Abstract
Primates show life history traits similar to those of cetaceans, such as small litter size, long gestation, long lactation, and long lifespan, in spite of striking contrasts in habitats, diet, mobility, and range size between them. Ecological factors (food and predation) may influence their life history traits in various ways, but social factors (social structure and reproductive strategies) may be more important for the life history of primates, in which both sexes live together even outside the breeding season. Group-living primates are classified into female-bonded species and female-dispersal species, based on the patterns of female dispersal after maturity. A comparison of life history parameters shows that female-dispersal species have a slower life history (gestation length, weaning age, age at first reproduction, and interbirth interval) than the female-bonded species, except for neonatal weight and weaning weight, which may be determined in relationship to female body weight. To elucidate factors promoting the slow life history, we focus on Atelinae and Hominidae (female-dispersal species) and examine their interspecific and intraspecific variation in social structure and male reproductive tactics in relationship to life history traits. Most Atelinae species form multimale and multifemale groups, and variation in their life history features may reflect relationships among males and their reproductive tactics. In howler monkeys, both males and females disperse, and infanticide by males may lead to a fast life history. In other Atelines, infanticide rarely occurs, although it has the effect of reducing interbirth interval. Forcible copulation by males occasionally occurs in spider monkeys. Variations in grouping among females reflecting their flexible foraging efforts according to distribution of high-quality foods may have some effects on the fast–slow continuum in the life history features of female Atelinae. Hominidae exhibit larger variations in life history features than Atelinae, probably because of their diverse social structure. Solitary nature and male reproductive tactics may have great influences on the life history of female great apes. Female orangutans, who usually live a solitary life, show the slowest life history. Maturing female orangutans need a longer time to establish their own home range and relationships with reproductive mates than female chimpanzees and gorillas, who transfer into other groups immediately after emigration. Female gorillas show the lowest age at first reproduction and the shortest interbirth interval. Intensive caretaking of the immature by male gorillas may facilitate early weaning, and infanticide by males may promote a prolonged bonding between a protector male and females to shorten the interbirth interval. Similar life history traits have been found in four long-term study sites of chimpanzees. Only females at Bossou show a fast life history, probably the result of high-quality foods and single male group composition under isolated conditions. The more frequent and stable association between females and males and more promiscuous mating in bonobos may facilitate the search for mating partners and lead to a shorter interbirth interval than chimpanzees. Frugivorous orangutans and chimpanzees may suffer more costs of female dispersal through decreased foraging efficiency than folivorous gorillas, and chimpanzees with fission–fusion grouping may suffer more social stress than gorillas in highly cohesive groups. Such differences may generally shape the fast–slow continuum of life history in female-dispersal primate species.

Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Life History Traits of Cetaceans and Primates
Life history is expressed as a scheduling of development and reproduction within a life cycle (Stearns 1992). Parameters of life history, such as gestation length, prenatal and postnatal growth rate, weaning age, age to first reproduction, interbirth interval, and lifespan, vary allometrically with body size (Read and Harvey 1989; Charnov 1991, 1993; Purvis and Harvey 1995). In mammals, large species take longer to grow to maturity, have a longer gestation, have a longer interval between births, and have fewer young per litter than smaller species. This “fast–slow continuum ” in the speed of life is also found among cetaceans and primates.
A cetacean is characterized by its large body size (for the blue whale, a body length of 31 m) and large neonatal mass (more than 15 % of maternal mass) because of its aquatic habitats and advanced feeding techniques, such as the filtering observed in mysticetes (Whitehead and Mann 2000). Among cetaceans, life history traits differ between mysticetes and odontocetes (Fig. 9.1). Although no difference is found in neonatal weight relative to adult female body weight, weaning age and age at first parturition of mysticetes are lower than those of odontocetes . The mysticetes show fast life history patterns and no obvious correlation between body size and the speed of their life history processes (Martin and Rothery 1993; Kasuya 1995). By contrast, the odontocetes generally follow the fast–slow continuum, possibly caused by predation pressure and food availability being variable with body size (Whitehead and Weilgart 2000; Whitehead and Mann 2000). Duration of lactation is extremely variable among cetacean species, ranging from 6 months in baleen whales to 6 years in bottlenose dolphins (Connor et al. 2000). Mysticetes produce milk with the highest fat content (30–53 %) among mammals and wean offspring at an earlier age than most of the odontocetes, irrespective of body length (Oftedal 1997). In odontocetes, the length of lactation has a positive correlation with body length, and weaning occurs gradually, probably the result of the necessity of learning how to feed on highly mobile prey (Mann and Smuts 1999). Food supply may affect the duration of lactation, which constitutes a constraint factor on the future reproduction of mothers (Martin and Rothery 1993; Mann et al. 2000). Cetaceans are gregarious and socially diverse, but most of their social structure is matrilineal , in which males disperse from their natal groups (Kasuya 1995). As observed in bottlenose dolphins and striped dolphins, kin-related females tend to associate and cooperate in rearing calves in a school (Miyazaki and Nishiwaki 1978; Shane et al. 1986). In some matrilineal social structures (e.g., pilot whales and killer whales), menopause has been found (Whitehead 1998). Patrilineal social structure has been found in Baird’s beaked whales, in which males reach maturity earlier and live longer than females (Kasuya and Jones 1984). Male Baird’s beaked whales care for weaned offspring, which is exceptional among cetaceans (Kasuya et al. 1997).
Life history traits of cetaceans and primates among mammals relative to female body weight: neonatal weight (a), weaning age (b), and age at first parturition (c). Open squares, mysticetes; filled squares, odontocetes; open triangles, anthropoids; filled triangles, prosimians; filled diamonds, other mammals. (Data from the list by Purvis and Harvey 1995)
Primates show life history traits similar to those of cetaceans, such as small litter size, long gestation, long lactation, and long lifespan, in spite of striking contrasts in habitats, diet, mobility, and range size between them (Harvey et al. 1987; Read and Harvey 1989; Ross 1998; Whitehead and Mann 2000). Primates lie at the slow end of the fast–slow continuum (Fig. 9.1; Table 9.1). Haplorhines have a slower life history than strepsirrhines of the same size (Purvis et al. 2003). Recent arguments have proposed determinant factors of the slow life history in primates, such as large brain size (Allman et al. 1993; Martin 1996), high risk of juvenile mortality (Janson and van Schaik 1993), nutritional risk (Borries et al. 2001; Altmann and Alberts 2003; Anderson et al. 2008), and arboreal lifestyle (Eisenberg 1981; Martin 1995), but no single factor seem to fully explain this (Harvey and Purvis 1999; van Schaik and Deaner 2002).
Ecological factors may influence the life history traits of primates in various ways (Kappeler et al. 2003). The low growth rate of primates may be caused by a negative association between mortality rates and growth rates, and juvenile vulnerability to food shortage and predation may shape their life history traits (Janson and van Schaik 1993). Primates may be adapted to the low mortality rates prevalent in their ancestral habitat (tropical forests), because other arboreal mammals, such as bats (Jones and MacLarnon 2001), also have low mortality rates. Primates living in the more unpredictable habitats have higher birthrates and earlier age at first reproduction (Ross 1998). The apes that are strictly distributed in and around the tropical forests have slow life history traits, whereas Old World monkeys living in variable habitats have relatively rapid life history traits. Forest macaques have a longer interbirth interval and a later age at first reproduction than opportunistic macaque species living in a variety of habitats, even in sympatric conditions (Ross 1992). However, the age of first reproduction in olive baboons is highly heritable (Williams-Blangero and Blangero 1995). Large intraspecific variations are also found in some life history traits. Female vervet monkeys may respond to limited access to food resources by delaying reproduction (Cheney et al. 1988). Female Japanese macaques and savanna baboons with high rank tend to mature earlier than females with low rank (Altmann et al. 1988; Gouzoules et al. 1982; Takahata et al. 1999). These observations may suggest that life history traits of primates may have evolved as a species-specific strategy as well as the immediate responses to environment changes.
Social structure and social behavior are also important for life history traits. Among mammals, primates have a unique social feature in that the two sexes live together even outside the breeding season. This lifestyle may result in diversity of social structure and may characterize the fast–slow continuum in relationship to social systems. Group size and socionomic sex ratio (the number of adult males per female within a group) can change feeding and reproductive strategies of both sexes and thereby affect life history parameters (Dunbar 1988; Sterck et al. 1997; Nunn and Pereira 2000). Female gregariousness, social relationships, or alloparental care of dependent infants may also change life history traits such as postnatal growth rate , weaning age , and interbirth interval (Fairbanks 1990; Stanford 1992; Van Noodwijk and van Schaik 2005). High infant growth rates do not appear to be correlated with environmental factors (diet, climate, or habitat) but with nonmaternal care, which allows mothers to increase birthrates by decreasing interbirth interval (Ross and MacLarnon 1995). Male reproductive strategies may constitute a strong selective force on life history traits. Infanticide by males promotes prolonged male–female association (van Schaik 2000) and complex male–infant relationships (Paul et al. 2000) and affects patterns of female movements between groups (Steenbeck 2000; Yamagiwa and Kahekwa 2001) and female reproductive biology (Watts 2000; van Noordwijk and van Schaik 2000; Yamagiwa et al. 2009). Recent findings show large variations in social structure and behavior between species and within species (Barton et al. 1996; Henzi and Barrett 2003; Doran et al. 2002; Yamagiwa et al. 2003). Life history traits are also easy to change, relatively independently, via selection (Kappeler et al. 2003). However, it is still unclear how such social variation is linked with life history variation.
In this chapter, we focus on female dispersal as the limiting factor of life history parameters. Group-living primates have been classified into female-bonded species and female-dispersal species (Wrangham 1980). Most of the macaques, cercopithecines , and Cebus monkeys form a group in which females remain during their entire life. They usually associate with kin-related females and form coalitions with them in agonistic contexts (Watanabe 1979; Silk 1982; Dunbar 1988; Harcourt 1992; Henzi and Barrett 1999). Cooperation and support of kin-related females increase female reproductive success. The linear dominance rank is stable among females and between kin-groups of females. Females of the kin-groups with higher rank have higher birthrates, younger age at first parturition, and lower infant mortality than females of kin-groups with lower rank (Drickamer 1974; Silk 1987; Itoigawa et al. 1992; Paul and Kuester 1996), although these tendencies are not consistent in some species (Cheney et al. 1988; Takahata et al. 1999). On the other hand, females of Hominidae and Atelinae usually leave their natal groups and spend their reproductive life without related females (Wrangham 1987; Yamagiwa 1999; Strier 1999a). Social relationships with males or unrelated females that they join are important for their reproductive success. The elder females or females joining earlier are dominant to younger females or those joining later (Goodall 1986; Watts 1991a; Idani 1991; Crockett and Pope 1993; Printes and Strier 1999; Nishimura 2003). However, intervention by males in conflicts (Watts 1997), sociosexual behavior among females (Kano 1992), and the fission–fusion nature of grouping (Wrangham and Smuts 1980; Goodall 1986; Strier 1992) reduce dominance effects and prevent females from having prolonged antagonistic interactions. Because of the lack of support from kin-related females, male reproductive strategies including infanticide may affect life history parameters in female-dispersal species (Strier 1999a; Harcourt and Stewart 2007). Here, we also examine inter- and intraspecific variation in social structure and male reproductive tactics in relationship to life history traits.
2 Life History Tactics of Female-Bonded and Female-Dispersal Species
Anthropoid primates form either female-bonded or female-dispersal groups, except for orangutans, in which both females and males spend a solitary life after maturity. Among them, we selected primate species with medium and large body size for comparisons of life history traits: 20 species for female-bonded and 15 species for female-dispersal (Table 9.2). The orangutan was included in the female-dispersal species because all females separate from their mothers before maturity. As life history parameters, gestation length, neonatal weight, weaning age, weaning weight, age at first parturition , and interbirth interval were used for interspecific comparison with reference to female body weight. We used the database constructed by Kappeler and Pereira (2003) and added to it some reliable data from recent reports. Most of the data were from observations on wild populations, but some data were from observations on provisioned or captive individuals. For statistical tests, we used the analysis of covariance (ANCOVA) in the Analyze/Fit Model section in JMPs.
Gestation length of female-dispersal species was significantly longer than that of female-bonded species (F = 69.05, p < 0.0001; Fig. 9.2a). Length of gestation of Atelinae (Ateles, Brachyteles) was far longer than that of Cercopithecinae (Cercopithecus, Macaca, Cercocebus, Lophocebus, Papio, Theropithecus) and Colobinae (Colobus, Semnopithecus). Gestation length of Hominidae was diverse and did not correlate with body weight among the four species. Neonatal weight tended to increase relative to female body weight, and no difference was found here between the female-bonded and the female-dispersal species (F = 106.86, p = 0.3877; Fig. 9.2b). Although no difference was found in weaning weight (F = 45.05, p = 0.9586; Fig. 9.2c), weaning ages of female-dispersal species were significantly higher than those of female-dispersal species (F = 34.28, p < 0.01; Fig. 9.2d). In primate species, weaning generally occurs around the time when infants reach about one third of adult body weight (Lee et al. 1991; Lee 1996). Female-dispersal species tend to have a higher age at first parturition (F = 23.97, p < 0.05; Fig. 9.2e) and longer interbirth interval (F = 56.63, p < 0.0001; Fig. 9.2f) than female-bonded species. These results suggest that female-dispersal species may have slower life history traits than female-bonded species. Neonatal weight and weaning weight may be determined in relationship to female body weight irrespective of female movement patterns. However, female dispersal and reproduction without help from kin-related females may result in a slow life history.
Life history traits of female-bonded and female-dispersal primate species (relative to female body weight): gestation length (a), neonatal weight (b), weaning age (c), weaning weight (d), age at first parturition (e), interbirth interval (f). Filled diamonds, female-bonded species; open diamonds, female-dispersal species
Female-dispersal species form various social structures, such as solitary, monogamous, polygynous, or multimale/multifemale groups. Which aspects of female dispersal or social features lead them to slow life history? To answer this question, we compared life history traits between genera, between species, and between populations within species of Atelinae and Hominidae, which have been extensively studied at several sites.
3 Factors Leading to Slow Life History of Atelinae and Hominidae
Atelinae and Hominidae are typical taxa in which all genera have common social features, such as the lack of female kin bonding and female dispersal from the natal group or mother (Goodall 1986; Rosenberger and Strier 1989; Strier 1999b; Harcourt and Stewart 2007). Atelines live in the tropical forests of Central and South America and are divided into four genera: howler monkeys (Alouatta), spider monkeys (Ateles), woolly monkeys (Lagothrix), and muriquis (Brachyteles). Hominidae (great apes), except for humans, are divided into three genera and live in the tropical forests of Asia (orangutans , Pongo) and Africa (gorillas , Gorilla; chimpanzees , Pan). Atelines usually form groups including multiple females and males. Although male howler monkeys also disperse from their natal groups, males of the other three Atelines remain in their natal groups to associate with kin-related males, as observed for chimpanzees and bonobos (Symington 1988; Strier 1999a). The social structures of the great apes are highly differentiated. Both male and female orangutans usually live alone and partially overlap their home range with neighboring individuals of the same sex (Galdikas 1984; Delgado and van Schaik 2000; van Schaik 1999). Gorillas form a cohesive group consisting of a mature male and several females with their offspring. Both female and male gorillas tend to emigrate from their natal groups, and only females immigrate into other groups in which they start reproduction (Yamagiwa and Kahekwa 2001; Stokes et al. 2003; Robbins et al. 2009). Chimpanzees form large groups including multiple males and females, and only females emigrate from their natal groups (Nishida 1979; Goodall 1986; Boesch and Boesch-Achermann 2000).
Analogous to the great apes, Atelines are characterized by the largest body weight and the slowest life history among neotropical primates (Table 9.3). The slower reproductive traits of woolly monkeys compared to howler monkeys , which have a similar body size, may reflect their differences in female reproductive costs in relationship to social features. Female howler monkeys can have more diverse options than female woolly monkeys , such as transferring into other groups, joining males to establish a new group, or remaining in their natal group to breed (Crockett and Pope 1993; Strier 1999a). Aggressive interactions among males over mating partners are frequent, and infanticide by males occurs in howler monkeys (Crockett and Seklic 1984; Agoramoorthy and Rudran 1995; Crockett and Janson 2000). In the Venezuelan red howler monkeys (Alouatta seniculus), infanticide reduces the interbirth interval, and the risk of infanticide increases with the number of females within a group (Crockett and Rudran 1987; Crockett and Janson 2000). Infanticide may prompt female emigration and lead to a fast life history. In the other three Ateline genera with male philopatry, however, infanticide rarely occurs. On the other hand, coalitional aggression by males to other males, including killing of immature males and forced copulations by males, have been reported in spider monkeys (Campbell 2003, 2006; Valeo et al. 2006; Gibson et al. 2008). There are large variations in the average interval between consecutive viable births of spider monkeys among long-term study sites [32.0 months in Mexico (Ramos-Fernandez 2003); 34.5 months in Peru (Symington 1988); 43.7 months in Columbia (Y. Shimooka, unpublished data)]. Within-group competition among males and their mating strategies , including sexual coercion , may change the interbirth interval of spider monkeys (Gibson et al. 2008; Shimooka et al. 2008).
A comparison of life history parameters in female great apes indicates that orangutans have the slowest and gorillas have the fastest life history (Table 9.4). These differences are inconsistent with female body weight, and the life history parameters vary with male mating strategies. There are two types of sexually mature male orangutan: “flanged” males , with fully developed secondary sexual features, cheek pads, long hair, and a throat sack, and “non-flanged” males , looking younger without these sexual features but actually having reached the adult age. Flanged males emit loud calls and maintain antagonistic relationships with each other, competing over access to females (Galdikas 1985; Rodman and Mitani 1987; van Schaik and van Hooff 1996). Non-flanged males occasionally travel in groups to follow the same females (van Schaik et al. 2004). The strong female mating preference for the dominant flanged males facilitates their exclusive mating, but roaming non-flanged males occasionally force females to mate with them. Females do not form prolonged consorts with either flanged or non-flanged males. Females with dependent infants rarely associate with males, which never take care of infants. The lack of the male’s care and protection may promote the female’s solitary travel and preclude early weaning and reproduction.
Gorillas form a cohesive group and have no territoriality with neighboring groups (Schaller 1963; Watts 1998; Tutin 1996; Yamagiwa et al. 1996; Bermejo 2004). High cohesiveness and one-male group composition may have promoted a rapid life history compared to other apes. The leading male monopolizes most of the copulations with fertile females and takes intensive care of the offspring before and after weaning (Fossey 1979; Stewart and Harcourt 1987; Fletcher 2001; Stewart 2001). These social features may facilitate weaning at an earlier age, shorter interbirth interval, and female reproduction at an earlier age for gorillas than for chimpanzees. Furthermore, infanticide by male gorillas occurs as a mating tactic to resume female estrus and thereby shorten interbirth interval (Watts 1989; Yamagiwa et al. 2009).
Female chimpanzees and bonobos copulate with multiple males and take care of their infants by themselves (Tutin 1979; Goodall 1986; Kano 1992). Female chimpanzees tend to associate or interact with other adults less frequently than males, and mothers with dependent infants rarely join males (Wrangham 1979; Nishida 1979; Boesch and Boesch-Achermann 2000). Infanticide occurs in chimpanzees, but promiscuous mating may reduce it, as do female tactics, with paternity confusion (Hasegawa 1989; Van Noordwijk and van Schaik 2000). Fission–fusion features and promiscuous mating may prevent males from monopolizing mating and lead to a slower life history than gorillas.
Life history parameters in female great apes vary with their social features. Female orangutans, who usually live a solitary life , show the slowest life history. Maturing females need to establish their own home range and relationships with reproductive mates after separation from their mothers. They need a longer time to attain these tasks than female chimpanzees and gorillas, who transfer into other groups immediately after emigration. Solitary travel for weeks or months by female chimpanzees or gorillas has rarely been observed (Wrangham 1979; Nishida 1979; Goodall 1986; Boesch and Boesch-Achermann 2000; Watts 2003; Stokes et al. 2003; Yamagiwa et al. 2003). Female chimpanzees and gorillas may easily find mates for reproduction in the group they join and thus may not need to establish their own ranging areas. Instead, they need to establish social relationships with unrelated conspecifics within the new group. Immigrant females usually are harassed by resident females in both chimpanzees and gorillas (Goodall 1986; Idani 1991; Furuichi 1997; Watts 1991a, 1994; Harcourt and Stewart 2007). Female gorillas get support from the leading males, who frequently intervene in conflicts among females (Watts 1997; Harcourt and Stewart 2007). Immigrant female bonobos first establish affiliative relationships with resident females through sociosexual behavior (Idani 1991; Kano 1992; Furuichi 1997; Hohmann et al. 1999). Although group life may facilitate female chimpanzees in starting or resuming reproduction earlier than do female orangutans, more complex social relationships within a group and unassisted caretaking may prevent them from having a fast life history.
The costs of female transfer may also prevent Atelinae from having rapid reproduction (Strier 1999a). Female woolly monkeys transfer between groups and give birth seasonally, and immigrant females are usually accepted peacefully (Nishimura 1994, 2003). The most frequent fission–fusion in grouping is found in spider monkeys (Symington 1990; Strier 1992). Female muriquis transfer throughout the year, and immigrant females are occasionally threatened by resident females and start copulation several months after immigration (Printes and Strier 1999; Strier and Ziegler 2000). Variations in grouping and relationships among females are the driving force of the fast–slow continuum in the life history of Atelinae.
4 Ecological Factors Versus Social Factors Influencing Life History Parameters in Hominidae
Ecological factors, such as nutritional conditions and predation risks, may also change life history parameters. Ecological risk aversion theory predicts that seasonal fluctuation in the availability of high-quality foods, increasing intraspecific feeding competition , and low predation risk, reducing mortality rate, may lead to a slow life history (Janson and van Schaik 1993). In this respect, great apes with low predation risk have a slower life history than Cercopithecus monkeys, and frugivorous orangutans and chimpanzees have a slower life history than folivorous gorillas. Spider monkeys, relying most heavily on fruits with the most frequent fission–fusion grouping, have the slowest life history among Atelinae (Symington 1990; Strier 1992; Shimooka 2005). Local variation within a genus or species in relationship to different environmental conditions may elucidate the ecological role in shaping life history parameters. The great apes have been intensively studied for many years at different sites and are the best subjects for examining these local variations.
Sumatran orangutans are more frugivorous and tend to associate more frequently than Bornean orangutans (van Schaik 1999; Wich et al. 1999; Delgado and van Schaik 2000). The association rate and time tend to increase with an increase in fruit availability (Sugardjito et al. 1987). However, the association of female orangutans or an increase in association of mother–offspring units along with an increase in food availability may not be linked with fast reproduction but does extend the interbirth interval (Wich et al. 2004). Two models tried to explain these tendencies. The ecological energetic model predicts that the lower and unpredictable energy availability in Borneo may lead to reproductive output scheduled according to sufficient energy availability during the period of fruit abundance (Knott 2001; Knott et al. 2009). The ecological life history model predicts that the higher energy availability and lower mortality in Sumatra may lead to slow life history (Wich et al. 2004, 2009). Another explanation is that the difference in life history may be caused by female response to the ratio of flanged/non-flanged males and to their different mating strategies. In Sumatra, where more non-flanged males (trying forcible mating) are available, females may have slow reproduction (Delgado and van Schaik 2000). In Borneo, where more flanged males (trying monopolization of mating) are available, females may have fast reproduction. However, more detailed observations on mating and reproduction are needed to verify these interpretations.
The slower physical maturation of frugivorous western gorillas compared to folivorous mountain gorillas may support the risk-aversion hypothesis (Breuer et al. 2009). The longer interbirth interval is also suggested for western gorillas, although no difference is observed in other reproductive parameters between them (Robbins et al. 2004). Between the subspecies of Gorilla beringei, some differences are observed in life history parameters (Table 9.4). In the montane forest of Kahuzi, G. b. graueri live at a lower altitude and have a more frugivorous diet than does G. b. beringei in the Virungas; they show slightly longer interbirth intervals and lower infant mortality than G. b. beringei (Yamagiwa et al. 2003). However, this difference may be caused by the presence or absence of infanticide by males, rather than ecological factors. Killing of infants by males occurred frequently in the Virunga gorilla population as a male reproductive tactic to hasten resumption of female estrus (Fossey 1984; Watts 1989). To avoid infanticide, females tend to travel with silverbacks (fully matured males) and to join a group with multiple males to seek more reliable protection (Watts 1996; Robbins 1999). These female strategies may reduce interbirth intervals in the Virungas. By contrast, infanticide has not been observed until recently in Kahuzi, and females occasionally form all-female groups for a prolonged period after the death of the leading silverbacks (Yamagiwa and Kahekwa 2001). Infant mortality (until the second year from birth) is higher for Virunga gorillas (33.9 %) than for Kahuzi gorillas (26.1 %), and 37 % of infant mortality in the Virungas was the result of infanticide (Watts 1991b; Yamagiwa et al. 2003). The interval between the death of an infant and a next birth for Virunga gorillas (1.0 years) is shorter than that for Kahuzi gorillas (2.2 years) (Yamagiwa et al. 2003). Female Virunga gorillas may find mates and resume reproduction more rapidly after the death of infants. Three cases of infanticide have recently been observed in a group of Kahuzi gorillas, and the birthrate in the groups is very high (Yamagiwa et al. 2009, 2011). The occurrence of infanticide may promote the fast life history of gorillas.
A comparison among four study sites of chimpanzees in natural habitats shows a similarity in interbirth interval (5.2–5.8 years on average). Age at first reproduction is also relatively constant among sites (13.2–14.3 years on average), except for chimpanzees at Bossou (10.9 years). Sugiyama (2004) attributed these findings to nutritional conditions. In Bossou, high-quality foods, such as fruits and nuts, are concentrated in the study group’s core area, and their tool using behavior may mitigate low nutrition during the period of fruit scarcity (Sugiyama 1997, 2004; Yamakoshi 1998). Small group size and isolated conditions may also speed the start of female reproduction in Bossou. Group size of Bossou chimpanzees is kept around 20, which is far smaller than those of Gombe, Mahale, and Tai (30–100). The study group of Bossou has been isolated from neighboring groups for 26 years, and most of the females started their first reproduction in their natal groups (Sugiyama 2004). The study group included only one adult male for more than 10 years (who stayed as an alpha male for 20 years), and the maturing males emigrated, probably because of increased competition with the leading male (Sugiyama 1999). Isolation from neighboring populations and the polygynous composition of the study group may have limited female mate choice to a single male and promoted their earlier start of reproduction. Females who gave birth in their natal group also were younger at their first birth than immigrant females in Mahale (Nishida et al. 2003). Females emigrating from their natal groups may suffer several costs for reproduction, such as reduced foraging efficiency in unknown ranges or harassment by resident females in the unfamiliar groups (Pusey 1980; Goodall 1986; Williams et al. 2002; Nishida et al. 2003).
Nutritional conditions also constitute limiting factors on the life history of great apes. Orangutans in captivity show faster reproduction than those in natural habitats (Knott and Kahlenberg 2007). Artificial feeding and grouping may permit faster growth and reproduction of orangutans than in their solitary natural habitats (Knott 2001). Rehabilitant free-ranging female orangutans also show an earlier age at first birth and a longer interbirth interval than wild females (Kuze et al. 2012). High energy intake from provisioning may enable their faster reproduction. The life history parameters of gorillas in natural habitats have been considered to be similar to those in captivity. A folivorous diet may supplement fruit scarcity, and the cohesive group formation with a one-male mating system may facilitate faster female reproduction at a level closer to their evolutionary potential (Tutin 1994; Harcourt and Stewart 2007). However, data on life history have come from folivorous mountain gorillas, and data in captivity are from frugivorous western gorillas. Recent studies on western gorillas in their natural habitats show a slower life history than captive gorillas, and regular provisioning may promote the faster life history of gorillas (Robbins et al. 2004; Breuer et al. 2009)
Female chimpanzees in captivity also tend to start reproduction earlier and have shorter interbirth intervals (Table 9.4). A rich nutritional condition from regular feeding, the limited selection of mates, and restricted movement may facilitate fast reproduction, as observed for Bossou chimpanzees. However, the interbirth interval at Bossou is longer than those in captivity and similar to those in natural habitats. Provisioned but free-ranging chimpanzees from the rehabilitation project in River Gambia National Park show a similar age at first reproduction and interbirth interval to those in natural habitats, rather than to those in captivity (Marsden et al. 2006). These observations suggest that limited movement under confined conditions may promote the shorter interbirth intervals of female chimpanzees in captivity.
5 Discussion
5.1 Costs of Female Transfer
In contrast to cetaceans, characterized by high mobility and animal diets in aquatic environments, the life history traits of primates with vegetarian diets are strongly linked with dispersal patterns of females. As primate socioecology predicts (Sterck et al. 1997), food availability and predation pressure shape female gregariousness and association between sexes, which in turn change life history parameters. Most cetaceans form matrilineal social groups, and kin-related females tend to cooperate in rearing calves in a school (Kasuya 1995; Shane et al. 1986). Food availability relative to body size may affect the duration of lactation and thereby female reproductive strategies (Martin and Rothery 1993; Whitehead and Weilgart 2000; Mann et al. 2000). By contrast, some primate species (Atelinae and Hominidae) form nonmatrilineal groups in which females transfer, and female dispersal may promote slow life history (Strier 1999a; Kappeler et al. 2003; Harcourt and Stewart 2007). Our study suggests that male reproductive tactics, adding to ecological factors, may affect the cost of female transfer and shape the fast–slow continuum in the life history traits of female-dispersal species.
In both Atelinae and Hominidae, high gregariousness among females observed in howler monkeys and gorillas is linked with faster life history than fission–fusion grouping in spider monkeys, muriquis, and chimpanzees and solitary travel in orangutans. In female-dispersal species of primates, in which females usually start reproductive life after separation from their mothers or from their natal groups, frequent association with other conspecifics and high group cohesiveness may reduce costs of female foraging and promote rapid reproduction. Solitary travel is costly in terms of the need for vigilance against predators and for finding and occupying high-quality food patches, and these costs may lead to delayed age at first reproduction and a longer interbirth interval. Association with other adults, especially with a single male that positively takes care of an infant, will decrease these costs and enable them to develop a faster life history.
In male philopatric species such as wooly monkeys, spider monkeys, muriquis, and chimpanzees, differences in life history may reflect a female’s flexible foraging efforts, according to the different spatiotemporal distribution of high-quality foods, as a means to attain reproductive success. Female atelines show large variations in grouping within and between species according to the distribution of high-quality foods (Symington 1990; Strier 1992; Shimooka 2005), and female chimpanzees also show large variations in grouping between and within species (Goodall 1986; Kano 1992; Boesch and Boesch-Achermann 2000). Female great apes may promote these variations as a way of coping with strong feeding competition because of their lower digestive ability for unripe fruits and mature leaves compared with Old World monkeys (Yamagiwa 2004). In both orangutans and chimpanzees, females with dependent infants tend to travel without other adult conspecifics (van Schaik 1999; Wrangham 1979), probably because of the higher costs of feeding competition. Female bonobos form larger parties than female orangutans and chimpanzees, and even females with dependent infants usually associate with other adult females and males. Large fruit patches available throughout the year and abundant fallback foods such as terrestrial herbs may mitigate the cost of grouping among bonobos (Wrangham 1986). Frequent sociosexual behavior may also reduce social tension induced by feeding competition among them (Kuroda 1980; Kano 1989; Kitamura 1989; Parish 1994). Female gorillas do not alter their grouping patterns in response to fruit availability, although they extend their daily path length with an increasing frugivorous diet (Goldsmith 1999; Doran et al. 2002; Yamagiwa et al. 2003).
5.2 Male Reproductive Tactics and Life History of Female Great Apes
Sexual coercion of males may have strong influences on life history traits in female-dispersal species of primates. Infanticide by males promotes faster life history in howler monkeys and gorillas (Crockett and Janson 2000; Harcourt and Stewart 2007; Yamagiwa et al. 2011). To avoid infanticide, females move and choose males with which to associate, and they stay with males who have the ability to protect their infants. These movements result in stable associations between females and the protector males that lead to a fast life history.
Infanticide may shape local variation in group composition within the genus Gorilla through female strategies against it. In the Virungas, where infanticide by extra-group males frequently occurs, females tend to transfer into multiple male groups , seeking more protection against infanticide (Watts 1989, 1996). The female preference for multimale groups may have increased the number of females within the multimale groups and enabled maturing males to have a mating opportunity (Robbins 1995, 2001; Watts 1996, 2000; Robbins and Robbins 2005). The dominant males tend to tolerate mating by kin-related subordinate males and cooperate to defend their groups against solitary males or other groups (Robbins 2001; Watts 2003). A recent genetic analysis of paternity in four multimale groups in the Virungas indicated that both dominant and subordinate males enjoyed reproductive success, with the dominants siring an average of 85 % of group offspring (Bradley et al. 2005). Philopatric males tend to start reproduction earlier and to sire more offspring than dispersal males (Robbins and Robbins 2005). Female choice of multimale groups may have prevented male dispersal from the natal group and consequently promoted faster life histories of both males and females at Virungas (Harcourt and Stewart 2007).
By contrast, eastern lowland gorillas rarely form multimale groups and females occasionally transfer with other females and immatures at Kahuzi, where infanticide has rarely been reported (Yamagiwa et al. 1993; Yamagiwa and Kahekwa 2001). Although long-term studies have not yet been conducted on western gorillas in the lowland tropical forests, very little infanticide has been reported (Stokes et al. 2003). Frugivorous diets may prevent gorillas from forming large multimale groups in the lowland forests, and females may not have the option of transferring into multimale groups (Yamagiwa et al. 2003). Females tend to prefer to transfer into smaller groups and remain together to accept new males after the death of the leading male (Stokes 2004). Recent DNA analysis suggested a network among related males in separate but neighboring groups instead of forming multimale groups in the population of western lowland gorillas at Mondika (Bradley et al. 2004). Consequently, by forming a network of related males in neighboring one-male groups, rather than forming multimale groups, frugivorous western gorillas may be able to avoid infanticide (Fig. 9.3).
Although infanticide rarely occurs in species with male philopatry, it functions to reduce interbirth interval and may shape interspecies variation in spider monkeys (Gibson et al. 2008; Shimooka et al. 2008). Their fission–fusion grouping is characterized by frequent associations among kin-related males within a group and communal defense by these males (Symington 1990, 1988; Strier 1992; Shimooka 2005). Within-group aggression among males and forced mating by males tend to occur in the groups of spider monkeys in which operational sex ratios were highly skewed toward males (Gibson et al. 2008). These observations suggest that competition among males over mates and male coercive mating may promote faster life history traits in atelines, although long-term data on individual demography are needed for further speculation.
By contrast, sexual coercion may promote a slow life history in the genus Pan. As do spider monkeys, both chimpanzees and bonobos form fission–fusion grouping and males tend to remain in their natal groups after maturity (Nishida 1979; Goodall 1986; Kano 1992). Females show promiscuous mating patterns , but their gregariousness and male mating tactics vary between species and across populations (Tutin 1979; Tutin and McGinnis 1981; Furuichi 1987). Male chimpanzees are generally more gregarious than females, and the most dominant males try to monopolize mating (Tutin 1979; Hasegawa and Hiraiwa-Hasegawa 1983; Boesch and Boesch-Achermann 2000). Males form a coalition for communal defense that occasionally results in fatal communal attacks in eastern chimpanzees (Goodall et al. 1979; Nishida et al. 1985; Watts et al. 2006). Infanticide by extra-group males also occurs as an extension of their territorial aggression (Goodall 1977; Hamai et al. 1992; Arcadi and Wrangham 1999; Watts and Mitani 2000; Muller 2007). Consequently, male eastern chimpanzees sire most of their offspring within their communities (Constable et al. 2001; Inoue et al. 2008; Wroblewski et al. 2009; Newton-Fisher et al. 2010). Infanticide by females was also observed in Gombe and Budongo and is considered as a female tactic for increased conflict over resources with new immigrants (Goodall 1977; Townsend et al. 2007; Muller 2007). These aggressions may promote solitary travel of females, especially females with suckling infants, to avoid infanticide, and thus may lead to slow reproduction.
There are considerable differences in coalitional formation and intergroup attacks by males between western and eastern chimpanzees (Goodall et al. 1979; Nishida et al. 1985; Herbinger et al. 2001; Wilson and Wrangham 2003; Watts et al. 2006; Lehman and Boesch 2003). Female western chimpanzees tend to associate and groom with each other frequently at both Bossou and Tai compared to eastern chimpanzees (Sugiyama 1988; Boesch 1991; Lehmann and Boesch 2008). Nevertheless, the interbirth interval parameters of female chimpanzees at both Tai and Bossou and the age of first reproduction at Tai are similar to those of female eastern chimpanzees (Table 9.4). However, the age of first reproduction at Bossou is far earlier than those of other sites, reflecting the conditions observed in captivity. The uniqueness of Bossou chimpanzees is shown by their one-male group composition, where only one male monopolizes mating for a prolonged period (Sugiyama 2004). This social situation is similar to that in captivity, where most groups include only one adult male. Variations in social features between western and eastern chimpanzees are also explained by the distribution of defendable high-quality food resources and predation risk (Wittig and Boesch 2003; Lehmann and Boesch 2008). High nutritional conditions lead to a shorter interbirth interval in captivity than in the wild (Table 9.4). The age of first reproduction may be influenced by both nutritional conditions and male mating tactics , especially the degree of mating monopolization.
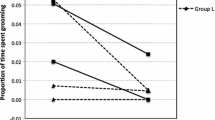
The influence of male mating tactics on the speed of life history is distinct between the two species of Pan. Male bonobos tend to associate with females rather than with other males, and their ranks reflect their mothers’ ranks because of the mothers’ strong support in agonistic conflicts between males (Kano 1992; Parish 1994; Furuichi 1997; Hohmann et al. 1999). Different groups of bonobos sometimes intermingle to stay together, and both females and males exhibit affiliative social interactions between groups (Idani 1990; Hashimoto et al. 2008). Weaker competition among male bonobos enables females to maintain stable association with males and may facilitate the search for mating partners and lead to a shorter interbirth interval than chimpanzees (Fig. 9.4).
In summary, male mating tactics may change the life history of the great apes in different ways. Female dispersal and independent reproduction from related conspecifics may enable them to form various social structures and flexible life history traits according to male mating strategies. Ecological factors basically shape the gregariousness of females in female-dispersal species, but they can choose from a wide variety of feeding strategies, from individual foraging to moving in cohesive groups. Males also take various mating tactics according to female movement and association patterns, which in turn also vary with male associations and mating strategies. Although the influences of these ecological and social factors on the life history of great apes differ between genera, between species, and between populations, the solitary nature may urge females to choose a slower life history, whereas stable associations between males and females may promote a faster life history (Fig. 9.5). Frugivorous orangutans and chimpanzees may suffer more costs of female movement through decreased foraging efficiency than folivorous gorillas, and chimpanzees with fission–fusion grouping may suffer more social stress than gorillas in highly cohesive groups. Such differences may generally shape the fast–slow continuum of life history in female-dispersal primate species.
References
Agoramoorthy G, Rudran R (1995) Infanticide by adult and subadult males in free-ranging red howler monkeys of Venezuela. Ethology 99:75–88
Allman JM, McLaughlin T, Hakeem A (1993) Brain weight and life-span in primate species. Proc Natl Acad Sci USA 90:118–122
Altmann J, Alberts SC (2003) Variability in reproductive success viewed from a life-history perspective in baboons. Am J Hum Biol 15:401–409
Altmann J, Hausfater G, Altmann SA (1988) Determinants of reproductive success in savanna baboons. In: Clutton-Brock TH (ed) Reproductive success. University of Chicago Press, Chicago, pp 403–418
Anderson HB, Emery Thompson M, Knott M, Perkins CD (2008) Fertility and mortality patterns of Bornean and Sumatran orangutans: is there a species difference in life history? J Hum Evol 54:34–42
Arcadi AC, Wrangham RW (1999) Infanticide in chimpanzees: review of cases and a new within-group observation from the Kanyawara study group in Kibale National Park. Primates 40:337–351
Barton RA, Byrne RW, Whiten A (1996) Ecology, feeding competition, and social structure in baboons. Bahav Ecol Sociobiol 38:321–329
Bermejo M (2004) Home-range use and intergroup encounters in western gorillas (Gorilla g. gorilla) at Lossi Forest, North Congo. Am J Primatol 64:223–232
Boesch C (1991) The effects of leopard predation on grouping patterns in forest chimpanzees. Behaviour 117:220–242
Boesch C, Boesch-Achermann H (2000) The chimpanzees of Tai Forest. Cambridge University Press, Cambridge
Borries C, Koenig A, Winkler P (2001) Variation of life history traits and mating patterns in female langur monkeys (Semnopithecus entellus). Behav Ecol Sociobiol 50:391–402
Bradley BJ, Doran-Sheehy DM, Lukas D, Boesch C, Vigilant L (2004) Dispersed male networks in western gorillas. Curr Biol 14:510–513
Bradley BJ, Robbins MM, Williamson EA, Steklis HD, Steklis NG, Eckhardt N, Boesch C, Vigilant L (2005) Mountain gorilla tug-of-war: silverbacks have limited control over reproduction in multimale groups. Proc Natl Acad Sci USA 102:9418–9423
Breuer T, Breuer-Ndoundou Hockemba M, Olejniczak C, Parnell RJ, Stokes EJ (2009) Physical maturation, life-history classes and age estimates of free-ranging western gorillas: insights from Mbeli Bai, Republic of Congo. Am J Primatol 71:106–119
Campbell CJ (2003) Female directed aggression in free-ranging Ateles geoffroyi. Int J Primatol 24:223–238
Campbell (2006) Lethal intragroup aggression by adult male spider monkeys (Ateles geoffroyi). Am J Primatol 68:1197–1201
Charnov EL (1991) Evolution of life history variation among female mammals. Proc Natl Acad Sci USA 88:1134–1137
Charnov EL (1993) Life history invariants: some explorations of symmetry in evolutionary ecology. Oxford University Press, Oxford
Cheney DL, Seyfarth RM, Andleman S, Lee PC (1988) Reproductive success in vervet monkeys. In: Clutton-Brock TH (ed) Reproductive success. University of Chicago Press, Chicago
Connor RC, Wells R, Mann J, Read A (2000) The bottlenose dolphin: social relationships in a fission–fusion society. In: Mann J, Connor R, Tyack P, Whitehead H (eds) Cetacean societies: field studies of whales and dolphins. University of Chicago Press, Chicago, pp 91–126
Constable JL, Ashley MV, Goodall J, Pusey AE (2001) Noninvasive paternity assignment in Gombe chimpanzees. Mol Ecol 10:1279–1300
Crockett CM, Janson CH (2000) Infanticide in red howlers: female group size, male membership, and a possible link to folivory. In: van Schaik CP, Janson CH (eds) Infanticide by males and its implications. Cambridge University Press, Cambridge, pp 75–98
Crockett CM, Pope TR (1993) Consequences of sex differences in dispersal for juvenile red howler monkeys. In: Pereira ME, Fairbanks LA (eds) Juvenile primates: life history, development, and behavior. Oxford University Press, Oxford, pp 104–118
Crockett CM, Rudran R (1987) Red howler monkey birth data. II: Interannual, habitat, and sex comparisons. Am J Primatol 13:369–384
Crockett CM, Seklic R (1984) Infanticide in red howler monkeys (Alouatta seniculus). In: Hausfater G, Hrdy SB (eds) Infanticide: comparative and evolutionary perspectives. Aldine De Gruyter, New York, pp 173–191
Delgado R, van Schaik CP (2000) The behavioral ecology and conservation of the orangutan (Pongo pygmaeus): a tale of two islands. Evol Anthropol 9:201–218
Doran DM, Jungers WL, Sugiyama Y, Fleagle JG, Heesy CP (2002) Multivariate and phylogenetic approaches to understanding chimpanzee and bonobo behavioral diversity. In: Boesch C, Hohmann G, Marchant LF (eds) Behavioural diversity in chimpanzees and bonobos. Cambridge University Press, Cambridge, pp 14–34
Drickamer LC (1974) A ten-year summary of reproductive data for free ranging Macaca mulatta. Folia Primatol 21:61–80
Dunbar RIM (1988) Primate social system. Helm, London
Eisenberg JF (1981) The mammalian radiations: an analysis of trends in evolution, adaptation, and behavior. Chicago University Press, Chicago
Fairbanks LA (1990) Reciprocal benefits of allomothering for female vervet monkeys. Anim Behav 40:553–562
Fedigan LM, Rose LM (1995) Inter-birth interval variation in three sympatric species of Neotropical monkey. Am J Primatol 37:9–24
Fletcher A (2001) Development of infant independence from the mother in wild mountain gorillas. In: Robbins MM, Sicotte P, Stewart KJ (eds) Mountain gorillas: three decades of research at Karisoke. Cambridge University Press, London, pp 153–182
Fossey D (1979) Development of the mountain gorilla (Gorilla gorilla beringei): the first thirty-six months. In: Hamburg DA, McCown ER (eds) The great apes. Benjamin/Cummings, Menlo Park, pp 138–184
Fossey D (1984) Infanticide in mountain gorillas (Gorilla gorilla beringei) with comparative notes on chimpanzees. In: Hausfater G, Hrdy SB (eds) Infanticide: comparative and evolutionary perspectives. Aldine, Hawthorne, pp 217–236
Furuichi T (1987) Sexual swelling, receptivity and grouping of wild pygmy chimpanzee females at Wamba, Zaire. Primates 28:309–318
Furuichi T (1997) Agonistic interactions and matrifocal dominance rank of wild bonobos (Pan paniscus) at Wamba. Int J Primatol 18:855–875
Furuichi T, Hashimoto C (2002) Why female bonobos have a lower copulation rate during estrus than chimpanzees. In: Boesch C, Hohmann G, Marchant LF (eds) Behavioral diversity in chimpanzees and bonobos. Cambridge University Press, New York, pp 156–167
Furuichi T, Idani G, Ihobe H, Kuroda S, Kitamura K, Mori A, Enomoto T, Okayasu N, Hashimoto C, Kano T (1998) Population dynamics of wild bonobos (Pan paniscus) at Wamba. Int J Primatol 19:1029–1043
Galdikas BMF (1984) Adult female sociality among wild orangutans at Tanjung Putting Reserve. In: Small MF (ed) Female primates: studies by women primatologists. Liss, New York, pp 217–235
Galdikas BMF (1985) Orangutan sociality at Tanjung Putting, Central Borneo. Int J Primatol 9:1–35
Gibson KN, Vick LG, Palma AC, Carrasco FM, Taub D, Ramos-Fernandez G (2008) Intra-community infanticide and forced copulation in spider monkeys: a multi-site comparison between Cocha Cashu, Peru and Punta Laguna, Mexico. Am J Primatol 70:485–489
Goldsmith ML (1999) Ecological constraints on the foraging effort of western gorillas (Gorilla gorilla gorilla) at Bai Hokou, Central African Republic. Int J Primatol 20:1–23
Goodall J (1977) Infant killing and cannibalism in free-living chimpanzees. Folia Primatol (Basel) 28:259–282
Goodall J (1986) The chimpanzees of Gombe. Belknap, Cambridge
Goodall J, Bandora A, Bergmann E, Busse C, Matama H, Mpongo E, Pierce A, Riss D (1979) Intercommunity interactions in the chimpanzee population of the Gombe National Park. In: Hamburg DA, McCown ER (eds) The great apes. Benjamin/Cummings, Menlo Park, pp 13–53
Gouzoules H, Gouzoules S, Fedigan LM (1982) Behavioral dominance and reproductive success in female Japanese monkeys (Macaca fuscata). Anim Behav 30:1138–1190
Hamai M, Nishida T, Takasaki H (1992) New records of within-group infanticide and cannibalism in wild chimpanzees. Primates 33:151–162
Harcourt AH (1992) Coalitions and alliances: are primates more complex than non-primates? In: Harcourt AH, de Waal FBM (eds) Coalitions and alliances in human and other animals. Oxford University Press, Oxford, pp 445–472
Harcourt AH, Stewart KJ (2007) Gorilla society: conflict, compromise and cooperation between the sexes. University of Chicago Press, Chicago
Harvey PH, Purvis A (1999) Understanding the ecological and evolutionary reasons for life history variation: mammals as a case study. In: McGlade J (ed) Advanced ecological theory: principles and applications. Blackwell, Oxford, pp 232–248
Harvey PH, Martin RD, Clutton-Brock TH (1987) Life histories in comparative perspective. In: Smuts BB, Cheney DL, Seyfarth RM, Wrangham RW, Struhsaker TT (eds) Primate societies. University of Chicago Press, Chicago, pp 181–196
Hasegawa T (1989) Sexual behavior of immigrant and resident female chimpanzees at Mahale. In: Heltne PG, Marquardt LA (eds) Understanding chimpanzees. Harvard University Press, Cambridge, pp 90–103
Hasegawa T, Hiraiwa-Hasegawa M (1983) Opportunistic and restrictive mating among wild chimpanzees in the Mahale Mountains, Tanzania. J Ethol 1:75–85
Hashimoto C, Tashiro Y, Hibino E, Mulabwa M, Yangonze K, Furuichi T, Idani G, Takenaka O (2008) Longitudinal structure of a unit-group of bonobos: male philopatry and possible fusion of unit-groups. In: Furuichi T, Thompson J (eds) The bonobos: behavior, ecology, and conservation. Springer, New York, pp 107–119
Henzi P, Barrett L (1999) The value of grooming to female primates. Primates 40:47–59
Henzi P, Barrett L (2003) Evolutionary ecology, sexual conflict, and behavioral differentiation among baboon populations. Evol Anthropol 12:217–230
Herbinger L, Boesch C, Rothe H (2001) Territory characteristics among three neighboring chimpanzee communities in the Tai National Park, Ivory Coast. Int J Primatol 32:143–167
Hohmann G, Gerloff U, Tautz D, Fruth B (1999) Social bonds and genetic ties: kinship, association, and affiliation in a community of bonobos (Pan paniscus). Behaviour 136:1219–1235
Idani G (1990) Relations between unit-groups of bonobos at Wamba, Zaire: encounters and temporary fusions. Afr Stud Monogr 11:153–186
Idani G (1991) Social relationships between immigrant and resident bonobo (Pan paniscus) females at Wamba. Folia Primatol (Basel) 57:83–95
Inoue E, Inoue-Murayama M, Vigilant L, Takenaka O, Nishida T (2008) Relatedness in wild chimpanzees: influence of paternity, male philopatry, and demographic factors. Am J Phys Anthropol 137:256–262
Itoigawa N, Tanaka T, Ukai N, Fujii H, Kurokawa T, Koyama T, Ando A, Watanabe Y, Imakawa S (1992) Demography and reproductive parameters of a free-ranging group of Japanese macaques (Macaca fuscata) in Katsuyama. Primates 33:49–68
Janson CH, van Schaik CP (1993) Ecological risk aversion in juvenile primates: slow and steady wins the race. In: Pereira ME, Fairbanks LA (eds) Juvenile primates: development and behavior. Oxford University Press, Oxford, pp 57–74
Jones KE, MacLarnon A (2001) Bat life histories: testing models of mammalian life history evolution. Evol Ecol Res 3:465–476
Kano T (1989) The sexual behavior of pygmy chimpanzees. In: Heltne PG, Marcquardt LA (eds) Understanding chimpanzees. Harvard University Press, Cambridge, pp 176–183
Kano T (1992) The last ape: pygmy chimpanzee behavior and ecology. Stanford University Press, Stanford
Kappeler PM, Pereira ME (eds) (2003) Primate life history and socioecology. University of Chicago Press, Chicago
Kappeler PM, Pereira ME, van Schaik CP (2003) Primate life histories and socioecology. In: Kappeler PM, Pereira ME (eds) Primate life histories and socioecology. Chicago University Press, Chicago, pp 1–23
Kasuya T (1995) Overview of cetacean life histories. In: Blix AS, Walløe, Ulltang Ø (eds) Whales, seals, fish and man: proceedings of the international symposium on the biology of marine mammals in the northeast Atlantic, Tromsø, Norway, 29 November–1 December 1994. Elsevier, Amsterdam, pp 481–497
Kasuya T, Jones LL (1984) Behavior and segregation of the Dall’s porpoise in the northwestern North Pacific Ocean. Sci Rep Whales Res Inst 35:107–128
Kasuya T, Balcomb K, Brownell RL Jr (1997) Life history of Baird’s whales off the Pacific coast of Japan. Rep Int Whaling Comm 47:969–979
Kirschofer R (1987) International studbook of the gorilla, Gorilla gorilla, 1985. Frankfurt Zoological Garden, Frankfurt
Kitamura K (1989) Genito-genital contacts in the pygmy chimpanzee (Pan paniscus). Afr Stud Monogr 10:46–67
Knott CD (2001) Female reproductive ecology of the apes: implications for human evolution. In: Ellison PT (ed) Reproductive ecology and human evolution. de Gruyter, New York, pp 429–463
Knott CD, Kahlenberg SM (2007) Orangutans in perspective. In: Campbell CJ, Fuentes A, MacKinnon KC, Panger M, Beader SK (eds) Primates in perspective. Oxford University Press, New York, pp 290–305
Knott CD, Thompson ME, Wich SA (2009) The ecology of female reproduction in wild orangutans. In: Wich SA, Utami SS, Mitra Setia T, van Schaik CP (eds) Orangutans: geographical variation in behavioral ecology and conservation. Oxford University Press, Oxford, pp 171–188
Kuroda S (1980) Social behavior of the pygmy chimpanzees. Primates 21:181–197
Kuroda S (1989) Developmental retardation and behavioral characteristics of pygmy chimpanzees. In: Heltne PG, Marquardt AE (eds) Understanding chimpanzees. Chicago Academy of Sciences, Chicago, pp 184–193
Kuze N, Sipangkui S, Malim T, Bernard H, Ambu L, Kohshima S (2008) Reproductive parameters over a 37-year period of free-ranging female Borneo orangutans at Sepilok Orangutan Rehabilitation Centre. Primates 49:126–134
Kuze N, Dellatore D, Banes GL, Pratje P, Tajima T, Russon AE (2012) Factors affecting reproduction in rehabilitant female orangutans: young age at first birth and short inter-birth interval. Primates 53:181–192
Lee PC (1996) The meaning of weaning: growth, lactation, and life history. Evol Anthropol 5:87–96
Lee PC, Majluf P, Gordon IJ (1991) Growth, weaning and maternal investment from a comparative perspective. J Zool 225:99–114
Lehman J, Boesch C (2003) Social influences on ranging patterns among chimpanzees (Pan troglodytes verus) in the Tai National Park, Cote d’Ivoire. Behav Ecol 14(5):642–649
Lehmann J, Boesch C (2008) Sexual differences in chimpanzee sociality. Int J Primatol 29:65–81
Mann J, Smuts BB (1999) Behavioral development of wild bottlenose dolphin newborns. Behaviour 136:529–566
Mann J, Connor RC, Barre LM, Heithaus MR (2000) Female reproductive success in wild bottlenose dolphins (Tursiops sp.): life history, habitat, provisioning, and group size effects. Behav Ecol 11:210–219
Marsden BS, Marsden D, Thompson EM (2006) Demographic and female life history parameters of free-ranging chimpanzees at the chimpanzee rehabilitation project, River Gambia National Park. Int J Primatol 27:391–410
Martin RD (1995) Phylogenetic aspects of primate reproduction: the context of advanced maternal care. In: Pryce CR, Martin RD, Skuse D (eds) Motherhood in human and nonhuman primates. Karger, Basel, pp 16–26
Martin RD (1996) Scaling of the mammalian brain: the maternal energy hypothesis. News Physiol Sci 11:149–156
Martin AR, Rothery P (1993) Reproductive parameters of female long-finned pilot whales (Globicephala melas) around the Faroe Islands. In: Donovan GP, Lockyer CH, Martin AR (eds) Biology of northern hemisphere pilot whales: a collection of papers. Reports of the International Whaling Commission, Special Issue 14. International Whaling Commission, Cambridge, pp 263–304
Miyazaki N, Nishiwaki M (1978) School structure of the striped dolphin off the Pacific coast of Japan. Sci Rep Whales Res Inst 30:65–115
Muller MN (2007) Chimpanzee violence: femmes fatales. Curr Biol 17:365–366
Newton-Fisher NE, Thompson ME, Reynolds V, Boesch C, Vigilant L (2010) Paternity and social rank in wild chimpanzees (Pan troglodytes) from the Budongo Forest, Uganda. Am J Phys Anthropol 142:417–428
Nishida T (1979) The social structure of chimpanzees of Mahale Mountains. In: Hamburg DA, McCown ER (eds) The great apes. Benjamin/Cummings, Menlo Park, pp 73–121
Nishida T, Hiraiwa-Hasegawa M, Hasegawa T, Takahata Y (1985) Group extinction and female transfer in wild chimpanzees in the Mahale Mountains. Z Tierpsychol 67:284–301
Nishida T, Corp N, Hamai M, Hasegawa T, Hiraiwa-Hasegawa M, Hosaka K, Hunt KD, Itoh N, Kawanaka K, Matsumoto-Oda A, Mitani JC, Nakamura M, Norikoshi K, Sakamaki T, Turner L, Uehara S, Zamma K (2003) Demography, female life history, and reproductive profiles among the chimpanzees of Mahale. Am J Primatol 59:99–121
Nishimura A (1994) Social interaction patterns of woolly monkeys (Lagothrix lagothricha): a comparison among the atelines. Sci Eng Rev Doshisha Univ 35(2):235–254
Nishimura A (2003) Reproductive parameters of wild female Lagothrix lagotricha. Int J Primatol 24:707–722
Nunn CL, Pereira ME (2000) Group histories and offspring sex ratios in ringtailed lemurs (Lemur catta). Behav Ecol Sociobiol 48:18–28
Oftedal OT (1997) Lactation in whales and dolphins: evidence of divergence between baleen- and toothed-species. J Mamm Gland Biol Neoplasia 2:205–230
Parish AR (1994) Sex and food control in the “uncommon chimpanzee:” how bonobo females overcome a phylogenetic legacy of male dominance. Ethol Sociobiol 15:157–179
Paul A, Kuester J (1996) Differential reproduction in male and female Barbary macaques. In: Fa JE, Lindburg DG (eds) Evolution and ecology of macaque societies. Cambridge University Press, Cambridge, pp 293–317
Paul A, Preuchoft S, van Schaik CP (2000) The other side of the coin: infanticide and the evolution of affiliative male–infant interactions in Old World primates. In: van Schaik CP, Janson CH (eds) Infanticide by males and its implications. Cambridge University Press, Cambridge, pp 269–292
Printes RC, Strier KB (1999) Behavioral correlates of dispersal in female muriquis (Brachyteles arachnoides). Int J Primatol 20:941–960
Purvis A, Harvey PH (1995) Mammalian life history evolution: a comparative test for Charnov’s model. J Zool 237:259–283
Purvis A, Webster AJ, Agapow P-M, Jones KE, Isaac NJB (2003) Primate life histories and phylogeny. In: Kappeler PM, Pereira ME (eds) Primate life histories and socioecology. Chicago University Press, Chicago, pp 25–40
Pusey AE (1980) Inbreeding avoidance in chimpanzees. Anim Behav 28:543–552
Read AF, Harvey PH (1989) Life history differences among the eutherian radiations. J Zool 219:329–353
Robbins MM (1995) A demographic analysis of male life history and social structure of mountain gorillas. Behaviour 132:21–47
Robbins MM (1999) Male mating patterns in wild multimale mountain gorilla groups. Anim Behav 57:1013–1020
Robbins MM (2001) Variation in the social system of mountain gorillas: the male perspective. In: Robbins MM, Sicotte P, Stewart KJ (eds) Mountain gorillas: three decades of research at Karisoke. Cambridge University Press, Cambridge, pp 29–58
Robbins AM, Robbins MM (2005) Fitness consequences of dispersal decisions for male mountain gorillas (Gorilla gorilla beringei). Behav Ecol Sociobiol 58:295–309
Robbins MM, Bermejo M, Cipolletta C, Magliocca F, Parnell RJ, Stokes E (2004) Social structure and life-history patterns in western gorillas (Gorilla gorilla gorilla). Am J Primatol 64:145–159
Robbins AM, Stoinski TS, Fawsett KA, Robbins MM (2009) Socioecological influences on the dispersal of female mountain gorillas: evidence of a second folivore paradox. Behav Ecol Sociobiol 63:477–489
Rodman PS, Mitani JC (1987) Orangutans: sexual dimorphism in a solitary species. In: Smuts BB, Cheney DL, Seyfarth RM, Wrangham RW, Struhsaker TT (eds) Primate societies. University of Chicago Press, Chicago, pp 146–154
Rosenberger AL, Strier KB (1989) Adaptive radiation of the ateline primates. J Hum Evol 18:717–750
Ross C (1992) life history patterns and ecology of macaque species. Primates 33:207–215
Ross C (1998) Primate life histories. Evol Anthropol 6:54–63
Ross C, MacLarnon A (1995) Ecological and social correlates of maternal expenditure on infant growth in haplorhine primates. In: Pryce C, Martin RD, Skuse D (eds) Motherhood in human and nonhuman primates: biological and social determinants. Karger, Basel, pp 37–46
Schaller GB (1963) The mountain gorilla: ecology and behavior. University of Chicago Press, Chicago
Shane SH, Wells RS, Wursig B (1986) Ecology, behavior and social organization of the bottlenose dolphin: a review. Mar Mamm Sci 2:34–63
Shimooka Y (2005) Sexual differences in ranging of Ateles belzebuth belzebuth at La Macarena, Colombia. Int J Primatol 26:385–406
Shimooka Y, Campbell CJ, Di Fiore A, Felton AM, Izawa K, Link A, Nishimura A, Ramos-Fernandez G, Wallace RB (2008) Demography and group composition of Ateles. In: Campbell CJ (ed) Spider monkeys: behavior, ecology, and evolution of the genus Ateles. Cambridge University Press, Cambridge, pp 329–348
Silk J (1982) Altruism among female Macaca radiata: explanations and analysis of patterns of grooming and coalition formation. Behaviour 79:162–168
Silk J (1987) Social behavior in evolutionary perspective. In: Smuts BB, Cheney DL, Seyfarth RM, Wrangham RW, Struhsaker TT (eds) Primate societies. University of Chicago Press, Chicago, pp 318–329
Sivert J, Karesh WB, Sunde V (1991) Reproductive intervals in captive female western lowland gorillas with a comparison to wild mountain gorillas. Am J Primatol 24:227–234
Stanford CB (1992) Costs and benefits of allomothering in wild capped langurs (Presbytes pileata). Behav Ecol Sociobiol 30:29–34
Stearns SC (1992) The evolution of life histories. Oxford University Press, Oxford
Steenbeck R (2000) Infanticide by males and female choice in wild Thomas’s langurs. In: van Schaik CP, Janson CH (eds) Infanticide by males and its implications. Cambridge University Press, Cambridge, pp 153–177
Sterck EHM, Watts DP, van Schaik CP (1997) The evolution of female social relationships in nonhuman primates. Behav Ecol Sociobiol 41:291–309
Stewart KJ (2001) Social relationships of immature gorillas and silverbacks. In: Robbins MM, Sicotte P, Stewart KJ (eds) Mountain gorillas: three decades of research at Karisoke. Cambridge University Press, London, pp 183–214
Stewart KJ, Harcourt AH (1987) Gorillas: variation in female relationships. In: Smuts B, Cheney D, Seyfarth R, Wrangham RW, Struhsaker T (eds) Primate societies. University of Chicago Press, Chicago, pp 155–164
Stokes EJ (2004) Within-group social relationships among females and adult males in wild western lowland gorillas (Gorilla gorilla gorilla). Am J Primatol 64:233–246
Stokes EJ, Parnell RJ, Olejniczak C (2003) Female dispersal and reproductive success in wild western lowland gorillas (Gorilla gorilla gorilla). Behav Ecol Sociobiol 54:329–339
Strier KB (1992) Ateline adaptations: behavioral strategies and ecological constraints. Am J Phys Anthropol 88:515–524
Strier KB (1999a) The atelines. In: Dolhinow P, Fuentes A (eds) The nonhuman primates. Mayfield, Mountain View, pp 109–122
Strier KB (1999b) Why is female kin bonding so rare? Comparative sociality of neotropical primates. In: Lee PC (ed) Comparative primate socioecology. Cambridge University Press, Cambridge, pp 300–319
Strier KB, Ziegler TE (2000) Lack of pubertal influences on female dispersal in muriqui monkeys, Brachyteles arachnoides. Anim Behav 59:849–860
Sugardjito J, te Boekhorst IJA, van Hooff JARAM (1987) Ecological constraints on the grouping of wild orangutans (Pongo pygmaeus) in the Gunung Leuser National Park, Sumatra, Indonesia. Int J Primatol 8:17–41
Sugiyama Y (1988) Grooming interactions among adult chimpanzees at Bossou, Guinea, with special reference to social structure. Int J Primatol 9:393–407
Sugiyama Y (1997) Social tradition and the use of tool-composites by wild chimpanzees. Evol Anthropol 6:23–27
Sugiyama Y (1999) Socioecological factors of male chimpanzee migration at Bossou, Guinea. Primates 40:61–68
Sugiyama Y (2004) Demographic parameters and life history of chimpanzees at Bossou, Guinea. Am J Primatol 124:154–165
Symington MM (1988) Demography, ranging patterns, and activity budgets of black spider monkeys (Ateles paniscus chamek) in the Manu National Park, Peru. Am J Primatol 15:45–67
Symington MM (1990) Fission–fusion social organization in Ateles and Pan. Int J Primatol 11:47–61
Takahata Y, Huffmann MA, Suzuki S, Koyama N, Yamagiwa J (1999) Why dominants do not consistently attain high mating and reproductive success: a review of longitudinal Japanese macaque studies. Primates 40:143–158
Townsend SW, Slocombe KE, Emery Thompson ME, Zuberbühler K (2007) Female-led infanticide in wild chimpanzees. Curr Biol 17:355–356
Tutin CEG (1979) Mating patterns and reproductive strategies in a community of wild chimpanzees (Pan troglodytes schweinfurthii). Behav Ecol Sociobiol 6:29–38
Tutin CEG (1994) Reproductive success story: variability among chimpanzees and comparisons with gorillas. In: Wrangham RW, McGrew WC, de Waal FBM, Heltne PG (eds) Chimpanzee cultures. Harvard University Press, Cambridge, pp 181–194
Tutin CEG (1996) Ranging and social structure of lowland gorillas in the Lopé Reserve, Gabon. In: McGrew WC, Marchant LF, Nishida T (eds) Great ape societies. Cambridge University Press, Cambridge, pp 58–70
Tutin CEG, McGinnis PR (1981) Chimpanzee reproduction in the wild. In: Graham CE (ed) Reproductive biology of the great apes. Academic Press, New York, pp 239–264
Udono T, Sasaoka S, Inoue M, Takenaka A, Takenaka O (1989) Breeding of the chimpanzee in Sanwakagaku Kenkyusho Reichorui Center and paternity discrimination. Primate Res 5:157 (in Japanese)
Valeo A, Schaffner CM, Vick L, Aureli F, Ramos-Fernandez G (2006) Intragroup lethal aggression in wild spider monkeys. Am J Primatol 68:732–737
Van Noodwijk MA, van Schaik CP (2005) Development of ecological competence in Sumatran orangutans. Am J Phys Anthropol 127:79–94
Van Noordwijk MA, van Schaik CP (2000) Reproductive patterns in eutherian mammals: adaptations against infanticide? In: van Schaik CP, Janson CH (eds) Infanticide by males and its implications. Cambridge University Press, Cambridge, pp 322–360
van Schaik CP (1999) The socioecology of fission–fusion sociality in orangutans. Primates 40:73–90
van Schaik CP (2000) Infanticide by male primates: the sexual selection hypothesis revisited. In: van Schaik CP, Janson CH (eds) Infanticide by males and its implications. Cambridge University Press, Cambridge, pp 27–60
van Schaik CP, Deaner RO (2002) Life history and cognitive evolution in primates. In: Tyack PL, de Waal FBM (eds) Animal social complexity. Harvard University Press, Cambridge
van Schaik CP, van Hooff JARAM (1996) Toward an understanding of the orangutan’s social system. In: McGrew WC, Marchant LF, Nishida T (eds) Great ape societies. Cambridge University Press, Cambridge, pp 3–15
van Schaik CP, Preuschoft S, Watts DP (2004) Great ape social systems. In: Russon AE, Begun DR (eds) The evolution of thought: evolutionary origins of great ape intelligence. Cambridge University Press, Cambridge, pp 190–209
Watanabe K (1979) Alliance formation in a free-ranging troop of Japanese macaques. Primates 20:459–474
Watts DP (1989) Infanticide in mountain gorillas: new cases and a reconsideration of the evidence. Ethology 81:1–18
Watts DP (1991a) Harassment of immigrant female mountain gorillas by resident females. Ethology 89:135–153
Watts DP (1991b) Mountain gorilla reproduction and sexual behavior. Am J Primatol 24:211–225
Watts DP (1994) Agonistic relationships between female mountain gorillas (Gorilla gorilla beringei). Behav Ecol Sociobiol 34:347–358
Watts DP (1996) Comparative socio-ecology of gorillas. In: McGrew WC, Marchant LF, Nishida T (eds) Great ape societies. Cambridge University Press, Cambridge, pp 16–28
Watts DP (1997) Agonistic interventions in wild mountain gorilla groups. Behaviour 134:23–57
Watts DP (1998) Long-term habitat use by mountain gorillas (Gorilla gorilla beringei). 1. Consistency, variation, and home range size and stability. Int J Primatol 19:651–680
Watts DP (2000) Causes and consequences of variation in male mountain gorilla life histories and group membership. In: Kappeler P (ed) Primate males. Cambridge University Press, Cambridge, pp 169–179
Watts DP (2003) Gorilla social relationships: a comparative overview. In: Taylor AB, Goldsmith ML (eds) Gorilla biology: a multidisciplinary perspective. Cambridge University Press, Cambridge, pp 302–327
Watts DP, Mitani JC (2000) Infanticide and cannibalism by male chimpanzees at Ngogo, Kibale National Park, Uganda. Primates 41:357–365
Watts DP, Muller M, Amsler S, Mbabazi G, Mitani JC (2006) Lethal intergroup aggression by chimpanzees in the Kibale National Park, Uganda. Am J Primatol 68:161–180
Whitehead H (1998) Cultural selection and genetic diversity in matrilineal whales. Science 282:1708–1711
Whitehead H, Mann J (2000) Female reproductive strategies of cetaceans: life histories and calf care. In: Mann J, Connor RC, Tyack PL, Whitehead H (eds) Cetacean societies: field studies of dolphins and whales. University of Chicago Press, Chicago, pp 219–246
Whitehead H, Weilgart L (2000) The sperm whale: social females and roving males. In: Mann J, Connor RC, Tyack PL, Whitehead H (eds) Cetacean societies: field studies of dolphins and whales. University of Chicago Press, Chicago, pp 154–172
Wich SA, Sterck EHM, Utami SS (1999) Are orangutan females as solitary as chimpanzee females? Folia Primatol (Basel) 70:23–28
Wich SA, Utami-Atmoko SS, Mitra Setia T, Rijksen HD, Schurmann C, van Hooff JARAM, van Schaik CP (2004) Life history of wild Sumatran orangutans (Pongo abelii). J Hum Evol 47:385–398
Wich SA, de Vries H, Ancrenaz M, Perkins L, Shumaker RW, Suzuki A, van Schaik CP (2009) Orangutan life history variation. In: Wich SA, Utami SS, Mitra Setia T, van Schaik CP (eds) Orangutans: geographical variation in behavioral ecology and conservation. Oxford University Press, Oxford, pp 65–75
Williams J, Pusey AE, Carlis JV, Farm BP, Goodall J (2002) Female competition and male territorial behaviour influence female chimpanzees’ ranging patterns. Anim Behav 63:347–360
Williams-Blangero S, Blangero J (1995) Heritability of age at first birth in captive olive baboons. Am J Primatol 37:233–239
Wilson ML, Wrangham RW (2003) Intergroup relations in chimpanzees. Annu Rev Anthropol 32:363–392
Wittig RM, Boesch C (2003) Food competition and linear dominance hierarchy among female chimpanzees in the Tai National Park. Int J Primatol 24:847–867
Wrangham RW (1979) Sex differences in chimpanzee dispersion. In: Hamburg DA, McCown ER (eds) The great apes. Benjamin/Cummings, Menlo Park, pp 480–489
Wrangham RW (1980) An ecological model of female-bonded primate groups. Behaviour 75:262–300
Wrangham RW (1986) Ecology and social relationships in two species of chimpanzees. In: Rubenstein DI, Wrangham RW (eds) Ecological aspects of social evolution: birds and mammals. Princeton University Press, Princeton, pp 352–378
Wrangham RW (1987) Evolution of social structure. In: Smuts BB, Wrangham RW, Cheney DL, Struhsaker TT, Seyfarth RM (eds) Primate societies. University of Chicago Press, Chicago
Wrangham RW, Smuts B (1980) Sex differences in the behavioral ecology of chimpanzees in the Gombe National Park, Tanzania. J Reprod Fertil Suppl 28:13–31
Wroblewski EE, Murray CM, Keele BF, Schumacher-Stankey JC, Hahn BH, Pusey AE (2009) Male dominance rank and reproductive success in chimpanzees, Pan troglodytes schweinfurthii. Anim Behav 77:873–885
Yamagiwa J (1999) Socioecological factors influencing population structure of gorillas and chimpanzees. Primates 40:87–104
Yamagiwa J (2004) Diet and foraging of the great apes: ecological constraints on their social organizations and implications for their divergence. In: Russon AE, Begun DR (eds) The evolution of thought: evolutionary origins of great ape intelligence. Cambridge University Press, Cambridge, pp 210–233
Yamagiwa J, Kahekwa J (2001) Dispersal patterns, group structure and reproductive parameters of eastern lowland gorillas at Kahuzi in the absence of infanticide. In: Robbins MM, Sicotte P, Stewart KJ (eds) Mountain gorillas: three decades of research at Karisoke. Cambridge University Press, London, pp 89–122
Yamagiwa J, Mwanza N, Spangenberg A, Maruhashi T, Yumoto T, Fischer A, Steinhauer BB (1993) A census of the eastern lowland gorillas Gorilla gorilla graueri in Kahuzi-Biega National Park with reference to mountain gorillas G. g. beringei in the Virunga region, Zaire. Biol Conserv 64:83–89
Yamagiwa J, Maruhashi T, Yumoto T, Mwanza N (1996) Dietary and ranging overlap in sympatric gorillas and chimpanzees in Kahuzi-Biega National Park, Zaire. In: McGrew WC, Marchant LF, Nishida T (eds) Great ape societies. Cambridge University Press, Cambridge, pp 82–98
Yamagiwa J, Kahekwa J, Basabose AK (2003) Intra-specific variation in social organization of gorillas: implications for their social evolution. Primates 44:359–369
Yamagiwa J, Kahekwa J, Basabose AK (2009) Infanticide and social flexibility in the genus Gorilla. Primates 50:293–303
Yamagiwa J, Basabose AK, Kahekwa J, Bikaba D, Ando C, Matsubara M, Iwasaki N, Sprague DS (2011) Long-term research on Grauer’s gorillas in Kahuzi-Biega National Park, DRC: life history, foraging strategies, and ecological differentiation from sympatric chimpanzees. In: Kappeler PM, Watts DP (eds) Long-term field studies of primates. Springer, New York, pp 385–412
Yamakoshi G (1998) Dietary responses to fruit scarcity of wild chimpanzees at Bossou, Guinea: possible implications for ecological importance of tool use. Am J Phys Anthropol 106:283–295
Yoshihara K (1999) Present situation of chimpanzees in Japan. Primate Res 15:267–271 (in Japanese)
Acknowledgments
This study was financed in part by Grants-in-Aid for Scientific Research by the Ministry of Education, Culture, Sports, Science, and Technology, Japan (No. 162550080, No. 19107007, and No. 24255010 to J. Yamagiwa), by Grant-in-Aid for Young Scientists (B) 19770213 to Shimooka. the Global Environmental Research Fund by Japanese Ministry of Environment (F-061 to T. Nishida, Japan Monkey Centre), the Kyoto University Global COE Program “Formation of a Strategic Base for Biodiversity and Evolutionary Research,” and Science and Technology Research Partnership for Sustainable Development of JST/JICA, and was conducted in cooperation with CRSN and ICCN. We appreciate to Dr. Kosei Izawa for his kind permission for the usage of a picture.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Japan
About this chapter
Cite this chapter
Yamagiwa, J., Shimooka, Y., Sprague, D.S. (2014). Life History Tactics in Monkeys and Apes: Focus on Female-Dispersal Species. In: Yamagiwa, J., Karczmarski, L. (eds) Primates and Cetaceans. Primatology Monographs. Springer, Tokyo. https://doi.org/10.1007/978-4-431-54523-1_9
Download citation
DOI: https://doi.org/10.1007/978-4-431-54523-1_9
Published:
Publisher Name: Springer, Tokyo
Print ISBN: 978-4-431-54522-4
Online ISBN: 978-4-431-54523-1
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)