) activity and signaling are attenuated by glycosylation and subsequent cell surface galectin-1 binding. J Biol Chem 283:33026–33035
Brockhausen I, Carver JP, Schachter H (1988) Control of glycoprotein synthesis. The use of oligosaccharide substrates and HPLC to study the sequential pathway for N-acetylglucosaminyltransferases I, II, III, IV, V and VI in the biosynthesis of highly branched N-glycans by hen oviduct membranes. Biochem Cell Biol 66:1134–1151
Christiansen MN, Chik J, Lee L et al (2014) Cell surface protein glycosylation in cancer. Proteomics 14(4–5):525–546. doi:10.1002/pmic
Dall’Olio F, Malagolini N, Trinchera M, Chiricolo M et al (2012) Mechanisms of cancer-associated glycosylation changes. Front Biosci (Landmark Ed) 17:670–699
Gu J, Taniguchi N (2008) Potential of N-glycan in cell adhesion and migration as either a positive or negative regulator. Cell Adh Migr 2:243–245
Hakomori S (2008) Glycosylation defining cancer malignancy: new wine in an old bottle. Proc Natl Acad Sci U S A 99:10231–10233
Inamori K, Endo T, Gu J et al (2004) N-Acetylglucosaminyltransferase IX acts on the GlcNAc β1,2-Manα1-Ser/Thr moiety, forming a 2,6-branched structure in brain O-mannosyl glycan. J Biol Chem 279:2337–2340
Kizuka Y, Kitazume S, Yoshida M et al (2011) Brain-specific expression of N-acetylglucosaminyltransferase IX (GnT-IX) is regulated by epigenetic histone modifications. J Biol Chem 286:31875–31884
Korekane H, Park JY, Matsumoto A et al (2013) Identification of ectonucleotide pyrophosphatase/phosphodiesterase 3 (ENPP3) as a regulator of N-acetylglucosaminyltransferase GnT-IX (GnT-Vb). J Biol Chem 288:27912–27926
Lau KS, Partridge EA, Grigorian A et al (2007) Complex N-glycan number and degree of branching cooperate to regulate cell proliferation and differentiation. Cell 129:123–134
Lee JK, Matthews RT, Lim JM et al (2012) Developmental expression of the neuron-specific N-acetylglucosaminyltransferase Vb (GnT-Vb/IX) and identification of its in vivo glycan products in comparison with those of its paralog, GnT-V. J Biol Chem 287:28526–28536
Taniguchi N, Korekane H (2011) Branched N-glycans and their implications for cell adhesion, signaling and clinical applications for cancer biomarker and in therapeutics. BMB Rep 44:772–781
Zhao Y, Sato Y, Isaji T et al (2008) Branched N-glycans regulate the biological functions of integrins and cadherins. FEBS J 275:1939–1948
Acknowledgment
The authors acknowledge many colleagues who shared the work with us for a longer period. We also apologize not to have cited relevant papers published by many excellent scientists.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Japan
About this entry
Cite this entry
Korekane, H., Taniguchi, N. (2014). Glycosylation in Cancer: Enzymatic Basis for Alterations in N-Glycan Branching. In: Endo, T., Seeberger, P., Hart, G., Wong, CH., Taniguchi, N. (eds) Glycoscience: Biology and Medicine. Springer, Tokyo. https://doi.org/10.1007/978-4-431-54836-2_183-1
Download citation
DOI: https://doi.org/10.1007/978-4-431-54836-2_183-1
Received:
Accepted:
Published:
Publisher Name: Springer, Tokyo
Online ISBN: 978-4-431-54836-2
eBook Packages: Springer Reference Biomedicine and Life SciencesReference Module Biomedical and Life Sciences